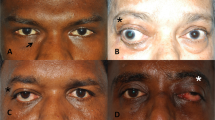
Abstract
Purpose
To describe the clinical manifestations and prognoses in 7 patients with invasive sino-orbital aspergillosis (ISOA).
Methods
This was a retrospective study of consecutive patients who were diagnosed as having ISOA at the Gifu University Hospital and Gifu Municipal Hospital between January 1993 and December 2015. Data were collected on demographics, initial manifestations, examination findings, treatments, clinical course, and outcomes.
Results
The median age of the 7 patients with ISOA was 68 years; 5 of them had diabetes. The initial symptoms were reduced blurred vision (57%), unilateral headaches (43%), unilateral abnormal sensations or numbness of the periorbital area (43%), and external ophthalmoplegia (43%). The medical department that the patients first visited was the ophthalmology department in 57% of the cases. The initial CT showed bone destruction in 71% and calcification in 14% of the patients. Six of the 7 cases were misdiagnosed. The definitive diagnosis of ISOA was made by histopathologic examinations of the biopsy specimens, with an average of 2.6 biopsies. All patients received aggressive antifungal treatments after the diagnosis. However, the final visual outcome was no light perception in 86% and death related to the ISOA in 43% of the patients. Patients who were older at the onset had lower survival rates.
Conclusions
The prognosis for patients with ISOA is poor in terms of both vision and life. Ophthalmologists are often the first examiner. ISOA should be considered in the differential diagnosis for patients with a gradually progressive orbital mass, unilateral headaches, numbness of the periorbital area, and a decrease in visual acuity of unknown origin.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Though a rare disease, the incidence of invasive sino-orbital aspergillosis (ISOA) is reportedly increasing owing to the growing number of aged individuals and immunocompromised patients who are treated with steroids or immunosuppressive drugs [1]. ISOA can progress to severe illness, with mortality rates of 40–80% [2–4].
ISOA has diverse signs and symptoms [5, 6]; the main clinical manifestations are headaches, eye and periorbital pain, facial paresthesia, blepharoptosis, visual disturbances, and ophthalmoplegia. Thus, ophthalmologists have a higher chance of being the first to examine patients with this disease than do otolaryngologists or neurosurgeons [6, 7]. Many ophthalmologists, however, are unfamiliar with the disease because of its rarity and difficulty of diagnosis. This lack of knowledge can lead to a delay in diagnosis or to misdiagnosis, which could lead to blindness and even death [4].
We report the clinical manifestations, examination findings, differential diagnoses, treatments, and outcomes of 7 patients with histopathologically confirmed ISOA.
Patients and methods
This was a retrospective study of 7 consecutive patients diagnosed as having ISOA at the Gifu University Hospital and Gifu Municipal Hospital between January 1993 and December 2015. The following information was collected from the medical records: age, sex, medical history, signs and symptoms, duration of symptoms, medical department first visited by the patient, initial visual acuity, computed tomography (CT) and magnetic resonance imaging (MRI) findings, initial diagnosis, number of biopsies, interval to definitive diagnosis, level of β-d-glucan, treatment, and outcome of vision and life. The study was approved by the institutional review board of each institute.
Results
Basic demographics
The median age of the 7 consecutive patients with localized ISOA was 68 years (range 26–76 years), and 6 of the 7 patients (85.7%) were aged over 50 years (Table 1). There were 6 men and 1 woman. Five patients (71.4%) had diabetes; 2, hypertension; 1, spinal disease; 1, stroke; and 1, hyperuricemia. Nobody suffered from diseases that led to immunodeficiency. All patients had unilateral sino-orbital aspergillosis (SOA) at the first presentation, and 3 patients (42.9%) developed bilateral lesions during the follow-up period. The diagnosis in case 3 was invasive Aspergillus sinusitis on the left side, which had been treated at another hospital 1.5 years earlier. The patient then developed it on the right side and visited our hospital. In 3 patients (42.9%), the HbA1c level was higher than the normal cut-off value of 6.5% at the initial presentation [Japan Diabetes Society (JDS)]. The medical department first visited by the patients was the ophthalmology department in 4 (57.1%), the neurosurgery department in 2 (28.6%), and the otorhinolaryngology department in 1 (14.3%). The median interval between the onset of symptoms and the first visit to our hospital was 69 days.
Initial signs and symptoms
The initial signs and symptoms were a decrease in vision in 4 (57.1%), unilateral headaches in 3 (42.9%), unilateral abnormal sensation or numbness in the periorbital area in 3 (42.9%), external ophthalmoplegia in 3 (42.9%), double vision in 2 (28.6%), and proptosis in 2 (28.6%) patients (Table 1). Additionally, 1 patient each had blurred vision, nasal congestion, staggering, and visual field abnormalities. The initial visual acuity was better than 20/30 in 5 (71.4%) and hand movements in 2 (28.6%) of the patients.
Imaging
Computed tomography (CT) and magnetic resonance imaging (MRI) were performed in all patients at the initial examination (Table 2). Five patients (71.4%) had ill-defined lesions. The initial CT findings indicated bone destruction in 5 patients (71.4%) (Fig. 1) and calcification in 1 patient (14.3%). The initial MRI examination showed hypointensity in the T1-weighted images in 1 patient (14.3%), and hypointensity in the T2-weighted images in 6 patients (85.7%) (Fig. 1). Although no CT and MRI abnormalities were detected in case 7 at the initial examination, mucosal hypertrophy in the sphenoidal sinus and soft tissue shadows in the cavernous sinus were detected 2 months later (Fig. 1). Intraorbital expansion of the lesions was confirmed in 2 (28.6%), intracranial extension in 2 (28.6%), and cavernous sinus invasion in 3 (42.9%) patients at the initial examination (Table 2).
Radiologic features in invasive sino-orbital aspergillosis. Computed tomography (left) and magnetic resonance imaging (middle and right). Case 1 A 29-year-old woman with double vision. CT (a) and T1-weighted MRI (b) showed a mass infiltrating into the left orbit and sinusitis in the ethmoid. Case 2 A 76-year-old man with temporal headache for 10 days. CT (c) and T1- and T2-weighted MRI (d, e) showed a soft tissue mass reaching the left apex. Case 3 A 67-year-old man with blurred vision for a few weeks. CT (f) and both enhanced T1- and enhanced T2-weighted MRI (g, h) showed a soft tissue mass reaching the right apex and extending into the cavernous sinus. Case 4 A 70-year-old man with temporal headache, nasal congestion, and staggering. CT (i) showed calcification in the left maxillary sinus. T1-weighted MRI (j) showed a heterogeneous mass. Enhanced T2-weighted MRI (k) showed thickening of the mucosa in the left maxillary sinus and a mass infiltrating the anterior cranial fossa. Case 5 A 57-year-old man with double vision and decrease in vision for 30 days. CT (l) and T1-weighted MRI (m) showed a homogeneous mass extending from the orbit to the anterior cranial fossa on the left. T2-weighted MRI (n) showed a hypointense mass in the muscle. Case 6 A 68-year-old man with temporal headache for a few weeks. CT (o) and T1-weighted MRI (p) showed a soft tissue mass in the left ethmoid and sphenoid sinuses. T2-weighted MRI (q) showed the mass enhanced. Case 7 A 73-year-old man with visual field abnormalities in the right eye. CT (r) and both T1- and T2-weighted MRI (s, t) showed no abnormal findings at presentation. CT (u) showed a soft tissue mass in the right sphenoid sinus 1.5 month after presentation. T1-weighted MRI showed enhancement of the sphenoid sinus on the right (v) and a hypointense mass around the right apex (w)
Initial diagnosis and treatments before definitive diagnosis
Tumor, idiopathic orbital inflammation, ocular sarcoidosis, lymphoma, bacterial sinusitis, Tolosa-Hunt syndrome, retrobulbar neuritis, and posterior ischemic optic neuropathy (PION) were considered as the initial diagnosis (Table 2). However, only 1 patient (case 4) was suspected by the radiologists to have a fungal infection because of the presence of dense calcification in the initial CT images.
The treatments before the definitive diagnosis were radiation in 2 (28.6%), systemic steroid in 4 (57.1%), and antibiotics in 2 (28.6%) patients with some overlapping treatments (Table 2).
Biopsy and definitive diagnosis
Biopsies were performed for all the patients other than the patient of case 3, and Aspergillus organisms were histopathologically detected in all the specimens (Table 3; Fig. 2). Diagnostic biopsy in case 3 was not performed at our institution because the patient had been histopathologically diagnosed as having Aspergillus sinusitis at another hospital 1.5 years earlier. The number of biopsies performed was 1 in 3 patients and more than 2 in 3 patients (average 2.6 times).
A definitive diagnosis of ISOA was made from the histopathologic examinations of the biopsy specimens except in case 3 (Table 3). Only for case 7, which was of the most recently diagnosed patient, was a polymerase chain reaction (PCR) test performed on formalin-fixed, paraffin-embedded sections to identify the Aspergillus species, and the results showed that A. fumigatus was the causative organism. Cases 1 to 6 were not identified to the level of the Aspergillus species because formalin-fixed, paraffin-embedded sections were not available. The median interval between the presentation at our institution and the time of definitive diagnosis for all the cases other than case 3 was 59 days (range 24–1018 days).
β-d-glucan and Aspergillus antigen
The serum β-d-glucan level just after the detection of Aspergillus was 45.3 to >300 pg/mL, which is higher than the standard range, in 5 of 6 patients (83.3%) (Table 2). There was no medical record of the serum β-d-glucan level in case 1. After the detection of the Aspergillus organisms, its antigen was positive in the blood of 3 of 4 patients (75%).
Treatments and outcomes
After the definitive diagnosis was made, all the patients were treated with several antifungal drugs (Table 3). The antifungal drugs used were intravenous amphotericin-B, oral itraconazole, intravenous micafungin, and intravenous or oral voriconazole. None of the patients had intraocular or intraorbital medical treatment and surgery.
The final outcome was no light perception, including phthisis bulbi, in 6 eyes (85.7%), and 20/600 in 1 eye (Table 4). There were signs of damage to the oculomotor nerve in 5 (71.4%), to the trochlear nerve in 4 (57.1%), to the trigeminal nerve in 4 (57.1%), and to the abducens nerve in 5 (71.4%) patients during the follow-up period. Five patients (71.4%) eventually had an intracranial extension of SOA; 3 of them (60%) died, and the average interval between the onset of symptoms and death was 327 days (range 187–433 days). In total, 4 of the 7 patients (57.1%) died during the follow-up period. The patient of case 3, who did not have an intracranial extension, unexpectedly died at 1555 days of follow-up, but the cause of death was not stated in the medical record. The patient of case 1 was followed for 3234 days but was lost after that. This patient also had depression and articulation disorder related to intracranial extension of Aspergillus and attempted suicide once during this period. The patients of cases 5 and 6 are still alive with no disease recurrence over 3129 and 2414 days of follow-up, respectively.
Discussion
We have presented the clinical manifestations and outcomes of 7 cases of ISOA, which is the largest number of cases among the previous reports in Japan. The reported risk factors for SOA were advanced age, diabetes mellitus, use of corticosteroids or immunosuppressive agents, hematologic malignancy, neutrophil defect, HIV infection, excessive environmental exposure to fungi, and transplantation [3, 4, 8]. In this study, 5 patients (71.4%) had diabetes mellitus and were aged over 65 years. However, none had diseases that compromise immunity.
The signs and symptoms of eyes with orbit and orbital apex SOA often precede those of the sinuses [2, 4]. Therefore, ophthalmologists have a greater chance to be the first to see patients with SOA [7]. Thurtell et al. [6] reported that 6 of 10 patients (60%) with SOA had initially visited an ophthalmologist, which is similar to our finding in 4 of 7 cases (57.1%).
The clinical manifestations of invasive fungal sinusitis including Aspergillus are varied and nonspecific [2]. The reported common signs and symptoms are proptosis [4, 6, 9], periocular swelling [9], pain located on one side of the head or retrobulbar area [1, 6, 8], disorders of eye movements [6], and decreased vision [4]. In our patients, the most common symptoms at presentation were blurred vision (4 patients [57.1%]), followed by unilateral headache (3 patients [42.9%]), unilaterally abnormal sensation or numbness of the periorbital area (3 patients [42.9%]), and external ophthalmoplegia (3 patients [42.9%]). Thus, persistent unilateral pain or abnormal sensation in the head or the retrobulbar area should alert clinicians to the possibility of SOA. According to the literature [4, 6, 10], symptoms related to sinus involvement such as rhinorrhea, epistaxis, nasal congestion, and nasal crusting are rare at presentation, which is consistent with our results.
Previous studies reported on the advantages of both CT and MRI in the diagnosis of suspected sino-orbital fungal infections (Table 5) [3–5, 11–14]. The presence of dense calcification is particularly suggestive of aspergillosis [4]. Calcifications in the CT images were reported to be present in 50% [11], 53% [12], 77% [13], and 88% [14] of patients with Aspergillus sinusitis. In the MR images of suspected aspergillosis, heterogeneous lesions and hypointense signals on both the T1- and the extremely T2-weighted images are reliable signs for this disease [3–5, 13]. However, calcification of the CT images was detected in only 1 of our patients (14%) and hypointense signals on both the T1- and the T2-weighted images were detected in another patient (14%) only. The reason for such differences in the incidence between our study and those of other studies is unknown. Although calcification in the CT images and hypointensity in the MR images are highly indicative of aspergillosis, it should be noted that their absence does not rule it out [7], as shown in our cases. Sinus aspergillosis is rare and difficult to distinguish from other similar diseases radiologically [14].
New diagnostic markers for invasive fungal infections, such as serum Aspergillus galactomannan and β-d-glucan, are reported to be useful for early diagnosis of invasive fungal infections [1, 15, 16]. In our study, the serum β-d-glucan level exceeded the normal limits in 83.3% of the patients. Thus, the level of serum β-d-glucan should be determined for patients having the early signs and symptoms of aspergillosis: unilateral and persistent headache, periorbital pain, facial numbness, ptosis, and disorders of ocular movements of unknown origin.
A definitive diagnosis of SOA is ultimately made by histopathologic examination of biopsy specimens with support from the clinical findings and CT and MR images. However, Dhiwakar et al reported a sensitivity of 33% for incisional biopsy from the paranasal sinuses [10]. Six of our patients with an average of 2.6 biopsies were finally diagnosed as having aspergillosis by the histopathologic findings. Similar to a previous report [10], only 3 of these patients (50.0%) were diagnosed after the first biopsy, and the others required repeated biopsies. The necessity of repeated biopsies may be due to 3 problems: the staining method, preconceptions, and sampling errors. Fungi including Aspergillus are frequently not stained with hematoxylin-eosin (HE), and special stains such as periodic acid-Schiff (PAS) are better in making them visible [4, 5, 7, 8, 17]. In case 1, the first to sixth biopsy specimens were stained with HE, and that of the seventh was stained with HE, PAS, and Grocott stains. Every section of the first to sixth biopsies revealed some abnormalities such as dense hyaline connective tissue with foreign body giant cells, fibrous connective tissue with inflammatory cell infiltration, and loose connective tissues without cell components. A definitive diagnosis of aspergillosis was made by the seventh biopsy specimen stained with PAS and Grocott staining, although the report of the seventh biopsy sections stained with HE were similar to those of the first to sixth biopsies. Aspergillus is generally considered to be a harmless saprophyte that is ubiquitous in our environment [18]. In case 2, Aspergillus organisms were detected on the first biopsy but were considered normal flora. The samples were reported to be inflammatory responses, and such preconceptions probably led to the misdiagnosis. Biopsies were taken 3 times in case 5, and all of the samples were stained with PAS. The histopathologic changes of the specimens were concluded to be tissue inflammatory responses, and Aspergillus organisms were finally detected on the fourth biopsy. This could probably be classified as a sampling error. PCR is also valuable in cases in which either the culture results are negative or culturing is not performed [19, 20]. In case 7, the most recent case, PCR was performed using formalin-fixed, paraffin-embedded sections, and A. fumigatus was detected.
SOA is often misdiagnosed as being a variety of diseases, such as optic neuritis, temporal arteritis, bacterial cellulitis, malignant tumor, lymphoma, idiopathic orbital inflammation, and the Tolosa-Hunt syndrome. Thus, it is often treated with steroids and radiation [4–6, 8]. In our study, 4 patients were initially treated with systemic steroids for presumed idiopathic orbital inflammation and/or Tolosa-Hunt syndrome, and 2 of 3 patients were misdiagnosed as having a malignant tumor and underwent radiation therapy. We recommend that biopsies be done to identify the causative organism before the use of steroids or radiation.
Thurtell et al. [6, 21] reported that amphotericin B is considered the gold standard agent to treat SOA; however, its use is limited because of the associated complications, such as nephrotoxicity. Itraconazole and voriconazole have now replaced amphotericin B. However, 4 of the 7 patients in our study treated with combinations of amphotericin B, itraconazole, and/or voriconazole eventually died, and 3 of these 4 patients died of intracranial extension. Before the introduction of antifungal therapy, these 3 patients received systemic steroid therapy for presumed inflammatory disease. The progression of the signs and symptoms should be carefully monitored during such empirical treatments. Even though antifungal agents are used, changes in the symptoms should be carefully monitored because of the increase in the number of fungi having low susceptibility to antifungal drugs [22, 23] and the problem of the penetration of antifungal drugs through the blood–brain barrier [2]. Newer antifungal agents are needed to control this severe infection. In 2016, Aggarwal et al. [21] reported that no definite treatment protocols existed for orbital aspergillosis, but a complete surgical debridement was recommended as the primary treatment in patients with orbital aspergillosis [21]. Involvement of the bone, blood vessels, and orbital structures, however, limits this approach. Radical surgical debridement of the sinuses and skull base area is rarely performed in Japan, unlike in advanced Western countries. Surgical intervention treatment might have rescued some of our patients.
Invasive fungal sinusitis has a very poor prognosis for both vision and life. The rate of permanent loss of light perception was reported to be 80% [24] and 100% [6]. The death rate was reported to be 50% [1], 60% [6], and 71% [8]. In our study, the rate of loss of light perception was 86% (6 of 7 eyes). The cause of the sudden death in case 3 was not known. Death related to SOA occurred in 3 of our 7 patients (43%). Differences between the deceased patients (cases 2, 4, and 7) and the surviving patients (cases 1, 5, and 6) in terms of the medical history or presence or absence of intracranial invasion were not observed, but the age at onset (median age, 73 vs 57 years) clearly differed between these 2 groups. Patients with older age at onset had a higher mortality rate, which is similar to previous reports [4, 9].
To conclude, ISOA has nonspecific clinical manifestations and poor prognoses for both vision and life. Diabetes mellitus and onset at age >65 years were risk factors for ISOA, and older age at onset may have a poor prognosis. Histopathologic examinations by repeated biopsies with support from the clinical findings and CT and MR images are necessary for a definitive diagnosis. Ophthalmologists are often the first to examine these patients, and ISOA should be considered in the differential diagnoses of patients with a gradually progressive orbital mass, unilateral persistent headache, unilateral numbness of the periorbital area, and decrease in visual acuity of unknown origin.
References
Lee DH, Yoon TM, Lee JK, Joo YE, Park KH, Lim SC. Invasive fungal sinusitis of the sphenoid sinus. Clin Exp Otorhinolaryngol. 2014;7:181–7.
Arndt S, Aschendorff A, Echternach M, Daemmrich TD, Maier W. Rhino-orbital-cerebral mucormycosis and aspergillosis: differential diagnosis and treatment. Eur Arch Otorhinolaryngol. 2009;266:71–6.
Johnson TE, Casiano RR, Kronish JW, Tse DT, Meldrum M, Chang W. Sino-orbital aspergillosis in acquired immunodeficiency syndrome. Arch Ophthalmol. 1999;117:57–64.
Pushker N, Meel R, Kashyap S, Bajaj MS, Sen S. Invasive aspergillosis of orbit in immunocompetent patients: treatment and outcome. Ophthalmology. 2011;118:1886–91.
Mauriello JA Jr, Yepez N, Mostafavi R, Barofsky J, Kapila R, Baredes S, et al. Invasive rhinosino-orbital aspergillosis with precipitous visual loss. Can J Ophthalmol. 1995;30:124–30.
Thurtell MJ, Chiu AL, Goold LA, Akdal G, Crompton JL, Ahmed R, et al. Neuro-ophthalmology of invasive fungal sinusitis: 14 consecutive patients and a review of the literature. Clin Exp Ophthalmol. 2013;41:567–76.
Levin LA, Avery R, Shore JW, Woog JJ, Baker AS. The spectrum of orbital aspergillosis: a clinicopathological review. Surv Ophthalmol. 1996;41:142–54.
Sivak-Callcott JA, Livesley N, Nugent RA, Rasmussen SL, Saeed P, Rootman J. Localized invasive sino-orbital aspergillosis: characteristic features. Br J Ophthalmol. 2004;88:681–7.
Mody KH, Ali MJ, Vemuganti GK, Nalamada S, Naik MN, Honavar SG. Orbital aspergillosis in immunocompetent patients. Br J Ophthalmol. 2014;98:1379–84.
Dhiwakar M, Thakar A, Bahadur S. Invasive sino-orbital aspergillosis: surgical decisions and dilemmas. J Laryngol Otol. 2003;117:280–5.
Kopp W, Fotter R, Steiner H, Beaufort F, Stammberger H. Aspergillosis of the paranasal sinuses. Radiology. 1985;156:715–6.
Thery A, Espitalier F, Cassagnau E, Durand N, Malard O. Clinical features and outcome of sphenoid sinus aspergillosis: a retrospective series of 15 cases. Eur Ann Otorhinolaryngol Head Neck Dis. 2012;129:179–84.
Chang T, Teng MM, Wang SF, Li WY, Cheng CC, Lirng JF. Aspergillosis of the paranasal sinuses. Neuroradiology. 1992;34:520–3.
Patel PJ, Kolawole TM, Malabarey TM, Hulailah A, Hamid F, Chakaki M. CT findings in paranasal aspergillosis. Clin Radiol. 1992;45:319–21.
Nakanishi W, Fujishiro Y, Nishimura S, Fukaya T. Clinical significance of (1→3)-beta-d-glucan in a patient with invasive sino-orbital aspergillosis. Auris Nasus Larynx. 2009;36:224–7.
Takahashi H, Hinohira Y, Hato N, Wakisaka H, Hyodo J, Ugumori T, et al. Clinical features and outcomes of four patients with invasive fungal sinusitis. Auris Nasus Larynx. 2011;38:289–94.
Procop GW, Wilson M. Infectious disease pathology. Clin Infect Dis. 2001;32:1589–601.
Glass RB, Hertzanu Y, Mendelsohn DB, Posen J. Paranasal sinus aspergillosis: a case report with computed tomogram findings. J Laryngol Otol. 1984;98:199–205.
Lau A, Chen S, Sorrell T, Carter D, Malik R, Martin P, et al. Development and clinical application of a panfungal PCR assay to detect and identify fungal DNA in tissue specimens. J Clin Microbiol. 2007;45:380–5.
Paterson PJ, Seaton S, McLaughlin J, Kibbler CC. Development of molecular methods for the identification of Aspergillus and emerging moulds in paraffin wax embedded tissue sections. Mol Pathol. 2003;56:368–70.
Aggarwal E, Mulay K, Menon V, Sundar G, Honavar SG, Sharma M. Isolated orbital aspergillosis in immunocompetent patients: a multicenter study. Am J Ophthalmol. 2016;165:125–32.
Alastruey-Izquierdo A, Melhem MS, Bonfietti LX, Rodriguez-Tudela JL. Susceptibility test for fungi: clinical and laboratorial correlations in medical mycology. Rev Inst Med Trop Sao Paulo. 2015;57(suppl 19):57–64.
Kaur S, Singh S. Biofilm formation by Aspergillus fumigatus. Med Mycol. 2014;52:2–9.
Choi HS, Choi JY, Yoon JS, Kim SJ, Lee SY. Clinical characteristics and prognosis of orbital invasive aspergillosis. Ophthal Plast Reconstr Surg. 2008;24:454–9.
Acknowledgements
We thank Professor Duco Hamasaki for editing this manuscript.
Conflicts of interest
H. Kawakami, None; K. Mochizuki, None; K. Ishida, None; K. Ohkusu, None.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kawakami, H., Mochizuki, K., Ishida, K. et al. Seven cases of localized invasive sino-orbital aspergillosis. Jpn J Ophthalmol 61, 179–188 (2017). https://doi.org/10.1007/s10384-016-0494-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-016-0494-5