Abstract
Invasive species may affect community diversity in several ways, causing species loss at local scales and promoting either biotic homogenization among sites within a region or increases in beta diversity among regions. If these losses or additions are restricted to specific functional groups or clades, it could mean the loss or modification of an ecosystem process, and cause important changes in native community structures. In this study, we assessed whether a bullfrog invasion is related to diversity changes in Atlantic Forest anuran communities. We compared communities from invaded and non-invaded sites, and tested for differences in composition and in the variability of composition, at both taxonomic and phylogenetic levels. The potential influence of pond and habitat features in our results was also assessed. We also tested if bullfrog invasion is promoting nestedness. We found no differences among anuran compositions from 57 invaded and non-invaded sites, across the three regions studied, either for taxonomic or for phylogenetic data. Habitat descriptors partially explained taxonomic composition. The taxonomic and phylogenetic compositions were also similar regarding dispersion variability. In addition, invaded sites did not show nested compositions from non-invaded sites. In sum, our results do not support the hypothesis that bullfrog invasion caused changes in diversity patterns of anuran communities. Our results contrast with studies conducted in other regions of the world, where invasive populations of bullfrogs seem to affect local native communities. In those places, bullfrogs find congeneric or confamilial species, which may be more affected by the invasion. In the Atlantic Forest, bullfrogs find suitable climatic conditions, and may face low biotic resistance since they find no congeneric species. These conditions may allow bullfrogs to coexist with native species without changes in diversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Species introduced into a new environment may affect the native diversity in several ways, ranging from decreases in native species fitness to complete changes in ecosystem functioning (Simberloff et al. 2005; Pyšek and Richardson 2010). In some cases, however, no apparent ecological effects have been detected, generally when the alien species fails to achieve invasive status (Blackburn et al. 2011; Zenni and Nuñez 2013). Of special concern are the pathways by which invasions may alter the diversity of native communities, and the consequences for community structure and ecosystem functioning (Olden and Poff 2003). For instance, invasions could lead to species loss or community enrichment, and both processes can profoundly alter community structure (Olden and Poff 2003; Sax and Gaines 2003; Winter et al. 2009).
Invasions may cause species loss at local scales, decreasing local (alpha) diversity. This effect on local diversity might also result in biotic homogenization, causing an increase of similarity in composition between invaded sites (Duncan et al. 2003; Ricciardi 2007). Biotic homogenization is likely to take place in a scenario where invaded sites present the same set of species, composed mostly of resistant species. These invaded sites, harboring resistant species, may also constitute nested compositions compared to non-invaded sites. Such community nestedness is likely to occur because non-invaded sites can support a more varied composition, including both resistant and susceptible species.
Studies of the effects of alien species on local diversity have usually focused on the potential changes of species composition and/or species loss (e.g., Qian and Ricklefs 2006; Lockwood 2006; Piazzi and Balata 2008; Stenert et al. 2012; Trentanovi et al. 2013). However, the vulnerability of a community to invasion could be more closely associated with its phylogenetic and functional diversity (Cianciaruso et al. 2009; Winter et al. 2009). Species that have co-occurred for long periods tend to be controlled by the same resource constraints and biotic interactions and to show some degree of divergence (Tilman 2004). Therefore, if species losses are restricted to certain functional groups or specific clades, the impacts on community structure are expected to be worryingly severe, and could mean the loss of an ecosystem process and/or of a historically-molded relationship (Winter et al. 2009). Furthermore, invasive species that are distantly phylogenetically related to species of native communities may take advantage of their distinct life-history traits, which may constitute a novelty in the invaded sites, and successfully establish new populations and attain high invasive status (Strauss et al. 2006). Accordingly, the phylogenetic structure of invaded communities and its relatedness to the invasive species are important issues to be addressed in studies on communities facing invasions.
In this study, we tested whether an invasive species can affect community composition at taxonomic and phylogenetic levels, by studying anuran pond communities from Atlantic Forest areas invaded by American bullfrogs (Lithobates catesbeianus). The bullfrogs are native to North America, but are invasive in more than 40 countries (Lever 2003). In some invaded sites, bullfrogs have displaced native amphibians and have been linked to species losses (Kraus 2009). Bullfrogs were introduced into the Brazilian Atlantic Forest in 1935, and the invasion process has been practically ignored since then (Both et al. 2011). Lithobates catesbeianus belongs to the family Ranidae (Pyron and Wiens 2011) and is distantly related to Neotropical clades occurring in the Atlantic Forest, with no confamilial member in the region.
We compared anuran compositions of communities of invaded and non-invaded sites in the Atlantic Forest. Specifically, we tested for (1) differences in composition and (2) in compositional variances (beta diversity), at both taxonomic and phylogenetic levels. Considering the history of bullfrog impacts in other invaded localities, we expected to find a low variance in species composition among the invaded ponds (i.e. low beta diversity). Additionally, we were interested in the potential mechanisms related to the hypothesized community changes in invaded versus non-invaded sites. We also tested (3) the hypothesis that the invaded communities were nested subsets, in terms of both taxonomic and phylogenetic composition, of the non-invaded communities.
Materials and methods
Study area and sampling
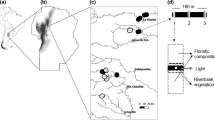
Amphibian diversity data were compiled from Both et al. (2014), who sampled ponds invaded and not invaded by bullfrogs in the Atlantic Forest, Brazil. Communities were sampled in three areas in the southern part of the Atlantic Forest. Study area A1 was located in the central part of the state of Rio Grande do Sul (29°25′46″–29°25′46″S/53°35′29″–53°12′44″W), and has Seasonal Deciduous Forest vegetation (IBGE 2004). Study area A2 was in the western part of the state of Santa Catarina (26°47′25″–27°09′24″S/52°58′57″–52°37′57″W), where both Seasonal Deciduous Forest and Mixed Ombrophilous Forest occur (IBGE 2004; Lucas and Fortes 2008). Study area A3 was located in eastern Santa Catarina (26°41′51″–27°02′27″S/49°15′48″–49°04′25″W) and is covered by Dense Ombrophilous Forest (IBGE 2004). All these areas have a highly suitable climate for L. catesbeianus (Giovanelli et al. 2008; Nori et al. 2011), and have the greatest density of bullfrog populations yet recorded in Brazil (Both et al. 2011). The forest in these areas is highly fragmented, as is the entire biome, which currently comprises <8 % of the original forest (Fundação SOS Mata Atlântica/INPE 2009). The general land use is farming (soybeans, corn, tobacco) and cattle grazing.
We selected 57 ponds suitable for bullfrog breeding from a larger survey of 90 ponds (Both et al. 2014). Ponds were natural or artificial, at least 50 cm deep and were filled in the three visits we did in the period of 18 m (one for pond selection and two for surveys). The 57 studied ponds were distributed in the three areas of study (A1: 16 invaded and 7 non-invaded ponds; A2: 10 invaded, 6 non-invaded; A3: 11 invaded, 7 non-invaded). We considered invaded ponds as those where bullfrogs displayed reproductive activities (calling males, amplexus, egg masses, tadpoles). Ponds without evidence of breeding were considered as non-invaded. Calling males were detected in 36 of the 37 invaded ponds, and their presence was highly associated with the presence of bullfrog tadpoles (X2 = 38.51, p < 0.001). Egg masses were observed in 12 ponds. In these three subtropical regions, bullfrogs can be seen all year around and concentrate reproductive activities during spring and summer (Kaefer et al. 2007). Male of bullfrogs are conspicuous and territorials. Data collection was planned to overlap breeding period (see below). Geographical coordinates and pond characteristics can be found in Online Resource 1.
Each pond was surveyed twice for post-metamorphic individuals and tadpoles. Surveys occurred in February–March (late summer) and October–November (spring) of 2010 and lasted about 25 days each. Those periods coincide with high native anuran and bullfrog breeding activities in the subtropical region (Kaefer et al. 2007; Both et al. 2008). On each day of a survey, four to six sites were examined for tadpoles in the daytime and for post-metamorphic individuals at night. Tadpoles were sampled in different microhabitats (e.g., margin with and without vegetation, center with and without vegetation) with dip-net sweeps (40 × 30 cm, mesh 0.02 mm), always done by the same collector. The effort was proportional to the pond size and heterogeneity (Shaffer et al. 1994). At least five sweeps were made in each microhabitat. Surveys were done between 09:00 and 19:00. Post-metamorphic surveys were begun 30 min after sunset, and we searched for individuals along the perimeters of breeding sites. Records of species presence were made using visual and auditory sampling. The effort employed for post-metamorphics was proportional to the size and complexity of the pond (Scott and Woodward 1994). Further details about sampling are available in Both et al. (2014) and anuran compositions are presented in Online Resource 2.
Pond and habitat descriptors
Depth was measured at each microhabitat sampled for tadpoles, and we used the maximal depth recorded to describe each pond (Both et al. 2014). Pond areas were measured at the two sampling events. To characterize the surrounding habitat for each pond, we used the distance to the closest forest fragment and accounted for the land use within a 400 m radius of each site. Land use classifications used were: grazing, crops, protected (nature reserves), or leisure (private properties without economic activities).
Phylogenetic tree
Knowledge of amphibian evolutionary relationships has improved greatly in the last 10 y, and major phylogenies are now available (Pyron and Wiens 2011; and see references herein). We opted to use the phylogenetic hypothesis of Pyron and Wiens (2011), as well as their proposed taxonomy. This hypothesis is based on molecular information for more than 2,800 species belonging to more than 400 genera. We extracted our subtree, including branch length information, in the R environment using the function prune.sample of the picante package (Kembel et al. 2012). Some taxa were not present in the source tree, and these were included in our subtree by collapsing terminal branches of the most closely related taxa (Fig. 1).
Data analyses
We tested our hypothesis that invaded and non-invaded sites would show distinct taxonomic and phylogenetic compositions through Permutational Multivariate Analysis of Variances (PERMANOVA) based on distances (Anderson 2001). We also tested PERMANOVA models accounting for potential confounding factors associated with invaded and non-invaded ponds: (1) distance to the next fragment, (2) land use (protected, leisure, crops, grazing), (3) pond type (natural, artificial) and (4) pond area. Tests were computed using Sørensen (taxonomic composition) and Phylosor (phylogenetic-weighted composition) dissimilarity matrices (Gower 1971; Bryant et al. 2008). For all models, we computed 9,999 permutations stratified within each study area. We used Non-metric Multidimensional Scaling (NMDS) to visually explore the variation of community composition among invaded and non-invaded ponds (Legendre and Legendre 1998).
Phylogenetic and taxonomic composition structure may not differ between invaded and non-invaded sites, even though they still may show distinct variability in composition (i.e., beta diversity). We assessed whether the invaded communities are more homogeneous in terms of variations in taxonomic and phylogenetic composition than non-invaded ones, using a test of homogeneity of multivariate dispersions (Anderson 2006). The test obtains centroids of the multivariate dispersions of invaded and non-invaded ponds and calculates the mean distance of ponds from their respective group centroid. Thus, we could test whether invaded sites show more homogeneous compositions (small distances from their group centroid) than non-invaded sites. The homogeneity tests were also computed on the same Sørensen and Phylosor dissimilarity matrices, and the significance was evaluated through 9,999 permutations stratified among the three study areas.
We assessed if the invaded sites harbor nested compositions in relation to non-invaded sites. We used the Nestedness metric based on Overlap and Decreasing Fill (NODF; Almeida-Neto et al. 2008) to evaluate nestedness in taxonomic composition, and a NODF extension, the phyloNODF (Melo et al. 2014), for phylogenetic composition. Both metrics order communities (e.g., rows of a site-by-species matrix) by a hypothesis supposedly to cause decreasing diversity, and compare if the hypothesized diversity-poor communities in fact show lower diversity than the hypothesized diversity-rich communities. If so, NODF quantifies the percentage of diversity in the diversity-poor communities that is shared with the diversity-rich communities. For the NODF, diversity is expressed as species richness. For the phyloNODF, diversity is expressed as phylogenetic diversity (PD, Faith 1992). In case the supposedly PD-poor communities in fact show lower PD than the supposedly PD-rich communities, phyloNODF is simply the percentage of the branch-lengths of the PD-poor communities that is shared with the PD-rich communities. Ponds may show nested faunas due to a number of other factors, such as size, depth and structural complexity, and these factors may add noise to the detection of the hypothesized nested effect caused by the invasive species. Therefore, we restricted nestedness analyses to pairs of ponds, one invaded and one non-invaded, as similar in size and depth as possible and no more than 20 km apart. Among the 57 ponds, we were able to select 15 pond pairs. Our hypothesis was that anuran faunas in invaded ponds are a subset of those present in non-invaded ponds. Accordingly, the first row of each of our 15 two-row matrices included the non-invaded pond (the hypothetically most species- or PD-rich pond). Null models for a two-row matrix should be very conservative, as only a limited number of randomized combinations are possible. Also, our hypothesis was general and not specific to each pair of ponds. Accordingly, we used an approach developed by Schneck et al. (2011) in which a one-tailed paired t test is used to compare observed nestedness to the mean of expected values according to a null model. These expected values were obtained from 9,999 runs of the equiprobable–equiprobable null model. All analyses were performed in the R environment (The R Development Core Team 2012).
Results
The species pool was composed of 34 anuran species, belonging to six families: Bufonidae, Hylidae, Alsodidae, Leptodactylidae, Leiuperidae and Microhylidae. The region A3 included two exclusive genera, Sphaenorhyncus and Bokermannohyla, although they belong to family Hylidae, represented in all areas (see Online Resource 2).
The NMDS ordination using taxonomic data indicated that area A3 was slightly different from the other two areas (Fig. 2a). However, the anuran compositions in invaded and non-invaded sites were similar within each study area. Similarly, the ordination for the phylogenetic data did not separate the invaded from the non-invaded ponds (Fig. 2b). The db-Manovas assessing anuran communities in invaded and non-invaded ponds did not show distinct taxonomic and phylogenetic compositions (F1,53 = 1.23, R2 = 0.01, p = 0.28; F1,55 = 0.83, R2 = 0.01, p = 0.66, respectively). Models comparing invaded and non-invaded ponds while accounting for habitat and pond features also show no effect of bullfrog invasion upon taxonomic or phylogenetic composition of anurans (F1,48 = 0.95, R2 = 0.01, p = 0.45; F1,48 = 0.71, R2 = 0.01, p = 0.72, respectively). Distance to the closest fragment (F1,4 8 = 2.16, R2 = 0.03, p = 0.05) and land use categories (grazing: F1,48 = 2.20, R2 = 0.03, p = 0.04; leisure: F1,48 = 4.34, R2 = 0.06, p < 0.001; protected: F1,48 = 4.38, R2 = 0.06, p < 0.001) partially explained taxonomic composition. No habitat or pond descriptor explained phylogenetic composition.
Ordination plots showing the first two axes of a NMDS analysis for taxonomic (a) and phylogenetic compositions (b) of 57 anuran communities in three study areas in the Atlantic Forest. Black circles represent anuran compositions from sites invaded by bullfrogs, and open circles represent compositions from non-invaded sites. Solid black polygon delimits distribution of communities from study area A1, hatched gray polygon delimits communities from A2, and hatched black polygon delimits communities from A3
Communities from invaded and non-invaded sites also did not differ regarding beta diversity for both data types analyzed (F1,55 = 0.37, p = 0.49; F1,55 = 0.20, p = 0.64; respectively for taxonomic and phylogenetic data; Fig. 3). Non invaded sites from study area A1 showed a remarkably low phylogenetic beta diversity when compared with invaded sites of the same area (Fig. 3). This pattern was caused because only a few clades were repeatedly present in these non-invaded sites. The hypothesis that invaded ponds harbor a nested subset of the species present in similar and nearby ponds was not corroborated (t1,14 = 0.217, p = 0.42; t1,14 = 0.942, p = 0.18, respectively for taxonomic and phylogenetic data).
Principal Coordinates Analyses of anuran communities from invaded and non-invaded ponds in three study areas (A1, A2, A3) of the Atlantic Forest. Taxonomic composition ordinations are displayed on the left, and phylogenetic composition ordinations are presented on the right. Black circles indicate the centroid of invaded sites, which are represented by open triangles. Gray circles indicate the centroids of non-invaded sites, which are represented by open circles. Polygons indicate the maximum dispersion of communities in each group
Discussion
The presence of established populations of invasive species has been linked to changes in native community structure, and many examples come from freshwater ecosystems (Moyle and Light 1996; Ricciardi and MacIsaac 2011). Most amphibians breed in freshwaters and are known to be affected by invasive fishes and amphibians (Kraus 2009). The American bullfrog is an aquatic amphibian with high fecundity rates and phenotypic plasticity, and is known to be a superior competitor in both its larval and adult life-history stages (Kats and Ferrer 2003). For these reasons, we hypothesized that we would find different anuran compositions in ponds with and without established bullfrog populations. However, we did not find evidence that bullfrog invasion affects the anuran taxonomic or phylogenetic composition in invaded ponds in the Atlantic Forest. The invaded ponds were not more homogeneous in species or clade compositions, and were not nested subsets of non-invaded ponds.
Taxonomic composition of anuran communities could only be explained by habitat descriptors. However, phylogenetic composition could not be associated with any predictors. These poor relationships between compositions and the environmental gradients were expected, since we only used waterbodies which potentially could support breeding populations of bullfrogs: ponds which hold water throughout the seasons and are more than 50 cm deep. Therefore, we partially controlled for pond permanence-area-depth gradients, which are known to be the main factors structuring amphibian communities in ponds (Wellborn et al. 1996). Using a higher number/kind of waterbodies, Both et al. (2014) assessed the covariation structure between spatial, landscape and pond feature descriptors explaining amphibian richness in these three areas and found that pond features are the main determinants of anuran richness in Southern Atlantic Forest. Here, we could associate composition changes with habitat gradients describing land use and distance to fragments. Interestingly, even in the presence of such gradients, bullfrogs did not affect anuran composition in ponds.
Most of the studies reporting bullfrog impacts on native amphibian communities were done in the northern hemisphere (Adams 1999; Adams and Wasson 2000; Pearl et al. 2004; Li et al. 2011). In these regions, bullfrogs find congeneric or confamilial species, and the similarity shared with them may result in direct species interactions and consequent displacement, reduction in fitness, or even extinction of native species at local scales (Kats and Ferrer 2003; Kraus 2009; and references therein). Indeed, alien amphibians have been successful colonizers in places where they find congeneric species, with an increased probability of establishment where congeneric richness is higher (Tingley et al. 2011). Alternatively, the lack of apparent effects of bullfrog populations on anuran communities in the Atlantic Forest may be explained by the limiting similarity hypothesis (MacArthur and Levins 1967). Specifically, bullfrogs may not affect anuran compositions in Atlantic Forest ponds because they exploit different resources in a complementary way compared to native species. Actually, this situation is predicted according to the stochastic niche theory proposed by Tilman (2004), who postulated that if an invasive species can survive, grow and reproduce using resources left unconsumed by the local community, it can overcome initial demographic stochastic filters and be “accepted” in the community (see also Stohlgern et al. 2006).
On the other hand, the opposite situation has also been proposed, where invasive species sharing less similarity with natives take advantage of their novelty, attaining high invasiveness and changing resident communities. This idea was first proposed by Darwin (1859) and is known as Darwin’s naturalization hypothesis. Strauss et al. (2006) tested if relatedness between native and invasive grass species influences the extent of impact on native communities, by comparing mean relatedness and invasiveness categories. They observed that invasive grasses that are less related to native communities were more invasive, and concluded that these grasses have more impact on native communities. It is important to note, however, that the bullfrog has been successful in invading areas containing both distantly and closely related species, becoming invasive in almost all areas compatible with their fundamental niche (Ficetola et al. 2007). The success of this species as an invader in such a wide range of conditions is probably due to multiple introduction events and/or human efforts, which assure multiple propagules and allow the species to overcome stochastic filters. In this study, however, we are not assuming that “invasiveness” means “impact.” Such an association between invasiveness and impact may be a valid route for some alien organisms, but not necessarily for all. Amphibians may be highly invasive and do not necessarily affect some native communities (present study; Greenlees et al. 2006; Brown et al. 2011).
Biotic homogenization driven by invasion has been reported for several communities facing different alien species. This process is expected to increase the similarity of communities at local scales, i.e., decrease beta diversity in invaded sites (Olden and Poff 2003). We were not able to detect the occurrence of biotic homogenization in this study, since the beta diversity among anuran communities did not differ between invaded and non-invaded sites. We suspect that this lack of difference between invaded and non-invaded sites could be partially due to the current status of the Atlantic Forest biome, which is highly fragmented, with dense human populations (Ribeiro et al. 2009). We speculate that the anuran communities studied are insensitive to the bullfrog invasion because they are already composed of resistant species, able to maintain viable populations in the face of fragmentation and anthropic activities. In fact, bullfrogs are benefited by such conditions (Adams et al. 2011), and disturbance gradients may be linked to native/alien coexistence (Hobbs and Huenneke 1992; Tilman 2004). In this study, composition changes occurred across habitat gradients describing land use and distance to fragments, but were not explained by bullfrog invasion. Ricotta et al. (2012) studied the effects of two different groups of alien plants, archaeophytes and neophytes, on phylo-betadiversity of native flora in urban landscapes in Europe. They found that only invasive archaeophyte species caused homogenization. Invasive species of neophytes were, in fact, associated with diversification. They attributed the contrasting effects of the two groups of invasive species on diversity to the time of introduction, region of origin, and association (or not) with anthropic activities. These results reinforce the possibility that the impacts of invasion may depend on the attributes of both the invasive and native species.
Moreover, we should not discard the hypothesis that the bullfrog changed the anuran compositions in these ponds in the past, displacing some taxa or clades. For instance, one candidate group that suffered displacement by bullfrogs is the species of Pseudis, which could be expected to occur in two of our study areas but were not encountered. Juveniles of bullfrogs are morphologically similar to adults of Pseudis. Also, species of Pseudis share with bullfrogs the aquatic habit (i.e., remain mostly within the waterbodies, not only in breeding seasons), and also have large tadpoles. The lack of historical sampling is the major challenge for understanding diversity changes at local scales (Sax and Gaines 2003). Similarly, we cannot say that bullfrog populations are not affecting communities at present, since they can do it through subtle processes that may result in community changes over the long term. For instance, in a previous study in the same ponds surveyed in this study, we found that bullfrogs have the potential to affect the acoustic niches of native species (Both and Grant 2012), which is directly linked to sexual selection in anurans and could affect evolutionary constraints over the long term (Ryan 1988). A series of consecutive studies has provided indications that bullfrogs may be effective vectors of lethal diseases, suggesting that they could contribute to amphibian species declines (Daszak et al. 2003; Toledo et al. 2006; Schloegel et al. 2009).
In conclusion, we did not find any evidence that invasive bullfrogs are causing changes in anuran compositions in the Atlantic Forest. This result was unexpected in view of the long list of impacts of bullfrogs on native frogs in other invaded areas around the world. Such contrasting results deserve further attention, since they may be linked to the phylogenetic structure of the invaded communities and the degree of relatedness of their species to bullfrogs. In the Atlantic Forest communities, bullfrogs find suitable climatic conditions, and may face low biotic resistance, exploiting available resources. This creates a situation where the species is similar enough to cross the environmental filter but divergent enough to avoid competition. In addition, the coexistence of bullfrogs and native species may be mediated through frequent disturbances and anthropic gradients.
References
Adams MJ (1999) Correlated factors in amphibian decline: exotic species and habitat change in western Washington. J Wildl Manage 63:1162–1171. doi:10.2307/3802834
Adams MJ (2000) Pond permanence and the effects of exotic vertebrates on anurans. Ecol Appl 10:559–568. doi:10.1890/1051-0761(2000)010[0559:PPATEO]2.0.CO;2
Adams MJ, Pearl CA, Galvan S, Mccreary B (2011) Non-native species impacts on pond occupancy by an anuran. J Wildl Manage 75:30–35. doi:10.1002/jwmg.29
Almeida-Neto M, Guimarães P, Guimarães PR, Loyola RD, Ulrich W (2008) A consistent metric for nestedness analysis in ecological systems: reconciling concept and measurement. Oikos 117:1227–1239. doi:10.1111/j.2008.0030-1299.16644.x
Anderson MJ (2001) Permutation tests for univariate or multivariate analysis of variance and regression. Can J Fish Aquat Sci 58:626–639. doi:10.1139/cjfas-58-3-626
Anderson MJ (2006) Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62:245–253. doi:10.1111/j.1541-0420.2005.00440.x
Blackburn TM, Pyšek P, Bacher S et al (2011) A proposed unified framework for biological invasions. Trends Ecol Evol 26:333–339. doi:10.1016/j.tree.2011.03.023
Both C, Grant T (2012) Biological invasions and the acoustic niche: the effect of bullfrog calls on the acoustic signals of white-banded tree frogs. Biol Lett 8:714–716. doi:10.1098/rsbl.2012.0412
Both C, Kaefer IL, Santos TG, Cechin STZ (2008) An austral anuran assemblage in the Neotropics: seasonal occurrence correlated with photoperiod. J Nat Hist 42:205–222. doi:10.1080/00222930701847923
Both C, Lingnau R, Santos-Jr A et al (2011) Widespread occurrence of the american bullfrog, Lithobates catesbeianus (Shaw, 1802) (Anura: Ranidae), in Brazil. South Am J Herpetol 6:127–134. doi:10.2994/057.006.0203
Both C, Madalozzo B, Lingnau R, Grant T (2014) Amphibian richness patterns in Atlantic Forest areas invaded by American bullfrogs. Austral Ecol. doi:10.1111/aec.1215
Brown GP, Phillips BL, Shine R (2011) The ecological impact of invasive cane toads on tropical snakes: field data do not support laboratory-based predictions. Ecology 92:422–431. doi:10.1890/10-0536.1
Bryant JB, Lamanna C, Morlon H, Kerkhoff AJ, Enquist BJ, Green JL (2008) Microbes on mountainsides: contrasting elevational patterns of bacterial and plant diversity. Proc Natl Acad Sci USA 105:11505–11511. doi:10.1073/pnas.0801920105
Cianciaruso MV, Silva IA, Batalha MA (2009) Diversidades filogenética e funcional: novas abordagens para a Ecologia de comunidades. Biota Neotrop 9:93–103
The R Development CoreTeam (2012) R:A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. Available from URL: http://www.R-project.org
Darwin C (1859) On the origin of species by means of natural selection. Murray, London
Daszak P, Cunningham AA, Hyatt AD (2003) Infectious disease and amphibian population declines. Divers Distrib 9:141–150. doi:10.1046/j.1472-4642.2003.00016.x
Duncan RP, Blackburn TM, Sol D (2003) The ecology of bird introductions. Annu Rev Ecol Evol Syst 34:71–98. doi:10.1146/annurev.ecolsys.34.011802.132353
Faith DP (1992) Conservation evaluation and phylogenetic diversity. Biol Conserv 61:1–10. doi:10.1016/0006-3207(92)91201-3
Ficetola GF, Thuiller W, Miaud C (2007) Prediction and validation of the potential global distribution of a problematic alien invasive species—the American bullfrog. Divers Distrib 13:476–485. doi:10.1111/j.1472-4642.2007.00377.x
Fundação SOS Mata Atlântica, INPE (2009) Atlas dos remanescentes florestais da Mata Atlântica, período 2005–2008. São Paulo
Giovanelli JGR, Haddad CFB, Alexandrino J (2008) Predicting the potential distribution of the alien invasive American bullfrog (Lithobates catesbeianus) in Brazil. Biol Invas 10:585–590. doi:10.1007/s10530-007-9154-5
Gower JC (1971) A general coefficient of similarity and some of its properties. Biometrics 27:857–871. doi:10.2307/2528823
Greenlees MJ, Brown GP, Webb JK et al (2006) Effects of an invasive anuran [the cane toad (Bufo marinus)] on the invertebrate fauna of a tropical Australian floodplain. Anim Conserv 9:431–438. doi:10.1111/j.1469-1795.2006.00057.x
Hobbs RJ, Huenneke LF (1992) Disturbance, diversity, and invasion: implications for conservation. Conserv Biol 6:324–337. doi:10.1046/j.1523-1739.1992.06030324.x
IBGE Instituto Brasileiro de Geografia e Estatística (2004) Mapa de vegetação do Brasil. http://www.ibge.gov.br/home/geociencias/default_prod.shtm. Accessed on 1 June 2012
Kaefer IL, Boelter RA, Cechin SZ (2007) Reproductive biology of the invasive bullfrog. Ann Zool Fenn 44:435–444
Kats LB, Ferrer RP (2003) Alien predators and amphibian declines: review of two decades of science and the transition to conservation. Divers Distrib 9:99–110. doi:10.1046/j.1472-4642.2003.00013.x
Kembel SW, Ackerly DD, Blomberg SP, Cornwell WK, Cowan PD, Helmus MR, Morlon H, Webb CO (2012) Picante, R tools for integrating phylogenies and ecology. R package version 1.5-2
Kraus F (2009) Alien reptiles and amphibians: a scientific compendium and analysis. Springer, The Netherlands
Legendre P, Legendre L (1998) Numerical ecology, 2nd edn. Elsevier, Amsterdam
Lever C (2003) Naturalized reptiles and amphibians of the world. Oxford University Press, Oxford
Li Y, Ke Z, Wang Y, Blackburn TM (2011) Frog community responses to recent American bullfrog invasions. Curr Zool 57:83–92
Lockwood JL (2006) Life in a Double-Hotspot: the transformation of Hawaiian passerine bird diversity following invasion and extinction. Biol Invas 8:449–457. doi:10.1007/s10530-005-6415-z
Lucas EM, Fortes VB (2008) Frog diversity in the Floresta Nacional de Chapecó, Atlantic Forest of southern Brazil. Biota Neotrop 8:51–61. doi:10.1590/S1676-06032008000300004
MacArthur R, Levins R (1967) The limiting similarity, convergence, and divergence of coexisting species. Am Nat 101:377–385. doi:10.2307/2459090
Melo AS, Cianciaruso MV, Almeida-Neto M (2014) treeNODF: nestedness to phylogenetic, functional and other tree-based diversity metrics. Method Ecol Evol 5:563–572
Moyle PB, Light T (1996) Biological invasions of freshwater: empirical rules and assembly theory. Biol Conserv 78:149–161. doi:10.1016/0006-3207(96)00024-9
Nori J, Urbina-Cardona JN, Loyola RD et al (2011) Climate change and American bullfrog invasion: what could we expect in South America? PLoS ONE 6:e25718. doi:10.1371/journal.pone.0025718
Olden JD, Poff NL (2003) Toward a mechanistic understanding and prediction of biotic homogenization. Am Nat 162:442–460. doi:10.1086/378212
Pearl CA, Adams MJ, Bury RB, McCreary B (2004) Asymmetrical effects of introduced bullfrogs (Rana catesbeiana) on native ranid frogs in Oregon. Copeia 2004:11–20. doi:10.1643/CE-03-010R2
Piazzi L, Balata D (2008) Invasion of alien macroalgae in different Mediterranean habitats. Biol Invas 11:193–204. doi:10.1007/s10530-008-9224-3
Pyron RA, Wiens JJ (2011) A large-scale phylogeny of Amphibia including over 2,800 species, and a revised classification of extant frogs, salamanders, and caecilians. Mol Phylogenet Evol 61:543–583. doi:10.1016/j.ympev.2011.06.012
Pyšek P, Richardson DM (2010) Invasive species, environmental change and management, and health. Annu Rev Environ Resour 35:25–55. doi:10.1146/annurev-environ-033009-095548
Qian H, Ricklefs RE (2006) The role of exotic species in homogenizing the North American flora. Ecol Lett 9:1293–1298. doi:10.1111/j.1461-0248.2006.00982.x
Ribeiro MC, Metzger JP, Martensen AC, Ponzoni FJ, Hirota MM (2009) The Brazilian Atlantic Forest: how much is left, and how is the remaining forest distributed? Implications for conservation. Biol Conserv 142:1141–1153. doi:10.1016/j.biocon.2009.02.021
Ricciardi A (2007) Are modern biological invasions an unprecedented form of global change? Conserv Biol 21:329–336. doi:10.1111/j.1523-1739.2006.00615.x
Ricciardi A, MacIsaac HJ (2011) Impacts of biological invasions on freshwater ecosystems. In: Richardson DM (ed) Fifty years of invasion ecology: the legacy of Charles Elton. Wiley-Blackwell, Oxford, pp 211–224
Ricotta C, La Sorte FA, Pyšek P et al (2012) Phylogenetic beta diversity of native and alien species in European urban floras. Glob Ecol Biogeogr 21:751–759. doi:10.1111/j.1466-8238.2011.00715.x
Ryan MJ (1988) Constraints and patterns in the evolution of anuran acoustic communication. In: Fritzsch B, Ryan M, Wilczynski W et al (eds) The evolution of the amphibian auditory system. Wiley, New York, pp 637–677
Sax DF, Gaines SD (2003) Species diversity: from global decreases to local increases. Trends Ecol Evol 18:561–566. doi:10.1016/S0169-5347(03)00224-6
Schloegel LM, Picco AM, Kilpatrick AM et al (2009) Magnitude of the US trade in amphibians and presence of Batrachochytrium dendrobatidis and ranavirus infection in imported North American bullfrogs (Rana catesbeiana). Biol Conserv 142:1420–1426. doi:10.1016/j.biocon.2009.02.007
Schneck F, Schwarzbold A, Melo AS (2011) Substrate roughness affects stream benthic algal diversity, assemblage composition, and nestedness. J N Am Benthol Soc 30:1049–1056. doi:10.1899/11-044.1
Scott J, Woodward BD (1994) Standard techniques for inventory and monitoring: surveys at breeding sites. In: Heyer WR, Donnelly MA, McDiarmid RW et al (eds) Measuring and monitoring biological diversity, standard methods for amphibians. Smithsonian Institution Press, Washington, pp 92–96
Shaffer HB, Alford RA, Woodward BD, Richards SJ, Altig RG, Gascon C (1994) Quantitative sampling of amphibian larvae. In: Heyer WR, Donnelly MA, McDiarmid RW et al (eds) Measuring and monitoring biological diversity, standard methods for amphibians. Smithsonian Institution Press, Washington, DC, pp 130–141
Simberloff D, Parker IM, Windle PN (2005) Introduced species policy, management, and future research needs. Front Ecol Environ 3:12–20. doi:10.2307/3868440
Stenert C, Bacca RC, Moraes AB et al (2012) Negative effects of exotic pine invasion on macroinvertebrate communities in southern Brazil coastal ponds. Mar Freshw Res 63:283–292. doi:10.1071/MF11169
Stohlgern TJ, Jarnevich C, Chong GW, Evangelista PH (2006) Scale and plant invasions: a theory of biotic acceptance. Preslia 78:405–426
Strauss SY, Webb CO, Salamin N (2006) Exotic taxa less related to native species are more invasive. Proc Natl Acad Sci USA 103:5841–5845. doi:10.1073/pnas.0508073103
Tilman D (2004) Niche tradeoffs, neutrality, and community structure: a stochastic theory of resource competition, invasion, and community assembly. Proc Natl Acad Sci USA 101:10854–10861. doi:10.1073/pnas.0403458101
Tingley R, Phillips BL, Shine R (2011) Establishment success of introduced amphibians increases in the presence of congeneric species. Am Nat 177:382–388. doi:10.1086/658342
Toledo LF, Britto FB, Araújo OGS, Giasson LMO, Haddad CFB (2006) The occurrence of Batrachochytrium dendrobatidis in Brazil and the inclusion of 17 new cases of infection. South Am J Herpetol 1:185–191
Trentanovi G, von der Lippe M, Sitzia T, Ziechmann U, Kowarik I, Cierjacks A (2013) Biotic homogenization at the community scale: disentangling the roles of urbanization and plant invasion. Divers Distrib 19:738–748. doi:10.1111/ddi.12028
Wellborn GA, Skelly DK, Werner EE (1996) Mechanisms creating community structure across a freshwater habitat gradient. Annu Rev Ecol Syst 27:337–363. doi:10.1146/annurev.ecolsys.27.1.337
Winter M, Schweiger O, Klotz S, Nentwig W, Andriopoulos P, Arianoutsou M, Basnou C, Delipetrou P, Didziulis V, Hejda M, Hulme PE, Lambdon PW, Pergl J, Pysek P, Roy DB, Kühn I (2009) Plant extinctions and introductions lead to phylogenetic and taxonomic homogenization of the European flora. Proc Natl Acad Sci USA 106:21721–21725. doi:10.1073/pnas.0907088106
Zenni RD, Nuñez MA (2013) The elephant in the room: the role of failed invasions in understanding invasion biology. Oikos 122:801–815. doi:10.1111/j.1600-0706.2012.00254.x
Acknowledgments
We are thankful for the helpful comments received by two anonymous reviewers and the editor on a previous version of this manuscript. The Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) provided a post-doctoral fellowship to CB (Proc. No. 159906/2012-1) and a research fellowship to ASM (Proc. No. 307479/2011-0).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Both, C., Melo, A.S. Diversity of anuran communities facing bullfrog invasion in Atlantic Forest ponds. Biol Invasions 17, 1137–1147 (2015). https://doi.org/10.1007/s10530-014-0783-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-014-0783-1