Abstract
A remarkable diversity of bioerosion trace fossils is reflected by the plethora of ichnotaxa that has been proposed for these structures during the past two centuries. Bioerosion traces include microborings, macroborings, grazing traces, attachment etchings, and predation traces. They occur in calcareous, siliceous, osteic, and xylic substrates, and are known or interpreted to be produced by tracemakers as diverse as bacteria, fungi, algae, invertebrates, and vertebrates. This review presents the status quo of an inventory of all bioerosion ichnotaxa currently recognized as valid, comprising 123 ichnogenera and 339 ichnospecies, including 45 combinationes novae, the majority of which on account of fossil sponge bioerosion traces formerly grouped within the sponge biotaxon Cliona. In addition, the spelling of several ichnotaxa has to be corrected, leading to eight nomina corrigenda, and three cases of primary or secondary homonymy require establishing nomina nova, i.e., the new ichnogenus name Irhopalia replacing Rhopalia Radtke, 1991, as well as the new ichnospecies names Entobia morrisi replacing E. glomerata (Morris, 1851) and Entobia tuberculata replacing E. mammillata Bromley and D’Alessandro, 1984, respectively. Ichnotaxa of dubious or invalid nomenclatural status currently include an additional 76 ichnogenera and 157 ichnospecies. The invalid ichnogenus Ipites is herein reinstated as new ichnogenus. Considering that only four valid (and one invalid) ichnofamilies had previously been established for bioerosion ichnotaxa, we here introduce a suite of 14 additional ichnofamilies: Gastrochaenolitidae, Talpinidae, Entobiaidae, Planobulidae, Ichnoreticulinidae, Saccomorphidae, Centrichnidae, Renichnidae, Podichnidae, Gnathichnidae, Circolitidae, Oichnidae, Belichnidae, and Machichnidae. During the past five decades, the number of valid bioerosion ichnotaxa has more than quadrupled, reflecting a boost in bioerosion research, but also indicating the need for ichnotaxonomic consolidation in concert with a revision of key ichnogenera. In this context, the aim of this overview is to call for feedback from the research community in order to foster completeness of this list and to provide ichnotaxonomic stability. Furthermore, we want to raise awareness of the existence of the listed ichnotaxa, many of which obviously have remained unconsidered or forgotten for a long time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Structures resulting from bioerosion of calcareous, siliceous, osteic, and xylic hard substrates classify as trace fossils and comprise various microborings, macroborings, grazing traces, attachment etchings and predation traces (e.g., Neumann 1966; Bromley 1994; Wisshak and Tapanila 2008; Tribollet et al. 2011). They are the work of a wide range of organisms across kingdoms and scales, and include traces made by bacteria, fungi, algae, invertebrates, and vertebrates. Trace fossils are named using a conceptual framework referred to as ichnotaxonomy, based on the Linnaean binominal nomenclature, with their names being governed by the ‘International Code of Zoological Nomenclature’ (International Commission on Zoological Nomenclature, ICZN 1999, in its current 4th edition; online version partially updated). Even though bioerosion ichnotaxa include the work not only of animals (explicitly protected by the ICZN), but also of plants, fungi, and bacteria (whose nomenclatural codes do not recognize trace fossils), they are commonly named within the framework of the ICZN as if they were protected (Bertling et al. 2004; Rindsberg 2012). Concepts and reviews on ichnotaxonomic principles, such as the validity and hierarchy of suitable ichnotaxobases, were provided, for instance by Pickerill (1994), Bertling et al. (2006), and Bertling (2007). Overview papers on trace-fossil systematics and the history of ichnotaxonomy were given by Magwood (1992), Bromley (1996), Knaust (2012), and Rindsberg (2012).
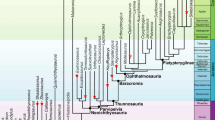
The dawn of taxonomical treatment of bioerosion trace fossils dates back almost two centuries (Fig. 1). Bronn (1837: p. 691) was the first to establish a still valid bioerosion ichnotaxon, Entobia, produced by excavating sponges. He did so, however, without introducing a species name, which was not a prerequisite at that time. The first of the many entobian ichnospecies was not described until 1843 as Entobia cretacea Portlock, 1843; it was subsequently designated as the type ichnospecies of Entobia by Häntzschel (1962). The first published and still valid bioerosion ichnospecies is Talpina ramosa von Hagenow, 1840, which was established together with Talpina solitaria von Hagenow, 1840, now cited as Trypanites solitarius (von Hagenow, 1840). Since these classical bioerosion trace fossils were established, more than a hundred ichnogenera and several hundred ichnospecies were described.
The timeline of bioerosion ichnotaxonomy, showing the number of valid as well as invalid ichnotaxa established per decade (bars) and cumulative per year (line). Arrows indicate landmark publications, initiations of workshop/conference series (IBW = International Bioerosion Workshop, WIT = Workshop on Ichnotaxonomy, ICHNIA = International Congress on Ichnology), and the various editions of the ‘Trace Fossils’ part of the ‘Treatise on Invertebrate Paleontology’ and the ‘International Code of Zoological Nomenclature’ (ICZN)
The primary objective of this contribution is to present an inventory of the currently recognized bioerosion ichnotaxa (Table 1), including taxa of dubious or invalid status (Table 2). While these lists were compiled, numerous redundant or otherwise invalid ichnotaxa were identified and consequently recombined, synonymized, and/or rejected. It is the very nature of such a taxonomic inventory that it is work in progress, and we hereby call for feedback from the research community. Input on errors of fact, conceptual pitfalls, and missing or potentially invalid ichnotaxa is highly appreciated: it would aid ichnotaxonomic stability and improve the completeness of this list. A web-based interactive bioerosion ichnotaxonomy database is currently under construction.
Concepts and methods
The present compilation is the result of an extensive literature query that primarily included searching and browsing the available publications on bioerosion trace fossils for records of ichnotaxa. In addition, a general Internet search was performed for all ichnogenera, and key online databases were consulted, such as the Index of Organism Names (ION; including all entries of the Zoological Record; http://www.organismnames.com), the Global Names Index (GNI; http://gni.globalnames.org), the Global Biodiversity Information Facility (GBIF; http://www.gbif.org/species), and ZooBank (http://zoobank.org). An earlier version of this list was presented as an interactive poster at the 9th International Bioerosion Workshop in Rome, October 2017, and feedback from the participants is considered in this review.
With respect to the definition of a trace fossil, we follow Bertling et al. (2006), defining it as ‘a morphologically recurrent structure resulting from the life activity of an individual organism (or homotypic organisms) modifying the substrate’. For confining bioerosion ichnotaxa, we acknowledge the original definition of the term by Neumann (1966), who defined bioerosion as ‘the removal of consolidated mineral or lithic substrate by the direct action of organisms’. We follow Bromley (1994), however, who refined the definition of bioerosion as ‘… the process by which animals, plants and microbes sculpt or penetrate surfaces of hard substrates’. With respect to traces in ‘hard substrate’, this definition includes bioerosion of calcareous substrates (biogenic and abiogenic carbonates), siliceous substrates (sensu lato; biogenic and abiogenic silica, silicate minerals and rocks, siliciclastic rocks), osteic substrates (bone, teeth, scales), and xylic substrates (wood, seeds, amber; soft plant tissues are excluded due to their consistency). These four principal substrate types (and more constrained substrates in some instances) are herein accepted as relevant ichnotaxobases on ichnospecies and especially on ichnogenus level, whereas ichnofamilies address primarily morphological categories across substrate types.
For clarification and complementation, Table 2 also lists those ichnotaxa that are not bioerosion ichnotaxa, but which were considered as such in the editions of the ‘Trace Fossils’ part of the ‘Treatise on Invertebrate Paleontology’ (Häntzschel 1962, 1975). The way ichnogenera and affiliated ichnospecies are presented largely follows the latest revision of the respective ichnotaxa. For those cases where (1) ichnotaxa are synonymized or otherwise rejected, (2) new combinations are established herein, or (3) nomenclatural acts are performed (introduction of nomina nova or emendations in spelling), this is briefly justified and discussed in a dedicated section.
Infrasubspecific varieties, such as Schloz’s (1972) Talpina ramosa var. curvata, recta, and subtilis, or the various varieties of Müller’s (1968) Conchifora zylindriformis, are not considered here, because such names are not regulated by the Code, if established after 1960 (ICZN 1999: Art. 15.2, 45.6.1). Similarly, the very few existing ichnosubspecies such as Müller’s (1977) Sedilichnus spongiophilus minus and maximus, are not considered herein, although such a subspecific rank is valid according to the Code (ICZN 1999).
The validity and availability of the ichnotaxa were assessed to the best of our knowledge following the jurisdiction of the ICZN (1999), thereby treating those ichnotaxa established in the years of ichnotaxonomic limbo (1931–1985) as if they were protected by the Code. Most relevant cut-off dates are the validity of extant animal traces and of illustrations without description prior to 1931, the validity of conditionally proposed ichnotaxa prior to 1961, and the validity of ichnogenera without a designated type ichnospecies, as well as nominal ichnotaxa established without designation of a holotype or syntypes prior to 2000.
As for traces of ctenostome bryozoans, there is a long-standing and still unresolved debate over which taxa to consider as biotaxa and which as ichnotaxa (e.g., Bertling 1995; Bromley 2004; Bertling et al. 2006; Rosso 2008; Buatois et al. 2017). The group remains in need of a revision that is beyond the scope of this paper. Consequently, we only list those taxa that were established as ichnotaxa in the first place (see Rosso 2008 for the latest review). Other (ichno-) genera that would need to be considered in a reevaluation include at least Electra Lamouroux, 1816; Terebripora d’Orbigny, 1847; Haimeina Terquem and Piette, 1865; Spathipora Fischer, 1866; Ropalonaria Ulrich, 1879; Vinella Ulrich, 1890; Penetrantia Silén, 1946; Immergentia Silén, 1946; Condranema Bassler, 1952; Foraripora Voigt and Soule, 1973; Casteropora Pohowsky, 1978; Cookobryozoon Pohowsky, 1978; Fischerella Pohowsky, 1978; Marcusopora Pohowsky, 1978; Orbignyopora Pohowsky, 1978; and Voigtella Pohowsky, 1978.
The distinction between true bioerosion traces and so-called ‘pseudoborings’ is of paramount importance. The latter are embedment structures termed ‘bioclaustrations’ (Palmer and Wilson 1988). They form when a living skeleton-secreting organism overgrows a living symbiont (sensu lato). This process has to be distinguished from ‘bioimmuration’ (Voigt 1972), where a dead organism is being overgrown. There is an ongoing discussion whether bioclaustration structures should be considered trace fossils. According to Bertling et al. (2006), they are not, because they were formed by means of a host reaction rather than active modification of the substrate by a tracemaker. It was (and is again here) consequently suggested that such structures be addressed as parataxa outside ichnotaxonomy in a yet to be defined ‘collective group’ within the framework of the ICZN. Nevertheless, numerous taxa have been established for bioclaustration structures prior to and after this rejection (see Tapanila and Ekdale 2007 and Klompmaker et al. 2014 for discussion), including the establishment of an ethological class ‘impedichnia’ by Tapanila (2005). This makes the standpoint expressed in Bertling et al. (2006) controversial.
Further complicating the matter, combinations of bioclaustration and bioerosion do occur, leading to so-called ‘compound boring-bioclaustrations’ (Tapanila and Ekdale 2007), as exemplified by Klemmatoica linguliforma Tapanila and Holmer, 2006 and Kanthyloma crusta Klompmaker et al., 2014. However, only those cases are herein considered as bioerosion trace fossils, where bioerosive action is predominant and identified as largely independent from the host reaction. These instances are the ichnogenus Tremichnus Brett, 1985 (see Wisshak et al. 2005), and the ichnospecies Gastrochaenolites vivus Edinger and Risk, 1994. All other bioclaustration structures proposed as trace fossils are not considered ichnotaxa here: Streptindytes Calvin, 1888; Gitonia Clarke, 1908; Hicetes Clarke, 1908;Chaetosalpinx Sokolov, 1948; Phragmosalpinx Sokolov, 1948; Diorygma Biernat, 1961; Torquaysalpinx Plusquellec, 1968; Helicosalpinx Oekentorp, 1969; Burrinjuckia Chatterton, 1975; Palaeophytobia Süss and Müller-Stoll, 1975; Protophytobia Süss, 1979; Catellocaula Palmer and Wilson, 1988; Clavatulicola Radwański and Bałuk, 1997; Eodiorygma Bassett et al., 2004; Klemmatoica Tapanila and Holmer, 2006; Caupokeras McKinney, 2009; Imbutichnus Santos et al., 2012; Ostiocavichnus Bohaty et al., 2012; Kanthyloma Klompmaker et al., 2014; Galacticus Klompmaker et al., 2016, and Thatchtelithichnus Zonneveld et al., 2015. The same applies to the related group of galls formed in calcareous skeletons, namely the ichnogenera Castexia Mercier, 1936; Endosacculus Voigt, 1959; and Heckerina Alekseev and Endelman, 1989.
The list of valid bioerosion ichnotaxa (Table 1) is organized in alphabetical order (ichnogenera) and year of publication (ichnospecies) instead of morphological criteria or higher taxonomic rank (existing and new ichnofamilies are characterized in a separate section). In addition, the type ichnospecies of each ichnogenus is indicated, as is the original combination of regrouped ichnospecies. The list is complemented by information about the nature of the bioeroded hard substrate, the general type of bioerosion trace, and the known or inferred tracemaker. Original publications are listed in the reference section.
The compilation of dubious or invalid ichnogenera and ichnospecies (Table 2) likewise is ordered alphabetically (ichnogenera) and according to the year of publication (ichnospecies). For each taxon, the reasoning for its present nomenclatural status and the respective reference are given.
This published work and the nomenclatural acts therein have been registered in ZooBank: http://zoobank.org/references/8FB87191-588F-47AD-8C0A-8957EAF3D6C8.
Bioerosion ichnofamilies
Only a limited number of the currently recognized bioerosion ichnotaxa is grouped within the higher systematic unit of an ichnofamily, although such a classification is supported by the ICZN (1999) and common for other groups of trace fossils (Bertling 2007). Some of the categories of architectural designs in trace fossils, as defined by Buatois et al. (2017), largely coincide with the currently recognized ichnofamilies (for instance the Dendrinidae), and were suggested by the latter authors as representing a suitable basis for the establishment of further ichnofamilies.
Acknowledging the demand for additional ichnofamilies that are primarily morphologically based (Bromley 1996; Bertling 2007) yet ecologically and ethologically meaningful, we additionally define a number of new ichnofamilies for bioerosion ichnotaxa accordingly. This way, emphasis is put on covering the most common and important ichnotaxa on the one hand, and on recognizing those groups that share enough morphological characters to allow inclusion of a fair amount of ichnogenera on the other hand. Several ichnogenera with a unique form remain without ichnofamily for the time being. Borings of ctenostome bryozoans are not considered here, because a revision of this group of bioerosion trace fossils is needed in order to clarify which of the established family names actually are to be regarded as ichnofamilies (see above).
Trypanitidae Mägdefrau, 1932
Type ichnogenus: Trypanites Mägdefrau, 1932.
Diagnosis: ‘Unbranched, straight tubes’ [original diagnosis, translated], here revised to: unbranched, cylindrical borings of approximately constant diameter with straight, winding, or spiraling course.
Members: Anobichnium Linck, 1949; Australocerambyx Peña, 1971; Carporichnus Genise, 1995; Eocavum Buchholz, 1986; Helicotaphrichnus Kern et al., 1974; Linckichnus Schlirf, 2006; Osprioneides Beuck and Wisshak in Beuck et al., 2008b; Paleobuprestis Walker, 1938; Paleoipidus Walker, 1938; Pecinolites Mikuláš and Dvořák, 2002; Spirichnus Fürsich et al., 1994; Stipitichnus Genise, 1995; Trypanites Mägdefrau, 1932; Tubulohyalichnus McLoughlin et al., 2009.
Ethological category: Domichnia (dwelling traces).
Rogerellidae Codez and de Saint-Seine, 1958
Type ichnogenus: Rogerella de Saint-Seine, 1951.
Diagnosis: ‘stalked slit, marginal bulge’ [original diagnosis, translated], here revised to: pouch-shaped borings.
Members: Aurimorpha Wisshak et al., 2008; Cubiculum Roberts et al., 2007; Cuenulites Rodríguez-Tovar et al., 2015; Cuniculichnus Höpner and Bertling, 2017; Petroxestes Wilson and Palmer, 1988; Rogerella de Saint-Seine, 1951; Sanctum Erickson and Bouchard, 2003; Umbichnus Martinell et al., 1999.
Ethological category: Domichnia (dwelling traces).
Remarks: Bromley and D’Alessandro (1987) considered the ichnogenus Zapfella de Saint-Seine, 1954 as a subjective junior synonym ofRogerella de Saint-Seine, 1951. They did not touch upon the question of synonymy at the ichnofamily level, even though Codez and de Saint-Seine (1958) erected the ichnofamily Zapfellidae in the same paper as their Rogerellidae, with Zapfella as its type ichnogenus. With synonymous type ichnogenera, the two ichnofamilies are synonymous as well. As it is up to the first reviser to clarify the synonymy of synchronously erected taxa (ICZN 1999: Art. 24.2), we here select Rogerellidae Codez and de Saint-Seine, 1958 as the valid ichnotaxon, rendering Zapfellidae invalid.
Dendrinidae Bromley et al., 2007
Type ichnogenus: Dendrina Quenstedt, 1849.
Diagnosis: Originally defined as ‘microborings having a rosetted or incompletely rosetted (i.e., fan-shaped) morphology, with or without a central or marginal main chamber’, slightly revised here to: dendritic or rosetted to fan-shaped borings, with or without a central or marginal main chamber.
Members: Abeliella Mägdefrau, 1937; Antodendrina, Wisshak, 2017; Calcideletrix Mägdefrau, 1937; Clionolithes Clarke, 1908; Dendrina Quenstedt, 1849; Dictyoporus Mägdefrau, 1937; Neodendrina Wisshak and Neumann, 2018; Nododendrina Vogel et al., 1987; Pyrodendrina Tapanila, 2008; Rhopalondendrina Wisshak, 2017.
Ethological category: Domichnia (dwelling traces), fodinichnia? (combined dwelling and feeding traces).
Osteichnidae Höpner and Bertling, 2017
Type ichnogenus: Osteichnus Höpner and Bertling, 2017.
Diagnosis: Originally defined as ‘non-branched borings in bone substrates irrespective of their orientation’, here revised to: cylindrical borings with fused U-loops.
Members: Asthenopodichnium Thenius, 1979; Canaliparva Furlong and McRoberts, 2014; Caulostrepsis Clarke, 1908; Maeandropolydora Voigt, 1965; Osteichnus Höpner and Bertling, 2017; Pseudopolydorites Głazek et al., 1971; Sertaterebrites Ekdale et al., 1989.
Remarks: Originally erected with osteic substrate as a main criterion, the ichnofamily can no longer be defined this way and is consequently redefined here based on the U-shaped morphology of its type ichnogenus.
Ethological category: Domichnia (dwelling traces).
Gastrochaenolitidae ifam. nov.
Type ichnogenus: Gastrochaenolites Leymerie, 1842.
Diagnosis: Unbranched, distally widened borings.
Members: Apectoichnus Donovan, 2018; Clavichnus Höpner and Bertling, 2017; Gastrochaenolites Leymerie, 1842; Palaeosabella Clarke, 1921; Phrixichnus Bromley and Asgaard, 1993; Teredolites Leymerie, 1842.
Ethological category: Domichnia (dwelling traces).
Talpinidae ifam. nov.
Type ichnogenus: Talpina von Hagenow, 1840.
Diagnosis: Branched cylindrical borings that may anastomose.
Members: Cunctichnus Fürsich et al., 1994; Cycalichnus Genise, 1995; Ipites igen. nov.; Lapispecus Voigt, 1970; Paleoscolytus Walker, 1938; Talpina von Hagenow, 1840; Xylonichnus Genise, 1995.
Ethological category: Domichnia (dwelling traces) or agrichnia (farming traces).
Entobiaidae ifam. nov.
Type ichnogenus: Entobia Bronn, 1837.
Diagnosis: Uni-camerate borings to multi-camerate boxwork borings, connected to the substrate surface by several apertures.
Members: Entobia Bronn, 1837; Unellichnus Breton, 2015a.
Ethological category: Domichnia (dwelling traces).
Remarks: The family name Entobiidae is already preoccupied by a family of copepods, established by Ho (1984) based on the type genus Entobius Dogiel, 1908. In order to avoid homonymy between family-group names, we follow recommendation 29A of the ICZN (1999) and use the entire ichnogeneric name as the stem for the new ichnofamily.
Planobolidae ifam. nov.
Type ichnogenus: Planobola Schmidt, 1992.
Diagnosis: Spherical, hemispherical or clavate microborings with a single or multiple connection(s) to the substrate surface.
Members: Cavernula Radtke, 1991; Cyclopuncta Elias, 1958; Granulohyalichnus McLoughlin et al., 2009; Planobola Schmidt, 1992.
Ethological category: Domichnia (dwelling traces).
Ichnoreticulinidae ifam. nov.
Type ichnogenus: Ichnoreticulina Radtke and Golubić, 2005.
Diagnosis: Systems of strongly ramifying microborings composed of cylindrical tunnels that often show intercalary, lateral, or terminal swellings.
Members: Conchocelichnus Radtke et al., 2016; Eurygonum Schmidt, 1992; Filuroda Solle, 1938; Ichnoreticulina Radtke and Golubić, 2005;Irhopalia nom. nov.; Orthogonum Radtke, 1991; Palaeomycelites Bystrov, 1956.
Ethological category: Domichnia (dwelling traces) or fodinichnia (combined dwelling and feeding traces).
Saccomorphidae ifam. nov.
Type ichnogenus: Saccomorpha Radtke, 1991.
Diagnosis: Spherical, sac-shaped, or multilobate microborings interconnected by thin tunnels.
Members: Polyactina Radtke, 1991; Saccomorpha Radtke, 1991.
Ethological category: Fodinichnia (combined dwelling and feeding traces).
Centrichnidae ifam. nov.
Type ichnogenus: Centrichnus Bromley and Martinell, 1991.
Diagnosis: Single to multiple, roughly circular depressions on the surface of hard substrates, shallower than wide, with individual grooves often arranged concentrically or excentrically.
Members: Augoichnus Arendt, 2012; Centrichnus Bromley and Martinell, 1991; Lacrimichnus Santos et al., 2003; Ophthalmichnus Wisshak et al., 2014; Solealites Uchman et al., 2018a; Tremichnus Brett, 1985.
Ethological category: Fixichnia (attachment traces).
Renichnidae ifam. nov.
Type ichnogenus: Renichnus Mayoral, 1987a.
Diagnosis: Spiral to elongate depressions on the surface of hard substrates, shallower than wide.
Members: Camarichnus Santos and Mayoral, 2006; Canalichnus Santos and Mayoral, 2006; Renichnus Mayoral, 1987; Spirolites Uchman et al., 2018a; Sulcichnus Martinell and Domènech, 2009.
Ethological category: Fixichnia (attachment traces).
Podichnidae ifam. nov.
Type ichnogenus: Podichnus Bromley and Surlyk, 1973.
Diagnosis: Multiple round or oval depressions on the surface of hard substrates, regularly spaced in a cluster.
Members: Finichnus Taylor et al., 2013; Flosculichnus Donovan and Jagt, 2005; Podichnus Bromley and Surlyk, 1973.
Ethological category: Fixichnia (attachment traces).
Gnathichnidae ifam. nov.
Type ichnogenus: Gnathichnus Bromley, 1975.
Diagnosis: Repetitive sets of linear, shallow grooves on the surface of hard substrates.
Members: Gnathichnus Bromley, 1975; Radulichnus Voigt, 1977.
Ethological category: Pascichnia (combined locomotion and grazing traces).
Circolitidae ifam. nov.
Type ichnogenus: Circolites Mikuláš, 1992.
Diagnosis: Circular to irregular-shaped depressions in hard substrates, with steep to overhanging marginal walls that may show sets of linear, shallow grooves.
Members: Circolites Mikuláš, 1992; Ericichnus Santos and Mayoral in Santos et al., 2015; Osteocallis Roberts et al., 2007; Planavolites Mikuláš, 1992.
Ethological category: Combined pascichnia (combined locomotion and grazing traces) and domichnia (dwelling traces).
Oichnidae ifam. nov.
Type ichnogenus: Oichnus Bromley, 1981.
Diagnosis: Complete circular penetrations or sets thereof in biogenic hard substrates, in some cases surrounded by shallow etchings.
Members: Dipatulichnus Nielsen and Nielsen, 2001; Kardopomorphos Beuck et al., 2008a; Lamniporichnus Mikulaš et al., 1998; Loxolenichnus Breton et al., 2017; Oichnus Bromley, 1981; Stellatichnus Nielsen and Nielsen, 2001.
Ethological category: Praedichnia (predation traces; but also in xylic seeds), occasionally combined with fixichnia (attachment traces).
Belichnidae ifam. nov.
Type ichnogenus: Belichnus Pether, 1995.
Diagnosis: Recurrent fracture patterns in shells and other skeletal material.
Members: Belichnus Pether, 1995; Bicrescomanducator Donovan et al. in Andrew et al., 2010.
Ethological category: Praedichnia (predation traces).
Machichnidae ifam. nov.
Type ichnogenus: Machichnus Mikuláš et al., 2006.
Diagnosis: Punctures to grooves, both of somewhat irregular outline, often in sets, in bone.
Members: Knethichnus Jacobsen and Bromley, 2009; Linichnus Jacobsen and Bromley, 2009; Machichnus Mikuláš et al., 2006; Mandaodonites Cruickshank, 1986; Nihilichnus Mikuláš et al., 2006.
Ethological category: Praedichnia (predation or scavenging traces).
Nomenclatural acts, invalidations, synonymizations and new combinations
This section contains the justification and discussion of all nomenclatural acts (i.e., introduction of nomina nova and nomina corrigenda), new combinations, and invalidations performed in this review, with represented ichnotaxa in alphabetical order.
Aggregatella Elliott, 1962, with its type ichnospecies A. pseudohieroglyphicus Elliott, 1962, was erected based on a thin-section and interpreted as a microcoprolite (pellet). However, these ‘small, flexuous curved or twisted elongate solid bodies’ are more likely microborings, but they cannot be identified with certainty based on the two-dimensional thin-section image. Accordingly, the two ichnotaxa must be considered nomina dubia.
Anellusichnus Santos et al., 2005 is herein regarded as subjective junior synonym of Centrichnus Bromley and Martinell, 1991 and its two ichnospecies are well accommodated within the original diagnosis of Centrichnus. Because they differ in morphology from the other ichnospecies, they are considered valid as new combinations.
Anoigmaichnus Vinn et al., 2014 and its type ichnospeciesA. odinsholmensis Vinn et al., 2014 include a cylindrical shaft with an elevated aperture, the latter interpreted as an embedment structure. Being mainly a boring, A. odinsholmensis resembles Trypanites weisei Mägdefrau, 1932, whose type material occasionally shows elevated apertures (so-called ‘Ringwälle’, Mägdefrau, 1932), and is therefore regarded as junior synonym of it.
Archaeomycelites Bystrow, 1959 and its type ichnospeciesA. odontophagus Bystrow, 1959 are herein synonymized with Palaeomycelites Bystrov, 1956 and its type ichnospecies P. lacustris Bystrov, 1956, respectively, based on identical shape of the borings; neither the position in bone/enamel nor facies should be used as ichnotaxobases (Bertling et al. 2006).
Asthenopodichnium fallax Francischini et al., 2016 does not belong to this ichnogenus, which is restricted to xylic substrates (Höpner and Bertling 2017). Rather, its substrate is calcareous; given this and the well-defined shape, it belongs to Petroxestes and is here considered a junior synonym of Petroxestes altera Jagt et al., 2009.
Asteriastoma Breton, 1992 and its type ichnospeciesA. cretaceum Breton, 1992 are subjective junior synonyms of Gnathichnus Bromley, 1975 and its type ichnospecies G. pentax Bromley, 1975, respectively, as suggested by Buatois et al. (2017). Asteriastoma merely constitutes a composite trace fossil of G. pentax oriented around a preexisting boring as a result of grazing and/or predation (see Bromley 1970b and Wisshak 2006 for examples).
Bascomella Morningstar, 1922, represented by its type ichnospecies B. gigantea Morningstar, 1922, was originally considered to be a ctenostome bryozoan boring composed of zooidal chambers and interconnecting stolons. Two additional ichnospecies were later proposed, namely B. fusiformis Condra and Elias, 1944 and B. subsphaerica Condra and Elias, 1944. Their actual composite nature of accidentally co-occurring acrothoracican borings and tubular borings of unknown origin was recognized by Elias (1957), who advocated redefiningBascomella as the pouch-shaped borings only. Restricting a composite ichnotaxon to one of its components, however, deviates from the original author’s intention: Morningstar (1922) meant to name the composite structure and hence, no single element can and may be isolated from it. The redefinition by Elias (1957) fails for this reason and consequently the composite ichnotaxon is invalidated, as it contains the work of more than one organism (e.g., Bertling et al. 2006). The taxonomic status of the other Bascomella ichnospecies is identical, even if they may contain different representatives ofRogerella de Saint-Seine, 1951 or other ichnogenera.
Belichnus Pether, 1995 was erected based on Holocene shells, but the determined age of 13.5–12.5 ka now corresponds to a Pleistocene age (Gradstein et al. 2012). Because no type ichnospecies was designated, B. monos is herein subsequently designated as type ichnospecies (ICZN 1999: Art. 69). Belichnus dusos Pether, 1995 was considered as a potential junior synonym of B. monos by Cadée and de Wolf (2013), which is formally confirmed herein.
Brachyzapfes Codez in Codez and de Saint-Seine, 1958 was synonymized with Rogerella de Saint-Seine, 1951 by Bromley and D’Alessandro (1987), based on close morphological resemblance. Accordingly, its type ichnospecies B. elliptica Codez in Codez and de Saint-Seine, 1958 is herein formally transferred to Rogerella as a new combination.
Brutalichnus Mikuláš et al., 2006, with the type ichnospecies B. brutalis Mikuláš et al., 2006, was erected on irregular though intense cracks in bones. They were claimed to have been produced by a carnivorous animal but do not exhibit tooth traces. The breakage is at least as likely due to diagenetic effects such as compaction, i.e., the biogenic nature of the structure remains to be demonstrated.
Caedichnus Stafford et al., 2015 and its type ichnospeciesC. spiralis Stafford et al., 2015 are phenomena of gastropod shell damage of various shape, which are consistent with the diagnosis of Bicrescomanducator Donovan et al. in Andrew et al., 2010. Caedichnus is therefore synonymized with that ichnogenus, maintaining its type ichnospecies as a new combination.
Calciroda Mayer, 1952 and its type ichnospeciesC. kraichgoviae Mayer, 1952 consists of ‘branched, horizontal tunnels running along the surface of organism hard parts, sometimes going vertically into the depth’ [original diagnosis, translated]. No holotype was designated, but the figured specimens indicate an overlapping or cross-cutting relationship of individual tunnels instead of true branching, in addition to unbranched specimens. Plewes (1996: pl. 13, Fig. 5) studied Mayer’s type material and confirmed that ‘…both branching points and avoidance by tunnels can be seen…’; she regarded Calciroda as junior synonym of Talpina von Hagenow, 1840. In comparison with the recently rediscovered type material of Talpina and its revision (Wisshak et al. 2017: Fig. 2.1), the branched specimens of the dubious (because composite) and partly eroded trace fossil C. kraichgoviae can be assigned to Talpina ramosa von Hagenow, 1840, whereas the unbranched specimens belong to Trypanites solitarius (von Hagenow, 1840). The second ichnospecies, C. tubulata Hary, 1975, mainly differs by its fill and preservation from branched C. kraichgoviae and thus is also synonymized with T. ramosa.
Chaetophorites Pratje, 1922, including the type ichnospecies C. gomontoides Pratje, 1922 and C. tenuis Mägdefrau, 1937, are nomina dubia. A neotype designation for lost type material of the former has proven infeasible (Glaub 1994), and C. tenuis is too poorly defined and illustrated to allow an unambiguous application. Chaetophorites appears to have represented a storage tank for various microborings, which have only become accessible to reliable morphological characterization with the advent of the epoxy casting method in the 1970s (Golubić et al. 1970). A third ichnospecies, C. cruciatus Mägdefrau, 1937, was later recognized as bryozoan trace fossil and was recombined by Voigt (1973) asImmergentia cruciata (Mägdefrau, 1937).
Cliona Grant, 1826 - > see Entobia Bronn, 1837 below.
Clionites Morris in Mantell, 1850 is an ichnogenus for sponge bioerosion traces with close affinity to Entobia Bronn, 1837. Its type ichnospecies C. conybeari Morris in Mantell, 1850 was consequently considered by Bromley (1970a) as a subjective junior synonym of E. cretacea Portlock, 1843, thus also synonymizing the two ichnogenera. We cannot judge C. parkinsoni Morris in Mantell, 1850 because we have not researched and investigated the holotype; it is only rudimentarily described, and the corresponding illustrations do not allow distinction of this ichnospecies. C. glomerata Morris, 1851 clearly is a cavernous sponge boring, and thus adequately grouped in the ichnogenusEntobia Bronn, 1837. As a result, it becomes a secondary junior homonym toE. glomerata (Michelin, 1846) comb. nov., another sponge bioerosion trace herein included in Entobia, thus demanding establishment of a nomen novum (see below underEntobia).
Clionoida Smith, 1910 and its type ichnospeciesC. arbiglandensis Smith, 1910 are nomina dubia, since the original illustration is poor and no type material could be tracked down in the two relevant Glasgow collections. Furthermore, no explicit definition is given for this ichnogenus.
Clionoides Fenton and Fenton, 1932, with C. thomasi Fenton and Fenton, 1932 as type ichnospecies, was erected for tubular and infrequently branched borings along the surface of shells. Furlong and McRoberts (2014) erroneously regarded the ichnogenus Palaeosabella Clarke, 1921 as a synonym of Clionoides, but disregarded the principle of priority (ICZN 1999: Art. 23). Moreover,Clionoides is a tubular boring without a club-shaped extension as in Palaeosabella. Although no holotype was specified by Fenton and Fenton (1932), their figures show shells with numerous tunnels and grooves running along or close to the surface. Branching occurs but can be regarded as false due to overlapping borings rather than true branching. Given that circumstance, Clionoides and C. thomasi can be defined as tubular borings along the surface of the substrate and thus become junior synonyms of Trypanites Mägdefrau, 1932 and T. solitarius (von Hagenow, 1840), respectively.C. utaturensis Ghare, 1982 describes ‘flexuous or more or less straight borings with circular openings’ in belemnite rostra, which is also consistent with the diagnosis of T. solitarius (von Hagenow, 1840) (see Bromley 1972), and is therefore included in it as another subjective junior synonym.
Conchifora zylindriformis Müller, 1968, including varieties, is synonymized with Trypanites solitarius (von Hagenow, 1840) based on the course more or less parallel to the curvature of the substrate surface.
Conchotrema Teichert, 1945 is an ichnogenus often discussed as probable synonym of Talpina von Hagenow, 1840 because of their close morphological similarity and comparable dimensions (e.g., Voigt 1972; Plewes 1996; Bromley 2004). However, to our knowledge its two ichnospecies were, never formally recombinedTalpina, and thus are formally recombined herein on grounds of the valid arguments put forward by these authors. It still needs to be clarified, however, whether the two ichnospecies are invalid junior synonyms or valid senior synonyms of other Palaeozoic ichnospecies of Talpina.
Cubiculum inornatus Xing et al., 2015 as well as the nomenC. levis Pirrone et al., 2014 are emended to corresponding gender of genus and species names, i.e., to C. inornatum and C. leve, respectively, as the gender of the ichnogenus is neuter (ICZN 1999: Art. 31.2).
Cunicularis isodiametricus Li, 1997 was established based on phosphatized natural casts of microborings and thus is to be considered an ichnotaxon as opposed to a cyanobacterium body fossil of the biotaxon Cunicularis Green et al., 1988. However, morphologically it is reminiscent of Endoconchia lata Runnegar in Bengtson et al., 1990, and thus herein considered a junior synonym of it.
Cylindricavus Borkar and Kulkarni, 1987, based on the type ichnospecies C. perplexus Borkar and Kulkarni, 1987, consists of ‘cylindrical, unbranched borings with variable disposition’ and occurs in freshwater carbonates. It is compared with Paleolithophaga (lapsus calami; junior synonym of Gastrochaenolites), but erected based on its freshwater instead of marine origin. However, facies or environment are not valid ichnotaxobases (Bertling et al. 2006). Moreover, freshwater borings are very rare compared to their marine counterparts, and bioerosion ichnotaxa of this size are essentially unknown. Various shapes and sizes of the figured C. perplexus suggest an alternative interpretation, e.g., as fossil root casts, and thus Cylindricavus is at present regarded as nomen dubium.
Cylindrocavites Ghare, 1982, based on its type ichnospecies C. cretacea Ghare, 1982, consists of cylindrical borings penetrating belemnite rostra perpendicular to their surface. Its form is identical with Trypanites Mägdefrau, 1932, and it is therefore regarded as junior synonym, following Pemberton et al. (1988). At the ichnospecies level, C. cretacea shares all diagnostic features with T. weisei Mägdefrau, 1932 and is herein synonymized with it.
Dekosichnus Genise and Hazeldine, 1995, with its type ichnospecies D. meniscatus Genise and Hazeldine, 1995, appears to be a junior synonym of Xylonichnus Genise, 1995. The diagnoses claim slight differences in the relative height and the more or less oval cross-section, but the figures of the type do not exhibit these characters. The greater density of borings of theDekosichnus type is not an ichnotaxobase (Bertling et al. 2006). Hence,D. meniscatus is herein transferred toXylonichnus as new combination.
Entobia Bronn, 1837 is by far the most speciose bioerosion ichnogenus. In the course of this compilation, it has received further accrual of former sponge biotaxa that actually describe empty fossil sponge bioerosion traces. These are to be considered bioerosion ichnotaxa, just as empty recent sponge borings described prior to 1931. This concerns species formerly in the invalid junior synonym Vioa Nardo, 1833: E. michelini (Nardo, 1845) comb. nov., E. nardina (Michelin, 1846) comb. nov., E. glomerata (Michelin, 1846) comb. nov., E. duvernoyi (Michelin, 1847) comb. nov., E. pectita (Michelotti, 1861) comb. nov., E. cerithii (Fraas, 1867) comb. nov., andE. catenata (Frič, 1883) comb. nov. Three further Vioa species were recently transferred to Entobia by Schönberg et al. (2017). Two Vioa ichnospecies are herein considered nomina dubia based on unspecific descriptions and lack of specific types, i.e., V. ostrearum Fraas, 1869 and V. millaris Frič, 1883. Another two Vioa ichnospecies, V. rependa Sismonda, 1871 and V. superficialis Sismonda, 1871, are herein considerednomina dubia, owing to vague descriptions and a poor or no illustration, respectively. Furthermore, the authorship of Sismonda (1871) is unclear: He apparently was the first to record the two taxa in question, but he cited Michelotti as their author. We were unable, however, to find the names in Michelotti’s work. In addition, numerous species of the valid sponge genus Cliona Grant, 1826 are here transferred to Entobia, namely E. irregularis (d’Orbigny, 1850) comb. nov., E. ramosa (d’Orbigny, 1850) comb. nov., E. perforata (Seguenza, 1882) comb. nov., E. intricata (Seguenza, 1882) comb. nov., E. peregrinator (Chapman, 1907) comb. nov., E. bullini (Annandale, 1920) comb. nov., E. radiciformis (Lehner, 1937), comb. nov., and E. microtuberum (Stephenson, 1941) comb. nov., the latter tentatively synonymized withE. megastoma (Fischer in d’Archiac et al., 1866) by Bromley and D’Alessandro (1984), a view that is not shared here. Two species of Cliona, C. duchassaingi Fischer, 1868 and C. studeri Mayer, 1872, are nomina nuda, since they were only listed without any description or indication. Yet four more Cliona species were described by Seguenza (1879), only the first one of which appears to be a fossil sponge boring, consecutively recombined as E. tubulosa (Seguenza, 1879) comb. nov. herein. The second one is recombined asMaeandropolydora vermicularis (Seguenza, 1879) comb. nov., the third is an unspecific assemblage of minute microborings and thus considered a nomen dubium, and the last one is an ascothoracid barnacle boring, recombined herein as Rogerella oostoma (Seguenza, 1879) comb. nov. However, according to Cleevely (1983) the collections of Seguenza were devastated during the 1908 Messina earthquake, rendering re-investigation of his types impossible. Three Cliona species established by Fischer in d’Archiac et al. (1866) and Fischer (1868) were considered nomina dubia by Bromley and D’Alessandro (1984). The type material, however, is accessible at the Paris Natural History Museum, and it is sufficiently well preserved to merit further investigation. Hence, these species are formally transferred to Entobia as E. falunica (Fischer in d’Archiac et al., 1866) comb. nov., E. praecursor (Fischer, 1868) comb. nov., and E. cerithiorum (Fischer, 1868) comb. nov. Finally, Cliona kelheadensis Smith, 1910 is considered a nomen dubium since its original illustration is poor and the type material appears to be lost. The ichnospecies E. glomerata (Morris, 1851) is clearly distinct from E. glomerata (Michelin, 1846) comb. nov., judged by the respective illustrations showing two very different forms of sponge borings. Given that Morris (1851) did not make any reference to Michelin (1846) when erecting his species, E. glomerata (Morris, 1851) becomes a secondary junior homonym ofE. glomerata (Michelin, 1846) comb. nov. The former thus requires a replacement name, and accordingly, E. morrisi nom. nov. is hereby introduced for E. glomerata (Morris, 1851). The name honours John Morris and his pioneering work on bioerosion trace fossils from the Upper Cretaceous of northern Europe. A similar case is E. mammillata Bromley and D’Alessandro, 1984, which is a secondary junior homonym to E. mammillata (Chapman, 1907) comb. nov., and is herein transferred from Cliona to Entobia. We introduce E. tuberculata nom. nov. as a replacement name, thereby largely retaining the original reference to the diagnostic mammillate hemispherical tubercles. In any case, in order to identify synonyms, the present plethora of 53 Entobia ichnospecies, and in particular the recently transferred early taxa, are in need of revision.
Feldmannius cavernosa (Casadío et al., 2001) is emended in spelling to read F. cavernosus (Casadío et al., 2001), in order to comply in gender with the generic name (ICZN 1999: Art. 31.2). This measure is deemed necessary as a result of Feldmannius Low and Guinot, 2010 having been introduced as a nomen novum for Feldmannia Casadío et al., 2001.
Gaspeichnus Hunt et al., 2018 and its type ichnospeciesG. complexus Hunt et al., 2018 are considered nomina dubia, since lithification of the substrate prior to emplacement of traces cannot be demonstrated, and the flattened traces likely represent burrows of uncertain affinity rather than borings.
Gitonia Clarke, 1908, defined by its type ichnospecies G. corallophila Clarke, 1908, is a bioclaustration and not a bioerosion trace (Oliver 1983). However,G. siphon Clarke, 1908, is a cylindrical bioerosion trace and was synonymized with Vermiforichnus clarkei by Cameron (1969b), which in turn is herein synonymized with Palaeosabella prisca (McCoy, 1855). Examination of Clarke’s (1908) type material of G. siphon will show whether it belongs to Palaeosabella Clarke, 1921 or to Trypanites Mägdefrau, 1932.
Glirotremmorpha entectus Collinson and Hooker, 2000 is emended to G. entecta, because the gender of the ichnogenus is feminine (ICZN 1999: Art. 31.2).
Hemicanalis Chiplonkar and Ghare, 1977, including its type ichnospecies H. reticulata Chiplonkar and Ghare, 1977, as well as the second ichnospecies H. prolius Chiplonkar and Ghare, 1977, are netlike, ramified burrows on the internal moulds of molluscan shells rather than bioerosion trace fossils. This way they have to be considered junior synonyms of Arachnostega Bertling, 1992 and A. fossiger (Fenton and Fenton, 1932).
Heterodontichnites Rinehart et al., 2006, with its type species H. hunti Rinehart et al., 2006, was supposed to differ from Mandaodonites coxi Cruickshank, 1986 by alleged round tooth traces of the latter and a more curved pattern. Owing to the incomplete preservation of the single specimen with Heterodontichnites, the latter difference cannot be substantiated, whereas the single tooth traces of Mandaodonites are ovoid, as in Heterodontichnites. With no significant difference remaining, the two ichnogenera and their type ichnospecies are respectively synonymized.
Ichnogutta erectus Botquelen and Mayoral, 2005 is emended to read I. erecta Botquelen and Mayoral, 2005 and to comply in gender with the generic name (ICZN 1999: Art. 31.2).
Ipites has been established by Karpiński (1962) explicitly noting twice the supposed close relationship to the Recent ‘Ips duplicatus Sahlb.’. The author introduces the name by stating: ‘The genus of the beetle I define with the name Ipites.’, i.e., without designating it as ‘igen. nov.’ or similar, after noting that this ‘is the first finding of brood galleries of a representatitve resembling the modern genus I. duplicatus Sahlb.’ Article 20 of the Code (ICZN 1999), however, states that ‘A name […] applied to fossils to distinguish them from extant members of that taxon, without clear evidence of intent to establish a new genus-group taxon, […] cannot be used as the valid name of a taxon.’ Ipites is not an available name therefore, leaving its sole ichnospecies name bobrowskianus without ichnogenus name. For this reason, we reintroduce Ipites as a new ichnogeneric name here and designate Ipites bobrowskianus (Karpiński, 1962) as its type ichnospecies. The etymology is based on the similarity of the trace fossil to the borings of the modern bark beetle genus Ips. We diagnose the ichnogenus as follows: Complex boring in xylic substrates, with regularly branched, larger central tunnels and numerous smaller tunnels radiating from the branches; the system lies parallel to the substrate surface. As differential diagnosis we remark that modern bark beetles exhibit a rather wide array of boring architecture, and informed by these patterns, we distinguish Ipites igen. nov. from Scolytolarvariumichnus Guo, 1991 with just one simple shaft in the centre.
Karethraichnus kulindros Zonneveld et al., 2015 is distinguished from K. lakkos Zonneveld et al., 2015 by ‘its cylindrical vs hemispherical morphology’. However, this difference can be explained as various developmental stages of the same boring, a situation analogous to the variable penetration depth found in ichnospecies ofOichnus. We follow the reasoning of Wisshak et al. (2015) and regard K. kulindros as a junior synonym of K. lakkos.
Knethichnus parallelum Jacobsen and Bromley, 2009 is emended to K. parallelus, because the gender of the ichnogenus is masculine (ICZN 1999: Art. 31.2).
Maeandropolydora filosa Chiplonkar and Ghare, 1977 is based on limited and poorly preserved material in a gastropod shell. The U-shaped tube has no specific orientation, a constant diameter and circular cross-section. As such it may be regarded as part of an originally more extensive boring assignable to M. sulcans Voigt, 1965 (cf. Bromley and D’Alessandro 1983).
Megascolytinus Petrov, 2013, with M. zherikhini Petrov, 2013 as its type ichnospecies, was established solely because of its large size, which should not be an ichnotaxobase (Bertling et al. 2006). In principle, its shape is identical withScolytolarvariumichnus Guo, 1991, but the ichnospecies deviates fromS. radiatus Guo, 1991. Therefore, it is herein cited asS. zherikhini (Petrov, 2013) comb. nov.
Mycelites Roux, 1887, with M. ossifragus Roux, 1887 as its type ichnospecies, was long deemed to be the only available ichnotaxon for fungal borings. Roux’ descriptions, at least in part, refer to organic material, as he writes about septa in channels. This excludes a void boring (a trace) but rather points to the presence of an organism. For this reason, the ichnogenus and ichnospecies are rejected. Mycelites conchifragus Schindewolf, 1962 was proposed only conditionally and is thus a nomen nudum (atelonym): ‘Simply in accordance with a handy label, I name them Mycelites conchifragus. If one wanted to attach species value to this name, the specimen […] may serve as a holotype’ [translated].
Mycobystrovia Goujet and Locquin, 1979 and its type ichnospecies M. lepidophaga Goujet and Locquin, 1979 are a taxa combining a fungus biotaxon and the probable boring of the same producer. The diagnosis makes it clear, however, that the name refers to the spores, and thus a fungus body fossil, whereas the boring is not described.
Nygmites Mägdefrau, 1937 and its type ichnospeciesN. solitarius (von Hagenow, 1840) are junior synonyms of Trypanites Mägdefrau, 1937 and T. solitarius (von Hagenow, 1840), respectively (Bromley 1972). Mägdefrau (1937) included N. pungens (Quenstedt, 1849) (= Terebripora pungens) and established the new ichnospecies N. sacculus, which differs from the other two ichnospecies by its pouch instead of cylinder shape. Consequently, N. sacculus was assigned toBrachyzapfes elliptica Codez and de Saint-Seine, 1958 by Seilacher (1968), synonymized with Zapfella pattei (de Saint-Seine, 1955) by Tomlinson (1969), and combined as Brachyzapfes sacculus (Mägdefrau, 1937) by Voigt (1972). Its nature as the boring of a cirriped was also revealed by Kennedy (1970). Based on its morphology, N. sacculus is herein transferred toRogerella de Saint-Seine, 1951 as new combination. A thorough review ofRogerella will show its ichnospecific affinity.
Osedacoides cretaceus Karl and Nyhuis, 2012 is a nomen dubium since it may be a valid ichnospecies, but the illustrated unique type specimen does not allow to decide whether it belongs to Karethraichnus Zonneveld et al., 2015. If this is the case it may form a senior synonym of one of the ichnospecies of Karethraichnus as well as of this ichnogenus.
Ostreoblabe Voigt, 1965 and its type ichnospeciesO. perforans Voigt, 1965 were originally described from the inner valve of oyster shells and are partly embedment structures and partly borings (Bromley 1970a, 2004). The borings are to be identified asPalaeosabella Clarke, 1921 and P. prisca (McCoy, 1855), respectively, thus being considered junior synonyms here.
Palaeachlya Duncan, 1876, (including the ichnospeciesP. perforans Duncan, 1876; P. tortuosa Etheridge, 1891; and P. torquis Etheridge, 1899), Palaeoperone Etheridge, 1891 (ichnospecies P. endophytica Etheridge, 1891), and Palaeopede Etheridge, 1899 (ichnospeciesP. whiteleggei Etheridge, 1899) were established as biotaxa, but the original descriptions and illustrations combine features of the tracemakers and their borings. The illustrations are, however, too unspecific to justify the status of the ichnotaxa; all are regarded as nomina dubia rather than algal or fungal body fossils; the type material needs to be researched and reinvestigated to clarify their status.
Palaeosabella arrogarum Plewes, 1996 was erected in an unpublished thesis and is therefore a nomen nudum.
Paleolithophaga Chiplonkar and Ghare, 1976 was synonymized with Gastrochaenolites Leymerie, 1842 by Kelly and Bromley (1984). Its type ichnospeciesP. andurensis Chiplonkar and Ghare, 1976 currently is a nomen dubium and would require the definition and inspection of a lectotype out of the several clusters of circular and incomplete borings contained on the type material. P. velasensis Badve and Ghare, 1984 appears to be a soft-bottom burrow with affinity toTisoa siphonalis de Serres, 1840, rather than a bioerosion ichnotaxon, and was synonymized with the latter (Knaust 2019).
Paleolithopholas Badve and Ghare, 1984 is defined as ‘circular to oblong’ borings. The figured material has to be assigned to Gastrochaenolites Leymerie, 1842 and is thus synonymized with this ichnogenus. Accordingly, its type ichnospecies P. raigadensis Badve and Ghare, 1984 is transferred to Gastrochaenolites, awaiting ichnotaxonomic revision to resolve potential synonymy with other ichnospecies in that ichnogenus.
Paleoscolytus sussexensis Jarzembowski, 1990 does not belong to this ichnogenus because it does not display the highly significant branched tunnels. Rather, it is a member of Scolytolarvariumichnus Guo, 1991, a scolytid boring with radial unbranched tunnels, leading to the new combination S. sussexensis (Jarzembowski, 1990).
Pennatichnus moguerenica Mayoral, 1988 is emended to P. moguerenicus, because the gender of the ichnogenus is masculine (ICZN 1999: Art. 31.2).
Planobola radicatus Schmidt, 1992 is emended to P. radicata, because the gender of the ichnogenus is feminine (ICZN 1999: Art. 31.2).
Polydorichnus Ishikawa and Kase, 2007 and its type ichnospecies P. subapicalis Ishikawa and Kase, 2007 are cylindrical borings in the columella of gastropod shells. They are identical with Helicotaphrichnus Kern et al., 1974 and H. commensalis Kern et al., 1974, respectively, and thus identified as junior synonyms.
Rhopalia Radtke, 1991 is an invalid junior homonym, because this genus name is preoccupied by the dipteran genus Rhopalia Macquart, 1838. This has already seen another junior homonym, the tunicate genus Rhopalia Norman, 1897 (now Rhopalaea Philippi, 1843). In consequence, we here introduce Irhopalia nom. nov., following a suggestion of the original author who had established Rhopalia (Radtke pers. comm. 2018). Irhopalia now includes the type ichnospecies Irhopalia catenata (Radtke, 1991) comb. nov., as well asI. spinosa (Radtke and Golubić, 2005) comb. nov. and I. clavigera (Golubić and Radtke, 2008) comb. nov.
Sedilichnus smiley Pokorný and Štofik, 2017 is a circular opening in a shell with an inner opening of crescentic outline. Following the review of Oichnus by Wisshak et al. (2015), this form represents an incomplete (unfinished) O. paraboloides, forming a junior synonym of it.
Seminolithes Hyde, 1953 is a subjective junior synonym of Rogerella de Saint-Seine, 1951, as already regarded by Bromley and D’Alessandro (1987). As a consequence, its type ichnospecies S. linii Hyde, 1953 (by monotypy) is now listed as the new combination Rogerella linii (Hyde, 1953).
Simonizapfes Codez and de Saint-Seine, 1958 is another subjective junior synonym of Rogerella de Saint-Seine, 1951, according to Bromley and D’Alessandro (1987), its type ichnospecies S. elongata Codez and de Saint-Seine, 1958 to be cited asR. elongata (Codez and de Saint-Seine, 1958) comb. nov. Likewise,S. davenporti Tomlinson, 1969 is transferred to Rogerella until a proper review of this ichnogenus is undertaken.
Specus Stephenson, 1952, based on the type ichnospecies S. fimbriatus Stephenson, 1952, includes curved or irregular, club-shaped borings and was synonymized with Trypanites Mägdefrau, 1932 by Bromley (1972). After Plewes (1996) reinstated Palaeosabella Clarke, 1921, Bromley (2004) suggested to synonymize Specus with that ichnogenus, while Furlong and McRoberts (2014) prefer synonymization with Clionoides Fenton and Fenton, 1932. With Clionoides herein considered a junior synonym of Trypanites (see above), however, we follow Bromley’s (2004) view and regard Specus as a junior synonym of Palaeosabella. S. fimbriatus is synonymous with P. prisca (McCoy, 1855), although a lectotype is still to be defined.
Spiracavites Chiplonkar and Ghare, 1977 was regarded a junior synonym of Trypanites Mägdefrau, 1932 by Pemberton et al. (1988), and its type ichnospeciesS. vermicularis Chiplonkar and Ghare, 1977 can well be assigned toT. solitarius (von Hagenow, 1840). The other two ichnospecies, S. radialis and S. marginaria Chiplonkar and Ghare, 1977 are recognized as morphological variations and thus synonymous with T. solitarius.
Spirichnus contentus Plewes, 1996 and S. vacillarum Plewes, 1996 were erected in an unpublished thesis and therefore are nomina nuda.
Stichus Etheridge, 1904 and its type ichnospeciesS. mermisoides Etheridge, 1904 are difficult to judge because the original illustrations lack resolution and spatial detail. We have not investigated the type material, but the structures described may not be bioerosion traces but diagenetically altered pores in the host shell. Consequently, both ichnotaxa are considered nomina dubia for the time being.
Talpina astartina Étallon in Thurmann and Étallon, 1864 is a nomen dubium, owing to a poor illustration and lost type material. Talpina scalariformis Ghare, 1982 does not comply with the characteristics of the ichnogenus Talpina von Hagenow, 1840. Instead, the parallel course of its tunnels and the rectangular branching pattern identify this ichnospecies as belonging to the ichnogenus Orthogonum Radtke, 1991, and it is here recombined accordingly. A closer investigation of the type material will be necessary to verify whether it is a senior synonym of O. giganteum Glaub, 1994.
Terebripora (?)portlocki Fischer, 1866 was introduced as a name referring toEntobia antiqua Portlock, 1843, thus representing an objective junior synonym only. However, E. antiqua indeed is a boring of a ctenostome bryozoan with affinity to Terebripora d’Orbigny, 1847.
Trypanites helicus Nielsen and Görmüş, 2004 was erected for planispirally tubular borings, thus not complying with the diagnosis ofTrypanites Mägdefrau, 1932. It is better accommodated in Helicotaphrichnus Kern et al., 1974, despite the different host organism, as the new combination H. helicus (Nielsen and Görmüş, 2004). The main difference from H. commensalis is its planispiral instead of trochospiral shape.
Trypetesa Norman, 1903 is a genus of acrothoracican body fossils and not an ichnogenus (e.g., Buatois et al. 2017). Similarly, Murphy and Williams (2013) recognized five species ofTrypetesa as valid acrothoracican cirriped species. T. caveata Tomlinson, 1963 and T. polonica Bałuk and Radwański, 1991, however, refer to borings and consequently are here transferred to Rogerella de Saint-Seine, 1951 as new combinations, until a review of that ichnogenus is performed.
Vermiforichnus Cameron, 1969a and its type ichnospecies V. clarkei Cameron, 1969a were established as replacement ichnotaxa for Palaeosabella Clarke, 1921 and its type ichnospecies P. prisca (McCoy, 1855), respectively. Cameron (1969a) followed McCoy’s (1855) original interpretation, repeated by Fenton and Fenton (1932) that the holotype ofPalaeosabella consists of a single sponge boring with a central cavity and radiating tunnels. This way it would belong toTopsentia Clarke, 1921 rather than a cluster of individual clavate worm borings. A thorough reinvestigation of McCoy’s holotype by Plewes (1996) and the present authors confirmsPalaeosabella and its type ichnospecies as valid ichnotaxa for individual clavate borings. A comparison of the holotypes ofV. clarkei Cameron (1969a) and P. prisca (McCoy, 1855) shows that P. prisca is the senior synonym ofV. clarkei, and as a consequence, Vermiforichnus is synonymous with Palaeosabella.
Ichnotaxonomy by numbers
A total of 714 bioerosion ichnotaxa have been described since 1837, 480 (67%) of which are currently considered as valid. These are grouped into 18 ichnofamilies and 123 ichnogenera, constituting a total of 339 ichnospecies. The latter include 179 (53%) macroborings, 97 (28%) microborings, 26 (8%) predation traces, 26 (8%) attachment etchings, and 11 (3%) grazing traces (Fig. 2a), described from calcareous (81%), xylic (8%), osteic (8%), and siliceous (3%) substrates (Fig. 2b). Their known or inferred tracemakers (Fig. 2c) are invertebrates (79%), bacteria (6%), fungi (5%), vertebrates (4%), plants (3%), or unknown (3%).
Those 234 ichnotaxa that are currently recognized as invalid include one ichnofamily, 76 ichnogenera, and 157 ichnospecies. Among the latter are 65 (41%) subjective junior synonyms, one (1%) objective junior synonym, one (1%) primary homonym, two (1%) secondary junior homonyms, 55 (35%) nomina dubia, 21 (13%) nomina nuda, 2 (1%) ichnotaxa that are trace fossils unrelated to bioerosion, and 10 (6%) taxa that actually are to be regarded as body fossils and thus are biotaxa. As for the current group of nomina dubia, it shall be noted that in several cases ichnotaxa can potentially be stabilized by reinvestigation, improved definition and enhanced illustration of the type material with designation of suitable lectotypes where applicable, or, in those cases where the type material remains lost, the definition of neotypes.
Bioerosion ichnotaxonomy appears largely as a European domain, with the most productive protagonists being Richard G. Bromley, who established no fewer than 50 valid bioerosion ichnotaxa between 1970 and 2013, and Max Wisshak (52 ichnotaxa since 2005). Other ichnotaxonomists who established a considerable amount of the currently valid bioerosion ichnotaxa are Gudrun Radtke (33 ichnotaxa since 1991), Eduardo Mayoral (30 ichnotaxa since 1987), Markus Bertling (23 ichnotaxa since 2017), Radek Mikuláš (19 ichnotaxa since 1992), Dirk Knaust (18 ichnotaxa herein), and Ana Santos (15 ichnotaxa since 2003). Together, these eight authors have established more than a third of all valid bioerosion ichnotaxa.
The quantitatively by far most productive decades (Fig. 1) were the 2010s (97 ichnotaxa so far), closely followed by the 2000s (96 valid bioerosion ichnotaxa), and the 1990s (92 ichnotaxa). Milestones in ichnotaxonomy were the two compilations of known bioerosion ichnogenera by Häntzschel (1962, 1975) in the ‘Trace Fossils’ volumes of the ‘Treatise on Invertebrate Paleontology’. This is also in accordance with the marked increase in bioerosion research and publications since Neumann’s (1966) definition of the term bioerosion (Schönberg and Tapanila 2006), and was furthermore catalyzed by the initiation of the series of International Bioerosion Workshops (IBW) by Richard Bromley in 1996, the Workshops on Ichnotaxonomy (WIT) by Markus Bertling in 1998, and the International Congress on Ichnology (ICHNIA) by Jorge Genise and others in 2004.
Conclusions and outlook
The past few decades have seen a boost in bioerosion research and the establishment of new ichnotaxa describing bioerosion trace fossils. In fact, the number of valid bioerosion ichnotaxa has nearly quadrupled since the time of the last compilation of ichnogenera in the ‘Trace Fossils’ part of the ‘Treatise on Invertebrate Paleontology’ by Häntzschel (1975). The present inventory includes a total of 339 valid ichnospecies, grouped into 123 ichnogenera and 18 ichnofamilies. Such an ichnotaxonomic ‘radiation’ inevitably bears a tendency for splitting and has created numerous redundant (synonymous) or otherwise invalid ichnotaxa. We see the present compilation as an expression of the current phase of ichnotaxonomic consolidation, by identifying synonymous ichnotaxa, lumping and recombining ichnotaxa, and invalidating or excluding taxa that do not comply with the definition of a trace fossil in general, or a bioerosion trace fossil in particular.
Together with measures performed herein, the list of invalid bioerosion ichnotaxa now includes 157 invalid ichnospecies, 76 rejected ichnogenera, and one invalid ichnofamiliy. However, there remains a strong demand for ichnotaxonomic revisions of certain groups of bioerosion trace fossils, such as the wealth of ichnotaxa and biotaxa that have been erected for traces of ctenostome bryozoans (revision in progress). Likewise, there is a need to reinvestigate the most speciose ichnogenera, foremost Entobia Bronn, 1837 (53 ichnospecies),Gastrochaenolites Leymerie, 1842 (15 ichnospecies; revision in progress),Rogerella de Saint-Seine, 1951 (14 ichnospecies), and Talpina von Hagenow, 1840 (13 ichnospecies). Ultimately, this process is expected to maintain ichnotaxonomic stability, to raise awareness of the inventory of available bioerosion ichnotaxa, and to improve the basis for utilizing bioerosion traces as paleoenvironmental indicators.
References
Alekseev A, Endelman L (1989) Association of ectoparasitic prosobranch gastropods with Upper Cretaceous echinoid Galerites. [Assotsiatsija ektoparaziticheskikh perednezhabernikh gastropod c pozdnemelovimi morskimi ezhami Galerites]. In: Kaljo DL (ed) Fossil and recent echinoderm researches. Akademia Nauk Estonoskoi SSR, Tallinn, pp 166–174
Andrew C, Howe P, Paul CR, Donovan SK (2010) Fatally bitten ammonites from the lower Lias Group (Lower Jurassic) of Lyme Regis, Dorset. Proc Yorkshire Geol Soc 58:81–94
Annandale N (1920) Observations on “Physia prinsepii,” Sowerby and on a clionid sponge that burrowed in its shell. Rec Geol Surv India 51:50–64
Arendt YA (2012) Traces of Augoichnus dituberculatus gen. et sp. nov. on Hypermorphocrinus magnospinosus from the Lower Permian of Cisuralia. Paleontol J 46:886–893
Badve RM, Ghare MA (1984) Holocene trace fossils from beach rock of Velas coast, Raigad district, Maharashtra. Biovigyanam 10:165–172
Bałuk W, Radwański A (1991) A new occurrence of fossil acrothoracican cirripedes: Trypetesa polonica sp. n. in hermitted gastropod shells from the Korytnica Basin (Middle Miocene: Holy Cross Mountains, Central Poland), and its bearing on behavioral evolution of the genus Trypetesa. Acta Geol Polonica 41:1–36
Bassett MG, Popov LE, Holmer LE (2004) The oldest-known metazoan parasite? J Paleontol 78:1214–1216
Bassler RS (1952) Taxonomic notes on genera of fossil and recent Bryozoa. J Wash Acad Sci 42:381–385
Bengtson S, Conway Morris S, Cooper BJ, Jell PA, Runnegar BN (1990) Early Cambrian fossils from South Australia. Mem Ass Aust Palaeontols 9:1–264
Bertling M (1992) Arachnostega n. ichnog. – burrowing traces in internal moulds of boring bivalves (Late Jurassic, northern Germany). Pal Z 66:177–185
Bertling M (1995) Ropalonaria arachne (Fischer, 1866) eine Bryozoen–Bohrspur aus dem norddeutschen Malm. Münstersche Forsch Geol Pal 77:357–362
Bertling M (2007) What´s in a name? Nomenclature, systematics, ichnotaxonomy. In: Miller W III (ed) Trace fossils: concepts, problems, prospects. Elsevier, Amsterdam, pp 81–91
Bertling M, Rindsberg AK, Schlirf M, Nielsen JK, Mikuláš R, Genise J, Nielsen KSS, Uchman A, Bromley RG (2004) Comments on the draft proposal to emend the Code with respect to trace fossils (2). Bull Zool Nomencl 61:37–39
Bertling M, Braddy SJ, Bromley RG, Demathieu GR, Genise JF, Mikuláš R, Nielsen JK, Nielsen KSS, Rindsberg AK, Schlirf M, Uchman A (2006) Names for trace fossils: a uniform approach. Lethaia 39:265–286
Beuck L, López Correa M, Freiwald A (2008a) Biogeographical distribution of Hyrrokkin (Rosalinidae, Foraminifera) and its host-specific morphological and textural trace variability. In: Wisshak M, Tapanila L (eds) Current developments in bioerosion. Springer, Berlin, Heidelberg, pp 329–360
Beuck L, Wisshak M, Munnecke A, Freiwald A (2008b) A giant boring in a Silurian stromatoporoid analysed by computer tomography. Acta Palaeontol Polonica 53:149–160
Biernat G (1961) Diorygma atrypophilia n. gen., n. sp.—a parasitic organism of Atrypa zonata Schnur. Acta Palaeontol Polonica 6:17–28
Blissett DJ, Pickerill RK (2007) Systematic ichnology of microborings from the Cenozoic White Limestone Group, Jamaica, West Indies. Scripta Geol 134:77–108
Boekschoten GJ (1970) On bryozoan borings from the Danian at Fakse, Denmark. In: Crimes TP, Harper JC (eds) Trace fossils. Geol J, Spec Issue 3. Seel House Press, Liverpool, pp 43–48
Bohatý J, Ausich WI, Nardin E, Nyhuis C, Schröder S (2012) Coral-crinoid biocoenosis and resulting trace fossils from the Middle Devonian of the Eifel Synclines (Rhenish Massif, Germany). J Paleontol 86:282–301
Borkar VD, Kulkarni KG (1987) Cylindricavus perplexus ichnogen. et ichnosp. nov. from the Gulbarga District, Karnataka State. Biovigyanam 13:98–103
Botquelen A, Mayoral E (2005) Early Devonian bioerosion in the Rade de Brest, Armorican Massif, France. Palaeontology 48:1057–1064
Branson CC (1937) Stratigraphy and fauna of the Sacajawea Formation, Mississippian, of Wyoming. J Paleontol 11:650–660
Breton G (1992) Les Goniasteridae (Asteroidea, Echinodermata) Jurassiques et crétacés de France. Taphonomie, systématique, biostratigraphie, paléobiogéographie, évolution. Bulletin Trimesteriel de la Société Géologique de la Normandie, h.s. Supplement 78:1–590
Breton G (2011) Le Gault du Perthois: terriers, phosphates et petites crottes. Bull Ass Géol Auboise 31:1–66
Breton G (2015a) Unellichnus calciforans igen. nov., isp. nov., boring in a Maastrichtian lithified seafloor from the Cotentin (France). Annales de Paléontologie 101:143–151
Breton G (2015b) Le type de l’ichnotaxon Podichnus donovani Breton, 2011 est un bryozoaire cyclostome. Bull Ass Geol Auboise 35:7–8
Breton G (2017) Les sclérobiontes des huîtres du Cénomanien supérieur du Mans (Sarthe, France). Ann Paleontol 103:173–183
Breton G, Néraudeau D, Cuenca-Boulat C (1992) Gnathichnus stellarum ichnosp. nov., trace de broutage d’un echinide du Campanien des Charentes (France). Rev Paleobiol 11:219–229
Breton G, Wisshak M, Néraudeau D, Morel N (2017) Parasitic gastropod bioerosion trace fossil on Cenomanian oysters from Le Mans, France and its ichnologic and taphonomic context. Acta Palaeontol Polonica 62:45–57
Brett CE (1985) Tremichnus: a new ichnogenus of circular-parabolic pits in fossil echinoderms. J Paleontol 59:625–635
Bromley RG (1970a) Borings as trace fossils and Entobia cretacea Portlock, as an example. In: Crimes TP, Harper JC (eds) Trace fossils. Geol J, Spec Issue 3. Seel House Press, Liverpool, pp 49–90
Bromley RG (1970b) Predation and symbiosis in some Upper Cretaceous clionid sponges. Bull Geol Soc Denmark 19:398–405
Bromley RG (1972) On some ichnotaxa in hard substrates, with a redefinition of Trypanites Mägdefrau. Paläontol Z 46:93–98
Bromley RG (1975) Comparative analysis of fossil and recent echinoid bioerosion. Palaeontology 18:725–739
Bromley RG (1981) Concepts in ichnotaxonomy illustrated by small round holes in shells. Acta Geol Hispanica 16:55–64
Bromley RG (1993) Predation habits of octopus past and present and a new ichnospecies, Oichnus ovalis. Bull Geol Soc Den 40:167–173
Bromley RG (1994) The palaeoecology of bioerosion. In: Donovan SK (ed) The palaeobiology of trace fossils. Wiley, London, pp 134–154
Bromley RG (1996) Trace fossils: biology, taphonomy and application, 2nd edn. Chapman & Hall, London
Bromley RG (2004) A stratigraphy of marine bioerosion. Geol Soc Lond Spec Publ 228:455–479
Bromley RG, Asgaard U (1993) Endolithic community replacement on a Pliocene rocky coast. Ichnos 2:93–116
Bromley RG, D’Alessandro A (1983) Bioerosion in the Pleistocene of southern Italy: ichnogenera Caulostrepsis andMaeandropolydora. Riv Italiana Paleontol Stratigr 89:283–309
Bromley RG, D’Alessandro A (1984) The ichnogenus Entobia from the Miocene, Pliocene and Pleistocene of southern Italy. Riv Italiana Paleontol Stratigr 90:227–296
Bromley RG, D’Alessandro A (1987) Bioerosion of the Plio-Pleistocene transgression of southern Italy. Riv Italiana Paleontol Stratigr 93:379–442
Bromley RG, D’Alessandro A (1989) Ichnological study of shallow-marine endolithic sponges from the Italian coast. Riv Italiana Paleontol Stratigr 95:279–314
Bromley RG, Martinell J (1991) Centrichnus, new ichnogenus for centrically patterned attachment scars on skeletal substrates. Bull Geol Soc Denmark 38:243–252
Bromley RG, Surlyk F (1973) Borings produced by brachiopod pedicles, fossil and recent. Lethaia 6:349–365
Bromley RG, Wisshak M, Glaub I, Botquelen A (2007) Ichnotaxonomic review of dendriniform borings attributed to foraminiferans: Semidendrina igen. nov. In: Miller W III (ed) Trace fossils: concepts, problems, prospects. Elsevier, Amsterdam, pp 518–530
Bromley RG, Uchman A, Kolodziej B, Kedzierski M (2009) Large chambered sponge borings on a Late Cretaceous abrasion platform at Cracow, Poland. Cretac Res 30:149–160
Bronn HG (1837) Lethaea Geognostica, Abbildungen und Beschreibungen der für die Gebirgs-Formationen bezeichnendsten Versteinerungen, Erster Band. Schweizerbart, Stuttgart
Bronn HG (1848) Index Palaeontologicus, 1. Abtheilung, 1. Hälfte. Schweizerbart, Stuttgart
Buatois LA, Wisshak M, Wilson MA, Mángano MG (2017) Categories of architectural designs in trace fossils: a measure of ichnodisparity. Earth Sci Rev 164:102–181
Buchholz H (1986) Die Höhle eines Spechtvogels aus dem Eozän von Arizona, USA (Aves, Piciformes). Verhandlungen des Naturwissenschaftlichen Vereins in Hamburg, N F 28:5–25
Bundschuh M, Balog SJ (2000) Fasciculus rogus nov. isp., an endolithic trace fossil. Ichnos 7:149–152
Bystrov AP (1956) On the damage caused by fungi in the skeletal elements of fossil animals. Vestnik Leningradskogo Universiteta, Seria Geologii i Geografii 18:30–46 [in Russian]
Bystrow AP (1959) The microstructure of skeleton elements in some vertebrates from lower Devonian deposits of the USSR. Acta Zool 40:59–83
Cadée GC, de Wolf P (2013) Belichnus traces produced on shells of the bivalve Lutraria lutraria by gulls. Ichnos 20:15–18
Calvin S (1888) On a new genus and new species of tubicolar Annelida. Am Geol 1:24–28
Cameron B (1969a) New name for Palaeosabella prisca (McCoy), a Devonian worm-boring, and its preserved probable borer. J Paleontol 43:189–192
Cameron B (1969b) Paleozoic shell-boring annelids and their trace fossils. Am Zool 9:689–703
Campbell SE, Kazmierczak J, Golubić S (1979) Palaeoconchocelis starmachii n. gen., n. sp., a Silurian endolithic rhodophyte (Bangiaceae). Acta Palaeontol Polonica 24:405–408
Casadío S, Marenssi S, Santillana S (2001) Endolithic bioerosion traces attributed to boring bryozoans in the Eocene of Antarctica. Ameghiniana 38:321–329
Casey R (1961) The stratigraphical paleontology of the Lower Greensand. Palaeontology 3:487–621
Chapman F (1907) New or little known Victorian fossils in the National Museum. Part IX - Some Tertiary species. Proc R Soc Vic 20:208–221
Chatterton BDE (1975) A commensal relationship between a small filter feeding organism and Australian Devonian spiriferid brachiopods. Paleobiology 1:371–378
Chiplonkar G, Ghare M (1976) Paleolithophaga, a new ichnogenus from south Indian Cretaceous rocks of Trichinopoly district. Biovigyanam 2:161–164
Chiplonkar G, Ghare M (1977) Serpulid and barnacle borings on South Indian Cretaceous fossils from Trichinopoly District, Tamil Nadu. Biovigyanam 3:193–204
Chow MM (1957) A new Cliona-like boring sponge from the Devonian of Hunan [in Chinese]. Acta Palaeontol Sinica 5:63–68
Chumakov NM, Dronov AV, Mikuláš R (2013) New ichnospecies of scratching traces from phosphatic nodules (Cenomanian, England). Stratigr Geol Correl 21:291–299
Clarke JM (1908) The beginnings of dependent life. N Y State Mus Bull 121:146–169
Clarke JM (1921) Organic dependence and disease, their origin and significance. N Y State Mus Bull 221–222:1–113
Cleevely RJ (1983) World palaeontological collections. Mansell Publishing, London
Codez J, de Saint-Seine R (1958) Révision des Cirripèdes acrothoraciques fossiles. Bull Soc Géol France (série 6) 7:699–719
Cole AR, Palmer TJ (1999) Middle Jurassic worm borings, and a new giant ichnospecies of Trypanites from the Bajocian/Dinantian unconformity, southern England. Proc Geol Ass 110:203–209
Collinson ME, Hooker JJ (2000) Gnaw marks on Eocene seeds: evidence for early rodent behaviour. Palaeogeogr Palaeoclimatol Palaeoecol 157:127–149
Condra GE, Elias MK (1944) Carboniferous and Permian ctenostomatous bryozoa. Bull Geol Soc Am 55:517–568
Cruickshank A (1986) Archosaur predation on an east African Middle Triassic dicynodont. Palaeontology 29:415–422
d’Archiac A, Fischer P, de Verneuil E (1866) Asie Mineure. Description physique de cette contrée—Paléontologie. Librairie Théodore Morgand, Paris
d’Orbigny A (1847) Voyage dans l’Amérique Méridionale. 5 [Zoophytes]:7–28
d’Orbigny A (1850) Prodrome de Paléontologie stratigraphique universelle des animaux mollusques et rayonnés, vol 2. V. Masson, Paris
de Araújo-Júnior HI, de Souza Barbosa FH, da Silva LHM (2017) Overlapping paleoichnology, paleoecology and taphonomy: analysis of tooth traces in a Late Pleistocene–early Holocene megafaunal assemblage of Brazil and description of a new ichnotaxon in hard substrate. Palaeogeogr Palaeoclimatol Palaeoecol 468:122–128
de Laubenfels MW (1955) Porifera. In: Moore RC (ed) Treatise on invertebrate paleontology, Part E—Porifera. University of Kansas Press, Lawrence, pp E21–E112
de Saint-Seine R (1951) Un Cirripèdes acrothoraciques du Crétacé:Rogerella lecointrei nov. gen., nov. sp. Comptes Rendus de l’Académie des Sciences 233:1051–1054
de Saint-Seine R (1954) Existence de Cirripèdes acrothoraciques dès de Lias: Zapfella pattei nov. gen., nov. sp. Bull Soc Géol France 6(série 4):447–451
de Saint-Seine R (1955) Les Cirripèdes acrothoraciques échinicoles. Bull Soc Géol France 6(série 5):299–303
de Serres M (1840) Description de quelques mollusques fossiles nouveaux des terrains infra-liassiques et de la craie compacte inferieure du Midi de la France. Ann Sci Nat (Zoologie) 14:5–25
Dogiel V (1908) Entobius loimiae n. g. n. sp., eine endoparasitische Copepode. Zool Anz 33:561–567
Dong Y, Ji Q, Wang S (1984) The discovery of microscopic trace fossils in the Sinian Doushantuo Formation in southwestern China [in Chinese]. Scientia Geol Sinica 1984:346–347
Donovan SK (2002) A new ichnospecies of Gastrochaenolites Leymerie from the Pleistocene Port Morant Formation of southeast Jamaica and the taphonomy of calcareous linings in clavate borings. Ichnos 9:61–66
Donovan SK (2018) A new ichnogenus for Teredolites longissimus Kelly and Bromley. Swiss J Palaeontol 137:95–98
Donovan SK, Jagt JWM (2002) Oichnus Bromley borings in the irregular echinoid Hemipneustes Agassiz from the type Maastrichtian (Upper Cretaceous, The Netherlands and Belgium). Ichnos 9:67–74
Donovan SK, Jagt JWM (2005) Flosculichnus tectus, an etched attachment scar from the Upper Cretaceous (Maastrichtian) of The Netherlands. Bulletin de l’Institut Royal des Sciences Naturelles de Belgique, Sciences de la Terre 75:207–210
Duchassaing P (1850) Animaux radiaires des Antilles. Plon Frères, Paris
Duchassaing de Fonbressin P, Michelotti GJ (1864) Spongiaires de la Mer Caraibe. De Erver Loosjes, Haarlem
Duncan PM (1876) On some unicellular algae parasitic within Silurian and Tertiary corals, with a notice of their presence in Calceola sandalina and other fossils. Q J Geol Soc 32:205–211
Edinger EN, Risk MJ (1994) Oligocene-Miocene extinction and geographic restriction of Caribbean corals: roles of turbidity, temperature, and nutrients. Palaios 9:576–598
Ekdale AA, Bromley RG (2001) Bioerosional innovation for living in carbonate hardgrounds in the Early Ordovician of Sweden. Lethaia 34:1–12
Ekdale AA, Brown FH, Feibel CS (1989) Nonmarine macroborings in early Pleistocene algal biolithites (stromatolites) of the Turkana Basin, northern Kenya. Palaios 4:389–396
Elias M (1957) Late Mississippian fauna from the Redoak Hollow formation of southern Oklahoma, part 1. J Paleontol 31:370–427
Elias M (1958) Late Mississippian fauna from the Redoak Hollow formation of southern Oklahoma, part 4. J Paleontol 32:1–57
Elias RJ (1980) Borings in solitary rugose corals of the Selkirk Member, Red River formation (late Middle or Upper Ordovician), southern Manitoba. Can J Earth Sci 17:272–277
Elliott GF (1962) More microproblematica from the Middle East. Micropaleontology 8:29–44
Erickson JM, Bouchard TD (2003) Description and interpretation ofSanctum laurentiensis, new ichnogenus and ichnospecies, a domichnium mined into Late Ordovician (Cincinnatian) ramose bryozoan colonies. J Paleontol 77:1002–1010
Étallon A (1859) Sur la classification des Spongiaires du Haut-Jura et leur distribution dans les étages. Actes de la Société Jurassienne d’Émulation 9:129–160
Étallon A (1864) Etudes paleontologiques sur le Jura graylois, terrains jurassiques moyen et supérieur. Mémoires de la Societé d’Émulation du Département de Doubs, 3. Série 8:221–506
Etheridge R (1891) On the occurrence of microscopic fungi, allied to the genus Palæachlya, Duncan, in the Permo-Carboniferous rocks of N. S. Wales and Queensland. Rec Geol Surv N S W 2:95–99
Etheridge R (1899) On two additional perforating bodies, believed to be thallophytic cryptograms, from the Lower Palaeocoic rocks of N. S. Wales. Rec Aust Mus 3:121–127
Etheridge R (1904) An endophyte (Stichus mermisoides) occurring in the test of a Cretaceous bivalve. Rec Aust Mus 5:255–257
Fenton CL, Fenton MA (1932) Boring sponges in the Devonian of Iowa. Am Midl Nat 13:42–54
Fischer MP (1866) Étude sur les Bryozoaires perforants de la famille des Térébriporides. Nouvelles Archives du Museum d’Histoire Naturelle de Paris 2:293–313
Fischer MP (1868) Recherches sur les Eponges perforantes fossiles. Nouvelles Archives du Museum d’Histoire Naturelle de Paris 4:16–170
Fischer MP (1875) D´un type de sarcodaires. J de Zoologie 4:530–533
Fraas O (1867) Aus dem Orient. Geologische Beobachtungen am Nil, auf der Sinai-Halbinsel und in Syrien. Ebner & Seubert, Stuttgart
Fraas O (1869) Begleitworte zur geognostischen Specialkarte von Württemberg - Atlasblatt Giengen mit den Umgebungen von Dischingen, Nattheim und Niederstotzingen. Königlich statistisch-topographisches Büro, Stuttgart
Francischini H, Paes Neto VD, Martinelli AG, Pereira VP, Marinho TS, Teixeira VPA, Ferraz MLF, Soares MB, Schultz CL (2016) Invertebrate traces in pseudo-coprolites from the Upper Cretaceous Marilia formation (Bauru Group), Minas Gerais State, Brazil. Cretac Res 57:29–39
Frest TJ, Strimple HL, Paul CRC (2011) The North American Holocystites fauna (Echinodermata, Blastozoa: Diploporita): paleobiology and systematics. Bull Am Paleontol 380:1–133
Frič A (1883) Studien im Gebiete der böhmischen Kreideformation. III. Die Iser-Schichten. Archiv für die naturwissenschaftlichen Landesdurchforschung von Böhmen 5:1–140
Fritsch A (1908) Problematica Silurica. Gerhard, Prag
Furlong CM, McRoberts CA (2014) Commensal borings from the Middle Devonian of central New York, ecologic and taxonomic review of Clionoides, Clionolithes, and Canaliparva n. ichnogen. J Paleontol 88:130–144
Fürsich FT, Palmer TJ, Goodyear KL (1994) Growth and disintegration of bivalve-dominated patch reefs in the Upper Jurassic of southern England. Palaeontology 37:131–171
Genise JF (1995) Upper Cretaceous trace fossils in permineralized plant remains from Patagonia, Argentina. Ichnos 3:287–299
Genise JF (2017) Ichnoentomology—insect traces in soils and Paleosols. Topics in Geobiology 37. Springer, Heidelberg Berlin
Genise JF, Hazeldine PL (1995) A new insect trace fossil in Jurassic wood from Patagonia, Argentina. Ichnos 4:1–5
Genise JF, Garrouste R, Nel P, Grandcolas P, Maurizot P, Cluzel D, Cornette R, Fabre A-C, Nel A (2012) Asthenopodichnium in fossil wood: different trace makers as indicators of different terrestrial palaeoenvironments. Palaeogeogr Palaeoclimatol Palaeoecol 365:184–191
Ghare MA (1982) Borings on belemnoid rostra from Utatur Group of Upper Cretaceous rocks of Trichinopoly district, Tamil Nadu. Geol Soc India 23:129–135
Ghare MA (1985) Clionid sponge borings from South Indian Cretaceous rocks, Tiruchirapally District, Tamil Nadu. Biovigyanam 112:177–182
Glaub I (1994) Mikrobohrspuren in ausgewählten Ablagerungsräumen des europäischen Jura und der Unterkreide (Klassifikation und Palökologie). Courier Forschungsinstitut Senckenberg 174:1–324
Głazek J, Marcinowski R, Wierzbowski A (1971) Lower Cenomanian trace fossils and transgressive deposits in the Cracow Upland. Acta Geol Polonica 21:433–448
Golubić S, Radtke G (2008) The trace Rhopalia clavigera isp. n. reflects the development of its makerEugomontia sacculata Kornmann, 1960. In: Wisshak M, Tapanila L (eds) Current developments in bioerosion. Springer, Berlin, Heidelberg, pp 95–108
Golubić S, Brent G, Le Campion T (1970) Scanning electron microscopy of endolithic algae and fungi using a multipurpose casting-embedding technique. Lethaia 3:203–209
Golubić S, Radtke G, Campbell SE, Lee S-J, Vogel K, Wisshak M (2014) The complex fungal microboring trace Saccomorpha stereodiktyon isp. nov. reveals the growth strategy of its maker. Ichnos 21:100–110
Goujet D, Locquin MV (1979) Découverte de spores fongiques dans les écailles de Poissons et d’Agnathes Paléozoïques: Mycobystrovia lepidophaga gen. et sp. nov. In: Proceedings of the 104e Congrès national des Sociétés savantes, Bordeaux, pp 87–99
Gradstein FM, Ogg JG, Schmitz M, Ogg G (2012) The Geologic Time Scale. Elsevier, Amsterdam
Grant RE (1826) Notice of a new zoophyte (Cliona celata, Gr.) from the Firth of Forth. Edinburgh New Philosoph J 1:78–81
Green JW, Knoll AH, Swett K (1988) Microfossils from Oolites and Pisolites of the Upper Proterozoic Eleonore Bay Group, Central East Greenland. J Paleontol 62:835–852
Gripp K (1967) Polydora biforans n. sp., ein in Belemniten-Rostren bohrender Wurm der Kreidezeit. Meyniana 17:9–10
Gruber A (1933) Bohrorganismen im oberen Muschelkalk. Geol Rundsch, Leipzig Festschrift 23a:263–266
Guo S (1991) A Miocene trace fossil of insect from Shanwang Formation in Linqu, Shandong. Acta Palaeontol Sinica 30:739–742
Haga T, Kurihara Y, Kase T (2010) Reinterpretation of the Miocene sea-snake egg Moniopterus japonicus as a boring of rock-boring bivalve Lithophaga (Mytilidae: Mollusca). J Paleontol 84:848–857
Hancock A (1849) On the excavating powers of certain sponges belonging to the genus Cliona; with descriptions of several new species, and an allied generic form. Ann Mag Nat Hist (2nd series) 3:321–348
Häntzschel W (1962) Trace fossils and problematica. In: Moore RC (ed) Treatise on Invertebrate Paleontology, Part W—Miscellanea. University of Kansas Press, Lawrence, pp W177–W245
Häntzschel W (1975) Trace fossils and problematica. In: Moore RC (ed) Treatise on invertebrate paleontology, Part W—Supplement I, trace fossils. University of Kansas Press, Lawrence, pp W1–W269
Hary A (1975) Inventaire des traces d’activité animale dans les sediments Mésozoiques du territoire Luxembourgeois. Publications du Service Géologique du Luxembourg 23:91–175
Hatai K, Masuda K, Noda H (1974) Marine fossils from the Moniwa Formation along the Natori River, Sendai, northeast Honshu, Japan, part 2. Problematica from the Moniwa Formation. Trans Proc Palaeontol Soc Jpn New Ser 95:364–371
Hillmer G, Schulz M (1973) Ableitung der Biologie und Ökologie eines Polychaeten der Oberkreide durch Analyse des Bohrganges Ramosulcichnus biforans (Gripp) nov. ichnogen. Mitt Geol-Paläontol Inst Univ Hamburg 42:5–24
Ho J-S (1984) New family of poecilostomatoid copepods (Spiophanicolidae) parasitic on polychaetes from southern California, with a phylogenetic analysis of nereicoliform families. J Crustacean Biol 4:134–146
Hofmann K (1996) Die mikro-endolithischen Spurenfossilien der borealen Oberkreide Nordwest-Europas und ihre Faziesbeziehungen. Geol Jb, A 136:1–151
Höpner S, Bertling M (2017) Holes in bones: ichnotaxonomy of bone borings. Ichnos 24:259–282
Hunt AP, Lucas SG, Klein K (2018) Late Triassic nonmarine vertebrate and invertebrate trace fossils and the pattern of the Phanerozoic record of vertebrate trace fossils. In: Tanner LA (ed) The Late Triassic World. Topics in Geobiology 46. Springer, Berlin, Heidelberg, pp 447–544
Hyde JE (1953) Mississippian formations of central and southern Ohio. Ohio Geol Surv, Bull 51:214–221
ICZN [International Commission for Zoological Nomenclature] (1999) International Code of Zoological Nomenclature, adopted by the International Union of Biological Sciences, 4th edn. International Trust for Zoological Nomenclature, London
Ishikawa M, Kase T (2007) Spionid bore hole Polydorichnus subapicalis new ichnogenus and ichnospecies: a new behavioral trace in gastropod shells. J Paleontol 81:1466–1475
Jacobsen AR, Bromley RG (2009) New ichnotaxa based on tooth impressions on dinosaur and whale bones. Geol Q 53:373–382
Jagt JWM, Neumann C, Donovan SK (2009) Petroxestes altera, a new bioerosional trace fossil from the upper Maastrichtian (Cretaceous) of northeast Belgium. Bulletin de l’Institut royal des Sciences naturelles de Belgique Sciences de la Terre 79:137–145
Jarzembowski EA (1990) A boring beetle from the Wealden of the Weald. In: Boucot A (ed) Evolutionary paleobiology of behaviour and coevolution. Elsevier, Amsterdam, pp 373–376
Karl H-V, Nyhuis CJ (2012) Ctenochelys stenopurus (Hay, 1905) (Testudines: Toxochelyidae) and Clidastes sp. (Squamata: Mosasauridae) from the upper Cretaceous of NW-Germany. Studia Palaeocheloniologica 4 (Studia Geologica Salamanticensia, volumen especial 9):129–142
Karl H-V, Gröning E, Brauckmann C (2012) Revision of Tropidemys seebachi Portis, 1878 (Testudines: Eucryptodira) from the Kimmeridgian (Late Jurassic) of Hanover (Northwestern Germany). Studia Palaeocheloniologica 4 (Studia Geologica Salamanticensia, volumen especial 9):11–24
Karpiński JJ (1962) Cast of the brood galleries of fossil beetle of the Scolytidae family from Oligocene/Miocene Sandstone at Osieczów (Lower Silesia). Pr Inst Geol 30:237–239
Kelly SR, Bromley RG (1984) Ichnological nomenclature of clavate borings. Palaeontology 27:793–807
Kennedy W (1970) Trace fossils in the Chalk environment. In: Crimes TP, Harper JC (eds) Trace fossils. Geol J, Spec Issue 3. Seel House Press, Liverpool, pp 263–282
Kern JP, Grimmer JC, Lister KH (1974) A new fossil spionid tube, Pliocene and Pleistocene of California and Baja California. J Paleontol 48:978–982
Kleemann K (2009) Gastrochaenolites hospitium isp. nov., trace fossil by a coral-associated boring bivalve from the Eocene and Miocene of Austria. Geol Carpath 60:339–342
Klompmaker AA, Artal P, van Bakel BWM, Fraaije RHB, Jagt JWM (2014) Parasites in the fossil record: a Cretaceous fauna with isopod-infested decapod crustaceans, infestation patterns through time, and a new ichnotaxon. PLoS ONE 9:e92551
Klompmaker AA, Portell RW, van der Meij ET (2016) Trace Fossil evidence of coral-inhabiting crabs (Cryptochiridae) and its implications for growth and paleobiogeography. Sci Rep 6:23443
Knaust D (2012) Trace fossil systematics. In: Knaust D, Bromley RG (eds) Trace fossils as indicators of sedimentary environments. Developments in Sedimentology 64. Elsevier, Amsterdam, pp 9–102
Knaust D (2019) The enigmatic trace fossil Tissoa de Serres, 1840. Earth Sci Rev 188:123–147
Lamouroux JVF (1816) Histoire des Polypiers Coralligènes Flexibles, Vulgairement Nommés Zoophytes. F. Poisson, Caen
Lees JH, Thomas A (1918) The Ste. Genevieve Marls near Fort Dodge and their fauna. Proc Iowa Acad Sci 25:599–616
Lehner L (1937) Fauna und Flora der frankischen albüberdeckenden Kreide. Palaeontographica 87A:159–165
Leymerie A (1842) Suite de mémoire sur le terrain Crétacé du département de l’Albe. Mem Soc Géol France 1(série 4):1–34
Li G (1997) Early Cambrian phosphate-replicated endolithic algae from Emei, Sichuan, SW China. Bull Nat Mus Nat Sci 10:193–216
Linck O (1949) Fossile Bohrgänge (Anobichnium simile n.g. n.sp.) an einem Keuperholz. N Jb Mineral Geol Paläontol, B 4–6:180–185
Loeblich AR, Tappan H (1964) Sarcodina, chiefly “Thecamoebians”, Foraminiferida. In: Moore RC (ed) Treatise on Invertebrate Paleontology, Part C - Protista/Protoctista 2. University of Kansas Press, Lawrence, pp C1–C900
Lopes RP, Pereira JC (2018) Molluscan grazing traces (ichnogenusRadulichnus Voigt, 1977) on a Pleistocene bivalve from southern Brazil, with the proposal of a new ichnospecies. Ichnos. https://doi.org/10.1080/10420940.2018.1532898
Low ME, Guinot D (2010) Feldmannius nom. nov. for Feldmannia Casadío, Marenssi & Santillana, “30-09-2001”, preoccupied by Feldmannia Guinot & Tavares, “28 septembre 2001”: a case of 48-hour precedence. Zootaxa 2568:67–68
Macquart PJM (1838) Diptères Exotiques Nouveaux ou Peu Connus, Tome prémier, 2eme partie. Roret, Paris
Mägdefrau K (1932) Über einige Bohrgänge aus dem Unteren Muschelkalk von Jena. Paläontol Z 14:150–160
Mägdefrau K (1937) Lebensspuren fossiler “Bohr” = Organismen. Beiträge zur naturkundlichen Forschung in Südwestdeutschland 2:54–67
Magwood JPA (1992) Ichnotaxonomy: a burrow by any other name…? In: Maples CG, West RR (eds) Trace Fossils. Palaeontological Society of America, Knoxville, pp 15–33
Malkowski K (1975) Attachment scars of the brachiopod Coenothyris vulgaris (Schlotheim, 1820) from the Muschelkalk of Upper Silesia. Acta Geol Polonica 25:277–283
Mantell GA (1850) A Pictorial Atlas of Fossil Remains, Consisting of Coloured Illustrations Selected from Parkinson’s “Organic Remains of a Former World” and Artis’s “Antediluvian Phytology”. H. G. Bohn, London
Marek J (1982) Runia runica ichnogen. et ichnosp. nov., a new sponge boring from the Silurian of Bohemia. Acta Universitatis Carolinae, Geologica 4:401–408
Martinell J, Domènech R (2009) Commensalism in the fossil record: Eunicid polychaete bioerosion on Pliocene solitary corals. Acta Palaeontol Polonica 54:143–154
Martinell J, Domènech R, Bromley RG (1999) Mysterious boring hidden within the hinge plates of heterodont bivalves. Bull Geol Soc Denmark 45:161–163
Mayer G (1952) Lebensspuren von Bohrorganismen aus dem Unteren Hauptmuschelkalk (Trochitenkalk) des Kraichgaus. N Jb Geol Paläontol, Mh 1952:450–456
Mayer K (1872) Systematisches Verzeichnis der Versteinerungen des Helvetian der Schweiz und Schwabens. Schabelitz’sche Buchhandlung, Zürich
Mayoral E (1987a) Acción bioerosiva de Mollusca (Gastropoda, Bivalvia) en el Plioceno inferior de la Cuenca del Bajo Guadalquivir. Rev Española Paleontol 2:49–58
Mayoral E (1987b) Stellichnus nov. icnogen., huellas de incrustación atribuidas a Paravinella nov. gen. (Bryozoa, Ctenostomata) de la Formación Arenas de Huelva (Plioceno inferior) en la Cuenca del Bajo Guadalquivir (Espana). Rev Española Paleontol 2:33–40
Mayoral E (1988) Microperforaciones (Tallophyta) sobre bivalvia del Plioceno del Bajo Guadalquivir. Importancia paleoecologica. Estudios Geologicos 44:301–316
Mayoral E (1991) Actividad bioerosiva de Briozoos Ctenostomados en el Ordovícico Superior de la zona cantábrica del Macizo Hespérico (Cabo Vidrias, Oviedo). Rev Española Paleontol 6:27–36
Mayoral E, Gutiérrez Marco JC, Martinell J (1994) Primas evidencas de briozoos perforantes (Ctenostomata) en braquiópodos Ordovícicos de los Montes de Toledo (Zona centroibérica Meridional, España). Rev Española Paleontol 9:185–194
McCoy F (1850) On some new genera and species of Silurian Radiata in the collection of the University of Cambridge. Ann Mag Nat Hist (2nd series) 6:270–290
McCoy F (1855) Description of the British Palæozoic Fossils in the Geological Museum of the University of Cambridge. Cambridge University Press, Cambridge
McKinney FK (2009) Bryozoan-hydroid symbiosis and a new ichnogenus,Caupokeras. Ichnos 16:193–201
McLoughlin N, Furnes H, Banerjee NR, Muehlenbachs K, Staudigel H (2009) Ichnotaxonomy of microbial trace fossils in volcanic glass. J Geol Soc London 166:1–11
Mercier J (1936) Zoothylacies d’Echinide fossile provoquées par un Crustacé: Castexia duvillei nov. gen., nov. sp. Bull Soc Géol France (série 5) 6:149–154
Michalík J (1977) Systematics and ecology of Zeilleria Bayle and other brachiopods in the uppermost Triassic of the West Carpathians. Geol Carpath 28:323–346
Michalík J (1980) A palaeoenvironmental and palaeoecological analysis of the West Carpathian part of the northern Tethyan nearshore region in the latest Triassic times. Riv Italiana Paleontol Stratigr 85:1047–1064
Michelin H (1846) Note sur différentes espèces du genre Vioa (famille des Spongiaires). In: Guérin-Méneville MD (ed) Zoologique, par la Société Cuvierienne. Au Bureau de la Revue Zoologique, Paris, pp 56–60
Michelin H (1847) Iconographie Zoophytologique. Bertrand, Paris [1840–1847; text + atlas]
Michelotti G (1861) Études sur le Miocène Inférieur de l’Italie septentrionale. Société Hollandaise des Sciences à Harlem, Les Héritiers Loosjes, Harlem
Mikuláš R (1992) Early Cretaceous borings from Štramberk (Czechoslovakia). Časopis pro Mineralogii a Geologii 37:297–312
Mikuláš R, Cílek V (1998) Terrestrial insect bioerosion and the possibilities of its fossilization (Holocene to Recent, Czech Republic). Ichnos 5:325–333
Mikuláš R, Dvořák Z (2002) Vrtby v drevitych tkanich stromovych kapradin rodu Tempskya v ceske kridove panvi [Borings in xylic tissues of the tree fern Tempskya in the Bohemian Cretaceous Basin, Czech Republic]. Zpravy o geologickych vyzkumech 2002:129–131
Mikuláš R, Dvorák Z, Pek I (1998) Lamniporichnus vulgaris igen. et isp. nov.: traces of insect larvae in stone fruits of hackberry (Celtis) from the Miocene and Pleistocene of the Czech Republic. J Czech Geol Surv 43:277–280
Mikuláš R, Kadlecová E, Fejfar O, Dvořák Z (2006) Three new ichnogenera of biting and gnawing traces on reptilian and mammalian bones: a case study from the Miocene of the Czech Republic. Ichnos 13:113–127
Morningstar H (1922) Pottsville Fauna of Ohio. Bull Geol Surv Ohio 25:1–312
Morris J (1851) Palaeontological notes. Ann Mag Nat Hist (2nd series) 8:85–90
Müller J (1851) Monographie der Petrefacten der Aachener Kreideformation. Henry & Cohen, Bonn
Müller G (1968) Bohr-Röhren von unbekannten Anneliden und anderen Organismen in unterdevonischen Brachiopodenklappen aus der Eifel und dem Siegerland, Rheinisches Schiefergebirge. Dissertation, Universität zu Köln
Müller AH (1977) Zur Ichnologie der subherzynen Oberkreide (Campan). Z Geol Wiss Berlin 5:881–897
Muñiz F, de Gibert JM, Esperante R (2010) First trace-fossil evidence of bone eating worms in whale carcasses. Palaios 25:269–273
Murphy AE, Williams JD (2013) New records of two trypetesid burrowing barnacles (Crustacea: Cirripedia: Acrothoracica: Trypetesidae) and their predation on host hermit crab eggs. J Mar Biol Ass U K 93:107–133
Muszer J, Uglik M (2013) Palaeoenvironmental reconstruction of the upper Visean Paprotnia Beds (Bardo Unit, Polish Sudetes) using ichnological and palaeontological data. Geol Q 57:365–384
Nardo GD (1833) Spongiariorum classificatio. Isis—encyclopädische Zeitschrift, vorzüglich für Naturgeschichte, vergleichende Anatomie und Physiologie 1833:519–524
Nardo GD (1845) Alcune tracce d’animali parassiti delle Placune. Atti della sesta riunione degli scienziati Italiani tenuta in Milano nel Settembre del MDCCCXLIV. Pirola, Milano, pp 428–430
Neumann AC (1966) Observations on coastal erosion in Bermuda and measurements of the boring rate of the sponge Cliona lampa. Limnol Oceanogr 11:92–108
Neumann C, Wisshak M (2009) Gastropod parasitism on Late Cretaceous to early Paleocene holasteroid echinoids – evidence from Oichnus halo isp. n. Palaeogeogr Palaeoclimatol Palaeoecol 284:115–119
Neumann C, Wisshak M, Bromley RG (2008) Boring a mobile domicile: an alternative to the conchiculous life habit. In: Wisshak M, Tapanila L (eds) Current Developments in Bioerosion. Springer, Berlin Heidelberg, pp 307–328
Nielsen JK (2002) Borings formed by Late Cretaceous endobiontic foraminifers within larger benthic foraminifers. Acta Palaeontol Polonica 47:673–678
Nielsen JK, Görmüş M (2004) Ichnotaxonomy and ethology of borings in shallow-marine benthic foraminifers from the Maastrichtian and Eocene of northwestern and southwestern Turkey. Riv Italiana Paleontol Stratigr 110:493–501
Nielsen KSS, Nielsen JK (2001) Bioerosion in Pliocene to late Holocene tests of benthic and planktonic foraminiferans, with a revision of the ichnogenera Oichnus and Tremichnus. Ichnos 8:99–116
Nielsen KSS, Nielsen JK, Bromley RG (2003) Palaeoecological and ichnological significance of microborings in Quaternary Foraminifera. Palaeontologica Electronica 6:1–13
Norman AM (1897) Museum Normanianum, or A catalogue of the Invertebrata of Europe, and the Arctic and North Atlantic oceans, which are contained in the collection of the Rev. Canon A. M. Norman. No. 9, Morton, Houghton-Le-Spring
Norman AM (1903) New generic names for some Entomostraca and Cirripedia. Ann Mag Nat Hist (7th series) 11:367–369
Oekentorp K (1969) Kommensalismus bei Favosiden. Münstersche Forsch Geol Paläontol 12:165–217
Olempska E, Wacey D (2016) Ambient inclusion trails in Palaeozoic crustaceans (Phosphatocopina and Ostracoda). Palaeogeogr Palaeoclimatol Palaeoecol 441:949–958
Oliver WA Jr (1983) Symbioses of Devonian rugose corals. Mem Ass Australasian Palaeontol 1:261–274
Paes Neto VD, Parkinson AH, Pretto FA, Soares MB, Schwanke C, Schultz CL, Kellner AW (2016) Oldest evidence of osteophagic behavior by insects from the Triassic of Brazil. Palaeogeogr Palaeoclimatol Palaeoecol 453:30–41
Palmer TJ, Palmer CD (1977) Faunal distribution and colonization strategy in a Middle Ordovician hardground community. Lethaia 10:179–199
Palmer TJ, Plewes C (1993) Borings and bioerosion in fossils. Geol Today 9:138–142
Palmer TJ, Wilson MA (1988) Parasitism of Ordovician bryozoans and the origin of pseudoborings. Palaeontology 31:939–949
Parkinson AH (2016) Traces of Insect Activity at Cooper’s D Fossil Site (Cradle of Humankind, South Africa). Ichnos 23:322–339
Pemberton SG, Jones B, Edgecombe G (1988) The influence ofTrypanites in the diagenesis of Devonian stromatopoids. J Paleontol 62:22–31
Peña LE (1971) Evidencia de insectos en maderas petrificadas halladas en lugares adyacentes al Estrecho de Magallanes. Annales del Museo de Historia Natural de Valparaíso 4:345–348
Pether J (1995) Belichnus new ichnogenus, a ballistic trace on mollusc shells from the Holocene of the Benguela region, South Africa. J Paleontol 69:171–181
Petrov AV (2013) New ichnotaxon Megascolytinus zherikhini (Coleoptera: Curculionidae: Scolytinae) from Upper Cretaceous deposits of Mongolia. Paleontol J 47:597–600
Philippi A (1843) Rhopalaea ein neues Genus der einfachen Ascidien. Archiv für Anatomie und Physiologie 1:45–47
Pickerill RK (1994) Nomenclature and taxonomy of invertebrate trace fossils. In: Donovan SK (ed) The Palaeobiology of Trace Fossils. Wiley, London, pp 3–42
Pickerill RK, Donovan SK, Portell RW (2001) Caulostrepsis spiralis isp. nov., Miocene Grand Bay Formation of Carriacou (Grenadines, Lesser Antilles). Ichnos 8:261–264
Pirrone CA, Buatois LA (2016) Bioeroded dinosaur bones: novel signatures of necrophagous activity in a Cretaceous continental environment. Ichnos 23:340–348
Pirrone CA, Buatois LA, Bromley RG (2014) Ichnotaxobases for bioerosion trace fossils in bones. J Paleontol 88:195–203
Plewes CR (1996) Ichnotaxonomic studies of Jurassic endoliths. Unpublished Dissertation, University of Wales, Aberystwyth
Plewes CR, Palmer T, Haynes J (1993) A boring foraminiferan from the Upper Jurassic of England and northern France. J Micropalaeontol 12:83–89
Pleydell SM, Jones B (1988) Boring of various faunal elements in the Oligocene-Miocene Bluff Formation of Grand Cayman, British West Indies. J Paleontol 62:348–367
Plusquellec Y (1968) Commensaux des tabulés et stromatoporoïdes du Dévonien armoricain. Annales de la Société Géologique du Nord 88:47–56
Pohowsky RA (1978) The boring ctenostomate Bryozoa: taxonomy and paleobiology based on cavities in calcareous substrata. Bull Am Paleontol 73:1–192
Pokorný R, Štofik M (2017) Evidence of bioerosive structures in Quaternary glaciomarine sediments from southwestern Iceland. Ichnos 24:204–221
Portlock JE (1843) Raport on the Geology of the County of Londonderry, and of Parts of Tyrone and Fermanagh. Milliken, Dublin
Pratje O (1922) Fossile kalkbohrende Algen (Chaetophorites gomontoides) in Liaskalken. Zentralblatt für Mineralogie, Geologie und Paläontologie [1922]:229–301
Price AW (1916) Notes on the Paleontology of Raleigh, Wyoming, McDowell and adjacent counties – Marine invertebrates from the Pottsville Series. In: Krebs CE, Teets DD, White IC (eds) Raleigh County and the Western Portions of Mercer and Summers Counties. West Virginia Geological Survey, Wheeling, pp 662–746
Quenstedt FA (1849) Petrefaktenkunde Deutschlands – Die Cephalopoden [text volume & atlas]. Ludwig Friedrich Fues, Tübingen
Quenstedt FA (1858) Der Jura. Laupp, Tübingen
Radtke G (1991) Die mikroendolithischen Spurenfossilien im Alt-Tertiär West-Europas und ihre palökologische Bedeutung. Courier Forschungsinstitut Senckenberg 138:1–185
Radtke G, Golubić S (2005) Microborings in mollusk shells, Bay of Safaga, Egypt: morphometry and ichnology. Facies 51:118–134
Radtke G, Glaub I, Vogel K, Golubić S (2010) A new dichotomous microboring, Abeliella bellafurca isp. nov., distribution, variability and biological origin. Ichnos 17:25–33
Radtke G, Campbell SE, Golubić S (2016) Conchocelichnus seilacheri igen. et isp. nov., a complex microboring trace of bangialean rhodophytes. Ichnos 23:228–236
Radwański A, Bałuk W (1997) A biogenic paradigm of shells of the clavatulid gastropods: the trace fossil Clavatulicola evaephilus ichnogen. et ichnosp. n. from the middle Miocene localities Korytnica (Holy Cross Mountains, Poland) and Grund (Vienna Basin, Austria). Acta Geol Polonica 47:199–210
Rasser MW, Vallon LH, Salvador RB (2016) Perforations of freshwater snail shells from the Miocene of Germany: Nihilichnus covichi n. isp. Ichnos 23:222–227
Rhumbler L (1909) Die Foraminiferen (Thalamophoren) der Plankton-Expedition. Lipsius & Tischer, Kiel
Rindsberg AK (2012) Ichnotaxonomy: finding patterns in a welter of information. In: Knaust D, Bromley RG (eds) Trace Fossils as Indicators of Sedimentary Environments. Developments in Sedimentology 64. Elsevier, Amsterdam, pp 45–78
Rinehart LF, Lucas SG, Spielmann JA (2006) Bite marks on tetrapod bones from the Upper Triassic Chinle Group representing a new ichnogenus. New Mexico Mus Nat Hist Sci Bull 37:160–163
Roberts EM, Rogers RR, Foreman BZ (2007) Continental insect borings in dinosaur bone: examples from the Late Cretaceous of Madagascar and Utah. J Paleontol 61:201–208
Robinson J, Lee D (2008) Brachiopod pedicle traces: recognition of three separate types of trace and redefinition of Podichnus centrifugalis Bromley & Surlyk, 1973. Fossils Strata 54:219–225
Rodríguez-Tovar FJ, Uchman A, Puga-Bernabéu A (2015) Borings in gneiss boulders in the Miocene (upper Tortonian) of the Sorbas Basin, SE Spain. Geol Mag 152:287–297
Rosso A (2008) Leptichnus tortus isp. nov., a new cheilostome etching and comments on other bryozoan-produced trace fossils. Studi Trentini di Scienze Naturali/Acta Geol 83:75–85
Roux W (1887) Über eine im Knochen lebende Gruppe von Fadenpilzen (Mycelites ossifragus). Zeitschrift für wissenschaftliche Zoologie 45:227–254
Rozhnov S (1989) New data about rhipidocystids (Eocrinoidea). In: Kaljo DL (ed) Fossil and recent echinoderm researches [In Russian]. Akademia Nauk Estonoskoi SSR, Tallinn, pp 38–57
Ruedemann R (1925) The Utica and Lorraine Formations of New York, Part 2. New York State Mus Bull 262:1–171
Ruggiero E, Raia P (2014) Oichnus taddeii, a new fossil trace produced by capulids on brachiopod shells. Spanish J Palaeontol 29:15–24
Sá AA, Gutiérrez-Marco JC (2015) Aroucaichnus igen. nov. y otros icnofósiles singulares del Ordovícico del Geoparque Arouca (Portugal). Boletín de la Sociedad Geológica del Perú 110:8–23
Santos A, Mayoral E (2006) Bioerosive structures of sclerozoan foraminifera from the lower Pliocene of southern Spain: a contribution to the palaeoecology of marine hard substrate communities. Palaeontology 49:719–732
Santos A, Mayoral E, Muñiz F (2003) New trace fossils produced by etching molluscs from the Upper Neogene of the southwestern Iberian Peninsula. Acta Geol Polonica 53:181–188
Santos A, Mayoral E, Muñiz F (2005) Bioerosion scars of acorn barnacles from the southwestern Iberian peninsula, Upper Neogene. Riv Italiana Paleontol Stratigr 111:181–189
Santos A, Mayoral E, Bromley RG (2011) Bioerosive structures from Miocene marine mobile-substrate communities in southern Spain, and description of a new sponge boring. Palaeontology 54:535–545
Santos A, Mayoral E, Baarli G, da Silva CM, Cacháo M, Johnson MD (2012) Symbiotic association of a pyrgomatid barnacle with a coral from a volcanic middle Miocene shoreline (Porto Santo, Madeira Archipelago, Portugal). Palaeontology 55:173–182
Santos A, Mayoral E, Villas E, Herrera Z, Ortega G (2014) First record of Podichnus in orthide brachiopods from the Lower Ordovician (Tremadocian) of NW Argentina and its relation to the early use of an ethological strategy. Palaeogeogr Palaeoclimatol Palaeoecol 399:67–77
Santos A, Mayoral E, Dumont CP, da Silva CM, Ávila SP, Baarli BG, Cachão M, Johnson ME, Ramalho RS (2015) Role of environmental change in rock-boring echinoid trace fossils. Palaeogeogr Palaeoclimatol Palaeoecol 432:1–14
Schindewolf OH (1962) Parasitäre Thallophyten in Ammoniten-Schalen. Paläontol Z 36:206–215
Schlaudt CM, Young K (1960) Acrothoracic barnacles from the Texas Permian and Cretaceous. J Paleontol 34:903–907
Schlirf M (2006) Linkichnus terebrans new ichnogenus et ichnospecies, an insect boring from the Late Triassic of the Germanic Basin, southern Germany. Ichnos 13:277–280
Schloz W (1972) Zur Bildungsgeschichte der Oolithenbank (Hettangium) in Baden-Württemberg. Arbeiten aus dem Institut für Geologie und Paläontologie der Universität Stuttgart 67:101–212
Schmidt H (1992) Mikrobohrspuren ausgewählter Faziesbereiche der tethyalen und germanischen Trias (Beschreibung, Vergleich und bathymetrische Interpretation). Frankfurter Geowissenschaftliche Arbeiten, A 12:1–228
Schönberg CHL, Tapanila L (2006) The bioeroding sponge Aka paratypica, a modern tracemaking analogue for the Paleozoic ichnogenus Entobia devonica. Ichnos 13:147–157
Schönberg CHL, Fang JKH, Carballo JL (2017) Bioeroding sponges and the future of coral reefs. In: Carballo J, Bell J (eds) Climate Change, Ocean Acidification and Sponges. Springer, Berlin Heidelberg, pp 179–372
Seguenza G (1879) Le formazioni Terziarie nella Provincia di Reggio (Calabria). Coi tipi del Salviucci, Rome
Seguenza G (1882) Paleontologia del Cretaceo Medio delle Provincie Meridionali d’Italia. Atti della Reale Accademia dei Lincei, Memorie della Classe di scienze fisiche, matematiche e naturali 12:101–214
Seilacher A (1968) Swimming habits of belemnites – recorded by boring barnacles. Palaeogeogr Palaeoclimatol Palaeoecol 4:279–285
Silén L (1946) On two new groups of Bryozoa living in shells of molluscs. Arkiv för Zoologi 38B:1–7
Sismonda E (1871) Matériaux pour servir à la Paléontologie du terrain tertiaire du Piémont. Memorie della Reale Accademia delle Scienze di Torino, Serie seconda 25:257–361
Smith J (1910) Carboniferous rocks of the Solway, Scotland. T Geol Soc Glasgow 14:30–59
Sokolov BS (1948) Kommensalizm i Favositid. Izvestija Imperatorskoj Akademii nauk, seria biologicheskaja 1:101–110
Solle G (1938) Die ersten Bohr-Spongien im europäischen Devon und einige andere Spuren. Senckenb Lethaea 20:154–178
Stafford ES, Dietl GP, Gingras MP, Leighton LR (2015) Caedichnus, a new ichnogenus representing predatory attack on the gastropod shell aperture. Ichnos 22:87–102
Stephenson LW (1941) The large invertebrate fossils of the Navarro Group of Texas. Publ Univ Texas 4101:1–641
Stephenson LW (1952) Larger invertebrate fossils of the Woodbine formation (Cenomanian) of Texas. U S Geol Surv, Prof Pap 242:1–226
Stiller F (2005) An Early Jurassic Talpina-dominated assemblage of borings in bivalve shells from southern Hunan, China, with remarks on the ichnogenus Talpina Hagenow, 1840. Acta Palaeontol Sinica 44:396–411
Süss H (1979) Durch Protophytobia cupressorum gen. nov., sp. nov. (Agromyzidae, Diptera) verursachte Markflecke in einem Holz von Juniperoxylon aus dem Tertiar von Süd-Limburg (Niederlande) und der Nachweis von Markflecken in einer rezenten Callitris- Art. Feddes Repertorium 90:165–172
Süss H, Müller-Stoll WR (1975) Durch Palaeophytobia platani n. g., n. sp. (Agromyzidae, Diptera) verursachte Markflecken im Holz fossiler Platanen aus dem ungarischen Miozän. Wissenschaftliche Zeitschrift der Humboldt-Universität Berlin, Mathematisch-Naturwissenschaftliche Reihe 24:515–519
Talent JA (1963) The Devonian of the Mitchell and Wentworth rivers. Geol Surv Victoria, Mem 24:1–118
Tapanila L (2005) Palaeoecology and diversity of endosymbionts in Palaeozoic marine invertebrates: trace fossil evidence. Lethaia 38:89–99
Tapanila L (2006) Devonian Entobia borings from Nevada, with a revision of Topsentopsis. J Paleontol 80:760–767
Tapanila L (2008) The medium is the message: imaging a complex microboring (Pyrodendrina cupra igen. n., isp. n.) from the early Paleozoic of Anticosti Island, Canada. In: Wisshak M, Tapanila L (eds) Current Developments in Bioerosion. Springer, Berlin Heidelberg, pp 123–145
Tapanila L, Ekdale A (2007) Early history of symbiosis in living substrates: trace-fossil evidence from the marine record. In: Miller W III (ed) Trace Fossils: Concepts, Problems, Prospects. Elsevier, Amsterdam, pp 345–355
Tapanila L, Holmer LE (2006) Endosymbiosis in Ordovician-Silurian corals and stromatoporoids: a new lingulid and its trace from eastern Canada. J Paleontol 80:750–759
Tapanila L, Roberts EM (2012) The earliest evidence of holometabolan insect pupation in conifer wood. PLoS ONE 7:e31668
Tavernier A, Golubić S (1993) Pliocene microboring assemblages in shells of the Purisima Formation (Capitola, California). Bollettino della Società Paleontologica Italiana, Special 1:405–414
Tavernier A, Campbell SE, Golubić S (1992) A complex marine shallow-water boring trace, Dendrorete balani n. ichnogen. et ichnospec. Lethaia 25:303–310
Taylor PD, Wilson MA, Bromley RG (1999) A new ichnogenus for etchings made by cheilostome bryozoans into calcareous substrates. Palaeontology 42:595–604
Taylor PD, Wilson MA, Bromley RG (2013) Finichnus, a new name for the ichnogenus Leptichnus Taylor, Wilson and Bromley 1999, preoccupied byLeptichnus Simroth, 1896 (Mollusca, Gastropoda). Palaeontology 56:456
Teichert C (1945) Parasitic worms in Permian brachiopod and pelecypod shells in Western Australia. Am J Sci 243:197–209
Terquem O (1855) Paléontologie de l’étage infèrieur de la formation liasique de la province de Luxembourg: Grand-Duché (Hollande), et de Hettange, du département de la Moselle. Mém Soc Géol France 2(série 5):219–343
Terquem O, Piette É (1865) Paléontologie du Lias Inférieur de l’Est de la France. Mém Soc Géol France, 2ème série 8:1–175
Thenius E (1979) Lebensspuren von Ephemeropteren-Larven aus dem Jung-Tertiär des Wiener Beckens. Ann Nat Mus Wien 82:177–188
Thenius E (1988) Lebensspuren von aquatischen Insektenlarven aus dem Jungtertiär Niederösterreichs. Beitr Paläontol Österr 14:1–17
Thomas AO (1911) A fossil burrowing sponge from the Iowa Devonian. Bull Lab Nat Hist State Univ Iowa 6:165–169
Thurmann J, Étallon A (1964) Lethea Bruntrutana ou études paléontologiques et stratigraphiques dur les terrains Jurassique supérieurs du Jura Bernois et en particulier des environs de Porrentruy. Nouveaux Mémoires de la Société Helvetique des Sciences Naturelles 20:355–500
Tomlinson JT (1953) A burrowing barnacle of the genus Trypetesa (order Acrothoracica). J Washington Acad Sci 43:373–381
Tomlinson JT (1963) Acrothoracican barnacles in Paleozoic myalinids. J Paleont 37:164–166
Tomlinson JT (1969) The burrowing barnacles (Cirripedia: Order Acrothoracica). U S Nat Mus Bull 296:1–162
Tribollet A, Radtke G, Golubić S (2011) Bioerosion. In: Reitner J, Thiel V (eds) Encyclopedia of Geobiology. Springer, Dordrecht, pp 117–134
Turquier Y (1967) Description d’un nouveau Trypetesa Norman (= Alcippe Hancock), cirripède acrothoracique des côtes françaises de la Manche. Cah Biol Mar 8:75–87
Turquier Y (1976) Etude de quelques cirripèdes acrothoraciques de Madagascar. II. Description de Trypetesa spinulosa n. sp. Bull Soc Zool France 101:559–574
Uchman A, Rattazzi B (2018) A new etching trace from Savignone Conglomerate (Oligocene), NW Italy, produced probably by limpet gastropods. Acta Geol Pol 68:651–662
Uchman A, Gaigalas A, Melesyte M, Kazakauska V (2007) The trace fossil Asthenopodichnium lithuanicum sp. nov. from Late Neogene brown-coal deposits. Lithuania. Geol Q 51:329–336
Uchman A, Stachacz M, Salamon K (2018a) Spirolites radwanskii igen. et isp. nov. - a vermetid gastropod etching trace from the middle Miocene rocky coast of the Paratethys. Poland. J Paleontol 92:883–895
Uchman A, Wisshak M, Rodríguez-Tovar FJ (2018b) The bivalve boringCuenulites amygdaloides nov. isp. in siliceous sponges from the Upper Cretaceous of Germany. Geobios 51:481–486
Ulrich EO (1879) Descriptions of new genera and species of fossils from the Lower Silurian about Cincinnati. J Cincinnati Soc Nat Hist 2:8–30
Ulrich EO (1890) New Lower Silurian Bryozoa. J Cincinnati Soc Nat Hist 12:173–198
Utinomi H (1962) Occurrence of a Trypetesa in Japan. Dohutsugaku Zasshi [Zoological Magazine, Tokyo] 71:399
Vaillant L (1909) Observations paléontologiques faites dans les sables Éocènes Landéniens aux environs d’Arras. Bull Soc Géol France 9:277–282
Vialov OS, Nessov LA (1974) Posmertnye povrezhdenija pancirej nekotorykh rannemelovykh cherepakh kosterazrushayushhimi organizmami [Postmortem injuries of shells of some early Cretaceous turtles by bone-damaging organisms]. Paleontologicheskij Sbornik 11:99–103
Vinn O, Wilson MA, Mõtus MA (2014) The earliest giant Osprioneides borings from the Sandbian (Late Ordovician) of Estonia. PLoS ONE 9:e99455
Vitális S (1961) Lebensspuren im Salgotarjaner Braunkohlenbecken. Annales Universitatis Scientiae Budapest Rolando Eötvös Nomintae, Sectio Geologica 1961:121–132
Vogel K, Golubić S, Brett CE (1987) Endolith associations and their relation to facies distribution in the Middle Devonian of New York State, USA. Lethaia 20:263–290
Voigt E (1959) Endosacculus moltkiae n. g. n. sp., ein vermutlicher fossiler Ascothoracide (Entomostr.) als Cystenbildner bei der Oktokoralle Moltkia minuta. Paläontol Z 33:211–223
Voigt E (1965) Über parasitische Polychaeten in Kreide-Austern sowie einige andere in Muschelschalen bohrende Würmer. Paläontol Z 39:193–211
Voigt E (1967) Über einen neuen acrothoraciden Cirripedier aus dem Essener Grünsand (Cenoman). Abh Nat Vereins Hamburg, N F 11:117–121
Voigt E (1970) Endolithische Wurm-Tunnelbauten (Lapispecus cuniculus n. g. n. sp. und Dodecaceria(?) sp.) in Brandungsgeröllen der oberen Kreide im nördlichen Harzvorlande. Geol Rundsch 60:355–380
Voigt E (1971) Fremdskulpturen an Steinkernen von Polychaeten-Bohrgängen aus der Maastrichter Tuffkreide. Paläontol Z 45:144–153
Voigt E (1972) Über Talpina ramosa v. Hagenow 1840, ein wahrscheinlich zu den Phoronidea gehöriger Bohrorganismus aus der Oberen Kreide, nebst Bemerkungen zu den übrigen bisher beschriebenen kretazischen “Talpina”-Arten. Nachrichten der Akademie der Wissenschaften II (Mathematisch-physikalische Klasse) [1972]:93–126
Voigt E (1973) lmmergentia cruciata (Mägdefrau) (Bryozoa, Ctenostomata) im Santon-Erz von Lengede-Broistedt bei Peine (BRD). Z Geol Wiss 1:991–1007
Voigt E (1975) Tunnelbaue rezenter und fossiler Phoronoidea. Paläontol Z 49:135–167
Voigt E (1977) On grazing traces produced by the radula of fossil and recent gastropods and chitons. Geol J, Spec Issue 9:335–346
Voigt E, Soule JD (1973) Cretaceous burrowing bryozoans. J Paleontol 47:21–33
von Hagenow F (1840) Monographie der Rügen’schen Kreide-Versteinerungen, II. Abtheilung: Radiarien und Annulaten. Nebst Nachträgen zur ersten Abtheilung. Neues Jahrbuch für Mineralogie, Geognosie, Geologie und Petrefakten-Kunde [1849]:630–672
Vosmaer GCJ (1887) Dr. H. G. Bronn’s Klassen und Ordnungen der Spongien (Porifera) wissenschaftlich dargestellt in Wort und Bild. C. F. Winter, Leibzig
Walker MV (1938) Evidence of Triassic insects in the Petrified Forest National Monument, Arizona. Proc U S Nat Mus 85:137–141
Wanner J (1938) Balanocrinus sundaicus n. sp. und sein Epöke aus dem Altmiocän der Insel Madura. N Jb Mineral Geol Paläontol, B 79:385–402
Wetherell NT (1852) Note on a new species of Clionites. Ann Mag Nat Hist 10:354–355
Wilson MA, Palmer TJ (1988) Nomenclature of a bivalve boring from the Upper Ordovician of the midwestern United States. J Paleontol 62:306–308
Wilson MA, Palmer TJ (1998) The earliest Gastrochaenolites (Early Pennsylvanian, Arkansas, USA): an upper Paleozoic bivalve boring? J Paleontol 72:769–772
Wisshak M (2006) High-latitude Bioerosion: The Kosterfjord Experiment. Lecture Notes in Earth Sciences 109. Springer, Berlin Heidelberg
Wisshak M (2008) Two new dwarf Entobia ichnospecies in a diverse aphotic ichnocoenosis (Pleistocene/Rhodes, Greece). In: Wisshak M, Tapanila L (eds) Current Developments in Bioerosion. Springer, Berlin Heidelberg, pp 213–233
Wisshak M (2017) Taming an ichnotaxonomical Pandora’s box: revision of dendritic and rosetted microborings (ichnofamily: Dendrinidae). Eur J Taxonomy 390:1–99
Wisshak M, Neumann C (2018) Large dendrinids meet giant clam: the bioerosion trace fossil Neodendrina carnelia igen. et isp. n. in a Tridacna shell from Pleistocene-Holocene coral reef deposits, Red Sea. Egypt. Fossil Record 21:1–9
Wisshak M, Porter D (2006) The new ichnogenus Flagrichnus—a palaeoenvironmental indicator for cold-water settings? Ichnos 13:135–145
Wisshak M, Tapanila L (2008) Current Developments in Bioerosion. Springer, Berlin Heidelberg
Wisshak M, Freiwald A, Lundälv T, Gektidis M (2005) The physical niche of bathyal Lophelia pertusa in a non-bathyal setting: environmental controls and palaeoecological implications. In: Freiwald A, Roberts JM (eds) Cold-Water Corals and Ecosystems. Springer, Berlin Heidelberg, pp 979–1001
Wisshak M, Seuß B, Nützel A (2008) Evolutionary implications of an exceptionally preserved Carboniferous microboring assemblage in the Buckhorn Asphalt lagerstätte (Oklahoma, USA). In: Wisshak M, Tapanila L (eds) Current Developments in Bioerosion. Springer, Berlin Heidelberg, pp 21–54
Wisshak M, Alexandrakis E, Hoppenrath M (2014) The diatom attachment scar Ophthalmichnus lyolithon igen. et isp. n. Ichnos 21:111–118
Wisshak M, Kroh A, Bertling M, Knaust D, Nielsen JK, Jagt JWM, Neumann C, Nielsen KSS (2015) In defence of an iconic ichnogenus – Oichnus Bromley, 1981. Ann Soc Geol Pol 85:445–451
Wisshak M, Titschack J, Kahl W-A, Girod P (2017) Classical and new bioerosion trace fossils in Cretaceous belemnite guards characterised via micro-CT. Fossil Record 20:173–199
Wisshak M, Meyer N, Radtke G, Golubić S (2018) Saccomorpha guttulata: a new marine fungal microbioerosion trace fossil from cool- to cold-water settings. Paläontol Z 92:525–533
Xing L, Parkinson A, Ran H, Pirrone C, Roberts E, Zhang J, Burns M, Wang T, Choiniere J (2015) The earliest fossil evidence of bone boring by terrestrial invertebrates, examples from China and South Africa. Hist Biol 28:1108–1117
Zamora S, Mayoral E, Esteve J, Gámez-Vintaned JA, Santos A (2011) Exoskeletal abnormalities in paradoxidid trilobites from the Cambrian of Spain, and a new type of bite trace. Bull Geosci 86:665–673
Zonneveld J-P, Bartels WS, Gunnell GF, McHugh LP (2015) Borings in early Eocene turtle shell from the Wasatch Formation, South Pass, Wyoming. J Paleontol 89:802–820
Acknowledgements
We gratefully acknowledge all colleagues from the bioerosion research community who brought overlooked ichnotaxa to our attention and discussed the validity of reputed ichnotaxa with us. These are Jorge Genise (Buenos Aires, Argentina), Kantimati Kulkarni (Agharkar, India), Eduardo Mayoral (Huelva, Spain), Matthew Riley (Cambridge, UK), Andrew Rindsberg (Livingston, USA), Ana Santos (Huelva, Spain), Ursula Toom (Tallinn, Estonia), Alfred Uchman (Krakow, Poland), Mark Wilson (Wooster, USA), and Li-Jun Zhang (Henan, China). We are particularly indebted to Christine Schönberg (Perth, Australia), who, during her ongoing revision of the bioeroding sponge genus Cliona, brought many actual bioerosion ichnospecies to light and to our notice. We thank Sebastian Teichert (Erlangen, Germany) for his valuable advice regarding the validity of Latin, latinized, or Greek spelling of various ichnotaxa and their combinations. Andrew Rindsberg (Livingston, USA) and Radek Mikuláš (Prague, Czech Republic) provided valuable reviews of this manuscript. Last but not least, we are indebted to our dear friend and mentor Richard G. Bromley (1939-2018) for his immense contribution to bioerosion research and ichnotaxonomy.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of a Topical Collection in Facies on Bioerosion: An interdisciplinary approach, guest edited by Ricci, Uchman, and Wisshak.
Rights and permissions
About this article
Cite this article
Wisshak, M., Knaust, D. & Bertling, M. Bioerosion ichnotaxa: review and annotated list. Facies 65, 24 (2019). https://doi.org/10.1007/s10347-019-0561-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10347-019-0561-8