Abstract
A mid-latitude coralgal bioconstruction (Tyrrhenian coast of Calabria, southern Italy) and associated sedimentary facies record climate and sea-level changes of this coastal area during the Pleistocene. The coralgal bioconstruction, characterized by Cladocora caespitosa bafflestone and red algae bindstone, associated with Spondylus gaederopus sp.-rich boundstone, formed as a result of submergence during Marine Isotope Stage 9 in a moderate shallow-marine environment at a depth of 6–20 m. The coast was affected by moderate to high-energy currents and occasional storms; the annual temperature was 12–18 °C. The lower part of the coralgal body is dominated by Cladocora and the upper part by Lithophylloid algae. The change was likely the result of the occasional influx of coarse clastic material partially buried the C. caespitosa colonies, but provided a suitable substrate for the pioneer encrusting organisms. During a subsequent glacial stage, sea-level fall combined with tectonic uplift induced the partial emergence of the coralgal body, and deposition of an overlying subaerial deposited breccia. A later resubmergence of the area, likely during the last interglacial period (MIS 5), is recorded as testified by the presence of endolithic microbialites in cavities within the coralgal bioconstruction and the breccia deposit. Microbialite formation took place in a marine environment with cooler water, with respect to the previous submergence phase, possibly during a time of early deglaciation. The low temperature, possibly combined with very shallow depths and an excess of nutrients, could have played a role in preventing further growth of C. caespitosa while favoring the precipitation of microbial carbonate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Autochthonous Quaternary bio-mediated carbonate deposits, in their global occurrence from cold to temperate and tropical latitudes, are sensitive recorders of past sea-level and environmental changes. Their accurate dating and geochemical characterization can be of prime importance to the understanding of the mechanisms driving glacial–interglacial cycles during Quaternary times (Camoin and Webster 2015). In particular, as a result of their growth within a narrow depth range, shallow-water coral bioconstructions can represent excellent sea-level indicators. High-resolution records of past global change are stored in the geochemical and physical parameters of the skeletons of bioconstructors (corals, algae, etc.), and these can be used to study ocean/atmosphere variability. In addition, changes in other environmental parameters such as light conditions, water energy, and nutrient levels, do affect these communities, which are sensitive to ecological changes affecting their environment. Moreover, of the biological indicators of environmental change, carbonate microbialites can be useful since they occur in shallow- to deep-water marine environments, in association with metazoan bioconstructors and as primary frame-builders; they may also occur in non-marine settings too, under any climatic conditions (Sprachta et al. 2001; Riding 2011; Perri et al. 2012a). However, it is only relatively recent that their use as paleoenvironmental indicators during the Quaternary has been recognized, and this has been limited to their occurrence within tropical coral-reef sequences and in periglacial continental environments (Camoin et al. 2006; Lacelle 2007).
This paper deals with Pleistocene shallow-water carbonate deposits, consisting of a coralgal bioconstruction, associated with bivalve-rich crusts within a submarine cave, and marine endolithic microbialites, cropping out at Punta Diamante, along the Tyrrhenian coast of Calabria, southern Italy. The deposit has formed since the Middle Pleistocene, during a period of time characterized by active tectonic uplift. The primary purpose of this study was to undertake a sedimentological and geochemical analysis of the carbonate deposits in order to obtain a detailed paleoecological interpretation of each depositional event, which, in turn, contributes to a better understanding of the paleoclimatic evolution of the area during the Quaternary.
Geological and morphological setting
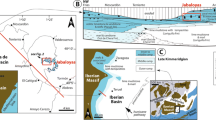
The study area, developing from present mean sea level up to 15 m above, is located close to the outlet of the Corvino River (Fig. 1a). It represents a small sea-stack of Jurassic/Lower Cretaceous meta-limestone, belonging to the ophiolitic Diamante-Terranova Unit (Amodio-Morelli et al. 1976; Cello et al. 1996), overlain by Quaternary deposits, the subject of this paper.
a The study area of Punta Diamante located close to the outlet of the Corvino River, on the hydrographic left. b Simplified sketch of the area showing the Quaternary sedimentary cover, lying on the rock basement, composed of: a coralgal bioconstruction, cave and the notch, and endolithic microbialites. Breccia deposits represent a single sedimentary event. Not to scale. c Aerial photo of Punta Diamante from the top of the northern cliff
In the scheme of Carobene et al. (1986), the examined area is considered a IV-order Pleistocene marine terrace, i.e., the lowest terrace observable in the neighborhood of Diamante, morphologically characterized by paleo-erosional forms, that is an abrasion platform, sub-vertical cliffs, and a cave (Fig. 1b, c). The abrasion platform, which gently dips (1–2°) toward the ESE, is elongated in an E–W direction for ~70 m, from the shoreline landward, and narrows from ~20 to ~3 m to the east. The sub-vertical cliffs are characterized by a quite deep wave-cut notch up to 2 m high, which gives an arcuate shape to the lower part of the cliff. Moreover, a Quaternary sedimentary cover can be recognized along the terrace, composed of: a coralgal bioconstruction (the main sedimentary body), cave bio-encrustations, breccia deposits, and endolithic microbialites (Fig. 1b, c).
The coralgal bioconstruction is represented by a residual deposit, 3–5 m thick and 15–20 m wide, of an original body that would have been larger and broadly domal/tabular in shape. The present body is also cut by modern erosional cliffs and an abrasion platform that allow observations and measurements of stratigraphic sections for the reconstruction of the Quaternary sedimentological history.
The bioconstruction is typified by an oligo-specific community of frame-builders formed by colonies of Cladocora caespitosa (L.) and encrusting Lithophylloideae red algae (Fig. 2). In particular, C. caespitosa autochthonous colonies are concentrated within the body mostly inward and at the bottom of the bioconstruction, forming a ~3-m-thick deposit passing laterally and seaward to scattered colonies (Fig. 2b). Encrusting red algae dominate the upper and lateral parts of the bioconstruction, and occur as bioclasts and encrustations in the coral-dominated part, which is limited by a syn-sedimentary erosional surface (Fig. 2a, c).
a Part of the coralgal bioconstruction lying on meta-limestone basement. This deposit is formed by b Cladocora caespitosa autochthonous colonies up to ~3 m high, and c encrusting red algae, mainly Titanoderma sp. The latter dominates the upper and lateral parts of bioconstruction; nevertheless, it occurs in the coral-dominated part, which is limited by an erosional surface
The cave, positioned laterally and inland with respect to the bioconstruction, is ~10 m2 wide and 3–4 m high (Fig. 3a). It has been cut into the meta-limestones, which are well exposed on the roof; the walls of the cave are covered by red algae dominated encrustation, locally rich in S. gaederopus bivalves and microbialites (Fig. 3b).
a View of the study site taken from abrasion platform. From the left, the cave underlying meta-limestone basement, the younger breccia deposit stratigraphically overlying the coralgal bioconstruction with an erosional surface. b Cave wall covered by red algae-dominated encrustation, locally rich in Spondylus gaederopus valves. c Breccia deposit formed mainly by meta-limestone varying from sub-angular to sub-rounded. d Laminated to thrombolytic micrite, containing marine fossils remains, fills a large and irregular cavity in the coralgal bioconstruction
A younger breccia deposit stratigraphically overlies the bioconstruction with an erosional contact (Fig. 3a). This mostly chaotic deposit appears truncated towards the open sea, eroded by modern wave action and continental water. A vague stratification is observable, associated with some clast imbrication, and a 10–20° westward (=seaward) dip, possible due to subaerial debris flows. Breccia also locally fills open paleo-fractures within both the bedrock and the bioconstruction. Fracturing is possibly linked to fault-related seismic activity, due to the Quaternary tectonic uplift of this Calabrian sector, still active today (Antonioli et al. 2009; Ferranti et al. 2006, 2009). Clasts forming the breccia, derived from the meta-limestone bedrock, vary from sub-angular to sub-rounded in shape and are poorly sorted, ranging in dimension from fine sand to blocks (Fig. 3c). The breccia deposit is generally clast-supported with little matrix, appearing highly porous, except for the lowest part of the deposit (where visible) where a laminated to thrombolytic micrite with marine fossils fills most of the pores and larger cavities (Fig. 3d).
Materials and methods
The study has been based on field observations mainly consisting of geological mapping coupled with measuring and sampling of stratigraphic sections. Microfacies analyses have been realized on polished samples and traditional thin-sections of the main lithological and/or biological assemblages recognized in the field. An open nomenclature has been used for fossil taxa because the focus is paleoecology.
A morphometric analysis of the Cladocora colonies was carried out according to their prevalent location: in transverse section, the individual corallite width was measured and the density of the bioconstruction was evaluated by counting the number of corallites per quadrat of 25 cm2. The ratio between the total cover of the corallites (number of corallites by their mean surfaces) and the quadrat size provides the percentage cover occupied by the corallites in a colony (corallite density). In longitudinal section, the general shape of the colonies has been observed and their heights measured. Colony morphology was described on more isolated colonies using the Is-index (Riegl 1995), as a degree of sphericity: Is-index = maximum height of colony/maximum diameter of colony.
Scanning electron microscope (SEM) studies of the micro/nanofacies were undertaken on polished thin-sections and freshly broken surfaces, using an FEI-Philips ESEM-FEG Quanta 200 F, operating in a range of 5–20 kV with a working distance between 6 and 15 mm. Samples were carbon- or gold-coated, respectively, depending on whether they were prepared for microanalysis or textural study with the SEM. Semi-quantitative element analyses of sub-micron-sized spots during SEM observations were obtained using an EDAX energy-dispersive X-ray spectrometer (EDS), operating at 20 kV with a working distance of 12 mm.
The bulk mineral composition of samples representative of breccia matrix, encrusting red algae, and C. caespitosa, was determined by XRD analyses using a Philips PW1730 diffractometer.
Carbon and oxygen isotope analyses were carried out on selected samples of Cladocora, red algae, and microbialites dissolving 8–10 mg of sample at 25 °C in 100 % orthophosphoric acid for 12 h.
Results
Four bio-sedimentary facies have been identified in the sedimentary bodies of Punta Diamante:
-
1.
C. caespitosa bafflestones;
-
2.
red algae bindstones;
-
3.
S. gaederopus-rich boundstones;
-
4.
endolithic microbialites;
The first two together form the coralgal bioconstruction unit.
C. caespitosa bafflestone
The colonies of C. caespitosa occur between 1 and 5.6 m above sea level. The most continuous coral body directly overlies Jurassic bedrock and is composed of contiguous colonies up to 30 cm high and about 50 cm in diameter (Is-index about 0.6), in part coalescent, forming a bank ~1.5 m in thickness (Fig. 2a, b). Colonies are sub-hemispherical and show a phaceloid morphology with a corallite density varying between 50 and 80 % and mean number of corallites per colony up to 81 (in 25 cm2); individual corallite diameter ranges between 5 and 8 mm with 6.5 as the mean value (Fig. 4a–c).
a Autochthonous, well-preserved, colonies of C. caespitosa. b, c Close-up views show the space among individual corallites completely filled by terrigenous detritus, mixed with patches of dense carbonate micrite and skeletal organisms. d, e Transverse section of a corallite shows scalloped-shape external ornamentations, few generations of septa, septa pinnacles, and tabulae. f Aragonitic needle-crystals spherulites, typical primary structures of C. caespitosa corallites, are well preserved in the lower part of the bank
Despite the general recrystallization due to diagenesis (see below), the corallites show scalloped-shape external ornamentations, few generations of septa, septa pinnacles, and tabulae (Fig. 4d, e). However, rarely, in the lower part of the bank are preserved genuine aragonitic needle-crystal spherulites, the typical primary skeletal structure of C. caespitosa corallites (Fig. 4f).
Sediment between the corallites varies from floatstone to, more rarely, rudstone, with several other organisms present (Fig. 5a). Serpulids up to 300 µm in diameter occur as individuals or more rarely in clusters (Fig. 5b) within the sediment, as well as encrusting lithophylloid red algae and corallites (Fig. 5c). Also present are rissoid gastropods up to 5 mm in height (Fig. 5d), benthic and encrusting foraminifera (Acervulina inhaerens Schultze, 1854) up to 1 mm in length (Fig. 5e), bryozoans (Fig. 5g), bivalve shell fragments (Fig. 5h), and echinoid debris (Fig. 5i). A variable amount of terrigenous detritus, mixed with patches of dense carbonate micrite and bioclasts, can be present, commonly forming a graded texture within cavities. Clasts vary from fine sand to medium pebble (<1 cm). Grains are poorly sorted, and range from angular to rarely sub-round shaped (Fig. 5a).
a Thin-section view of C. caespitosa bafflestone shows phaceloid corallites with sub-circular section. These occur even as bifurcated (e.g., upper right of the photo). Sediment among them is characterized by terrigenous clasts up to 1 mm in length and numerous skeletal remains. b Serpulids partially filled by sediment, which form geopetal structures. c Several layers of red algae encrust corallite. d Recrystallized gastropod in longitudinal section. e Benthic foraminifera. f Encrusting foraminifera associated to red algae. g Bryozoan. h Recrystallized bivalve shell remain. i Echinoid test
Red algae bindstone
Lithophylloidae crustose coralline red algae occur as a secondary frame-builder associated with C. caespitosa bafflestone. Titanoderma sp. (Nägeli, 1858) is the most common species, with subordinate Lithophyllum sp. (Philippi, 1837). However, laterally seaward and mainly upward (following a major erosional surface) with respect to the coral domain, the red algae create flat to domal centimeter-thick bindstone units, and become the dominant frame-builder for the rest of the bioconstruction (Fig. 2a, c), with the red algal bodies showing a variable thickness (1 m in average). Overall, red algae represent at least 80 % by volume of the entire coralgal deposit.
At the micro-scale, thick multi-layered algal thalli are irregularly arranged, forming roughly lenticular, porous crusts, with micro-scale erosion surfaces between (Fig. 6a, b).
a Thin-section photo shows multi-layered, irregularly arranged red algae belonging to Titanoderma sp. in which voids are filled by sediment. b Close-up view of algal perithallus in which conceptacles occur. c Algae encrusting flat pebbles. d Detrital pocket enclosed among algal layers. Detritus is composed mainly by sub-angular to sub-rounded terrigenous clasts in micritic matrix. e Rare peloidal micrite occurring in a pocket. f Mould of a vermetid gastropod belonging to Petaloconchus sp.
Algae mainly encrust larger clasts (generally flat pebbles), probably because of their greater stability (Fig. 6c). Lenses of detrital material are common within the algal layers, some showing grading, composed of terrigenous grains, angular to rarely sub-rounded, up to 1–2 mm in diameter, and rarer bioclasts, enclosed within a brownish-to yellowish-colored micrite (Fig. 6d). The last, also locally prevalent in floatstones, appears aphanitic and rarely peloidal (Fig. 6e).
Remains of well-preserved fossils are common: benthic foraminifera (Acervulina inhaerens), along with fragments of echinoids and bivalves, and moulds of vermetid gastropods belonging to Petaloconchus sp. (Fig. 6f).
Spondylus gaederopus-rich boundstone
This bio-sedimentary deposit represents the main component of the bioencrustation within the cave, which is partially roofed by the meta-limestone basement. Numerous right valves of the bivalve S. gaederopus (L.) are cemented on the calcareous crust coating the paleocave walls (Fig. 3b); detrital left valves, recognizable by their reddish external color (Dardano 1992), are also present within the deposit (Fig. 7a). Both valves are ornate, having a radial sculpture with 8–9 stronger ribs, which are armed with hollow, irregularly shaped and often very prominent spines (Lamprell 1986; Poppe and Goto 1993). The interstitial areas between two ribs are densely filled with small prickles (Lamprell 1998). The ovoid valves are inequivalve, with the left flat and the right convex, as well as thicker (Delamotte and Vardala-Theodorou 2001). The internal margin of both valves is crenulated (Lamprell 2001). The external surface of the left valve is reddish to violet, whereas the right valve, likewise both the internal valves surface, is white (Dardano 1992).
The S. gaederopus bivalves are included in a lithified boundstone composed of red algae and microbialites. Encrusting red algae form rhodoliths up to ≈5 cm in diameter; irregular laminae are arranged in more or less concentric layers around a core generally made up of bioclastic sediment (Fig. 7b). Cellular structures in the red algae are hard to recognize through neomorphism, and only a few conceptacles occur; these are either partially or totally filled by detritus or secondary cements. Associated microbialitic boundstone is composed of peloidal to aphanitic micrite that fills remaining cavities (Fig. 7d). Coarse-to-fine terrigenous sediment, composed by grains mostly of 1 mm in diameter and rarely up to 5 cm, is patchily included in the boundstone. Several other organisms, such as serpulids, foraminifera, gastropods, bryozoans, remains of echinoids and bivalves, contribute to the formation of the biofacies.
Endolithic microbialite
Several cavities within the bioconstruction, as well as most of the pores of the lowest parts of the breccia deposit nearest the sea, are partially to totally filled with micritic carbonate deposits, reddish to yellow in color (Figs. 3c, 8a). Lamination is generally even to gently undulating, forming small-scale domes facing into the open space of the partially filled cavities, with a fabric varying from laminated to massive, partly thrombolytic (Fig. 8b, c). The deposit can line isopachously some cavities or occupy their lower part like a geopetal structure. However, most of the cavities are completely filled by the micrite.
a Field picture shows endolithic microbialite filling partially voids present in the lower part of the breccia deposit. b Small domes, up to 5 cm in diameter, formed by micrite, occur in open spaces within breccia deposit. c Endolithic microbialite with laminate fabric. d Thin-section view shows an intricate micro-fabric in which and e SEM micrographs of a sponge spicule remain and an internal model of a gastropod testify the presence of several skeletal organisms within endolithic microbialite
At the micro-scale, the micrite mainly has a peloidal fabric, even if aphanitic, and micro-thrombolytic fabrics are present. Peloids and grumuleax are composed of darker micrite surrounded by microsparite (Fig. 8d). Terrigenous clasts are also bound into the micrite, as well as small shells (or fragments) of molluscs and sponge spicules, confirming the marine origin of the deposit (Fig. 8e).
Micrite crystals observed with the SEM are composed of aggregates of subhedral to euhedral crystals 0.5–2 μm in dimension. These crystals show a particular nano-structure resulting from the coalescence of nano-spheres of carbonate, 80–100 nm in diameter; this is a feature typical of microbial carbonates (see below) (Fig. 9a, b).
Diagenesis and composition
Relatively weak diagenetic alteration has affected the carbonates forming the observed bio-sedimentary facies. Corals, originally composed of aragonite, have generally been replaced by mosaics of equant low-Mg calcite (Mg ~ 3.5 mol%). The fabric is rarely drusy, with dog tooth-like calcite crystals ranging from 50 to 100 μm in thickness along the inner edge of some cavities (Fig. 10a). In rare cases, original acicular aragonite crystals are preserved, forming the typical spherulites of the coral skeleton; their composition includes Mg ~ 1.1 % and Sr ~ 1.3 % (mole) with a few moles of Na and S (Fig. 4f).
The skeletons of red algae are generally relatively well preserved, showing the original microstructure; the calcite composition reaches Mg ~ 7 (mole%).
Micrite is commonly well preserved and composed of low-Mg calcite (Mg ~ 2 mol%), including that composing the microbialites where, obviously, it is the main component, except for local patches of coarse neomorphic calcite (Fig. 10b). Finally, later dissolution has created bio-molds and karstic micro-conduits, as well as some smoothing of dog-tooth spar cements (Fig. 10c).
The whole pattern of the diagenetic products suggests a main phase of phreatic meteoric water, followed by emergence, during which secondary porosity was created through dissolution.
Carbon and oxygen isotope analyses from samples of red algae, microbialites, partially aragonitic corallites and late calcite cements are plotted in Fig. 11. The oxygen values vary from low to moderately negative (−1.4 and −4.0 ‰). The coral carbonate shows the least negative value, whereas other facies are similar, with the most negative values belonging to the microbialites.
The carbon values, all negative, show a more significant variation, ranging from −3.0 and −10.3 ‰. The general trend of carbon and oxygen values is covariant, with again the corals showing the least negative values and the microbialites and red algae the more negative ones.
Discussion and interpretation
The coralgal bioconstruction and the cave crust
Cladocora caespitosa is the main native zooxanthellate and constructional coral of the Mediterranean Sea and represents a good climatic indicator; it is generally regarded as a warm temperature fauna. Moreover, studies of the ecology of this species suggest that C. caespitosa could be a useful indicator of climate change and consequently a proxy for past climate fluctuations (Peirano et al. 2004; Silenzi et al. 2005; Montagna et al. 2007). Large banks or ‘reefs’ of C. caespitosa are recorded from the Mediterranean area since at least the Late Pliocene (Aguirre and Jiménez 1998) but it is during the Pleistocene that the species reaches the widest distribution, in relation to the warmer climatic phases of interglacial times (Peirano et al. 2004). Its present geographical distribution has decreased with respect to fossil occurrences, likely due to climate change and human impact (Laborel 1987; Peirano et al. 1997). Nevertheless, it is reported throughout the Mediterranean Sea (Zibrowius 1980; Laborel 1987; Koukouras and Kühlmann 1991; Kühlmann et al. 1991) and is described, in particular, from all the seas surrounding the Italian coast, i.e., Tyrrhenian sea (Morri et al. 1994; Silenzi et al. 2005), Adriatic sea (Schiller 1993; Montagna et al. 2007; Kružić 2005; Kružić and Benković 2008; Kružić and Požar-Domac 2003), and Ionian Sea (Lumare 1996).
In general, colonies are characterized by distinct corallites, each having its own wall, independent of the others, and growing in a continuous rectilinear way, with growth rates ranging from 1.30 mm/year to 6.2 mm/year (Peirano et al. 1999; Kružic and Požar-Domac 2003). Since this species has symbiotic algae (zooxanthellae), it is restricted to the euphotic zone, ranging in depth from a few meters to around 50 m (Casado-Amezua et al. 2011; Bernasconi et al. 1997). With respect to other environmental parameters, however, it seems to be quite tolerant, colonizing substrates from hard to soft, living in low to high turbulent waters (Zibrowius 1980; Schiller 1993), and being well adapted to turbid coastal environments with high sedimentation rates (Külhmann 1996).
Kružić and Benković (2008), studying three distinct recent C. caespitosa banks in the Croatian Adriatic Sea, showed that the lowest values of the Is-index (colony morphology) are primarily caused by strong sea-bottom currents. In particular, sea currents in the range of 0.7–1.0 m/s are correlated with an Is-Index of 0.55–0.65, as well as with a corallite number value <80, and a calix diameter >5.0 mm (up to >6.0). Such biometric values also show a positive correlation with the highest values of light intensity and turbidity (as a sum of dispersed organic matter and sediment) measured in the Mljet bank (between 6 and 18 m of depth), of the Croatian sites. Biometric indicators obtained from the Punta Diamante bank closely approach those of Mljet bank, suggesting a likely similar environment.
The association of the C. caespitosa with encrusting red algae as the main frame-builder, with secondary inhabitants consisting of mainly encrusting forams, benthic forams, serpulids, and bryozoans, implies a moderate- to high-energy euphotic environment. Interstitial detrital sediment, mainly graded sand with some gravel and fine matrix, confirms a possible frequent effect of storms (transition zone) instead of a more wave-dominate (shoreface) domain.
The red algae dominated facies shows an increase in grain-size and prevalence of encrusting organisms like foraminifers (Acervulina inhaerens) and vermetids, with respect to the Cladocora facies. Coralline assemblages composed of Lithophylloids mostly occur in shallow-water environments of temperate seas. Coralline assemblages dominated by Lithophyllum sp. cover the rocky surfaces and make up the algal nodules in the upper 10–12 m at many Mediterranean localities (Hamel and Lemoine 1953; Cormaci et al. 1985; Adey 1986; Di Geronimo et al. 1993; Basso 1998). Lithophylloids are the main algal components in bryomol or rhodalgal lithofacies characteristic of shallow temperate carbonates in the Neogene deposits of the western Mediterranean (Braga and Aguirre 2001). Chamberlain and Irvine (1994) reported L. pustulatum (Lamouroux) Foslie (as Titanoderma pustulatum (J.V. Lamouroux) Nägeli as predominantly littoral in the British Isles, and it has been recorded by Woelkerling (1996) mainly to depths of 5 m. However, Lithophyllum species and species groups are common to depths of about 20 m (Bosence 1991), but they can also be found on the deeper seafloor (Canals and Ballesteros 1997). Camoin et al. (2006) interpreted a Lithophylloid assemblage with Acervulina inhaerens as indicative of moderate to high energy and depths in the range of 10–20 m, although in a tropical system.
The cave crust with the bivalves can be considered a lateral variation of the algal-rich facies. In fact, a substantial similarity exists between the two facies, even if the more protected environment of the cave (avoided by corals) with its decreased light intensity and water energy, led to the deposition of finer grained material and growth of serpulid worms and microbialite, which are typical of restricted cryptic and shaded environments in protected areas (Camoin et al. 1999; Jones and Hunter 1995; Martindale 1992). The facies is also rich of S. gaederopus that lives in the infralittoral zone, mainly in shallow waters up to 50 m deep (Maier and Titschack 2010).
The endolithic microbialites
Microbialites can be of key interest in the reconstructions of depositional environments and biological successions, and some have provided an insight into Quaternary environmental changes because of their widespread occurrence in Late Pleistocene to Holocene reef frameworks (Montaggioni and Camoin 1993; Reitner 1993). However, very little is known of the role and presence of microbialites within mid-latitude coral and coralgal bioherms.
Microbialites occur in all of the studied facies, but particularly in the cave crust. Those clearly associated with the coralgal bioconstruction are represented by small-scale aphanitic to peloidal pore fillings, which formed syn-sedimentary or very early post-deposition.
Of particular interest are the microbialites that “colonize” the coralgal bioconstruction and the breccia deposits. As deposition of the continental breccia postdates the bioconstruction, microbialite formation represents the last depositional event that took place after the emergence of the bioconstruction and breccia deposition, during a possible subsequent submersion. Moreover, the microbial deposit preferentially develops along a fracture line (a possible fault) that affects the bioconstruction.
The internal structure of microbialites ranges from aphanitic, clotted aggregates to dense forms with laterally roughly planar to wavy lamination (Fig. 8). Small grains trapped in the microbial fabrics include terrigenous material, marine bioclasts, and sponge spicules. The occurrence of such grains suggests that the surfaces of the crusts were at least periodically sticky, perhaps mucilaginous. The relative scarcity of extraneous particles in crusts suggests that sediment trapping was much less important than in-place bio-mediated precipitation of micritic carbonate, most probably associated with living or decaying microorganisms. Microbialites, in fact, can form as the result of the availability of organic matter residue within interstitial cavities that provide a substrate for bacterial activity which, altering porewater alkalinity in the microenvironment, induces carbonate precipitation (Dupraz et al. 2009). It is well known that such a process produces a particular ultrastructure in modern and ancient carbonate microbialites, consisting of coalescing sub-spherical nano-crystals (e.g., Camoin et al. 1999, 2006; Perri and Spadafora 2011; Perri et al. 2012a, b). A similar ultrastructure is well preserved in the observed microbialites (Fig. 9); moreover, the negative values (−7.74 and −9.30) of the δ13C indicate that carbon dioxide utilized in calcite precipitation was organic in origin (Hoefs 2009, 2011).
Environmental significance of bio-sedimentary and diagenetic events
The microbialites are characteristically involved in a biological succession and sedimentary sequence that includes the coralgal bioconstruction formation, a phase of emergence with the development of continental breccia, and the formation of the endolithic microbialites.
Similar Pleistocene deposits have been reported along the Tyrrhenian coast of Basilicata and Calabria (Carobene and Dai Pra 1990, 1991). Their occurrence has been used to identify ancient Quaternary shorelines and to reconstruct the tectonic and climatic evolution of these coastal margins during the Middle Pleistocene, in a timespan encompassing Marine Isotope Stages 9 and 7 (Carobene and Dai Pra 1990).
Previous authors have referred to the Punta Diamante deposit as a biocalcarenite with C. caespitosa (Copat Marconi et al. 1981; Carobene et al. 1986; Carobene and Dai Pra 1990), or to a Cladocora-bearing biocalcarenite (Carobene and Dai Pra 1991) associated with limestone clasts, quartz, mica grains and rock/fossil fragments (especially Lithothamnium sp.) (Carobene and Ferrini 1993). The 230Th/234U radiometric analysis performed on C. caespitosa fragments has provided an age of ≥306 ka BP, suggesting the formation of the colonies during the MIS 9 (Carobene et al. 1986), when the sea level was about 20 m higher relative to its present position. During successive environmental changes, possibly induced by tectonic movements and/or climatic fluctuations, a sandy gravel deposit formed, occluding and partially burying the C. caespitosa colonies. These new mobile substrates represented a possibility for encrusting red algae to overlie corals, which competed for survival in the same environment for a brief period, during which algae and corals alternated in a chaotic sequence.
During the Middle-Late Pleistocene, intense climatic variations, as glacial stages, led to the formation of cryoclasts. Moraine, cirques, and periglacial deposits present in the hinterland mountains confirm the presence of glacial ice (Boenzi and Palmentola 1975; Hughes et al. 2006). Frost weathering associated with active faulting, fractured the meta-limestone basement to form a thick clastic breccia, which covered the coralgal bioconstruction. Uplift of the area and contemporaneous regression of the sea (due to glaciation) caused the (most probably partial) emergence of the coralgal bioherm.
Subsequently, renewed submergence affected the area, which can be related to a new interglacial stage, possibly MIS 5e. It is of note that Carobene and Dai Pra (1991) recognized several marine and continental units post-Cladocora-bearing biocalcarenite, as well as abrasion platforms, wave-cut notches, caves, and Lithodomus holes. The authors dated these elements to the main peak of the last interglacial period (substage 5e–Eutyrrhenian).
The alternation of emergence and submergence affecting the whole sedimentary body is confirmed by the diagenetic story: drusy to equant isopachous cements found in C. caespitosa bafflestone indicate marine phreatic conditions, possibly influenced by continental fresh water influx, as suggested by the slightly negative δ18O. This was likely related to activity of the paleo-Corvino River that partially eroded into the coral buildup. Secondary porosity, as evidenced by the dissolution of many bioclasts, notably the corallites and bivalves, occurs especially in the marine breccia, likely the result of fresh water influx. Bioerosion structures, produced by boring marine organisms, are widespread in the breccia, as well as being common across the whole outcrop.
C. caespitosa is well adapted to the sea temperature seasonality of the Mediterranean Sea, which experiences large changes in temperature, from 8 °C during winter up to 28 °C in summer (Schiller 1993; Kružić 2005; Rodolfo-Metalpa et al. 2006). It is well known that skeletal aragonite of corals provides chemical and isotopic records that can be interpreted in terms of seawater temperature, salinity, and isotopic composition, as well as oceanic upwelling and anthropogenic input. Indeed, the δ18O, δ13C, Sr/Ca, and Mg/Ca records derived from coral aragonite are widely used as proxies for paleoclimatic and paleoenvironmental reconstructions. Such proxies, typically derived from tropical corals, have been tested on recent Mediterranean C. caespitosa by Silenzi et al. (2005). The authors report that the Sr/Ca ratios show a significant correlation with the sea-surface temperature (SST) observed between the years 1982 and 2000, whereas the δ18O and Mg/Ca ratio data do not appear to fit well with the SST observed. Here, in the areas of coral aragonite preservation, a Sr/Ca ratio of ~13 (mmol/mol), Mg/Ca ~4.7 (mmol/mol) and δ18O-1,4 ‰ (PDB) were measured. Such values, although semi-quantitative (EDS), are comparable with those obtained by Silenzi et al. (2005), suggesting a range of annual temperature between 12° and 18°, although the Sr/Ca ratio of Diamante corals could indicate a cooler temperature (<10°).
In the Quaternary reef system of French Polynesia, shallower reefal microbialites are a late-stage encrustation of the dead parts of coral colonies and/or of associated encrusting organisms (red algae and foraminifers), in turn creating surface crusts (Camoin et al. 1999, 2006). Deeper slope microbialites mainly correspond to the final stage of a biological succession in which shallow-water corals and associated encrusting organisms are replaced by deeper-water assemblages of red algae and foraminifers, thus indicating an overall deepening sequence. Both biological successions record changes in water quality, and especially an increase in nutrients; in fact, in shallow-water settings, increased alkalinity, and nutrient availability in interstitial waters were related to surface fluxes, terrestrial groundwater seepage, and periodic runoff, whereas slope environments were exposed to continuous upwelling of deeper waters during sea-level rise (Camoin et al. 1999, 2006).
A substantial difference exists between the microbialite of French Polynesia and the microbialites of Punta Diamante, since the latter postdate both the coralgal bioconstruction and the continental breccia; in fact, their origin is related to a new phase of submergence, implying that the microbialites may have formed in a quite different environment. A further difference exists in the stable isotope composition. Microbialites from those tropical islands and many others (e.g., Papeete in Camoin et al. 1999; Mauritius and Mayotte in Camoin et al. 1997; Jamaica in Land and Goreau 1970; Heron Island in Camoin et al. 1999 and Vanuatu in Cabioch et al. 2006), display carbon and oxygen isotope mean values of +2.89 ‰ δ13C, and −0.16 ‰ δ18O. These values are respectively typical of non-enzymatic fractionation and of precipitates at equilibrium with normal sea water (Schidlowski 2000). Microbialites from Punta Diamante display mean carbon and oxygen isotope values of –8.5 ‰ δ13C, and −3.5 ‰ δ18O. The carbon value confirms an input of CO2 from microbially degraded organic matter, whereas the negative value of oxygen could suggest the presence of an ice cap. However, a slight freshwater input cannot be excluded (Peirano et al. 2009), possibly during a phase of early deglaciation, since diagenetic mixing with fresh water is excluded on the grounds of the microbialites being marine and not recrystallized. During this stage, in fact, the last recorded in the Punta Diamante deposits, surface influx of sediment (from rivers) led to the trapping of reworked rock fragments in microbialites and the interlayering of fine-grained sands with microbial crusts. It is reasonable to forecast an increased input of detrital organic matter into the shallow-water environment at this time, and this may have significantly hampered the accretion of coral reefs and favored the growth of algal turf (Hallock 1988; Mutti and Hallock 2003), and, eventually, the development of microbialites (Sprachta et al. 2001).
Conclusions
A paleoecological study of the mid-latitude coralgal bioconstruction of Punta Diamante, along the northern Tyrrhenian coast of Calabria, and associated biofacies, has led to a more precise interpretation of the geological history of this coastal sector during the late Middle Pleistocene and possibly early Late Pleistocene. The Quaternary sedimentary cover consists of a coralgal bioconstruction, dominated by C. caespitosa bafflestone, a red algae bindstone, and a S. gaederopus-rich boundstone, breccia deposit and endolithic microbialites.
Field observations coupled with microfacies and isotope analyses undertaken on selected samples of the main sedimentary units document the chronological succession of the facies, an alternation of phases of submergence and emergence related to climate change, and the marine paleoenvironmental parameters during the submergence phases.
During the first submergence phase (MIS 9) the coralgal bioconstruction formed in a moderate shallow-marine environment at a depth between 6 and 20 m, swept by moderate- to high-energy sea currents and occasionally affected by storm surges; the general range of annual temperature was between 12 and 18 °C. The biological succession within the coralgal body from C. caespitosa to red algae in the upper part of the bioconstruction was likely the result of occasional influx of coarse clastic sediment that partially buried the C. caespitosa colonies and provided a suitable base for the pioneer encrusting organisms.
During a subsequent glacial stage, the sea-level lowering combined with tectonic uplift induced the partial emergence of the coralgal body that was then overlain by the breccia unit.
Finally, a second submergence phase affected the area, likely during the last interglacial period (isotope stage 5), as testified by the presence of endolithic microbialite preferentially deposited inside a fracture that affects the bioconstruction, thus postdating both the coralgal bioconstruction and the breccia. Unlike modern tropical microbialites that usually represent the late/final stage of a biological succession during a relative sea-level rise/deepening, the endolithic microbialite records a clearly separate phase of deposition, more recent in time than the previous sedimentary sequence. Its formation occurred in a marine environment characterized by cooler water, with respect to the previous submergence phase, possibly during an early stage of deglaciation. The low temperature, possibly combined with a very shallow-water depth and an excessive amount of organic matter, could have played a role in preventing a further C. caespitosa colonization while favoring the in-place precipitation of microbial carbonate.
References
Adey WH (1986) Coralline algae as indicators of sea-level. In: van de Plassche O (ed) Sea-level research: a manual for the collection and evaluation of data. Free University of Amsterdam, Amsterdam, pp 229–279
Aguirre J, Jiménez AP (1998) Fossil analogues of the present day ahermatypic Cladocora caespitosa coral banks: sedimentary setting, dwelling community, and taphonomy (Late Pliocene, W Mediterranean). Coral Reefs 17:203–213
Amodio-Morelli L, Bonardi G, Colonna V, Dietrich D, Giunta G, Ippolito F, Liguori V, Lorenzoni S, Paglionico A, Perrone V, Piccarretta G, Russo M, Scandone P, Zanettin-Lorenzoni E, Zuppetta A (1976) L’arco calabro-peloritano nell’orogene appenninico-maghrebde. Mem Soc Geol Ital 17:1–60
Antonioli F, Ferranti L, Fontana A, Amorosi A, Bondesan A, Braitenberg C, Dutton A, Fontolan G, Furlani S, Lambeck K, Mastronuzzi G, Monaco C, Spada G, Stocchi P (2009) Holocene relative sea-level changes and vertical movements along the Italian and Istrian coastlines. Quatern Int 206:102–133
Basso D (1998) Deep rhodolith distribution in the Pontian Islands, Italy: a model for the paleoecology of a temperate sea. Palaeogeogr Palaeoclimat Palaeoecol 137:173187
Bernasconi MP, Corselli C, Carobene L (1997) A bank of the scleractinian coral Cladocora caespitosa in the Pleistocene of the Crati valley (Calabria, southern Italy): growth versus environmental conditions. Boll Soc Paleontol Ital 36(1–2):53–61
Boenzi F, Palmentola G (1975) Osservazioni sulle tracce glaciali della Calabria. Boll Soc Geol Ital 94:961–977
Bosence DWJ (1991) Coralline algae: mineralization, taxonomy, and palaeoecology. In: Riding R (ed) Calcareous algae and stromatolites. Springer, Heidelberg, pp 98–113
Braga JC, Aguirre J (2001) Coralline algal assemblages in upper Neogene reef and temperate carbonates in southern Spain. Palaeogeogr Palaeoclimatol Palaeoecol 175:27–41
Cabioch G, Camoin G, Webb GE, Le Cornec F, Garcia Molina M, Pierre C, Joachimski MM (2006) Contribution of microbialites to the development of coral reefs during the last deglacial period: case study from Vanuatu (South-West Pacific). Sed Geol 185:277–295
Camoin GF, Webster JM (2015) Coral reef response to Quaternary sea-level and environmental changes: state of the science. Sedimentology 62:401–428
Camoin GF, Colonna M, Montaggioni LF, Casanova J, Faure G, Thomassin BA (1997) Holocene sea level changes and reef development in southwestern Indian Ocean. Coral Reefs 16:247–259
Camoin G, Gautret P, Montaggioni LF, Cabioch G (1999) Nature And Environmental Significance Of Microbialites In Quaternary Reefs: the Tahiti Paradox. Sed Geol 126:271–304
Camoin G, Cabioch G, Eisenhauer A, Braga JC, Hamelin B, Lericolais G (2006) Environmental significance of microbialites in reef environments during the last deglaciation. Sed Geol 185:277–295
Canals M, Ballesteros E (1997) Production of carbonate particles by phytobenthic communities on the Mallorca-Menorca shelf, northwestern Mediterranean sea. Deep-Sea Res II 44:611–629
Carobene L, Dai Pra G (1990) Genesis, chronology and tectonics of the Quaternary marine terraces of the Tyrrhenian coast of northern Calabria (Italy). Their correlation with climatic variations. Il Quaternario 3:75–94
Carobene L, Dai Pra G (1991) Middle and upper Pleistocene sea level highstands along the Tyrrhenian coast of Basilicata (southern Italy). Il Quaternario 4(1a):173–202
Carobene L, Ferrini G (1993) Morphological, sedimentological and tectonics features of Diamante-M. Carpinoso marine terrace flight (Tyrrhenian coast of northern Calabria, Italy)). Earth Surf Proc Land 18:225–239
Carobene L, Dai Pra G, Gewelt M (1986) Niveaux marins du Pléistocène moyen-supérieur de la cote tyrrhénienne de la calabre (italie méridionale). Datations 230th/234u et tectonique récente. Z Fürgeomorphol 62(suppl):141–158
Casado-Amezua P, Garcia-Jiménez R, Kersting DK, Templado J, Coffroth JA, Merino P, Acevedo I, Machordom A (2011) Development of microsatellite markers as a molecular tool for conservation studies of the Mediterranean reef builder coral Cladocora caespitosa (Anthozoa, Scleractinia)). J Hered 102(5):622–626
Cello G, Invernizzi C, Mazzoli S (1996) Structural signature of tectonic processes in the Calabrian Arc, southern Italy: evidence from the oceanic-derived Diamante-Terranova unit. Tectonics 15(1):187–200
Chamberlain YM, Irvine LM (1994) Lithophylloideae. In: Irvine LM, Chamberlain YM (eds) Seaweeds of the British Isles. 1. Rhodophyta. Part 2B, Corallinales, Hildebrandiales. Natural History Museum, London, pp 59–112
Copat Marconi G, Lena G, Zezza F (1981) Segnalazione del ritrovamento e della datazione di Cladocora caespitosa nel livello dei due-quattro metri nel tratto di costa Calabro-Lucana tra Maratea e Belvedere Marittimo. Rend Soc Geol Ital 4:373–374
Cormaci M, Furnari G, Giaccone G, Colonna P, Mannino AM (1985) Metodo sinecologico per la valutazione degli apporti inquinanti nella rada di Augusta (Siracusa). Boll Accad Gioenia Sci Nat 18:829–850
Dardano M (1992) Notes on Spondylus ictericus (Reeves, 1856). Hawaiian Shell News 40:1
Delamotte M, Vardala-Theodorou E (2001) Shells from the Greek Seas. The Goulandris Natural History Museum, Athens, p 323
Di Geronimo R, Alongi G, Giaccone G (1993) Formazione organogene a Lithophyllum lichenoides Philippi (Rhodophyta, Corallinales) nel Mesolitorale di Capo S. Alessio (Sicilia orientale). Boll Accad Gioenia Sci Nat 26:145–172
Dupraz C, Reid RP, Braissant O, Decho AW, Norman RS, Visscher PT (2009) Processes of carbonate precipitation in modern microbial mats. Earth Sci Rev 96:141–162
Ferranti L, Antonioli F, Mauz B, Amorosi A, Dai Pra G, Mostronuzzi G, Monaco C, Orru P, Pappalardo M, Radtke U, Renda P, Romano P, Sanso P, Verrubbi V (2006) Markers of the last interglacial sea-level high stand along the coast of Italy: tectonic implications. Quatern Int 145–146:30–54
Ferranti L, Santoro E, Mazzella ME, Morelli D (2009) Active transpression in the northern Calabria Apennines, southern Italy. Tectonophyiscs 476:226–251
Hallock P (1988) The role of nutrient availability in bioerosion: consequences to carbonate buildups. Palaeogeogr Palaeoclimatol Palaeoecol 63(1–3):275–291
Hamel G, Lemoine MP (1953) Corallinacées de France et d’Afrique du Nord. Arch Mus Hist Nat Paris Sér 7(1):15–136
Hoefs J (2009) Stable isotope geochemistry. Springer, Berlin, pp 1–286. doi:10.1007/978-3-540-70708-0
Hoefs J (2011) Isotopes and Geobiology. In: Reitner J, Thiel V (eds) Encyclopedia of Geobiology, Encyclopedia of Earth Science Series. Springer, Heidelberg, pp 511–516
Hughes PD, Woodward JC, Gibbard PL (2006) Quaternary glacial history of the Mediterranean mountains. Prog Phys Geogr 30(3):334–364
Jones B, Hunter IG (1995) Vermetids buildups from Grand Cayman. Br West Indies J Coast Res 11:973–983
Koukouras A, Kühlmann DHH (1991) Rasenkorallen als Biotope in der Agais. Naturwissenschaftliche Rundschau 44(11):444–445
Kružić P (2005) Ecology of the coral Cladocora caespitosa (Linnaeus, 1767) and its banks in the Adriatic Sea. Ph.D. thesis, University of Zagreb, pp 1–198
Kružić P, Benković L (2008) Bioconstructional features of the coral Cladocora caespitosa (Anthozoa, Scleractinia) in the Adriatic Sea (Croatia). Mar Ecol (Berl) 29:125–139. doi:10.1111/j.1439-0485.2008.00220.x
Kružić P, Požar-Domac A (2003) Banks of the coral Cladocora caespitosa (Anthozoa, Scleractinia) in the Adriatic Sea. Coral Reefs 22(4):536
Külmann DHH (1996) Preliminary report on Holocene sub-marine accumulation of Cladocora caespitosa (L., 1767) in the Mediterranean. Gottinger Arb Geol Paläont Sb2:65–69
Külmann DHH (1996) Preliminary report on Holocene sub-marine accumulation of Cladocora caespitosa (L., 1767) in the Mediterranean. Göttinger Arbeiten zur Geologie und Paläontologie Sb2, 65-69 Kühlmann DHH, Chintiroglou C, Koutsoubas D, Koukouras A (1991) Korallenriffe in Mittelmeer? Naturwissenschaftliche Rundschau 44(8):316
Laborel J (1987) Marine biogenic constructions in the Mediterranean, a review. Sci Rep Port-Cros Natl Park 13:97–126
Lacelle D (2007) Environmental setting, (micro) morphologies and stable C–O isotope composition of cold climate carbonate precipitates-a review and evaluation of their potential as paleoclimatic proxies. Quatern Sci Rev 26:1670–1689. doi:10.1016/j.quascirev.2007.03.011
Lamprell KL (1986) Spondylus—spiny oyster shells of the world. Backhuys, Leiden
Lamprell KL (1998) Recent Spondylus species from the Middle East and adjacent regions, with the description of two new species. Vita Marina 45(1–2):41–60
Lamprell KL (2001) Spondylids from the Mediterranean Sea and Atlantic Ocean (Mollusca: Bivalvia: Spondylidae). Mem Queens Mus 46:611–622
Land LS, Goreau TF (1970) Submarine lithification of Jamaican reefs. J Sed Petrol 40:457–462
Lumare F (1996) Sulla scogliera a Cladocora di Crotone e le sue biocenosi. Rend Accad Natl XL 4:101–131
Maier E, Titschack J (2010) Spondylus gaederopus: a new Mediterranean climate archive—based on high-resolution oxygen and carbon isotope analyses. Palaeogeogr Palaeoclimatol Palaeoecol 291:228–238
Martindale W (1992) Calcified epibionts as palaeoecological tools: examples from the Recent and Pleistocene reefs of Barbados. Coral Reefs 11:167–177
Montaggioni LF, Camoin GF (1993) Stromatolites associated with coralgal communities in Holocene high-energy reefs. Geology 21:149–152
Montagna P, McCulloch M, Mazzoli C, Silenzi S, Odorico R (2007) The non-tropical coral Cladocora caespitosa as the new climate archive for the Mediterranean: high-resolution (weekly) trace element systematics. Quat Sci Rev 26:441–462. doi:10.1016/j.quascirev.2006.09.008
Morri C, Peirano A, Bianchi CN, Sassarini M (1994) Present-day bioconstructions of the hard coral, Cladocora caespitosa (L.) (Anthozoa, Scleractinia), in the Eastern Ligurian Sea (NW Mediterranean). Biol Mar Mediterr 1(1):371–372
Mutti M, Hallock P (2003) Carbonate systems along nutrient and temperature gradients: some sedimentological and geochemical constraints. Int J Earth Sci 92:465–475
Peirano A, Morri C, Mastronuzzi G, Nike Bianchi C (1997) The coral Cladocora caespitosa (Anthozoa, Scleractinian) as a bioherm builder in the Mediterranean Sea. Mem Descr Carta Geol d’Ital 52:59–74
Peirano A, Morri C, Bianchi CN (1999) Skeleton growth and density pattern of the temperate, zooxanthellate scleractinian Cladocora caespitosa from the Ligurian Sea (NW Mediterranean). Mar Ecol Prog Ser 185:195–201
Peirano A, Morri C, Bianchi CN, Aguirre J, Antonioli F, Calzetta G, Carobene L, Mastronuzzi G, Orrù P (2004) The Mediterranean coral Cladocora caespitosa: a proxy for past climate Fluctuations? Glob Planet Change 40:195–200. doi:10.1016/S0921-8181(03)00110-3
Peirano A, Kružić P, Mastronuzzi G (2009) Growth of Mediterranean reef of Cladocora caespitosa (L.) in the Late Quaternary and climate inference. Facies 55:325–333
Perri E, Spadafora A (2011) Evidence of microbial biomineralization in modern and ancient stromatolites. In: Seckbach J, Tewari V (eds) The stromatolites: interaction of microbes with sediments, Cellular origin, life in extreme habitats and astrobiology, vol 18. Springer, Berlin, pp 631–649
Perri E, Tucker ME, Spadafora A (2012a) Carbonate organo-mineral micro- and ultrastructures in sub-fossil stromatolites: Marion Lake, South Australia. Geobiology 10:105–117
Perri E, Manzo E, Tucker M (2012b) Multi-scale study of the role of the biofilm in the formation of minerals and fabrics in calcareous tufa. Sed Geol 185:267–275
Poppe GT, Goto Y (1993) European seashells. Hemmen, Wiesbaden, p 2
Reitner J (1993) Modern cryptic microbialite/metazoan facies from Lizard Island (Great Barrier Reef, Australia), formation and concepts. Facies 29:3–40
Riding R (2011) Microbialites, stromatolites, and thrombolites. In: Reitner J, Thiel V (eds) Encyclopedia of Geobiology, Encyclopedia of Earth Science Series. Springer, Heidelberg, pp 635–654
Riegl B (1995) Effects of sand deposition on scleractinian and alcyonacean corals. Mar Biol 121:517–526
Rodolfo-Metalpa R, Richard C, Allemand D, Bianchi CN, Morri C, Ferrier-Pagès C (2006) Response of zooxanthellae in symbiosis with the Mediterranean corals Cladocora caespitosa and Oculina patagonica to elevated temperatures. Mar Biol 150:45–55
Schidlowski M (2000) Carbon isotopes and microbial sediments. In: Riding R, Awramik SM (eds) Microbial sediment. Springer, Berlin, pp 84–95
Schiller C (1993) Ecology of the symbiotic coral Cladocora caespitosa (L.) (Faviidae, cleractinia) in the Bay of Piran (Adriatic Sea): I distribution and biometry. PSZN: Mar Ecol 14(3):205–219
Silenzi S, Bard E, Montagna P, Antonioli F (2005) Isotopic and elemental records in a non-tropical coral (Cladocora caespitosa): discovery of a new high-resolution climate archive for the Mediterranean Sea. Glob Planet Change 49:94–120. doi:10.1016/j.gloplacha.2005.05.005
Sprachta S, Camoin G, Golubic S, Le Campion Th (2001) Microbialites in a modern lagoonal environment: nature and distribution (Tikehau Atoll, French Polynesia). Palaeogeogr Palaeoclimatol Palaeoecol 175:103–124
Woelkerling WJ (1996) Subfamily Lithophylloideae. In: Womersley HBS (ed) The marine benthic flora of Southern Australia. Rhodophyta Part IIIB. Australian Biological Resources Study, Canberra, pp 214–237
Zibrowius H (1980) Les scléractiniaires de la méditerranée et de l’Atlantique nord-oriental. Mém l’Inst Océanograph 11:1–284
Acknowledgments
We are indebted to Daniela Basso, University of Milano-Bicocca, for valuable advice concerning identification of red algae. Helpful, constructive, outside review of the manuscript was made by Maurice Tucker, University of Bristol. We also thank Petar Kružić and an anonymous reviewer for their useful comments. Funds were provided by the University of Calabria (MIUR Grant ex-60 %).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bernasconi, M.P., Cefalà, M. & Perri, E. Mid-latitude coralgal bioconstruction and endolithic microbialites: environmental significance during Quaternary climate variations. Facies 61, 19 (2015). https://doi.org/10.1007/s10347-015-0449-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10347-015-0449-1