Abstract
A native to Europe, Poa annua now has a cosmopolitan distribution and is invasive in the sub-Antarctic. As a major weed in temperate turf, there has been considerable investment in research of the species, but little is known about its ecology in the sub-Antarctic, particularly its reproductive ecology and population dynamics. We characterised the seed bank of this invasive species in the sub-Antarctic, by quantifying seed density, depth, persistence and viability. Poa annua seed bank density was correlated with elevation, animal disturbance, soil wetness and soil depth, but most strongly with P. annua cover. Seed bank density was greatest (132,000 seeds m−2) at low altitude coastal sites where P. annua is abundant but declined with increasing altitude to <2600 seeds m2. Seed was most abundant within the top 3 cm of the soil and decreased with soil depth. Seed viability declined over time, from an initial viability of 81 to <3 % after 2 years in the soil. This study demonstrates that whilst P. annua seed banks can be dense, the seed bank is shallow (<10 cm) with low persistence and viability. This first detailed study on the in situ seed bank profile of P. annua in the sub-Antarctic helps us understand the distribution and persistence of this invasive weed and is essential information for the development and implementation of future management. These findings, such as low seed bank persistence, challenge current thinking about eradication or control feasibility in the region.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Current legislation and management plans call for the eradication and control of invasive species in the sub-Antarctic and Antarctic (de Villiers et al. 2006; Parks and Wildlife Service 2006; Hughes and Convey 2012). There has been a number of successful eradications across the region over the last 30 years; however, these have mostly targeted vertebrate pests (Convey and Lebouvier 2009; Terauds et al. 2014). Only five non-native plant species have been successfully eradicated from sub-Antarctic islands (McGeoch et al. 2015) and 11 non-native plant species from Antarctica (see Hughes et al. 2015). All of these incursions were either a single plant or very small populations with restricted distributions, and for most there was no sign of flowering or seed production (Hughes et al. 2015). Typically most of these eradications were undertaken on an opportunistic or ad hoc basis. There have been several failed and numerous ongoing eradication attempts for non-native plants on sub-Antarctic islands (Shaw 2013) and in Antarctica (Hughes et al. 2015; Molina-Montenegro et al. 2015). In many of these cases ongoing seedling emergence from the seed bank has prevented successful eradication (Cooper et al. 2011; Shaw 2013; Molina-Montenegro et al. 2015). The recent attempted eradication of Poa pratensis in Antarctica (Hughes et al. 2015) is likely to be a success because the small population has never produced fertile seed and therefore has no seed bank (Pertierra et al. 2013).
It is essential to have some ecological knowledge of established non-native plants, in particular their phenological strategies and physiological capacities, when designing eradication strategies (Hughes et al. 2015). Seed bank dynamics are particularly important as they drive regeneration, based on seed density, persistence and viability. It is essential to determine if removal of above ground material and exhaustion of the seed bank are realistic, as well as the timeframe required for such management initiatives (Vranjic et al. 2000; Grundy and Jones 2002; Panetta and Timmins 2004; Gioria and Osborne 2010).
Poa annua is the most widespread non-native plant in the sub-Antarctic, occurring on all major islands (Frenot et al. 2005; Shaw et al. 2010; McGeoch et al. 2015) and also growing in the Antarctic Peninsula (Convey et al. 2006; Hughes et al. 2010; Wódkiewicz et al. 2014; Chwedorzewska et al. 2015). Poa annua is native to Europe but it now has a cosmopolitan distribution (Heide 2001) and occurs within most climates with the exception of arid zones (Beard 1970). It is well studied in temperate areas, where it is a major weed of turf grass (Wu et al. 1987; Christians 2006; Lush 1988). Across the sub-Antarctic, P. annua is thought to have been introduced and spread by humans, given its occurrence around sites of human activity (Frenot et al. 1999, 2005). Poa annua is a primary coloniser of disturbed sites (Frenot et al. 1997) and competes with (Molina-Montenegro et al. 2012, 2016) and displaces native species (Frenot et al. 2001, 2005; Scott and Kirkpatrick 2008). The species is a successful invader globally as it has a wide ecological amplitude, reproduces under a wide variety of environmental conditions, has high fecundity, displays a range of biotypes (Walton 1975; Beard et al. 1978; Mitich 1998; Heide 2001) and has the capacity for rapid evolution of variable life history traits (Till-Bottraud et al. 1990). Poa annua can be perennial or annual, although it is widely assumed that most populations of P. annua in the Antarctic and sub-Antarctic are perennial (Ellis et al. 1971; Walton 1975; Selkirk et al. 1990; Frenot et al. 2005).
Poa annua reproduces primarily by seed (Beard 1970). In temperate, watered, fertilized sites (such as turf grass) the soil seed bank can contain up to 210,000 seeds m−2 (Lush 1988). In Antarctic terrestrial communities, such as the South Shetland Islands, the seed bank density is 5,000 (Wódkiewicz et al. 2013) to 10,000 seeds m−2 (Wódkiewicz et al. 2014). While there has been no quantification of seed bank density in the sub-Antarctic, P. annua on Iles Kerguelen produces 11 fertile non-dormant seeds per plant (Frenot et al. 1997). Poa annua seed viability is high (Hutchinson and Seymour 1982) and seeds can be viable for up to four years (Warwick 1979). Perennial biotypes do not generally exhibit seed dormancy, while dormancy may be several years in annual biotypes (Warwick 1979; Callahan and McDonald 1992; Breuninger 1993).
It is important to determine where in the soil profile seed is buried, as this influences seed viability, recruitment and species persistence (Benvenuti et al. 2001; Grundy et al. 2003). There is little information available on the vertical distribution of the P. annua seed bank. In temperate areas, it is reported to be concentrated in the top soil horizon (0–4 cm) (Graham and Hutchings 1988) and seed has been found to be more dominant in the thatch than the soil (Lush 1988), but can also be found deeper in the soil profile (Akinola et al. 1998). Dynamic soil processes influence seed burial and therefore influence population persistence (Chambers and MacMahon 1994).
On sub-Antarctic Macquarie Island (54°30′ S, 158°57′ E), P. annua is the most common and widespread non-native plant (Copson 1984; Scott and Kirkpatrick 2008; Williams et al. 2013). The island is of high conservation value due to its abundant wildlife and unique geology. It consists of an elongated plateau 200–300 m high, surrounded by steep coastal slopes. Forty two native vascular plant species have been recorded and seven non-native vascular species (de Salas and Baker 2015). Most non-natives have been removed (Copson and Whinam 2001; DPIPWE unpublished data), however Cerastium fontanum, Stellaria media and P. annua remain widespread. The distribution of P. annua expanded with the invasive rabbit population due to its ability to tolerate grazing and the reduced competition from heavily grazed, native plants (Bergstrom et al. 2009; Scott and Kirkpatrick 2013; Whinam et al. 2014). Immediately after the eradication of rabbits on Macquarie Island (Terauds et al. 2014), changes in the vegetation dynamics occurred (Shaw et al. 2011), but the long term response of P. annua and consequent effects on native species are not known. There is an increasing interest by managers across the Antarctic and sub-Antarctic region to eradicate and/or control invasive species (McGeoch et al. 2015), in particular P. annua (Commonwealth of Australia 2014; Hughes et al. 2015) given its prevalence and potential impacts (Molina-Montenegro et al. 2012) on these high conservation ecosystems.
To understand the invasion success of P. annua in the region, it is essential to quantify the species’ seed bank dynamics specifically in the Macquarie Island ecosystem. Globally, seed bank density, depth distribution, persistence and viability of P. annua can vary significantly and studies have shown that seed banks can vary across small spatial scales, i.e. within a site (Lush 1988), including Antarctic populations (Wódkiewicz et al. 2014). Altitude drives vegetation composition and plant growth on Macquarie Island. Given the altitudinal range, associated vegetation, variable topography and the dynamic nature of soils on Macquarie Island (Selkirk et al. 1990; Selkirk-Bell and Selkirk 2013) we aim to understand how the density, depth distribution, persistence and viability of seed of P. annua will vary across the island in response to changing environmental characteristics. Information presented here is essential to understand the invasion biology of P. annua and to determine effective control methods (Vranjic et al. 2000; Grundy and Jones 2002; Panetta and Timmins 2004; Gioria and Osborne 2010). This information will inform future management, given that previous non-native plant management in the region has failed because of the persistent seed banks (see Shaw 2013).
Materials and methods
Study sites
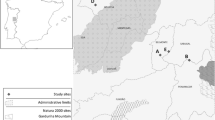
Six sites were located across an altitudinal and P. annua density gradient on Ma cquarie Island (Fig. 1). Two sites, Bauer Bay Beach and Tractor Rock, were selected at low altitude (<50 m above sea level) with high P. annua cover (>60 %). Two sites, Bauer Bay Slope and Doctor’s Track, were selected at mid altitude (100–150 m above sea level) with medium P. annua cover (15–50 %). Two sites, Lower Boot Hill and Upper Boot Hill were selected at high altitude (>250 m above sea level) with low P. annua cover (<10 %). Poa annua residence time was greater than 35 years across all sites (Copson 1984).
Inset Location of Macquarie Island in the Southern Ocean (Australian Antarctic Data Centre 2005); and location of seed bank study sites on Macquarie Island
Environmental characteristics varied across the six study sites. Location, elevation and aspect were determined by handheld GPS (Garmin GPSMap 78) (Table 1). Soil depth was measured ten times at each site by inserting a graduated steel rod into the soil to bedrock or a depth that was greater than the length of the rod (85 cm). Animal disturbance (i.e. old rabbit burrowing, trampling by wildlife) was visually estimated and determined as low—soil and vegetation intact (value of 1); medium—some soil and vegetation disturbance (value of 2) or high—much of the soil and vegetation heavily disturbed (value of 3). Exposure was inferred based on conditions experienced, prevailing winds and topography and was determined to be low—sheltered from prevailing winds (value of 1); medium—some exposure to prevailing winds (value of 2) or high—exposure to much of the prevailing winds (value of 3). Wetness was visually estimated as low—site dry, no surface water (value of 1); medium—soil slightly waterlogged (value of 2) or high—soil saturated, surface water visible (value of 3). Soil type was broadly determined in the field. Variation in site characteristics can be seen in Fig. 2 and Table 1.
Seed bank density
In February 2013 ten 1 m × 1 m plots were randomly located at each site. Within each plot, vegetation was assessed. The species present, percentage of the plot area covered by each species, and maximum heights of three individual plants of each species were recorded. The number of reproductive tillers was counted for 20 random plants at each site to give a mean tiller density/plant/site. A single P. annua plant was collected from the centre of each plot, rinsed of soil, oven dried and the dry weight recorded. A soil core (50 mm diameter and 100 mm depth) was collected from the centre of each plot for seed bank measurements using a stainless-steel corer. Core locations were determined by handheld GPS. Cores were then stored in zip lock bags and refrigerated at 4 °C. An additional ten cores were collected from the same locations in February 2014 to investigate inter-annual variability.
Cores collected in 2013 were analysed in October/November 2013 (9 months after collection) and those collected in 2014 were analysed in February/March 2014 (<1 month after collection). Each core was sectioned sequentially into ten 1 cm deep subsamples, excluding cores collected at one high altitude site which, due to gravel content, did not remain intact. Each sub-sample was placed in a petri dish with a small amount of distilled water and the number of intact seeds of P. annua counted using a dissection microscope at 10× magnification. Seed counts were combined for 0–3, 3–7 and 7–10 cm depth subsamples for statistical analysis.
Seed persistence and viability
Ripe seeds of P. annua were collected in late January 2013 from dispersing seed heads at a variety of sites on Macquarie Island, air dried, mixed well and used for the 2013–2014 assessment of persistence and viability. Seeds that appeared plump, hard and brown under a magnifying lens were selected for burial and a subset tested for viability. Viability was determined by soaking three replicates of 50 seeds in water overnight, slicing them longitudinally through the embryo, and immersing them in a 1 % solution of triphenyltetrazolium chloride in covered petri dishes for at least 20 h (Peters 2000). Seeds were then assessed under a binocular microscope to determine viability (indicated by red/pink staining of the embryo). In instances where some seed staining occurred but the whole embryo was not stained, seeds were not considered viable. This viability testing gave an indication of the initial viability of the buried P. annua seed. Twenty five fresh P. annua seeds were placed between two disks (70 mm diameter and 5 mm thick) of sandy-peat soil sourced from Razor West on Macquarie Island (Fig. 1) and placed in one hundred seed burial bags (100 mm × 100 mm made of Nitex 500 µm 38 % open nylon mesh). The Razor West soil had been sterilised with steam at 115 °C for 2 h to kill any resident seeds. Twenty bags were buried at each site—Bauer Bay Beach, Bauer Bay Slope, Doctor’s Track, Tractor Rock and Lower Boot Hill—in a 4 × 5 grid pattern 0.5 m apart, each in a 15 cm square, 10 cm deep hole that was refilled with soil.
In January 2014 seed was collected and viability tested using the same method as in 2013. However soil used in burial bags was collected from each proposed seed burial site in this experiment, to include all site differences in the treatments. Soil was oven dried at 40 °C and any seed removed and then approximately 15 cm3 mixed with 25 fresh seeds and placed inside the nylon bag. Bags were buried at Bauer Bay Beach, Bauer Bay Slope, Doctor’s Track, Lower Boot Hill, Tractor Rock and Upper Boot Hill in a 4 × 5 cm grid pattern with 10 bags at each of 1 and 5 cm depths.
In January 2014, at each site, 10 of the seed bags buried in 2013 were exhumed and remaining seed counted to give an indication of persistence, and seed viability tested using the method described above. In April 2015 the remaining seed bags buried in 2013 and all seed bags buried in 2014 were exhumed and remaining seeds counted and viability tested. A subset of the seeds present in the seed bank soil cores collected in 2014 was also tested for viability using the method above to give an indication of the viability of the soil seed bank at the time of core collection. Poa annua seeds from depths 0–2, 4–6 and 8–10 cm were grouped together and 3 replicates of 5 or more seeds from samples where there was sufficient seed present (n ≥ 20) were viability tested.
Data analysis
Statistical analyses were carried out using R version 3.1.1 (R Core Team 2014). All results, with the exception of the relationship between seed bank density and percentage cover were analysed using analysis of variance (ANOVA). Variances were checked by plotting residual versus fitted values to confirm the homogeneity of the data and normality was checked by quantile–quantile comparison plots. Significant means were separated using Tukey’s highest significant difference test. Data were transformed to account for non-normal distribution. Seed bank density, P. annua cover and seed viability data were square root transformed. Seed bank depth data were log transformed.
Non-linear regression was used to determine the relationship between P. annua seed bank density and percentage cover of P. annua. A Mitscherlich function was fitted: Y = a × (1 − exp(−b × X)), where Y is the cover, X is the seed bank density, a is the upper asymptote and b is the slope parameter. Bivariate linear regression was used to investigate the relationships between seed bank and environmental characteristics where significance was indicated by a P value less than 0.05.
Results
Seed bank density
There was no significant difference in the seed bank density of cores collected in 2013 and 2014 (P > 0.05) so the average of the 2 years is presented. Seed bank density did differ significantly between sites (P < 0.001) (Table 2). Tractor Rock had a significantly higher seed bank density than all other sites, with approximately 132,000 seeds m−2. The seed bank density at Bauer Bay Beach was much lower, with around 37,000 seeds m−2. Seed bank densities at Bauer Bay Slope, Doctor’s Track and Lower Boot Hill were around 15,000, 10,000 and 2,000 seeds m−2, respectively. No seed was observed at Upper Boot Hill, despite the presence of a small number of reproducing P. annua plants, therefore it was removed from the statistical analyses.
Bivariate linear regression (Table 3) indicated that (higher) seed bank density was negatively correlated with increasing elevation (P < 0.001, R 2 = 0.382) and wetness (P < 0.001, R 2 = 0.537) and positively correlated with increasing P. annua cover (P < 0.001, R 2 = 0.512), animal disturbance (P = 0.001, R 2 = 0.177) and soil depth (P = 0.001, R 2 = 0.199). Poa annua cover and wetness showed a stronger correlation with seed bank density than other variables. Seed bank density was not significantly related to aspect (P = 0.857, R 2 = 0.001) or exposure (P = 0.267, R 2 = 0.022).
Further investigation into the correlation between seed bank density and P. annua cover in the form of non-linear regression analysis indicated a significant relationship of increasing P. annua seed bank density with increasing P. annua cover (P < 0.001, R 2 = 0.670) (Fig. 3). The trend was largely attributed to the variation in cover and seed bank density at Tractor Rock. Percentage P. annua cover was well correlated with site altitude (Table 2). The low altitude, coastal site of Tractor Rock (seed bank density of 132,000 seeds m−2), had a high percentage cover of P. annua (70–80 %; Table 2). The mid altitude sites of Bauer Bay Slope and Doctor’s Track which had an intermediate seed bank size (10,000–15,000 seeds m−2) had moderate cover of P. annua (14–21 %) while the high altitude Lower Boot Hill and Upper Boot Hill sites had small or non-existent seed bank densities (<2,600 seeds m−2) and a low cover of P. annua (<5 %). Although low altitude Bauer Bay Beach had a relatively high cover of P. annua (48–67 %), seed bank density was only moderate (37,000 seeds m−2). Poa annua cover was lower in the second year (2014) at all sites (P ≤ 0.001). The mean dry weight and number of reproductive tillers per P. annua plant also varied between sites. Low altitude plants were large and had a high number of reproductive tillers (Table 1) while high altitude plants were considerably smaller with few reproductive tillers per plant. Plants at mid altitude were intermediate in size and reproductive capacity (Table 1).
Seed bank vertical distribution
Seed density varied significantly with site and depth (P < 0.001) but not with year (P = 0.059) (Table 2). In 2013, all sites except Tractor Rock had significantly higher seed densities in the top 3 cm of the soil than in the bottom 3 cm (P ≤ 0.001). For Tractor Rock, seed density was higher in the top 3 cm than in the bottom 3 cm, but due to high variation in the data there was no statistical significance.
In 2014, Bauer Bay Slope and Doctor’s Track had significantly more seed in the surface soil depths than the lower soil depths (P < 0.05) but there was no statistically significant difference in the seed densities between the top and bottom soil depths at Tractor Rock, Bauer Bay Beach and Lower Boot Hill (P > 0.05). Seed density was generally greater in the top few centimetres of the soil at all sites and declined with depth but high variation in the data often resulted in no significance between consecutive depths. Tractor Rock and Bauer Bay Beach had a high proportion of seed in the mid layers in comparison with the other sites.
Seed persistence and viability
The amount of buried seed remaining (persistence) was significantly lower after 2 years than after 1 year (P < 0.001) (Table 2). After a year of burial, the percentage of seed remaining had declined to approximately 80 % or less and after 2 years of burial had declined to approximately 50 % or less. Although there was a decline in the number of seeds found between 1 and 2 years of burial at each site, the difference was not significant (P > 0.05) at any site. Although ANOVA showed the percentage of seed remaining was significantly different between sites (P = 0.001), this was mainly determined by the data for Bauer Bay Slope which had significantly higher seed persistence than Tractor Rock (P < 0.001) and Doctor’s Track (P = 0.035) over both years.
Initial viability of seeds buried in 2013 was 81.5 ± 1.5 %. This declined after 1 year (2014) to less than 15 % at all sites, except Bauer Bay Slope (28 ± 17 %) (Table 2). After 2 years (2015) viability had declined to less than 3 % at all sites. There were some significant differences in the seed viability between sites (P < 0.001). Seed at Bauer Bay Beach and Bauer Bay Slope had significantly higher post-burial viability than Tractor Rock (P = 0.027 and 0.001, respectively) and Doctor’s Track (P < 0.001 and <0.001, respectively), but was similar to that at Lower Boot Hill (P > 0.05). Seed at Lower Boot Hill had significantly higher viability than Doctor’s Track (P = 0.004) but was similar to that at Tractor Rock (P > 0.05). Seed at Tractor Rock and Doctor’s Track had similarly low viability (P > 0.05).
The persistence of P. annua seed in the soil after 1 year (2014) of burial was assessed at different depths (Table 2). Although persistence and viability of seed at 10 cm depth could not be statistically compared to seed at 1 and 5 cm depths due to slight differences in the burial method, given that initial seed viabilities were similar in both experiments, and that seasonal differences are small in the maritime climate of Macquarie Island, it is reasonable to compare (non-statistically) all three burial depths. There was no significant difference in the percentage of seed remaining between 1 and 5 cm burial depths at any site (P > 0.05) and only Bauer Bay Beach was significantly different to the other sites having a higher percentage of seed remaining than at all other sites (P < 0.05). There was a similar proportion of seed remaining at 10 cm burial depth to 1 and 5 cm for all sites.
The viability of seed buried for 12 months (at 1 and 5 cm depth) decreased from initial viability of 89.7 ± 1.5 % to below 15 % at all sites and burial depths (Table 2). There was no significant difference in seed viability between 1 cm and 5 cm depth (P = 0.148), however site was a significant contributor to differences in the percentage of viable seed. Bauer Bay Beach had a significantly higher proportion of viable seed than Doctor’s Track and Lower Boot Hill (P < 0.01) and Upper Boot Hill had significantly higher seed viability than Tractor Rock, Doctor’s Track, Bauer Bay Slope and Lower Boot Hill (P < 0.01). A higher proportion of seed remained viable at 10 cm depth than 1 or 5 cm.
The viability of P. annua seed in the soil cores was variable. Seed collected from Lower Boot Hill had the highest mean viability at 50 ± 70 %. Seed collected from Tractor Rock had a mean viability of 15 ± 30 %. Bauer Bay Beach, Bauer Bay Slope and Doctor’s Track had lower seed viabilities of less than 7 ± 16 %. However variance was high and so there was no significant difference between sites (P > 0.05).
Discussion
Poa annua is a successful invader of the sub-Antarctic. Successful invaders typically display characteristics such as high fecundity, efficient dispersal, high physiological tolerance, high phenotypic plasticity, relatively high growth rates, an ability to evade natural enemies, fitness homeostasis, high competitive ability, vegetative reproduction, short juvenile period and short intervals between abundant seed production (Rejmanek and Richardson 1996; Pyšek and Richardson 2008; Moravcova et al. 2015).
Poa annua on Macquarie Island has been shown to be an early coloniser, have efficient dispersal (Copson 1984) and tolerate disturbance (Scott and Kirkpatrick 1994) and grazing by responding with rapid growth rates (Copson 1984; Taylor 1955; Whinam et al. 2014). Further to this we have now quantified that (on Macquarie Island) plants show high fecundity, producing dense, somewhat persistent seed banks. We also show that P. annua has high plasticity in growth form and can grow and reproduce across a range of environmental gradients, all of which drives its success as an invader.
Seed bank density and vertical distribution
Poa annua can produce dense seed banks (Mengistu 1999). Even in Antarctica, P. annua seed bank density has been recorded up to 10,000 m−2 (Wódkiewicz et al. 2014). In this study on Macquarie Island, seed bank densities varied mainly in response to P. annua cover. Where there was high P. annua cover, seed bank densities were high (up to 132,000 seeds m−2), and conversely where P. annua cover was low, seed bank density was low.
Seed bank densities were reduced at the high elevation sites due to reduced plant growth and reproductive performance which is typical of high altitude areas (Hunter and Grant 1971; Arroyo et al. 1999; Barcikowski et al. 1999; Hautier et al. 2009). Plants at high altitude were smaller with few reproductive tillers in comparison with plants at low altitude. High altitude sites were also characterised by shallow gravely soil which hinders seed entrapment (Chambers et al. 1991; Chambers and MacMahon 1994) and elevated soil moisture which can reduce seed viability (Schafer and Chilcote 1970; Fenner 2000). These sites are also exposed to the prevailing winds and experience much higher wind speeds than other sites (Tweedie and Bergstrom 2000). Low vegetation cover reduces the opportunity for seed to be trapped (Nelson and Chew 1977; van Tooren 1988; Aguiar and Sala 1994; Wódkiewicz et al. 2014). Cryoturbation is also common at high altitude sites (Bergstrom and Selkirk 1999). While this can aid in the incorporation of seed into the seed bank (van Tooren 1988; Grundy and Jones 2002), the low seed production and strong winds blowing seeds away at these sites discount the role of cryoturbation at these sites. Conversely, at low altitude coastal sites, disturbance promotes growth and reproduction of P. annua (Walton 1975; Hutchinson and Seymour 1982; Erskine et al. 1998; Molina-Montenegro et al. 2014) in addition to greater drainage, lower exposure and denser vegetation cover enhancing seed burial at these sites (Nelson and Chew 1977; van Tooren 1988; Chambers et al. 1991; Aguiar and Sala 1994; Chambers and MacMahon 1994; Fenner 2000; Wódkiewicz et al. 2014).
It is well documented (Moore and Wein 1977; Fenner 1985; Guo et al. 1998; Chang et al. 2001; Reuss et al. 2001; Holmes 2002) that seed of many species are generally concentrated in the top few centimetres of the soil and density declines with depth. In a chalk grassland in the United Kingdom, seed of P. annua was found to be concentrated within the top 4 cm of the soil (Graham and Hutchings 1988). This was also observed for mid and high altitude sites on Macquarie Island where soils were typically peat/gravel mixtures. However, at low altitude coastal sites less aggregated sandy soils enable seeds to move deeper down the soil profile (Chambers et al. 1991; Guo et al. 1998). The dynamic nature of the coastal soils driving seed to reach deeper depths (Fenner 2000) can likewise bring seeds to the surface where they can germinate.
Seed persistence and viability
After 2 years in the soil seed bank, the majority of seed was non-viable. While this does not indicate lack of dormancy of the seed per se, the perennial biotype of P. annua does not have dormant seed (Warwick 1979; Lush 1988; Callahan and McDonald 1992) and plants on sub-Antarctic Kerguelen do not exhibit seed dormancy (Frenot and Gloaguen 1994). Persistence and viability of P. annua seed was enhanced deeper in the soil profile, as is typical for many species (Dawson and Bruns 1975; Egley and Chandler 1978, 1983; Zorner et al. 1984; Gleichsner and Appleby 1989; Miller and Nalewaja 1990; Mennan and Zandstra 2006). However for management purposes it is vital to acknowledge that even after 2 years some seed remained viable.
Implications
Poa annua plants on Macquarie Island possess many of the characteristics that are common to invasive species, explaining its success in the sub-Antarctic. Our research also suggests that non-native species with similar characteristics to P. annua—high fecundity, wide tolerance, high plasticity—will also show potential to be invasive in the sub-Antarctic.
Information on the seed bank dynamics of an invasive species informs the impact of the species on native biota. High density seed banks of invasive species, similar to P. annua at coastal sites on Macquarie Island, impact both on native seed banks and standing plant communities (Hejda et al. 2009; Vila et al. 2011; Gioria et al. 2014). Poa annua has been documented to germinate at lower temperatures than native grasses, giving it a competitive advantage (Frenot and Gloaguen 1994). Poa annua also competes with species native to the Antarctic Peninsula (Molina-Montenegro et al. 2012).
For any future management of P. annua in the sub-Antarctic, or indeed Antarctica, information on the seed bank dynamics of P. annua is valuable. For example, if single attempts are made to remove above ground P. annua with the aim of preventing further seed set, a residual seed bank will still remain. However, viability is low and only a small proportion will be viable after several years, so efforts should focus on ongoing control of the emergent seedlings for at least two subsequent years. The depth and density of the seed bank will also influence whether chemical or non-chemical control methods are more appropriate (Hobbs and Humphries 1995; Steinke and Stier 2002; Visser et al. 2010).
Further research should focus on in situ and ex situ management to further inform control of this highly invasive species in the sub-Antarctic and broader region. There are several cases of failed plant eradication and control attempts in the sub-Antarctic due to seed bank persistence. Information on seed bank dynamics would increase the success of control programs. Findings such as low persistence of viable seeds challenge current thinking about eradication or control feasibility in the region and allow management programs to be carefully tailored to specific seed bank dynamics and achieving more successful and efficient control. The characteristics of P. annua seed banks contribute to its success as an island wide invader on Macquarie Island, and more broadly throughout the sub-Antarctic and Antarctic.
References
Aguiar MR, Sala OE (1994) Competition, facilitation, seed distribution and the origin of patches in a Patagonian Steppe. Oikos 70:26–34. doi:10.2307/3545695
Akinola MO, Thompson K, Hillier SH (1998) Development of soil seed banks beneath synthesized meadow communities after seven years of climate manipulations. Seed Sci Res 8:493–500. doi:10.1017/S0960258500004463
Arroyo M, Cavieres LA, Castor C, Humaña AM (1999) Persistent soil seed bank and standing vegetation at a high alpine site in the central Chilean Andes. Oecologia 119:126–132. doi:10.1007/s004420050768
Australian Antarctic Data Centre (2005) Map 13143: location of Macquarie Island in relation to Australia and Antarctica [black and white]. Australian Antarctic Division, Australia
Barcikowski A, Lyżwińska R, Zarzycki K (1999) Growth rate and biomass production of Deschampsia antarctica Desv. in the Admiralty Bay Region, South Shetland Islands, Antarctica. Pol Polar Res 20:301–311
Beard J (1970) An ecological study of annual bluegrass. US Golf Assoc Green Sect Rec 8:13–18
Beard JB, Rieke PE, Turgeon AJ, Vargas JMJ (1978) Annual bluegrass (Poa annua L.): description, adaptation, culture and control. In: Michigan State University, Agricultural Experiment Station Research Report. # 352. Michigan State University Agricultural Experiment Station, East Lansing
Benvenuti S, Macchia M, Miele S (2001) Quantitative analysis of emergence of seedlings from buried weed seeds with increasing soil depth. Weed Sci 49:528–535. doi:10.1614/0043-1745(2001)049[0528:QAOEOS]2.0.CO;2
Bergstrom D, Selkirk P (1999) Bryophyte propagule banks in a feldmark on subantarctic Macquarie Island. Arct Antarct Alp Res 31:202–208. doi:10.2307/1552610
Bergstrom DM, Lucieer A, Kiefer K, Wasley J, Belbin L, Pedersen TK, Chown SL (2009) Indirect effects of invasive species removal devastate World Heritage Island. J Appl Ecol 46:73–81. doi:10.1111/j.1365-2664.2008.01601.x
Breuninger J (1993) Poa annua control in Bentgrass greens. Golf Course Manag 61:68–73
Callahan LM, McDonald ER (1992) Effectiveness of bensulide in controlling two annual bluegrass (Poa annua) subspecies. Weed Technol 6:97–103
Chambers JC, MacMahon JA (1994) A day in the life of a seed: movements and fates of seeds and their implications for natural and managed systems. Annu Rev Ecol Syst 25:263–292. doi:10.2307/2097313
Chambers JC, MacMahon JA, Haefner JH (1991) Seed entrapment in alpine ecosystems: effects of soil particle size and diaspore morphology. Ecology 75:1668–1677. doi:10.2307/1940966
Chang ER, Jefferies RL, Carleton TJ (2001) Relationship between vegetation and soil seed banks in an Arctic coastal marsh. J Ecol 89:367–384. doi:10.2307/3072282
Christians N (2006) Control options: What’s next for Poa annua control? Grounds Maint 41:28–30
Chwedorzewska K, Gielwanowska I, Olech M, Molina-Montenegro MA, Wódkiewicz M, Galeria H (2015) Poa annua L. in the maritime Antarctic: an overview. Polar Rec 261:637–643. doi:10.1017/S0032247414000916
Commonwealth of Australia (2014) Heard Island and McDonald Islands marine reserve management plan 2014–2024. Department of the Environment, Canberra
Convey P, Lebouvier M (2009) Environmental change and human impacts on terrestrial ecosystems of the sub-Antarctic islands between their discovery and the mid-twentieth century. Pap Proc R Soc Tasman 143:33–44
Convey P, Frenot Y, Gremmen N, Bergstrom DM (2006) Biological invasions. In: Bergstrom DM, Convey P, Huiskes AHL (eds) Trends in Antarctic terrestrial and limnetic ecosystems. Springer, Dordrecht, pp 193–220. doi:10.1007/1-4020-5277-4_10
Cooper J, Cuthbert R, Gremmen N, Ryan PG, Shaw JD (2011) Earth, fire and water: applying novel techniques to eradicate the invasive plant, procumbent pearlwork Sagina procumbens, on Gough Island, a World Heritage Site in the South Atlantic. In: Veitch CR, Clout MN, Towns, DR (eds) Island invasives: eradication and management: proceedings of the international conference on island invasives, Auckland, New Zealand, pp 162–165
Copson GR (1984) An annotated atlas of the vascular flora on Macquarie Island. ANARE Res Notes 18:1-70
Copson G, Whinam J (2001) Review of ecological restoration programme on subantarctic Macquarie Island: pest management progress and future directions. Ecol Manag Restor 2:129–138. doi:10.1046/j.1442-8903.2001.00076.x
Core Team R (2014) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Dawson JH, Bruns VF (1975) Longevity of barnyardgrass, green foxtail, and yellow foxtail seeds in soil. Weed Sci 23:437–440. doi:10.2307/4042354
de Salas M, Baker M (2015) A census of the vascular plants of Tasmania and index to the student’s flora of Tasmania and flora of Tasmania online. Tasmanian Herbarium, Tasmanian Museum and Art Gallery, Hobart
de Villiers MS, Cooper J, Carmichael N, Glass JP, Liddle GM, McIvor E, Micol T, Roberts A (2006) Conservation management at Southern Ocean islands: towards the development of best-practice guidelines. Polarforschung 75:113–131. doi:10.1007/s003000050423
Egley GH, Chandler JM (1978) Germination and viability of weed seeds after 2.5 years in a 50-year buried seed study. Weed Sci 26:230–239. doi:10.2307/4042705
Egley GH, Chandler JM (1983) Longevity of weed seeds after 5.5 years in the Stoneville 50-year buried-seed study. Weed Sci 31:264–270. doi:10.2307/4043807
Ellis WM, Lee BTO, Calder DM (1971) A biometric analysis of populations of Poa annua L. Evolution 25:29–37. doi:10.2307/2406497
Erskine PD, Bergstrom DM, Schmidt S, Stewart GR, Tweedie CE, Shaw JD (1998) Subantarctic Macquarie Island: a model ecosystem for studying animal-derived nitrogen sources using 15N natural abundance. Oecologia 117:187–193. doi:10.2307/4222149
Fenner M (1985) Seed Ecology. Chapman and Hall, London
Fenner M (ed) (2000) Seeds: The Ecology of Regeneration in Plant Communities, 2nd edn. CABI Publishing, Wallingford
Frenot Y, Gloaguen J-C (1994) Reproductive performance of native and alien colonizing phanerogams on a glacier foreland, Iles Kerguelen. Polar Biol 14:473–481. doi:10.1007/bf00239052
Frenot Y, Gloaguen JC, Tréhen P (1997) Climate change in Kerguelen Islands and colonization of recently deglaciated areas by Poa kerguelensis and Poa annua. In: Battaglia B, Valencia J, Walton DWH (eds) Antarctic communities: species, structure and survival. Cambridge University Press, Cambridge
Frenot Y, Aubry M, Misstet MT, Gloaguen JC, Gourret JP, Lebouvier M (1999) Phenotypic plasticity and genetic diversity in Poa annua L. (Poaceae) at Crozet and Kerguelen Islands (subantarctic). Polar Biol 22:302–310. doi:10.1007/s003000050423
Frenot Y, Gloaguen JC, Masse L, Lebouvier M (2001) Human activities, ecosystem disturbance and plant invasion in subantarctic Crozet, Kerguelen and Amsterdam Islands. Biol Conserv 101:33–50. doi:10.1016/S0006-3207(01)00052-0
Frenot Y, Chown SL, Whinam J, Selkirk PM, Convey P, Skotnicki M, Bergstrom DM (2005) Biological invasions in the Antarctic: extent, impacts and implications. Biol Rev 80:45–72. doi:10.1017/S1464793104006542
Gioria M, Osborne B (2010) Similarities in the impact of three large invasive plant species on soil seed bank communities. Biol Invasions 12:1671–1683. doi:10.1007/s10530-009-9580-7
Gioria M, Jarosik V, Pyšek P (2014) Impact of invasions by alien plants on soil seed bank communties: emerging patterns. Perspect Plant Ecol Evol Syst 16:132–142. doi:10.1017/S0032247414000916
Gleichsner JA, Appleby AP (1989) Effect of depth and duration of seed burial on ripgut brome (Bromus rigidus). Weed Sci 37:68–72. doi:10.2307/4044758
Graham DJ, Hutchings MJ (1988) Estimation of the seed bank of a chalk grassland ley established on former arable land. J Appl Ecol 25:241–252. doi:10.2307/2403622
Grundy AC, Jones NE (2002) Weed Management Handbook. Blackwell Science Ltd, Osney
Grundy AC, Mead A, Burston S (2003) Modelling the emergence response of weed seeds to burial depth: interactions with seed density, weight and shape. J Appl Ecol 40:757–770. doi:10.1046/j.1365-2664.2003.00836.x
Guo Q, Rundel PW, Goodall DW (1998) Horizontal and vertical distribution of desert seed banks: patterns, causes, and implications. J Arid Environ 38:465–478. doi:10.1006/jare.1997.0353
Hautier Y, Randin CF, Stocklin J, Guisan A (2009) Changes in reproductive investment with altitude in an alpine plant. J Plant Ecol-UK 2:125–134. doi:10.1093/jpe/rtp011
Heide OM (2001) Flowering responses of contrasting ecotypes of Poa annua and their putative ancestors Poa infirma and Poa supina. Ann Bot 87:795–804. doi:10.1006/anbo.2001.1406
Hejda M, Pyšek P, Jarosik V (2009) Impact of invasive plants on the species richness, diversity and composition of invaded communities. J Ecol 97:393–403. doi:10.1111/j.1365-2745.2009.01480.x
Hobbs R, Humphries S (1995) An integrated approach to the ecology and management of plant invasions. Conserv Biol 9:761–770. doi:10.1046/j.1523-1739.1995.09040761.x
Holmes PM (2002) Depth distribution and composition of seed-banks in alien-invaded and uninvaded fynbos vegetation. Austral Ecol 27:110–120. doi:10.1046/j.1442-9993.2002.01164.x
Hughes KA, Convey P (2012) Determining the native/non-native status of newly discovered terrestrial and freshwater species in Antarctica—current knowledge, methodology and management action. J Enviro Manage 93:52–66. doi:10.1016/j.jenvman.2011.08.017
Hughes KA, Convey P, Maslen NR, Smith RIL (2010) Accidental transfer of non-native soil organisms into Antarctica on construction vehicles. Biol Invasions 12:875–891. doi:10.1007/s10530-009-9508-2
Hughes K, Pertierra L, Molina-Montenegro M, Convey P (2015) Biological invasions in terrestrial Antarctica: what is the current status and can we respond? Biodivers Conserv 24:1031–1055. doi:10.1007/s10531-015-0896-6
Hunter R, Grant SA (1971) The effect of altitude on grass growth in East Scotland. J Appl Ecol 8:1–19. doi:10.2307/2402123
Hutchinson CS, Seymour GB (1982) Poa annua L. J Ecol 70:887–901. doi:10.2307/2260111
Lush WM (1988) Biology of Poa annua in a temperate zone golf putting green (Agrostis stolonifera/Poa annua). II. The seed bank. J Appl Ecol 25:989–997. doi:10.2307/2403760
McGeoch MA, Shaw JD, Terauds A, Lee JE, Chown SL (2015) Monitoring biological invasion across the broader Antarctic: a baseline and indicator framework. Global Environ Chang 32:108–125. doi:10.1016/j.gloenvcha.2014.12.012
Mengistu LW (1999) Genetic diversity and herbicide resistance in annual bluegrass (Poa annua L.). Oregon State University, Oregon
Mennan H, Zandstra B (2006) The effects of depth and duration of seed burial on viability, dormancy, germination, and emergence of ivyleaf speedwell (Veronica hederifolia). Weed Technol 20:438–444. doi:10.1614/WT-05-090R.1
Miller SD, Nalewaja JD (1990) Influence of burial depth on wild oats (Avena fatua) seed longevity. Weed Technol 4:514–517. doi:10.2307/3987500
Mitich LW (1998) Annual bluegrass (Poa annua L.). Weed Technol 12:414–416
Molina-Montenegro MA, Carrasco-Uraa F, Rodrigo C, Convey P, Valladares F, Gianoli E (2012) Occurence of the non-native annual blugrass on the Antarctic mainland and its negative effects on native plants. Conserv Biol 26:717–723. doi:10.1111/j.1523-1739.2012.01865.x
Molina-Montenegro MA, Carrasco-Uraa F, Acuna-Rodrigues I, Oses R, Torres-Diaz C, Chwedorzewska KJ (2014) Assessing the importance of human activities for the establishment of the invasive Poa annua in Antarctica. Polar Res 33:214–225. doi:10.3402/polar.v33.21425
Molina-Montenegro M, Pertierra L, Razeto-Barry P, Díaz J, Finot V, Torres-Díaz C (2015) A recolonization record of the invasive Poa annua in Paradise Bay, Antarctic Peninsula: modeling of the potential spreading risk. Polar Biol 38:1091–1096. doi:10.1007/s00300-015-1668-1
Molina-Montenegro MA, Galleguillos C, Oses R, Acuna-Rodriguez I, Lavin P, Gallardo-Cerda J, Torres-Diaz C, Diex B, Pizarro GE, Atalo C (2016) Adaptive phenotypic plasticity and competitive ability deployed under a climate change scenario may promote the invasion of Poa annua in Antarctica. Biol Invasions 18:603–618. doi:10.1007/s/10530-015-1033-x
Moore JM, Wein RW (1977) Viable seed populations by soil depth and potential site recolonization after disturbance. Can J Bot 55:2408–2412. doi:10.1139/b77-274
Moravcova L, Pyšek P, Jarosik V, Pergl J (2015) Getting the right traits: reproductive and dispersal characteristics predict the invasiveness of herbaceous plant species. PLoS ONE 10:e0123634. doi:10.1371/journal.pone.0123634
Nelson JF, Chew RM (1977) Factors affecting seed reserves in the Mojave Desert ecosystem, Rock Valley, Nye County, Nevada. Am Midl Nat 97:300–320
Panetta F, Timmins S (2004) Evaluating the feasibility of eradication for terrestrial weed incursions. Plant Prot Q 19:5–11
Parks and Wildlife Service (2006) Macquarie Island nature reserve and world heritage area management plan. Parks and Wildlife Service, Department of Tourism Arts and the Environment, Hobart
Pertierra LR, Lara F, Benayas J, Hughes KA (2013) Poa pratensis L., current status of the longest-established non-native vascular plant in the Antarctic. Polar Biol 36:1473–1481. doi:10.1007/s00300-013-1367-8
Peters J (ed) (2000) Tetrazolium Testing Handbook: Contribution No. 29 to the Handbook on Seed Testing, revised 2000. AOSA, Las Cruces
Pyšek P, Richardson D (2008) Invasive Plants. Elsevier, Oxford
Rejmanek M, Richardson D (1996) What attributes make some plant species more invasive? Ecology 77:1655–1661. doi:10.2307/2265768
Reuss SA, Buhler DD, Gunsolus JL (2001) Effects of soil depth and aggregate size on weed seed distribution and viability in a silt loam soil. Appl Soil Ecol 16:209–217. doi:10.1016/S0929-1393(00)00115-3
Schafer DE, Chilcote DO (1970) Factors influencing persistence and depletion in buried seed populations. II. The effects of soil temperature and moisture. Crop Sci 10:342–345. doi:10.2135/cropsci1970.0011183X001000040007x
Scott JJ, Kirkpatrick JB (1994) Effects of human trampling on the sub-Antarctic vegetation of Macquarie Island. Polar Rec 30:207–220. doi:10.1017/S003224740002427X
Scott JJ, Kirkpatrick JB (2008) Rabbits, landslips and vegetation change on the coastal slopes of subantarctic Macquarie Island, 1980-2007: implications for management. Polar Biol 31:409–419. doi:10.1007/s00300-007-0367-y
Scott JJ, Kirkpatrick JB (2013) Changes in the cover of plant species associated with climate change and grazing pressure on the Macquarie Island coastal slopes, 1980–2009. Polar Biol 36:127–136. doi:10.1007/s00300-012-1243-y
Selkirk P, Seppelt R, Selkirk D (1990) Subantarctic Macquarie Island: Environment and Biology. Cambridge University Press, Cambridge
Selkirk-Bell JM, Selkirk PM (2013) Vegetation-banked terraces on subantarctic Macquarie Island: a reappraisal. Arct Antarct Alp Res 45:261–274. doi:10.1657/1938-4246-45.2.261
Shaw JD (2013) Southern Ocean Islands invaded: Conserving biodiversity in the world’s last true wilderness. In: Foxcroft L, Pyšek P, Richardson DM, Genovesi P (eds) Plant invasions in protected areas: PATTERNS, problems and challenges. Invading nature—Springer Series in invasion ecology, vol 7. Springer, Berlin, pp 449–472
Shaw JD, Spear D, Greve M, Chown SL (2010) Taxonomic homogenization and differentiation across Southern Ocean Islands differ among insects and vascular plants. J Biogeog 37:217–228. doi:10.1111/j.1365-2699.2009.02204.x
Shaw JD, Terauds A, Bergstrom DM (2011) Rapid commencement of ecosystem recovery following aerial baiting on sub-Antarctic Macquarie Island. Ecol Manag Restor 12:241–244. doi:10.1111/j.1442-8903.2011.00611.x
Steinke K, Stier J (2002) Tolerance of supina bluegrass to pre and post-emergence herbicides. J Environ Hortic 20:118–121
Taylor BW (1955) The flora, vegetation and soils of Macquarie Island. Australian National Antarctic Research Expeditions Reports, B (2) No. 19. Botany, Hobart
Terauds A, Doube J, McKinlay J, Springer K (2014) Using long-term population trends of an invasive herbivore to quantify the impact of management actions in the sub-Antarctic. Polar Biol 37:833–843. doi:10.1007/s00300-014-1485-y
Till-Bottraud I, Wu L, Harding J (1990) Rapid evolution of life history traits in populations of Poa annua L. J Evol Biol 3:205–224. doi:10.1046/j.1420-9101.1990.3030205.x
Tweedie CE, Bergstrom D (2000) A climate change scenario for surface air temperature at subantarctic Macquarie Island. Paper presented at the Proceedings of the VII SCAR international biology symposium, Christchurch, New Zealand, 31st October–4th September
van Tooren BF (1988) The fate of seeds after dispersal in Chalk Grassland: the role of the bryophyte layer. Oikos 53:41–48. doi:10.2307/3565661
Vila M, Espinar JL, Hejda M, Hulme PE, Jarosik V, Maron JL, Pergl J, Schaffner U, Sun Y, Pyšek P (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14:702–708. doi:10.1111/j.1461-0248.2011.01628.x
Visser P, Louw H, Cuthnert R (2010) Strategies to eradicate the invasive plant procumbent pearlwort Sagina procumbens on Gough Island, Tristan da Cunha. Conserv Evid 7:116–122
Vranjic JA, Groves RH, Willis AJ (2000) Environmental weed management systems. In: Sindel BM (ed) Australian weed management systems. RG and FJ Richardson, Melbourne
Walton DWH (1975) European weeds and other alien species in the sub-Antarctic. Weed Res 15:271–282. doi:10.1111/j.1365-3180.1975.tb01135.x
Warwick S (1979) The biology of Canadian weeds: 37 Poa annua L. Can J Plant Sci 59:1053–1066. doi:10.4141/cjps79-165
Whinam J, Fitzgerald N, Visoiu M, Copson G (2014) Thirty years of vegetation dynamics in response to a fluctuating rabbit population on sub-Antarctic Macquarie Island. Ecol Manag Restor 15:41–51. doi:10.1111/emr.12076
Williams L, Kristiansen P, Shaw JD, Sindel BM, Wilson SC (2013) Weeds down under: invasion of the sub-Antarctic wilderness of Macquarie Island. Plant Prot Q 28:71–72
Wódkiewicz M, Galera H, Chwedorzewska KJ, Gielwanowska I, Olech M (2013) Diaspores of the introduced species Poa annua L. in soil samples from King George Island (South Shetlands, Antarctica). Arct Antarct Alp Res 45:1–5. doi:10.1657/1938-4246-45.3.415
Wódkiewicz M, Ziemianski M, Kwiecien K, Chwedorzewska K, Galera H (2014) Spatial structure of the soil seed bank of Poa annua L.—alien species in the Antarctica. Biodivers Conserv 23:1339–1346. doi:10.1007/s10531-014-0668-8
Wu L, Till-Bottraud I, Torres A (1987) Genetic differentiation in temperature-enforced seed dormancy among golf course populations of Poa annua L. New Phytol 107:623–631. doi:10.1111/j.1469-8137.1987.tb02932.x
Zorner PS, Zimdahl RL, Schweizer EE (1984) Effect of depth and duration of seed burial on kochia (Kochia scoparia). Weed Sci 32:602–607. doi:10.2307/4043975
Acknowledgments
This project was funded by the Australian Antarctic Science Program, project AAS 4158 and an Australian Postgraduate Award through the University of New England. We thank the Tasmanian Parks and Wildlife Service for granting us access to Macquarie Island, the Australian Antarctic Division for logistical support and Luis Rodriguez Pertierra for field support. We thank Dr. Dana Bergstrom and two anonymous reviewers for feedback on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Williams, L.K., Kristiansen, P., Sindel, B.M. et al. Quantifying the seed bank of an invasive grass in the sub-Antarctic: seed density, depth, persistence and viability. Biol Invasions 18, 2093–2106 (2016). https://doi.org/10.1007/s10530-016-1154-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-016-1154-x