Summary
Iron deficiency and anaemia are common in colorectal cancer. Replacement with oral or intravenous iron effectively treats this deficiency. However, mechanistic and population studies suggest that excess iron promotes colorectal carcinogenesis. Growing research into gut microbiota and dysbiosis suggests one explanation for this association. Iron is growth limiting for many pathogenic bacteria and may promote a shift in the ratio of pathogenic to protective bacteria. This may increase the toxic bacterial metabolites, promoting inflammation and carcinogenesis. This has important implications as we seek to correct anaemia in our patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
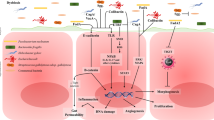
Iron deficiency and iron excess both have important implications for colorectal cancer (CRC). Iron is an essential trace element, most commonly recognised for its function when incorporated into haem proteins to form haemoglobin. However, it is found in all cells and involved in many cellular processes, acting as a cofactor for enzymes, in iron-sulphur clusters, haem groups (haemoglobin, myoglobin) and in cytochromes essential for oxidative phosphorylation in mitochondria [1]. Despite this ubiquity, excess iron is highly toxic and judicious homeostasis is essential to protect the human body from the damaging oxidative effects of iron (see Fig. 4) [2].
Iron deficiency
Iron deficiency in colorectal cancer results from an imbalance of absorption versus demand for iron. This results in the depletion of iron stores and finally an iron-deficient state. 60 % of patients with colorectal cancer have iron deficiency at presentation [3].
The main mechanisms for development of iron deficiency in colorectal cancer are pathological blood loss from gastrointestinal bleeding due to the cancer and functional iron deficiency, whereby normal iron stores cannot be mobilised due to hepcidin-mediated iron sequestration and restriction of iron induced by chronic inflammation [4, 5].
Clinically this manifests as iron deficiency anaemia and for those undergoing surgery, this anaemia increases mortality, morbidity, length of stay and bloods transfusions [6–8]. Further, blood transfusions are themselves an independent risk factor for mortality, morbidity and cancer recurrence [9, 10]. These deleterious outcomes reinforce the need to correct iron deficiency in order to reduce these risks. This can be achieved with oral (enteral) or intravenous (parenteral) iron, although concerns exist whether iron supplementation may in fact be harmful.
Iron excess
From the 1980s onwards, a link between both dietary iron and body iron stores causing colorectal cancer had been proposed [2, 11–13]. Meat consumption (especially red meat and processed meat) has been associated with colorectal cancer [14].
Stevens et al. looked at a population of 14,000 adults from 1971 that were followed up for between 10 and 13 years. They found that total iron binding capacity was lower and transferrin saturation significantly higher in men that developed cancer, suggesting high body iron stores increased cancer risk. No significant difference was found in women [11].
Specifically in colorectal cancer, Nelson performed a systematic review of 33 human studies examining iron exposure and colorectal cancer risk. He concluded that three quarters of studies support the association of iron and increased colorectal cancer risk. However, it is not clear whether excess dietary iron (and hence bioavailable iron in the lumen of the gut) or excess total body iron stores are most implicated in the apparent increase in CRC [15].
Conversely, reduction in iron can decrease cancer risk. As early as 1963, Weinberg proposed that the body restricts iron from neoplastic cells in a similar way to microbial infection [16]. In 2008, Edgren et al. examined the effect of repeated blood donation (and hence iron loss) on cancer risk. They demonstrated that iron loss of > 2.7 g compared to iron loss of < 0.75 g decreased cancer risk (combined odds ratio, OR = 0.70, 95 % confidence interval, CI = 0.58 to 0.84) [17].
Further evidence linking molecular changes in iron transport and colorectal cancer exists. Brookes et al. [18] investigated iron transport in human colorectal cancer. They demonstrated increased expression of the iron import proteins duodenal cytochrome b (DCYTB), divalent metal transporter 1 (DMT1) and transferrin receptor (TfR1; Fig. 1 and 2). Furthermore, iron export was reduced due to decreased expression and abnormal localisation of hephaestin (HEPH) and ferroportin-1 (FPN; [18]; Fig. 3). The net effect of these changes would increase intracellular iron in tumour cells.
Iron loading also increases Wnt-signalling, a major oncogenic pathway in colorectal cancer cell line models. However, this only occurs in mutant APC cell lines (Caco-2 and SW480), while wild-type APC cell lines (HEK-293 and human primary fibroblasts) show no increase [19]. Further, in murine models depletion of luminal iron reduces intestinal tumorigenesis and, conversely, increasing luminal iron strongly promotes tumorigenesis. Of particular importance, systemic iron replacement with an iron-depleted diet did not promote tumorigenesis [20]. Murine ulcerative colitis models similarly showed no increase in colon carcinogenesis with systemic iron supplementation when compared to iron-enriched diets [21, 22]. In humans, healthy volunteers given oral iron supplementation showed a 40 % increase in free radical production in faeces [23].
Transferrin receptor 1 (TfR1) immunohistochemistry (Thermo Scientific [USA], Biorbyt [UK], Novus biologicals [USA] #13-6800 TfR1 mouse Ab 1:200). a Normal human colonic mucosa demonstrating luminal apical membranous staining for TfR1 (black arrow). b Human colorectal adenocarcinoma demonstrating overexpression and mis-localisation of TfR1 (more intense cytoplasmic staining with no propensity for the apical membrane; © Oliver Ng, Medical University of Vienna, 2016)
Divalent metal transporter 1 (DMT1) immunohistochemistry (Biorbyt, UK #orb5976 DMT1 rabbit Ab 1:250) a Normal human colonic mucosa demonstrating luminal apical membranous staining for DMT1 (black arrow) b Human colorectal adenocarcinoma demonstrating overexpression and mis-localisation of DMT1 (diffuse cytoplasmic staining with no propensity for the apical membrane; © Oliver Ng, Medical University of Vienna, 2016)
Ferroportin (FPN) immunohistochemistry (Novus biologicals [USA] NBPI 21502 rabbit Ab 1:500). a Normal human colonic mucosa demonstrating cytoplasmic and strong basolateral membranous staining for FPN (black arrow). b Human colorectal adenocarcinoma demonstrating and mis-localisation of FPN from the basolateral membrane (© Oliver Ng, Medical University of Vienna, 2016)
Iron and microbiota
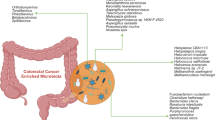
One theory for why iron increases colorectal cancer risk is that unabsorbed dietary iron promotes disruption of the gut microbiota (dysbiosis) causing a shift in the ratio of protective to pathogenic bacteria [24].
Research in this field has increased during the last 10 years, largely driven by advances in DNA sequencing (especially of highly conserved hypervariable regions of the 16 S rRNA genes in bacteria), which, combined with real-time quantitative PCR techniques, allows characterisation and quantification of microbiota biodiversity [25, 26].
Specific bacteria have been implicated in carcinogenesis, including Streptococcus bovis, Bacteroides, Enterococcus faecalis and Clostridia [27–30]. These bacteria produce genotoxic metabolites, such as hydrogen sulphide and secondary bile salts, which likely promote inflammation and carcinogenesis [31, 32].
In contrast, gut protective Bifidobacterium longum and Lactobacillus acidophilus appear to inhibit carcinogenesis by forming a protective barrier against colonisation by pathogenic bacteria [33, 34]. Bifidobacteriaceae bind iron to their surface reducing free radical formation and bioavailable iron for pathogenic bacteria [35]. Lactobacilli also appear to reduce the mutagenic effects of bile acids [36]. Butyrate production by these bacteria is also thought to be anti-carcinogenic and may be altered by dysbiosis [37].
Eradication of bacteria with antibiotic treatment or breeding germ free mice in colitis-associated cancer models significantly decreases the incidence of colon cancer and alters gut microbiota [38, 39].
Iron is essential for growth, virulence and ability to colonise the gut for many of the pathogenic bacterial species [40]. Bacteria dependent on iron have iron-binding siderophores that enable them to absorb free iron or scavenge iron from haemoglobin or transferrin. Importantly, the protective Lactobacilli bacteria do not have siderophores and grow independently of iron, instead requiring manganese for growth [41].
As a response to infection, iron-withholding strategies (mediated through hepcidin) play a key role in host defence [42]. Described as hypoferraemia of inflammation, this sequestration of iron in macrophages denies iron to invading microorganisms [43].
Anaemic African children supplemented with iron demonstrate an increase in the ratio of pathogenic enterobacteria compared to protective Bifidobacteria and Lactobacilli, with an associated increase in inflammation [44, 45].
Further support for this theory is found in in vitro and animal studies. Low-iron in vitro cultures of faecal microbiota favour the growth of Lactobacillus species and a decrease in Clostridium and Bacteroides species [46]. Similar findings were seen in rat’s gut microbiota with iron-deficient and replete diets [47]. Mice fed iron-deprived diets also increased the population of Lactobacilli in the gut [48].
Genetic modification of iron metabolism genes in mice to increase (iron regulatory protein 2 ) or decrease (hereditary haemachromatosis Hfe) iron resulted in an abundance of beneficial Lactobacillus species with IRP-2 knockouts and a predominant Enterococcus faecium species in Hfe-/- mice [49].
The route of iron supplementation also appears to be important. Werner et al. used a Crohn’s disease-like ileitis mouse model and gave either iron sulphate-containing diet or an iron-free diet, with or without iron injections, for 11 weeks. They showed that an iron sulphate-free diet prevented development of Crohn’s ileitis through alteration of the composition of microbiota in the gut. Importantly, systemic iron repletion via injections did not abrogate the protective effect of luminal iron depletion [50].
These findings have recently been demonstrated in humans as well. In a study that randomised inflammatory bowel disease patients to oral or intravenous iron therapy for anaemia, those receiving oral iron therapy showed a significant decrease in the abundance of bacterial communities (especially in Crohn’s disease) when compared with intravenous iron [51].
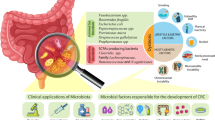
Clinical relevance
It is likely, that colonic microbiota and iron are only a part of a more complex interplay of diet, immunology and iron biology that play a role in colorectal carcinogenesis.
However, this acknowledgement that iron is proinflammatory has already gained recognition within gastroenterology and the treatment of inflammatory bowel disease with oral iron is not recommended in active disease [52].
For surgeons and anaesthetists looking to optimise their patient’s haemoglobin and correct iron deficiency anaemia prior to surgery, we should now be considering the harmful effects of high-dose oral iron. Instead, we should be considering more bioavailable low-dose preparations of oral iron or giving intravenous iron that avoids the gut altogether. Would this avoid the harmful imbalance of colonic microbiota that research suggests occurs when luminal iron is abundant?
References
Beard JL, Dawson H, Pinero DJ. Iron metabolism: a comprehensive review. Nutr Rev. 1996;54(10):295–317.
Huang X. Iron overload and its association with cancer risk in humans: evidence for iron as a carcinogenic metal. Mutat Res. 2003;533(1–2):153–71.
Beale AL, Penney MD, Allison MC. The prevalence of iron deficiency among patients presenting with colorectal cancer. Colorectal Dis. 2005;7(4):398–402.
Sadahiro S, Suzuki T, Tokunaga N, Mukai M, Tajima T, Makuuchi H, Saito T. Anemia in patients with colorectal cancer. J Gastroenterol. 1998;33(4):488–94.
Little JBE, Gardner L. Anemia of chronic disease. In: Hoffman RBE, Silberstein L, Heslop H, Weitz J, Anastasi J, editors. Hematology: basic principles and practice, 6th edn. Philadelphia: Elsevier Saunders; 2013. pp. 450–6.
Carson JL, Duff A, Poses RM, Berlin JA, Spence RK, Trout R, Noveck H, Strom BL. Effect of anaemia and cardiovascular disease on surgical mortality and morbidity. Lancet. 1996;348(9034):1055–60.
Beattie WS, Karkouti K, Wijeysundera DN, Tait G. Risk associated with preoperative anemia in noncardiac surgery: a single-center cohort study. Anesthesiology. 2009;110(3):574–81.
Leichtle SW, Mouawad NJ, Lampman R, Singal B, Cleary RK. Does preoperative anemia adversely affect colon and rectal surgery outcomes? J Am Coll Surg. 2011;212(2):187–94.
Cata JP, Wang H, Gottumukkala V, Reuben J, Sessler DI. Inflammatory response, immunosuppression, and cancer recurrence after perioperative blood transfusions. Br J Anaesth. 2013;110(5):690–701.
Bernard AC, Davenport DL, Chang PK, Vaughan TB, Zwischenberger JB. Intraoperative transfusion of 1U to 2U packed red blood cells is associated with increased 30-day mortality, surgical-site infection, pneumonia, and sepsis in general surgery patients. J Am Coll Surg. 2009;208(5):931–937.e2, discussion 938–939 doi:10.1016/j.jamcollsurg.2008.11.019.
Stevens RG, Jones DY, Micozzi MS, Taylor PR. Body iron stores and the risk of cancer. N Engl J Med. 1988;319(16):1047–52.
Nelson RL, Davis FG, Sutter E, Sobin LH, Kikendall JW, Bowen P. Body iron stores and risk of colonic neoplasia. J Natl Cancer Inst. 1994;86(6):455–60.
Wurzelmann JI, Silver A, Schreinemachers DM, Sandler RS, Everson RB. Iron intake and the risk of colorectal cancer. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive. Oncology. 1996;5(7):503–7.
Chao A, Thun MJ, Connell CJ, McCullough ML, Jacobs EJ, Flanders WD, Rodriguez C, Sinha R, Calle EE. Meat consumption and risk of colorectal cancer. JAMA. 2005;293(2):172–82.
Nelson RL. Iron and colorectal cancer risk: human studies. Nutr Rev. 2001;59(5):140–8.
Weinberg ED. Iron in neoplastic disease. Nutr Cancer. 1983;4(3):223–33.
Edgren G, Reilly M, Hjalgrim H, Tran TN, Rostgaard K, Adami J, Titlestad K, Shanwell A, Melbye M, Nyren O. Donation frequency, iron loss, and risk of cancer among blood donors. J Natl Cancer Inst. 2008;100(8):572–9.
Brookes MJ, Hughes S, Turner FE, Reynolds G, Sharma N, Ismail T, Berx G, McKie AT, Hotchin N, Anderson GJ, et al. Modulation of iron transport proteins in human colorectal carcinogenesis. Gut. 2006;55(10):1449–60.
Brookes MJ, Boult J, Roberts K, Cooper BT, Hotchin NA, Matthews G, Iqbal T, Tselepis C. A role for iron in Wnt signalling. Oncogene. 2008;27(7):966–75.
Radulescu S, Brookes MJ, Salgueiro P, Ridgway RA, McGhee E, Anderson K, Ford SJ, Stones DH, Iqbal TH, Tselepis C, et al. Luminal iron levels govern intestinal tumorigenesis after Apc loss in vivo. Cell Rep. 2012;2(2):270–82.
Seril DN, Liao J, Ho KL, Warsi A, Yang CS, Yang GY. Dietary iron supplementation enhances DSS-induced colitis and associated colorectal carcinoma development in mice. Dig Dis Sci. 2002;47(6):1266–78.
Seril DN, Liao J, Yang CS, Yang GY. Systemic iron supplementation replenishes iron stores without enhancing colon carcinogenesis in murine models of ulcerative colitis: comparison with iron-enriched diet. Dig Dis Sci. 2005;50(4):696–707.
Lund EK, Wharf SG, Fairweather-Tait SJ, Johnson IT. Oral ferrous sulfate supplements increase the free radical-generating capacity of feces from healthy volunteers. Am J Clin Nutr. 1999;69(2):250–5.
Zhu Q, Gao R, Wu W, Qin H. The role of gut microbiota in the pathogenesis of colorectal cancer. Tumour Biol. 2013;34(3):1285–300.
Matsuki T, Watanabe K, Fujimoto J, Takada T, Tanaka R. Use of 16S rRNA gene-targeted group-specific primers for real-time PCR analysis of predominant bacteria in human feces. Appl Environ Microbiol. 2004;70(12):7220–8.
Ott SJ, Musfeldt M, Ullmann U, Hampe J, Schreiber S. Quantification of intestinal bacterial populations by real-time PCR with a universal primer set and minor groove binder probes: a global approach to the enteric flora. J Clin Microbiol. 2004;42(6):2566–72.
Sobhani I, Tap J, Roudot-Thoraval F, Roperch JP, Letulle S, Langella P, Corthier G, Tran Van Nhieu J, Furet JP. Microbial dysbiosis in colorectal cancer (CRC) patients. PLOS ONE. 2011;6(1):e16393.
Gold JS, Bayar S, Salem RR. Association of Streptococcus bovis bacteremia with colonic neoplasia and extracolonic malignancy. Arch Surg. 2004;139(7):760–5.
Moore WE, Moore LH. Intestinal floras of populations that have a high risk of colon cancer. Appl Environ Microbiol. 1995;61(9):3202–7.
Huycke MM, Gaskins HR. Commensal bacteria, redox stress, and colorectal cancer: mechanisms and models. Exp Biol Med. 2004;229(7):586–97.
Nicholson JK, Holmes E, Kinross J, Burcelin R, Gibson G, Jia W, Pettersson S. Host-gut microbiota metabolic interactions. Science. 2012;336(6086):1262–7.
O’Keefe SJ, Chung D, Mahmoud N, Sepulveda AR, Manafe M, Arch J, Adada H, van der Merwe T. Why do African Americans get more colon cancer than Native Africans? J Nutr. 2007;137(1 Suppl):175S–182S.
Rowland IR, Bearne CA, Fischer R, Pool-Zobel BL. The effect of lactulose on DNA damage induced by DMH in the colon of human flora-associated rats. Nutr Cancer. 1996;26(1):37–47.
McIntosh GH, Royle PJ, Playne MJ. A probiotic strain of L. acidophilus reduces DMH-induced large intestinal tumors in male Sprague-Dawley rats. Nutr Cancer. 1999;35(2):153–9.
Kot E, Bezkorovainy A. Binding of ferric iron to the cell walls and membranes of Bifidobacterium thermophilum: effect of free radicals. J Agric Food Chem. 1999;47(11):4606–10.
Lidbeck A, Nord CE, Gustafsson JA, Rafter J. Lactobacilli, anticarcinogenic activities and human intestinal microflora. Eur J Cancer Prev. 1992;1(5):341–53.
Vipperla K, O’Keefe SJ. The microbiota and its metabolites in colonic mucosal health and cancer risk. Nutr Clin Pract. 2012;27(5):624–35.
Klimesova K, Kverka M, Zakostelska Z, Hudcovic T, Hrncir T, Stepankova R, Rossmann P, Ridl J, Kostovcik M, Mrazek J, et al. Altered gut microbiota promotes colitis-associated cancer in IL-1 receptor-associated kinase M‑deficient mice. Inflamm Bowel Dis. 2013;19(6):1266–77.
Engle SJ, Ormsby I, Pawlowski S, Boivin GP, Croft J, Balish E, Doetschman T. Elimination of colon cancer in germ-free transforming growth factor beta 1‑deficient mice. Cancer Res. 2002;62(22):6362–6.
Andrews SC, Robinson AK, Rodriguez-Quinones F. Bacterial iron homeostasis. FEMS Microbiol Rev. 2003;27(2–3):215–37.
Weinberg ED. The Lactobacillus anomaly: total iron abstinence. Perspect Biol Med. 1997;40(4):578–83.
Nemeth E, Rivera S, Gabayan V, Keller C, Taudorf S, Pedersen BK, Ganz T. IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J Clin Invest. 2004;113(9):1271–6.
Ganz T, Nemeth E. Iron homeostasis in host defence and inflammation. Nat Rev Immunol. 2015;15(8):500–10.
Zimmermann MB, Chassard C, Rohner F, N’Goran EK, Nindjin C, Dostal A, Utzinger J, Ghattas H, Lacroix C, Hurrell RF. The effects of iron fortification on the gut microbiota in African children: a randomized controlled trial in Cote d’Ivoire. Am J Clin Nutr. 2010;92(6):1406–15.
Jaeggi T, Kortman GA, Moretti D, Chassard C, Holding P, Dostal A, Boekhorst J, Timmerman HM, Swinkels DW, Tjalsma H, et al. Iron fortification adversely affects the gut microbiome, increases pathogen abundance and induces intestinal inflammation in Kenyan infants. Gut. 2015;64(5):731–42.
Dostal A, Fehlbaum S, Chassard C, Zimmermann MB, Lacroix C. Low iron availability in continuous in vitro colonic fermentations induces strong dysbiosis of the child gut microbial consortium and a decrease in main metabolites. FEMS Microbiol Ecol. 2013;83(1):161–75.
Dostal A, Chassard C, Hilty FM, Zimmermann MB, Jaeggi T, Rossi S, Lacroix C. Iron depletion and repletion with ferrous sulfate or electrolytic iron modifies the composition and metabolic activity of the gut microbiota in rats. J Nutr. 2012;142(2):271–7.
Tompkins GR, O’Dell NL, Bryson IT, Pennington CB. The effects of dietary ferric iron and iron deprivation on the bacterial composition of the mouse intestine. Curr Microbiol. 2001;43(1):38–42.
Buhnik-Rosenblau K, Moshe-Belizowski S, Danin-Poleg Y, Meyron-Holtz EG. Genetic modification of iron metabolism in mice affects the gut microbiota. Biometals. 2012;25(5):883–92.
Werner T, Wagner SJ, Martinez I, Walter J, Chang JS, Clavel T, Kisling S, Schuemann K, Haller D. Depletion of luminal iron alters the gut microbiota and prevents Crohn’s disease-like ileitis. Gut. 2011;60(3):325–33.
Lee T, Clavel T, Smirnov K, Schmidt A, Lagkouvardos I, Walker A, Lucio M, Michalke B, Schmitt-Kopplin P, Fedorak R, et al. Oral versus intravenous iron replacement therapy distinctly alters the gut microbiota and metabolome in patients with IBD. Gut. 2016; doi:10.1136/gutjnl-2015-309940.
Dignass AU, Gasche C, Bettenworth D, Birgegard G, Danese S, Gisbert JP, Gomollon F, Iqbal T, Katsanos K, Koutroubakis I, et al. European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases. J Crohns Colitis. 2015;9(3):211–22.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
O. Ng declares that he has no competing interests.
Ethical standards
This article does not contain any studies with human participants or animals performed by the author.
Rights and permissions
About this article
Cite this article
Ng, O. Iron, microbiota and colorectal cancer. Wien Med Wochenschr 166, 431–436 (2016). https://doi.org/10.1007/s10354-016-0508-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10354-016-0508-4