Abstract
A novel aerobic bacterium, KC1T, was isolated from a salt mine in Kuche county, Xinjiang province, China. Cells were observed to be Gram-positive, rod-shaped, endospore-forming and motile with flagella. Strain KC1T was found to grow at 25–45 °C (optimum 37 °C), pH 6.5–9.0 (optimum 8.0) and NaCl 0–10 % (v/v) (optimum 4 %). The major fatty acids were identified as anteiso-C15:0 and anteiso-C17:0. Menaquinone-7 (MK-7) was found to be the predominant isoprenoid quinone. The cell-wall diamino acid was found to be meso-diaminopimelic acid. Polar lipid analysis revealed the presence of phosphatidylglycerol and a glycolipid. The 16S rRNA gene sequence of strain KC1T showed low similarity (<96 %) to other validly named species. The phylogenetic trees showed that strain KC1T is closely related to Bacillus azotoformans DSM 1046T and Bacillus methanolicus DSM 16454T. Both these type strains showed 95.4 % 16S rRNA gene sequence similarity to strain KC1T. The DNA G+C content of strain KC1T was determined to be 39.0 mol%. On the basis of its phenotypic, chemotaxonomic and genotypic characteristics, strain KC1T is considered to represent a novel species of the genus Bacillus, for which the name Bacillus salitolerans sp. nov. is proposed. The type strain is KC1T (=JCM 19760T = CGMCC 1.12810T).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Bacillus, belonging to the family Bacillaceae, phylum Firmicutes, was firstly proposed by Cohn (1872). It is one of the largest bacterial genera, including around 200 validly named species and 7 subspecies (http://www.bacterio.net/bacillus.html). The species of this genus are phenotypically and genotypically heterogeneous (Priest 1993; Slepecky and Hemphill 2006). In general, Bacillus species are obligate aerobes or facultatively anaerobic, spore-forming, rod-shaped, Gram-positive, and have low G+C content (Claus and Berkeley 1986). Most members have MK-7 as the major respiratory quinone; iso-C15:0 and anteiso-C15:0 as the major fatty acids; and meso-diaminopimelic acid as the diagnostic diamino acid in the peptidoglycan. Bacillus species have been isolated from various habitats, including air (Shivaji et al. 2006), ancient tombs (Gatson et al. 2006), brown alga (Ivanova et al. 2004), desert sands (Zhang et al. 2011), forest soils (Chen et al. 2011), freshwater (Baik et al. 2010), ginseng root (Qiu et al. 2009), hot springs (Nazina et al. 2004), marine sediments (You et al. 2013; Zhu et al. 2014), sea water (Yoon et al. 2003), sandy soil (Lee et al. 2008) and a soda lake (Reddy et al. 2015). Many can be found in saline habitats (Amoozegar et al. 2009; Arahal et al. 1999; Lee et al. 2006; Lim et al. 2006; Pappa et al. 2010; Shi et al. 2011; Xue et al. 2008).
During our surveys of bacterial diversity of a salt mine in Kuche county, Akesu area in Xinjiang province of China, a novel isolate named KC1T was obtained. Based on a polyphasic taxonomic approach, we propose that strain KC1T represents a novel species of genus Bacillus.
Materials and methods
Bacterial strains and culture condition
Strain KC1T was isolated from a salt mine sample (salt crystal, obtained at 41°43′0″N, 82°57′33″E) in Xinjiang province, China. Marine broth 2216 (MB, Difco) supplemented with 2.0 % of agar (MA) was used for bacterial isolation. For bacterial enrichment about 3 g of salt sample was suspended in 30 ml MB (28 °C, 2 days) which was further diluted and spread on MA plates. The plates were incubated at 28 °C for 2 days. A cream colony was isolated, purified and identified. The isolate was routinely grown on MA plates at 28 °C and maintained at −80 °C with 25 % (v/v) glycerol.
Biomass for chemotaxonomic and molecular studies was obtained following growth in shake flasks with MB at 28 °C for 2 days. Bacillus azotoformans DSM 1046T, Bacillus methanolicus DSM 16454T and Bacillus subtilis subsp. subtilis DSM 10T (included as the type species of the genus Bacillus) were obtained from the Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH (DMSZ; Germany) and selected for physiological, biochemical and chemotaxonomic characteristics comparison.
Morphological, physiological and biochemical characterisation
Cell morphology and motility were observed with an optical microscope (BX40; Olympus) and by transmission electron microscopy (JEM-1230; JEOL) (Huo et al. 2010). Gram staining was performed by following the method outlined by Dong and Cai (2001). To determine the growth conditions of strain KC1T, 0–15 % (w/v) NaCl concentrations with intervals of 1 % were used. The ranges of pH and temperature for growth were determined according to Zhang et al. (2013).
Catalase and oxidase activities, nitrate and nitrite reduction were tested according to Dong and Cai (2001). Indole, methyl red and Voges-Proskauer tests, H2S production as well as hydrolysis of aesculin, casein, gelatin and starch were performed as described by Zhu (2011). To analyze the use of accessory electron acceptors, sodium thiosulfate (20 mM), sodium sulfate (20 mM), sodium sulfite (5 mM), sodium nitrite (5 mM) and sodium nitrate (20 mM) were respectively added to the sterile MB. Oxygen was removed as described by Grishchenkov et al. (2000). The same medium lacking l-cysteine and resazurin was used as aerobic control. Other enzyme activities, physiological and biochemical characteristics were further determined by using the API ZYM and API 20NE kits, using distilled water supplemented with 4 % (w/v) NaCl to suspend the cells. Acid production tests were determined using API 50CH kits, using modified MB from which yeast extract and peptone were omitted and 0.05 g/L yeast extract was added. Utilisation of single carbon sources for growth was carried out using Biolog GP2 96-well Microplates (Biolog) for the oxidation of 95 organic carbon sources in modified MB. All API tests were performed according to the manufacturer’s instructions (bioMérieux). Antibiotic sensitivity tests were determined on MA plates with antibiotic discs containing the following amounts (μg per disc, unless indicated): amikacin (30), amoxicillin (10), bacitracin (0.04 IU), cefoxitin (30), cefotaxime (30), chloramphenicol (30), ciprofloxacin (5), erythromycin (15), gentamicin (10), kanamycin (30), neomycin (30), nitrofurantoin (300), norfloxacin (10), novobiocin (30), nystatin (100), polymyxin (300 IU), rifampicin (5), sulfamethoxazole (300), tobramycin (10) and vancomycin (30).
Chemotaxonomic characterisation
For fatty acid methyl esters (FAMEs) analysis, late exponential phase cells of strains KC1T, B. azotoformans DSM 1046T, B. methanolicus DSM 16454T and B. subtilis subsp. subtilis DSM 10T were harvested from MB. FAMEs were obtained from freeze-dried cells as described by Kuykendall et al. (1988) and their identification and quantification were performed using the Sherlock Microbial Identification System (MIDI) with the standard MIS Library Generation Software version 4.5 (Microbial ID). Polar lipids of strain KC1T and the three reference strains were extracted and examined by TLC on silica gel 60 F254 aluminium-backed thin-layer plates (10 × 10 cm, Merk 5554) and further analysed using previously described procedures (Minnikin et al. 1984). The solvent system chloroform/methanol/water (65:24:4, by vol.) was used in the first dimension and chloroform/glacial acetic acid/methanol/water (80:15:12:4, by vol.) was used in the second dimension. The TLC plates were sprayed with sulfuric acid/ethanol (1:1, v/v) and heated at 120 °C for 10 min to reveal total lipids. Isoprenoid quinones and the isomer type of the diamino acid in the cell wall peptidoglycan were determined as described by Komagata and Suzuki (1987).
Molecular studies
The 16S rRNA gene was amplified and analysed as described previously (Xu et al. 2007). PCR products were cloned into the vector pMD19-T (TaKaRa) and then sequenced. The sequence was submitted to GenBank to search for similar sequences by using EzTaxon-e server (Kim et al. 2012) and BLAST (http://www.ncbi.nlm.nih.gov/BLAST/). Multiple alignments with sequences of the most closely related bacteria and calculations of levels of sequence similarity were carried out using CLUSTAL W1.8 (Thompson et al. 1994). Phylogenetic analyses were performed using the following tree-making algorithms: the neighbour-joining (Saitou and Nei 1987), the maximum-parsimony (Fitch 1971) and the maximum-likelihood (Felsenstein 1981) methods using the MEGA 5 program package (Tamura et al. 2011). Evolutionary distances were calculated according to the algorithm of Kimura’s two-parameter model (Kimura 1980) for the neighbour-joining method. The DNA G+C content was determined by reversed-phase HPLC as described by Mesbah and Whitman (1989).
Results and discussion
Morphological, physiological and biochemical characteristics
Colonies of strain KC1T on MA plate after 2 days incubation were observed to be 0.5–1.5 mm in diameter, cream, circular and elevated. The cell morphology of strain KC1T is shown in Fig. S1. Cells were observed to be Gram-positive, rod-shaped (0.5–0.9 × 1.7–4.6 μm), endospore-forming and motile with flagella. Catalase and oxidase activities, methyl red test, H2S production and hydrolysis of casein were found to be positive. Indole and Voges-Proskauer tests, hydrolysis of starch and gelatin, nitrate reduction, as well as the production of arginine dihydrolase, lysine decarboxylase, ornithine decarboxylase and urease were found to be negative. The physiological and biochemical characteristics of strain KC1T that differentiate it from the reference strains B. azotoformans DSM 1046T, B. methanolicus DSM 16454T and B. subtilis subsp. subtilis DSM 10T are shown in Table 1. The detailed physiological and biochemical characteristics of strain KC1T are given in the species description. Strain KC1T was found to be resistant to bacitracin, nystatin and polymyxin but sensitive to amikacin, amoxicillin, cefoxitin, cefotaxime, chloramphenicol, ciprofloxacin, erythromycin, gentamicin, kanamycin, neomycin, nitrofurantoin, norfloxacin, novobiocin, rifampicin, sulfamethoxazole, tobramycin and vancomycin.
Chemotaxonomy results
The major fatty acids of strain KC1T were identified as anteiso-C15:0 (56.6 %), anteiso-C17:0 (11.1 %), iso-C15:0 (4.6 %), iso-C17:0 (4.6 %), C18:0 (4.5 %) and C16:0 (4.2 %); the complete fatty acid profiles of strain KC1T and the three reference strains are summarised in Table 2. The major polar lipids were identified as phosphatidylglycerol and a glycolipid, whereas B. methanolicus DSM 16454T and B. subtilis subsp. subtilis DSM 10T contained diphosphatidylglycerol in addition to phosphatidylglycerol and glycolipid. B. azotoformans DSM 1046T contained diphosphatidylglycerol, phosphatidylethanolamine and minor amounts of glycolipid which is different from strain KC1T (Fig. S2). MK-7 was found to be the predominant menaquinone. The cell wall peptidoglycan was found to contain meso-diaminopimelic acid as the diamino acid.
Molecular characterisation
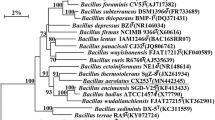
The almost complete 16S rRNA gene sequence (1509 bp) of strain KC1T was obtained (GenBank accession number KF751884). Based on 16S rRNA gene sequence analysis, strain KC1T was found to show high sequence similarities with B. azotoformans DSM 1046T (95.4 %) and B. methanolicus DSM 16454T (95.4 %), followed by B. horikoshii DSM 8719T (95.3 %), B. andreesenii DSM 23947T (95.2 %), B. bataviensis DSM 15601T (95.1 %) and B. drentensis DSM 15600T (95.0 %). Levels of 16S rRNA gene sequence similarity between strain KC1T and other species of the genus Bacillus were found to be below 95 %. Phylogenetic trees revealed that strain KC1T clusters within the genus Bacillus and to be closely related to B. azotoformans DSM 1046T and B. methanolicus DSM 16454T (Fig. 1, Figs. S3 and S4). The genomic G+C content of strain KC1T was determined to be 39.0 mol%.
Taxonomic conclusion
The following observations support the conclusion that strain KC1T belongs to the genus Bacillus: strain KC1T is Gram-positive, motile, rod-shaped and endospore-forming; the major fatty acid is anteiso-C15:0; the major polar lipid is phosphatidylglycerol; the predominant menaquinone is MK-7 and the cell wall peptidoglycan diamino acid is meso-diaminopimelic acid; strain KC1T was phylogenetically clustered within the genus Bacillus and closest to strains B. azotoformans DSM 1046T and B. methanolicus DSM 16454T; the G+C content of the genomic DNA was determined to be 39.0 mol%. However, strain KC1T shows significant different physiological and biochemical characteristics from the type strains of the most closely related species B. azotoformans DSM 1046T and B. methanolicus DSM 16454T, and compared with B. subtilis subsp. subtilis DSM 10T, with regard to morphological features, growth conditions (temperature, pH and NaCl ranges), hydrolysis of casein and gelatin, acid production from carbohydrates, enzyme activity and antibiotic sensitivities (Table 1). Moderate differences in fatty acid composition were also observed between strain KC1T and the three reference species. The amount of anteiso-C15:0 in strain KC1T was notably higher than in the three reference strains and the amount of iso-C15:0 was notably lower in the three reference strains. The detailed fatty acid profiles of the four strains are displayed in Table 2. Differences in the polar lipid profiles were also found among the four strains as shown in Fig. S2. Moreover, the 16S rRNA gene sequence analysis indicated that strain KC1T shared lower than 96 % sequence similarities to other validly named species in genus Bacillus.
Based on the phenotypic, chemotaxonomic and genotypic characteristics described above, strain KC1T can be concluded to represent a novel species within genus Bacillus, for which the name Bacillus salitolerans sp. nov. is proposed.
Description of Bacillus salitolerans sp. nov.
Bacillus salitolerans (sa.li.to’le.rans. L. n. sal, salis salt; L. part. adj. tolerans tolerating; N.L. part. adj. salitolerans salt tolerating).
Cells are Gram-positive, strictly aerobic, motile by means of flagella, rod-shaped (0.5–0.9 × 1.7–4.6 μm) and endospore-forming. Colonies are 0.5–1.5 mm in diameter, cream, circular and elevated on MA plates after 2 days incubation. Growth occurs at 25–45 °C (optimum 37 °C), pH 6.5–9.0 (optimum 8.0) and NaCl 0–10 % (v/v) (optimum 4 %). Growth does not occur under anaerobic conditions with sodium thiosulfate, sodium sulfite, sodium sulfate, sodium nitrite and sodium nitrate as electron acceptors. Catalase and oxidase activities, methyl red test, H2S production and hydrolysis of casein are positive. Indole and Voges-Proskauer tests, nitrate reduction, hydrolysis of starch and gelatin, as well as the production of arginine dihydrolase, lysine decarboxylase, ornithine decarboxylase and urease are negative. Positive in the Biolog GP2 MicroPlate for the oxidation of acetic acid, N-acetylglucosamine, adenosine-5′-monophosphate, l-alaninamide, l-alanine, l-alanyl-glycine, amygdalin, d-araviyol, l-arabinose, arbutin, l-asparagine, 2, 3-butanediol, d-cellobiose, β-cyclodextrin, l-fructose, d-fructose, d-fructose-6-phosphate, d-galactose, gentiobiose, d-glucose, α-d-glucose-1-phosphate, d-glucose-6-phosphate, l-glutamic acid, d-l-α-glycerol phosphate, glycerol, glycogen, α-hydroxybutyric, γ-hydroxybutyric acid, p-hydroxy-phenylacetic, m-inositol, inulin, α-ketoglutaric acid, lactamide, d-lactic acid methyl ester, α-d-lactose, lactulose, d-malic acid, l-malic acid, maltose, mannan, d-mannitol, d-mannose, d-melibiose, β-methyl-d-galactoside, 3-methyl-glucose, palatinose, propionic acid, d-psicose, putrescine, pyruvic acid, l-pyroglutamic acid, d-raffinose, l-rhamnose, sedoheptulosan, d-sorbitol, succinamic acid, succinic acid, succinic acid mono-methyl ester, thymidine, thymidine-5′-monophosphate, trehalose, Tween 80 and uridine. Other substrates in the Biolog GP2 MicroPlate are negative. According to the API 50 CH system, positive for acid production from N-acetyl-β-d-glucosamine, aesculin, l-arabinose, d-fructose, 2-ketogluconate, 5-ketogluconate, maltose, d-mannitol, d-mannose, α-methyl-d-glucopyranoside, sucrose, trehalose and d-xylose; other substrates of the API 50 CH system are not converted to acid. With the API ZYM and API 20 NE systems, acid phosphohydrolase, alkaline phosphatase, cystine arylamidase, esterase (C4), esterase lipase (C8), α-glucosidase, leucine arylamidase, naphthol-AS-BI-phosphohydrolase are positive. Lipase (C14) is weakly positive. N-acetyl-β-glucosaminidase, α-chymotrypsin, α-fucosidase, α-galactosidase, β-galactosidase, β-glucosidase, β-glucuronidase, α-mannosidase, trypsin and valine arylamidase are negative. The major fatty acids are anteiso-C15:0 and anteiso-C17:0. Menaquinone-7 (MK-7) is the predominant isoprenoid quinone. The cell wall peptidoglycan contains meso-diaminopimelic acid as the diamino acid. Major polar lipids are phosphatidylglycerol and a glycolipid. The DNA G+C content of the type strain is 39.0 mol% (HPLC).
The type strain KC1T (=JCM 19760T = CGMCC 1.12810T) was isolated from a salt mine taken from Kuche county, Xinjiang province, China. The GenBank accession number of the 16S rRNA gene sequence of the type strain is KF751884.
References
Amoozegar MA, Sánchez-Porro C, Rohban R, Hajighasemi M, Ventosa A (2009) Bacillus persepolensis sp. nov., a moderately halophilic bacterium from a hypersaline lake. Int J Syst Evol Microbiol 59:2352–2358
Arahal DR, Márquez MC, Volcani BE, Schleifer KH, Ventosa A (1999) Bacillus marismortui sp. nov., a new moderately halophilic species from the Dead Sea. Int J Syst Bacteriol 49(2):521–530
Bae SS, Lee JH, Kim SJ (2005) Bacillus alveayuensis sp. nov., a thermophilic bacterium isolated from deep-sea sediments of the Ayu Trough. Int J Syst Evol Microbiol 55:1211–1215
Baik KS, Lim CH, Park SC, Kim EM, Rhee MS, Seong CN (2010) Bacillus rigui sp. nov., isolated from wetland fresh water. Int J Syst Evol Microbiol 60:2204–2209
Chen YG, Hao DF, Chen QH, Zhang YQ, Liu JB, He JW, Tang SK, Li WJ (2011) Bacillus hunanensis sp. nov., a slightly halophilic bacterium isolated from non-saline forest soil. Antonie Van Leeuwenhoek 99:481–488
Claus D, Berkeley CW (1986) The genus Bacillus. In: Sneath PHA (ed) Bergey’s manual of systematic bacteriology, vol 2. Williams and Wilkins, Baltimore, pp 1105–1139
Cohn F (1872) Untersuchungen über bacterien. Beitr Biol Pflanz 1:127–244 (in German)
Dong XZ, Cai MY (2001) Determination of biochemical properties. In: Dong XZ, Cai MY (eds) Manual for the systematic identification of general bacteria. Science Press, Beijing (in Chinese), pp 370–398
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376
Fitch WM (1971) Toward defining the course of evolution: minimum change for a specific tree topology. Syst Zool 20:406–416
Gatson JW, Benz BF, Chandrasekaran C, Satomi M, Venkateswaran K, Hart ME (2006) Bacillus tequilensis sp. nov., isolated from a 2000-year-old Mexican shaft-tomb, is closely related to B. subtilis. Int J Syst Evol Microbiol 56:1475–1484
Grishchenkov VG, Townsend RT, McDonald TJ, Autenrieth RL, Bonner JS, Boronin AM (2000) Degradation of petroleum hydrocarbons by facultative anaerobic bacteria under aerobic and anaerobic conditions. Process Biochem 35:889–896
Huo YY, Xu XW, Cui HL, Wu M (2010) Gracilibacillus ureilyticus sp. nov., a halotolerant bacterium from a saline-alkaline soil. Int J Syst Evol Microbiol 60:1383–1386
Ivanova EP, Alexeeval YA, Zhukova NV, Gorshkova NM, Buljan V, Nicolau DV, Mikhailov VV, Christen R (2004) Bacillus algicola sp., nov., a novel filamentous organism isolated from brown alga Fucus evanescens. Syst Appl Microbiol 27:301–307
Kim OS, Cho YJ, Lee K, Yoon SH, Kim M, Na H, Park SC, Jeon YS, Lee JH, Yi H, Won S, Chun J (2012) Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Komagata K, Suzuki K (1987) Lipid and cell-wall analysis in bacterial systematics. Method Microbiol 19:161–207
Kuykendall LD, Roy MA, O’Neill JJ, Devine TE (1988) Fatty acids, antibiotic resistance, and deoxyribonucleic acid homology groups of Bradyrhizobium japonicum. Int J Syst Bacteriol 38:358–361
Lee JC, Lim JM, Park DJ, Jeon CO, Li WJ, Kim CJ (2006) Bacillus seohaeanensis sp nov., a halotolerant bacterium that contains l-lysine in its cell wall. Int J Syst Evol Microbiol 56:1893–1898
Lee JC, Lee GS, Park DJ, Kim CJ (2008) Bacillus alkalitelluris sp. nov., an alkaliphilic bacterium isolated from sandy soil. Int J Syst Evol Microbiol 58:2629–2634
Lee GH, Rhee MS, Chang DH, Kwon KK, Bae KS, Yang SH, Kim BC (2014) Bacillus solimangrovi sp. nov., isolated from mangrove soil. Int J Syst Evol Microbiol 64:1622–1628
Lim JM, Jeon CO, Kim CJ (2006) Bacillus taeanensis sp. nov., a halophilic Gram-positive bacterium from a solar saltern in Korea. Int J Syst Evol Microbiol 56:2903–2908
Mesbah M, Whitman WB (1989) Measurement of deoxyguanosine/thymidine ratios in complex mixtures by high-performance liquid chromatography for determination of the mole percentage guanine+cytosine of DNA. J Chromatogr 479:297–306
Minnikin DE, Odonnell AG, Goodfellow M, Alderson G, Athalye M, Schaal A, Parlett JH (1984) An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J Microbiol Meth 2:233–241
Nazina TN, Lebedeva EV, Poltaraus AB, Tourova TP, Grigoryan AA, Sokolova DSH, Lysenko AM, Osipov GA (2004) Geobacillus gargensis sp. nov., a novel thermophile from a hot spring, and the reclassification of Bacillus vulcani as Geobacillus vulcani comb. nov. Int J Syst Evol Microbiol 54:2019–2024
Pappa A, Sánchez-Porro C, Lazoura P, Kallimanis A, Perisynakis A, Ventosa A, Drainas C, Koukkou A (2010) Bacillus halochares sp. nov., a halophilic bacterium isolated from a solar saltern. Int J Syst Evol Microbiol 60:1432–1436
Pham HTV, Kim J (2014) Bacillus thaonhiensis sp. nov., a new species, was isolated from the forest soil of kyonggi university by using a modified culture method. Curr Microbiol 68:88–95
Pichinoty F, Barjac HD, Mandel M, Asselineau J (1983) Description of Bacillus azotoformans sp. nov. Int J Syst Bacteriol 33:660–662
Priest FG (1993) Systematics and ecology of Bacillus. In: Sonenshein AL, Hoch JA, Losick R (eds) Bacillus subtilis and other gram-positive bacteria. American Society for Microbiology, Washington, DC, pp 3–33
Qiu F, Zhang X, Liu L, Sun L, Schumann P, Song W (2009) Bacillus beijingensis sp. nov. and Bacillus ginseng sp. nov., isolated from ginseng root. Int J Syst Evol Microbiol 59:729–734
Reddy SV, Thirumala M, Farooq M, Sasikala C, Ramana CV (2015) Bacillus lonarensis sp. nov., an alkalitolerant bacterium isolated from a soda lake. Arch Microbiol 197(1):27–34
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Shi R, Yin M, Tang SK, Lee JC, Park DJ, Zhang YJ, Kim CJ, Li WJ (2011) Bacillus luteolus sp. nov., a halotolerant bacterium isolated from a salt field. Int J Syst Evol Microbiol 61:1344–1349
Shivaji S, Chaturvedi P, Suresh K, Reddy GSN, Dutt CBS, Wainwright M, Narlikar JV, Bhargava PM (2006) Bacillus aerius sp. nov., Bacillus aerophilus sp. nov., Bacillus stratosphericus sp. nov. and Bacillus altitudinis sp. nov., isolated from cryogenic tubes used for collecting air samples from high altitudes. Int J Syst Evol Microbiol 56:1465–1473
Slepecky R, Hemphill E (2006) The genus Bacillus. Nonmedical. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The Prokaryotes, vol 4. Springer, New York, pp 530–562
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and paximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acid Res 22:4673–4680
Xu XW, Wu YH, Zhou Z, Wang CS, Zhou YG, Zhang HB, Wang Y, Wu M (2007) Halomonas saccharevitans sp. nov., Halomonas arcis sp. nov., and Halomonas subterranea sp. nov., halophilic bacteria isolated from hypersaline environments of China. Int J Syst Evol Microbiol 57:1619–1624
Xue Y, Ventosa A, Wang X, Ren P, Zhou P, Ma Y (2008) Bacillus aidingensis sp. nov., a moderately halophilic bacterium isolated from Ai-Ding salt lake in China. Int J Syst Evol Microbiol 58:2828–2832
Yoon JH, Kim IG, Kang KH, Oh TK, Park YH (2003) Bacillus marisflavi sp. nov. and Bacillus aquimaris sp. nov., isolated from sea water of a tidal flat of the Yellow Sea in Korea. Int J Syst Evol Microbiol 53:1297–1303
You ZQ, Li J, Qin S, Tian XP, Wang FZ, Zhang S, Li WJ (2013) Bacillus abyssalis sp. nov., isolated from a sediment of the South China Sea. Antonie Van Leeuwenhoek 103(5):963–969
Zhang L, Wu GL, Wang Y, Dai J, Fang CX (2011) Bacillus deserti sp. nov., a novel bacterium isolated from the desert of Xinjiang, China. Antonie Van Leeuwenhoek 99:221–229
Zhang WY, Fang MX, Zhang WW, Xiao C, Zhang XQ, Yu ZP, Zhu XF, Wu M (2013) Extensimonas vulgaris gen. nov., sp. nov., a novel member of the family Comamonadaceae. Int J Syst Evol Microbiol 63:2062–2068
Zhu XF (2011) Modern experimental technique of microbiology. Zhejiang University Press (English translation), Hangzhou
Zhu D, Tanabe SH, Xie C, Honda D, Sun J, Ai L (2014) Bacillus ligniniphilus sp. nov., an alkaliphilic and halotolerant bacterium isolated from sediments of the South China Sea. Int J Syst Evol Microbiol 64:1712–1717
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 31170001).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, WY., Hu, J., Zhang, XQ. et al. Bacillus salitolerans sp. nov., a novel bacterium isolated from a salt mine in Xinjiang province, China. Antonie van Leeuwenhoek 108, 443–451 (2015). https://doi.org/10.1007/s10482-015-0497-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-015-0497-0