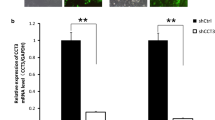
We studied the effect of CCDC3 on the viability of human breast cancer cell line MDA-MB-231. The levels of CCDC3 mRNA and the corresponding protein in MDA-MB-231, MCF-7, T-47D, and HCC1937 cell lines were measured by reverse transcription quantitative real-time PCR and Western blotting. Since MDA-MB-231 cells had higher expression of mRNA CCDC3 and CCDC3 protein, we used this cell line for transfection with small interfering RNA by lentivirus. Cell Counting Kit-8 and clone formation assay were used to detect the effects of CCDC3 knockdown on cell viability; flow cytometry was used to detect the effects of CCDC3 knockdown on cell apoptosis and cell cycle. In MDA-MB-231 cell line, the CCDC3 protein level was significantly down-regulated after CCDC3 knockdown in comparison with the control group (p<0.05). The cell viability and the number of clones in the CCDC3 knockdown group were significantly reduced (p<0.05), while the apoptosis rate significantly increased (p<0.05). Thus, after CCDC3 knockdown, cell viability is weakened in MDA-MB-231 cells, and cell apoptosis rate is increased. Therefore, CCDC3 gene is promising as a new candidate target for BC treatment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fisusi FA, Akala EO. Drug Combinations in Breast Cancer Therapy. Pharm. Nanotechnol. 2019;7(1):3-23. doi: https://doi.org/10.2174/2211738507666190122111224
Chen Z, Wu W, Huang Y, Xie L, Li Y, Chen H, Li W, Yin D, Hu K. RCC2 promotes breast cancer progression through regulation of Wnt signaling and inducing EMT. J. Cancer. 2019;10(27):6837-6847. doi: https://doi.org/10.7150/jca.36430.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021;71(1):7-33. doi: https://doi.org/10.3322/caac.21654
Jin L, Han B, Siegel E, Cui Y, Giuliano A, Cui X. Breast cancer lung metastasis: Molecular biology and therapeutic implications. Cancer Biol. Ther. 2018;19(10):858-868. doi: https://doi.org/10.1080/15384047.2018.1456599
Kobayashi S, Fukuhara A, Taguchi T, Matsuda M, Tochino Y, Otsuki M, Shimomura I. Identification of a new secretory factor, CCDC3/Favine, in adipocytes and endothelial cells. Biochem. Biophys. Res. Commun. 2010;392(1):29-35. doi: https://doi.org/10.1016/j.bbrc.2009.12.142
Ugi S, Maeda S, Kawamura Y, Kobayashi MA, Imamura M, Yoshizaki T, Morino K, Sekine O, Yamamoto H, Tani T, Rokushima M, Kashiwagi A, Maegawa H. CCDC3 is specifically upregulated in omental adipose tissue in subjects with abdominal obesity. Obesity (Silver Spring). 2014;22(4):1070-7. doi: https://doi.org/10.1002/oby.20645
Liao W, Liu H, Zhang Y, Jung JH, Chen J, Su X, Kim YC, Flores ER, Wang SM, Czarny-Ratajczak M, Li W, Zeng SX, Lu H. Ccdc3: A New P63 Target Involved in Regulation Of Liver Lipid Metabolism. Sci Rep. 2017;7(1):9020. doi: https://doi.org/10.1038/s41598-017-09228-8
Eberlein A, Kalbe C, Goldammer T, Brunner RM, Kuehn C, Weikard R. Analysis of structure and gene expression of bovine CCDC3 gene indicates a function in fat metabolism. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2010;156(1):19-25. doi: https://doi.org/10.1016/j.cbpb.2010.01.013
Azad AK, Chakrabarti S, Xu Z, Davidge ST, Fu Y. Coiled-coil domain containing 3 (CCDC3) represses tumor necrosis factor-α/nuclear factor κB-induced endothelial inflammation. Cell. Signal. 2014;26(12):2793-800. doi: https://doi.org/10.1016/j.cellsig.2014.08.025
Emens LA. Breast Cancer Immunotherapy: Facts and Hopes. Clin Cancer Res. 2018;24(3):511-520. doi: https://doi.org/10.1158/1078-0432.CCR-16-3001
Zhang XF, An MZ, Ma YP, Lu YM. Regulatory effects of CCDC3 on proliferation, migration, invasion and EMT of human cervical cancer cells. Eur. Rev. Med. Pharmacol. Sci. 2019;23(8):3217-3224. doi: https://doi.org/10.26355/eurrev_201904_17680
Hill DA, Friend S, Lomo L, Wiggins C, Barry M, Prossnitz E, Royce M. Breast cancer survival, survival disparities, and guideline-based treatment. Breast Cancer Res. Treat. 2018;170(2):405-414. doi: https://doi.org/10.1007/s10549-018-4761-7
Tang T, Guo C, Xia T, Zhang R, Zen K, Pan Y, Jin L. LncCCAT1 Promotes Breast Cancer Stem Cell Function through Activating WNT/β-catenin Signaling. Theranostics. 2019;9(24):7384-7402. doi: https://doi.org/10.7150/thno.37892
Zheng A, Song X, Zhang L, Zhao L, Mao X, Wei M, Jin F. Long non-coding RNA LUCAT1/miR-5582-3p/TCF7L2 axis regulates breast cancer stemness via Wnt/β-catenin pathway. J. Exp. Clin. Cancer Res. 2019;38(1):305. doi: https://doi.org/10.1186/s13046-019-1315-8
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mao, J., Lu, Q., Li, P. et al. CCDC3 Gene Regulates the Proliferation of Breast Cancer Cells. Bull Exp Biol Med 174, 653–658 (2023). https://doi.org/10.1007/s10517-023-05763-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05763-9