Abstract
Amblyseius orientalis (Ehara) (Acari: Phytoseiidae) is an effective predatory mite for spider mite control on fruit trees in China. In recent decades, it has been produced massively at a commercial natural enemy producer, feeding on the storage mite Carpoglyphus lactis (L.). In the predator production process, the ratio of predatory mites to their prey was found to be critical for the population increase of A. orientalis in large-scale rearings. In this study, we investigated the predatory capacity of A. orientalis on various developmental stages of the prey C. lactis, and the effect of prey numbers on predator reproduction. The maximum predation rate of A. orientalis adults on C. lactis adults was 2.21 per day at the lowest density of five prey adults, and on C. lactis eggs it was 45.07 at the highest density of 60 prey eggs. The preference index Ci of A. orientalis on C. lactis eggs and adults was 0.4312 and − 0.9249, respectively, suggesting that A. orientalis preferred eggs to adults. Amblyseius orientalis could reproduce when it preyed on either eggs or deutonymphs of C. lactis. However, the fecundity of the predatory mites is not always proportional to the provided prey number. Higher density of prey deutonymphs resulted in lower fecundity, whereas more prey eggs resulted in higher fecundity of A. orientalis. Therefore, our study indicated that the choice of suitable density and developmental stage of prey can significantly improve A. orientalis production on a large scale.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Amblyseius orientalis (Ehara) (Acari: Phytoseiidae) is a widely distributed and effective predatory mite for spider mite control in fruit production in China (Sheng et al. 2014; Zheng et al. 2008). Previously, it was used exclusively as a spider mite control agent (Zhang et al. 1992; Zheng et al. 2008); however, in the past decade, it was found to be effective against thrips and whiteflies as well (Sheng et al. 2014; Yang et al. 2018). Thus, the mass rearing and large-scale production of this native predatory mite is gradually attracting attention. Early studies of A. orientalis rearing involved host plants and spider mites. However, it was not practical for large-scale production of A. orientalis in a natural enemy factory, as it was a long and costly process (Zhang et al. 2015). Some studies have attempted to rear predatory mites on pollen, but the cost was too high and results were not satisfactory (Liao and Zhu 1985).
Carpoglyphus lactis (L.) is a fast-reproducing storage pest mite which has been widely used as an alternative prey (Knapp et al. 2018; Ramachandran et al. 2021; Wang et al. 2008; Wu et al. 2009). It can be used for successful large-scale rearing of many commercial predatory mites, such as Amblyseius swirskii (Athias-Henriot) (Nguyen et al. 2013; Zhang and Zhang 2021). Our laboratory has reported that A. orientalis feeding on C. lactis can complete development, mating, and reproduction (Sheng et al. 2014). Compared with diets of Bemisia tabaci (Zhang et al. 2015), Panonychus citri (Zhang 1990) and castor pollen (Liao and Zhu 1985), A. orientalis feeding on C. lactis had a shorter preoviposition period and higher reproduction rate (Sheng et al. 2014). Due to diet compatibility, C. lactis appears a suitable alternative diet for large-scale production and field applications of A. orientalis.
Instead of spider mites, few studies have investigated whether there is a preference of A. orientalis to developmental stages of prey C. lactis as well as their predatory capacity on C. lactis. Moreover, we notice that the density of prey to A. orientalis is influential to predator population increase during both laboratory observation and factory production (personal observation). Although mass rearing method was considered with no negative impact on predation and development of the predatory mite (Saemi et al. 2017), many studies have already revealed the critical effect of prey on predation and female fecundity of the predators (Werling et al. 2013; Osman et al. 2016; Rasmy and Abou-Elella 2002). If prey numbers were much higher than predator numbers, they could inhibit the expansion of A. orientalis populations instead (personal observation).
In this study, we investigated the predatory capacity of A. orientalis on different developmental stages of the prey C. lactis, the stage preference and the effect of prey numbers on predator reproduction. The aim of this study was to uncover the potential reason of low population growth of A. orientalis when using C. lactis as an alternative prey in large-scale production.
Materials and methods
Mite rearing
Amblyseius orientalis was collected from soybean fields in Changli Research Institute of Pomology, Hebei Academy of Agricultural and Forestry Sciences (119°09′E, 39°43′N). The colony had been maintained on C. lactis for 10 years in the Laboratory of Predatory Mites, Institute of Plant Protection, Chinese Academy of Agricultural Sciences (Yan et al. 2022). Carpoglyphus lactis feeding on yeast were provided by Shoubonong Biotechnology Company (Beijing, China). All mites were reared in an incubator (RXZ, Ningbo, China) at 25 ± 1 °C, 70 ± 5% RH and L14:D10 photoperiod.
The predatory capacity of Amblyseius orientalis
To investigate the predatory capacity of A. orientalis on C. lactis at different densities, we transferred C. lactis adults and deutonymphs into small arenas (Yan et al. 2022). Briefly, the arena was a tiny device with two tightly clipped layers (modified Munger cage): a transparent acrylic board (30 × 20 × 3 mm) with a 1 cm diameter hole in the middle sealed by nylon mesh with glue, one piece of rectangular glass (30 × 20 × 1 mm) on top. The prey densities were set at 5, 10 and 20 adults or deutonymphs per arena. Amblyseius orientalis female adults were starved for 24 h in a plastic box before being transferred separately to each arena with C. lactis adults or deutonymphs. All treatments were repeated with 30 A. orientalis adults. The consumed number of both stages of C. lactis was recorded within 24 h.
Similarly, we investigated the predatory capacity of A. orientalis adults and larvae on eggs of C. lactis at different densities. We collected sufficient C. lactis eggs to set three densities of 30, 45 and 60 per arena. Amblyseius orientalis females were starved for 24 h and then separated into the arena with different densities of prey eggs. The same process was repeated, with one newly hatched A. orientalis larva per arena, and three prey densities of C. lactis were set at 10, 20 and 30 eggs per arena.
Stage preference of Amblyseius orientalis to prey
We studied the developmental prey stage preference when A. orientalis was exposed to egg, nymph, and adult in the same arena. We collected one starved A. orientalis female adult and transferred it to the arena with 10 eggs, 10 deutonymphs and 10 adults of C. lactis. The predation number of A. orientalis on each developmental stage of C. lactis was recorded after 6, 12 and 24 h. The experiment was replicated 30×.
Effect of developmental stage and density of Carpoglyphus lactis on predator fecundity
We moved one gravid A. orientalis adult into the arena with densities of 30, 45 and 60 eggs or 5, 10 and 20 deutonymphs of C. lactis. Each density was repeated 30×. These arenas were changed daily with the same number of eggs or deutonymphs of C. lactis, respectively. We then recorded the number of consumed eggs or deutonymphs of C. lactis daily and the oviposition of each A. orientalis for 7 consecutive days.
Statistical analysis
Kruskal-Wallis tests with pairwise comparisons were used to examine the effects of different C. lactis densities on predatory capacity, prey preference and fecundity of A. orientalis (α = 0.05). All analyses were run in SPSS v.25.0 and results were visualized in GraphPad v.8.02.
In the preference experiment, the prey preference index (Ci) of predators to various stages was expressed as Ci = (Qi-Fi) / (Qi+Fi) (Ivlev et al. 1962; Zhou and Chen 1987), where Fi is the proportion of the ith prey type in the environment, and Qi is the proportion of predation of the ith prey type by the predator. If Ci = 0, the predator had no preference for the ith prey. If 0 < Ci < 1, the predator had a positive preference for the ith prey, and if -1 < Ci < 0, the predator had a negative preference for the ith prey.
Results
The predatory capacity of Amblyseius orientalis on Carpoglyphus lactis
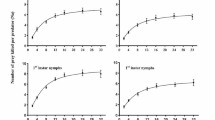
Prey densities significantly affected predation but in different trends (Fig. 1). The maximum number of C. lactis adults preyed upon by A. orientalis females was 2.21 at the lowest density of five mites per arena (χ2 = 12.436, df = 2, p = 0.002; Fig. 1A). In contrast, only 0.80 killed prey was observed at the density of 20 C. lactis adults per arena. The opposite tendency was observed when A. orientalis fed on deutonymphs: at the highest density of 20 mites per arena the maximum predation number was 6.44, which was significantly more than 4.07 in the group of five prey mites per arena (χ2 = 22.414, df = 2, p < 0.001; Fig. 1B). When testing A. orientalis adults and larvae on C. lactis eggs, the consumption number increased with prey densities. The maximum predation number of A. orientalis adults was 45.07 at the highest density of 60 eggs per arena, in contrast to the predation of 25.30 on 30 eggs per arena (χ2 = 44.869, df = 2, p < 0.001; Fig. 1C). Similarly, the maximum predation number of A. orientalis larvae was 13.08 at the highest density of 30 eggs per arena, which was more than the 7.62 in an arena with 10 prey eggs (χ2 = 24.536, df = 2, p < 0.001; Fig. 1D). In total, taking C. lactis eggs and deutonymphs as preys, the predation of A. orientalis adults tended to increase with prey density, whereas the predation number tended to decrease when feeding on high density of C. lactis adults (χ2 = 26.336, df = 1, p < 0.001; Fig. 2).
Predatory capacity (number of prey items consumed) of Amblyseius orientalis on Carpoglyphus lactis at different densities. Predation of A A. orientalis adults on C. lactis adults, B A. orientalis adults on C. lactis deutonymphs, C A. orientalis adults on C. lactis eggs, and D A. orientalis larvae on C. lactis eggs. Each dot represents an individual. Horizontal lines indicate the mean (± SD) of biological replicates. Means capped with different letters are significantly different (Kruskal–Wallis test: p < 0.05)
Stage preference
Amblyseius orientalis significantly preferred eggs to deutonymphs and adults of C. lactis (Table 1). Of the C. lactis eggs 81.8% was consumed, the largest percentage. The positive index Ci of 0.431 indicated a preference of A. orientalis for C. lactis eggs. On the contrary, C. lactis adults were the least chosen with only 1.3%. The negative index Ci of −0.925 suggested that A. orientalis have a strong aversion towards adult prey.
In the 24 h preference experiment, the numbers of consumed C. lactis eggs in the three time periods were 3.87, 5.87, and 8.43, which were significantly higher than the respective adult numbers of 0, 0.04, and 0.09 (Fig. 3). Apparently, A. orientalis preying on eggs of C. lactis continued at each time period.
Effect of prey developmental stage and density on Amblyseius orientalis fecundity
The above experiment revealed different preferences of A. orientalis to eggs and deutonymphs of C. lactis, so we further tested the effects of eggs and deutonymphs at different densities on A. orientalis fecundity. The maximum predation was 4.15 killed prey at the highest density of 20 deutonymphs per day (χ2 = 8.107, df = 2, p = 0.017; Fig. 4A). However, the highest reproduction with 2.13 eggs per day occurred at the density of five prey deutonymphs (χ2 = 21.487, df = 2, p < 0.001; Fig. 4C). When preyed on eggs, 38.62 eggs were consumed at the highest density of 60 eggs per arena, significantly more than 31.50 and 26.94 eggs at the other two density groups (χ2 = 32.833, df = 2, p < 0.001; Fig. 4B). The corresponding maximum reproduction was 1.90 eggs at the density of 45 prey items (χ2 = 14.869, df = 2, p = 0.001; Fig. 4D).
Predatory capacity (no. prey items consumed) and fecundity (no. eggs laid) of Amblyseius orientalis adult females on eggs and deutonymphs of Carpoglyphus lactis offered at three densities. Predation on C. lactis A deutonymphs and B eggs. Fecundity on C. lactis C deutonymphs and D eggs. Each dot represents an individual. Horizontal lines indicate the mean (± SD) of biological replicates. Means capped with different letters are significantly different (Kruskal−Wallis test: p < 0.05)
Predation rate and reproduction of A. orientalis were only correlated when prey density was high (Fig. 5). When prey number was lower, although not correlated, it did not decrease reproduction.
Discussion
The results of our study showed that A. orientalis female adults had strong predatory capacities on C. lactis eggs and deutonymphs, but not on adults. In the preference experiment, the Ci of 0.42 to C. lactis eggs showed a preference, whereas the Ci of −0.92 to adults indicated aversion. This is consistent with what we observed during practical rearing. Some studies also revealed predatory mites preferred eggs as prey (Furuichi et al. 2005; Ganjisaffar and Perring 2015; Moghadasi et al. 2013; Naeem et al. 2017; Song et al. 1995). Aversion of adult prey by the predators might be based on semiochemical compounds – an interesting study showed that Suidasia medanensis after attack could release defense volatiles that deter subsequent attack from the predatory mite A. swirskii (Midthassel et al. 2016). Whether such chemical compounds are also involved in the interaction of C. lactis and A. orientalis, or whether perhaps a physical barrier is in play in the developmental stage preference, remains to be elucidated.
Body size and mobility affects A. orientalis predation. In the experiment, the starved A. orientalis can prey on C. lactis adults and deutonymphs, both of similar size as the predator; however, the predation time was much longer than when preying on eggs. Besides body size, other factors might also affect predation, such as behavioral contact with the prey. Eggs obviously do not move, but predation of A. orientalis on mobile stages of C. lactis is relatively time-consuming, as the prey try to escape and avoid predation. Catching same size C. lactis adults was difficult for A. orientalis, as it was observed trying to grasp and prey on C. lactis. Apparently, the comparatively large body size, strong mobility and relatively thick tegument of C. lactis adults result in predation difficulty. Compared with C. lactis adults, the thin egg shell is easy to be pierced by the mouthparts of A. orientalis.
The development and reproduction of predators require crucial nutrients (Harvey et al. 2012; Williams and Roane 2007; Woods et al. 2020). Compared with high-protein prey, high-lipid prey had negative effects on the survival and reproduction of the web-building spider Hylyphantes graminicola (Wen et al. 2020). The predatory mite Parasitus consanguineus (Parasitidae), separately feeding on two prey species, the mushroom flies Megaselia halterata and Lycoriella ingenua, significantly differed in survival, female longevity and fecundity (Szlendak and Lewandowski 2009). When providing additive proteins or saccharides to prey mites in mass rearings, the phytoseiid predatory mite Neoseiulus barkeri presented higher fitness with shorter developmental durations and higher fecundity than those fed on basic diet (Huang et al. 2013). These results indicate that the food of the prey mites has a major impact on the nutritional value of the prey mites for predatory mites. In our study, C. lactis eggs could provide sufficient nutrition for A. orientalis reproduction, suggesting the developmental stage preference was not risky to predators in a long run. Our results are also consistent with a previous study that the eggs are sufficiently nutritious to support the predatory mite to complete reproduction and egg prey preference does not pose a survival risk to predators (Moghadasi et al. 2013). Neoseiulus californicus has been reported to prey eggs of five spider mite species which deposited on a variety of plants with similar performance (Gotoh et al. 2006). All this evidence indicates prey eggs are sufficient to support growth and development of the predatory mites.
In the current study, we found that fecundity of A. orientalis could be lower when providing higher density of C. lactis adults. It means that the ratio of prey and predator is critical during predatory mite rearing. As discussed above, we hypothesize that the energy supplied by the prey may be less than the energy consumed by A. orientalis during predation. Other explanations might be that the predators are deterred to prey C. lactis by an alarm pheromone or physical barrier. In mass-production, we should quantify prey density and strictly control the ratio of C. lactis to A. orientalis in inoculation to ensure the population growth of predatory mites. In short, our study elucidated the sensitive characteristics of A. orientalis to prey density, and provided a solution for stable population growth of predatory mites in mass rearing.
References
Furuichi H, Oku K, Yano S, Takafuji A, Osakabe M (2005) Why does the predatory mite Neoseiulus womersleyi Schicha (Acari: Phytoseiidae) prefer spider mite eggs to adults? Appl Entomol Zoolog 40:675–678. https://doi.org/10.1303/aez.2005.675
Ganjisaffar F, Perring TM (2015) Prey stage preference and functional response of the predatory mite Galendromus flumenis to Oligonychus pratensis. Biol Control 82:40–45. https://doi.org/10.1016/j.biocontrol.2014.12.004
Gotoh T, Tsuchiya A, Kitashima Y (2006) Influence of prey on developmental performance, reproduction and prey consumption of Neoseiulus californicus (Acari: Phytoseiidae). Exp Appl Acarol 40(3–4):189–204. https://doi.org/10.1007/s10493-006-9032-3
Harvey JA, Cloutier J, Visser B, Ellers J, Wäckers FL, Gols R (2012) The effect of different dietary sugars and honey on longevity and fecundity in two hyperparasitoid wasps. J Insect Physiol 58(6):816–823. https://doi.org/10.1016/j.jinsphys.2012.03.002
Huang H, Xu X, Lv J, Li G, Wang E, Gao Y (2013) Impact of proteins and saccharides on mass production of Tyrophagus putrescentiae (Acari: Acaridae) and its predator Neoseiulus barkeri (Acari: Phytoseiidae). Biocontrol Sci Technol 23(11):1231–1244. https://doi.org/10.1080/09583157.2013.822849
Ivlev VS, Scott D, Winberg GG, Fry FEJ, Ricker WR (1962) Experimental Ecology of the feeding of fishes. Chesapeake Sci 3(1):56–58. https://doi.org/10.2307/1350423
Knapp M, Houten YM, Baal EV, Groot TV (2018) Use of predatory mites in commercial biocontrol: current status and future prospects. Acarologia 58:72–82. https://doi.org/10.24349/ACAROLOGIA/20184275
Liao YM, Zhu ZM (1985) Study on the feeding habits of Amblyseius orientalis. Jiangxi Plant Prot 3:17–18
Midthassel A, Leather S, Wright DJ, Baxter IH, Farman DI, Cork A (2016) An astigmatid defense volatile against a phytoseiid mite. Entomol Exp Appl 158:97–107. https://doi.org/10.1111/eea.12382
Moghadasi M, Saboori A, Allahyari H, Golpayegani A (2013) Prey stages preference of different stages of Typhlodromus bagdasarjani (Acari: Phytoseiidae) to Tetranychus urticae (Acari: Tetranychidae) on rose. Persian J Acarol 2:531–538. https://doi.org/10.22073/PJA.V2I3.10047
Naeem T, Bajwa BE, Mazhar MS, Jamil N, Khan SB (2017) Prey stage preference and predatory potential of Typhlodromus divergentis (Acari: Phytoseiidae) against two spotted spider mites, Tetranychus urticae (Acari: Tetranychidae). J Agric Res 55:661–669
Nguyen DT, Vangansbeke D, Lü X et al (2013) Development and reproduction of the predatory mite Amblyseius swirskii on artficial diets. Biol Control 58:369–377. https://doi.org/10.1007/s10526-012-9502-y
Osman MA, Abou-Elella GM, Rasmy AH (2016) Life table parameters of the predatory mite, Cydnoseius negevi (Swirski & amitai) affected by prey density of Tetranychus urticae koch (acari: Phytoseiidae, tetranychidae). Egypt J Biol Pest Control 26(4):827–832
Ramachandran D, Lindo Z, Meehan ML (2021) Feeding rate and efficiency in an apex soil predator exposed to short-term temperature changes. Basic Appl Ecol 50(Suppl):87–96. https://doi.org/10.1016/j.baae.2020.11.006
Rasmy AH, Abou-Elella GM (2002) Effect of prey density on functional and numerical responses of the predatory mite typhlodromus negevi (acari: phytoseiidae). Springer Neth. https://doi.org/10.1007/978-94-017-0611-7_31
Saemi S, Rahmani H, Kavousi A, Chi H (2017) Group-rearing did not affect the life table and predation rate of Phytoseiulus persimilis (Acari: Phytoseiidae) fed on Tetranychus urticae. Syst Appl Acarol 22(10):1698. https://doi.org/10.11158/saa.22.10.11
Sheng FJ, Wang ED, Xu XN, Wang BM (2014) Life table of experimental population of Amblyseius orientalis feeding on Carpoglyphus lactis. Chin J Biol Control 30(02):194–198. https://doi.org/10.16409/j.cnki.2095-039x.2014.02.004
Song Y, Ha T, Chung D, Heong KL (1995) The predatory behavior of green mirid bug, Cyrtorhinus lividipennis Reuter, on brown planthopper eggs in different temperature conditions. Korean J Appl Entomol 34(3):234–242
Szlendak E, Lewandowski M (2009) Development and reproductive capacity of the predatory mite parasitus consanguineus (Acari: Parasitidae) reared on the larval stages of Megaselia halterata and Lycoriella ingenua. Exp Appl Acarol 47(4):285–292. https://doi.org/10.1007/s10493-008-9218-y
Wang BM, Wang ZQ, Wu ZY, Wang XW, Fan QH (2008) The occurrence and control of Carpoglyphus lactis (Linnaeus) (Acari:Carpoglyphidae). J Biosaf 17(02):156–160
Wen L, Jiao X, Liu F, Zhang S, Li D (2020) High-lipid prey reduce juvenile survivorship and delay egg laying in a small linyphiid spider Hylyphantes graminicola. J Exp Biol. https://doi.org/10.1242/jeb.237255
Werling BP, Lowenstein DM, Straub CS, Claudio G (2013) Multi–predator effects produced by functionally distinct species vary with prey density. J Insect Sci 1:30. https://doi.org/10.1673/031.012.3001
Williams L 3rd, Roane TM (2007) Nutritional ecology of a parasitic wasp: food source affects gustatory response, metabolic utilization, and survivorship. J Insect Physiol 53(12):1262–1275. https://doi.org/10.1016/j.jinsphys.2007.06.017
Woods MJ, Conlong DE, Ngomane N, Gillespie DY, Hoffman LC, Pieterse E (2020) The development of an improved artificial diet for the mass rearing of Eldana saccharina Walker (Lepidoptera: Pyralidae). J Sci Food Agric 100(13):4678–4687. https://doi.org/10.1002/jsfa.10466
Wu ZY, Zhang XH, Jia L, Fan QH (2009) Effects of five preys on growth and reproduction of lasioseius sp. J Fujian Agric Forestry Univ (Natural Sci Edition) 38:581–584
Yan H, Zhang B, Li ZP (2022) Phenotypic plasticity of predatory mite Amblyseius orientalis in response to diet switch. Syst Appl Acarol 27(6):1098–1108. https://doi.org/10.11158/saa.27.6.9
Yang JY, Sheng FJ, Song ZW, Lv JL, Xu XN, Li DS, Wang ED (2018) Functional responses of Amblyseius orientalis and A. tsugawai to eggs and 1st instar of Bemisia tabaci. Chin J Biol Control 34(02):214–219. https://doi.org/10.16409/j.cnki.2095-039x.2018.02.006
Zhang SY (1990) Research on the biology and foods of Amblyseius orientalis. Nat Enemies Insects 12(1):21–24
Zhang K, Zhang ZQ (2021) The dried fruit mite Carpoglyphus lactis (Acari: Carpoglyphidae) is a suitable alternative prey for Amblyseius herbicolus (Acari: Phytoseiidae). Syst Appl Acarol 26(11):2167–2217. https://doi.org/10.11158/saa.26.11.15
Zhang SY, Cao XW, Han ZQ, Wu WN (1992) Research on natural control of two spider mites by Amblyseius orientalis (Acari: Phytoseiidae) in apple orchards. Nat Enemies Insects 14(1):21–24
Zhang XX, Lv JL, Hu Y, Wang BM, Chen X, Xu XN, Wang ED (2015) Prey preference and life table of Amblyseius orientalis on Bemisia tabaci and Tetranychus cinnabarinus. PLoS ONE 10(10):e0138820. https://doi.org/10.1371/journal.pone.0138820
Zheng FQ, Zhang XH, Mo TL, Zheng JQ, Wu JB (2008) Ecological niches and guilds of main insect pests and their natural enemies on apple trees. Acta Ecol Sin 28:4830–4840
Zhou JZ, Chen CM (1987) Quantitative measurement of selectivity of predator for prey. Acta Ecol Sin 7(1):50–56
Acknowledgements
This work was supported by National Key R&D Program of China (2022YFD1401200) and Beijing Agriculture Innovation Consortium (BAIC01-2022).
Author information
Authors and Affiliations
Contributions
JW and YL: Investigation, formal analysis, Writing—original draft preparation, reviewing and editing. FS: Investigation and formal analysis. BZ: Conceptualization, Fformal analysis, supervision, writing—original draft preparation, reviewing and editing. EW and XX: Validation, supervision and editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, J., Liu, Y., Sheng, F. et al. Predatory mite Amblyseius orientalis prefers egg stage and low density of Carpoglyphus lactis prey. Exp Appl Acarol 90, 267–276 (2023). https://doi.org/10.1007/s10493-023-00805-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-023-00805-3