Abstract
This study aims at estimating the potential of the hydrogen stable isotope (δ2H) analysis of chironomid remains (HC) to reconstruct past changes in the methane (CH4) cycle in lakes. A rearing experiment was first designed to assess the contribution of hydrogen derived from diet to the chironomid biomass and the offset between larvae and their HC. Results from the rearing experiment show that hydrogen stable isotope applied to the chironomid HC seems to be a powerful geochemical tracer because a large proportion (about 85 %) of the total hydrogen in the chironomid biomass is derived from the diet and that the application of a corrected factor to the HC values allows estimating the δ2H of the chironomid larvae. We have then tested the interest of the HC δ2H analysis in addition to other approaches to reconstruct CH4 production and consumption along a sediment core covering the last 1500 years and collected in the deepest part of Lake Remoray (Jura Mountains, France). δ13C and δ2H analyses were performed on both HC and sedimentary organic matter and were combined with an analysis of ancient DNA (aDNA) of methane-oxidizing bacteria (MOB). Results show that MOB is an important part of the total bacteria aDNA all along the studied period. Moreover, δ13C data confirm that MOB may be an important source of carbon for the chironomid biomass (between 20 and 54 %). Finally, δ2H values appear fastidious to obtain in the paleolimnological context; however, this tracer may provide evidence that CO2 reduction is the main dominant process of methanogenesis in Lake Remoray. δ2H analysis could be useful to distinguish the long-term evolution and the relative contribution of the two pathways of methanogenesis. However, δ2H rearing experiments are needed to better estimate the offset and the contribution values.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Freshwater lakes contribute up to 16 % of the global methane (CH4) emissions (Bastviken et al. 2008) and have a non-negligible play in the current global warming (Kirschke et al. 2013). Methanogenesis is a pathway of organic matter degradation under anoxic conditions (Rudd and Hamilton 1978), which caused by Archean bacteria. According to the very high trophic fractionation of methanogens, biogenic CH4 is known to exhibit very low δ13C values (currently from −70 to −50 ‰; Whiticar 1999). Methanogenesis involves two pathways: carbon dioxide reduction and acetate fermentation. According to their metabolic synthesis, biogenic CH4 exhibits also very low δ2H values (from −400 to −250 ‰ for the acetate fermentation and from −250 to −150 ‰ for the carbon dioxide reduction; Whiticar et al. 1986; Whiticar 1999). The environmental conditions allowing the control of the type of substrate used during the methanogenesis are very different. Indeed, the quality of organic matter and the temperature could influence the prevalence of these two processes (Glissman et al. 2004; Mandic-Mulec et al. 2012).
However, up to 75 % of this gas is used as a source of energy and carbon by methane-oxidizing bacteria (MOB; Schubert et al. 2010). MOB has also a high trophic fractionation, leading to very low δ13C values of this organism (from −90 to −60 ‰; Summons et al. 1994; Eller et al. 2007). Very few studies have focused on the hydrogen isotopic values for MOB, but they have suggested that MOB has a much more depleted δ2H signatures than CH4 (e.g., Burke et al. 1988; Liptay et al. 1998). MOB can then be assimilated by metazoan organisms, as a complementary food, supporting then the higher trophic level of the lake food web (Ravinet et al. 2010). Lake sediments are known to be “hot spots” of CH4 production (Bastviken et al. 2004). Some evidences of MOB contribution to the benthic metazoan have been highlighted and particularly for the chironomid larvae (Diptera). In fact, Jones et al. (2008) have shown that up to 80 % of their biomass can derive from MOB assimilation in case of hypoxic hypolimnia. The recognition of the trophic reliance on CH4 has provided a new perspective on lacustrine trophic functioning (e.g., Jones et al. 2008), but it is still poorly studied. The understanding of such mechanism is hampered by the lack of monitoring data (Naeher et al. 2014). Therefore, long-term reconstructions of past CH4 dynamics in aquatic food web are needed.
The use of lake sediments as ecological archives allows providing reliable information about the temporal evolution of CH4 cycle contribution to the benthic food web (van Hardenbroek et al. 2009). Non-biting midges (Chironomidae, Diptera) are ubiquitous organisms whose larvae grow in various environments, including lake sediment. The most chitinous part of their exoskeletons (head capsule, “HC”) is preserved well in surface sediment. The HC chemical composition stays stable for several millennia (Verbruggen et al. 2010). δ13C analysis of these remains is considered to be a reliable descriptor for tracking temporal changes in sources of carbon assimilated by chironomid larvae. Indeed, the δ13C offset between larvae and head capsules is low (Heiri et al. 2012; Frossard et al. 2013), and the trophic fractionation (difference in δ13C values between food and larvae) is assumed to be negligible or very low (Goedkoop et al. 2006; Frossard et al. 2013). Recently, it was demonstrated that δ13C of chironomid remains could be used to highlight incorporation of CH4-derived carbon (van Hardenbroek et al. 2009). In addition, the quantitative analysis of ancient DNA (hereafter “aDNA”) of MOB has demonstrated a strong correlation between an increase in the percent of MOB in the total bacteria present in sediment and a decrease in δ13C values of HC (Belle et al. 2014). This methodology allows collecting reliable information about past MOB dynamics and transfers into benthic food web (by reconstruction of chironomid paleodiet). However, the long-term temporal evolution of the biogenic CH4 synthesis remains poorly understood and needs the calibration of other descriptors such as hydrogen stable isotopes.
Deines et al. (2009) have shown that it could be possible to track the type of substrate used during the methanogenesis by δ2H analysis of consumers. The use of hydrogen stable isotopes as a secondary trophic tracer in paleolimnological study needs the calibration of two assumptions. In fact, hydrogen from a biomass of consumer derives both from diet and water, and it is necessary to decipher the relative contribution of each sources. In addition, in paleolimnological studies, we have to check that the δ2H analysis of the chironomid remains is a relevant proxy for the δ2H value of larvae. Several studies have focused on the quantification of the relative contribution of hydrogen derived from the diet to the chironomid biomass. By δ2H analyses of both water and chironomid larvae in an experiment, previous studies have shown that between 53 and 70 % of hydrogen in chironomid larvae are derived from their diet (Wang et al. 2009; Soto et al. 2013), making hydrogen isotopes a potential excellent trophic tracer. However, these studies used a constant food source and water with very different δ2H signature (labeled water). In this context, it was not possible to assess the relationship between the diet contribution values and the food sources. Complementary rearing experiments are needed to better estimate this contribution. In addition, no study exists concerning the second assumption and this is the first time that the relationship between δ2H of larvae and their chitinous remains was studied.
An experiment was designed (1) to study the offset between the δ2H of larvae and their HC and (2) to assess the contribution of hydrogen derived from different diet sources to the organic matter composition of living chironomid larvae. Based on these results, δ2H and δ13C analyses of subfossil chironomid remains were performed in a sediment core collected in the deepest part of Lake Remoray (Jura Mountains, eastern France) and were combined with MOB aDNA analysis to understand the evolution of CH4 cycle in this lake over the last 1500 years.
Materials and methods
Dietary origin of chironomid δ2H
Rearing experiment conditions
Chironomus riparius egg masses were supplied by an ecotoxicology laboratory (IRSTEA, Lyon) and cultured in a 50 L aquarium with approximately 5 cm of fine silica sand. Larvae were grown at approximately 20 °C with a photoperiod of 16-h light/8-h dark and fed with Tetramin (commercial fish food from Tetra).
Ten egg masses were isolated in a 100-mL beaker until hatching. Then, larvae (n = 100) belonging to the pelagic first instar were introduced into a 250-mL beaker, with 2 cm of fine silica sand and 200 mL of water. Larvae were then fed using three different types of food that were reduced to fine powder, specifically Tetramin (flakes of leftover fish), Oat (terrestrial plant) and Groenlandia densa (freshwater plant) at a rate of 0.4 mg of carbon per larva per day (Doi et al. 2007). Additionally, 150 mL of tap water (with a conductivity of approximately 350 µS cm−1) was replenished daily to prevent the evaporation and increase in ammonium concentration. The study was maintained in the dark to prevent any algae development. Three replicates per treatment were performed. To carry out a potential gradient in the experimental chamber, the position of the nine culture media was randomly distributed. Culture media were maintained at 20 °C using a thermostatic cabinet (Aqua® Lytic).
Sample collection
Three δ2H measurements were performed per each food sources. δ2H analysis was also performed on the larvae, HC and water in each growing medium. Before pupation, four larvae belonging to the fourth instar were collected and introduced into a beaker with distilled water over 24 h to allow gut clearance (Feuchtmayr and Grey 2003). Water samples were also taken at the end of the experiment (between 18 and 23 days) and centrifuged (5 min at 145,000 rotations per minute) to remove any particulate organic matter. Then, water samples were conditioned into a 2-mL collection tube and stored at 4 °C. The substrate of each beaker was sieved through a 200 µm mesh; HC belonging to the fourth instar were picked from the residue. The number of HC collected varied between 40 and 55 to reach the minimal mass of 0.3 mg. Solid samples (larvae, HC and food sources) were then dried at 60 °C for 72 h prior to the isotopic analysis (see “Stable isotope analysis” section below).
Paleolimnological application to Lake Remoray
Study site
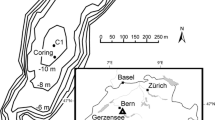
Lake Remoray (46°46′12″ N; 6°15′49″ E) is located in the Jura Mountains in eastern France. The water depth of this moderate size lake (95 ha) reaches 27 m (Fig. 1a). The bedrock of its catchment is composed by carbonate substratum. This lake is dimictic, and its tropholytic zone has been hypoxic at the end of summer and during winter stratification (personal unpublished data), since ca. AD 1970. Borderelle et al. (2008) have shown that Chironomus sp. larvae from a 20 m water depth exhibited low δ13C values (around −47.9 ‰), suggesting a potentially high transfer of CH4-derived carbon through a benthic food web.
A 186-cm-long sediment core was collected from the deepest part of Lake Remoray using a gravity corer (UWITEC, 90 mm of diameter) during the autumn of 2013 (Fig. 1b). Sediment dating was performed using a radionuclide method (210Pb, 137Cs and 241Am) and two additional radiocarbon dates (leaves; Beta Analytic, USA). Age–depth was modeled using a simple linear interpolation (Clam package for R; Blaauw 2010).
Subfossil chironomid and organic matter sampling
Because genus belonging to the Chironomini tribe has a high affinity to the MOB development (van Hardenbroek et al. 2012), carbon isotope analyses of Sergentia coracina-type remains were considered. The first 176.5 cm were cut with a sample thickness of 2, 3 and 4 cm until a depth in the core of 26, 50 and 176.5 cm, respectively (except a sample of 3.5 cm thicknesses between 162 and 165.5 cm). Sediment samples were pre-treated with NaOH (10 %) and HCl (10 %) solutions, following the recommendation of van Hardenbroek et al. (2009). Then, samples were sieved through a 100 µm mesh. S. coracina-type HC belonging to instar 4 were carefully hand-picked under a stereomicroscope. HC were then pooled to reach the minimal mass for δ13C analysis (20 µg for approximately 10 HC, hereafter “δ13CHC”) and introduced into tin capsules. According to the minimal mass required for the analyses and HC concentration, 38 analyses were performed for carbon isotopes.
Sedimentary organic matter was also analyzed on the same samples that received HC analysis (38 samples). Prior to δ13C analysis, the samples were treated to remove any carbonate content, following recommendations by Belle et al. (2014): fumigation for 6 h under vacuum desiccation in a 12 M HCl solution. Approximately 6 mg of pre-treated sediment was introduced into tin capsules for the analysis of sedimentary organic matter δ13C (hereafter “δ13COM”).
The temporal evolutions of δ13CHC and δ13COM allow the definition of clusters with similar trend. Hydrogen stable isotopes were performed in five samples within these homogeneous zones. For HC δ2H analysis, the minimal mass of 0.3 mg (requiring between 150 and 200 subfossil HC) was introduced into silver capsules for (hereafter) “δ2HHC”. The sedimentary organic matter δ2H analysis was performed on the same samples as the HC hydrogen analysis (five samples), and approximately 1 mg of sediment without pre-treatment was introduced into silver capsules (hereafter “δ2HOM”).
aDNA of methanotroph analysis
Methanotroph aDNA was analyzed from 24 samples (1 cm thickness) taken from the Rem13_P1 core. The aDNA extraction was performed on approximately 300 mg of wet sediment using PowerSoil® DNA Isolation Kit, followed by purification (PowerClean® DNA Clean-Up Kit; from MO BIO Laboratories, Inc.). Primer sequences of methanotroph type I and methanotroph type II were the sequences described by Belle et al. (2014), and total bacteria primers sequences were: total bacteria sens: 5′-GATACCCTGGTAGTCCACGC-3′; total bacteria reverse: 5′-GGCCCCCGTCAATTCCTT-3′. Quantitative polymerase chain reaction (qPCR) conditions were performed, following recommendations by Belle et al. (2014). The sum of methanotroph quantities were expressed relative to total bacteria according to Livak and Schmittgen (2001).
Stable isotope analysis
Carbon isotope analyses were conducted with an Isotope Ratio Mass Spectrometer interfaced with an Elemental Analyzer (EA-IRMS) at INRA Nancy (Champenoux). The results are expressed as the delta notation with Vienna Pee Dee Belemnite as the standard: δ13C (‰) = (R sample/R standard − 1) × 1000 (‰), where R = 13C/12C. Replication of sample measurements from standards gave analytical errors (1σ) of ±0.12 ‰ (n = 7) for HC analyses.
For hydrogen isotope analyses, chitin samples were introduced (approximately 1 mg for larvae and sedimentary organic matter and 0.3 mg for HC) into silver capsules. According to Wassenaar and Hobson (2003), the capsules were left open for a period ≥7 days to allow the exchangeable hydrogen in the sample to fully equilibrate with the moisture in the laboratory air. The analytical process also included analysis of standards (BWB-II, whale baleen; and RSPB EGG, egg shell membrane standard with non-exchangeable δ2H values of −108 ± 4 and −99 ‰, respectively), which allowed the correction of the influence of exchangeable hydrogen pollution by laboratory moisture. This correction assumed that the standards and our samples (HC, larvae, sedimentary organic matter, and food sources) should contain the same proportion of exchangeable hydrogen. Analyses were conducted on an EA-IRMS at Iso-Analytical (UK). The results are expressed as the delta notation with Vienna Standard Mean Ocean Water as the standard: δ2H (‰) = (R sample/R standard − 1) × 1000 (‰), where R = 2H/1H. Replication of sample measurements from standards gave analytical errors (1σ) of ±1.16 ‰ (n = 14) for solid analyses and ±1.68 ‰ (n = 8) for water analyses.
Rearing data analysis
The contribution of hydrogen from food to the chironomid larvae biomass was assessed by the use of the Bayesian mixing model proposed by Solomon et al. (2009):
In the equation, δ2Hlarvae, δ2Hwater and δ2Hfood j are the hydrogen isotopic signatures of chironomid larvae, water and food measured for each experimental condition and ϕ food j is the proportion of the food source j in the diet of chironomid larvae. In our rearing experience, ϕ food j is assumed to be 100 % because larvae were fed with a unique food source per experimental condition; The proportion of the H derived from food can be calculated with the proportion of H in chironomid larvae tissues that is derived from water (1 − ω water). Student’s t test was performed to assess the significance of the difference in the contribution of hydrogen derived from the diet to the chironomid biomass.
Paleolimnological data analysis
We quantified the contribution of carbon originated from MOB to the chironomid biomass by the use of a simple two-source isotope-mixing model (Phillips and Gregg 2001) modified to take into account the difference in δ13C values between chironomid larvae and their HC (−0.9 ‰; Frossard et al. 2013).
In this equation, trophic fractionation between food and chironomid larvae is assumed to be negligible (Frossard et al. 2013). The δ13C values of sedimentary organic matter (δ13COM) were assumed to represent those of a mixture of all carbon sources available for chironomid larvae, whereas the δ13C values of MOB (δ13CMOB) were obtained from the literature and ranged from −90 to −60 ‰ (Eller et al. 2007; Jones et al. 2008).
The temporal evolution of paleolimnological data (MOB proportion, δ13CHC, δ13COM, and MOB contribution) was smoothed using a local polynomial regressions (loess parameters: size of the neighborhood = 40 % of the data, degree = 2). All statistical analyses and graphical display were performed using R.2.15.1 statistical software (R Core Team 2012).
Results
Rearing experiment
δ2H values of food sources and water
Tetramin, Oat and G. densa have 42, 48 and 40 % carbon content, respectively (Goedkoop et al. 2006; Doi et al. 2007; this study). The δ2H values of the three food sources significantly differed (Kruskal–Wallis, p < 0.05), with values for Tetramin, Oat and G. densa of −126.6 ± 1.1, −104.2 ± 4.9 and −140.5 ± 4.9 ‰, respectively. The δ2H values of water in the experiment beakers were −58.4 ± 1.2, −56.2 ± 0.4 and −54.7 ± 1 ‰ for Tetramin, Oat and G. densa feeding conditions, respectively (Fig. 2). There was a significant effect of the rearing conditions on the δ2H values of the water (Kruskal–Wallis, p < 0.05).
δ2H offset between larvae and HC
Unfortunately, the very low hydrogen content in a few samples (<10 µg) has prevented the obtention of the δ2H values for 2 HC samples from G. densa feeding experiments and 1 HC sample from Tetramin as food. Nevertheless, all the 6 offsets calculated between larvae and their HC at the fourth instar were very negative and ranged from −32 to −13.5 ‰ (Table 1; Fig. 2). These offsets show that the HC were significantly enriched in 2H compared to the whole larvae.
Contribution of hydrogen from food to the chironomid biomass
The δ2H values of larvae were −115.9 ± 1.6 ‰ from the Tetramin feeding experiment, whereas Tetramin and water exhibit δ2H values of −126.6 ± 1.1, and −58.4 ± 1.2 ‰, respectively. Applying the equation proposed by Solomon et al. (2009), the mean proportion of H in larval tissues originated from Tetramin was 84.2 ± 2.7 % (1 − ω water = (δ2Hlarvae − δ2Hwater)/δ2Hfood − δ2Hwater) × 100, in triplicate. In the experiment with Groenlandia as food, the mean proportion of larval H originated from food was 86.6 ± 5.6 % (Fig. 2). These values were not significantly different (Student’s t test, p > 0.05), and the average value was 85.4 ± 4.1 %. In the Oat experiment, we observed unexpected results. The mean δ2H value of larvae was lower (−120.2 ± 5.6 ‰) than the δ2H values of both water (−56.2 ± 0.4 ‰) and Oat (−104.2 ± 4.9 ‰) (Fig. 2). Consequently, the larvae have mainly fed on an alternative (or complementary) food with a lower δ2H value than Oat.
Application to Lake Remoray
MOB abundance
The sediment core collected in the deepest part of Lake Remoray covered the last 1500 years (Fig. 1c). The MOB aDNA is in relative high proportion, and the average value is around 15 % of the total bacteria aDNA (Fig. 3a). The temporal evolution reveals values ranging from 5 to 34.5 %. There is no statistical difference in the MOB proportion between A and B samples (Student’s t test, p < 0.05).
In situ variability of δ13C and δ2H values of subfossil chironomid
The δ13C values of the chironomid remains (HC) are very negative all along the sediment core (ranging from−43.9 to −38 ‰; Fig. 3b), and much more depleted in 13C than the sedimentary organic matter (ranging from −33.4 to −28.1 ‰; Fig. 3b). Thus, the differences between the δ13COM and δ13CHC values are very high and range from 6.5 to 17 ‰ (Fig. 3b). A simple two-source isotope-mixing model (δ13C of sedimentary organic matter and δ13C of MOB with a value of −90 and −60 ‰) was performed to estimate the contributions of MOB to the chironomid biomass along the core. These estimations showed a temporal trend with values ranging from 10 to 27 % with MOB values of −90 ‰; and from 20 to 54 % with MOB value of −60 ‰ (Fig. 3c).
Only two samples (among five) were analyzed for the hydrogen stable isotope and determined by the HC concentration in the Rem13_P1 core (A from 20 to 57 cm; and B from 90 to 150 cm). The δ2HOM values were −103.2 and −98.2 ± 4.51 ‰, and the δ2HHC values were −107.5 and −107.8 ‰ for samples A and B, respectively. The δ13C values (δ13CHC, δ13COM), and δ2HOM were then averaged for comparison in order to cover the range of the two HC δ2H samples.
It is possible to estimate the isotopic values of larvae using the offsets between larvae and HC (Frossard et al. 2013 for δ13C; for δ2H offset see part “δ2H offset” section below). These offsets are −0.9 ‰ for the δ13C values and ranged from −32 to −15.3 ‰ for δ2H values. According to these results, the evaluation of the larvae isotopic signatures from sample A is −41.1 ± 2.24 ‰ for δ13C and −131.6 ± 6.1 ‰ for δ2H (Table 2). For sample B, the δ13C and δ2H values of larvae are evaluated about −41.8 ± 0.72 and −132.1 ± 6.6 ‰, respectively.
The trophic fractionation between chironomid larvae and their source is considered to be negligible for carbon isotopes (Heiri et al. 2012; Frossard et al. 2013), and the “assimilated mixture” δ13C values are −41.1 ± 2.24 and −41.8 ± 0.72 ‰ for the two samples (Table 2; Fig. 4). The evaluation of the δ2Hassimilated mixture is calculated by the following equation:
Based on the results from this study, (1 − ω water), which is the proportion of hydrogen in chironomid larvae derived from the diet, is approximately 85 % (Fig. 2b). Because lower values of this proportion are reported in the literature (Wang et al. 2009; Soto et al. 2013), the lowest value of 53 % has been also taken into account to include the uncertainty of our results in the calculated δ2H of assimilated mixture. In the equation, the δ2H of water has been assumed to be constant and at about −70 ‰ in Jura lakes (δ2H value of water from Lake Saint-Point hypolimnion; Leroux 2010). The calculated δ2H values for the assimilated mixture ranged between −186.5 ± 12.4 and −142.6 ± 7.8 ‰, and between −187.1 ± 12.4 and −143 ± 7.8 ‰ for the A and B samples, respectively (Table 2).
Biplot representing δ13C and δ2H values of the isotopic baseline of potential organic matter available for the chironomid feeding. CR corresponds to the CH4 isotopic values from CO2 reduction and AF from acetate fermentation (Whiticar et al. 1986; Whiticar 1999). AOM corresponds to aquatic organic matter (Vuorio et al. 2006; Hondula et al. 2013); and TOM to terrestrial organic matter (Meyers and Ishiwatari 1993; Doucett et al. 2007); SOM to sedimentary organic matter (data from this study); AM to assimilated mixture (calculated values from this study) and CL to chironomid larvae (deduced values from HC of this study)
Discussion
Rearing experiment
δ2H offset
The study analyzing the difference between larvae and their HC at the fourth instar revealed a range of δ2H offset between −31.2 and −13.5 ‰ (Fig. 2a). The significant depletion in 2H of larvae compared to their HC could be explained by the difference in chemical composition of these tissues. In fact, the exoskeleton of an arthropod, including chironomid HC, is mainly composed of chitin, a polysaccharide chain. The whole body of larvae corresponds to two biochemical matrices: exoskeleton and soft tissues (including muscle, fatty tissues…). The chemical composition of soft tissue is more complex than that of exoskeleton. The main components of the soft tissues include proteins, lipids, carbohydrates and chitin (approximately 50, 14, 23 and 4 % of dry mass, respectively, Yurkowski and Tabachek 1979). It is widely recognized that lipids exhibit very low δ2H values (Estep and Hoering 1980; DeNiro and Epstein 1978). The higher lipid content in larval tissues may induce the depletion of approximately −25 ‰, when compared to the δ2H values of HC. Several studies have also highlighted the influence of the lipid content on explaining the differences in δ2H values between tissues in the same animal (Hobson et al. 1999; Graham et al. 2014). Moreover, Soto et al. (2013) have shown that lipid extraction before deuterium analysis of chironomid larvae induces an increase in the δ2H values, and they have suggested extracting the lipid before bulk tissue analysis. In this study, the δ2H analysis was performed on bulk tissue for both HC and larvae. It is possible that a change in the lipid content in larvae can induce a change in the offset value. Nevertheless, the correction of the HC δ2H value by an offset, ranging from −31.2 to −13.5 ‰, can be proposed as first values to provide a reliable estimation of the larval δ2H value.
Contribution of the hydrogen from diet to the chironomid larvae
This experiment was designed with three isotopically distinct foods (with a range of −36 ‰) and growing water with a low δ2H variation (<3 ‰). This configuration allows the assessment of the contribution of hydrogen derived from food, which is transferred to the chironomid biomass assuming that this contribution is not food-dependent. In fact, Solomon et al. (2009) have shown that this contribution depends on the trophic level of the consumer which is constant in our rearing experiment. Low δ2H variation for water and highly variable δ2H of food mimic natural conditions were diet sources that can strongly vary over time (Grey et al. 2004) and space (Belle et al. 2015) for each consumer. This contribution reached approximately 85 % (Fig. 2b; 85.4 ± 4.1 %) for two of the three treatments (Tetramin and G. densa). Prior to this study, two estimates were reported for the contribution of diet to the chironomid larvae δ2H values. Wang et al. (2009) found that 69 % of hydrogen in chironomid was derived from the diet, whereas Soto et al. (2013) estimated this contribution at 53 %. Applying to C. riparius larvae, the formula proposed by Solomon et al. (2009) to calculate this contribution from the trophic level of the consumer leads to a contribution of 75 % considering the trophic level equal to 1 (after producers). The range of values is large, and many uncertainties are still unresolved (e.g., influence of the temperature on this proportion, etc.). In addition, other factors could affect the experimental conditions in the growing media (evaporation, algal development, bacterial development, etc.).
The Oat experiment illustrates well the difficulties in maintaining constant the “iso-integrity” of food available for chironomid larvae. Microbial colonization, enhanced by the high mineral element content of Oat, could induce an alteration of the δ2H of food (Goedkoop et al. 2006) or can be preferentially assimilated by larvae (Akerblom and Goedkoop 2003). In this way, biofilm formations were seen in the growing media during the chironomid development. It is impossible on the basis of available data to better understand what happened in the growing media. However, these results could provide evidence of the selective feeding behavior of chironomid larvae.
These uncertainties encouraged us to take into account the lowest and the highest values of contribution (53 and 85 %) to reconstruct the δ2H signature of assimilated mixture by chironomid larvae from the A and B samples in the Remoray’s core.
Past CH4 cycle in Lake Remoray
MOB contribution to the chironomid biomass
qPCR analysis of aDNA allows highlighting the very high contribution of MOB to the total bacteria aDNA extracted from sediment samples. The relative proportion reaches approximately 13 % (Fig. 3a). The living MOB community mainly grows at the sediment/water interface (Rahalkar et al. 2009; Schubert et al. 2011; Ho et al. 2011), and this development is limited by the availability of CH4 (Hershey et al. 2015). As a consequence, results from the present study suggest that both CH4 production and CH4 oxidation rates were very high in Lake Remoray over the studied period. In addition, the availability of the MOB should significantly influence the chironomid paleodiet (Belle et al. 2014).
Carbon isotope analysis provides evidence of the incorporation of a very 13C-depleted organic matter into chironomid biomass because the δ13C value of the assimilated mixture appears much lower than that of sedimentary organic matter (−42 vs. −30 ‰; Table 2). The complementary assimilation of CH4-derived carbon by chironomid larvae is evident (Kohzu et al. 2004; Doi et al. 2006). Moreover, it is widely recognized that MOB can contribute significantly to the chironomid biomass (Jones et al. 2008 and references therein). The results from the mixing model suggest a variable contribution to the chironomid biomass, but these values are always superior to 10 % (from 10 to 27 % with MOB values of −90 ‰; and from 20 to 54 % with MOB value of −60 ‰). Despite the uncertainty about the MOB contribution to the chironomid biomass, the results clearly show that transfer of biogenic CH4 into the benthic food web of Lake Remoray is important all along the studied period (the last 1500 years).
Methanogenesis pathway and δ2H
It was very fastidious and time consuming to reach the minimal mass needed for the hydrogen analyses (three of five samples were below the limit of quantification of 10 µg of hydrogen), and this mass required a large number of HC (more than 150 subfossil HC per sample). The hydrogen isotope analysis could provide complementary information about the methanogenesis pathway in context, where CH4 oxidation and transfer into the food web was provided by other descriptors (MOB aDNA and δ13CHC in this methodology).
As concluded by several previous works, it is possible to distinguish the type of substrate (acetate or carbon dioxide) used during methanogenesis. Indeed, acetate fermentation allows the production of very 2H-depleted CH4 (currently between −400 and −250 ‰), whereas carbon dioxide reduction produces a less 2H-depleted CH4 (currently ranging from −250 to −170 ‰; Whiticar et al. 1986; Whiticar 1999). The trophic fractionation for the hydrogen isotopes is still poorly studied, but it is known that MOB exhibits a higher fractionation for hydrogen than for the carbon (Coleman et al. 1981; Sessions et al. 2002), suggesting that the δ2H values of MOB should be much more negative than CH4 δ2H values. However, Sessions et al. (2002) have also shown that an important part (up to 50 %) of the hydrogen content in lipids of MOB derives from environment water, inducing an opposite effect to the trophic fractionation. These observations make it difficult to reliably estimate the MOB δ2H values.
However, in the present study, the estimated assimilated mixture was moderately 2H depleted compared to the sedimentary organic matter (between −186 and −142 ‰ against −100 ‰, Table 2). The present results from the mixing models applied to δ13C values have shown that this dietary corresponds to a mixture between MOB and sedimentary organic matter (likely a ratio ranging between 10:90 and 54:46; Fig. 3). Nevertheless, the moderately negative signature of the assimilated mixture (between −186 and −142 ‰) may suggest that in Lake Remoray, the dominant methanogenesis pathway may be CO2 reduction, which seems to coincide with what is known about environmental conditions required for this pathway of methanogenesis. In fact, this hydrogenotrophic pathway of CH4 synthesis has also been previously reported from deep stratified lakes similar to Lake Remoray (Lojen et al. 1999; Ogrinc et al. 2008). In addition, the catchment area of Lake Remoray is very large and could induce an important flow of terrestrial organic matter with a low index of degradability (δ13C values of sedimentary organic carbon −28 ‰; Fig. 3c). Hydrogenotrophic pathway seems to correspond to the anoxic degradation of refractory organic matter under low temperatures (Sugimoto and Wada 1993; Mandic-Mulec et al. 2012), whereas acetoclastic methanogenesis corresponds to degradation of labile organic matter.
Conclusion and future challenges
A rearing experiment was conducted to calibrate the use of hydrogen stable isotopes to reconstruct the past CH4 cycle in lake. Results show that the contribution of hydrogen derived from diet to the chironomid biomass is important (around 85 %), making the hydrogen stable isotopes an excellent trophic tracer. This value is the highest in the literature and highlights the complexity of the metabolic processes involved in the chironomid feeding behavior. Moreover, the range of offset between larvae and HC seems to vary from −31.2 to −13.5 ‰. The use of the extreme values in paleolimnological studies should allow the estimation of δ2H value of the chironomid larvae and reflect the uncertainty of this value. More rearing experiments are needed to better estimate these two prerequisites, in particular the use of other food sources (e.g., with MOB culture similar to experiment designed by Deines et al. 2007), and different chironomid species. In addition, complementary studies are needed to assess the potential effect of acid pre-treatment on the HC δ2H values (similar to Perga 2009). In addition, recent advance in mass spectrometry could allow the work with very low mass of hydrogen inducing the decrease in the HC mass required (desirable for a wide use in paleolimnological studies).
The application in the paleolimnological study of Lake Remoray appears fastidious and time consuming. However, the use of the range of offsets and the range of diet contribution allow the estimation of the δ2H values of the assimilated mixture by chironomid larvae and these values are not enough negative to highlight the implication of the acetoclastic pathway. In this context, this new paleotracer may provide evidence that CO2 reduction was the main dominant process of methanogenesis in Lake Remoray over the studied period. In conclusion, hydrogen stable isotope analysis could be useful to distinguish the long-term evolution and the relative contribution of the two pathways of methanogenesis and offers a powerful package to study the past CH4 cycle with combined analyses of δ13C and MOB aDNA.
References
Akerblom N, Goedkoop W (2003) Stable isotopes and fatty acids reveal that Chironomus riparius feeds selectively on added food in standardized toxicity tests. Environ Toxicol Chem 22:1473–1480
Bastviken D, Cole J, Pace M, Tranvik L (2004) Methane emissions from lakes: dependence of lake characteristics, two regional assessments, and a global estimate. Glob Biogeochem Cycles. doi:10.1029/2004GB002238
Bastviken D, Cole JJ, Pace ML, Van de Bogert MC (2008) Fates of methane from different lake habitats: connecting whole-lake budgets and CH4 emissions. J Geophys Res. doi:10.1029/2007JG000608
Belle S, Parent C, Frossard V et al (2014) Temporal changes in the contribution of methane-oxidizing bacteria to the biomass of chironomid larvae determined using stable carbon isotopes and ancient DNA. J Paleolimnol 52:215–228. doi:10.1007/s10933-014-9789-z
Belle S, Millet L, Gillet F et al (2015) Assemblages and paleo-diet variability of subfossil Chironomidae (Diptera) from a deep lake (Lake Grand Maclu, France). Hydrobiologia. doi:10.1007/s10750-015-2222-4
Blaauw M (2010) Methods and code for “classical” age-modelling of radiocarbon sequences. Quat Geochronol 5:512–518. doi:10.1016/j.quageo.2010.01.002
Borderelle A-L, Gilmette C, Lovy C et al (2008) Macroinvertebrate δ13C variability analysis for the assessment of lake trophic functioning. Fundam Appl Limnol 172:289–300. doi:10.1127/1863-9135/2008/0172-0289
Burke RA, Barber TR, Sackett WM (1988) Methane flux and stable hydrogen and carbon isotope composition of sedimentary methane from the Florida Everglades. Glob Biogeochem Cycles 2:329–340. doi:10.1029/GB002i004p00329
Coleman DD, Risatti JB, Schoell M (1981) Fractionation of carbon and hydrogen isotopes by methane-oxidizing bacteria. Geochim Cosmochim Acta 45:1033–1037. doi:10.1016/0016-7037(81)90129-0
Deines P, Grey J, Richnow H-H, Eller G (2007) Linking larval chironomids to methane: seasonal variation of the microbial methane cycle and chironomid δ13C. Aquat Microb Ecol 46:273
Deines P, Wooller MJ, Grey J (2009) Unravelling complexities in benthic food webs using a dual stable isotope (hydrogen and carbon) approach. Freshw Biol 54:2243–2251
DeNiro MJ, Epstein S (1978) Influence of diet on the distribution of carbon isotopes in animals. Geochim Cosmochim Acta 42:495–506. doi:10.1016/0016-7037(78)90199-0
Doi H, Kikuchi E, Takagi S, Shikano S (2006) Selective assimilation by deposit feeders: experimental evidence using stable isotope ratios. Basic Appl Ecol 7:159–166. doi:10.1016/j.baae.2005.04.011
Doi H, Kikuchi E, Takagi S, Shikano S (2007) Changes in carbon and nitrogen stable isotopes of chironomid larvae during growth, starvation and metamorphosis. Rapid Commun Mass Spectrom 21:997–1002. doi:10.1002/rcm.2925
Doucett RR, Marks JC, Blinn DW et al (2007) Measuring terrestrial subsidies to aquatic food webs using stable isotopes of hydrogen. Ecology 88:1587–1592
Eller G, Deines P, Krüger M (2007) Possible sources of methane-derived carbon for chironomid larvae. Aquat Microb Ecol 46:283–293
Estep MF, Hoering TC (1980) Biogeochemistry of the stable hydrogen isotopes. Geochim Cosmochim Acta 44:1197–1206. doi:10.1016/0016-7037(80)90073-3
Feuchtmayr H, Grey J (2003) Effect of preparation and preservation procedures on carbon and nitrogen stable isotope determinations from zooplankton. Rapid Commun Mass Spectrom 17:2605–2610. doi:10.1002/rcm.1227
Frossard V, Belle S, Verneaux V et al (2013) A study of the δ13C offset between chironomid larvae and their exuvial head capsules: implications for palaeoecology. J Paleolimnol 50:379–386. doi:10.1007/s10933-013-9732-8
Glissman K, Chin K-J, Casper P, Conrad R (2004) Methanogenic pathway and archaeal community structure in the sediment of eutrophic lake dagow: effect of temperature. Microb Ecol 48:389–399. doi:10.1007/s00248-003-2027-2
Goedkoop W, Akerblom N, Demandt MH (2006) Trophic fractionation of carbon and nitrogen stable isotopes in Chironomus riparius reared on food of aquatic and terrestrial origin. Freshw Biol 51:878–886. doi:10.1111/j.1365-2427.2006.01539.x
Graham CT, Harrison SSC, Harrod C (2014) Differences in the contributions of dietary water to the hydrogen stable isotope ratios of cultured Atlantic salmon and Arctic charr tissues. Hydrobiologia 721:45–55. doi:10.1007/s10750-013-1636-0
Grey J, Kelly A, Ward S et al (2004) Seasonal changes in the stable isotope values of lake-dwelling chironomid larvae in relation to feeding and life cycle variability. Freshw Biol 49:681–689. doi:10.1111/j.1365-2427.2004.01217.x
Heiri O, Schilder J, Van Hardenbroek M (2012) Stable isotopic analysis of fossil chironomids as an approach to environmental reconstruction: state of development and future challenges. Fauna Nor. doi:10.5324/fn.v31i0.1436
Hershey AE, Northington RM, Hart-Smith J et al (2015) Methane efflux and oxidation, and use of methane-derived carbon by larval Chironomini, in arctic lake sediments. Limnol Oceanogr 60:276–285. doi:10.1002/lno.10023
Ho A, Lüke C, Cao Z, Frenzel P (2011) Ageing well: methane oxidation and methane oxidizing bacteria along a chronosequence of 2000 years. Environ Microbiol Rep 3:738–743. doi:10.1111/j.1758-2229.2011.00292.x
Hobson KA, Atwell L, Wassenaar LI (1999) Influence of drinking water and diet on the stable-hydrogen isotope ratios of animal tissues. Proc Natl Acad Sci USA 96:8003–8006
Hondula KL, Pace ML, Cole JJ, Batt RD (2013) Hydrogen isotope discrimination in aquatic primary producers: implications for aquatic food web studies. Aquat Sci 76:217–229. doi:10.1007/s00027-013-0331-6
Jones RI, Carter CE, Kelly A et al (2008) Widespread contribution of methane-cycle bacteria to the diets of lake profundal chironomid larvae. Ecology 89:857–864
Kirschke S, Bousquet P, Ciais P et al (2013) Three decades of global methane sources and sinks. Nat Geosci 6:813–823. doi:10.1038/ngeo1955
Kohzu A, Kato C, Iwata T et al (2004) Stream food web fueled by methane-derived carbon. Aquat Microb Ecol 36:189–194. doi:10.3354/ame036189
Leroux A (2010) Caractérisation et évolution des flux détritiques et authigènes en contexte lacustre carbonaté au cours du Tardiglaciaire et de l’Holocène (Lac Saint-Point, Haute-Chaîne du Jura): implications paléoclimatiques et paléoenvironnementales. Phdthesis, Université de Franche-Comté
Liptay K, Chanton J, Czepiel P, Mosher B (1998) Use of stable isotopes to determine methane oxidation in landfill cover soils. J Geophys Res 103:8243–8250. doi:10.1029/97JD02630
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Lojen S, Ogrinc N, Dolenec T (1999) Decomposition of sedimentary organic matter and methane formation in the recent sediment of Lake Bled (Slovenia). Chem Geol 159:223–240. doi:10.1016/S0009-2541(99)00032-7
Mandic-Mulec I, Gorenc K, Petrišic MG et al (2012) Methanogenesis pathways in a stratified eutrophic alpine lake (Lake Bled, Slovenia). Limnol Oceanogr 57:868–880. doi:10.4319/lo.2012.57.3.0868
Meyers PA, Ishiwatari R (1993) Lacustrine organic geochemistry—an overview of indicators of organic matter sources and diagenesis in lake sediments. Org Geochem 20:867–900. doi:10.1016/0146-6380(93)90100-P
Naeher S, Niemann H, Peterse F et al (2014) Tracing the methane cycle with lipid biomarkers in Lake Rotsee (Switzerland). Org Geochem 66:174–181. doi:10.1016/j.orggeochem.2013.11.002
Ogrinc N, Žagar M, Faganeli J et al (2008) Methane Formation in a Remote Mountain Lake (Lake Planina, NW Slovenia). Geomicrobiol J 25:346–356. doi:10.1080/01490450802402786
Perga M-E (2009) Potential of δ13C and δ15N of cladoceran subfossil exoskeletons for paleo-ecological studies. J Paleolimnol 44:387–395. doi:10.1007/s10933-009-9340-9
Phillips DL, Gregg JW (2001) Uncertainty in source partitioning using stable isotopes. Oecologia 127:171–179. doi:10.1007/s004420000578
R Core Team (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Rahalkar M, Deutzmann J, Schink B, Bussmann I (2009) Abundance and activity of methanotrophic bacteria in littoral and profundal sediments of Lake Constance (Germany). Appl Environ Microbiol 75:119–126. doi:10.1128/AEM.01350-08
Ravinet M, Syväranta J, Jones RI, Grey J (2010) A trophic pathway from biogenic methane supports fish biomass in a temperate lake ecosystem. Oikos 119:409–416. doi:10.1111/j.1600-0706.2009.17859.x
Rudd JW, Hamilton RD (1978) Methane cycling in a eutrophic shield lake and its effects on whole lake metabolism. Limnol Oceanogr 23:337–348
Schubert CJ, Lucas F, Durisch-Kaiser E et al (2010) Oxidation and emission of methane in a monomictic lake (Rotsee, Switzerland). Aquat Sci 72:455–466
Schubert CJ, Vazquez F, Lösekann-Behrens T et al (2011) Evidence for anaerobic oxidation of methane in sediments of a freshwater system (Lago di Cadagno). FEMS Microbiol Ecol 76:26–38. doi:10.1111/j.1574-6941.2010.01036.x
Sessions AL, Jahnke LL, Schimmelmann A, Hayes JM (2002) Hydrogen isotope fractionation in lipids of the methane-oxidizing bacterium Methylococcus capsulatus. Geochim Cosmochim Acta 66:3955–3969. doi:10.1016/S0016-7037(02)00981-X
Solomon CT, Cole JJ, Doucett RR et al (2009) The influence of environmental water on the hydrogen stable isotope ratio in aquatic consumers. Oecologia 161:313–324. doi:10.1007/s00442-009-1370-5
Soto DX, Wassenaar LI, Hobson KA (2013) Stable hydrogen and oxygen isotopes in aquatic food webs are tracers of diet and provenance. Funct Ecol 27:535–543. doi:10.1111/1365-2435.12054
Sugimoto A, Wada E (1993) Carbon isotopic composition of bacterial methane in a soil incubation experiment: contributions of acetate and CO2H2. Geochim Cosmochim Acta 57:4015–4027. doi:10.1016/0016-7037(93)90350-6
Summons RE, Jahnke LL, Roksandic Z (1994) Carbon isotopic fractionation in lipids from methanotrophic bacteria: relevance for interpretation of the geochemical record of biomarkers. Geochim et Cosmochim Acta 58:2853–2863. doi:10.1016/0016-7037(94)90119-8
van Hardenbroek M, Heiri O, Grey J et al (2009) Fossil chironomid δ13C as a proxy for past methanogenic contribution to benthic food webs in lakes? J Paleolimnol 43:235–245. doi:10.1007/s10933-009-9328-5
van Hardenbroek M, Lotter AF, Bastviken D et al (2012) Relationship between δ13C of chironomid remains and methane flux in Swedish lakes. Freshw Biol 57:166–177. doi:10.1111/j.1365-2427.2011.02710.x
Verbruggen F, Heiri O, Reichart G-J, Lotter AF (2010) Chironomid δ18O as a proxy for past lake water delta O-18: a Late glacial record from Rotsee (Switzerland). Quat Sci Rev 29:2271–2279. doi:10.1016/j.quascirev.2010.05.030
Vuorio K, Meili M, Sarvala J (2006) Taxon-specific variation in the stable isotopic signatures (δ13C and δ15N) of lake phytoplankton. Freshw Biol 51:807–822. doi:10.1111/j.1365-2427.2006.01529.x
Wang YV, O’Brien DM, Jenson J et al (2009) The influence of diet and water on the stable oxygen and hydrogen isotope composition of Chironomidae (Diptera) with paleoecological implications. Oecologia 160:225–233. doi:10.1007/s00442-009-1303-3
Wassenaar LI, Hobson KA (2003) Comparative equilibration and online technique for determination of non-exchangeable hydrogen of keratins for use in animal migration studies. Isot Environ Health Stud 39:211–217. doi:10.1080/1025601031000096781
Whiticar MJ (1999) Carbon and hydrogen isotope systematics of bacterial formation and oxidation of methane. Chem Geol 161:291–314
Whiticar MJ, Faber E, Schoell M (1986) Biogenic methane formation in marine and freshwater environments: CO2 reduction vs. acetate fermentation—isotope evidence. Geochim Cosmochim Acta 50:693–709. doi:10.1016/0016-7037(86)90346-7
Yurkowski M, Tabachek JL (1979) Proximate and amino acid composition of some natural fisch foods. In: Halver JE, Tiews K (eds) Finfish nutrition and fishfeed technology. Heenemann GmbH & Co, Berlin, pp 435–448
Acknowledgments
This study is a part of “Zone Atelier Arc Jurassien”. Financial support for this study was provided by Conseil Regional de Franche-Comté and OREAS project. We thank Christian Hossann and Claude Bréchet (INRA Nancy, Champenoux) for assistance in the stable isotope analysis of carbon, and Ian Begley and Steve Brooks (Iso-Analytical, UK) for assistance with the deuterium analysis. We also address our gratitude to Laurie Murgia and Vincent Bichet (Chrono-Environnement, Besançon) for technical assistance during the coring survey.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Piet Spaak.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix 1
δ2H values of chironomid feeding experiment (DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Belle, S., Verneaux, V., Millet, L. et al. A case study of the past CH4 cycle in lakes by the combined use of dual isotopes (carbon and hydrogen) and ancient DNA of methane-oxidizing bacteria: rearing experiment and application to Lake Remoray (eastern France). Aquat Ecol 49, 279–291 (2015). https://doi.org/10.1007/s10452-015-9523-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-015-9523-6