Abstract
Objective
To design and synthesize a novel near-infrared (NIR) fluorescent probe based on indocyanine Green (ICG), that can be applied in imaging living cells.
Results
A highly fluorescent novel NIR fluorescent probe (IR-793) was synthesized in two steps. IR-793 had better fluorescence and optical stability than ICG. In addition, no obvious cytotoxicity effect of IR-793 was observed and cell viability was above 75% at the maximum concentration (120 nM). IR-793 also exhibited good performance in imaging living A549 cells.
Conclusion
IR-793, a novel NIR fluorescent probe that is stable, low-cost, highly fluorescent and low cytotoxicity, has been designed and synthesized for imaging living cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Near-infrared (NIR) fluorescence imaging is a powerful tool in cell biology, pharmacology, and disease diagnosis (Mather 2009). NIR probes fluoresce from 600 to 900 nm, which can decrease background fluorescence and allow deep tissue penetration (Zhang et al. 2010). Therefore, NIR fluorescent probes are applied in disease diagnosis and biomarker development (Rasmussen et al. 2009).
At present, only a few NIR imaging probes have received approval for clinical use from FDA (Luo et al. 2011). Indocyanine Green (ICG), which is one of the NIR fluorescent probes approved by FDA, has low toxicity, negligible side effects, and is widely used for monitoring cardiac function, hepatic output, and retinal angiography. In addition, ICG has been recently used as a potential photosensitizer (Mamoon et al. 2009; Funayama et al. 2012; Sevick-Muraca 2012). However, the applications of ICG have several limitations, including low quantum yield, optical instability in the body, and unrestrained leakage in blood vessels. Thus, many research groups have focused on the synthesis of ICG derivatives to improve the brightness, solubility, photostability, and optical stability for both in vitro and in vivo imaging (Xu et al. 2007; Chang et al. 2008).
We have designed and synthesized various ICG derivatives as NIR imaging probes for non-invasive in vivo imaging research. A series of NIR fluorescence probes, namely, IR-780, IR-789, and MP-Cy, which exhibit high sensitivity, good photostability, and excellent cell membrane permeability, have been identified by our laboratory (Sun et al. 2013, 2014, 2015). In the current work, considering the aforementioned limitations of ICG, we have designed and synthesized a novel NIR fluorescent probe IR-793 which has better water solubility and optical stability in serum when applied in cellular analysis in an aqueous environment. Herein, we describe IR-793, a NIR fluorescent dye with high extinction coefficients, can provide good photostability with high extinction coefficients and show promising application in biomedical diagnosis.
Materials and methods
Materials and instruments
1H-NMR data were recorded using an ACF-300/500 MHz NMR spectrometer (Bruker) under ambient temperature in DMSO-d6, and tetramethylsilane was used as internal standard reference. Molecular weight was determined using a LC/MS system (Agilent). 2,3,3-Trimethylindolenine-5-sulfonic acid, p-methylbenzyl bromide, 2-chloro-1-formyl-3-hydroxymethylenecyclohexene, and ICG were purchased from Sigma-Aldrich (Shanghai). The A549 cell line (human lung adenocarcinoma epithelial cell line) was obtained from ATCC. All reagents and solvents were commercially available and used without further purification, unless otherwise indicated.
Synthesis and characterization of IR-793
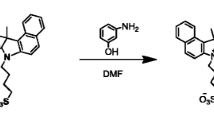
Scheme 1 shows the synthetic route of the designed heptamethine cyanine dye IR-793. Compound 1, 2,3,3-trimethylindolenine-5-sulfonic acid (0.68 g, 2.45 mmol), and compound 2, p-methylbenzyl bromide (0.54 g, 2.96 mmol), were dissolved in 10 ml toluene. The solution was stirred under argon for 12 h at 85 °C. Vacuum filtration was performed to isolate the resulting precipitate and obtain a crude solid that was recrystallized to yield compound 3, which is a pink solid (0.70 g, yield 83.1 %). Compound 3, 1-p-methylbenzyl bromide-2,3,3-trimethylindolenine-5-sulfonic acid (0.65 g, 1.90 mmol), compound 4, 2-chloro-1-formyl-3-hydroxymethylenecyclohexene (0.16 g, 0.95 mmol), and anhydrous sodium acetate (0.90 mmol, 0.16 g) were dissolved in 16 ml acetic anhydride. The solution was stirred under argon for 1 h at 75 °C. The resulting solution was added into the methyl tert-butyl ether to yield a crude solid. The crude residue was purified via column chromatography on silica gel using dichlormethane/methanol (10:1, Rf = 0.40), which yielded IR-793 (240 mg, 30.8 %) as a dark green solid. The IR-793 fractions of high chemical purity were collected and tested through 1H-NMR spectroscopy using ACF-300/500 MHz NMR spectrometers. Then, the compound of IR-793 was further evaluated with a quadrupole time-of-flight LC/MS system.
Absorption spectra
Absorption spectra were obtained using a UV–Vis spectrophotometer at room temperature and within the spectral range of 600–900 nm. Probe IR-793 (1 ml, 0.01 mM) and ICG (1 ml, 0.01 mM) were studied via absorption spectroscopy in PBS, methanol solutions, or serum.
Fluorescence spectra
Fluorescence spectra were determined using a spectrofluorimeter. Probe IR-793 (0.1 ml, 0.01 mM) and ICG (0.1 ml, 0.01 mM) were separately added into the 1 ml color comparison tubes and studied through fluorescence spectroscopy in PBS, methanol solutions, or serum.
Optical stability
The optical stability of IR-793 in serum was determined through the previously reported method that used fluorescence spectroscopy (799 nm excitation, continuous wavelength of 600–900 nm emission) (Nakayama et al. 2003; Zhang et al. 2010). A 10 μM IR-793 was incubated in fetal bovine serum at 37 °C for 2 h to evaluate the stability in serum. Fluorescence intensities were detected in 10 ml color comparison tubes using a Cary Eclipse spectrofluorometer (740 nm excitation, 825 nm emission) every 10 min for 1 h, then at 2 h. All the experiments were performed in triplicates, and data were plotted as mean values ± SD.
Cell culture
Human lung cancer cells (A549) were cultured in RPMI-1640 supplemented with 10 % (v/v) fetal calf serum, 1 % penicillin, and 1 % streptomycin. Cells were grown routinely in a monolayer culture at 37 °C under 5 % CO2 humidified atmosphere in T25 cell culture flasks. Cells were split in 1:3 ratios at 80 % confluence.
Cytotoxicity assay
MTT assay was conducted to assess the cytotoxicity of IR-793. The A549 cells (5000 cells per well) were seeded in 96-well plates for 24 h before treatment. The culture medium in each well was replaced with 200 μl IR-793-supplemented treatment medium. Various concentrations of 10, 20, 40, 60, 80, and 120 nm IR-793 were tested. A medium without IR-793 was used as the control. Cells were further incubated for 24 and 48 h. After the incubation period, the cells were washed thrice with PBS before adding 100 μl MTT working solution at 0.5 mg ml−1, followed by incubation for 4 h at 37 °C with 5 % (v/v) CO2. The remaining MTT solution was removed after 4 h, and 100 ml DMSO was added into each well to dissolve the formazan crystals, followed by incubation at 37 °C with 5 % CO2 for 15 min. The absorbance of each well at 490 nm was recorded using a plate reader. Cell viability was determined by obtaining the signal from the treated groups over the control groups without irradiation. All experiments were performed in triplicates, and data were plotted as mean values ± SD.
Confocal imaging in living cells
A549 cells were grown on glass-bottom culture dishes. After incubation with IR-793 (10 µM) and ICG (10 µM) in FBS-free 1640 medium at 37 °C for 30 min and washing thrice with PBS buffer (pH 7.4), the cells were immediately imaged using a confocal microscope with an objective lens (×20). Excitation was at 793 nm when the cells were incubated with IR-793 and 805 nm when the cells were incubated with ICG.
Results and discussion
Characterization of IR-793
IR-793 was synthesized using a simple method (Scheme 1). The structure of IR-793 was confirmed via 1H-NMR and MS. 1H-NMR (400 MHz, CD3OD): δ(ppm) = 1.78 (s, 14H), 2.32 (s, 6H), 2.53–2.56 (t, J = 6 Hz, 4H), 5.40 (s, 4H), 6.32–6.35 (d, J = 14 Hz, 2H), 7.16–7.22 (m, 8H), 7.35–7.37 (d, J = 8.4 Hz, 2H), 7.87–7.89 (d, J = 8.4 Hz, 2H), 7.97 (s, 1H), 8.39–8.43 (d, J = 14.4 Hz); time-of-flight MS m/z: 822.2[M] (1H-NMR and MS spectra of IR-793, as shown in Supplementary Fig. 1) confirmed the formation of IR-793.
Optical properties of IR-793
We detected the absorption and emission spectrum of IR-793 in methanol, serum, and PBS (Fig. 1a, b). The maximal absorption and emission wavelength of IR-793 in serum showed a red shift compared with that in methanol and PBS, as shown in Fig. 1a. The emission intensity was considerably higher in serum than that in methanol and PBS, as shown in Fig. 1b. The absorption and emission peak of IR-793 in different media (methanol, serum, and PBS) were in the NIR region (600–900 nm). The excitation and emission spectra of IR-793 were located at 793 and 830 nm in methanol, 779 and 810 nm in PBS, and 799 and 823 nm in serum, which emitted a similar emission peak in methanol to Indocyanine Green (ICG), as shown in Fig. 1c, d. Moreover, the total fluorescence intensities of IR-793 and ICG in the serum were maintained at a stable level for 120 min, which indicated that IR-793 was stable similar to ICG in the serum (Fig. 2). However, the fluorescence intensity of IR-793 in the serum was higher than that of the ICG at the same concentration. These results suggested that IR793 had promising potential as a NIR probe in biomedical imaging.
Cytotoxicity of IR-793
When using probes in living cells, their biocompatibility is always the first property to consider (Funayama et al. 2012). Therefore, cytotoxicity was measured via MTT assay to quantify the living A549 cells in the wells after incubation with various concentrations of IR-793. The results indicated that the cell viability of A549 cells was not altered by IR-793 unlike with ICG, even in the wells with the highest probe concentration (120 nm). The bar graph in Fig. 3 indicated cell viability to be above 75 % at the maximum concentration of 120 nm after 24 and 48 h of incubation. Cytotoxicity assay proved that IR-793 had no apparent toxicity to living cells. These results proved that IR-793 probe was safe in living cells.
The viability of A549 cells cultured with IR-793 in comparison with ICG at the same dose incubated for 24 and 48 h. Solid bars represent ICG, while dotted bars represent IR-793. Data were presented as the mean cell viability with standard deviation after triplicate analysis. P value < 0.05 is indicated by one asterisk
In vitro cell imaging
A confocal microscopy analysis was performed to study the applicability of IR-793 in biological systems compared with ICG. A549 cells were inoculated in IR-793 at a concentration of 10 µM with good cell viability for 30 min. The distribution of the probe within the cells was observed under a fluorescence microscope following excitation at 793 nm. Figure 4 showed that IR-793 and ICG can permeate into the membrane, and the fluorescence intensities varied within single cells. The studies showed that IR-793 demonstrated excellent membrane permeability and good photostability.
Confocal fluorescence images of living cells, A549 cells. a Confocal fluorescence images of living cells A549 incubated with 10 μM IR-793 at 37 °C for 30 min, b bright field image of living A549 cells, c confocal fluorescence images of living cells A549 incubated with 10 μM ICG at 37 °C for 30 min, and d bright field image of living A549 cells
Conclusion
The need to develop new, reliable, and low-cost NIR fluorescence probes for cell imaging and disease diagnosis is increasing. In the present study, we developed a modified procedure for the chemical synthesis of a highly fluorescent, novel NIR fluorescence probe (IR-793) to improve the overall yield for potential large-scale preparation. IR-793 exhibited better water solubility and optical stability in serum than ICG when applied in cellular analysis in an aqueous environment. The excellent water solubility of IR-793 ensured its benefit for in vivo imaging and its biocompatibility with cells (Amiot et al. 2008). IR-793 demonstrated outstanding optical properties and photostability in serum, which allowed living cells with minimal NIR background fluorescence from the hosts to achieve excellent signal-to-background ratio. Furthermore, IR793 had low cytotoxicity to living A549 cells. In conclusion, the aforementioned preliminary evaluation results indicated that IR-793 has higher prospects as a biocompatible NIR fluorescent sensor for sensitive intracellular imaging compared with ICG. This simple and sensitive NIR fluorescent probe will be considerably useful to biomedical researchers on biological systems, such as in the real-time monitoring of cells and image-guided oncologic surgery, as a fluorescent probe with chemically conjugated specific ligands. In our future work, we will focus on systematic investigations of photo-dependent cytotoxic activities to broaden the application of IR-793.
References
Amiot CL, Xu S, Liang S, Pan L, Zhao JX (2008) Near-infrared fluorescent materials for sensing of biological targets. Sensors 8:3082–3105
Chang SK, Rizvi I, Solban N, Hasan T (2008) In vivo optical molecular imaging of vascular endothelial growth factor for monitoring cancer treatment. Clin Cancer Res 14(13):4146–4153
Funayama T, Sakane M, Abe T, Ochiai N (2012) Photodynamic therapy with indocyanine green injection and near-infrared light irradiation has phototoxic effects and delays paralysis in spinal metastasis. Photomed Laser Surg 30:47–53
Luo S, Zhang E, Su Y, Cheng T, Shi C (2011) A review of nir dyes in cancer targeting and imaging. Biomaterials 32:7127–7138
Mamoon AM, Gamal-Eldeen AM, Ruppel ME, Smith RJ, Tsang T, Miller LM (2009) In vitro efficiency and mechanistic role of indocyanine green as photodynamic therapy agent for human melanoma. Photodiagnosis Photodyn Ther 6:105–116
Mather S (2009) Molecular imaging with bioconjugates in mouse models of cancer. Bioconjug Chem 20:631–643
Nakayama A, Bianco AC, Zhang CY, Lowell BB, Frangioni JV (2003) Quantitation of brown adipose tissue perfusion in transgenic mice using near-infrared fluorescence imaging. Mol Imaging 2:37–49
Rasmussen JC, Tan IC, Marshall MV, Fife CE, Sevick-Muraca EM (2009) Lymphatic imaging in humans with near-infrared fluorescence. Curr Opin Biotechnol 20:74–82
Sevick-Muraca EM (2012) Translation of near-infrared fluorescence imaging technologies: emerging clinical applications. Annu Rev Med 63:217–231
Sun C, Wang P, Li L, Zhou G, Zong X, Hu B, Zhang R, Cai J, Chen J, Ji M (2013) A new near-infrared neutral PH fluorescent probe for monitoring minor PH changes and its application in imaging of hepg2 cells. Appl Biochem Biotechnol 172:1036–1044
Sun C, Wu Y, Cai J, Wang P, Zong X, Zhou G, Li L, Ji M (2014) Synthesis of a near-infrared fluorescent probe and its application in imaging of mcf-7 cells. Biotechnol Lett 36:1203–1207
Sun C, Cai J, Chen J, Wu Y, Wang P, Zhou G, Zong X, Chen B, Lv Y, Ji M (2015) The synthesis of a novel near-infrared fluorescent probe and its application in imaging of living cells. Appl Biochem Biotechnol 175:1644–1650
Xu H, Baidoo K, Gunn AJ, Boswell CA, Milenic DE, Choyke PL, Brechbiel MW (2007) Design, synthesis, and characterization of a dual modality positron emission tomography and fluorescence imaging agent for monoclonal antibody tumor-targeted imaging. J Med Chem 50:4759–4765
Zhang C, Liu T, Su Y, Luo S, Zhu Y, Tan X, Fan S, Zhang L, Zhou Y, Cheng T, Shi C (2010) A near-infrared fluorescent heptamethine indocyanine dye with preferential tumor accumulation for in vivo imaging. Biomaterials 31:6612–6617
Acknowledgments
This work is supported by National High-tech Research and Development Project (863 Project, 2013AA032205), Industry Project of Jiangsu Science-technology Support Plan (BE2013840), Science and Technology Development Program of Suzhou (ZXY201412), the Graduate Research and Innovation Program of Jiangsu Province in China (KYLX15-0170).
Supporting information
Supplementary Figure—1H-NMR and MS of IR-793.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xing, J., Zhou, G., Sun, C. et al. Synthesis and characterization of a novel near-infrared fluorescent probe for applications in imaging A549 cells. Biotechnol Lett 38, 1851–1856 (2016). https://doi.org/10.1007/s10529-016-2179-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-016-2179-z