Abstract
The worldwide decrease in fishery stocks in recent years is related to the degradation of coastal environments and to the increases in capture effort and extreme natural events. Restocking dilapidated populations with artificial hatchery-reared larvae has arisen in recent years as a viable technique for the reparation and maintenance of stocks of commercially exploited species. One of the first steps in larvae cultivation is the evaluation of an appropriate diet for rearing. We aimed to test viable diets for feeding zoea larvae of the crab Menippe nodifrons and the swimming crab Callinectes danae. We tested five live food treatments, three microalgae (Tetraselmis gracilis, Chaetoceros calcitrans, and Thalassiosira weissflogii), the rotifer Brachionus plicatilis, and Artemia sp. nauplii. The most suitable feeding protocol for rearing M. nodifrons larvae was a combined diet of enriched B. plicatilis up to the zoea III instar with the introduction of hatched Artemia sp. nauplii onwards to the megalopa stage. Larvae of C. danae showed high mortality rates and none achieved the zoea III phase on any of the diets tested. Although our results for C. danae rearing were not conclusive, the data provide additional information on improper rearing diets for this species for future research. Future studies testing other food sources are necessary to improve larvae production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Globally, crab fishery is a vast industry, with ~ 180 million tons of global capture production in 2017 (FAO Fisheries and Aquaculture 2020). However, there is little information related to the real impact caused by industrial fishing on the stocks of exploited species (Da Nóbrega Alves and Nishida 2003; Becker 2008; Castilho-Westphal et al. 2008). Rodrigues et al. (2000) related the decline in the capture of the mangrove crab Ucides cordatus (Linnaeus, 1763) on the coast of São Paulo and Paraná states to a possible decrease of stocks. The same phenomenon is observed throughout the Brazilian coast, in the states of Pará, Paraíba, Bahia, and Santa Catarina (Da Nóbrega Alves and Nishida 2003; Souto 2007; Wunderlich et al. 2008; Freitas et al. 2015). As observed for U. cordatus, a decrease in the stock of many commercially exploited species is expected due to the rise in capture effort from market demand, together with habitat damage and extreme climate events in future decades (Da Nóbrega Alves and Nishida 2003; Bell et al. 2005; Becker 2008; Bell et al. 2008). The captive production of Brachyura can be rather scarce, due to difficulties throughout the species’ life span. The low rate of larval survival is associated with a high predation rate combined with poor environmental tolerance (Silva 2002; Agh and Sorgeloos 2005).
Most marine Brachyuran species exhibit planktotrophic larvae (Pechenik 1999; Anger 2001; Vieira 2006). These species produce a greater number of offspring able to reproduce per unit energy invested, besides increased cannibalism, territorialism, and slow growth (Pechenik 1999). These characteristics can hinder the species’ aquaculture economically and technically. The most common way of exploitation is direct gathering or fishing of wild individuals (Anger 2001; Carvalho 2010).
Due to decreases in the world’s stocks, measures to enhance and conserve the populations of depleted species have been developed. Restocking by artificial hatchery-reared larvae has been emerging as a viable technology of population recovery and conservation of different coastal areas (Silva 2002; Silva 2007; Becker 2008; Bell et al. 2008). Stock recomposition is based on obtaining larvae from ovigerous females from the wild and growing cultures of zoea larvae to megalopa stage. The strong positive geotaxis of megalopa increases the success of larval recruitment, it being the suitable stage for the release of larvae (Silva 2007; Becker 2008). Additionally, release of juveniles and later stages can enhance restocking (Bell et al. 2005).
The first step is the evaluation of an appropriate larval diet for rearing. The size of food in relation to larval feeding appendages (maxillipeds) is a key factor in proper feeding of larvae. Possible disproportionality can lead to a nutritional deficit, prolonged development time, and increased mortality rate (Harms and Seeger 1989; Anger 2001). Due to the absence of predators and steady conditions, larval survival reaches higher rates than those in nature. Food availability, which fluctuates seasonally in the environment, regulates larval hatching and growth. Decapod larvae hatch as lecithotrophs, although with a limited reserve. As such reserve ends, the now planktotrophic larvae must reach their optimum biomass. Although larvae can withstand periods of food deprivation, at a low feeding rate, larval development can be delayed, and growth and survival are reduced (Stanton and Sulkin 1991; Anger 2001). Low-cost larval production allied to a viable survival rate is the utmost intention (Bell et al. 2005; Becker 2008). This method will assert the supply of larvae to reach full potential, by attaining optimal stocking density of the species in later larval mass rearing.
Although larval culture already shows positive outcomes, there is no consolidation of its techniques, mainly in terms of successful feeding and breeding protocols of a range of species. The design of technically viable diets that provide the necessary nutritional requirements for each species is a key factor in establishing successful population recovery methods (Becker 2008). Through established larval breeding protocols, restocking of estuarine/marine species with larval development may be possible. We aimed to evaluate the effect of different diets on the survival rate and development time of the stone crab Menippe nodifrons Stimpson, 1859 and the swimming crab Callinectes danae Smith, 1869, from zoea I to megalopa stage, clarifying which diet is the most suitable. We used three microalgae species, Tetraselmis gracilis (Kylin) Butcher, 1959; Chaetoceros calcitrans (Paulsen) Takano, 1968; and Thalassiosira weissflogii Hustedt, 1926; the rotifer Brachionus plicatilis (Müller, 1786) (enriched with T. gracilis); and nauplii of Artemia sp. Leach, 1819 as a food source. We hypothesised that larval survival rate and time to develop in both species is directly linked to the most suitable diet. A microalgal and/or rotifer diet could be the most suitable for rearing both species, as similar results were found for closely related species (Sulkin 1975; Sorgeloos and Léger 1992; Abrunhosa et al. 2002; Brown 2002; Ruscoe et al. 2004; Waiho et al. 2018).
Menippe nodifrons inhabits rocky shores and banks of Phragmatopoma sp. and possesses great potential for exploitation due to its relatively large size and developed cheliped musculature (Melo 1996; Oshiro 1999; Fransozo et al. 2000; Rodrigues-Alves et al. 2013). The species is widely distributed and has potential economic importance. Menippe nodifrons exhibits five zoea phases (uncommonly six phases) and one megalopa stage (Scotto 1979; Anger 2001). Callinectes danae inhabits estuarine waters near mangroves and open sea regions near sandy beaches (Sforza et al. 2010). The species displays a contiguous geographic distribution from Florida to the Brazilian state of Rio Grande do Sul (Melo 1996; Marochi et al. 2013). Actively exploited by artisanal fishery, the species is commonly captured as a bycatch of shrimp fishing (Costa and Negreiros-Fransozo 1998; Severino-Rodrigues et al. 2001; Marochi et al. 2013). The general larval development lasts six to eight zoea instars with a megalopa stage, with an optimal survival rate at salinity near 30 PSU (Costlow and Bookhout 1968; Sankarankutty et al. 1999).
Materials and methods
Collection area and conditioning of females
Seven ovigerous M. nodifrons females, mean carapace width 51.66 ± 3.78 mm, were manually sampled on a rocky shore in Itanhaém, São Paulo, Brazil (24° 12′ 06.9″ S 46° 48′ 41.3″ W), and two ovigerous C. danae females, mean carapace width 89.5 ± 7.77 mm, were collected by trawling (23 mm fishnet mesh size) in São Vicente, São Paulo, Brazil (23° 58′ 28.6″ S 46° 22′ 57.1″ W). We sampled only females with eggs in an advanced stage of embryo development, indicated by their greyish colour (Costa and Negreiros-Fransozo 1998; Oshiro 1999). The advanced period is preferred to avoid the interference of laboratory conditions on embryo development. The females were conditioned in glass aquariums (290 × 120 × 185 mm) with 500 ml of artificial marine water (salinity 30 PSU and 25 ± 1 °C) and a photoperiod of 12 : 12 h (light : dark). The artificial marine water was obtained by dissolving artificial refined sea salt (Blue Treasure Reef Sea Salt, Qingdao Sea-Salt Aquarium Technology Co., Ltd) in deionized water.
Diet cultivation
The food source for Brachyuran larvae should meet specific criteria to be an appropriate diet, such as being of suitable size to be easily captured and digested, having a satisfactory concentration (to supply enough food and avoiding excess feeding) and containing enough nutrients (Sui et al. 2008). The microalgae species strains were inoculated in culture medium (filtered seawater with nutrients) at 24 ± 1 °C, photoperiod 24 : 0 h (light : dark), and constant aeration, based on Guillard’s F/2 protocols (for more detail, see Duerr et al. 1998). The chosen culture medium is the one most used in marine algae culture (Derner et al. 2006). The three microalgae species were chosen based on their previous application in the larval diet of closely related species and their widespread usage in aquaculture (Brown 2002; Becker 2008; Lober and Zeng 2009).
The rotifers were cultivated at the same temperature and photoperiod as the microalgae and were fed on T. gracilis. Microalgae, as prey, exhibit a positive influence on the reproduction and nutritional value of B. plicatilis and are considered a source of the rotifer enrichment (Duerr et al. 1998; Lubzens et al. 2001; Becker 2008). Although the rotifer B. plicatilis is not a natural prey for Brachyura larvae, it is commonly used in crustacean larvae culture (Shields and Lupatsch 2012). It exhibits slow swimming activity, an accelerated reproduction rate, and tolerance of a high culture density (Suantika et al. 2000). Artemia sp. has been used almost universally as a key zooplanktonic live prey since the 1960s in aquaculture hatchery, even that it is not a natural prey for Brachyura larvae (Abrunhosa et al. 2002; Shields and Lupatsch 2012). Dormant Artemia sp. eggs can be stored for long periods and then used, requiring only 24 to 48 h of incubation, which makes them the least labour-intensive live diet accessible for larval cultures (Shields and Lupatsch 2012).
Experimental design
After hatching, zoea I larvae from all females of each species were mixed in 500 ml of water in the same conditions as mentioned before (salinity 30 PSU and 25 ± 1 °C). This procedure was used to avoid the effect of maternal origin on the results (Simith et al. 2014). The zoea I larvae were transferred individually to 12-well cellular culture plates (Kasvi®) with 5 ml of water (salinity 30 PSU and 25 ± 1 °C). Hatchery in individual experimental units decreases cannibalism and competition between recently metamorphosed megalopae on other larvae still at zoea stage. At these controlled conditions, even larvae with developmental delay and slow growth can succeed in reaching further phases (Becker 2008). Five different diets were used as treatments: the microalgae Tetraselmis gracilis [T], Chaetoceros calcitrans [C], and Thalassiosira weissflogii [W]; the rotifer Brachionus plicatilis (enriched with T. gracilis) [R]; and freshly hatched Artemia sp. nauplii [A]. For each treatment, 96 zoea I (replica) larvae were used, totalling 480 M. nodifrons and C. danae larvae. After positive initial results in the M. nodifrons hatchery, a diet of B. plicatilis was tested on the first three zoea phases followed by the addition of Artemia sp. from then on [RA] using 96 zoea I larvae. The cellular culture plates with larvae were stocked in a germination chamber (model 347 CDG, FANEM Ltd.®) with temperature (25 ± 1 °C) and photoperiod (12 : 12 h light : dark) controlled. Every 24 h, the live larvae were relocated to a new well containing fresh saltwater and new food was provided. For feeding, we used the following concentrations: 400,000 ind/ml of microalgae, 40 ind/ml of rotifers, and 0.6 ind/ml of Artemia sp. nauplii (Becker 2008; Cottens et al. 2014). The contents of the old wells were then analysed in the search for ecdysis. Survival data and instar development were obtained daily, indicating the progress of larval development and the larval mortality rate. The experiment ended when all larvae died or reached the megalopa stage. To facilitate data analysis and discussion, the results are shown at two moments of development: from zoea I to zoea III and from zoea I to megalopa stage.
Statistical analysis
Normality (Shapiro–Wilk) and variance homogeneity (Bartlett) tests were performed. The effects of diet on the survival rate and development duration were evaluated through a permutational analysis of variance (PERMANOVA) with a pairwise post hoc PERMANOVA test. The rate of larval survival (number of larvae alive), time for metamorphosis, and maximum time for mortality were used as response variables, and the diet treatments as explanatory variables. Survival curves were obtained using the Kaplan–Meier test. For all analysis, data were analysed at two different moments of development: from zoea I to zoea III and from zoea I to megalopa stage. Analyses were performed in the R environment (R Development Core Team 2018) using the ‘survival’ (Therneau 2017) and ‘vegan’ (Oksanen 2018) packages. The PERMANOVA post hoc test was performed using PRIMER (Anderson et al. 2008).
Results
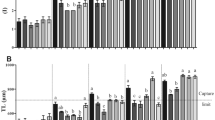
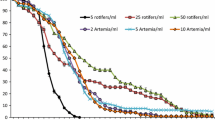
Diet affected M. nodifrons larvae survival and development time from zoea I to zoea III (p < 0.05), except for [T] × [C] (p = 0.43). Only larvae fed [R], [A], and [RA] diets developed to zoea III. Forty-one larvae on the [R] diet reached this phase, with a survival rate of 40% and development time of 10 ± 2.64 days. Five larvae on the [A] diet survived to ZIII, with a survival rate of 4.8% and development time of 14.75 ± 3.70 days. The [RA] treatment showed a 55.2% larval survival rate and development time of 12.65 ± 1.18 days (Fig. 1 and Table 1). The longevity of larvae fed a microalgae diet was short. Only [T] treatment larvae advanced to ZII. Survival rate dropped 45% on the third day and all larvae died on the eighth day. The [W] and [C] treatments ended on the seventh and sixth days, respectively. The survival rate of larvae on both diets dropped on the fifth (45.84%) and fourth (52.08%) days. Metamorphosis from ZI to ZII started on the fourth day for all diets.
Diet also affected M. nodifrons larval survival and development time from zoea I to megalopa stage (p < 0.05) (Fig. 2 and Table 1). Only larvae on the [A] and [RA] diets reached the megalopa stage. On the [A] diet, two larvae (2.08%) moulted to megalopa, in an average time of 29.5 ± 0.70 days (Fig. 2 and Table 1). On the [RA] diet, three larvae (5.88%) reached the megalopa stage, in an average time of 34.33 ± 8.39 days and with a mean survival rate of 15.33% (Fig. 2 and Table 1).
Diet also affected the survival of C. danae (p < 0.05). No larvae reached the zoea III stage. On the [T], [C], [W], and [A] diets, the mean larval survival time was 3.56 ± 1.22 days overall, without any larvae reaching ZII. Only two larvae fed the [R] diet reached the ZII phase, with a mean survival time of 6.59 ± 2.69 days (Fig. 3 and Table 2).
Discussion
The survival and duration of development from zoea I to megalopa stage of M. nodifrons and C. danae are affected by diet. Our results with M. nodifrons suggest that diets using different organisms at different life stages are ideal for larval development. We accept our initial hypothesis for M. nodifrons that the most suitable diet for larval development also shows a similar development time. On the other hand, none of the diets tested was effective for C. danae larvae. The most suitable feeding protocol for rearing M. nodifrons larvae is a combined diet of B. plicatilis up to the zoea III instar, with the introduction of hatched Artemia sp. nauplii onwards to the megalopa stage. The [RA] diet should be also tested for mass culture rearing of M. nodifrons. Although our results for rearing C. danae were not conclusive, the data provide additional information on improper diets for rearing the species for future research.
The most suitable diet for larval development until zoea III of M. nodifrons is the enriched B. plicatilis [R] diet. The nutritional needs of larvae alter at each stage as a function of their development (Souza and Costa 2006). An inadequate food source can generate disturbances in both larval development time and survival (Carvalho 2010). For the [R] diet, the minor mortality rate in the first days was related to fast development until the zoea III stage. The rotifer’s size (~ 250 to 300 μm) compared with the size of zoea I–zoea III M. nodifrons larvae (~ 0.55 to 0.80 mm) and easy laboratory culture makes it a fit diet for pre-zoea III larvae (Scotto 1979; Harvey and Epifanio 1997; Waiho et al. 2018). The rotifer diets were not suitable for feeding C. danae larvae. While other factors are also significant, the disparity in size between larvae and prey is also likely the key factor in this case. Sulkin (1975) suggested that Callinectes sapidus first-stage larvae are not able to ingest rotifers (> 120 μm) due to the difficulty in breaking down the prey into pieces. The same can be inferred for small C. danae larvae.
We chose an enriched rotifer diet to improve the overall larval survival rate. Rotifer enrichment maintains the general health and development of Brachyuran larvae (Ben-Amotz et al. 1987; Baylon 2009; Waiho et al. 2018). Brachionus plicatilis are non-selective filter feeders, ingesting any particle up to 12–15 μm, ideal for the bioencapsulation of any fatty acid-rich algae (Becker 2008). The alga used to enrich our [R] treatment (T. gracilis) showed a high level of essential fatty acids (EFAs). EFAs such as highly unsaturated fatty acids (HUFAs) or polyunsaturated fatty acids (PUFAs) are crucial to the growth and survival of marine fish and crustacean larvae (Lourenço et al. 1997; Selvakumar and Umadevi 2014). The use of EFA enrichment in a high-density rotifer diet at initial stages minimizes metamorphosis time, reduces larval development time, and standardizes the larval growth rate (Becker 2008).
The mixed rotifer plus Artemia sp. diet [RA] (400–500 μm) was the most suitable for rearing M. nodifrons from zoea III onwards to megalopa stage. Although Artemia nauplii are an extremely convenient source of food, they are naturally low in EFAs. Thus, larval survival is likely enhanced on an Artemia diet combined with other fatty acid-enriched food sources (Agh and Sorgeloos 2005; Beder et al. 2018). Ruscoe et al. (2004) suggested that larvae fed an Artemia mono-diet take longer to reach megalopa and endure higher levels of mortality. The diet is also not suitable for rearing C. danae. In previous studies on the rearing of Callinectes sapidus and Callinectes similis, early-stage larvae were able to consume only parts of the Artemia nauplii and individuals no larger than 110 μm (Sulkin and Epifanio 1975; Bookhout and Costlow 1977). Therefore, Artemia nauplii are only appropriate live food in terms of size and swimming speed for post-ZIII instars of M. nodifrons larval development.
Our data suggest a non-exclusive microalgae diet for both M. nodifrons and C. danae larvae. Both species suffered a high larval mortality rate allied to a lack of development for all microalgae treatments. Our main hypothesis is related to the disproportion of the size of microalgae and larval feeding appendages. Tetraselmis gracilis, C. calcitrans, and T. weissflogii are small (~ 9.59 μm, ~ 7 μm, and 4–32 μm, respectively; Olenina et al. 2006; Da Silva Gorgônio et al. 2013) in comparison to M. nodifrons (~ 0.55 mm) and C. danae (~ 0.40 mm) zoea I larvae. Likely, the larvae were not able to feed on the [W] and [C] diets, dying by the end of their reserves, at ZI. It is unlikely that the food quantity was insufficient because we used a high concentration commonly used in larvae aquaculture (Becker 2008; Cottens et al. 2014). The [T] treatment was the only one under which larvae developed into the ZII phase. The genus Tetraselmis exhibits increased mobility due to its flagellated morphology and was the largest microalga used (Bicudo and Menezes 2006). The predatory behaviour of larvae and their preference for mobile food (Anger 2001) may stimulate them to feed, leading to ZII development under this treatment. Prior research showed only improved outcomes in treatments with a combination of an algal and non-algal diet (Sulkin 1975). Brown mono-microalgal diets may also cause a deficiency of some essential nutrients, decreasing growth and general survival (Harms and Seeger 1989; Seixas 2016). Our survey brings new information about a combination of a non-algal diet.
The most suitable feeding protocol for rearing M. nodifrons larvae is a combined diet of B. plicatilis up to the zoea III instar with the introduction of hatched Artemia sp. nauplii onwards to the megalopa stage. The [RA] diet can be also tested for mass culture rearing of M. nodifrons. Although our results on C. danae rearing were not conclusive, the data provide additional information on unsuitable diets for rearing the species for future research. Based on widespread species production, different microalga genera with a high concentration of fatty acids can be tested, such as Pavlova, Skeletonema, Chlorella, Isochrysis, and Porphyridium (Muller-Feuga 2000; Brown 2002), or other holoplanktonic and meroplanktonic organisms.
References
Abrunhosa FA, Silva-Neto AA, Melo MA, Carvalho LO (2002) Importância da alimentação e do alimento no primeiro estágio larval de Ucides cordatus cordatus (Linnaeus, 1763) (Decapoda: Ocypodidae). Rev Cienc Agron 33(2):5–9
Agh N, Sorgeloos P (2005) Handbook of protocols and guidelines for culture and enrichment of live food for use in larviculture. Urmia, Iran
Anderson MJ, Gorley RN, Clarke KR (2008) Permanova+ for PRIMER: guide to software and statistical methods. PRIMER-E, Plymouth
Anger K (2001) Crustacean issues. The biology of decapod crustacean larvae. Helgoland, Germany
Baylon JC (2009) Appropriate food type, feeding schedule and Artemia density for the zoea larvae of the mud crab, Scylla tranquebarica (Crustacea: Decapoda: Portunidae). Aquacult 288(3–4):190–195
Becker AG (2008) Utilização de organismos-alimento na larvicultura do caranguejo-uçá, Ucides cordatus (Linnaeus, 1763) (Crustacea, Brachyura, Ocypodidae). Dissertation, Federal University of Paraná
Beder AM, Copeman LA, Eckert GL (2018) The effects of dietary essential fatty acids on the condition, stress response, and survival of the larvae of the red king crab Paralithodes camtschaticus Tilesius, 1815 (Decapoda: Anomura: Lithodidae). J Crustacean Biol 38(6):728–738
Bell JD, Munro JL, Nash WJ, Rothlisberg PC, Loneragan NR, Ward RD (2005) Advances in marine biology: restocking and stock enhancement of marine invertebrate fisheries. London, UK
Bell JD, Leber KM, Blankenship HL, Loneragan NR, Masuda R (2008) A new era for restocking, stock enhancement and sea ranching of coastal fisheries resources. Rev Fish Sci 16(1–3):1–9
Ben-Amotz A, Fishler R, Schneller A (1987) Chemical composition of dietary species of marine unicellular algae and rotifers with emphasis on fatty acids. Mar Biol 95(1):31–36
Bicudo, CEDM, Menezes, M (2006) Gêneros de algas de águas continentais do Brasil: chave para identificação e descrições. São Carlos, São Paulo
Bookhout CG, Costlow JD (1977) Larval development of Callinectes similis reared in the laboratory. B Mar Sci 27(4):704–728
Brown MR (2002) Nutritional value and use of microalgae in aquaculture. In: Cruz-Suárez, Ricque-Marie D, Tapia-Salazar M, Simoes G (ed) Avances en Nutrición Acuícola, Quintana Roo, México, pp 281–292
Carvalho AL (2010) Avaliação do efeito da salinidade e alimentação no desenvolvimento inicial em larvas de três espécies de caranguejos de importância econômica em laboratório Dissertation, Rural Federal University of Rio de Janeiro
Castilho-Westphal GG, Ostrensky A, Pie MR, Boeger WA (2008) Estado da arte das pesquisas com o caranguejo-uçá, Ucides cordatus. Arch Vet Sci 13(2):151–166
Costa TM, Negreiros-Fransozo ML (1998) The reproductive cycle of Callinectes danae Smith, 1869 (Decapoda, Portunidae) in the Ubatuba region, Brazil. Crustaceana 71(6):615–627
Costlow JD, Bookhout CG (1968) The complete larval development of the land-crab, Cardisoma guanhumi Latreille in the laboratory (Brachyura, Gecarcinidae). Crustaceana Supplement 2:259–270
Cottens KF, Silva UAT, Ventura R, Ramos FM, Ostrensky A (2014) Cultivo de larvas de Ucides cordatus (Linnaeus, 1763) sob diferentes intensidades luminosas. Arq Bras Med Vet Zootec 66(5):1464–1470
Da Nóbrega Alves RR, Nishida AK (2003) Aspectos socioeconômicos e percepção ambiental dos catadores de caranguejo-uçá Ucides cordatus (L. 1763) (Decapoda, Brachyura) do estuário do Rio Mamanguape, Nordeste do Brasil. Interciencia 28(1):36–43
Da Silva Gorgônio CM, Aranda DAG, Couri S (2013) Morphological and chemical aspects of Chlorella pyrenoidosa, Dunaliella tertiolecta, Isochrysis galbana and Tetraselmis gracilis microalgae. Nat Sci 5(7):783–791
Derner RB, Ohse S, Villela M, Carvalho SM, Fett R (2006) Microalgas, produtos e aplicações. Cienc Rural 36(6):1959–1967
Duerr E, Molnar A, Sato V (1998) Cultured microalgae as aquaculture feeds. J Mar Biotechnol 6(2):65–70
FAO Fisheries and Aquaculture. Fishery Statistical Collections: Global Production Statistics 1950-2017 (2020) Food and Agriculture Organization of the United Nations. http://www.fao.org/fishery/statistics/global-production/query/en. Accessed 06 January 2020
Fransozo A, Bertini G, Corrêa M (2000) Population biology and habitat utilization of the stone crab Menippe nodifrons Stimpson, 1859 (Decapoda, Xanthidae) in Ubatuba region, Brazil. In: Klein JCV, Schram FR (ed) The biodiversity crisis and Crustacea - Proceedings of the Fourth International Crustacean Congress. Rotterdam, pp 275–281
Freitas A, Furtado-Júnior I, Tavares MCS, Borcem ER (2015) Análise socioeconômica e esforço de pesca na captura do caranguejo-uçá–Ucides cordatus (Crustacea: Ucididae) –na Reserva Extrativista Maracanã–costa amazônica do Brasil. Bol Mus Para Emílio Goeldi Cienc Hum 10(3):711–722
Harms J, Seeger B (1989) Larval development and survival in seven decapod species (Crustacea) in relation to laboratory diet. J Exp Mar Bio Ecol 133(1–2):129–139
Harvey EA, Epifanio CE (1997) Prey selection by larvae of the common mud crab Panopeus rherbstii Milne-Edwards. J Exp Mar Biol Ecol 217:79–91
Lober M, Zeng C (2009) Effect of microalgae concentration on larval survival, development and growth of an Australian strain of giant freshwater prawn Macrobrachium rosenbergii. Aquacult 289:95–100
Lourenço SO, Marquez UML, Mancini-Filho J, Barbarino E, Aidar E (1997) Changes in biochemical profile of Tetraselmis gracilis I. Comparison of two culture media. Aquaculture 148(2–3):153–168
Lubzens E, Zmora O, Barr Y (2001) Biotechnology and aquaculture of rotifers. Hydrobiologia 446–447:337–353
Marochi MZ, Moreto TF, Lacerda MB, Trevisan A, Masunari S (2013) Sexual maturity and reproductive period of the swimming blue crab Callinectes danae Smith, 1869 (Brachyura: Portunidae) from Guaratuba Bay, Paraná State, southern Brazil. Nauplius 21(1):43–52
Melo, GAS (1996). Manual de Identificação dos Brachyura (Caranguejos e Siris) do Litoral Brasileiro. Editora Plêiade, São Paulo
Muller-Feuga A (2000) The role of microalgae in aquaculture: situation and trends. J Appl Phycol 12(3–5):527–534
Oksanen J (2018) R package: community ecology package. Version 2.5–2. https://cran.r-project.org/package=vegan. Accessed 14 June 2018
Olenina I, Hajdu S, Edler L, Andersson A, Wasmund N, Busch S, Göbel J, Gromisz S, Huseby S, Huttunen M, Jaanus A, Kokkonen P, Ledaine I, Niemkiewicz E (2006) Biovolumes and size-classes of phytoplankton in the Baltic Sea. HELCOM Balt Sea Environ Proc 106:1–44
Oshiro LMY (1999) Aspectos reprodutivos do caranguejo guaia, Menippe nodifrons Stimpson (Crustacea, Decapoda, Xanthidae) da Baía de Sepetiba, Rio de Janeiro, Brasil. Rev Bras Zool 16(3):827–834
Pechenik JA (1999) On the advantages and disadvantages of larval stages in benthic marine invertebrate life cycles. Mar Ecol Prog Ser 177:269–297
R Development Core Team (2018) R: a language and environment for statistical computing. Vienna (Austria): R Foundation for Statistical Computing. http://www.R-project.org. Accessed 14 June 2018
Rodrigues AMT, Branco EJ, Saccardo SA, Blankensteyn AA (2000) Explotação do caranguejo Ucides cordatus (Decapoda: Ocypodidae) e o processo de gestão participativa para normatização da atividade na região sudeste-sul do Brasil. Bol Inst Pesca 26(1):63–78
Rodrigues-Alves DF, De Paiva B-AS, Fransozo V, Bertini G, Cobo VJ (2013) Importance of biogenic substrates for the stone crab Menippe nodifrons Stimpson, 1859 (Brachyura: Eriphioidea). Lat Am J Aquat Res 41:459–467
Ruscoe IM, Williams GR, Shelley CC (2004) Limiting the use of rotifers to the first zoeal stage in mud crab (Scylla serrata Forskål) larval rearing. Aquaculture 231:517–527
Sankarankutty C, Hong SY, Kim KB (1999) Description of laboratory reared first zoea of Callinectes danae Smith (Crustacea, Decapoda, Portunidae). Rev Bras Zool 16:45–49
Scotto LE (1979) Larval development of the Cuban stone crab, Menippe nodifrons (Brachyura, Xanthidae), under laboratory conditions with notes on the status of the family Menippidae. Fish B-NOAA 77(2):359–386
Seixas PPH (2016) Desenvolvimento larval do Caranguejo Uçá, Ucides cordatus (Linnaeus, 1763) alimentado com diferentes dietas. Dissertation, Rural Federal University of Rio de Janeiro
Selvakumar P, Umadevi K (2014) Enhanced lipid and fatty acid content under photoheterotrophic condition in the mass cultures of Tetraselmis gracilis and Platymonas convolutae. Algal Res 6:180–185
Severino-Rodrigues E, Pita JB, Graça-Lopes R (2001) Pesca artesanal de Siris (Crustacea, Decapoda, Portunidae) na região estuarina de Santos e São Vicente (SP), Brasil. Bol Inst Pesca 27(1):7–19
Sforza R, Nalesso RC, Joyeux JC (2010) Distribution and population structure of Callinectes danae (decapoda: portunidae) in a tropical Brazilian estuary. J Crustac Biol 30(4):597–606
Shields RJ, Lupatsch I (2012) Algae for aquaculture and animal feeds. J Anim Sci 21(1):23–37
Silva UAT (2002) Cultivos Experimentais de Caranguejo-Uçá, Ucides cordatus (Linnaeus, 1763). Dissertation, Federal University of Paraná
Silva UAT (2007) Recuperação populacional de caranguejo-uçá, Ucides cordatus (Linnaeus, 1763), através da liberação de formas imaturas em áreas antropicamente pressionadas. Dissertation, Federal University of Paraná
Simith DJB, Pires MAB, Abrunhosa FA, Maciel CR, Diele K (2014) Is larval dispersal a necessity for decapod crabs from the Amazon mangroves? Response of Uca rapax zoeae to different salinities and comparison with sympatric species. J Exp Mar Bio Ecol 457:22–30
Sorgeloos P, Léger P (1992) Improved larviculture outputs of marine fish, shrimp and prawn. J World Aquacult Soc 23(4):251–264
Souto JFB (2007) Uma abordagem etnoecológica da pesca do caranguejo, Ucides cordatus, Linnaeus, 1763 (Decapoda: Brachyura). Biotemas 20:69–80
Souza K, Costa R (2006) Efeito de diferentes dietas sobre a muda dos estágios iniciais de Ucides cordatus (Linnaeus, 1763) (Decapoda: Ocypodidae). Bol Mus Para Emílio Goeldi Ser Cienc Nat Bol Mus 1:97–102
Stanton JL, Sulkin SD (1991) Nutritional requirements and starvation resistance in larvae of the brachyuran crabs Sesarma cinereum (Bosc) and S. reticulatum (Say). J Exp Mar Bio Ecol 152(2):271–284
Suantika G, Dhert P, Nurhudah M, Sorgeloos P (2000) High-density production of the rotifers Brachionus plicatilis in a recirculation system; consideration of water quality, zootechnical and nutritional aspect. Aquac Eng 21:201–214
Sui LY, Wille M, Wu XG, Cheng YX, Sorgeloos P (2008) Effect of feeding scheme and prey density on survival and development of Chinese mitten crab Eriocheir sinensis zoea larvae. Aquac Res 39(6):568–576
Sulkin SD (1975) The significance of diet in the growth and development of larvae of the blue crab, Callinectes sapidus Rathbun, under laboratory conditions. J Exp Mar Bio Ecol 20(2):119–135
Sulkin SD, Epifanio CE (1975) Comparison of rotifers and other diets for rearing early larvae of the blue crab, Callinectes sapidus Rathbun. Estuar Coast Mar Sci 3(1):109–113
Therneau T (2017) Survival: survival analysis, including penalized likelihood. R package version 2.36–10. http://CRAN.R-project.org/package=survival. Accessed 14 June 2018
Vieira, RRR (2006) Identificação, abundância e distribuição das fases larvais das espécies de Brachyura (Crustacea, Decapoda) no estuário da lagoa dos patos e região costeira adjacente. Dissertation, Federal University of Rio Grande do Sul
Waiho K, Fazhan H, Quinitio ET, Baylon JC, Fujaya Y, Azmie G, Wu Q, Shi X, Ikhwanuddinb M, Ma H (2018) Larval rearing of mud crab (Scylla): what lies ahead. Aquaculture 493:37–50
Wunderlich AC, Pinheiro MA, Rodrigues AMT (2008) Biology of the mangrove uca crab, Ucides cordatus (Crustacea: Decapoda: Brachyura), in Babitonga Bay, Santa Catarina, Brazil. Rev Bras Zool 25(2):188–198
Acknowledgements
We are grateful to Dr. Ubiratã de Assis Teixeira da Silva and M.Sc. Camila Prestes dos Santos Tavares, for microalgae inocula.
Funding
This work was supported by the São Paulo Research Foundation (FAPESP: 2017/07406-3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed by the authors. All biological samples collected for the present study complied with the current laws of the Brazilian Federal Government and experiments were conducted with the permission of the Brazilian Institute of Environment and Renewable Natural Resources (IBAMA) (Authorization # 60095-1-DIFAP/IBAMA, 12 September 2017).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guarizo, M., Costa, T.M. & Marochi, M.Z. Effect of diet during larval development of Menippe nodifrons Stimpson, 1859 and Callinectes danae Smith, 1869. Aquacult Int 28, 1969–1980 (2020). https://doi.org/10.1007/s10499-020-00569-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-020-00569-2