Abstract
Tetranychus urticae Koch (Acari: Tetranychidae) is a major pest species in numerous crops including hop (Humulus lupulus L.). Substantial T. urticae infestation was observed to occur in this recently introduced crop in Brazil. The adoption of less suitable cultivars to the pest species is highly desirable for integrated pest management. We used free-choice trials and two-sex life table analysis to determine the preference and population growth of T. urticae under laboratory conditions using three of hop cultivars currently expanding in Brazil (Mantiqueira, Victoria, and Yakima Gold). We also estimated the density of non-glandular trichomes and lupulin glands found on the abaxial leaf surface of these cultivars and correlated them with performance parameters of T. urticae. Mantiqueira appeared to be the least preferred by adult females for attractiveness and oviposition suggesting existence of antixenosis on this cultivar. Female immature stages developed slower on Yakima Gold and Mantiqueira, but no difference was observed between the latter and Victoria. Fecundity and longevity were significantly lower on Mantiqueira than on Victoria and Yakima Gold. No significant differences were observed among cultivars for intrinsic rate of increase (r), finite rate of increase (λ), and net rate of reproduction (R0), suggesting the absence of antibiosis. Although, lupilin gland densities were higher on Mantiqueira and Yakima Gold than on Victoria, no significant correlations were observed between these defensive traits and performance parameters of T. urticae. However, 30-day population simulations of T. urticae suggest that Yakima Gold is the least susceptible, Mantiqueira is moderately susceptible, and Victoria is highly susceptible.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hop (Humulus lupulus L., Cannabaceae) is an economically important crop for the brewing industry, where it is used to impact flavour and aroma of beer, and has also drawn attention in recent years due to its potential pharmaceutical applications. (Chadwick et al. 2006; Wang et al. 2008; Piraneo et al. 2015; Kryou et al. 2017; Bocquet et al. 2018). It is a dioeciously perennial specialty crop that is planted in female monoculture and its young shoots are also edible, whereas its lignified stems are used in paper and textile fibre manufacturing (Neve 1991; Rodrigues et al. 2015). Hops have been grown in temperate areas for centuries, mostly in the Northern Hemisphere. In 2017, the cropped area worldwide was over 91 thousand ha, and production was 148,603 tons; the largest producers are Germany, USA, Czech Republic, and China (FAOSTAT 2017).
Although Brazil is currently the third-largest beer producer in the world, its hop production is marginal, and almost 100% of lupulin (a main substance of hop) is imported from Germany and USA (Bebert 2017; Marcusso and Müller 2018). One of the reasons for low hop production in Brazil is the assumption that this crop is not adapted to tropical edaphoclimatic conditions, but recent attempts to grow it locally have been successful (Sarnighausen et al. 2017). The success of hop growing in Brazil will reduce beer production costs thereby making local breweries more competitive (Silva 2019). As a traditional agricultural country, Brazil has great potential for hop cultivation in both family and large-scale farming (Silva 2019). However, recent field observations have demonstrated the potential of two-spotted spider mite (TSSM), Tetranychus urticae Koch (Acari: Tetranychidae) to become one of the main pests on hop in Brazil.
In the main hop-producing countries, TSSM is already considered one of the key pests that achieve the largest population densities mainly in drought periods and in protected environments (Weihrauch 2005; Mahaffee et al. 2009; Woods et al. 2014). Exclusively phytophagous, this mite infests preferentially the abaxial surface of leaves, as well as hop cones (Walsh et al. 2015). TSSM feeds on cell content, causing chlorotic spots that reduce the photosynthetic capacity, increasing tissue necrosis, premature leaf fall, and eventually cause the death of infested plants (Walsh et al. 2015).
Chemical control is the main method for controlling TSSM in hop crops (Ako et al. 2004; Weihrauch 2005; Wu et al. 2019). However, the efficacy of that method is not always acceptable. Intensification of acaricide sprays can promote pests to evolve pesticide resistance. Some of the main causes that can lead to such evolution in TSSM are innumerable detoxification genes, its rapid development, and high reproductive capacity due to arrhenotoky parthenogenesis (Helle and Pijnacker 1985; Van Leeuwen et al. 2010, 2015; Grbić et al. 2011). For this reason, alternative control measures have been sought worldwide including biological control using predatory mites and insects (James 2003; Calderwood et al. 2015), alone or combined with acaricides (Lilley et al. 1999; James and Price 2002; James 2003; Iskra et al. 2019), as well as crop intercropping (Barber et al. 2003; Calderwood et al. 2017; Campbell 2018).
Another interesting approach is the use of mite resistant cultivars (Gonzalez-Dominguez et al. 2015; Costa et al. 2017; Gong et al. 2018; Savi et al. 2019a,b). Resistant hop cultivars could keep TSSM population below economic damage level. This may reduce spraying with acaricides, thereby avoiding negative environmental impact and poisoning risks to farmers and consumers (Savi et al. 2019a). It has already shown to be promising for some crops such as soybeans (Sedaratian et al. 2009, 2011), eggplant (Khanamani et al. 2013), cucumber (Maleknia et al. 2015), common beans (Shoorooei et al. 2018), strawberry (Gong et al. 2018; Rostamini et al. 2018), tomatoes (Savi et al. 2019a,b), and peanuts (Melville et al. 2019).
Pest populations are affected by several factors, including constitutive morphological and chemical traits of their host plants (Khanamani et al. 2015; Savi et al. 2019b). Presence of trichomes is one of the factors that can play an important role in cultivar resistance (Savi et al. 2019b). Two trichomes types have been identified in hop plants grouped as non-glandular (arrows) and glandular (lupulin glands) (Wang et al. 2008; Patzak et al. 2015). Non-glandular trichomes interfere mostly physically with pests, whereas glandular trichomes play an important role in cultivar resistance to pests by releasing defense compounds such as bitter acids (humulone and lupulone), polyphenols, essential oils (mono- and sesquiterpenes) and flavonoids compounds (Wang et al. 2008; Biendl et al. 2014; Undas et al. 2018). These compounds may cause antixenosis (mite behavior changes reducing or ending host colonization) and/or antibiosis (pest fitness reduction by reducing survival, oviposition, and development). However, there are few studies on pest-resistant hop cultivars in the literature (Regev and Cone 1975; Peters and Berry 1980; Lawanprasert 1994). Knowing the performance of a pest on different cultivars can be useful for practical purposes in integrated pest management (IPM).
Life table parameters provide important information as to how a pest species is affected by plant cultivars. Various studies have evaluated the life table of TSSM (Sedaratian et al. 2009; Khanamani et al. 2015) based on a traditional life table analysis (female age-specific life table). This method considers only females, disregarding the male population and variation in developmental rates among individuals of a population. According to Chi and Liu (1985), this may result in errors in the estimation of life table parameters. Given these limitations, Chi and Liu (1985) and Chi (1988) developed a theoretical model of life table analysis. It considers ages or stages and development rates of both sexes. Such design allows deeper knowledge on arthropod biology and population growth parameters, which are fundamental for pest management efficiency. Additionally, the construction of life tables with this model can assist in pest population projections.
Here, we used free-choice trials and the two-sex life table to access the preference and population performance of the TSSM on three hop cultivars under cultivation in Brazil (Mantiqueira, Victoria, and Yakima Gold). We also quantified the densities of non-glandular and glandular trichomes found on those cultivars in order to check possible correlation with the performance of TSSM.
Materials and methods
TSSM colony
To establish a mite colony, TSSM individuals were collected from soybeans (Glycine max L.) at the College of Agricultural and Veterinary Science, Jaboticabal Campus, University of São Paulo State (FCAV/UNESP), Brazil. The mites were then kept on jack beans (Canavalia ensiformis L.) for several generations in an air-conditioned chamber (25 ± 1 °C, 70 ± 10% RH, and 12-h photoperiod).
Hop cultivars
The study included three hop cultivars (Mantiqueira, Victoria, and Yakima Gold), chosen for their adaptability to Brazilian edaphoclimatic conditions and their continuous expansion in Brazil. Vegetative part cuttings of these cultivars obtained from the business group Hops Brazil (Table 1) were planted in Styrofoam tray cells, filled with Bioplant® substrate (coconut fibre, pine bark, bovine manure, sawdust, vermiculite, rice husks, ashes, agricultural plaster, calcium carbonate, magnesium, magnesium thermo-phosphate, and fertilizers) and kept in a greenhouse. Irrigation was carried out via automatic sprinkling 4× a day (at 8, 11, 14, and 17 h). Fertilizer [250 mL per 10 g dilution tray of monoammonium phosphate fertilizer (MAP) in 1 L water] was applied 2 weeks after planting rhizomes. Seedlings were transplanted individually in 4-L pots, filled at 80% with a mixture of similar proportions of soil, sand, and tanned bovine manure, previously sterilized in autoclave at 120 °C for 3 h. Each plant was fertilized every 15 days with 5 g urea (44% N) and 5 g potassium chloride (58% KCl). The plants were kept in a greenhouse without control of environmental conditions and irrigated daily by manual watering. The plants were not treated with pesticides.
Free-choice test
Dual choice tests were performed to assess TSSM attractiveness and egg-laying preference in each cultivar. We used 30-mm leaf discs made of leaves detached from the median third of the canopy of 7-month-old plants. Discs were placed close together, with the abaxial surface facing upward, due to mite preference. Glass coverslips (18 × 18 mm) were glued between two discs to facilitate mite movement across them. Each set of two leaflets was placed on a filter paper disc superimposed on a nylon foam layer (1 cm high), moistened with distilled water (Fig. 1), and set in a Petri dish (10 cm diameter, 1.5 cm high). Ten 6-day-old TSSM females were taken from the stock rearing and released into the coverslip centre. These units were kept in an air-conditioned chamber, under the same conditions as the stock colonies. After 24 h, the females and eggs found in each disc were counted. The former was expressed in percentage, whereas the latter was expressed in per capita oviposition rate per day obtained dividing the number of eggs with the number of mites on the disc. The experimental design was completely randomized, and 20 replicates were performed for each pair of dual-choice assay.
The number of adult females found in each cultivar was analysed by an observed vs. expected χ2 test at 5% probability using Proc FREQ in SAS. Oviposition data were transformed to √(x + 0.5), to make data more normally distributed and homogenous, but we reported the averages and standard errors for non-transformed data. Differences between cultivars with respect to average oviposition rates were tested by means of t-tests for paired observations (α = 0.05) using the computer program R (R Core Team 2012). The experiment-wise risk of committing at least one type 1 error (i.e., rejecting the null-hypothesis of no difference though it may be true) after pairwise tests is \(1-{(1-\alpha )}^{k}\). With α = 0.05 and k = 3 (numbers of pairwise tests), this experiment-wise error becomes 14.3%. In order to evite this type 1 error, the Bonferroni adjustment was performed after pairwise tests by using the formula α′ = α/k = 0.05/3 = 0.0167. Therefore, only P values lower than 0.0167 should be considered as significant when three pairwise tests are conducted.
Population performance
Before assessing population performance, TSSM specimens maintained on jack beans (stock rearing) were transferred to plants of the respective cultivars (Mantiqueira, Victoria and Yakima Gold) to be tested, onto which they were reared for three generations, for adaptation in order to suppress potential previous-diet effects (Awmack and Leather 2002).
Experimental units were made in Petri dishes (5 × 2 cm) using a nylon foam layer (1 cm thick) covered by a cotton-wool thin layer, which was moistened with distilled water. A 3-cm-diameter leaf disc made with leaves excised from the median third of the canopy of 7- to 8-month-old plants was placed into the cotton, with their abaxial surface up. To obtain eggs of uniform age, a gravid adult female of unknown age was transferred from the stock colony of the corresponding cultivar to leaf discs. Six h later, the female and extra eggs were removed, leaving a single egg per disc. For each cultivar, a cohort of 60 eggs of TSSM was used. These units were kept in air-conditioned chamber, under the same conditions mentioned in the stock colonies. Upon reaching adulthood, mites were sexed and transferred to new experimental units to form couples. For this purpose, some males (of unknown age) were taken from the stock colony of the corresponding genotype.
The units were examined at every 24 h under a stereomicroscope to determine the duration of each developmental stage and survivorship. Mite size, moving ability and presence of exuviae were considered to determine ecdysis occurrence (Laing 1969). The following parameters were determined: adult pre-oviposition period (APOP: period between adult emergence and its first oviposition), total pre-oviposition period (TPOP: period between first oviposition of parent and offspring generation), oviposition days (number of days in which oviposition occurred), longevity of each sex, sex ratio and fecundity. Leaf discs were replenished with new ones at every 3 days to maintain leaf quality for the mites.
In units where males died before females, other males from the stock colony were used to replace them. Data on males that came from stock colonies and mites that died on the strip of cotton wool while trying to escape were not used in the statistical analyses. Eggs laid were removed after every observation period. The experiment was considered finished when all mites had died.
The software TWOSEX-MSChart by Chi (2019a, b; available at http://140.120.197.173/Ecology/prod02.htm) was used to estimate development and reproduction from raw data and to calculate population parameters, using the procedure ‘two-sex life table’ (Chi and Liu, 1985; Chi, 1988). The following parameters were estimated: age-stage–specific survival rate (sxj), age-specific survival rate (lx), age-specific fecundity (mx), age-specific maternity (lxmx), age-specific life expectancy (exj), age-stage reproductive value (vxj), net reproduction rate (R0), intrinsic rate of increase (r), stage-specific fecundity (fxj), finite rate of increase (λ), and average generation time (T). In these parameters, j stands for the stage of an individual (i.e., j = 1 corresponds to eggs, 2 to larvae, 3 to protonymphs, 4 to deutonymphs, and 5 to adults) and x stands for age (in days). The formulas used to calculate these parameters are included in Table 2. Standard errors of development, fecundity, reproduction period, and population parameters were also estimated using the bootstrap method, following the procedure proposed by Huang and Chi (2012). During this procedure, data of each of these biological parameters were re-sampled 100,000 times. Differences among cultivars were compared by the paired bootstrap test, based on the confidence interval of differences (Efron and Tibshirani 1993; Akköprü et al. 2015).
TSSM population was projected by combining life table data in accordance with the theory of ‘age stage, life table two-sex’ (Chi 1990). The TIMING-MSChart software (Chi 2019b) was used to simulate TSSM population growth on the three cultivars for 30 days, using an initial population of 10 newly laid eggs. The 30-day population variability was projected using life tables of 0.025 and 0.975 percentiles of the 100,000-bootstrap results of the finite ratio of increase (λ) as described by Huang et al. (2018).
Non-glandular and glandular trichomes
The traits protecting the plants against spider mites were assessed by estimating the density of non-glandular (arrows) and glandular (lupulin glands) trichomes for each cultivar. For this purpose, the trichomes were counted under a stereomicroscope in 20 areas of 1 × 1 cm on the abaxial surface (where the mites prefer to stay) of leaves excised from the median third of the canopy of 8-month-old plants of each cultivar.
Trichome density data did not follow a normal distribution, even after transformation. Thus, we used a generalized linear model (GLM) with quasi-Poisson distribution to compare trichome numbers among cultivars. The best model-fit for was assessed by half-normal plots with simulation envelopes, using the ‘hnp’ package in R (Demétrio et al. 2014). If significant, multiple comparisons by Tukey's test (α = 0.05) for trichome density data among hop cultivars, using ‘glht’ function in ‘multcomp’ package of R (R Development Core Team 2012), with P value adjustments (Hothorn et al. 2008).
Correlation between parameters
Pairwise correlations of the parameters (attractiveness, oviposition preference, fecundity, development time, population projection, and lupulin and non-glandular trichome densities) were estimated for the cultivars using the corr.test function of the R psych package (method = Pearson). The correlations were visualized by the corrplot R package.
Principal component analysis (PCA)
Significant data on attractiveness, oviposition rate per capita, fecundity, development time, and lupulin density of the three cultivars were standardized (normal distribution, mean = 0, variance = 1) for exploratory multivariate analysis, and PCA was carried out using STATISTICA software v.7 (Statsoft 2004). Correlation between parameter matrices was assessed by Bartlett's sphericity test, and number of main components was defined by means of the broken stick criterion. Through PCA, we could determine the explanatory variables for each evaluated cultivar (Hair et al. 2006).
Results
Attractiveness of cultivars
The dual-choice tests revealed (Fig. 2) that the percentage of female mites settling on leaflets of Victoria was higher than on Mantiqueira leaflets (χ2 = 6.56, df = 1, P = 0.01), whereas the tests involving Yakima Gold versus Mantiqueira (χ2 = 1.58, df = 1, P = 0.21) and Victoria versus Yakima Gold (χ2 = 0.42, df = 1, P = 0.52) indicated no preference. The difference between Victoria and Mantiqueira was also significant after a Bonferroni adjustment.
Average (± SE) percentages of attracted adult female two-spotted spider mites on leaflet discs of each hop cultivar 24 h after release in dual-choice leaf disc assay (initial number of females = 28; number of replicates = 10), contrasting Mantiqueira versus Victoria, Mantiqueira versus Yakima Gold, and Victoria versus Yakima. The asterisk (*) indicates significant preference for one of the options (χ2 tests: df = 1, P < 0.05; n.s., P > 0.05). The difference between Victoria and Mantiqueira was also significant after a Bonferroni adjustment (α/k = 0.05/3 = 0.017)
A similar pattern was observed with respect to the dual-choice tests for the per capita oviposition rate per day (Fig. 3). Without a Bonferroni adjustment, it was significantly higher on Victoria than on Mantiqueira (t = 2.05, df = 19, P = 0.04), but the P value was not smaller than α/k = 0.05/3 = 0.017 (Bonferroni adjustment). The differences between Yakima Gold vs. Mantiqueira (t = 1.72, df = 19, P = 0.10), and Victoria vs. Yakima Gold (t = 0.93, df = 19, P = 0.36) suggested no preference.
Average (± SE) per capita daily oviposition rate of two-spotted spider mites on each hop cultivar within 24 h of mite release in dual-choice leaf disc assay, contrasting Victoria versus Mantiqueira, Yakima Gold versus Mantiqueira, and Yakima Gold versus Victoria. The P values are based on two-sample t tests; no significant differences between cultivars were observed after Bonferroni adjustment (α/k = 0.05/3 = 0.017)
Population performance
When males and females were combined, no significant differences among cultivars for the duration of the egg, larval, and protonymph stages were noted. Male immature stages (egg to deutonymph) also had no differences among cultivars. Development of female immature stages was slower on Yakima Gold (9.8 days) than on Victoria (9.0 days), and no difference was observed between the latter and Mantiqueira (9.1 days). Survival during the immature stages was above 90% in all cultivars (Table 3).
Pre-oviposition periods (APOP and TPOP) were longer on Yakima Gold (1.5 and 11.3 days, respectively) than on Victoria (1.2 and 10.1 days) and they were in between on Mantiqueira (1.4 and 10.5 days) (Table 4). Female longevity and oviposition days were significantly longer on Yakima Gold (29.6 and 16.4 days, respectively) and Victoria (28.5 and 15.8 days) than on Mantiqueira (23.4 and 11.0 days) (Table 4). Also fecundity was higher on Yakima Gold (78.6 eggs/female) and Victoria (84.5) than on Mantiqueira (55.9). Male longevity (26–28 days) and sex ratio (0.5–0.6) did not differ among the cultivars.
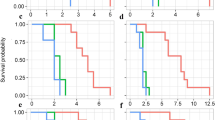
The survival rate curves (Fig. 4a) show the proportion of TSSM surviving at age x of stage j (sxj) in relation to the initial number of eggs (60) on the three hop cultivars. The observed overlaps between proportions of the distinct stages are due to individual differences in developmental speed. The proportions reached their maximum values due to recruitment from the previous stage and dropped due to concurrent mortality and moulting to the next stage. Newly laid egg had 47, 59 and 60% probability to survive until reaching adulthood being a female and 45, 37 and 36% probability to survive until reaching adulthood being a male, respectively, on Victoria, Mantiqueira and Yakima Gold.
Considering all development stages together, the probability of a mite surviving until age x is represented by age specific survival (lx) (Fig. 4b). On Mantiqueira, 45.1% of mites were alive until the 25th day of age, whereas on Victoria and Yakima Gold, survival rates were 62.7 and 66.0%, respectively.
Age-stage specific fecundity (fxj, daily number of eggs produced per female of age x) and age-specific fecundity (mx, average daily fecundity per individuals, i.e., this number is divided by all individuals of age x) curves fluctuated throughout the oviposition period (Fig. 4b). The highest peak of fxj was observed on Victoria (7.52) on the 12th day, followed by Yakima Gold (6.03) on the 14th day, whereas the lowest peak occurred on Mantiqueira (5.63) on the same day.
The highest average daily fecundity per individual (mx = 3.77 eggs) was observed on Yakima Gold on the 14th day (Fig. 4b), as well as the highest age-specific maternity (lxmx = 3.6 offspring) (Fig. 4b).
Life expectancy (exj) indicates the time that individuals at age x and stage j are expected to live in each hop cultivar (Fig. 5a). Life expectancy of newly laid eggs (e01) is equal to mean longevity. The values of e01 (newly laid eggs) were 27.7, 26.7, and 24.3 days on Yakima Gold, Victoria, and Mantiqueira, respectively, whereas a 20-day-old adult female lived for an extra 9 days on Mantiqueira and Victoria, and 11 days on Yakima Gold (Fig. 5a).
Age-stage reproductive value (vxj) describes the contribution of a mite at age x and stage j to the next generation (Fig. 5b). Females close to peak reproduction contributed more to the following population when compared to mites at other ages and stages. The reproductive value of females peaked on the 11th day on Mantiqueira (v11 = 20.49) and Victoria (v11 = 27.89), but on the 14th day on Yakima Gold (v14 = 23.13) (Fig. 5b).
The average generation time (T) was highest on Yakima Gold and lowest on Mantiqueira; the intermediate value on Victoria did not differ significantly from either of the two (Table 5). No significant differences were observed for R0 (32.90–47.18), r (0.220–0.227), and λ (1.247–1.255) (Table 4). The 30-day population projection from 10 newly laid eggs was higher on Mantiqueira (3502.1) and Victoria (3526.9) and lower on Yakima Gold (2782.0 mites) (Fig. 6). The 2.5 and 97.5 percentiles of the intrinsic rate of increase (r) used to simulate population growth variability revealed that population size can vary from 1876 to 5516 mites on Mantiqueira, from 1667 to 6317 on Victoria, and from 1476 to 4864 on Yakima Gold (Fig. 6).
Non-glandular (arrows) and glandular trichomes (lupulin glands)
No significant differences were observed among cultivars with respect to the density of non-glandular trichomes (F2, 57 = 0.555, P = 0.58) (Table 6). The densities of lupulin glands (glandular trichomes) were higher on Mantiqueira (190.1 trichomes/cm2) and Yakima Gold (193.1) than on Victoria (140.1) (F2, 57 = 3.959, P = 0.025) (Table 6).
Correlation between parameters
Results of the pairwise correlation analyses for the measured parameters indicated a positive association between the number of mites settling on a given cultivar and the per capita oviposition rate per day in free-choice bioassays (r = 0.81, P < 0.0001) (Fig. 7). A negative correlation was found between the developmental period of female immatures and the predicted population size after 30 days on the studied cultivars (r = − 0.82, P = 0.043). No other significant correlation was found (Fig. 7).
Pairwise correlations between preference test parameters, mite population performance and trichome densities. Non-significant (P > 0.05) correlation coefficients (r) are crossed out. The size and color differences of the symbols in the graph varied according to the value of correlation coefficients. Parameters: Pref.atr preference for attractiveness, Pref.ovip preference for egg-laying, Dev.period development time length, Ovip.days oviposition days, Pop.proj population projection, Lup.glands lupulin glands, Non-gland. non-glandular trichomes
Principal component analysis (PCA)
Principal component analysis indicated that the studied cultivars were different from each other (Fig. 8). On the first canonical axis (PC1), we verified an accumulated variation of 65.9% of the original parameters represented by positive and significant correlations, such as adult females settled (0.98), per capita oviposition rate per day (0.99), and fecundity (0.92). These parameters were the most relevant on Victoria. On Mantiqueira, the most relevant parameters were lower fecundity, shorter oviposition days, and lower longevity in positive scores for the second axis (PC2). Although PC2 was not significant (38.1%) for most parameters, the only relevant information associated with PC2 was the female immature development period (− 0.97). This parameter was most relevant for Yakima Gold.
Principal components analysis ordination diagram of preference and population performance parameters of Tetranychus urticae, and density and type of trichomes on the three hop cultivars. Parameters: Pref.fem preferred by adult females, Pref.ovip preferred for egg-laying, Dev.period development time length, Longev. total longevity, Ovi.days oviposition days, Fecund. fecundity rate, Lupulina lupulin glands, Non-gland. non-glandular trichomes, APOP adult pre-oviposition period
Discussion
This study determined the preference of TSSM for colonization and population performance on three potentially relevant cultivars for hop production in Brazil. Although Yakima Gold did not differ statistically from Mantiqueira, the latter appeared to be less suitable for initial colonization of adult females. This result suggests that Mantiqueira possesses a certain level of antixenosis resistance against TSSM. Seki (2016) and Shoorooei et al. (2018) reported that a possible cause for antixenosis is the insufficient level of attractive stimuli provided by the plants. Jones et al. (1996, 2010) reported that the colupulone substance found in hop leaves, the main component of the specific bitter acids group such as beta-acid, has a repellent effect on TSSM and reduces their oviposition.
Part of the population growth parameters of TSSM obtained in this study is comparable to those found in previous studies with the same mite. For instance, the duration of TSSM immature development reported by Peters and Berry (1980) on five hop genotypes (Cascade, Comet, Puggle, Talisman, and L-16) at 25 °C lasted 11.81–13.01 days and was close to that of our study, which in turn was higher than that reported by Lawanprasert (1994) for another four hop cultivars (EG-86-23, J-83-5, Huller Bitterer and Pride of Ringwood) ranged from 5.4 to 5.6 days. These authors also reported longevity ranging from 22.9 to 25 days at 25 °C, which is nearly identical to the longevities found in our study. Fecundity (63–151.8 eggs/female) and sex ratio (71% females), reported by Mayberry (1968) and Lawanprasert (1994), were higher than those found in this study. In addition, the intrinsic rate of increase (r) and the net reproduction rate (R0) reported by the same authors, under similar conditions, were higher (0.225–0.269 and 39.469–60.333, respectively) than found in our study. These differences might be due to (subtle) differences between, e.g., cultivars, experimental conditions and methods used for estimating life table parameters.
Our findings highlight that TSSM development time, pre-oviposition, longevity, and fecundity differed among the cultivars. Plants can resist attacks by herbivorous arthropods by prolonging their developmental time and by reducing their fecundity and longevity, and/or by attracting the pest’s natural enemies (Fürstenberg-Hägg et al. 2013; Xu et al. 2019). In our study, although the immature developmental times of TSSM on the various cultivars were very close, mites on Yakima Gold nevertheless developed slower than on the two other cultivars. Interestingly, Silva (2019) reported that Yakima Gold had a lower content of oleic composition (1.5 mL/100 g) compared to Mantiqueira (2.5 mL/100 g) and Victoria (2.8 ml/100 g). It suggests that differences in the oleic content composition of hops cultivars may affect TSSM immature development time. Melville et al. (2019) also found a correlation between peanut breeding lines that carry high oleic traits and susceptibility to Tetranychus ogmophallos Ferreira and Flechtmann. Our results corroborate also those of Čerenak et al. (2009), who observed a positive relationship between the amount of essential oil compounds in hop cultivars with resistance or susceptibility to powdery mildew.
The shortest period for adult pre-oviposition (APOP) on Victoria could be advantageous to TSSM as faster reproduction increases population growth rates. Yet, the lowest TSSM fecundity levels on Mantiqueira can be partially attributed to its leaf amino acid composition, which has proved to directly affect herbivorous arthropod fecundity (Weihrauch and Moreth 2005). Islam et al. (2017) found that adult females need suitable sources of nitrogen for the full development of their ovaries and suitable sources of carbohydrates for energy production. In terms of these compounds, low quantity and quality may decrease herbivore fecundity (Awmack and Leather 2002; Maleknia et al. 2016; Melville et al. 2019).
The slower TSSM immature development on Yakima Gold and the shorter longevity and higher fecundity on Mantiqueira are likely to lead to differences in population growth rate and finite rate of increase. However, this was not the case, based on the absence of statistical differences between cultivars with respect to parameters determining their potential population growth. Thus, in spite of differences among cultivars, these differences combined were insufficient to convincingly demonstrate that Yakima Gold showed antibiosis against TSSM. This matches results of Gong et al. (2018) on strawberries indicating that also non-attractive cultivars can be damaged by spider mites.
Based on unpublished personal observations, we claim that the three studied cultivars are susceptible to TSSM attacks, and that some cultivars are more susceptible than others. Our 30-day population projection estimates for TSSM indicated that Yakima Gold was the cultivar with the lowest population levels compared with the two other cultivars despite this species had the highest net reproductive rate (R0). This emphasizes the importance of developmental time, rather than fecundity, for population growth. Our study showed a significant correlation between TSSM development time and the population projection.
Leaf morphology, such as non-glandular trichome (arrows) or glandular trichomes (lupulin glands) densities, had previously been reported as factors affecting TSSM preference and/or performance on hops (Peters and Berry 1980; Jin et al. 2016). In our study, we found significant differences only for the number of glandular trichomes among studied cultivars. However, the differences were rather small and may have been insufficient to impact TSSM preference or performance, as indicated by the absence of significant correlations between lupulin glands and the measured parameters. Some secondary metabolites such as α- and β-pinene, ε-murolene, and germacrene D are reported in hop glandular trichomes related to resistance against Phorodon humuli (Schrank) (Hemiptera: Aphididae), another pest in hop crops (Undas et al. 2018). However, further studies are needed to assess the role of these compounds in the resistance of hop cultivars against TSSM.
Although all susceptible, the cultivars could be classified by distribution in a multivariate analysis diagram. Victoria was rated as highly susceptible for being more attractive and allowing greater oviposition, as well as providing short development time and longer oviposition days. Cultivar Yakima Gold can be classified as less susceptible to TSSM, as it leads to a longer development time, which is strongly associated with reduced population growth. In turn, Mantiqueira can be classified as moderately susceptible as it was the least attractive and experienced rather low oviposition, longevity and fecundity. In short, our findings indicate that as Yakima Gold was the least susceptible to TSSM, this cultivar should, all other things being the same, be preferred when compared to Mantiqueira and Victoria cultivars in order to reduce mite problems. Our results also indicate that monitoring for TSSM in Mantiqueira and Victoria crops should be more intensive, given their higher susceptibility to this mite pest.
References
Akköprü EP, Atlihan R, Okut H, Chi H (2015) Demographic assessment of plant cultivar resistance to insect pests: a case study of the dusky-veined walnut aphid (Hemiptera: Callaphididae) on five walnut cultivars. J Econ Entomol 108:378–387. https://doi.org/10.1093/jee/tov011
Ako M, Borgemeister C, Poehling HM, Elbert A, Nauen R (2004) Effects of neonicotinoid insecticides on the bionomics of two-spotted spider mite Tetranychus urticae Koch (Acari: Tetranychidae). J Econ Entomol 97:1587–1594. https://doi.org/10.1603/0022-0493-97.5.1587
Awmack CS, Leather SR (2002) Host plant quality and fecundity in herbivorous insects. Ann Rev Entomol 47:817–844. https://doi.org/10.1146/annurev.ento.47.091201.145300
Barber A, Campbell CAM, Crane H, Lilley R, Tregidga E (2003) Biocontrol of twospotted spider mite Tetranychus urticae on dwarf hops by the phytoseiid mites Phytoseiulus persimilis and Neoseiulus californicus. Biocontrol Sci Technol 13:275–284. https://doi.org/10.1093/jee/tov011
Bebert S (2017) Conheça a produção de lúpulo brasileiro. https://revistagloborural.globo.com › conheca-producao-de-lupulo-brasileiro. Acessed 10 Oct 2019
Biendl M, Engelhard A, Forster A, Gahr A, Lutz A, Mitter W, Schmidt R, Schönberger C (2014) Hops: their cultivation, composition and usage, 1st edn. Fachverlag Hans Carl, Nuremberg
Bocquet L, Sahpaz S, Rivière C (2018) An overview of the antimicrobial properties of hop. In: Mérillon JM, Rivière C (eds) Natural antimicrobial agents, vol 19. Springer, Cham, pp 31–54
Calderwood L, Lewins S, Darby H (2015) Survey of northeastern hop arthropod pests and their natural enemies. J Integr Pest Manag 6:1–14. https://doi.org/10.1093/jipm/pmv017
Calderwood L, Cubins J, Vesty D, Darby H (2017) Effect of drive row ground covers on hop (Rosales: Cannabaceae) yard arthropod pests in Vermont, USA. Environ Entomol 46:183–190
Campbell CAM (2018) Influence of companion planting on damson hop aphid Phorodon humuli, two spotted spider mite Tetranychus urticae, and their antagonists in low trellis hops. Crop Prot 114:23–31. https://doi.org/10.1016/j.cropro.2018.08.014
Čerenak A, Kralj D, Javornik B (2009) Compounds of essential oils as markers of hop resistance (Humulus lupulus) to powdery mildew (Podosphaera macularis). Acta Agric Slov 93:267–273. https://doi.org/10.2478/v10014-009-0015-z
Chadwick LR (2006) The pharmacognosy of Humulus lupulus L. (hops) with an emphasis on estrogenic properties. Phytomedicine 13:119–131
Chi H (1988) Life-table analysis incorporating both sexes and variable development rates among individuals. Environ Entomol 17:26–34. https://doi.org/10.1093/ee/17.1.26
Chi H (1990) Timing of control based on the stage structure of pest populations: a simulation approach. J Econ Entomol 83:1143–1150. https://doi.org/10.1093/jee/83.4.1143
Chi H (2019a) TWOSEX-MSChart: a computer program for the age-stage, two-sex life table analysis. http://140.120.197.173/ecolo gy/Downl oad/Twose x-MSCha rt.rar. Accessed 14 Nov 2019
Chi H (2019b) Timing-MSChart: Computer program for population projection based on age-stage, twosex life table. National Chung Hsing University, Taichung, Taiwan. http://140.120.197.173/Ecology/prod02.htm. Accessed 20 Nov 2019
Chi H, Liu H (1985) Two new methods for the study of insect population ecology. Bull Inst Zool Acad Sin 24:225–240
Costa AF, Teodoro PE, Bhering LL, Fornazier MJ, Andrade JS, Martins DS, Zanuncio Junior JS (2017) Selection of strawberry cultivars with tolerance to Tetranychus urticae (Acari: Tetranychidae) and high yield under different managements. Genet Mol Res. https://doi.org/10.4238/gmr16029599
Demétrio CGB, Hinde J, Moral RA (2014) Models for overdispersed data in entomology. In: Ferreira CP, Godoy WAC (eds) Ecological modeling applied to entomology. Springer, Switzerland, pp 219–259. https://doi.org/10.1007/978-3-319-06877-0_9
Efron B, Tibshirani RJ (1993) An introduction to the bootstrap. Chapman & Hall, New York
FAOSTAT (2017) Food and agriculture organization of the United Nations, Rome, Italy. http://faostat3.fao.org/home/E. Accessed 2 Nov 2019
Fisher RA (1930) The genetical theory of natural selection: a complete, variorum. Oxford University Press, Oxford
Fürstenberg-Hägg J, Zagrobelny M, Bak S (2013) Plant defense against insect herbivores. Int J Mol Sci 14:10242–10297. https://doi.org/10.3390/ijms140510242
Gong YJ, Chen JC, Zhu L, Cao LJ, Jin GH, Hoffmann AA, Zhong CF, Wang P, Lin G, Wei SJ (2018) Preference and performance of the two-spotted spider mite Tetranychus urticae (Acari: Tetranychidae) on strawberry cultivars. Exp Appl Acarol 76:185–196. https://doi.org/10.1007/s10493-018-0295-2
Gonzalez-Dominguez SG, Santillan-Galicia MT, Gonzalez-Hernandez V, Suarez Espinosa J, Gonzalez-Hernandez H (2015) Variability in damage caused by the mite Tetranychus urticae (Trombidiformes: Tetranychidae) Koch on three varieties of strawberry. J Econ Entomol 108:1371–1380. https://doi.org/10.1093/jee/tov084
Goodman D (1982) Optimal life histories, optimal notation, and the value of reproductive value. Am Nat 119:803–823
Grbic M, Van Leeuwen T, Clark RM, Rombauts S, Rouze P, Grbic V et al (2011) The genome of Tetranychus urticae reveals herbivorous pest adaptations. Nature 479:487–492. https://doi.org/10.1038/nature10640
Hair JF Jr, Black WC, Babin BJ, Anderson RE, Tatham RL (2006) Multivariate data analysis, 6th edn. Pearson Prentice Hall, New Jersey
Helle W, Pijnacker LP (1985) Parthenogenesis, chromosomes and sex. In: Helle W, Sabelis MW (eds) Spider mites their biology, natural enemies and control, vol 1A. Elsevier, Amsterdam, pp 129–138
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363. https://doi.org/10.1002/bimj.200810425
Huang YB, Chi H (2011) The age-stage, two-sex life table with an offspring sex ratio dependent on female age. J Agric for 60:337–345
Huang YB, Chi H (2012) Age-stage, two-sex life tables of Bactrocera cucurbitae (Coquillett) (Diptera: Tephritidae) with a discussion on the problem of applying female age-specific life tables to insect populations. Insect Sci 19:263–273. https://doi.org/10.1111/j.1744-7917.2011.01424.x
Huang HW, Chi H, Smith CL (2018) Linking demography and consumption of Henosepilachna vigintioctopunctata (Coleoptera: Coccinellidae) fed on Solanum photeinocarpum (Solanales: Solanaceae): with a new method to project the uncertainty of population growth and consumption. J Econ Entomol 20:1–9. https://doi.org/10.1093/jee/tox330
Iskra AE, Woods JL, Gent DH (2019) Stability and resiliency of biological control of the twospotted spider mite (Acari: Tetranychidae) in hop. Environ Entomol 48:894–902. https://doi.org/10.1093/ee/nvz071
Islam MT, Jahan M, Gotoh T, Ullah MS (2017) Host-dependent life history and life table parameters of Tetranychus truncates (Acari: Tetranychidae). Syst Appl Acarol 22:2068–2082. https://doi.org/10.11158/saa.22.12.4
James DG (2003) Pesticide susceptibility of two coccinellids (Stethorus punctum picipes and Harmonia axyridis) important in biological control of mites and aphids in Washington hops. Biocontrol Sci Technol 13:253–259. https://doi.org/10.1080/0958315021000073510
James DG, Price TS (2002) Fecundity in twospotted spider mite (Acari: Tetranychidae) is increased by direct and systemic exposure to imidacloprid. J Econ Entomol 95:729–732. https://doi.org/10.1603/0022-0493-95.4.729
Jin GH, Gong YJ, Qian ZW, Zhu L, Wang ZH, Chen JC, Wei SJ (2016) Selectivity and fitness of the two-spotted spider mite, Tetranychus urticae (Acarina: Tetranychidae) to different varieties of eggplant. Acta Entomol Sin 59:328–336. https://doi.org/10.1007/s10493-018-0295-2
Jones G, Campbell CAM, Pye BJ, Maniar SP, Mudd A (1996) Repellent and oviposition deterring effects of hop beta acids on the two-spotted spider mite Tetranychus urticae. Pest Manag Sci 47:165–169. https://doi.org/10.1002/(SICI)1096-9063(199606)47:2%3c165::AID-PS392%3e3.0.CO;2-R
Jones G, Campbell CAM, Hardie J, Pickett JA, Pye BJ, Wadhams LJ (2010) Integrated management of two-spotted spider mite Tetranychus urticae on hops using hop ß-acids as an antifeedant together with the predatory mite Phytoseiulus persimilis. Biocontrol Sci Technol 13:241–252. https://doi.org/10.1080/0958315021000073501
Khanamani M, Fathipour Y, Hajiqanbar H (2013) Population growth response of Tetranychus urticae to eggplant quality: application of female age-specific and age-stage, two-sex life tables. Int J Acarol 39:638–648
Kyrou I, Christou A, Panagiotakos D, Stefanaki C, Skenderi K, Katsana K, Tsigos C (2017) Effects of a hops (Humulus lupulus L.) dry extract supplement on self-reported depression, anxiety and stress levels in apparently healthy young adults: a randomized, placebo-controlled, double-blind, crossover pilot study. Hormones 16:171–180. https://doi.org/10.14310/horm.2002.1738
Laing JE (1969) Life history and life table of Tetranychus urticae Koch. Acarologia 11:32–42
Lawanprasert A (1994). Response of hop cultivars to two-spotted spider mite infestation. Doctorate of Philosophy Thesis, University of Tasmania, 471f.
Lilley R, Campbell C, Ridout M (1999) Vertical dispersal of the two-spotted spider mite Tetranychus utricae, and the predatory mite Phytoseiulus persimilis on dwarf hops. Agric for Entomol 1:111–117. https://doi.org/10.1046/j.1461-9563.1999.00015.x
Mahaffee WF, Pethybridge SJ, Gent DH (eds) (2009) Compendium of hop diseases, arthropod pests, and disorders. APS Press, St. Paul
Maleknia B, Fathipour Y, Soufbaf M (2016) How greenhouse cucumber cultivars affect population growth and two-sex life table parameters of Tetranychus urticae (Acari: Tetranychidae). Int J Acarol 42:70–78. https://doi.org/10.1080/01647954.2015.1118157
Marcusso EF, Müller, CV. (2018). Anuário da Cerveja no Brasil 2018: Crescimento e Inovação. http://www.agricultura.gov.br/assuntos/inspecao/produtos-vegetal/arquivos/in-no-72-de-16-de-novembro-de-2018.pdf. Acessed 2 Nov 2019
Mayberry TW (1968) Varietal resisitance of hops, Humulus lupulus (Linn.), to the two-spotted spider mite, Tetranychus urticae (Koch). Master Thesis, Oregon State University, Oregon. 75 f
Melville CC, Zampa SF, Savi PJ, Michelotto MD, Andrade DJ (2019) Peanut cultivars display susceptibility by triggering outbreaks of Tetranychus ogmophallos (Acari: Tetranychidae). Exp Appl Acarol 78:295–314. https://doi.org/10.1007/s10493-019-00385-1
Neve RA (ed) (1991) Hops. Chapman and Hall, London, pp 1–272
Patzak J, Krofta K, Henychová A, Nesvadba V (2015) Number and size of lupulin glands, glandular trichomes of hop (Humulus lupulus L.), play a key role in contents of bitter acids and polyphenols in hop cone. Int J Food Sci Technol 50:1864–1872. https://doi.org/10.1111/ijfs.12825
Peters K, Berry R (1980) Effect of hop leaf morphology on two spotted spider mite. J Econ Entomol 73:235–238
Piraneo T, Bull J, Morales M, Lavine LC, Walsh DB, Zhu F (2015) Molecular mechanisms of Tetranychus urticae chemical adaptation in hop fields. Sci Rep 5:17090. https://doi.org/10.1038/srep17090
R Core Team (2012) R: a language and environment for statistical computing. R Development Core Team, Vienna
Regev S, Cone W (1975) Chemical differences in hop varieties vs. susceptibility to the two spotted spider mite. Environ Entomol 4:697–700. https://doi.org/10.1093/ee/4.5.697
Rodrigues MA, Morais JS, de Castro JPM (Eds.) (2015). Jornadas de lúpulo e cerveja: novas oportunidades de negócio: livro de atas. Bragança: Instituto Politécnico, CIMO. http://hdl.handle.net/10198/11625.Acessed 10 Oct 2019
Rostami N, Maroufpoor M, Sadeghi A, Ghazi MM, Atlihan R (2018) Demographic characteristics and population projection of Phytonemus pallidus fragariae reared on different strawberry cultivars. Exp Appl Acarol 76:473–496
Sarnighausen P, Sarnighausen VCR, Dal Pai A. O Lúpulo e a oportunidade do Agronegócio no Brasil. 6a Jornada de Iniciação Científica da FATEC de Botucatu, 23 a 27 de outubro de 2017, Botucatu, São Paulo, Brasil.
Savi PJ, de Moraes GJ, Melville CC, Andrade DJ (2019a) Population performance of Tetranychus evansi (Acari: Tetranychidae) on African tomato varieties and wild tomato genotypes. Exp Appl Acarol 77:555–570. https://doi.org/10.1007/s10493-019-00364-6
Savi PJ, de Moraes GJ, Boiça Junior AL, Melville CC, Carvalho RF, Lourenção AL, Andrade DJ (2019b) Impact of leaflet trichomes on settlement and oviposition of Tetranychus evansi (Acari: Tetranychidae) in African and South American tomatoes. Syst Appl Acarol 24:2559–2576. https://doi.org/10.11158/saa.24.12.19
Sedaratian A, Fathipour Y, Moharramipour S (2009) Evaluation of resistance in 14 soybean genotypes to Tetranychus urticae (Acari: Tetranychidae). J Pest Sci 82:163–170. https://doi.org/10.1007/s10340-008-0235-8
Sedaratian A, Fathipour Y, Moharramipour S (2010) Comparative life table analysis of Tetranychus urticae (Acari: Tetranychidae) on 14 soybean genotypes. Insect Sci 18:541–553. https://doi.org/10.1111/j.1744-7917.2010.01379.x
Seki K (2016) Leaf morphology-assisted selection for resistance to two-spotted spider mite Tetranychus urticae Koch (Acari: Tetranychidae) in carnations (Dianthus caryophyllus L.). Pest Manag Sci 72:1926–1933. https://doi.org/10.1002/ps.4231
Shoorooei M, Hoseinzadeh AH, Maali-Amiri R, Allahyari H, Torkzadeh-Mahani M (2018) Antixenosis and antibiosis response of common bean (Phaseolus vulgaris) to two-spotted spider mite (Tetranychus urticae). Exp Appl Acarol 74:365–381. https://doi.org/10.1007/s10493-018-0240-4
Silva CT (2019) Caracterizações químicas dos primeiros cultivares de lúpulo (Humulus lupulus L.) produzidos no brasil. Vitória: Universidade Federal do Espírito Santo. 90f. Mestrado em Agroquímica.
Statsoft I (2004) STATISTICA (data analysis software system), version 7. http://www.stats oft.com. Accessed 25 Nov 2019
Tuan SJ, Lee CC, Chi H (2014a) Erratum: Population and damage projection of Spodoptera litura (F.) on peanuts (Arachis hypogaea L.) under different conditions using the age-stage, two-sex life table. Pest Manag Sci 70:805–813. https://doi.org/10.1002/ps.3618
Tuan SJ, Lee CC, Chi H (2014b) Population and damage projection of Spodoptera litura (F) on peanuts (Arachis hypogaea L) under different conditions using the age-stage, two-sex life table. Pest Manag Sci 70:19–36. https://doi.org/10.1002/ps.3618
Undas AK, Weihrauch F, Lutz A, van Tol R, Delatte T, Verstappen F, Bouwmeester H (2018) The use of metabolomics to elucidate resistance markers against damson-hop aphid. J Chem Ecol 44:711–726. https://doi.org/10.1007/s10886-018-0980-y
Van Leeuwen T, Vontas J, Tsagkarakou A, Dermauw W, Tirry L (2010) Acaricide resistance mechanisms in the two-spotted spider mite Tetranychus urticae and other important Acari: a review. Insect Biochem Mol Biol 40:563–572. https://doi.org/10.1016/j.ibmb.2010.05.008
Van Leeuwen T, Tirry L, Yamamoto A, Nauren R, Dermauw W (2015) The economic importance of acaricides in the control of phytophagous mites and an update on recent acaricide mode of action research. Pestic Biochem Phys 121:12–21. https://doi.org/10.1016/j.pestbp.2014.12.009
Walsh DB, Gent DH, Barbour JD, Boydston RA, George AE, James DG, Sirrine JR (eds) (2015) Field guide for integrated pest management in hops. Washington Hop Commission, Moxee
Wang G, Tian L, Aziz N, Broun P, Dai X, He J, Dixon RA (2008) Terpene biosynthesis in glandular trichomes of hop. Plant Physiol 148:1254–1266. https://doi.org/10.1104/pp.108.125187
Weihrauch F (2005) Evaluation of a damage threshold for two-spotted spider mites, Tetranychus urticae Koch (Acari: Tetranychidae), in hop culture. Ann Appl Biol 146:501–509. https://doi.org/10.1111/j.1744-7348.2005.040163.x
Weihrauch F, Moreth L (2005) Behavior and population development of Phorodon humuli (Schrank) (Homoptera: Aphididae) on two hop cultivars of different susceptibility. J Insect Behav 18:693–705. https://doi.org/10.1007/s10905-005-7020-9
Woods J, Dreves A, James D, Lee J, Walsh D, Gent DH (2014) Development of biological control of Tetranychus urticae (Acari: Tetranychidae) and Phorodon humuli (Hemiptera: Aphididae) in Oregon hop yards. J Econ Entomol 107:570–581. https://doi.org/10.1603/EC13488
Wu M, Adesanya AW, Morales MA, Walsh DB, Lavine LC, Lavine MD, Zhu F (2019) Multiple acaricide resistance and underlying mechanisms in Tetranychus urticae on hops. J Pest Sci 92:543–555. https://doi.org/10.1007/s10340-018-1050-5
Xu D, Wang K, Zhang Y, Wang H, Wu Q, Wang S (2019) The performance of Tetranychus urticae on five melon cultivars is correlated with leaf thickness. Syst Appl Acarol 24:645–658. https://doi.org/10.11158/saa.24.4.10
Acknowledgements
To the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES-Financial Code 001—for granting a doctoral scholarship to the first and second authors.
Author information
Authors and Affiliations
Contributions
Conceptualization: PJS, RFG and DJA. Methodology, Investigation and Analysis: PJS. Writing: PJS, RFG, PJS, STSM, LTB, GJM and DJA. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Savi, P.J., Gonsaga, R.F., de Matos, S.T.S. et al. Performance of Tetranychus urticae (Acari: Tetranychidae) on three hop cultivars (Humulus lupulus). Exp Appl Acarol 84, 733–753 (2021). https://doi.org/10.1007/s10493-021-00643-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-021-00643-1