Abstract
Purpose
To describe the clinical features and treatment outcomes of severe retinopathy in eyes with incontinentia pigmenti (IP) of infants within a few months of birth.
Study design
Retrospective clinical study.
Methods
Six eyes of three patients (6-day-old girl, 5-month-old girl, and 14-day-old boy) with IP were examined and treated under general anesthesia. Ophthalmologic examinations were performed including images from wide-angle fluorescein angiography (FA), swept-source optical coherence tomography (OCT), and OCT angiography (OCTA).
Results
Ophthalmoscopy showed prominent vascular tortuosity in five eyes, retinal hemorrhages in four eyes, and incomplete vascular development in two eyes. FA showed extensive avascularity including the posterior pole of the retina in all cases except one eye. Prompt and intensive laser photocoagulation stabilized the pre-proliferative severe retinopathy in five eyes; however, foveal structure and vessel anomalies were detected in three of six eyes by OCT and two of five eyes by OCTA.
Conclusion
Severe retinopathy in the neonatal period and infancy was present not only in the periphery but also in the posterior pole including the fovea, which might be related to retinal vascular maldevelopment. It is, therefore, recommended that wide-angle fundus FA examination be performed in the early postnatal period to detect early signs of severe retinopathy in infants with IP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Incontinentia pigmenti (IP), known as Bloch-Sulzberger syndrome, was described first in 1906 by Garrod [1]. A rare X-linked dominantly inherited syndrome mostly seen in females yet fatal in males, has an incidence of 1 in 40,000 newborns; in most cases, IP results from mutations in the IKBKG gene, responsible for impaired signaling of nuclear factor kappa B, a critical transcription factor that up-regulates the immune system and prevents apoptosis [2]. Female infants with IP develop characteristic abnormalities of the skin, central nervous system (CNS), bones, hair, teeth, and eyes. Approximately 25–77% of cases are associated with ophthalmic manifestations, which primarily occur in the retina, such as vascular occlusion, neovascularization, vitreoretinal hemorrhages, and exudative and tractional retinal detachments [3,4,5]. Although the pathogenesis of retinopathy in IP is quite different from that of retinopathy of prematurity (ROP); vascular developmental disorders, severe vaso-occlusion, and subsequent neovascularization progress rapidly, similar to severe ROP. Based on phenotypic similarities in other progressive vitreoretinal diseases such as ROP and familial exudative vitreoretinopathy, application of laser photocoagulation to the peripheral ischemic retina is recommended to cause regression of fibrovascular proliferation and prevent tractional retinal detachment in IP [6, 7]. However, the indications of prophylactic ablation remain unclear, and few reports provide a detailed description of the clinical features and treatment outcomes in severe cases [8,9,10,11,12]. We report three infants with IP who developed severe retinopathy with extensive avascularity and foveal anomaly within a few months of birth and their favorable outcomes associated with prompt and intensive laser photocoagulation for these progressive retinopathies.
Materials and methods
The institutional review board of the National Center for Child Health and Development approved this study (permit no. 1676). The legal guardians of the three infants provided informed consent.
The medical records of the three infants with IP were reviewed retrospectively. The data collected included age, sex, decimal best-corrected visual acuity (BCVA), fundus examinations, fluorescein angiography (FA), swept-source optical coherence tomography (SS-OCT), and OCT angiography (OCTA) (DRI OCT-1, Topcon Corporation), and medical and family histories. Fundus photography and wide-angle FA were performed using the RetCam imaging system (Massie Research Laboratories). The foveas were evaluated using SS-OCT and OCTA, in which the superficial plexus was analyzed in the 3 × 3 mm area around the fovea.
We determined the severity of the retinopathy based on the zones of the extent of the avascular areas and a plus sign such as vascular dilation and tortuosity in the posterior pole for active retinopathy as described in the International Classification of ROP [13]. We defined “pre-proliferative retinopathy” as zone I retinopathy with or without a plus sign and zone II retinopathy with a plus sign as in pre-threshold ROP. We judged that the retinopathy with a prominent plus sign, vascular maldevelopment indicating hypoplasia with dysplasia of the retinal vasculature, shunting vessels, and retinal hemorrhage, corresponded to aggressive posterior retinopathy resembling that of ROP (AP-ROP) [13, 14]. Dense photocoagulation was applied to all avascular areas even those in the vascular arcade of the posterior pole, as is done to treat the AP-ROP [14].
Results
We examined six eyes of three patients (6-day-old girl, 5-month-old girl, and 14-day-old boy) who presented with retinopathy with IP that had been diagnosed by pediatricians and dermatologists based on the characteristic skin lesions and confirmed by skin biopsy. Two patients had malformed teeth, but no cranial or other complications were apparent in the three cases. The clinical features, history, FA, OCT, and OCTA findings of the six eyes are summarized in Table 1.
Case 1
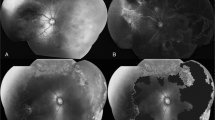
A 6-day-old female infant with IP was referred to our hospital for ophthalmologic examination. She was born at 39 weeks and 1-day gestation and weighed 3016 g with a normal APGAR score. The family history was negative for anything notable; her mother had no history of spontaneous abortion. At the initial examination, the anterior segment was normal, but ophthalmoscopy showed prominent retinal arterial narrowing and tortuosity, venous dilation in zone I, and marked vascular maldevelopment bilaterally with shunting vessels and multiple retinal hemorrhages indicating aggressive posterior retinopathy (Fig. 1a, b). An examination was performed under general anesthesia the same day. FA showed extensive avascular retina involving the fovea OU. Extremely severe retinal vascular occlusion involving the fovea, especially OD, had occurred, and the retinal vasculature was limited to around the optic disc region, in which prominent anastomoses of anomalous vessels were observed (Fig. 1c, d). Analogous to the treatment rationale for AP-ROP, the avascular retinas OU were treated immediately with intensive laser photocoagulation applied to almost the entire retina from the posterior pole to the ora serrata. Indirect argon laser photocoagulation was applied to all avascular retinas and anomalous shunt vessels detected by FA (972 spots OD; 715 spots OS; 0.3 s; 260–360 mW; near confluent, 1 application diameter of spacing). Power settings varied according to the light mark of the laser spots to avoid excessive coagulation. In order to cover the skipped lesions and residual anomalous vessels with a plus sign after 3 days, we repeated the laser treatment of the extensive retinal avascularity including the posterior pole (293 spots OD; 405 spots OS; 0.3 s; 260–360 mW; confluent, 1 application diameter of spacing) and stabilized the aggressive posterior retinopathy within about 3 months without fibrovascular proliferation (Fig. 1e, f). Unfortunately, the laser scars expanded near the macula. Follow-up examinations of the treated aggressive retinopathy in IP were scheduled twice weekly during the first and second weeks, once weekly during the next 2 weeks, once every 2 weeks for 2 months, once monthly up to the sixth month, then once every 3 months–6 years of age. OCT performed at 5 years of age showed a degenerated macula OD due to enlargement of the laser scar. In the left eye, the macula appeared normal on the OCT images (Fig. 2a), but OCTA showed abnormal retinal capillaries in the foveal avascular zones (Fig. 2b).
Case 1. Fundus photographs and fluorescein angiography (FA) images OD (a, c) and OS (b, d). Severe vascular occlusion (arrows) and multiple retinal hemorrhages (arrowheads) are seen in zone I (a, b). FA in the early phase shows that almost all retinal perfusion is disrupted, except for abnormal vasculature around optic disc region that resulted in irregular anastomoses (arrows) (c). Tiny retinal perfusion with anastomosis is preserved OS, in which capillary dropout is observed (arrowheads) (d). Intensive two times of laser photocoagulation stabilized the activity of the severe retinopathy OD (e) and OS (f)
Postoperative optical coherence tomography (OCT), OCT angiography (OCTA) (3 × 3 mm, asterisks indicate temporal retina) in the superficial plexus images in case 1 (a, b) at 5 years of age, case 2 (c, d) at 10 months of age, and case 3 (e–h) at 1 month of age. Case 1 OS, the foveal structure with bulging is preserved as seen on an OCT image (a), but there are abnormal retinal capillaries of foveal avascular zones seen on OCTA images (b). Case 2 OD, although thinning of retinal layers is seen due to creeping of laser scars adjacent to the macula, foveal structure with bulging appears to be preserved on an OCT image (c), and the vascular density in the superficial plexus (d) is preserved on an OCTA image despite the extensive avascular area. Case 3 OS, the ellipsoidal zone of the macula is discontinuous on an OCT image (e), and OCTA shows that the macular vascular density in the superficial plexus (f) is decreased compared to the right eye (g, h)
At 2 years of age the cycloplegic refraction was −4.00 diopters (D) cyl −0.50 D Ax 180° OD and emmetropia OS. Refractive correction for myopic astigmatism OD and amblyopia treatment with patching resulted in a BCVA of 1.2 OU, but at 6 years of age it was 0.2 OD and 0.9 OS due to latent nystagmus. She maintained orthophoria but did not show stereopsis. Although the retina had extensive avascularity during the neonatal period, the patient did not have seizures, developmental delays, or other complications up to 6 years of age. No cerebral abnormalities were found by magnetic resonance imaging (MRI). At the last visit, malformation of the teeth was observed.
Case 2
A 5-month-old female infant with IP was referred to our hospital for ophthalmologic examination. She was born at 38 weeks and 1-day gestation and weighed 2390 g with a normal APGAR score. The family history revealed that her mother was diagnosed with IP and had retinopathy OS treated with laser photocoagulation. Although the patient had no symptoms and was apparently normal up to the age of 3 months, she developed the characteristic blistering rash accompanied by prominent hyperpigmentation on her limbs and trunk at the age of 4 months. At the initial visit to our hospital, the anterior segment was normal but ophthalmoscopy showed dilation and tortuosity of the retinal vessels with numerous patchy retinal hemorrhages bilaterally (Fig. 3a, b). An examination was performed under general anesthesia the same day. FA showed extensive retinal avascularity in zone I that reached the fovea OU (Fig. 3c, d). Analogous to the treatment rationale for AP-ROP, the avascular retinas and anomalous vessels detected by FA including the posterior poles OU were treated immediately with intensive indirect argon laser photocoagulation (1579 spots OD; 1146 spots OS; 0.3 s; 240 mW; confluent, 1 application diameter of spacing). The severe retinopathy stabilized within 3 months without fibrovascular proliferation (Fig. 3e, f). The avascular retina that extended close to the macula also was treated with intense laser, which resulted in thinning and degeneration of the retinal layers. However, the patient developed good bilateral fixation and following behavior at the age of 22 months, supported by the preserved foveal structure with bulging OU detected by OCT and OCTA (Fig. 2c, d). No cerebral or other complications developed.
Case 2. Fundus photographs and fluorescein angiography (FA) images OD (a, c) and OS (b, d). Mild dilation and tortuosity of the retinal vessels (arrows) with multiple retinal hemorrhages (arrowheads) are seen in zone I OU (a, b). FA shows extensive avascularity in zone I (arrowheads) adjacent to the fovea bilaterally (c, d). The early phase of FA shows prominent vascular occlusion and anastomosis OS (d). FA-guided intensive laser photocoagulation stabilized the retinopathy (e, f)
Case 3
A 14-day-old male infant with a diagnosis of IP confirmed by skin biopsy was referred to our hospital for ophthalmologic examination. He was born at 42 weeks and 2 days gestation and weighed 3862 g with a normal APGAR score. The family history was negative, except for one spontaneous abortion by his mother. Chromosomal analysis to identify XXY, the major cause of IP in boys, was negative, suggesting somatic mosaicism, another reported cause in male infants [15]. At the initial visit, the anterior segments were normal bilaterally, but ophthalmoscopy showed prominent vascular tortuosity and anastomoses involving the posterior pole of the retina OS (Fig. 4a, b). One week after the initial visit, the patient was examined under general anesthesia. FA showed minimal avascular retina at the periphery within zone II OD but extensive retinal avascularity throughout 360° of the retina reaching zone I OS (Fig. 4c, d). It was determined that the zone I retinopathy with a plus sign OS was pre-proliferative, whereas the zone II retinopathy with a minimal plus sign OD was non-proliferative but possibly progressed to pre-proliferative retinopathy. The avascular retinas OU were treated immediately with indirect argon laser photocoagulation (329 spots OD; 765 spots OS; 0.2 s; 200 mW; near confluent, 1 application diameter of spacing), and further progression of the avascularity did not occur after the initial treatment and stabilized within 1 month (Fig. 4e, f). Although the retinopathy was not as aggressive compared with the other two cases, OCT showed an abnormal foveal structure with a discontinuous ellipsoidal zone OS (Fig. 2e). OCTA detected decreased vascular density in the superficial plexus (Fig. 2f). OCT showed mild foveal hypoplasia at the age of 1 month (Fig. 2g) OD with minimal avascularity in the periphery, and OCTA detected normal vascular density in the superficial plexus (Fig. 2h). The patient developed good bilateral fixation and following behavior at the age of 18 months. MRI showed no CNS abnormalities or other severe systemic complications, except for minor malformation of the teeth at 8 months of age.
Case 3. Fundus photographs and fluorescein angiography (FA) images OD (a, c) and OS (b, d). Peripheral avascular retina (arrowheads) with minimum vascular tortuosity is seen OD (a), while vascular tortuosity and anastomosis (arrows) are prominent in the temporal retina extending to the posterior pole OS (b). FA shows almost normal posterior-midperipheral perfusion with avascularity and anastomosis in the peripheral retina (arrowheads) within zone II OD (c). However, FA in the early phase reveals extensive vascular occlusion (arrows) of the temporal retina reaching zone I OS (d). Laser photocoagulation applied to the avascular retina OU stabilized the retinopathy (e, f)
Discussion
In the current cases, within a few months after birth, severe retinal vascular occlusion occurred and FA revealed extensive retinal avascularity involving the fovea OU of cases 1 and 2 and OS of case 3. We determined the severity of the retinopathy based on the zones of the extent of the avascular areas and a plus sign such as vascular dilation and tortuosity in the posterior pole for active retinopathy as described in the International Classification of ROP [13]. To clarify the rationale and pattern of laser treatment, we also defined pre-proliferative retinopathy as similar to pre-threshold ROP and aggressive posterior retinopathy as resembling AP-ROP [13, 14]. In this series, we successfully treated these eyes with intensive laser photocoagulation following diagnosis during the pre-proliferative phase of retinopathy. Previously, only a few reports have described successful treatment of severe IP with early laser photocoagulation. One IP case had disc neovascularization, indicating retinal ischemia, and was treated with panretinal photocoagulation, which resulted in regression of the neovascularization [11]. In another case, the avascular retina extended to the posterior pole similar to a current case and was treated with intensive laser photocoagulation, with stabilization without regrowth [12]. In the current study, especially in case 1, extremely severe retinal vascular occlusion involving the fovea developed and only anomalous retinal vasculature with vessel anastomoses in the limited area around the optic disc was observed, but it did not progress to a retinal detachment and stabilized. Even in severe cases related to retinal vascular maldevelopment in neonates, prompt and intensive laser may prevent the vision-threatening sequelae of neovascularization, fibrosis, exudation, and retinal detachment.
Wide-angle fundus examination with FA is believed to be useful for establishing the diagnosis, treatment, and follow-up of IP in infants with active retinopathy [16, 17]. FA may be essential for detecting the extent of the retinal avascularity and estimating the severity of the retinopathy. The current cases also suggest that pre-proliferative intensive laser treatment confirmed by FA imaging seems essential to avoid severe fibrovascular proliferation that leads to retinal detachment.
It is debatable whether aggressive laser treatment is needed for a non-aggressive type of retinopathy. The right eye of case 3 might have had a non-proliferative stage of retinopathy and it is doubtful whether immediate treatment was necessary. However, we have experienced several cases of infants referred to our hospital with refractory tractional retinal detachment and could not postpone laser treatment for the other five eyes which presented with active progressive retinopathy. Previously, Chen et al. [10] performed an observational cohort study of 42 female patients with IP and report that three of four eyes developed a retinal detachment despite prophylactic ablation to the nonperfused retina in the youngest group of patients (≤2.5 years). They speculate that in the youngest cases, laser or cryotherapy may have led to tractional retinal detachment resulting from contraction of the fibrovascular tissue or perhaps the patients had more severe disease and were already at high risk for a retinal detachment. The authors recommend that prophylactic ablation should be limited to patients with progression of neovascularization, vitreous traction, or vitreous hemorrhage. However, we think ablation should be performed before the proliferative stage for severe retinopathy in infants to prevent tractional retinal detachments. Jandeck et al. [9] report successful treatment of severe retinopathy in IP with laser photocoagulation and additional vitrectomy and advocate early coagulation treatment for patients with large avascular areas and beginning fibrovascular proliferation.
It has not been clarified whether aggressive laser photocoagulation extending to the posterior retina, even close to the fovea, is necessary for adequate treatment. The use of intravitreal antivascular endothelial growth factor in a case with IP is reported, but the efficacy has not been determined [18, 19]. To decrease the adverse effects in the posterior retina, adjuvant or alternative treatment to laser photocoagulation, especially for severe cases, should be considered. First and foremost, because the incidence of occlusive events peaks in the neonatal period, ophthalmologists should evaluate infants with IP soon after diagnosis. Although there was a family carrier, it was unusual that case 2 had no symptoms up to 3 months of age and the fundus examination was delayed. We recommend informing patients with IP that every female child be screened by ophthalmologists starting at the neonatal period.
It should be noted that previous IP cases with severe retinopathy are reported to have CNS complications, which suggests the close relationship of maldevelopment between the retina and brain [8, 9, 11]; MRI findings are important for screening for CNS complications. In our cases, no CNS complications developed, which indicates that severe retinopathy is not necessarily accompanied by simultaneous cerebral complications. It is unclear whether such an extensive vascular occlusion occurred during the neonatal period or whether the prominent vascular maldevelopment during the fetal period was accompanied by IP.
Foveal irregularity is an important cause of visual loss among patients with IP. Previous FA and OCT studies have reported an absent or abnormal fovea even in cases with peripheral retinopathy alone [20,21,22], discussed as a result of vascular remodeling after vascular occlusive events in the macula [20]. A recent study reports decreased vascular density, abnormal vascular loops, and flow loss in the macula detected on OCTA images [23]. In the current cases, the left eyes of cases 1 and 3 had postoperative foveal capillary abnormalities detected by OCTA. In addition, in the left eye of case 3, although the vascular occlusion occurred mainly in the peripheral retina, OCT showed a discontinuous ellipsoidal zone and decreased vascular density of the macula. However, no foveal involvement was detected in the OCT or OCTA images obtained in case 2, in which an extensive avascular area was found in the posterior pole when the patient was 5 months old. Since the retinopathy in cases 1 and 3 developed extremely early, i.e., within 3 weeks of the neonatal period, macular vessel maldevelopment may tend to occur in early infancy, when macular formation is insufficient. Further investigation is needed to clarify the association between foveal abnormalities and retinal vascular pathogenesis in cases with IP.
The current retrospective non-comparative study had several limitations. We experienced rare cases of IP especially with severe retinal vasculopathy and advocate that prompt aggressive laser photocoagulation may be needed for similar cases to avoid proliferative vitreoretinopathy; however the ideal timing and pattern of laser treatment have not been established. We applied laser photocoagulation to all avascular retinas and anomalous shunt vessels detected by FA to treat the aggressive retinopathy in cases 1 and 2 as for AP-ROP; however, performing laser near the macula may amunt to overtreatment. Preoperative OCT and OCTA images were not obtained; therefore we could not accurately evaluate the effect of the treatment on the foveal abnormalities. In addition, we determined that laser treatment similar to ROP is indicated for pre-proliferative retinopathy, but pre-threshold retinopathy in IP remains unclear without prospective randomized comparative studies. Because the pathogeneses of retinopathy of IP and ROP differ, it should be clarified in future studies if the treatment rationale for ROP is appropriate for retinopathy associated with IP.
References
Garrod AE. Peculiar pigmentation of the skin of an infant. Trans Clin Soc Lond. 1906;39:216.
Smahi A, Courtois G, Vabres P, Yamaoka S, Heuertz S, Munnich A, et al. Genomic rearrangement in NEMO impairs NF-kappaB activation and is a cause of incontinentia pigmenti. The International Incontinentia Pigmenti (IP) Consortium. Nature. 2000;405:466–77.
Goldberg MF, Custis PH. Retinal and other manifestations of incontinentia pigmenti (Bloch-Sulzberger syndrome). Ophthalmology. 1993;100:1645–54.
O’Doherty M, Mc Creery K, Green AJ, Tuwir I, Brosnahan D. Incontinentia pigmenti-ophthalmological observation of a series of cases and review of the literature. Br J Ophthalmol. 2011;95:11–6.
Swinney CC, Han DP, Karth PA. Incontinentia pigmenti: a comprehensive review and update. Ophthalmic Surg Lasers Imaging Retina. 2015;46:650–7.
Nishimura M, Oka Y, Takagi I, Yamana T, Kitano A. The clinical features and treatment of the retinopathy of Bloch-Sulzberger syndrome (incontinentia pigmenti). Jpn J Ophthalmol. 1980;24:310–9.
Ranchod TM, Trese MT. Regression of retinal neovascularization after laser photocoagulation in incontinentia pigmenti. Retina. 2010;30:708–9.
Meallet MA, Song J, Stout JT. An extreme case of retinal avascularity in a female neonate with incontinentia pigmenti. Retina. 2004;24:613–5.
Jandeck C, Kellner U, Foerster MH. Successful treatment of severe retinal vascular abnormalities in incontinentia pigmenti. Retina. 2004;24:631–3.
Chen CJ, Han IC, Tian J, Muñoz B, Goldberg MF. Extended follow-up of treated and untreated retinopathy in incontinentia pigmenti: analysis of peripheral vascular changes and incidence of retinal detachment. JAMA Ophthalmol. 2015;133:542–8.
Shah GK, Summers CG, Walsh AW, Neely KA. Optic nerve neovascularization in incontinentia pigmenti. Am J Ophthalmol. 1997;124:410–2.
Batioglu F, Ozmert E. Early indirect laser photocoagulation to induce regression of retinal vascular abnormalities in incontinentia pigmenti. Acta Ophthalmol. 2010;88:267–8.
An International Committee for the Classification of Retinopathy of Prematurity. The international classification of retinopathy of prematurity revisited. Arch Ophthalmol. 2005;123:991–9.
Yokoi T, Hiraoka M, Miyamoto M, Yokoi T, Kobayashi Y, Nishina S, et al. Vascular abnormalities in aggressive posterior retinopathy of prematurity detected by fluorescein angiography. Ophthalmology. 2009;116:1377–82.
Kenwrick S, Woffendin H, Jakins T, Shuttleworth SG, Mayer E, Greenhalgh L, et al. Survival of male patients with incontinentia pigmenti carrying a lethal mutation can be explained by somatic mosaicism or Klinefelter syndrome. Am J Hum Genet. 2001;69:1210–7.
Shaikh S, Trese MT, Archer SM. Fluorescein angiographic findings in incontinentia pigmenti. Retina. 2004;24:628–9.
DeVetten G, Ells A. Fluorescein angiographic findings in a male infant with incontinentia pigmenti. J AAPOS. 2007;11:511–2.
Lin KL, Hirose T, Kroll AJ, Lou PL, Ryan EA. Prospects for treatment of pediatric vitreoretinal diseases with vascular endothelial growth factor inhibition. Semin Ophthalmol. 2009;24:70–6.
Shah PK, Bachu S, Narendran V, Kalpana N, David J, Srinivas CR. Intravitreal bevacizumab for incontinentia pigmenti. J Pediatr Ophthalmol Strabismus. 2013;50:52–4.
Goldberg MF. Macular vasculopathy and its evolution in incontinentia pigmenti. Trans Am Ophthalmol Soc. 1998;96:55–65.
Basilius J, Young MP, Michaelis TC, Michaelis TC, Hobbs R, Jenkins G, et al. Structural abnormalities of the inner macula in incontinentia pigmenti. JAMA Ophthalmol. 2015;133:1067–72.
Mangalesh S, Chen X, Tran-Viet D, Viehland C, Freedman SF, Toth CA. Assessment of the retinal structure in children with incontinentia. Retina. 2017;37:1568–74.
Liu TYA, Han IC, Goldberg MF, Linz MO, Chen CJ, Scott AW. Multimodal retinal imaging in incontinentia pigmenti including optical coherence tomography angiography: findings from an older cohort with mild phenotype. JAMA Ophthalmol. 2018;136:467–72.
Acknowledgements
We thank the patients and their parents/guardians for participating in this study. This work was supported by grants from the National Center for Child Health and Development 28-2 and the Japan Agency for Medical Research and Development 19gk0110034h0002.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
S. Nakao, None; S. Nishina, None; S. Tanaka, None; T. Yoshida, None; T. Yokoi, None; N. Azuma, None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corresponding Author: Noriyuki Azuma
Electronic supplementary material
Below is the link to the electronic supplementary material. Supplemental Fig.1a, 1b, 1c, 1d, 3a, 3b, 3c, 3d, 4a, 4b, 4c, 4c-2, 4d are provided to show the wide angle of views.
About this article
Cite this article
Nakao, S., Nishina, S., Tanaka, S. et al. Early laser photocoagulation for extensive retinal avascularity in infants with incontinentia pigmenti. Jpn J Ophthalmol 64, 613–620 (2020). https://doi.org/10.1007/s10384-020-00768-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-020-00768-7