Abstract
Genetic identity and morphological features of a human biting tick, Amblyomma testudinarium, were determined for the first time in Taiwan. Morphological features of adult male and female ticks of Am. testudinarium were observed and photographed by a stereo- microscope. The genetic identity was analyzed by comparing the sequences of mitochondrial 16S ribosomal DNA gene obtained from 18 strains of ticks representing 10 species of Amblyomma, and four outgroup species of Dermacentor and Rhipicephalus ticks. Nine major clades could be easily distinguished by neighbour-joining analysis and were congruent by maximum-parsimony method. All these Am. testudinarium ticks collected from Taiwan and Japan were genetically affiliated to a monophyletic group with highly homogeneous sequence (99.8–100% similarity), and can be discriminated from other species of Amblyomma and other genera of ticks (Dermacentor and Rhipicephalus) with a sequence divergence ranging from 6.9 to 23.9%. Moreover, intra- and inter-species analysis based on the genetic distance (GD) values indicated a lower level (GD < 0.003) within the same lineage of Am. testudinarium ticks collected from Taiwan and Japan, as compared with other lineage groups (GD > 0.108) of Amblyomma ticks, as well as outgroup (GD > 0.172) species. Our results provide the first distinguished features of adult Am. testudinarium ticks and the first genetic identification of Am. testudinarium ticks collected from humans in Taiwan. Seasonal prevalence, host range, and vectorial capacity of this tick species in Taiwan need to be further clarified.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ticks are obligate hematophagous arthropods that parasitize every class of vertebrates in almost every region of the world and it may act as vectors with the ability to transmit various pathogens including bacteria, rickettsiae, and protozoan (Balashov 1972). There are three generally recognized families of ticks: the Ixodidae, the Argasidae, and the Nuttalliellidae (Oliver 1989; Sonenshine 1991). Based on studies of valid names of tick species, the Amblyomma genus comprising 130 species was recognized as the second largest group of Ixodidae ticks (Horak et al. 2002; Barker and Murrell 2008; Guglielmone et al. 2010). In addition, the global importance of ticks was determined by their ability to transmit a variety of tick-borne pathogens associated with human diseases and by direct damage to livestock due to their feeding behaviour (Jongejan and Uilenberg 2004). The impact of ticks and tick-borne pathogens on human and animal health becomes a major concern in public health.
Amblyomma testudinarium is a rare tick species reported only in Asia countries including Malaysia, India, Japan, Korea, Eastern China and Taiwan (Luh and Woo 1950; Nagahana and Matuo 1962; Dhanda and Rao 1964; Maa and Kuo 1966; Suzuki et al. 1990; Robbins 2005). It has previously been reported as a common ectoparasite of several species of reptiles, wild cats, dogs and wild boars (Motoi et al. 2013; Tateno et al. 2015; Suh et al. 2016), and occasionally on humans (Estrada-Pena and Jongejan 1999). This tick species was detected with various pathogens including Borrelia, rickettsia, Hepatozoon and tick-borne virus (Motoi et al. 2013; Ho et al. 2014; Sun et al. 2015; Tateno et al. 2015; Suh et al. 2016; Yun et al. 2016). The medical and veterinary importance with the recent emergence of human babesiosis (Shih et al. 1997), Lyme borreliosis (Shih and Chao 1998; Chao et al. 2011), canine babesiosis (Lee et al. 2010) and rodent babesial infection (Chao et al. 2017b) in Taiwan raises the focus of research attention on vector ticks. Although the hard ticks of Ixodes granulatus, I. ovatus and Rhipicephalus sanguineus had been identified as the incriminated vector ticks for the transmission of Borrelia and Babesia in Taiwan (Chao et al. 2009, 2016, 2017a), the morphological features and genetic identity of Am. testudinarium collected from humans in Taiwan have never been reported.
It may be that the tick species of Am. testudinarium collected from Taiwan is a genetically distinct lineage, as compared with the existing common ticks of Amblyomma species around the world and the potential of genetic variation in relation to the geographical distribution may also exist among these Am. testudinarium ticks characterized with similar morphology. Thus, the objective of this study intends to investigate the genetic identity of Am. testudinarium ticks collected from Taiwan, and to analyze the phylogenetic relationships between and within the species of Amblyomma ticks by analyzing the mitochondrial 16S rDNA gene. The genetic divergence of Am. testudinarium ticks collected from Taiwan was also analyzed by comparing with other tick species identified from various geographical sources which have been documented in GenBank.
Materials and methods
Collection and identification of tick specimen
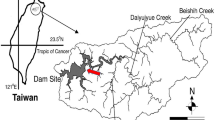
A total of nine adult Am. testudinarium (7 females and 2 males) were collected from attached or infested humans of different localities in Taiwan (22°17′N, 120°52′E; 23°16′N, 120°51′E; 23°04′N, 120°47′E; 23°28′N, 120°54′E; 24°10′N, 121°24′E; 25°06′N, 121°35′E; 25°05′N, 121°35′E) (Fig. 1). All these adult ticks were removed from attached or infested humans (Table 1; Fig. 2), and were subsequently stored in 70% ethanol or plastic vials until further analysis. All tick specimens of Am. testudinarium were identified to species level on the basis of their morphological characteristics, as described previously (Shih and Chao 2011). Ultrastructural observations by stereo-microscope were used to delineate the morphological features of adult Am. testudinarium ticks in Taiwan. Briefly, tick specimens were cleaned by sonication in 70% ethanol solution for 5–10 min and then washed twice in sterile distilled water. Afterwards, each tick specimen was placed on a glass slide and photographed using a stereo-microscope (SMZ 1500, Nikon, Tokyo, Japan) equipped with a fiber lamp. The external features of the Am. testudinarium ticks were recorded for species identification.
DNA extraction from tick specimen
Total genomic DNA was extracted from individual tick specimens used in this study. Briefly, tick specimens were cleaned by sonication for 3–5 min in ethanol solution and then washed twice in sterile distilled water. Afterwards, the individual tick specimen dissected into pieces was placed in a microcentrifuge tube filled with 180-μL lysing buffer solution supplied with a DNeasy Tissue Kit (catalogue no. 69506, Qiagen, Taipei, Taiwan) and then homogenized with a TissueLyser II (catalogue no. 85300, Qiagen, Germany), instructed by the manufacturer. The homogenate was centrifuged at room temperature and the supernatant fluid was further processed by a DNeasy Tissue Kit, as instructed by the manufacturer. After filtration, the filtrate was collected and the DNA concentration was determined spectrophotometrically with a DNA calculator (Nanovue Plus Spectrophotometer).
DNA amplification by polymerase chain reaction (PCR)
DNA samples extracted from the tick specimens were used as a template for PCR amplification. A specific primer set of 16S+1 (5′-CTGCTCAATGATTTTTTAAATTGCTGTGG-3′) corresponding to the 3′ end of the mitochondrial 16S rDNA and 16S−1 (5′-CCGGTCTGAACTCAGATCAAGT-3′) corresponding to the 5′ end of the mitochondrial 16S rDNA were designed to target the mitochondrial 16S rDNA gene, as described previously (Black and Piesman 1994). All PCR reagents and Taq polymerase were obtained and used as recommended by the supplier (Takara Shuzo, Japan). Briefly, a total of 0.2-μmol of the appropriate primer set and adequate amounts of template DNA were used in each 50-μl reaction mixture. In contrast, adequate amounts of sterile distilled water were added for serving as a negative control. PCR amplification was performed with a Perkin-Elmer Cetus thermocycler (GeneAmp system 9700) and was amplified for 10 cycles with the conditions of denaturation at 92 °C for 1 min, annealing at 48 °C for 1 min, and extension at 72 °C for 90 s., then followed by 32 cycles with the conditions of denaturation at 92 °C for 40 s, annealing at 54 °C for 35 s, and extension at 72 °C for 90 s. Thereafter, amplified DNA products were electrophoresed on 2% agarose gels in Tris-Borate-EDTA (TBE) buffer and visualized under ultraviolet (UV) light after staining with ethidium bromide. A DNA ladder (1-kb plus, catalogue no. 10787-018, Invitrogen, Taipei, Taiwan) was used as the standard marker for comparison. A negative control of distilled water was included in parallel with each amplification.
Sequence alignments and phylogenetic analysis
After purification (QIAquick PCR Purification Kit, catalog No. 28104), sequencing reaction was performed with 25 cycles under the same conditions and same primer set of initial amplification by dye-deoxy terminator reaction method using the Big Dye Terminator Cycle Sequencing Kit in an ABI Prism 377-96 DNA Sequencer (Applied Biosystems, Foster City, CA, USA). The resulting sequences were initially edited by BioEdit software (V5.3) and aligned with the CLUSTAL W software (Thompson et al. 1994). Thereafter, the aligned sequences of 3 ticks of Taiwan were further analyzed by comparing with other 15 strains of tick specimens based on the different genus and different geographical origin of Amblyomma, Dermacentor, and Rhipicephalus ticks that are available in GenBank. Phylogenetic analysis was performed by neighbour-joining (NJ) compared with maximum parsimony (MP) methods to estimate the phylogeny of the entire alignment using MEGA 6.0 software package (Tamura et al. 2013). The genetic distance values of inter- and intra-species variations were also analyzed by the Kimura two-parameter model (Kimura 1980). All phylogenetic trees were constructed and performed with 1000 bootstrap replications to evaluate the reliability of the construction, as described previously (Felsenstein 1985).
Nucleotide sequence accession numbers
The nucleotide sequences of PCR-amplified mitochondrial 16S rDNA genes of three strains of Am. testudinarium ticks determined in this study have been registered and assigned the following GenBank accession numbers: strains TWKL-Amt1 (KX685426), TWKL-Amt2 (KX685427), and TWKL-Amt3 (KX685428), respectively. For phylogenetic analysis, a total of 18 strains of ticks representing ten species of Amblyomma, and four outgroup species of Dermacentor (D. andersoni and D. variabilis) and Rhipicephalus (R. sanguineus and R. appendiculatus) ticks were included for comparison and their GenBank accession numbers are shown in Table 2.
Results
Morphological identification of Amblyomma testudinarium ticks collected from attached humans
The ultrastructural features of adult stage of male and female Am. testudinarium tick characterized with one pair of long-narrow palp, oval-shaped body, four pairs of legs, and rectangular-shaped basis capituli (Fig. 3a, d). The rhombus-shaped scutum (long-arrow indicated on Fig. 3a) of female tick bears metallic yellowish markings with deep punctations and one pair of protrude eyes (short-arrows indicated on Fig. 3a) located at the lateral margin of rhombus-shaped scutum. In contrast, the oval-shaped scutum covered whole body of male tick bears one pair of protrude eyes (short-arrows indicated on Fig. 3d) located at the lateral margin near leg and clear festoom located at the bottom of scutum (long-arrow indicated on Fig. 3d). Ventral view of female Am. testudinarium shows the prolonged hypostome equipped with numerous sharp teeth of four/four dentition (long-arrow indicated on Fig. 3c), as compared to the curved teeth of male tick (long-arrow indicated on Fig. 3f). The internal spurs of short triangular observed only on coxae I (long-arrows indicated on Fig. 3b, e) and absent on coxae II–IV of female and male ticks. The external spurs of distinct cone-like were observed on coxa I of female, and coxae I and IV of male (short-arrows indicated on Fig. 3b, e). The external spurs of broad triangular were observed on coxae II–IV of female and coxae II–III of male (arrowheads indicated on Fig. 3b, e). In addition, the genital aperture of female Am. testudinarium was situated at level between coxae II and III (short-arrow indicated on Fig. 3b). In contrast, the genital aperture of male Am. testudinarium was situated at level between coxae II (short-arrow indicated on Fig. 3e).
Light micrographs of female (a–c) and male (d–f) Amblyomma testudinarium ticks collected from Taiwan. a, d Dorsal view of female and male ticks characterized with one pair of long-narrow palp, oval-shaped body, four pairs of legs, rectangular-shaped basis capituli. a The rhombus-shaped scutum (long-arrow) of female tick bears metallic yellowish markings with deep punctations and one pair of protrude eyes (short-arrows) located at the lateral margin of rhombus-shaped scutum. d The oval-shaped scutum covered whole body of male tick bears one pair of protrude eyes (short-arrows) located at the lateral margin near leg and clear festoom located at the bottom of scutum (long-arrow). b, e Ventral view of female and male Am. testudinarium shows the internal spurs of short triangular observed only on coxae I (long-arrows) and absent on coxae II–IV of female and male ticks. The external spurs of distinct cone-like were observed on coxa I of female, and coxae I and IV of male (short-arrows), and the external spurs of broad triangular were observed on coxae II–IV of female and coxae II–III of male (arrowheads). In addition, the genital aperture was situated at level between coxae II and III of female, and at level between coxae II of male (short-arrow). c, f Ventral view of female Am. testudinarium shows the prolonged hypostome equipped with numerous sharp teeth of four/four dentition (long-arrow), as compared to the curved teeth of male tick (long-arrow)
Sequence alignment and genetic analysis
To clarify the genetic identity of Am. testudinarium ticks of Taiwan, the sequences of mitochondrial 16S rDNA fragments of 3 Taiwan strains of Am. testudinarium performed by this study were aligned and compared with the downloaded sequences of 11 different geographical strains of Amblyomma ticks, and 4 outgroup strains of Dermacentor and Rhipicephalus ticks from GenBank. Results indicate that the lengths of the aligned sequences were measured from 365 to 380 bp, and the nucleotide sequences between the 3 strains of Am. testudinarium of Taiwan were highly homogeneous (100% similarity). All these Am. testudinarium ticks of Taiwan and Japan were genetically affiliated to a monophyletic group with highly homogeneous sequence (99.8–100% similarity), and can be discriminated from other species of Amblyomma ticks and other genera (Dermacentor and Rhipicephalus) ticks with a sequence divergence ranging from 6.9 to 23.9% (Table 3). In addition, intra- and inter-species analysis based on the genetic distance (GD) values indicated a lower level (GD < 0.003) within the same lineage of Am. testudinarium ticks collected from Taiwan and Japan, as compared with other lineage group (GD > 0.108) of Amblyomma ticks, as well as outgroup species (GD > 0.172) of Dermacentor and Rhipicephalus ticks (Table 3).
Phylogenetic analysis of tick specimens
Phylogenetic relationships based on the sequence alignment of mitochondrial 16S rDNA were performed to demonstrate the genetic divergence among 18 strains of ticks investigated in this study. Bootstrap analysis was used to analyze the repeatability of the clustering of specimens represented in phylogenetic trees. Phylogenetic trees constructed by both NJ (Fig. 4) and MP (Fig. 5) analyses showed congruent basal topologies with nine major branch of distinguished clades (Figs. 4, 5). All these Am. testudinarium ticks of Taiwan constitute a monophyletic clade closely affiliated to the same lineage group of Am. testudinarium ticks, and can be easily discriminated from other lineage groups of Amblyomma ticks with a bootstrap value of 98 in NJ analysis (Fig. 4). The phylogenetic tree of MP analysis was identical to the NJ tree and strongly support the separation of different lineages between the Am. testudinarium from Taiwan and the other lineage groups of Amblyomma ticks with a bootstrap value of 91 (Fig. 5). These results reveal a lower genetic divergence within the same species of Am. testudinarium ticks from Taiwan, but a higher genetic variations among different lineage or genus of Amblyomma ticks.
Phylogenetic relationships based on the 16S ribosomal DNA (rDNA) gene sequences between three strains of Amblyomma testudinarium ticks from Taiwan, two strains of Am. testudinarium from Japan and Thailand, and nine other strains belonging to nine species of Amblyomma. Two species of Dermacentor and Rhipicephalus ticks served as outgroup comparison. The trees were constructed and analyzed by neighbour-joining (NJ) method using 1000 bootstraps replicates. Numbers at the nodes indicate the percentages of reliability of each branch of the tree. Branch lengths are drawn proportional to the estimated sequence divergence
Phylogenetic relationships based on the 16S ribosomal DNA (rDNA) gene sequences between three strains of Amblyomma testudinarium ticks from Taiwan, two strains of Am. testudinarium from Japan and Thailand, and nine other strains belonging to nine species of Amblyomma. Two species of Dermacentor and Rhipicephalus ticks served as outgroup comparison. The trees were constructed and analyzed by maximum parsimony (MP) method using 1000 bootstraps replicates. Numbers at the nodes indicate the percentages of reliability of each branch of the tree. Branch lengths are drawn proportional to the estimated sequence divergence
Discussion
This study provides the first distinguished colour figures of adult Am. testudinarium ticks in Asia and describes the first genetic identification based on the mitochondrial 16S ribosomal gene among Am. testudinarium ticks collected from Taiwan. Although species determination and differentiation of Amblyomma ticks have traditionally been based on morphological features of the adult stages of these ticks, a DNA-based approach provides the feasibility to investigate the genetic variance at the individual base-pair level and gives much more direct pathway for measuring the genetic diversity between and within species of Amblyomma ticks (Black and Piesman 1994; Burger et al. 2012; Lopes et al. 2016). In previous investigations, sequence analysis of the mitochondrial 16S rDNA have been used to distinguish and to assess the phylogenetic relationships of diverse species of closely related Ixodes, Rhipicephalus and Amblyomma ticks by comparing their nucleotide variations of the mitochondrial 16S rDNA (Black and Piesman 1994; Chao et al. 2009; Burger et al. 2012; Erster et al. 2013; Lopes et al. 2016; Zemtsova et al. 2016). Indeed, results from this study demonstrate that the nucleotide composition of the mitochondrial 16S rDNA derived from these Am. testudinarium ticks collected from Taiwan and Japan is genetically affiliated to a monophyletic group with highly homogeneous sequence (99.8–100% similarity), and can be discriminated from the same lineage group of Am. testudinarium tick from Thailand, other species of Amblyomma ticks and other genera (Dermacentor and Rhipicephalus) ticks with a sequence divergence ranging from 6.9 to 23.9% (Table 3; Fig. 4). Although this tick species had been recorded in Taiwan (Robbins 2005), no genetic characteristics were available for species identification. Thus, our study demonstrates the first molecular evidence confirming the genetic identity of Am. testudinarium ticks collected in Taiwan and provides the first convincing sequences (GenBank accession numbers: KX685426–KX685428) of Am. testudinarium ticks in Taiwan.
The geographical distribution of Am. testudinarium in Taiwan and eastern Asia needs to be further defined. In previous reports, this tick species has been found majority on reptilian hosts, wild cats and wild boars in northeastern Asia (i.e., Korea and Japan) (Motoi et al. 2013; Tateno et al. 2015; Suh et al. 2016), and human infestation may occasionally occur (Estrada-Pena and Jongejan 1999). In this report, seven female and two male ticks attached or infested on humans were collected from various localities in the lower and higher mountain areas of mainland Taiwan (Fig. 1), and these specimens provide the first evidence regarding the feeding activity of Am. testudinarium on humans in Taiwan. Because of increased international trade in wild reptiles in Taiwan (Shiau et al. 2006), accidental transportation of ticks on imported reptiles may contribute to the introduction of exotic ticks into Taiwan from its adjacent Countries. Indeed, accidental transportation of Am. cordiferum and Aponomma varanense ticks on a king cobra snake from Malaysia to Taiwan has also been reported (Norval et al. 2009). Thus, the geographical dissemination of Am. testudinarium tick throughout the eastern Asia is highly anticipated. Further investigation on the identification of Am. testudinarium tick in those areas may facilitate our understanding of geographical distribution of Am. testudinarium ticks in eastern Asia.
The natural cycle of life stages of Am. testudinarium ticks in Taiwan remains unknown. This tick species is known as a reptilian tick in northeastern Asia, and most reported collections are from snakes. Indeed, previous studies indicated that eight species of snakes have been found parasitized by this tick species and this distinctive species is almost exclusively a reptile tick (Suh et al. 2016). In this report, all these adult females and males of Am. testudinarium ticks were collected from infested humans throughout the mainland Taiwan. However, there is no collection of larval and nymphal stages of Am. testudinarium for further identification. Because the feeding habitat for the snake depends mainly on catching of rodents or small mammal. Thus, it is postulated that reptilian host of snakes and small mammal of rodents may serve as infested hosts for adult and immature stages of Am. testudinarium in Taiwan, respectively. Further investigation focused on the seasonal prevalence and the host specificity of various stages of Am. testudinarium ticks would help to reveal the developmental cycle of Am. testudinarium ticks in Taiwan.
In conclusion, this study describes the first report of feeding activity of Am. testudinarium tick on humans and provides the first genetic identification of the mitochondrial 16S rDNA gene of Am. testudinarium ticks collected from the mainland Taiwan. Further study focused on the feeding habitat and vectorial capacity of this distinctive species of Am. testudinarium tick may enhance our understanding of the life cycle and epidemiological feature of tick-borne pathogens transmitted by Am. testudinarium tick in Taiwan.
References
Balashov YS (1972) Bloodsucking ticks (Ixodoidea)-vectors of diseases of man and animals. Misc Publ Entomol Soc Am 8:268–305
Barker SC, Murrell A (2008) Systematics and evolution of ticks with a list of valid genus and species names. In: Bowman AS, Nuttall PA (eds) Ticks: biology, disease and control. Cambridge University Press, Cambridge
Black WC IV, Piesman J (1994) Phylogeny of hard- and soft-tick taxa (Acari: Ixodida) based on mitochondrial 16S ribosomal DNA sequences. Proc Natl Acad Sci USA 91:10034–10038
Burger TD, Shao RF, Beati L, Miller H, Barker SC (2012) Phylogenetic analysis of ticks (Acari: Ixodida) using mitochondrial genomes and nuclear rRNA genes indicates that the genus Amblyomma is polyphyletic. Mol Phylogenet Evol 64:45–55
Chao LL, Wu WJ, Shih CM (2009) Molecular analysis of Ixodes granulatus, a possible vector tick for Borrelia burgdorferi sensu lato in Taiwan. Exp Appl Acarol 48:329–344
Chao LL, Chen YJ, Shih CM (2011) First isolation and molecular identification of Borrelia burgdorferi sensu stricto and B. afzelii from skin biopsies of patients in Taiwan. Int J Infect Dis 15:e182–e187
Chao LL, Yeh ST, Hsieh CK, Shih CM (2016) First detection and molecular identification of Babesia vogeli from Rhipicephalus sanguineus (Acari: Ixodidae) in Taiwan. Exp Appl Acarol 68:539–551
Chao LL, Liao HT, Ho TY, Shih CM (2017a) First detection and molecular identification of Babesia gibsoni from Rhipicephalus sanguineus ticks. Acta Trop 166:356–362
Chao LL, Yu WC, Shih CM (2017b) First detection and molecular identification of Babesia microti in Rattus losea captured from the offshore Kinmen Island of Taiwan. Ticks Tick-Borne Diseases 8:313–319
Dhanda V, Rao TR (1964) A report on the collection of ixodid ticks made in the North East Agency frontier, India. Indian Med Res 52:1139–1153
Erster O, Roth A, Wolkomirsky R, Leibovich B, Shkap V (2013) Comparative analysis of mitochondrial markers from four species of Rhipicephalus (Acari: Ixodidae). Vet Parasitol 198:364–370
Estrada-Pena A, Jongejan F (1999) Ticks feeding on humans: a review of records on human-biting Ixodoidea with special reference to pathogen transmission. Exp Appl Acarol 23:685–715
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 52:1119–1134
Guglielmone AA, Robbins RG, Apanaskevich DA et al (2010) The Argasidae, Ixodidae and Nuttalliellidae (Acari: Ixodida) of the world: a list of valid species names. Zootaxa 2528:1–28
Ho J, Ling F, Chai C, Lu Y, Yu X, Lin J, Sun J, Chang Y, Ye X, Gu S, Pang W, Wang C, Zheng X, Jiang J, Chen Z, Gong Z (2014) Prevalence of Borrelia burgdorferi Sensu Lato in ticks from Eastern China. Am J Trop Med Hyg 92(2):262–266
Horak IG, Camicas JL, Keirans JE (2002) The Argasidae, Ixodidae and Nuttalliellidae (Acri: Ixodida): a world list of valid tick names. Exp Appl Acarol 28:27–54
Jongejan F, Uilenberg G (2004) The global importance of ticks. Parasitology 129:S3–S14
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Lee CC, Hsieh YC, Huang CC et al (2010) Sequence and phylogenetic analysis of the thrombospondin-related adhesive protein (TRAP) gene of Babesia gibsoni isolates from dogs in Taiwan. J Vet Med Sci 72:1329–1335
Lopes MG, Junior JM, Foster RJ, Harmsen BJ, Sanchez E, Martins TF, Quigley H, Marcili A, Labruna MB (2016) Ticks and rickettsiae from wildlife in Belize, Central America. Parasites Vectors 9:62
Luh PL, Woo WC (1950) A list of Chinese ticks. Chin J Entomol 1:195–222
Maa TC, Kuo JS (1966) Catalogue and bibliography of ticks and mites parasitic on vertebrates in Taiwan. Q J Taiwan Museum 19:373–413
Motoi Y, Asano M, Inokuma H, Ando S, Kawabata H, Takano A, Suzuki M (2013) J Vet Med Sci 75(3):262–267
Nagahana M, Matuo K (1962) Two tick species parasitic on the human body. Med Biol Tokyo 62:119–121
Norval G, Robbins RG, Kolonin G, Shiao PJ, Mao JJ (2009) Unintentional transport of ticks into Taiwan on a king cobra (Ophiophagus hannah). Herpetol Notes 2:203–206
Oliver JH Jr (1989) Biology and systematics of ticks (Acari: Ixodida). Annul Rev Eco Syst 20:397–430
Robbins RG (2005) The ticks (Acari: Ixodidae: Argasidae, ixodidae) of Taiwan: a synonymic checklist. Proc Entomol Soc Wash 10:245–253
Shiau TW, Hou PC, Wu SH, Tu MC (2006) A survey of alien pet reptiles in Taiwan. Taiwania 51:71–80
Shih CM, Chao LL (1998) Lyme disease in Taiwan: primary isolation of Borrelia burgdorferi-like spirochetes from rodents in Taiwan area. Am J Trop Med Hyg 59:687–692
Shih CM, Chao LL (2011) A catalog of Ixodidae ticks of Taiwan. National Defense Medical Center, Taipei
Shih CM, Liu LP, Chung WC et al (1997) Human babesiosis in Taiwan: asymptomatic infection with a Babesia microti-like organism in a Taiwanese woman. J Clin Microbiol 35:450–454
Sonenshine DE (1991) Biology of ticks. Oxford University Press, New York
Suh JH, Kim HC, Yun SM, Lim JW, Kim JH, Chong ST, Kim DH, Kim HT, Kim H, Klein TA, Johnson JL, Lee WJ (2016) Detection of SFTS virus in Ixodes nipponensis and Amblyomma testudinarium (Ixodida: Ixodidae) collected from reptiles in the Republic of Korea. J Med Entomol 53(3):584–590
Sun J, Lin J, Gong Z, Chang Y, Ye X, Gu S, Pang W, Wang C, Zheng X, Hou J, Ling F, Shi X, Jiang J, Chen Z, Lv H, Chai C (2015) Detection of spotted fever group Rickettsiae in ticks from Zhejiang Province, China. Exp Appl Acarol 65:403–411
Suzuki N, Yamaguti N, Saruta T, Hashiguchi Y (1990) Twelve cases of tick-bite among humans in kochi Prefecture. Jpn J Saint Zool 41:71–74
Tamura K, Stecher G, Peterson D et al (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Tateno M, Sunahara A, Nakanishi N, Izawa M, Matsuo T, Setoguchi A, Endo Y (2015) Molecular survey of arthropod-borne pathogens in ticks obtained from Japaneses wildcats. Ticks Tick-Borne Dis 6:281–289
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nuc Acids Res 22:4673–4680
Yun SM, Lee YJ, Choi WY, Kim HC, Chong ST, Chang KS, Coburn JM, Klein TA, Lee WJ (2016) Molecular detection of severe fever with thrombocytopenia syndrome and tick-borne encephalitis viruses in ixodid ticks collected from vegetation, Republic of Korea, 2014. Ticks Tick-Borne Dis 7:970–978
Zemtsova GE, Apanaskevich DA, Reeves WK, Hahn M, Snellgrove A, Levin ML (2016) Phylogeographical of Rhipicephalus sanguineus sensu lato and its relationships with climatic factors. Exp Appl Acarol 69:191–203
Acknowledgements
This work was supported in part by grants from Center for Infectious Disease and Cancer Research (KMU-TP105E32) and the Kaohsiung Medical University Research Foundation (KMU-M106007), Kaohsiung Medical University, Kaohsiung, Taiwan, ROC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chao, LL., Lu, CW., Lin, YF. et al. Molecular and morphological identification of a human biting tick, Amblyomma testudinarium (Acari: Ixodidae), in Taiwan. Exp Appl Acarol 71, 401–414 (2017). https://doi.org/10.1007/s10493-017-0119-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-017-0119-9