Abstract
Vertebrates play key roles as seed dispersers, herbivores, and top predators in tropical ecosystems. Therefore, obtaining population estimates for these species and understanding the factors that affect them are essential for wildlife management since changes in their populations have consequences for entire ecosystems. Vertebrate abundances in tropical forest may be related to habitat characteristics, resource seasonality, and human pressure. However, how ecological variables and human pressure concurrently influence animal abundances is not well understood. We investigated the associations between the number of records of vertebrates (ground-dwelling birds and medium- and large-sized mammals) and habitat features, food availability, and human pressure in a sustainable protected area in the Brazilian Amazon of western Pará, Brazil. Our study design included the recording of animals at 38 camera trap stations, sampling of environmental variables (canopy cover, leaf area index, tree height, and local altitude) and food resources (fruit or prey biomass), and measurement of a hunting pressure proxy (distance from human settlements). Our results indicated that groups responded in different ways: omnivorous mammals were affected positively by local altitude, canopy openness, and leaf area index; game birds were affected positively by local altitude and leaf area index; ungulates were affected negatively by local altitude and positively by food resources; and large rodents were affected only by food resources (positively). In contrast, insectivorous mammals and mesopredators were not affected by any variable we tested. Surprisingly, no groups responded to distance from human access, although the low number of records of large species, such Tapirus terrestris and Dicotyles tajacu, suggests that the sampled area may suffer from significant hunting pressure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Birds and mammals are responsible for seed dispersal of more than 80% of tropical tree species (Peres and Van Roosmalen 2002; Almeida-Neto et al. 2008), which is key to plant recruitment and is a driver of vegetation structure (Schupp et al. 2010). Furthermore, mammals and birds play an essential role as seed predators and herbivores, contributing to the maintenance of forest diversity (Villar et al. 2020b). Such vertebrates occupy various trophic levels, from primary consumers to top predators, connecting complex ecological networks (Vidal et al. 2013), fostering energy flow and biochemical cycling, and returning nutrients to the soil (Villar et al. 2020a).
Additionally, tropical forest birds and mammals also provide ecosystem services. Through seed dispersal, they promote the recruitment of hardwood trees that capture more carbon and help to reduce the effects of climate change (Bello et al. 2015). Numerous plants that provide timber and non-timber forest products depend on animals for their recruitment (Haugaasen et al. 2010; Brocardo et al. 2018). Predators control the populations of species that cause damage to agriculture or are disease transmitters (O’Bryan et al. 2018). In addition, large vertebrates, mainly mammals and birds, are an important protein source for people living in tropical ecosystems throughout the world (Jerozolimski and Peres 2003; Nasi et al. 2011).
These tropical ecosystems are heterogeneous landscapes where environmental characteristics may change spatially and temporally (Alves et al. 2010; Fadini et al. 2021), creating a gradient of habitats that influences the presence and abundance of animal species. Mammals and birds may respond to vegetation type (Haugaasen and Peres 2007; Galetti et al. 2009; Tardio and Da Silveira 2015), to food and water resource (Keuroghlian et al. 2004; Tardio and Da Silveira 2015; Rabelo et al. 2019; Weiler et al. 2020), to water regime seasonality (Mamede and Alho 2006), to altitudinal gradient (Morais et al. 2019), and also to competitor and predator presence (Newsome et al. 2017).
In addition to natural features, birds and mammals in tropical ecosystems have their presence and abundance affected by human pressures such as deforestation (Kinnaird et al. 2003; Weiler et al. 2020), habitat fragmentation (Crooks et al. 2017), fires (Barlow and Peres 2006), logging (Brodie et al. 2015), roadkill (Cáceres 2011; Rosa and Bager 2012), and overhunting (Benítez-López et al. 2017; Sampaio et al. 2022). Overhunting is a particularly powerful threat because it may even affect animal populations in large continuous habitats (Peres and Palacios 2007; Galetti et al. 2017; Rosa et al. 2021c). There are many reported cases of human impacts being related to human access, notably via distance effects (Antunes et al. 2016; Prasniewski et al. 2022). Therefore, determining the factors that contribute to the presence and abundance of animals is necessary to evaluate the effects of human pressures (Jorge et al. 2013).
In this study, we investigated the effects of environmental characteristics (e.g., food source, canopy openness) and human pressure on ground-dwelling medium- and large-sized mammals and birds in a protected area of the Amazon Forest. The Amazon Forest is the world’s largest tropical forest and one of the areas richest in vertebrates (Hawkins et al. 2007; Schipper et al. 2008). It has had an influence on the evolutionary history and biodiversity of the entire Neotropical region (Antonelli et al. 2018). Improving our understanding of the concurrent role of ecological variables and human pressure on animal abundances can assist in planning effective conservation and management strategies (Michalski et al. 2015).
Accordingly, we tested the following hypotheses:
-
H1. Response to local resources: Animals change habitat use according to resource availability; therefore, we expected the number of animal records to be higher in sites with more food resources during the survey period.
-
H2. Habitat structure: The occurrence of animals is related to habitat characteristics. We expected animal records to be positively or negatively related to structure, depending on the group of species studied. For example, sites with higher leaf density might have a greater abundance of birds and mammals in response to the increase in refuge availability.
-
H3. Human pressure: The distance from human access (e.g., roads, rivers) and settlements is related to hunting pressure in the Amazon Forest and thus to vertebrate occurrence. Therefore, we expected the number of terrestrial bird and mammal records to increase with increasing distance from roads and human settlements.
Materials and methods
Study area
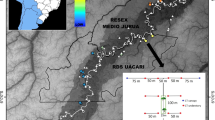
This study was carried out in the Tapajós National Forest (TNF), a protected area covering 527,319 ha (IUCN category VI—protected area with sustainable use of natural resources) (ICMBio 2019), located on the right bank of the Tapajós River in the Tapajós Endemism Area of the Amazon Forest, western Pará state, Brazil (Silva et al. 2005). The altitude ranges from 8 to 330 m, with a predominance of dense and open rain forests. Mean annual temperature is 25.5 °C, mean annual rainfall is 1820 mm, and there is a 4-month dry season between August and November, although normally, there is some precipitation in all months of the year (ICMBio 2019).
Pre-Columbian settlements have been present in this region for at least 4500 years (Stenborg et al. 2012; Maezumi et al. 2018). European colonization began in the seventeenth century with the foundation of Portuguese villages (Santarém and Aveiro) (IBGE 1957), although colonization intensified in the early twentieth century with the start of the rubber boom (ICMBio 2019). The TNF was established in 1974 as a sustainable protected area for the conservation and sustainable use of natural resources. Traditional communities living within its limits were allowed to remain and exploit natural resources (ICMBio 2019). Approximately 9000 people live in the TNF in 31 communities, some of which are recognized as indigenous people (ICMBio 2019). The construction of the Cuiabá-Santarém highway (BR-163) in 1970 changed the landscape around the TNF, leading to loss of forest cover along the highway and access from unpaved roads, which increased with the expansion of soy bean culture in this region after the mid-1990s (Fearnside 2007; Garrett et al. 2013). Due to forest loss and habitat fragmentation in this landscape, the TNF is considered essential to the conservation of mammals in the region (Sampaio et al. 2010; Rosa et al. 2021c).
Currently, the TNF is managed through a reduced-impact timber-harvest system, which is restricted to areas defined by a Management Plan established by the Chico Mendes Institute for Biodiversity Conservation (ICMBio), the governmental agency responsible for the administration of federal protected areas in Brazil, with the participation of local communities living in the TNF. The TNF residents also extract non-timber products to consume and sell and practice subsistence hunting. Although subsistence hunting is only allowed for residents, the TNF suffers from poaching by outsiders (ICMBio 2019) Fig.1.
Study design
We sampled mammals and game birds with camera traps, a method widely used to monitor wildlife in tropical forests (Ahumada et al. 2013; Jansen et al. 2014). Our sampling design was composed of four Rapid Assessments and Long-term Ecological Research modules (RAPELD) established to monitor biodiversity in the TNF as part of the Brazilian Program for Biodiversity Research (PPBio) (Magnusson et al. 2005; Rosa et al. 2021a). Each is rectangular in shape, 1 km in width, and 5 km in length and has 10 regularly spaced plots (250-m length and variable width following the terrain contour) separated by a minimum distance of 1 km (Magnusson et al. 2013). We installed a single unbaited camera trap (Bushnell 12Mp Natureview Cam Essential HD Low Glow®, n = 12; Primus Proof Cam 3 Review®, n = 5; and Moultrie A5 Low Glow Game Camera®, n = 3) deployed out of the RAPELD module trail (5 to 20 m), with a total of ten camera trap stations per module (Fig. 2). The position of each camera trap was recorded with a GPS device (Garmin 62S, Garmin International Inc., Kansas, USA). We installed the cameras on trees, 30–40 cm from the ground, close to paths used by animals, and programmed them to record photographs (three photographs per trigger) or videos (10 s), with a 1-s delay, operating for 24 h/day for 34 days. Due to the number of camera traps being less than the number of stations and logistical reasons, we sequentially sampled the modules between July and December 2019 (dry season). We obtained data for only 38 stations because two camera traps failed to work.
Response variables
We used the number of records of game birds and medium- (> 1 kg) and large-sized (> 20 kg) mammals recorded per camera trap as surrogates for animal abundance (Haugaasen and Peres 2007; Galetti et al. 2009; Hawes and Peres 2014; Hong et al. 2015; Michalski et al. 2015; Alvarenga et al. 2018; Scabin and Peres 2021). Although the use of camera trap records as a measure of animal abundance does not account for imperfect and variable detection (Sollmann et al. 2013), some studies have shown that they are correlated (Rovero and Marshall 2009; Parsons et al. 2017; Palmer et al. 2018). Observations of the same species at a camera trap station were considered independent records only after an interval of 24 h between them. When more than one individual of a species was recorded in a single event, we counted them and considered the sum of individuals as the number of records. We did that because group size may be affected by habitat characteristics and human pressure (Reyna-Hurtado et al. 2016), and so the variation in group size could be a response to environmental changes or human pressure in our study area.
Species were identified using standardized guides (Reis et al. 2010, 2015). We grouped them according to their diets and whether they were used as game by humans as follows: terrestrial game birds, omnivorous mammals, insectivorous mammals, large rodents, ungulates, mesopredators, and top predators (Ahumada et al. 2011; Kutt and Gordon 2012; Michalski et al. 2015; Paredes et al. 2017). Game birds include all large ground-dwelling birds; omnivorous mammals include mammals that have omnivorous diet and are lesser preferred by hunters in our study region (Torres et al. 2021); large rodents include two preferred rodents by hunters (Torres et al. 2021); insectivorous mammals included ant eaters and armadillos; the latter ones are a preferential hunted group (Torres et al. 2021) and, despite including other food items in their diet, have a predominant consumption of insects and other invertebrates (Sikes et al. 1990; da Silveira Anacleto 2007; Whitaker et al. 2012); ungulates included hoofed mammals, a very preferred group by hunters (Torres et al. 2021); and finally, mesopredators and top predators include, respectively, medium and large feline species found in our study area (Table 1).
Explanatory variables
Food resources
At the end of each survey, two observers collected all fruits and seeds on the ground in plots (250-m long and 1-m wide) adjacent to each camera trap (5 to 20 m). Fruits and seeds were dried in an oven at 70 °C for 48 h and weighed (Silva et al. 2019). We used dry fruit/seed biomass as an explanatory variable for game birds, omnivorous mammals, large rodents, ungulates, and insectivorous mammals. For the last group, we included fruit biomass as a proxy of insect abundance, since insects are positively related to fruit availability (Pizo and Oliveira 2000; Szinwelski et al. 2015), and Dasypus species consume fruits as well as invertebrates. We used the biomass of the prey (Dasypus spp., omnivores, large rodents, and game birds) recorded by the camera traps as food resources for mesopredators. To obtain the total biomass for each camera trap, we multiplied the mean body mass of a given species by the number of records and summed this for all species. Biomass data were log-transformed to improve model fit.
Habitat structure
We chose habitat features that have been widely shown to impact ecology, density, and abundance of ground-dwelling mammals and birds, including canopy openness, mean height of trees, leaf area in the lower forest stratum (a between 0 and 15 m high), and local altitude. The first three variables were measured along each plot (250 m) using a light detection and ranging (LIDAR) ground portable device (Model LD90-3100VHS-FLP, Riegl, Horn, Austria), which scans the environment with a laser for rapid measurement of multiple variables quantifying forest structure (see Torralvo et al. 2020). Thus, the value for each plot represents a mean value obtained along its length for canopy openness (in percentage), leaf area (index), and height tree (meters). We measured the local altitude (meters, at sea level (a.s.l.)) with a hand-held GPS device (Garmin 62S) and checked it using Google Earth, which provides reliable and accurate elevation data (Wang et al. 2017).
Human pressure
We used the distance (meters) of camera trap stations from human residences and roads as an index of human pressure, since hunting activities are commonly linked to distance from such access points (Peres and Lake 2003). We plotted the positions of residences and roads with the aid of Google Earth images and field inspections. Distances from camera trap stations to residences and roads were measured with Qgis 3.4.1.5 (QGIS.org 2021).
Data analysis
We undertook all analyses in R version 3.6.2 (R Core Team 2019). To investigate the influence of the explanatory variables on the number of records of each functional group, we used generalized linear models (GLM) with MASS package (Ripley et al. 2021). We also tested generalized linear mixed models (GLMM), with RAPELD modules as a random factor to account for within-module autocorrelation. However, including the random factor did not improve our models (resulting in larger values of Akaike’s information criterion for small samples (AICc)); thus, we opted for simpler models. Before running the models, we tested the multicollinearity between the explanatory variables using the variance inflation factor (VIF) function (vif.cca) in the vegan package (Oksanen et al. 2019). Due to a high VIF, we excluded two variables (tree height and distance from roads) with VIF > 5 (Zuur et al. 2009). Tree height was significantly related to local altitude; therefore, we retained the latter because in addition to influences on forest structure, it may be a surrogate for other habitat characteristics, such as litter deposition, soil type, and water drainage (Costa and Magnusson 2010). Distance from roads and distance from residences were also related; we retained the latter because we considered it being more important since it represented the constant presence of people who might engage in hunting (Torres et al. 2021).
We used the negative binomial distribution with log link instead of the Poisson distribution because of overdispersion, with the exception of the data for mesopredators, for which we used the binomial distribution due to the low number of records (absence or presence). We tested the model adjustment the residual diagnostic in the DHARMa package (Hartig and Lohse 2020). Analysis of the spatial autocorrelation of residuals (ncf package; Bjornstad 2020) indicated that the game bird group was spatially autocorrelated when using a negative binomial distribution; therefore, we log-transformed the raw data to obtain an approximately normal distribution and used it in a model assuming Gaussian errors, which resulted in residuals without spatial autocorrelation. The pseudo-R2 statistics of the models were calculated using Efron’s pseudo-R2 function available in the performance package (Lüdecke et al. 2021). We plotted partial regressions using the visreg package (Breheny et al. 2020).
Results
We recorded a total of 21 medium- and large-sized mammal species and eight game bird species (Table 1). The number of records ranged from 2 to 59 per camera trap station (mean ± SD = 21.7 ± 15.08). The functional group most recorded was large rodents with 411 records (mean = 10.8 ± 8.8; range = 0–40), present at 37 of the 38 camera trap stations, followed by game birds with 193 records (mean = 5.07 ± 6.9; range = 0–38; present at 36 stations), ungulates with 89 records (mean = 2.34 ± 4.8; range = 0–27; present at 22 stations), insectivorous mammals with 62 records (mean = 1.63 ± 2.2; range = 0–12; present at 26 stations), omnivorous mammals with 54 records (mean = 1.42 ± 2.3; range = 0–8; present at 18 stations), mesopredators with 11 records (mean = 0.28 ± 0.45; range = 0–1; present at 11 stations), and top predators with only 5 records (mean = 0.13 ± 0.41; range = 0–2; present at 4 stations).
The local altitude ranged from 83 to 209 m a.s.l. (mean = 149.9 ± 39.4 m a.s.l.), leaf area index from 1.21 to 3.09 (mean = 2.01 ± 0.43), canopy openness from 15.2 to 69.6% (mean = 42.1 ± 11.5%), distance from human residences from 1.6 to 6.4 km (mean = 3.85 ± 1.25 km), fruit biomass from 0 to 6.77 kg (mean = 0.91 ± 1.59 kg), and prey biomass (food resource for mesopredators) from 2.1 to 267.4 kg (mean = 61.7 ± 55.3 kg).
Large rodents were significantly positively affected by food resources. Game birds responded positively to local altitude and leaf area index. Ungulates were recorded more at lower altitudes and were positively affected by fruit biomass. Omnivorous mammals were positively affected by leaf area index, canopy openness, and local altitude. Insectivorous mammals and mesopredators did not respond to any variable. The low number of records of top predators did not allow the analysis for this group. Surprisingly, distance from human residences did not affect any functional group (Table 2; Fig. 3).
Discussion
Our results show different patterns in animal records in the same continuous forest according to differences in local habitat characteristics. The habitat heterogeneity of tropical ecosystems, and especially in the Amazon, is considered a key element in maintaining high levels of biodiversity and community structure (Haugaasen and Peres 2007; Ahumada et al. 2011; Rojas-Ahumada et al. 2012; Maximiano et al. 2020).
The results supported our first hypothesis only for large rodents and ungulates, which were more frequent in the plots with high fruit biomass, indicating that these groups of animals may respond more intensively to changes in the availability of this resource than those in other groups. The diets of Neotropical ungulates and of large rodents are highly frugivorous (Salas and Fuller 1996; Silvius and Fragoso 2003; Keuroghlian et al. 2004), and they are the main taxa involved in the removal of fruits and seeds from the forest floor (Galetti et al. 2015; Camargo-Sanabria and Mendoza 2016; Brocardo et al. 2018; Rosa et al. 2021b). The higher records of ungulates at sites with high fruit biomass may result from the concentration of their activities near fruiting trees (Beck–King et al. 1999; Foerster and Vaughan 2002; Silvius and Fragoso 2003; Keuroghlian et al. 2004; Tardio and Da Silveira 2015). Concurrently, the absence of effect of fruit biomass on game birds and omnivorous mammals may be related to use of other food resources by these groups (Jordano 2000), such as arthropods, which could be more important than fruits for some species. In addition, the largest fruit biomass was found in plots with high incidence of Attalea, whose fruits on the ground are consumed mainly by large rodents and ungulates (Silvius 2002).
Although other groups did not respond to fruit biomass, they were significantly affected by habitat structure, corroborating our second hypothesis. Local altitude positively affected game birds, most likely as an indirect effect of vegetation changes along the altitudinal gradient, which may be related to the type and amount of foodstuffs. For example, Capaverde and collaborators found positive relation between altitude and the number of fruit plants and the insect biomass in Central Amazon (2018). Game birds also positively responded to higher leaf area index, a metric which correlates with high vegetation density in the understory (from zero to 15 m) and so which may provide protection from predators and opportunities for nest hiding by such species (Estrada et al. 2002; Depalma and Mermoz 2019). The protection provided by vegetation may also explain the positive relationship between omnivorous mammals and altitude and leaf area index, since they use dense vegetation to avoid predators (Dutra et al. 2011; Gorini et al. 2012). In addition, a larger net leaf area could also be linked to greater availability of foraging sites. Records of omnivores were dominated by Didelphis marsupialis and Nasua nasua (74% and 18% of the records, respectively). Both are scansorial species, able to climb and forage in understory vegetation (Beisiegel and Mantovani 2006; Rossi et al. 2010). Omnivore records were also related to canopy openness, which may be associated with increases in such food sources as fruits, small vertebrates, and arthropods (Levey 1988; Basset et al. 2001; Silveira et al. 2010; Garda et al. 2013; Tardio and Da Silveira 2015).
In contrast to game birds and omnivorous mammals, ungulates were negatively affected by local altitude, and insectivorous mammals (armadillos and ant eaters) also tended to have a negative relationship with local elevation. Since lower-altitude sites may be closer to streams (Capaverde Jr et al. 2018), they may provide greater access to water resources, a factor important for explaining the abundance of large Neotropical mammals (Michalski et al. 2015; Paredes et al. 2017; Antunes et al. 2019; Weiler et al. 2020). In addition, the lower-elevation sites also show differences in vegetation composition and soil moisture (Costa and Magnusson 2010). The quality of the understory vegetation is important for browser species, such as Mazama spp. and Tapirus terrestris (Salas and Fuller 1996; Gayot et al. 2004). Armadillos (insectivorous) and peccaries (ungulates) both forage at sites with humid soil and avoid high temperatures (Desbiez et al. 2009; Goulart et al. 2009).
Mesopredators (Leopardus spp.) did not respond to any measured variable. Previous studies have shown this group to be affected by forest cover (Harveson et al. 2004; Goulart et al. 2009; Wang et al. 2019); however, with our study site being a continuous area, forest cover may have varied little between sampling plots. Furthermore, these species are generalists in terms of their habitat use, with high mobility and large home ranges (Dillon and Kelly 2008; Kasper et al. 2016); therefore, micro-scale habitat features may have few effects on their presence. In addition, the greater part of the diet of Leopardus spp. is based on small mammals (< 1 kg) (Wang 2002), a group not investigated in our study. The different Leopardus species may also have different habitat requirements, and grouping them together may have masked these specificities.
No groups responded to distance from human settlements; thus, our third hypothesis (human pressure) was not supported. Several previous studies have demonstrated that human access or settlements can negatively impact animal populations and alter their habitat use in tropical forests (Peres and Lake 2003; Cruz et al. 2014; Antunes et al. 2016; Benítez-López et al. 2017; Morais et al. 2019). Hunters in the Amazon walk up to 9 km into the forest (Peres and Lake 2003); therefore, our result must be interpreted with caution, since the maximum distance between plots and human residences or road access was just over 6 km, implying that all our sampling plots may be similarly impacted by human pressure. For instance, Peres and Lake (2003) showed that some ungulates and game birds may need more than 4–5 km to be released from hunting pressure in the Amazon forest, while Prasniewski and colleagues (2022) showed that the effects of accessibility may extending up to 10 km inside the forest, in an analysis of incidence of illegal activities, including poaching, in two protected areas of the Atlantic Forest. Hunting is common in our study area region, with large rodents, ungulates, insectivorous mammals, and game birds being the main prey consumed by local people (Torres et al. 2018, 2021). The few records of the largest species (Tapirus terrestris and Dicotyles tajacu) and the absence of Tayassu pecari in our plots may indicate a defaunation process linked to hunting pressure (Peres and Palacios 2007; Antunes et al. 2016; Galetti et al. 2017; Rosa et al. 2021c).
Conclusion
Our results indicate that habitat features are important predictors of vertebrate presence in forested areas of the Amazon Basin, although different groups of animals respond in different ways. This finding reinforces the role of habitat heterogeneity in animal assemblage structure. Because environmental characteristics vary in their importance to different groups, conservation strategies demand the protection of a diversified range of contiguous habitats. Large reserves are widely used as a conservation tool in the Brazilian Amazon, and although they are normally large enough to include a heterogeneous landscape, 62% of the protected surface of Brazilian protected areas in Amazon is destined for sustainable use of natural resources (Veríssimo et al. 2011), as is our study site. This means that there is a likelihood of habitat quality changing over time due to human use; therefore, we recommend constant monitoring to evaluate how biodiversity responds to anthropogenic changes. Future planning must account for such eventualities. We also recommend future studies to address the distances traveled to hunt as means of better understanding the impact of humans on animal populations in this region.
Data availability
The datasets generated during the current study are available as supplemental material.
References
Ahumada JA, Hurtado J, Lizcano D (2013) Monitoring the status and trends of tropical forest terrestrial vertebrate communities from camera trap data: a tool for conservation. PLoS ONE 8:e73707
Ahumada JA, Silva CEF, Gajapersad K et al (2011) Community structure and diversity of tropical forest mammals: data from a global camera trap network. Philos Trans R Soc B Biol Sci 366:2703–2711
Almeida-Neto M, Campassi F, Galetti M et al (2008) Vertebrate dispersal syndromes along the Atlantic forest: broad-scale patterns and macroecological correlates. Glob Ecol Biogeogr 17:503–513
Alvarenga GC, Ramalho EE, Baccaro FB et al (2018) Spatial patterns of medium and large size mammal assemblages in várzea and terra firme forests, Central Amazonia, Brazil. PLoS One 13:e0198120. https://doi.org/10.1371/journal.pone.0198120
Alves LF, Vieira SA, Scaranello MA et al (2010) Forest structure and live aboveground biomass variation along an elevational gradient of tropical Atlantic moist forest (Brazil). For Ecol Manage 260:679–691
Antonelli A, Zizka A, Carvalho FA et al (2018) Amazonia is the primary source of Neotropical biodiversity. Proc Natl Acad Sci 115:6034–6039
Antunes AC, Baccaro F, Caetano Andrade VL et al (2019) Igapó seed patches: a potentially key resource for terrestrial vertebrates in a seasonally flooded forest of central Amazonia. Biol J Linn Soc 128:460–472
Antunes AP, Fewster RM, Venticinque EM et al (2016) Empty forest or empty rivers? A century of commercial hunting in Amazonia. Sci Adv 2:e1600936
Barlow J, Peres CA (2006) Effects of single and recurrent wildfires on fruit production and large vertebrate abundance in a central Amazonian forest. Biodivers Conserv 15:985–1012
Basset Y, Charles E, Hammond DS, Brown VK (2001) Short-term effects of canopy openness on insect herbivores in a rain forest in Guyana. J Appl Ecol 38:1045–1058
Beck-King H, von Helversen O, Beck-King R (1999) Home range, population density, and food resources of Agouti paca (Rodentia: Agoutidae) in Costa Rica: a study using alternative methods 1. Biotropica 31:675–685
Beisiegel BM, Mantovani W (2006) Habitat use, home range and foraging preferences of the coati Nasua nasua in a pluvial tropical Atlantic forest area. J Zool 269:77–87
Bello C, Galetti M, Pizo MA et al (2015) Defaunation affects carbon storage in tropical forests. Sci Adv 1:e1501105
Benítez-López A, Alkemade R, Schipper A (2017) The impact of hunting on tropical mammal and bird populations. Science (80- ) 356:180–183
Bjornstad ON (2020) ncf: spatial covariance functions. https://cran.r-project.org/web/packages/ncf/ncf.pdf. Accessed 1 Feb 2022
Breheny P, Burchett W, Breheny MP (2020) Package ‘visreg’. https://cran.r-project.org/web/packages/visreg/visreg.pdf. Accessed 1 Feb 2022
Brocardo CR, Pedrosa F, Galetti M (2018) Forest fragmentation and selective logging affect the seed survival and recruitment of a relictual conifer. For Ecol Manage 408:87–93. https://doi.org/10.1016/j.foreco.2017.09.046
Brodie JF, Giordano AJ, Ambu L (2015) Differential responses of large mammals to logging and edge effects. Mamm Biol 80:7–13
Cáceres NC (2011) Biological characteristics influence mammal road kill in an Atlantic Forest-Cerrado interface in south-western Brazil. Ital J Zool 78:379–389
Camargo-Sanabria AA, Mendoza E (2016) Interactions between terrestrial mammals and the fruits of two neotropical rainforest tree species. Acta Oecologica 73:45–52
Capaverde UD Jr, Pereira LG, do A, Tavares V da C, et al (2018) Subtle changes in elevation shift bat-assemblage structure in Central Amazonia. Biotropica 50:674–683
Costa FRC, Magnusson WE (2010) The need for large-scale, integrated studies of biodiversity–the experience of the Program for Biodiversity Research in Brazilian Amazonia. Nat Conserv 8:3–12
Crooks KR, Burdett CL, Theobald DM et al (2017) Quantification of habitat fragmentation reveals extinction risk in terrestrial mammals. Proc Natl Acad Sci 114:7635–7640
Cruz P, Paviolo A, Bó RF et al (2014) Daily activity patterns and habitat use of the lowland tapir (Tapirus terrestris) in the Atlantic Forest. Mamm Biol 79:376–383
da Rosa CA, Bager A (2012) Seasonality and habitat types affect roadkill of neotropical birds. J Environ Manage 97:1–5
da Silva JMC, Rylands AB, Da Fonseca GAB (2005) The fate of the Amazonian areas of endemism. Conserv Biol 19:689–694
da Silveira Anacleto TC (2007) Food habits of four armadillo species in the Cerrado area, Mato Grosso. Brazil Zool Stud 46:529
Depalma DM, Mermoz ME (2019) Ground nesting birds in roadside borders of the Argentine Pampas: habitat use and predation risk of artificial nests. Rev Bras Ornitol 27:261–274
Desbiez ALJ, Santos SA, Keuroghlian A, Bodmer RE (2009) Niche partitioning among white-lipped peccaries (Tayassu pecari), collared peccaries (Pecari tajacu), and feral pigs (Sus scrofa). J Mammal 90:119–128
Dillon A, Kelly MJ (2008) Ocelot home range, overlap and density: comparing radio telemetry with camera trapping. J Zool 275:391–398
Dutra HP, Barnett K, Reinhardt JR et al (2011) Invasive plant species alters consumer behavior by providing refuge from predation. Oecologia 166:649–657
Estrada A, Rivera A, Coates-Estrada R (2002) Predation of artificial nests in a fragmented landscape in the tropical region of Los Tuxtlas, Mexico. Biol Conserv 106:199–209
Fadini RF, Brocardo CR, Rosa C et al (2021) Long-term standardized ecological research in an Amazonian savanna: a laboratory under threat. An Acad Bras Cienc 93
Fearnside PM (2007) Brazil’s Cuiabá-Santarém (BR-163) highway: the environmental cost of paving a soybean corridor through the Amazon. Environ Manage 39:601
Foerster CR, Vaughan C (2002) Home range, habitat use, and activity of Baird’s tapir in Costa Rica. Biotropica 34:423–437
Galetti M, Bovendorp RS, Guevara R (2015) Defaunation of large mammals leads to an increase in seed predation in the Atlantic forests. Glob Ecol Conserv 3:824–830. https://doi.org/10.1016/j.gecco.2015.04.008
Galetti M, Brocardo CR, Begotti RA et al (2017) Defaunation and biomass collapse of mammals in the largest Atlantic forest remnant. Anim Conserv 20:270–281. https://doi.org/10.1111/acv.12311
Galetti M, Giacomini HC, Bueno RS et al (2009) Priority areas for the conservation of Atlantic forest large mammals. Biol Conserv 142:1229–1241. https://doi.org/10.1016/J.BIOCON.2009.01.023
Garda AA, Wiederhecker HC, Gainsbury AM et al (2013) Microhabitat variation explains local-scale distribution of terrestrial Amazonian lizards in Rondônia, Western Brazil. Biotropica 45:245–252
Garrett RD, Lambin EF, Naylor RL (2013) The new economic geography of land use change: Supply chain configurations and land use in the Brazilian Amazon. Land Use Policy 34:265–275
Gayot M, Henry O, Dubost G, Sabatier D (2004) Comparative diet of the two forest cervids of the genus Mazama in French Guiana. J Trop Ecol 31–43
Gorini L, Linnell JDC, May R et al (2012) Habitat heterogeneity and mammalian predator–prey interactions. Mamm Rev 42:55–77
Goulart FVB, Cáceres NC, Graipel ME et al (2009) Habitat selection by large mammals in a southern Brazilian Atlantic Forest. Mamm Biol 74:182–190
Hartig F, Lohse L (2020) Package DHARMa. https://cran.r-project.org/web/packages/DHARMa/DHARMa.pdf. Accessed 1 Feb 2022
Harveson PM, Tewes ME, Anderson GL, Laack LL (2004) Habitat use by ocelots in south Texas: implications for restoration. Wildl Soc Bull 32:948–954
Haugaasen JMT, Haugaasen T, Peres CA et al (2010) Seed dispersal of the Brazil nut tree ( Bertholletia excelsa) by scatter-hoarding rodents in a central Amazonian forest. J Trop Ecol 26:251–262. https://doi.org/10.1017/S0266467410000027
Haugaasen T, Peres CA (2007) Vertebrate responses to fruit production in Amazonian flooded and unflooded forests. Biodivers Conserv 16:4165
Hawes JE, Peres CA (2014) Fruit-frugivore interactions in Amazonian seasonally flooded and unflooded forests. J Trop Ecol 381–399
Hawkins BA, Diniz-Filho JAF, Jaramillo CA, Soeller SA (2007) Climate, niche conservatism, and the global bird diversity gradient. Am Nat 170:S16–S27
Hong M, Yuan S, Yang Z et al (2015) Comparison of microhabitat selection and trace abundance of giant pandas between primary and secondary forests in Liziping Nature Reserve, China: effects of selective logging. Mamm Biol 80:373–379
IBGE (1957) Enciclopédia dos municípios brasileiros -, vol XIV. IBGE, Rio de Janeiro
ICMBio (2019) Instituto Chico Mendes de Conservação da Biodiversidade. Plano de Manejo Floresta Nacional do Tapajós. Brasília
Jansen PA, Ahumada J, Fegraus E, O’Brien T (2014) TEAM: a standardised camera trap survey to monitor terrestrial vertebrate communities in tropical forests. In: Meek PD, Fleming P, Ballard G et al (eds) Camera trapping: wildlife research and management, 1st edn. Csiro Publishing, Melbourne, pp 263–270
Jerozolimski A, Peres CA (2003) Bringing home the biggest bacon: a cross-site analysis of the structure of hunter-kill profiles in Neotropical forests. Biol Conserv 111:415–425. https://doi.org/10.1016/S0006-3207(02)00310-5
Jordano P (2000) Fruits and frugivory. In: Fenner M (ed) Seeds: the ecology of regeneration in plant communities. CABI Books, CABI International., pp 125–166
Jorge MLSP, Ribeiro MC, Ferraz KMPMB (2013) Mammal defaunation as surrogate of trophic cascades in a biodiversity hotspot. Biol Conserv 163:49–57. https://doi.org/10.1016/j.biocon.2013.04.018
Kasper CB, Schneider A, Oliveira TG (2016) Home range and density of three sympatric felids in the Southern Atlantic Forest, Brazil. Brazilian J Biol 76:228–232
Keuroghlian A, Eaton DP, Longland WS (2004) Area use by white-lipped and collared peccaries (Tayassu pecari and Tayassu tajacu) in a tropical forest fragment. Biol Conserv 120:411–425. https://doi.org/10.1016/j.biocon.2004.03.016
Kinnaird MF, Sanderson EW, O’Brien TG et al (2003) Deforestation trends in a tropical landscape and implications for endangered large mammals. Conserv Biol 17:245–257
Kutt AS, Gordon IJ (2012) Variation in terrestrial mammal abundance on pastoral and conservation land tenures in north-eastern A ustralian tropical savannas. Anim Conserv 15:416–425
Levey DJ (1988) Tropical wet forest treefall gaps and distributions of understory birds and plants. Ecology 69:1076–1089
Lüdecke D, Ben-Shachar MS, Patil I et al (2021) Performance: an R package for assessment, comparison and testing of statistical models. J Open Source Softw 6:3139
Maezumi SY, Alves D, Robinson M et al (2018) The legacy of 4,500 years of polyculture agroforestry in the eastern Amazon. Nat Plants 4:540–547
Magnusson WE, Braga-Neto R, Pezzini F, et al (2013) Biodiversidade e Monitoramento Ambiental Integrado: O Sistema RAPELD na Amazônia. Attema, Santo André
Magnusson WE, Lima AP, Luizão R et al (2005) RAPELD: a modification of the Gentry method for biodiversity surveys. Biota Neotrop 5:19–24
Mamede SB, Alho CJR (2006) Response of wild mammals to seasonal shrinking-and-expansion of habitats due to flooding regime of the Pantanal, Brazil. Brazilian J Biol 66:991–998
Maximiano MF de A, d’Horta FM, Tuomisto H, et al (2020) The relative role of rivers, environmental heterogeneity and species traits in driving compositional changes in southeastern Amazonian bird assemblages. Biotropica
Michalski LJ, Norris D, de Oliveira TG, Michalski F (2015) Ecological relationships of meso-scale distribution in 25 neotropical vertebrate species. PLoS One 10:e0126114. https://doi.org/10.1371/journal.pone.0126114
Morais TA, da Rosa CA, de Azevedo CS et al (2019) Factors affecting space use by wild boars (Sus scrofa) in high-elevation tropical forests. Can J Zool 97:971–978
Nasi R, Taber A, Van VN (2011) Empty forests, empty stomachs? Bushmeat and livelihoods in the Congo and Amazon Basins. Int for Rev 13:355–368
Newsome TM, Greenville AC, Ćirović D et al (2017) Top predators constrain mesopredator distributions. Nat Commun 8:1–7
O’Bryan CJ, Braczkowski AR, Beyer HL et al (2018) The contribution of predators and scavengers to human well-being. Nat Ecol Evol 2:229–236
Oksanen J, Guillaume BF, Michael F et al (2019) Vegan: community ecology package. https://cran.r-project.org/web/packages/vegan/vegan.pdf. Accessed 1 Feb 2022
Palmer MS, Swanson A, Kosmala M et al (2018) Evaluating relative abundance indices for terrestrial herbivores from large-scale camera trap surveys. Afr J Ecol 56:791–803
Paredes OSL, Norris D, de Oliveira TG, Michalski F (2017) Water availability not fruitfall modulates the dry season distribution of frugivorous terrestrial vertebrates in a lowland Amazon forest. PLoS ONE 12:e0174049
Parsons AW, Forrester T, McShea WJ et al (2017) Do occupancy or detection rates from camera traps reflect deer density? J Mammal 98:1547–1557
Peres CA, Lake IR (2003) Extent of nontimber resource extraction in tropical forests: accessibility to game vertebrates by hunters in the Amazon basin. Conserv Biol 17:521–535
Peres CA, Palacios E (2007) Basin-wide effects of game harvest on vertebrate population densities in Amazonian forests: implications for animal-mediated seed dispersal. Biotropica 39:304–315
Peres CA, Van Roosmalen M (2002) Primate frugivory in two species-rich Neotropical forests: implications for the demography of large-seeded plants in overhunted areas. In: Levey DJ, Silva WR, Galetti M (eds) Seed dispersal and frugivory: ecology, evolution and conservation. Cab International Oxford (United Kingdom), Oxford, pp 407–421
Pizo MA, Oliveira PS (2000) The use of fruits and seeds by ants in the Atlantic Forest of southeast Brazil 1. Biotropica 32:851–861
Prasniewski VM, Szinwelski N, Bertrand AS et al (2022) Brazil’s Iguaçu National Park threatened by illegal activities: predicting consequences of proposed downgrading and road construction. Environ Res Lett
QGIS.org (2021) QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org. Accessed 1 Feb 2022
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Rabelo RM, Aragón S, Bicca-Marques JC (2019) Prey abundance drives habitat occupancy by jaguars in Amazonian floodplain river islands. Acta Oecologica 97:28–33
Reis ML, Raíces DSL, Martins JFV et al (2015) Monitoramento da biodiversidade: Região 4 - Guia de identificação de espécies alvo de aves e mamíferos. GKNORONHA, Brasília
Reis NR, Peracchi AL, Fregonezi MN, Rossaneis BK (2010) Mamíferos do Brasil: Guia de Identificação, 1st edn. Technical Books Editora, Rio de Janeiro
Reyna-Hurtado R, Beck H, Altrichter M et al (2016) What ecological and anthropogenic factors affect group size in white-lipped peccaries (Tayassu pecari)? Biotropica 48:246–254
Ripley B, Venables B, Bates D et al (2021) Package ‘MASS’. https://cran.r-project.org/web/packages/MASS/MASS.pdf. Accessed 1 Feb 2022
Rojas-Ahumada DP, Landeiro VL, Menin M (2012) Role of environmental and spatial processes in structuring anuran communities across a tropical rain forest. Austral Ecol 37:865–873
Rosa C, Baccaro F, Cronemberger C et al (2021a) The program for biodiversity research in Brazil: the role of regional networks for biodiversity knowledge, dissemination, and conservation. An Acad Bras Cienc 93. https://doi.org/10.1590/0001-3765202120201604
Rosa C, Hegel CGZ, Passamani M (2021b) Seed removal of Araucaria angustifolia by native and invasive mammals in protected areas of Atlantic Forest. Biota Neotrop 21
Rosa DP, Brocardo CR, Castro AB et al (2021c) Species-rich but defaunated: the case of medium and large-bodied mammals in a sustainable use protected area in the Amazon. Acta Amaz 54:323–333
Rossi RV, Bianconi GV, Carmignotto AP, Miranda CL (2010) Ordem Didelphimorphia. Technical Books Editora, Rio de Janeiro
Rovero F, Marshall AR (2009) Camera trapping photographic rate as an index of density in forest ungulates. J Appl Ecol 46:1011–1017
Salas LA, Fuller TK (1996) Diet of the lowland tapir (Tapirus terrestris L.) in the Tabaro River valley, southern Venezuela. Can J Zool 74:1444–1451
Sampaio R, Lima AP, Magnusson WE, Peres CA (2010) Long-term persistence of midsized to large-bodied mammals in Amazonian landscapes under varying contexts of forest cover. Biodivers Conserv 19:2421–2439
Sampaio R, Morato RG, Abrahams MI et al (2022) Physical geography trumps legal protection in driving the perceived sustainability of game hunting in Amazonian local communities. J Nat Conserv 67:126175
Scabin AB, Peres CA (2021) Hunting pressure modulates the composition and size structure of terrestrial and arboreal vertebrates in Amazonian forests. Biodivers Conserv 30:3613–3632
Schipper J, Chanson JS, Chiozza F et al (2008) The status of the world’s land and marine mammals: diversity, threat, and knowledge. Science (80- ) 322:225–30. https://doi.org/10.1126/science.1165115
Schupp EW, Jordano P, Gómez JM (2010) Seed dispersal effectiveness revisited: a conceptual review. New Phytol 188:333–353. https://doi.org/10.1111/j.1469-8137.2010.03402.x
Sikes RS, Heidt GA, Elrod DA (1990) Seasonal diets of the nine-banded armadillo (Dasypus novemcinctus) in a northern part of its range. Am Midl Nat 383–389
Silva JV, Barbosa RI, Citó AC (2019) Estimando taxas de decomposição e fator de estabilização da liteira usando o método tbi (tea bag index). Boa Vista
Silveira JM, Barlow J, Louzada J, Moutinho P (2010) Factors affecting the abundance of leaf-litter arthropods in unburned and thrice-burned seasonally-dry Amazonian forests. PLoS ONE 5:e12877
Silvius KM (2002) Spatio-temporal patterns of palm endocarp use by three Amazonian forest mammals: granivory or ‘grubivory’? J Trop Ecol 18:707–723
Silvius KM, Fragoso JMV (2003) Red-rumped Agouti (Dasyprocta leporina) Home range use in an Amazonian forest: implications for the aggregated distribution of forest trees. Biotropica 35:74–83
Sollmann R, Mohamed A, Samejima H, Wilting A (2013) Risky business or simple solution–relative abundance indices from camera-trapping. Biol Conserv 159:405–412
Stenborg P, Schaan DP, Lima AMA (2012) Precolumbian land use and settlement pattern in the Santarém region, lower Amazon. Amaz - Rev Antropol 4:222–250
Szinwelski N, Rosa CS, de Castro Solar RR, Sperber CF (2015) Aggregation of cricket activity in response to resource addition increases local diversity. PLoS ONE 10:e0139669
Tardio BMR, Da Silveira R (2015) The role of forest structure and human occupation in structuring mammal assemblages in oligotrophic ecosystems of C entral A mazonia. Austral Ecol 40:318–330
Torralvo K, Magnusson WE, Lima A, Rosa CA (2020) Dados de estrutura da vegetação obtidos com LIDAR portátil de chão em parcelas permanentes de módulos RAPELD na Floresta Nacional (FLONA) do Tapajós- PA. In: PPBioAmOc.577.2. https://ppbiodata.inpa.gov.br/metacat/metacat/PPBioAmOc.577.2/default
Torres CP, Morsello C, Parry L et al (2018) Landscape correlates of bushmeat consumption and hunting in a post-frontier Amazonian region. Environ Conserv 45:315–323. https://doi.org/10.1017/S0376892917000510
Torres PC, Morsello C, Parry L, Pardini R (2021) Forest cover and social relations are more important than economic factors in driving hunting and bushmeat consumption in post-frontier Amazonia. Biol Conserv 253:108823
Veríssimo A, Rolla A, Vedoveto M, Futada S de M (2011) Áreas Protegidas na Amazônia Brasileira: avanços e desafios. Instituto Socioambiental, São Paulo
Vidal MM, Pires MM, Guimarães PR Jr (2013) Large vertebrates as the missing components of seed-dispersal networks. Biol Conserv 163:42–48
Villar N, Paz C, Zipparro V et al (2020a) Frugivory underpins the nitrogen cycle. Funct Ecol 35:357–368
Villar N, Siqueira T, Zipparro V et al (2020b) The cryptic regulation of diversity by functionally complementary large tropical forest herbivores. J Ecol 108:279–290. https://doi.org/10.1111/1365-2745.13257
Wang B, Rocha DG, Abrahams MI et al (2019) Habitat use of the ocelot (Leopardus pardalis) in Brazilian Amazon. Ecol Evol 9:5049–5062
Wang E (2002) Diets of ocelots (Leopardus pardalis), margays (L. wiedii), and oncillas (L. tigrinus) in the Atlantic rainforest in southeast Brazil. Stud Neotrop Fauna Environ 37:207–212
Wang Y, Zou Y, Henrickson K et al (2017) Google Earth elevation data extraction and accuracy assessment for transportation applications. PLoS ONE 12:e0175756
Weiler A, Núñez K, Silla F (2020) Forest matters: use of water reservoirs by mammal communities in cattle ranch landscapes in the Paraguayan Dry Chaco. Glob Ecol Conserv e01103
Whitaker JO, Ruckdeschel C, Bakken L (2012) Food of the armadillo Dasypus novemcinctus L. from Cumberland Island. GA Southeast Nat 11:487–506
Zuur A, Ieno EN, Walker N et al (2009) Mixed effects models and extensions in ecology with R. Springer Sci Bus Media
Acknowledgements
We thank the Instituto Brasileiro de Conservação da Biodiversidade (ICMBio) for providing permits (SISBIO no. 67787-3). DCPR received a master’s fellowship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). CR received a post-doctoral fellowship from Conselho Nacional para o Desenvolvimento Científico e Tecnológico (CNPq). CRB received a post-doctoral fellowship from CAPES (CAPES, Brazil, Finance Code 001). ABC received a doctoral fellowship from CNPq. The English review of the manuscript was supported by Edital 03/2021/PROPPIT/UFOPA–Programa de Apoio à Produção Científica Qualificada–PAPCIQ/UFOPA. Adrian A Barnett helped with the English language. We thank the two anonymous reviewers for their comments that helped to improve the manuscript.
Funding
This work was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), PROCAD-AM (proc. # 88881.314420/2019-01), and PELD (LTER) POPA by the Conselho Nacional para o Desenvolvimento Científico e Tecnológico (CNPq) (proc. # 441443/2016-8).
Author information
Authors and Affiliations
Contributions
The study conception and design were performed by Rodrigo F. Fadini, Carlos R. Brocardo, Dian P. Rosa, Clarissa Rosa, Arlison C. Bezerra, and William E. Magnusson. Data collection was performed by Dian P. Rosa, Arlison C. Bezerra, Carlos R. Brocardo, Clarissa Rosa, Kelly Torralvo, and Rodrigo F. Fadini. The analyses were performed by Carlos R. Brocardo, Rodrigo F. Fadini, and Pedro Pequeno. The first draft of the manuscript was written by Carlos R. Brocardo and Rodrigo F. Fadini, while all authors commented on subsequent versions. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brocardo, C.R., Rosa, D.C.P., Castro, A.B. et al. Responses of ground-dwelling birds and mammals to local environmental variables and human pressure in an Amazonian protected area. Eur J Wildl Res 69, 48 (2023). https://doi.org/10.1007/s10344-023-01677-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-023-01677-z