Abstract
There is a well-known relationship between porous materials performance in a given process and their textural properties. These properties include specific surface area, among others, where the most widely used experimental technique to determine them is gas adsorption. Although the most used adsorptive gas is N2 at 77 K up to atmospheric pressure, its quadrupole moment generates specific interactions with surface groups, as silanols in silica materials, causing a preferential orientation effect on the adsorbed N2 molecule affecting the specific surface area value. In this sense, we analyzed the adsorption–desorption isotherms at 77 K of nanoporous silica materials using different adsorptives. From these data, we obtained the specific surface area (SBET) values of the samples by applying the BET method with the IUPAC recommendations for each gas. The selected materials were MCM-41, MCM-48, SBA-15, and SBA-16, and the adsorptives used were Ar, O2, and CH4, along with N2. Among the chosen adsorptives, Ar and CH4 do not have a quadrupole moment, whereas this value is present for N2 and O2, being the latter four times smaller than nitrogen. In addition, at 77 K, both Ar and CH4 are below their triple-point temperature, while N2 and O2, which are above their triple-point temperature, are in the same thermodynamic state. Taking the SBET obtained by Ar at 77 K as the referential value of each sample, the corresponding molecular transversal areas of the other adsorptives were estimated. It was found that the variation of transversal area for the N2 molecule at 77 K on silica materials was between 0.133 and 0.149 nm2 (below its common value of 0.162 nm2). In contrast, in the case of the O2 molecule at 77 K, this value was almost constant, with an average of 0.123 nm2. These results showed that the quadrupole moment of the O2 does not play an important role in the interaction with surface silanol groups present in the samples, making oxygen at 77 K a potential and reliable adsorptive to determine the specific surface area of silica materials.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Several industrial processes (e.g., obtention of chemical products, transformation, and elimination of pollutants) involve catalysis. Other processes with environmental and energy interest require gas adsorption (e.g., capture, storage, and separation of gases). In both applications, catalysts and adsorbents play an essential role. Their performance can be explained partly due to their textural properties. Among these materials, those with mesoporous structure (2–50 nm) allow a better diffusion of many molecules of industrial, environmental, and energetic interest [1]. Particularly, ordered mesoporous materials (OMM) have been vastly used in the applications mentioned above due to their high specific surface area and porosity [2], along with the possibility of functionalizing the surface with several functional groups for a given application [3]. Those materials are considered as model adsorbents because of the symmetry presented at the mesoscopic level, making them suitable to match experimental data with theoretical models, in particular for textural characterization (e.g., measurement of the specific surface area and pore size distribution) [4].
The relevance of the textural properties, referred to the specific surface area and porosity, of porous materials in determined application has been demonstrated and studied by several researchers, in applications, such as the adsorption of: gases [5,6,7,8,9,10,11,12,13,14], vapors [15,16,17,18,19], organic and pharmaceutical compounds [20,21,22,23,24,25,26,27], dyes [28,29,30], proteins [31,32,33], heavy metals [34,35,36], and diverse catalytic applications as well [37,38,39,40,41,42,43,44,45]. The performance of the porous materials in the applications mentioned above are mainly related with the apparent surface area, which according to the IUPAC [46] is a “fingerprint” of the adsorbents.
N2 adsorption–desorption at 77 K has been considered the standard technique to assess the textural properties of porous materials, mainly for its availability as probe gas and cryogenic liquid [46]. Nonetheless, several researchers [47,48,49,50,51] and even the IUPAC, advice that the permanent quadrupole moment of N2 molecule presents specific interactions with surface groups, e.g., the silanol surface groups present in the silica materials, causing an effect of preferential orientation in the adsorbed N2 molecules. For this case, the IUPAC recommends the use of Ar at 87 K, due to its null quadruple moment. On the other hand, the use of adsorptives different than N2 at different temperatures in adsorption–desorption studies, can be useful to obtain complementary information regarding the texture and/or porous structure of the materials, for instance, Table S1 (supplementary information) shows reports regarding the adsorption of different gases apart from N2 using silica OMM samples, where some of them were used to characterize these materials. From this table we can observe that all of the gases can be studied at 77 K, which is interesting because of the accesibility of liquid N2 as cryogenic system. This table also shows that there are few reports with O2, CH4 and CO, although the use of latter is not recommended for rutinary characterization due to its high toxicity. In particular, the older reports showed only the O2 and CH4 adsorption isotherm, without obtaining textural information from the data obtained with these adsorptives [52,53,54,55,56,57],whereas the most recent report [58] only examines the relationship between the pore imperfections and the nature of the adsorption/desorption branch by measuring adsorption/desorption subloops and the temperature dependence of hysteresis loops. Finally, it is noteworthy to stress that Ar at 77 K is limited to analyze materials with pore diameters below 15 nm [59], which might be assumed also to CH4 because it is at the same thermodynamic state than Ar at 77 K.
In this work, we used different adsorptives such as N2, Ar, O2, and CH4, at 77 K, with different quadrupolar moments and thermodynamic properties to obtain the adsorption–desorption isotherms. From there, we used the corresponding data to estimate the specific surface area of silica OMM, such as SBA-15, SBA-16, MCM-41, and MCM-48 by the BET method, analyzing the results to understand the effect of each gas in the calculation of the SBET.
2 Materials and methods
2.1 OMM synthesis
The reagents used in the synthesis of the different OMM were: cetyltrimethylammonium bromide (CTAB, C16H33N(CH3)3Br, Merck), Pluronic P123® (EO20PO70EO20, Sigma Aldrich), and Pluronic® F127 (EO106PO70EO106, Sigma Aldrich) as surfactants; Tetraethyl orthosilicate (TEOS, SiC8H20O4, Merck) as silica source; water and ethanol (EtOH, CH3CH2OH) as solvents; sodium hydroxide (NaOH, Cicarelli); hydrochloric acid (HCl, Biopack) and ammonium hydroxide (NH4OH, Biopack) as catalysts; and n-butanol (n-BuOH, C4H9OH) as a structure-directing agent.
The different surfactant:silica molar ratios used for the synthesis of the samples were chosen based on the most typically reported ratios for each OMM (0.12, 0.3, 0.017, and 0.0025 for MCM-41, MCM-48, SBA-15, and SBA-16, respectively). From these values, ratios above and below were used to obtain a representative number of samples with similar nature but different textural properties. The obtained materials were characterized by TEM and XRD, and the results are presented in the Supplementary Information.
2.1.1 MCM-41
The synthesis of these OMM was performed at room temperature using non-hydrothermal conditions, based on the procedure reported by Barrera et al. [60]. The reagents used were: CTAB, TEOS, NaOH, and water. The molar ratio used in this synthesis was: xCTAB:1TEOS:0.6NaOH:100H2O, where x corresponds to the molar ratio CTAB/TEOS (surfactant:silica).
In a typical synthesis, CTAB is dissolved in water, then 1 mol L−1 NaOH is added to the mixture, keeping a vigorous stirring until complete dissolution of the CTAB (ca. 2 h). Afterward, TEOS is added dropwise to the solution under constant stirring. Then, the resulting solution is kept under stirring for 24 h at room temperature. The solids produced are filtered and washed with abundant distilled water until a conductivity < 10 μS cm−1, dried at 60 °C for 12 h and calcined at 550 °C for six hours using a heating rate of 1 °C min−1. In order to study the influence of the surfactant:silica ratio, a series of MCM-41 materials were synthesized, varying the CTAB/TEOS molar ratio as follows: 0.07, 0.12, and 0.17. The samples were named based on these ratios, i.e., MCM-41_A, MCM-41_B, and MCM-41_C, respectively.
2.1.2 MCM-48
The synthesis of MCM-48 was carried out at room temperature under non-hydrothermal conditions, based on the procedure reported by Schumacher et al. [61]. The reagents used were: CTAB, TEOS, NH4OH, water, and EtOH. The molar ratio used in this synthesis was: xCTAB:1TEOS:12.5 NH4OH:54EtOH:417H2O, where x corresponds to the CTAB/TEOS molar ratio.
A typical synthesis can be described as follows: CTAB is dissolved in water and EtOH, then an NH3 aqueous solution (25% w/w) is added to the mixture. Vigorous stirring was kept until complete dissolution of the CTAB, producing a transparent solution. Afterward, the required quantity of TEOS was added to the solution; immediately, this solution is stirred at 1000 rpm for 10 min. The resulting cloudy solution is kept unstirred for 24 h at room temperature. The solids produced are filtered and washed with abundant distilled water until a conductivity below 10 μS cm−1. The solids are dried at 60 °C for 12 h and calcined at 550 °C for 6 h using a heating rate of 1 °C min−1. For this material, the CTAB/TEOS molar ratio was varied as follows: 0.2, 0.3, and 0.4. The samples were named as follows: MCM-48_A, MCM-48_B, and MCM-48_C, respectively.
2.1.3 SBA-15
This OMM was synthesized under non-hydrothermal conditions, as reported by Barrera et al. [60]. The reagents used were: P123, TEOS, HCl, and H2O. The molar ratio used in this synthesis was: xP123:1TEOS:6HCl:140H2O, where x corresponds to the molar ratio P123/TEOS.
The synthesis consists of the dissolution of P123 in an HCl 2 mol L−1 (pH ca. 1) aqueous solution, under vigorous stirring at 40 °C for 2 h or complete dissolution of the surfactant. Then, TEOS is added dropwise, and the stirring is kept for 4 h at the same temperature. Afterward, this solution is aged at 40 °C, without stirring for 20 h, then the solution is heated at 80 °C and kept for 48 h. The solids produced are filtered and washed with abundant distilled water until a conductivity below 10 μS cm−1. The samples are dried at 60 °C for 12 h and calcined at 550 °C for 6 h using a heating rate of 1 °C min−1. In this case, P123/TEOS molar ratio was varied as follows: 0.013, 0.017, and 0.021. The samples were named SBA-15_A, SBA-15_B, and SBA-15_C, respectively.
2.1.4 SBA-16
SBA-16 was synthesized under non-hydrothermal conditions, based on the procedure reported by Stevens et al. [62]. The reagents utilized were F127, TEOS, HCl, water, and n-BuOH. The molar ratios used in this synthesis were: xF127:1TEOS:0.88HCl:1.69n-BuOH:110H2O, where x corresponds to the molar ratio F127/TEOS.
A typical synthesis can be described as follows: F127 is suspended in HCl (concentrated) and n-BuOH and vigorously stirred at 45 °C until the complete dissolution of F127. Then, TEOS is added dropwise at the same stirring and temperature, and the resulting cloudy solution is stirred for 24 h at 45 °C. Later, the aging stage is carried out at 100 °C under reflux. The solids produced are filtered and washed with abundant distilled water until a conductivity below 10 μS cm−1. The solids are then dried at 60 °C for 12 h and calcined at 550 °C for 6 h using a heating rate of 1 °C min−1. In this case, F127/TEOS molar ratio was varied as follows: 0.0017, 0.0025, and 0.0033. The samples were named SBA-16_A, SBA-16_B, and SBA-16_C, respectively.
2.2 Gas adsorption measurements
N2 (99.999%), Ar (99.999%), O2 (99.8%) and CH4 (99.995%) adsorption–desorption isotherms at 77 K were measured in a manometric adsorption equipment ASAP 2000 from Micromeritics. Before each analysis, the samples were outgassed at 150 °C for 12 h. The experiments were performed at the temperature of liquid nitrogen, which was 76.8 K on average during the experiments. Under this condition, the experimental average saturation pressures of the different adsorptives, po, were 712, 181, 142, and 8.8 Torr, for N2, Ar, O2, and CH4, respectively. In each case, the saturation pressure was continuously measured during the experiment, taking special care to the thermodynamic state of the adsorptives (see Table 1). Considering that the measured range in term of the absolute pressure for Ar, O2 and CH4 adsorption at 77 K is lower than 200 Torr is necessary to have pressure transducers of 1 Torr, 10 Torr, and 1000 Torr in order to obtain more reliable and accurate data. In addition, N2 and Ar adsorption–desorption isotherms at 77.4 and 87.3 K, respectively, were measured for SBA-15_B in Autosorb iQ analyzer from Quantachrome Instruments.
2.3 Thermodynamic conditions and properties of the adsorptives
The thermodynamic states of the selected adsorptives are displayed in Table 1.
Under experimental conditions (77 K), the adsorptives are in the thermodynamic state of subcritical gases (T/Tc < 1). Additionally, Ar and CH4 are at a temperature below their triple point, behaving as a subcooled liquid (T/TTP < 1). Besides performing the adsorption experiments with adsorptives presenting different thermodynamic conditions, the selection of the adsorptives was based on their molecular dimensions as well as their quadrupole moments (Table 1) to study the effect of these parameters on the specific surface area of the silica OMM. As is shown in Table 1, Ar and CH4 have a quadrupole moment of zero, whereas N2 presents a quadrupole moment four times larger than O2.
2.4 Specific surface area
The specific surface area of the silica OMM materials, SBET, was determined using the standard BET method (Eq. 1) [66] and taking into account the criteria suggested by the IUPAC [46].
where nads (in mmol g−1) is the amount adsorbed at a relative pressure p/po, nm (in mmol g−1) is the monolayer capacity, and C is the adsorbate–adsorbent interaction constant.
For the evaluation of SBET, it is necessary to know the value of the transversal area of the adsorbed molecule on the surface (σ). This value can be obtained (in nm2) using Eq. (2) proposed by Emmet and Brunauer [67].
where f is the packing factor of adsorbed molecules, M is the molecular weight (in g mol−1) of the adsorptive, ρ (in g cm−3) is the adsorptive density in the liquid state, and NA is Avogadro’s constant (in mol−1). Assuming that the adsorbed molecules adopt a hexagonal close-packed structure, the packing factor takes a value of 1.091 [51]. Table 2 presents the customary σ values of the adsorptives to determine the specific surface area (SBET) of the silica OMM materials studied.
3 Results and discussion
3.1 Isotherm analysis with different adsorptives
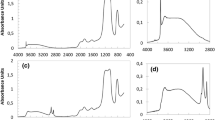
Figure 1 shows the experimental N2, Ar, O2, and CH4 adsorption–desorption isotherms at 77 K of the studied MCM-41 materials, which are Type IV according to the IUPAC classification [46], typical of this type of materials. It can be seen that after the capillary condensation in the primary mesopores, there is an almost horizontal plateau, suggesting no significant presence of secondary mesopores (mesoporosity related to disorder in the OMM).
Another worth-mentioning aspect is that with all gases, except N2, the materials present a slight hysteresis loop (insets in Figs. 1b, c, and d). The reversibility of the N2 isotherm in the region of capillary condensation/evaporation is because the MCM-41 primary mesopore size is below the nitrogen capillary critical pore size (ca. 4–5 nm), and this phenomenon occurs at a relative pressure below the critical value for nitrogen (i.e., 0.42) [56, 69, 70]. The Ar and O2 isotherms (Fig. 1b and c) exhibit a small hysteresis loop, closing at a critical relative pressure of ca. 0.25 and 0.2 for Ar and O2, respectively. This fact is because the primary mesopore size of the samples is larger than the critical pore size of these adsorptives (ca. 2.5–3.3 nm) [54, 56, 71]. Regarding CH4 isotherms (Fig. 1d), their behavior is similar to that of Ar and O2, i.e., they present a small hysteresis loop, closing at a critical relative pressure of 0.12, which is lower than the other adsorptives, and it could be related to its low saturation pressure. From this result, it can be inferred that CH4 critical pore size must be similar to those values of Ar and O2 (i.e., between 2.5 and 4 nm) or even less than 2.5 nm, to our knowledge, there are no reported values of methane critical pore size. In general, the beginning of the capillary condensation is close to the mono-multilayer adsorption region,particularly for CH4, the capillary condensation is overlapped with the mono-multilayer adsorption likely related to CH4 low saturation pressure (8.8 Torr).
Figure 2 displays the N2, Ar, O2, and CH4 adsorption–desorption isotherms at 77 K of MCM-48 materials. The isotherms are Type IV, similar to those obtained for MCM-41. Here, a well-defined stage of capillary condensation is also observed, and the plateau after the ending of this stage indicates a low amount of secondary mesopores.
The behavior of the hysteresis loops of the isotherms is similar to that in the MCM-41, where the N2 isotherms are reversible in the capillary condensation/evaporation stage, and the presence of small hysteresis loops for the other gases (insets in Fig. 2b, c and d). These results support the hypothesis that the critical capillary size of CH4 at 77 K is between 2.5 and 4 nm.
Figure 3 shows the N2, Ar, O2, and CH4 adsorption–desorption isotherms at 77 K for the SBA-15. This type of materials also present a Type IV(a) isotherms. These isotherms exhibit a Type H1 hysteresis loop with well-defined stages of capillary condensation/evaporation, which are characteristic of materials with uniform mesopore sizes and a high degree of ordering [46]. N2 and O2 adsorption isotherms show an increase in the amount adsorbed at high relative pressures, indicating the presence of larger mesopores (ca. 40 nm), which cannot be seen by Ar at 77 K given that this adsorptive only can provide information of pore sizes below 15 nm, as reported previously [71, 72]. Considering that CH4 and Ar at 77 K have the same thermodynamic state, i.e., both are below their triple point, it is probable that CH4 can also measure pores sizes up to the same magnitude order (ca. 15 nm).
Figure 4 displays the experimental N2, Ar, O2, and CH4 adsorption–desorption isotherms at 77 K of SBA-16 materials. These materials also present Type IV(a) isotherms, where SBA-16_B and SBA-16_C samples show an important ordering degree of the mesoporous structure (according to the isotherm shape). Whereas the SBA-16_A sample exhibits isotherms with a less pronounced capillary condensation stage, indicating that this material presents a wider mesopore size distribution.
With all the adsorptives evaluated, the isotherms of SBA-16_B and SBA-16_C samples present Type H2(a) hysteresis loops, characteristic of materials with an important network effect in their pore structure, and where the desorption branch can be affected by cavitation phenomenon [73]. In the SBA-16_A sample, the N2 isotherm exhibits a Type H2(b) hysteresis loop. Whereas with the other adsorptives, this sample presents a Type H5 hysteresis loop, associated with the presence of pore-blocking and cavitation phenomena due to open and blocked pores [46].
3.2 Specific surface area analysis with different adsorptives
Table 3 presents the specific surface areas (SBET), and C constant (see Eq. 1) of silica OMM obtained from N2, Ar, O2, and CH4 adsorption data at 77 K, using their customary molecular areas. It can be seen from the values of C constant that the order of adsorbate–adsorbent interaction energies changes when analyzing OMMs with narrow and large mesopore sizes. It is noteworthy that in all cases, despite O2 presents a quadrupole moment, it has an adsorbate-adsorbent interaction similar to Ar, which has no quadrupole moment.
Table 3 shows SBET values of silica OMM using different adsorptives, obtained by using the criteria discussed in Sect. 3.3, and it can be observed that there is variation among them. Thommes et al. [46] stated that at 77 K, the formation of the structure of the Ar monolayer depends on the adsorbent surface chemistry. The samples studied in this work have the same surface chemistry, therefore, in this case, it is expected that the Ar monolayer at 77 K presents the same structure. Therefore, we can consider the SBET value with Ar at 77 K as reference for all samples (SBET-Ar). Based on the previous statement, we observe that: (i) N2 overestimates the SBET between 8 and 22% (14% of total variation), (ii) O2 overestimates the SBET between 13 and 16% (3% of total variation), and (iii) CH4 presents a variation of SBET values of ± 8% (16% of total variation). Furthermore, in order to evaluate these variations, for each sample, we calculate the transversal areas (σ) of N2, O2, and CH4 at 77 K to obtain the same SBET-Ar value. The obtained σ values for N2, O2, and CH4 at 77 K are displayed in Fig. 5, reflecting the behavior of the results presented in Table 3.
It can be observed in Fig. 5 that both N2 and CH4 at 77 K are the adsorptives that present the highest variation of σ values. The results that can be highlighted from Fig. 5 are:
-
(i)
N2 at 77 K presents calculated σ values between 0.133 and 0.149 nm2, where the lower limit is close to the reported values of 0.13–0.135 nm2 for the case of oxides (silica) using Ar at 77 K as reference [51], and TEM analysis for amorphous silica [74]. This result was compared with the obtained using Ar at 87 K as reference (σ = 0.142 nm2) for the SBA-15_B sample (see Figure S3 in the Supplementary Information), finding a σ value of 0.135 nm2 for N2 at 77 K, which is within the obtained range, thus demonstrating the validity of our results. This variation range (which is below the customary value of 0.162 nm2) is because N2 molecule presents the highest permanent quadrupole moment among the studied adsorptives (see Table 1, producing specific interactions with surface silanol groups (Si–OH of silica OMM,these interactions cause a preferential orientation of the N2 molecule adsorbed on the surface of silica OMM, as it was previously reported by Rouquerol et al. [50] and Rouquerol et al. [51]. For instance, if N2 molecules are capable of interact vertically with the –OH groups of the silica surface, the σ value of this configuration becomes 0.11 nm2 [51]. In this sense, depending on the number of surface silanol groups on the silica, there will be an adsorbed monolayer on the surface, composed by a certain amount of N2 molecules oriented vertically (with 0.11 nm2), and others oriented horizontally (with 0.162 nm2), with an average σ value below 0.162 nm2, which is in agreement with the results here obtained. When the percentage of vertically oriented molecules is calculated, values between 25 and 56% (corresponding to 0.149 and 0.133 nm2, respectively) are estimated.
-
(ii)
For CH4 at 77 K, σ values between 0.143 and 0.167 nm2 were found, and the value used initially (0.156 nm2) was within this range. Methane is a three-dimensional molecule (having a tetrahedral shape), symmetric, and does not present quadrupole moment (Table 2). Therefore the variation found in the σ values is directly related to the errors in the determination of the amount adsorbed in the monolayer (nm). Specifically, for MCM-41 and MCM-48 materials, having narrow mesopores, its capillary condensation stage is close (or superposed) to the mono-multilayer filling region (as discussed above); this makes difficult to find a suitable p/po region to apply the BET equation, which considers only the mono-multilayer filling.
-
(iii)
In the case of O2 at 77 K, the calculated σ values (between 0.121 and 0.125 nm2) are almost constant, with an average value of 0.123 nm2, which is lower than the customary value of 0.141 nm2. The size and shape of the O2 molecule are very similar to N2, and at 77 K, both molecules, are in the same thermodynamic state (vapor); the only difference between them is that O2 has a quadrupole moment four times lower than N2 (see Table 2), suggesting that this electrostatic interaction does not contribute significantly to the total interaction with the silanol groups present in OMM. This result is relevant because O2 at 77 K can be considered as an interesting adsorptive for the SBET determination of porous solids (using a σ value of 0.123 nm2), complementing the reported by Jagiello and collaborators [75, 76] who used O2 at 77 K as a probe adsorptive to obtain the pore size distribution of different carbon materials. Also, this adsorptive has the advantage (with respect to Ar at 77 K) that the temperature of analysis is above its triple-point temperature, which means that it does not behave like a subcooled liquid.
3.3 Considerations for the S BET calculation
In order to guarantee a consistent SBET value, the criteria proposed by Rouquerol et al. [77] and included in the last IUPAC recommendations [46] must be considered. One of them is the plotting of nads(1-p/po) vs. p/po (normalized adsorption isotherm, Fig. 6) to find the p/po value where the curve reaches its maximum, representing the upper limit for the application of the BET equation. When this criterion is applied, typical curves as shown in Figs. 6c and d are observed.
Nonetheless, in porous materials with well-defined narrow mesopores, the capillary condensation stage is close to the mono-multilayer filling region; in these cases, the maximum of the normalized adsorption isotherm is where the stage of capillary condensation finishes, as shown in Figs. 6a and b. Thus, for these cases, the p/po maximum value must be the inflection point where the capillary condensation starts (marked by an arrow in Figs. 6a and b for Ar as an example).
In Fig. 6a and b, it can be observed that in the normalized adsorption isotherm, with Ar the separation between the mono-multilayer adsorption and the capillary condensation regions is the highest. Whereas with CH4, it can be observed that the normalized adsorption isotherm presents the highest proximity between the capillary condensation and the filling of the mono-multilayer regions. This analysis agrees with the fact that the measurement of the specific surface area using the BET method is better defined with adsorptives such as Ar and O2.
4 Conclusions
In this work, we measured and analyzed the gas adsorption isotherms of various OMM using N2, Ar, O2, and CH4 as adsorptives at 77 K and evaluated the SBET. Both pore volumes and pore size distribution will be analyzed in future papers.
Regarding the SBET evaluation, we conclude the following:
-
(i)
The quadrupole moment present in N2 interacts with the OMM silanol groups causing a preferential orientation and affecting the σ values significantly; thus, the SBET is overestimated.
-
(ii)
CH4 adsorption at 77 K on the OMM with narrow mesopores did not present a clear “knee” formation (B point), because the capillary condensation stage occurs along with the mono-multilayer adsorption (possibly due to its low saturation pressure, ca. 9 mm Hg).
-
(iii)
The main conclusion of this work is that in O2 at 77 K, the obtained σ values were almost constant, its low quadrupole moment (four times lower than N2), which does not interact with the OMM silanol groups, and the ease of accessing cryogenic liquids to reach this temperature, make O2 a reliable adsorptive to calculate the SBET of silica OMM.
References
Taguchi, A., Schüth, F.: Ordered mesoporous materials in catalysis. Microporous Mesoporous Mater. 77, 1–45 (2005). https://doi.org/10.1016/j.micromeso.2004.06.030
Davis, M.E.: Ordered porous materials for emerging applications. Nature 417, 813–821 (2002). https://doi.org/10.1038/nature00785
Hoffmann, F., Cornelius, M., Morell, J., Fröba, M.: Silica-based mesoporous organic-inorganic hybrid materials. Angew. Chem. Int. Ed. 45(20), 3216–3251 (2006). https://doi.org/10.1002/anie.200503075
Ravikovitch, P.I., Neimark, A.V.: Characterization of micro- and mesoporosity in SBA-15 materials from adsorption data by NLDFT method. J. Phys. Chem. B 105(29), 6817–6823 (2001). https://doi.org/10.1021/jp010621u
De Araújo, M.J.G., Villarroel-Rocha, J., De Souza, V.C., Sapag, K., Pergher, S.B.C.: Carbon foams from sucrose employing different metallic nitrates as blowing agents: application in CO2 capture. J. Anal. Appl. Pyrolysis 141, 103627 (2019). https://doi.org/10.1016/j.jaap.2019.05.016
Bastos-Neto, M., Canabrava, D.V., Torres, A.E.B., Rodriguez-Castellón, E., Jiménez-López, A., Azevedo, D.C.S., Cavalcante, C.L., Jr.: Effects of textural and surface characteristics of microporous activated carbons on the methane adsorption capacity at high pressures. Appl. Surf. Sci. 253(13), 5721–5725 (2007). https://doi.org/10.1016/j.apsusc.2006.12.056
Gadiou, R., Saadallah, S.-E., Piquero, T., David, P., Parmentier, J., Vix-Guterl, C.: The influence of textural properties on the adsorption of hydrogen on ordered nanostructured carbons. Microporous Mesoporous Mater. 79(1–3), 121–128 (2005). https://doi.org/10.1016/j.micromeso.2004.10.034
Karatepe, N., Orbak, I., Yavuz, R., Özyuǧuran, A.: Sulfur dioxide adsorption by activated carbons having different textural and chemical properties. Fuel 87(15–16), 3207–3215 (2008). https://doi.org/10.1016/j.fuel.2008.06.002
Li, J., Cheng, S., Zhao, Q., Long, P., Dong, J.: Synthesis and hydrogen-storage behavior of metal–organic framework MOF-5. Int. J. Hydrogen Energy 34(3), 1377–1382 (2009). https://doi.org/10.1016/j.ijhydene.2008.11.048
Montiel-Centeno, K., Barrera, D., Villarroel-Rocha, J., Moreno, M.S., Sapag, K.: CMK-3 nanostructured carbon: effect of temperature and time carbonization on textural properties and H2 storage. Chem. Eng. Commun. 206(11), 1–15 (2019). https://doi.org/10.1080/00986445.2019.1615469
Montiel-Centeno, K., Barrera, D., Villarroel-Rocha, J., Moreno, M.S., Sapag, K.: Hierarchical nanostructured carbons as CO2 adsorbents. Adsorption 25, 1287–1297 (2019). https://doi.org/10.1007/s10450-019-00089-3
Parra, J.B., Ania, C.O., Arenillas, A., Rubiera, F., Palacios, J.M., Pis, J.J.: Textural development and hydrogen adsorption of carbon materials from PET waste. J. Alloys Compd. 379(1–2), 280–289 (2004). https://doi.org/10.1016/j.jallcom.2004.02.044
Saha, D., Deng, S., Yang, Z.: Hydrogen adsorption on metal-organic framework (MOF-5) synthesized by DMF approach. J. Porous Mater. 16, 141–149 (2009). https://doi.org/10.1007/s10934-007-9178-3
Wahby, A., Silvestre-Albero, J., Sepúlveda-Escribano, A., Rodríguez-Reinoso, F.: CO2 adsorption on carbon molecular sieves. Microporous Mesoporous Mater. 164, 280–287 (2012). https://doi.org/10.1016/j.micromeso.2012.06.034
Janus, R., Piotr, K., Dudek, B., Piwowarska, Z., Kochanowski, A.: Removal of methyl–ethyl ketone vapour on polyacrylonitrile-derived carbon/mesoporous silica nanocomposite adsorbents. Microporous Mesoporous Mater. 145, 65–73 (2011). https://doi.org/10.1016/j.micromeso.2011.04.029
Pui, W.K., Yusoff, R., Aroua, M.K.: A review on activated carbon adsorption for volatile organic compounds (VOCs). Rev. Chem. Eng. 35(5), 649–668 (2018). https://doi.org/10.1515/revce-2017-0057
Xin, L., Huiling, L., Siqi, H., Zhong, L.: Dynamics and isotherms of water vapor sorption on mesoporous silica gels modified by different salts. Kinet. Catal. 51, 754–761 (2010). https://doi.org/10.1134/S0023158410050186
Yates, M., Blanco, J., Martin-Luengo, M.A., Martin, M.P.: Vapour adsorption capacity of controlled porosity honeycomb monoliths. Microporous Mesoporous Mater. 65, 219–231 (2003). https://doi.org/10.1016/j.micromeso.2003.08.009
Zhang, X., Gao, B., Fang, J., Zou, W., Dong, L., Cao, C., Zhang, J., Li, Y., Wang, H.: Chemically activated hyrochar as an effective adsorbent for volatile organic compounds (VOCs). Chemosphere 218, 680–686 (2019). https://doi.org/10.1016/j.chemosphere.2018.11.144
Almazán-Almazán, M.C., López-Domingo, F.J., Domingo-García, M., Léonard, A., Pérez-Mendoza, M., Pirard, J.P., López-Garzón, F.J., Blacher, S.: Influence of carbon xerogel textural properties on the dynamic adsorption of methyl iodide. Chem. Eng. J. 173(1), 19–28 (2011). https://doi.org/10.1016/j.cej.2011.07.027
Barrera, D., Villarroel-Rocha, J., Sapag, K.: Synthesis and textural characterization of a templated nanoporous carbon from MCM-22 zeolite and its use as adsorbent of amoxicillin and ethinylestradiol. Adsorption 20, 967–976 (2014). https://doi.org/10.1007/s10450-014-9640-x
Fathy, N.A., Sayed Ahmed, S.A., Abo El-enin, R.M.M.: Effect of activation temperature on textural and adsorptive properties for activated carbon derived from local reed biomass: Removal of p-nitrophenol. Environ. Res. Eng. Manag. 59(1), 10–22 (2012)
Haque, E., Khan, N.A., Talapaneni, S.N., Vinu, A., JeGal, J.-G., Jhung, S.-H.: Adsorption of phenol on mesoporous carbon CMK-3: effect of textural properties. Bull. Korean Chem. Soc. 31(6), 1638–1642 (2010). https://doi.org/10.5012/bkcs.2010.31.6.1638
Hartmann, M., Vinu, A., Chandrasekar, G.: Adsorption of vitamin E on mesoporous carbon molecular sieves. Chem. Mater. 17(4), 829–833 (2005). https://doi.org/10.1021/cm048564f
Huang, J., Jin, X., Mao, J., Yuan, B., Deng, R., Deng, S.: Synthesis, characterization and adsorption properties of diethylenetriamine-modified hypercrosslinked resins for efficient removal of salicylic acid from aqueous solution. J. Hazard. Mater. 217–218, 406–415 (2012). https://doi.org/10.1016/j.jhazmat.2012.03.053
Moral-Rodríguez, A.I., Leyva-Ramos, R., Ania, C.O., Ocampo-Pérez, R., Isaacs-Páez, E.D., Carrales-alvarado, D.H., Parra, J.B.: Tailoring the textural properties of an activated carbon for enhancing its adsorption capacity towards diclofenac from aqueous solution. Environ. Sci. Pollut. Res. 26, 6141–6152 (2019). https://doi.org/10.1007/s11356-018-3991-x
Pires, J., Carvalho, A., Carvalho, M.B.D.: Adsorption of volatile organic compounds in Y zeolites and pillared clays. Microporous Mesoporous Mater. 43(3), 277–287 (2001). https://doi.org/10.1016/S1387-1811(01)00207-4
Durán-Jiménez, G., Stevens, L.A., Hodgins, G.R., Uguna, J., Ryan, J., Binner, E.R., Robinson, J.P., Durán-jiménez, G., Stevens, L.A., Hodgins, G.R., Uguna, J., Ryan, J., Binner, E.R., Robinson, J.P.: Fast regeneration of activated carbons saturated with textile dyes: textural, thermal and dielectric characterization. Chem. Eng. J. 378, 121774 (2019). https://doi.org/10.1016/j.cej.2019.05.135
Graham, N., Chen, X.G., Jayaseelan, S.: The potential application of activated carbon from sewage sludge to organic dyes removal. Water Sci. Technol. 43(2), 245–252 (2001). https://doi.org/10.2166/wst.2001.0096
Liu, G., Yang, R., Li, M.: Liquid adsorption of basic dye using silica aerogels with different textural properties. J. Non. Cryst. Solids. 356(4–5), 250–257 (2010). https://doi.org/10.1016/j.jnoncrysol.2009.11.019
Ania, C.O., Gomis-berenguer, A., Dentzer, J., Vix-guterl, C.: Nanoconfinement of glucose oxidase on mesoporous carbon electrodes with tunable pore sizes. J. Electroanal. Chem. 808, 372–379 (2018). https://doi.org/10.1016/j.jelechem.2017.09.009
Katiyar, A., Ji, L., Smirniotis, P., Pinto, N.G.: Protein adsorption on the mesoporous molecular sieve silicate SBA-15: effects of pH and pore size. J. Chromatogr. A 1069, 119–126 (2005). https://doi.org/10.1016/j.chroma.2004.10.077
Vinu, A., Murugesan, V., Tangermann, O., Hartmann, M.: Adsorption of cytochrome c on mesoporous molecular sieves: Influence of pH, pore diameter, and aluminum incorporation. Chem. Mater. 16(16), 3056–3065 (2004). https://doi.org/10.1021/cm049718u
Atia, A.A., Donia, A.M., Abou-el-enein, S.A., Yousif, A.M.: Studies on uptake behaviour of copper (II) and lead (II) by amine chelating resins with different textural properties. Sep. Purificat. Technol. 33(3), 295–301 (2003). https://doi.org/10.1016/S1383-5866(03)00089-3
García-Sánchez, A., Alastuey, A., Querol, X.: Heavy metal adsorption by different minerals: application to the remediation of polluted soils. Sci. Total Environ. 242(1–3), 179–188 (1999). https://doi.org/10.1016/S0048-9697(99)00383-6
Tufo, A.E., Larralde, A.L., Villarroel-Rocha, J., Sapag, K., Sileo, E.E.: Synthesis and characterization of pure and Al-substituted akaganeites and evaluation of their performance to adsorb As(V). J. Environ. Chem. Eng. (2018). https://doi.org/10.1016/j.jece.2018.10.009
Aguado, J., Serrano, D.P., Escola, J.M., Peral, A.: Catalytic cracking of polyethylene over zeolite mordenite with enhanced textural properties. J. Anal. Appl. Pyrol. 85(1–2), 352–358 (2009). https://doi.org/10.1016/j.jaap.2008.10.009
Campelo, J.M., García, A., Luna, D., Marinas, J.M.: Textural properties, surface chemistry and catalytic activity in cyclohexene skeletal isomerization of acid treated natural sepiolites. Mater. Chem. Phys. 24(1–2), 51–70 (1989). https://doi.org/10.1016/0254-0584(89)90045-X
Faria, P.C.C., Órfao, J.J.M., Pereira, M.F.R.: Ozone decomposition in water catalyzed by activated carbon: influence of chemical and textural properties. Ind. Eng. Chem. Res. 45(8), 2715–2721 (2006). https://doi.org/10.1021/ie060056n
Hong, E., Kim, C., Lim, D.-H., Cho, H.-J., Shin, C.-H.: Catalytic methane combustion over Pd/ZrO2 catalysts: effect of crystalline structure and textural properties. Appl. Catal. B Environ. 232(15), 544–552 (2018). https://doi.org/10.1016/j.apcatb.2018.03.101
Iwamoto, M., Tanaka, Y., Sawamura, N., Namba, S.: Remarkable effect of pore size on the catalytic activity of mesoporous silica for the acetalization of cyclohexanone with methanol. J. Am. Chem. Soc. 125(43), 13032–13033 (2003)
Khodakov, A.Y., Griboval-constant, A., Bechara, R., Zholobenko, V.L.: Pore size effects in Fischer Tropsch synthesis over cobalt-supported mesoporous silicas. J. Catal. 206(2), 230–241 (2002). https://doi.org/10.1006/jcat.2001.3496
Suzuki, T.M., Yamamoto, M., Fukumoto, K., Akimoto, Y., Yano, K.: Investigation of pore-size effects on base catalysis using amino-functionalized monodispersed mesoporous silica spheres as a model catalyst. Microporous Mesoporous Mater. 251(2), 249–257 (2007). https://doi.org/10.1016/j.jcat.2007.08.010
Tichit, D., Lhouty, M.H., Guida, A., Chiche, B.H., Figueras, F., Auroux, A., Bartalini, D., Garrone, E.: Textural properties and catalytic activity of hydrotalcites. J. Catal. 151(1), 50–59 (1995). https://doi.org/10.1006/jcat.1995.1007
Vanhove, D., Zhuyong, Z., Makambo, L., Blanchard, M.: Hydrocarbon selectivity in Fischer-Tropsch synthesis in relation to textural properties of supported cobalt catalysts. Appl. Catal. 9(3), 327–342 (1984). https://doi.org/10.1016/0166-9834(84)80004-4
Thommes, M., Kaneko, K., Neimark, A.V., Olivier, J.P., Rodriguez-Reinoso, F., Rouquerol, J., Sing, K.S.W.: Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report ). Pure Appl. Chem. 87(9–10), 1051–1069 (2015). https://doi.org/10.1515/pac-2014-1117
De Torre, L.E.C., Flores, E.S., Bottani, E.J.: Nitrogen physisorption on modified silica surfaces. Langmuir 16(4), 1896–1901 (2000)
Ehrburger-Dolle, F., Holz, M., Lahaye, J.: Use of N2, Ar and CO2 adsorption for the determination of microporosity and surface fractal dimension of carbon blacks and silicas. Pure Appl. Chem. 65(10), 2223–2230 (1993). https://doi.org/10.1351/pac199365102223
McClellan, A.L., Harnsberger, H.F.: Cross-sectional areas of molecules adsorbed on solid surfaces. J. Colloid Interface Sci. 23(4), 577–599 (1967). https://doi.org/10.1016/0021-9797(67)90204-4
Rouquerol, J., Rouquerol, F., Grillet, Y., Boudellal, M.: In: Gregg, S.J., Sing, K.S.W., Stoeckli, H.F. (eds.) Characterization of Porous Solids. The Society of Chemical Industry, London (1979)
Rouquerol, F., Rouquerol, J., Sing, K.S.W., Llewellyn, P., Maurin, G.: Adsorption by Powders and Porous Solids: Principles, Methodology and Applications. Elsevier, Oxford (2014)
Branton, P.J., Hall, P.G., Sing, K.S.W.: Physisorption of nitrogen and oxygen by MCM-41, a model mesoporous adsorbent. J. Chem. Soc. Chem. Commun. (1993). https://doi.org/10.1039/C39930001257
Edler, K.J.: Template induction of supramolecular structure: Synthesis and characterisation of the mesoporous molecular sieve, MCM-41. PhD Thesis, Australian National University, Canberra (1997). https://doi.org/10.25911/5d7a27505b4ec
Morishige, K., H., Fujii, M., Uga, D. : Capillary critical point of argon, nitrogen, oxygen, ethylene, and carbon dioxide in MCM-41. Langmuir 13(13), 3494–3498 (1997)
Morishige, K., Nakamura, Y.: Nature of adsorption and desorption branches in cylindrical pores. Langmuir 20(11), 4503–4506 (2004). https://doi.org/10.1021/la030414g
Sonwane, C.G., Bhatia, S.K., Calos, N.: Experimental and theoretical investigations of adsorption hysteresis and criticality in MCM-41: Studies with O2, Ar, and CO2. Ind. Eng. Chem. Res. 37(6), 2271–2238 (1998). https://doi.org/10.1021/ie970883b
Branton, P.J., Hall, P.G., Sing, K.S.W., Reichert, H., Schüth, F., Unger, K.K.: Physisorption of argon nitrogen and oxygen by MCM-41 a model mesoporous adsorbent. J. Chem. Soc. Faraday Trans. 90(19), 2965–2967 (1994). https://doi.org/10.1039/FT9949002965
Morishige, K.: Nature of adsorption hysteresis in cylindrical pores: effect of pore corrugation. J. Phys. Chem. C 120(39), 22508–22514 (2016). https://doi.org/10.1021/acs.jpcc.6b07764
Thommes, M., Köhn, R., Fröba, M.: Sorption and pore condensation behavior of pure fluids in mesoporous MCM-48 silica MCM-41 silica SBA-15 silica and controlled-pore glass at temperatures above and below the bulk triple point. Appl. Surf. Sci. 196(1–4), 239–249 (2002). https://doi.org/10.1016/S0169-4332(02)00062-4
Barrera, D., Villarroel-Rocha, J., Marenco, L., Oliva, M.I., Sapag, K.: Non-hydrothermal synthesis of cylindrical mesoporous materials: influence of the surfactant/silica molar ratio adsorption science & technology. Adsorpt. Sci. Technol. 29, 975–988 (2011). https://doi.org/10.1260/0263-6174.29.10.975
Schumacher, K., Ravikovitch, P.I., Chesne, ADu., Neimark, A.V., Unger, K.K.: Characterization of MCM-48 materials. Langmuir 16(10), 4648–4654 (2000). https://doi.org/10.1021/la991595i
Stevens, W.J.J., Mertens, M., Mullens, S., Thijs, I., Van Tendeloo, G., Cool, P., Vansant, E.F.: Formation mechanism of SBA-16 spheres and control of their dimensions. Microporous Mesoporous Mater. 93(1–3), 119–124 (2006). https://doi.org/10.1016/j.micromeso.2006.02.015
Govinda Raju, G.: Gaseous Electronics: Tables, Atoms, and Molecules. CRC Press, Boca Raton (2017)
Reid, R., Poling, B.E., Prauznits, J.M.: The Properties of Gases and Liquids. McGraw-Hill Companies, New York (1987)
Sing, K.S.W., Williams, R.T.: The use of molecular probes for the characterization of nanoporous adsorbents. Part. Part. Syst. Charact. 21, 71–79 (2004). https://doi.org/10.1002/ppsc.200400923
Brunauer, S., Emmett, P.H., Teller, E.: Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 60, 309–319 (1938). https://doi.org/10.1021/ja01269a023
Emmett, P.H., Brunauer, S.: The use of low temperature van der Waals adsorption isotherms in determining the surface area of iron synthetic ammonia catalysts. J. Am. Chem. Soc. 59(8), 1553–1564 (1937). https://doi.org/10.1021/ja01287a041
Yamashita, Y., Kato, M.: Viscoelastic properties of polycrstalline solid methane and carbon dioxide. Geophys. Res. Lett. 24, 1327–1330 (1997). https://doi.org/10.1029/97GL01205
Cychosz, K.A., Guillet-Nicolas, R., García-Martínez, J., Thommes, M.: Recent advances in the textural characterization of hierarchically structured nanoporous materials. Chem. Soc. Rev. 46(2), 389–414 (2017)
Cychosz, K.A., Thommes, M.: Progress in the physisorption characterization of nanoporous gas storage materials. Engineering 4(4), 559–566 (2018). https://doi.org/10.1016/j.eng.2018.06.001
Kruk, M., Jaroniec, M.: Determination of mesopore size distribution from Argon adsorption data at 77 K. J. Phys. Chem. B 106(18), 4732–4739 (2002). https://doi.org/10.1021/jp0137423
Thommes, M., Köhn, R., Fröba, M.: Sorption and pore condensation behavior of nitrogen, argon, and krypton in mesoporous MCM-48 silica materials. J. Phys. Chem. B 104(33), 7932–7943 (2000). https://doi.org/10.1021/jp994133m
Landers, J., Gor, G.Y., Neimark, A.V.: Density functional theory methods for characterization of porous materials. Colloids Surf. A 437, 3–32 (2013). https://doi.org/10.1016/j.colsurfa.2013.01.007
Jelinek, L., Kováts, E.: True surface areas from nitrogen adsorption experiments. Langmuir 10, 4225–4231 (1994). https://doi.org/10.1021/la00023a051
Jagiello, J., Kenvin, J.: Consistency of carbon nanopore characteristics derived from adsorption of simple gases and 2D-NLDFT models. Advantages of using adsorption isotherms of oxygen (O2) at 77 K. J. Colloid Interface Sci. 542(15), 151–158 (2019). https://doi.org/10.1016/j.jcis.2019.01.116
Jagiello, J., Kenvin, J., Ania, C., Parra, J.B., Celzard, A., Fierro, V.: Exploiting the adsorption of simple gases O2 and H2 with minimal quadrupole moments for the dual gas characterization of nanoporous carbons using 2D-NLDFT models. Carbon 160, 164–175 (2020). https://doi.org/10.1016/j.carbon.2020.01.013
Rouquerol, J., Llewellyn, P., Rouquerol, F.: Is the BET equation applicable to microporous adsorbents? Stud. Surf. Sci. Catal. 160, 49–56 (2007). https://doi.org/10.1016/S0167-2991(07)80008-5
Acknowledgements
The authors want to thank UNSL and CONICET for the financial support of this work.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Villarroel-Rocha, J., Barrera, D., Arroyo-Gómez, J.J. et al. Insights of adsorption isotherms with different gases at 77 K and their use to assess the BET area of nanoporous silica materials. Adsorption 27, 1081–1093 (2021). https://doi.org/10.1007/s10450-021-00339-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-021-00339-3