Abstract
The main focus of this work was to study the use of freshwater wetlands by 12 waterbirds (4 resident species and 8 Palearctic migrant species) during the wintering period in West Bengal, India. From morning to evening hours, the time budget of waterbirds changed significantly. The differences in diurnal activities between migrants and residents were statistically significant. Foraging niche overlaps were calculated based on foraging habitats and techniques; five migrant species had exceedingly high niche overlaps (habitats 0.80–0.99; techniques 0.79–0.99). Overlap among resident birds or migrant species was much lower (habitats 0.01–0.56; techniques 0.01–0.66). Niche dimensions like the foraging habitats (1.26–3.47) and foraging techniques (1.13–4.54) and these two dimensions together (1.64–8.63) calculated the niche breadths. A mean Euclidean distance of 5.63 in Cluster analysis constructed on two niche dimensions reliably defined four distinct guilds. The post hoc test highlighted significant differences between the intra-guild niche breadths. Proportional differences in the uses of foraging habitats and techniques and temporal variations in foraging activities accommodated residents and winter migrants in the study sites. Such information would be crucially important for the conservation and effective management strategies of Indian waterbirds and their habitats, situated along the East Asian-Australian and Central Asian Flyways, which are significant tropical wintering grounds for migratory waterbirds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat attributes and inter- and intra-specific interactions play crucial roles in structuring communities in a habitat (Pianka 1974). For waterbirds, available foraging resources and foraging areas play a pivotal role in shaping community structure (Weller 1999). Detailed information on habitat quality and community structures would be imperative to formulate conservation strategies, especially for the migratory waterbirds that use highly fractured wintering habitats along the flyways (Hanski and Gilpin 1997). Wetland birds that migrate southwards along Central Asia Flyway (CAF) and East Asian-Australasian Flyway (EAAF) through tropical areas forage and rest at their wintering sites and accumulate sufficient fat reserves before leaving for colder regions of breeding sites at higher latitudes (Li et al. 2009; Dhanjal-Adams et al. 2017; MoEFCC, Government of India 2018). In the wintering months, both resident waterbirds and Palearctic migrants use the wetlands of West Bengal as their foraging or staging grounds (Chatterjee et al. 2020a; Mukherjee et al. 2020). Pérez-Crespo et al. (2013) recorded that the waterbird communities showed a complex structure owed to a decent number of variables that affected both intra and interspecific interactions. Based on resource utilization of the species within a habitat, the community can be divided into distinct ecological units are called “guilds” (Root 1967). Simberloff and Dayan (1991) defined guilds as the “building blocks” of a specific community. These “building blocks” are composed of species that use similar resources (food and nutrition) within a defined physical habitat in a comparable manner. However, differences in use of foraging habitat, feeding techniques, food preferences, and time of foraging play crucial roles in resource partitioning in these birds (Polla et al. 2018). Every year billions of waterbirds migrate to India, along the CAF and EAAF, from their breeding grounds, flying past thousands of kilometers, exhibiting astonishing feats of endurance. These winter guests share the feeding and fattening habitats with resident species. Fernández and Hamilton (2015) reported that a comparison of resource sharing and guild structure for migratory and resident waterbirds during wintering period was important for a better understanding of species’ dynamics in the invaded habitat. However, these long-distance migratory movements are energetically costly and little is being studied on how such costs are compensated at the fattening grounds during winter with niche adjustments with the resident waterbirds. Soriano-Redondo et al. (2020) showed that in migratory flying species, like birds and bats, there was an adaptive allocation of resources for growth and reproduction, apart from survival; staggered utilization of such allocated resources through time reduced the direct mortality costs of migration. Therefore, at the community level, the winter migrant species should maximize their foraging resource utilization to compensate for energy expenses during migratory movements. Eyres et al. (2020) reported that seasonal niche overlap values were overall relatively low and the niche occupied by migrants was never identical between seasons in passerine birds.
Habitat heterogeneity is one of the foremost aspects determining the use of wetlands habitats in a harmonizing way by both resident and migratory waterbirds in good numbers (Perez-Garcia et al. 2014). This present study has been conducted to study the use of Indian freshwater wetlands (of West Bengal, eastern India) by 12 waterbirds species (4 residents and 8 Palearctic migrants) populations during the wintering period (from October through March). The study analyzes species-specific diurnal time-activity budgets and compared behavior between residents and migrants during the wintering period. The present study, therefore, comprehensively comments on the time-activity budget, feeding techniques, habitat use, niche sharing, and guild structure reflecting resource partitioning within and between residents and wintering waterbirds. This study aims to answer three important questions: (1) whether there are any temporal variations in diurnal activities within and between migratory and resident waterbirds or not, (2) whether the resident and migratory bird species are sharing the same feeding guild or not, and (3) whether niche utilization could explain the co-occurrence of resident and migratory waterbirds at the wintering/staging grounds.
Materials and methods
Study areas
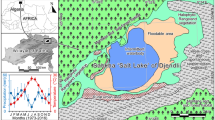
Four study sites are selected that consistently shelter populations of waterbirds in West Bengal (in the districts of Purulia and Bankura), India. Adra Sahebbandh (Site 1; 23°28′N, 86°42′E) is harboring a good number of winter waterbird species; timely, water hyacinth management and proper protection make it suitable for wintering habitat for bird migrants. Purulia Sahebbandh (Site 2; 23°33′N, 86°35′E), the other wetland located in Purulia district, although facing pollution through municipal wastewater inputs and anthropogenic pressures which lead to degradation of habitat quality, shelters a sizable population of waterbirds. Kadamdeuli dam (Site 3; 23°10′N, 86°85′E) and Gangdoa Dam (Site 4; 23°40′N, 87°08′E) are located on Shilabati River and the Shali River, respectively, in Bankura district; these locations are far away from human interference which make these sites suitable wintering grounds for both migratory and resident waterbirds. Figure 1 shows the location map, and further descriptions of the study sites are elaborated in Appendix. We have added the checklist of prevalent aquatic macrophytes, insects, mollusks, and fish collected using standard methods (collection, preservation, and identification) following the comprehensive works of Mandal and Mukherjee (2017), Sao (2016), Mondal and Patra (2015), Roy et al. (2013), and Nandi et al. (2007), from the wetlands under study in Appendix. Importance of the study sites as wintering habitats on EAAF and CAF along with the richness and abundance of waterbirds are already published (Mukherjee et al. 2022).
Waterbird species under study
Twelve prevalent waterbird species were studied; out of these twelve, eight were Palearctic migrants, and four were residents to the eastern part of the Indian subcontinent. Previous studies by Nandi et al. (2004) and Khan et al. (2016) also recorded sizeable populations of these birds to winter in the sites under the present investigation. Identification and nomenclature of avifauna was done following Ali and Ripley (1987), Grimmett et al. (2011), and Kazmierczak and van Perlo (2000). Four-letter alpha codes for the birds’ names were used following Pyle and DeSante (2003). The name of these waterbirds, their alpha codes, and conservation status are given in Table 1.
Field observations on time activity
For recording diurnal time activity of waterbirds, a total period of 64 days (2 days per wetland per month from November through February during the wintering period of migratory waterbirds for the years 2018–2019 and 2019–2020) were invested at 4 study sites. Selected four winter months had sufficiently large (each species density ≥ 30) waterbird populations (including winter migrants) for behavior study. According to Morrison (1984), thirty was the minimum sample size required for analyzing waterbird behavior. Later, Liordos (2010) also suggested that this sample size was mandatory. The duration between 6.00 AM and 5.00 PM of the s sampling day, i.e., 11 h, was divided into four equal parts of 2 h 45 min. The ad libitum binocular observations and video footage recorded the scan sample of the behavior. On each sampling day, the scan sampling data of three durations of 30 min, randomly selected within each of these four sections of 2 h 45 min, were used. Therefore, altogether 64 observations on time activity, spending 192 h, were made, and the mean values in percentages were represented.
Behavioral categories were recorded following Green et al. (1999), and these were feeding (diving and interval between two dives); resting (included sleeping behavior without head-on-back and with head-on-back and also eyes open or closed, loafing); preening (included comfort, bathe, wing-flap, head-shake, wing-shake, stretch); swimming (included searching/scanning, and flying); and others (included alert, intra- and interspecific interactions social interaction).
Only the first/initial observation on each individual of waterbird was considered (following the initial observation method described by Liordos 2010). Time-activity was quantified by the scan-sample approach (Martin and Bateson 1993; Losito et al. 1989). Ali et al. (2016) also suggested using instantaneous or scan sampling for studying the behavior of individuals in groups. Behavior of every individual in a group was documented at fixed time intervals (30 s) during the scan sampling following Altman (1974) during the selected diurnal hours. Observations were made from two favored (because of their accessibility and unimpeded yet secretive view) vantage points at each site, using Nikon Fieldscope (25–75 × 82 ED), Olympus (8 × 40 DPS I) binoculars, and Sony RX10 IV camera.
Water depth and foraging habitat
Foraging resources (vegetation) were mapped during August–September, ahead of wintering period of migratory waterbirds, to avoid habitat disturbance. We presumed that within the study period, there would not be considerable differences in the distribution of foraging resources in the concerned habitats. We followed the methodology, as elaborated by Chatterjee et al. (2020b), for mapping of foraging resources at the study sites. Foraging habitats of these waterbirds (mainly divers) were characterized and categorized conferring to water depth and types of associated vegetation. Average water depth was measured from one shore of the wetland to the other shore at an interval of 1 m along two transects (one along the width and the other one along the length of the wetland; the distance between adjacent transect lines was 2 m, both for width-wise and length-wise lines). Weight and a string marked in a meter were dipped from boat to measure the depth of waterbodies. Mean of these recordings depicted the average depth of a foraging site. Portions of water bodies with < 1.5 m water depth characterized as shallow water regions, whereas deep water regions represented by water level > 1.5 m (Gopal and Sah 1995). As the wintering period under study did not experience any heavy rain, it was presumed that the measured depth did not differ during the field observations.
Works of Liordos (2010) and Peŕez-Crespo et al. (2013) were followed for the categorization of the foraging habitats based on the water depth and type of associated vegetation. Five major foraging habitats for both resident and migratory waterbirds were identified in four study sites.
-
1.
Deep water with floating vegetation (DWFV): This was the habitat that had deep water (i.e., > 1.5 m depth) covered by floating vegetation.
-
2.
Deep water with a clear surface (DWCS): This was the area of wetland having deep water (i.e., > 1.5 m depth) without any emergent or floating vegetation. This area consisted of the deepest portion of the water bodies.
-
3.
Shallow water with floating vegetation (SWFV): The depth of this habitat was < 1.5 m; it had copious floating vegetation predominantly consisted of water hyacinth.
-
4.
Shallow water with a clear surface (SWCS): The depth of this habitat is < 1.5 m. This area was devoid of any emergent and floating vegetation.
-
5.
Shoreline with hydrophytic vegetation (SLHV): This habitat consisted of predominantly mudflats at the shoreline with vegetation.
Foraging techniques employed by the waterbirds
Feeding behaviours were categorized by the foraging techniques employed by these waterbirds following the works by Liordos (2010) and Peŕez-Crespo et al. (2013). Eight major foraging behaviours were identified:
-
1.
Diving (DI): The bird temporarily vanishes underwater to forage
-
2.
Upending or Tipping (UP): It is a feeding technique where the bird moderately dips in a vertical position while feeding; however, its tail and legs remain above the water surface.
-
3.
Beak-dip (BD): In this technique birds mainly feed by dipping its beak in to the water. Dipping of beak can be either partly or in full.
-
4.
Head-dip (HD): This technique involves dipping the head including the beak in water for foraging. In this case, the eyes too are submerged while foraging.
-
5.
Neck-dip (ND): This foraging technique uses dipping up to the neck under water level, fully or partially at the foraging site.
-
6.
Filtering or gleaning (FI): In this technique, the bird held its beak in plane of water surface, while submerging only the mandible for straining the food particles from the water surface.
-
7.
Picking (PI): This foraging technique is characterized by picking the food substances mainly from the top of the muddy shorelines.
-
8.
Grazing (GR): When birds eat floating or marginal macrophytes.
Niche dimension and guild structure
Two separate days, for two predetermined time durations (one from 7 to 10 am, and other from 2 to 5 pm) were invested to specially record the foraging behavior of the species from the selected vantage points at the study sites covering each habitat type. However, during the study on diurnal time activities, the foraging behavior of waterbirds was also carefully and critically recorded. Foraging habitats and foraging techniques were recorded for a specific waterbird in consultation with the work of Ali and Ripley (1968). Not less than 30 foraging observations for each of the 12 waterbird species were used for the present study. Data were arranged into three matrices: (i) feeding habitat (12 species × 5 habitat variable), (ii) feeding technique (12 species × 8 feeding technique), and (iii) combined foraging habitat and foraging technique (12 species × 40 possible combinations for both the dimensions). Out of 40 possible combinations, 25 combinations were feasible rejecting combinations that were not practical (like shoreline with hydrophytic vegetation and diving). Both niche breadth and niche overlap were tabulated following these matrices, and these matrices also used for assigning foraging guilds (Liordos 2010; Chatterjee et al. 2020b).
Niche breadth and niche overlap calculations
The one-dimensional matrices of both niche dimensions were used for calculating the foraging niche breadth and niche overlap. The foraging niche breadth of a species was calculated using the formula derived by Levins (1968):
where Pi was the proportion of observation in individual category (i) within a specific niche dimension (i.e., habitat and technique), i.e., Pi is the proportion of exploitation of a particular resource by the species under study in a specific niche dimension.
Foraging niche overlap (o) between every pair of waterbird species and within each niche dimension was calculated by means of the index of Pianka (1974):
where Pij was the proportional values of exploitation of resource i by species j and k, respectively (i.e., jth and kth species). This index was ranged from values between 0 and 1. The value of 1 signified complete niche overlap between the species pair under contemplation and 0 denoted complete isolation.
Assigning guild for the waterbirds in the community
For assigning a guild to 12 waterbirds selected for the study cluster analysis was used in the present study, considering three original matrices (for foraging habitats, foraging techniques, and bi-dimensional). These matrices were subjected to arcsine transformation, as recommended by Fowler and Cohen (1990), where y = arcsine × 0.5 which was used to represent proportion and to decrease Kurtosis of the distribution of a variable (Peŕez-Crespo et al. 2013).
Statistical analyses
Hierarchical cluster analysis, using an unweighted pair group method (UPGM), assigned guilds of waterbirds based on the niche dimensions, foraging habitats, and foraging techniques. Post hoc with Tukey’s honestly significant difference (Tukey HSD) test (level of significance at p < 0.05) compared the differences in niche overlap between all the guilds to highlight the significant differences between the guilds (Winer 1971). One-way analysis of variance (ANOVA) at a significance level of p ≤ 0.01 and calculated partial ŋ2 for effect size determined whether different activities of waterbirds did significantly vary during diurnal hours or not (Kabasakal et al. 2017). We used STATISTICA 8.0, PAST 4.03 softwares for statistical analyses.
Results
The diurnal time-activity budget of eight Palearctic migrants and four resident birds were analyzed and five major diurnal activities: resting (included sleeping behavior without head-on-back and with head-on-back and eyes open or closed, loafing), swimming (included searching/scanning, and flying), feeding (dabbling, diving and interval between two dives); preening (included comfort, bathe, wing-flap, wing-shake, head-shake, stretch), and others (included alert, intra- and interspecific interactions social interaction) were recorded and represented in Fig. 2. In diurnal hours, the maximum percentage of time was allotted to resting (ranging from 39.9 to 70.0%) by most of the migratory waterbirds except GCGR. In GCGR, the highest percentage of day-time was allotted to swimming (44.9 ± 10.25%), followed by feeding (26.1 ± 11.60%). Most of the migratory species spent the morning hours (6.00 to 8.45) in resting, while they were engaged in different other activities during midday and afternoon hours. In resident waterbirds, the maximum percentage of diurnal hours was also allotted to swimming (ranging from 39.9 to 49.0%) except LWDU. LWDU spent the maximum percentage of diurnal time in resting (58.4 ± 32.11%). From morning hours through the evening, the time-activity budget of waterbirds changed significantly (one-way ANOVA F = 32.29; p < 0.01; partial ŋ2 = 0.701); further, significant differences in diurnal activities were also noted between migrants and residents as they behaved differently during daytime (one-way ANOVA F = 67.38; p < 0.01; partial ŋ2 = 0.530).
Foraging techniques and foraging habitats used by the waterbirds were represented in Fig. 3. Among eight migratory waterbirds, five birds (RCPO, TUDU, GCGR, COPO, and FEDU) most frequently used DI (on average 82.6%) as a foraging technique mainly from DWCS (on average 58.9%) and SWCS (on average 16.6%). Two resident birds, namely LIGR and EUCO, also used DI as the most prevalent foraging technique (93.0 ± 1.95% and 61.8 ± 2.49%, respectively). However, these two birds mainly used SWCS (79.6%) followed by DWCS (12.3%). NOPI used UP most frequently (52.7 ± 3.29%) followed by BD (13.1 ± 1.56%). Both EUWI and CPGO used HD as primary foraging technique (59.1 ± 4.01% and 37.3 ± 2.98%, respectively), followed by ND (17.9 ± 2.10%) in the case of EUWI while UP (18.9 ± 1.23%) for CPGO. These three birds, namely, NOPI, EUWI, and CPGO, primarily used SWFV (on average 67.5%) as their favored foraging habitat. GADW and LWDU, on th3 other hand, used all the eight foraging techniques. UP (31.4 ± 2.78%) and HD (26.9 ± 3.10%) were the two most frequently used foraging techniques in GADW. However, LWDU primarily used HD (28.0 ± 2.69%) followed by DI (17.2 ± 1.39%). GADW used SWFV (39.1 ± 3.42%) primarily followed by SWCS (24.3 ± 3.21%), while LWDU used SLHV (82.5 ± 5.62%) as main foraging habitat followed by SWFV (8.9 ± 1.94%).
Percentage population of waterbirds using specific foraging habitat a and percentage of time allocated for using different foraging techniques by waterbirds b. (Foraging habitats: DWFV, deep water with floating vegetation; DWCS, deep water with clear surface; SWFV, shallow water with floating vegetation; SWCS, shallow water with clear surface; SLHV, shoreline with hydrophytic vegetation) (Foraging techniques: DI, diving; UP, upending; HD, head-dip; BD, beak-dip; ND, neck-dip; FI, filtering; PI, picking; GR, grazing)
Foraging niche overlap was calculated basing on foraging habitats and techniques; exceedingly higher niche overlaps (0.80–0.99) for foraging habitats were observed in between five migrant species, namely, RCPO, TUDU, COPO, FEDU, and GCGR (Table 2). Higher foraging habitat overlap was also recorded in between the remaining three migrant species, namely, GADW, EUWI, and NOPI. A resident species, CPGO, also showed high foraging habitat overlap with three migrant species, namely, GADW, EUWI, and NOPI. Overlaps in between other resident birds or with migrant species were much less (0.01–0.56). Niche overlaps for foraging techniques were somewhat different; higher niche overlaps (0.79–0.99) for foraging techniques in between five migrant species, namely, RCPO, TUDU, COPO, FEDU, and GCGR, were, however, similar as observed for foraging habitats (Table 2). Similar high overlaps for foraging techniques, as for habitats, were also recorded in between GADW, EUWI, and NOPI. However, two resident species, namely EUCO and LIGR, showed exceedingly high (0.94–1.00) overlap values with RCPO, TUDU, GCGR, COPO, and FEDU. Another two resident species, CPGO and LWDU, showed similar high overlap with EUWI. High overlaps in foraging technique in between two resident species pairs, namely, EUCO–CPGO, and LWDU–CPGO were recorded. Foraging technique overlaps in between other resident birds or with migrant species were much less (0.01–0.66).
Foraging niche breadth based on feeding habitats (varied from 1.26–3.47) exhibited that GADW had the highest niche breadth followed by EUWI, NOPI, FEDU, and COPO (Table 3). However, the lowest value of the same was noted for CPGO followed by LIGR and EUCO. Foraging niche breadth constructed on feeding techniques (varied from 1.13 to 4.54) had different results; LWDU showed the highest niche breadth based on foraging techniques, followed by GADW, CPGO, and NOPI. We recorded comparable higher values for EUWI, EUCO, and RCPO. Narrower niche breadths, based on feeding techniques, were detected for GCGR, LIGR, TUDU, and FEDU. Considering both feeding dimensions, GADW showed the widest niche breadth (8.63) followed by LWDU (8.12), whereas the narrowest niche breadth showed by GCGR (1.64) followed by LIGR (1.69).
Cluster analysis based on foraging habitats and foraging techniques (i.e., bi-dimensional niche dimensions) projected that the mean Euclidean distance of 5.63 steadfastly defined four waterbird species clusters (Fig. 4). Thereby, the waterbirds were categorized in four separate guilds, namely, generalist dabblers (Guild 1), shallow water dabblers (Guild 2), generalist divers (Guild 3), and clear surface divers (Guild 4). Guild 1, which was categorized by a wide niche breadth considering both foraging dimensions, represented by two waterbirds, namely, GADW and LWDU. Guild 1 and Guild 2 contained three species (EUWI, CPGO, and NOPI) that showed the next higher bi-dimensional foraging niche breadth. Predominantly diver species, like COPO, FEDU, TUDU, RCPO, and EUCO, that occupied Guild 3 had further less niche breadth, while the occupants of the Guild 4, namely, LIGR and GCGR, had the minimum breadth and hence were considered as clear surface divers. Guild 1 showed highest mean niche breadths based on feeding habitat, while Guild 4 showed the narrowest breadth for the same. The post hoc analysis with Tukey HSD test on four guilds attested the significant differences between the intra-guild niche breadths constructed on both the dimensions. Niche breadth of Guild 1 (GADW and LWDU) was significantly different from the other three guilds: Guild 2 (EUWI, CPGO, and NOPI), Guild 3 (COPO, FEDU, TUDU, RCPO, and EUCO), and Guild 4 (LIGR and GCGR) (2.245, 3.128, and 3.935 respectively at p < 0.05). However, niche breadth Guild 2 was significantly different from Guild 1, and Guild 4 (− 2.245 and 1.690, respectively, at p < 0.05), and the niche breadth of Guild 3 was significantly different from only Guild 1 (− 3.128 at p < 0.05).
Discussion
Time activity budgets together with habitat study were expedient in outlining suitable conservation strategies (Das et al. 2011). The quantum of time apportioned for different activities is critically important to understand the demands for specific resources and also to identify the challenges the waterbirds may face in wintering habitats. Foraging habitat requirements and/or foraging techniques vary between waterbird species, more specifically between different foraging guilds. Different factors, such as the physical conditions of the individuals of the species, community organization, foraging resources, and environmental conditions are the major determinants of time-activity budgets (Paulus 1988).
In the present study, the time-activity budget and the niche dimensions of waterbirds were analyzed to emphasize the resource partitioning among migratory and resident birds. A detailed account of the diurnal time activity budget explored the temporal aspect of resource partitioning between the waterbird species. Previous studies by Liordos (2010) and Chatterjee et al. (2020b) emphasized the utilization of resources from viable foraging habitats and the employment of different foraging techniques determined the resource partitioning. Resting, locomotion and feeding were the dominant diurnal activities of ducks; these three activities together constituted over 93% of all activity (Green et al. 1999). Our result confirmed this report. Our study recorded that all waterbirds spent much of their daytime in three major activities, namely resting, swimming, and feeding. Interestingly, either resting or swimming dominated the diurnal activities while time invested in active foraging was never higher than those two activities. Diurnal time-activity budget can be ambiguous (Ali et al. 2016) as habitat use can be very different at night (Green 1998); however, knowledge about behavioral requirements and habitat use in daytime could aid in proper management of the wintering habitats. In the present study, we recorded that the percentage of daytime invested in feeding during winter months was lower than the other two major activities (resting and swimming), and results were consistent with previous researches (Paulus 1988; Thomson et al. 1988; Ali et al. 2016). Thomson et al. (1988) recorded that resting, swimming and preening comprised 50–99% of the cumulative time budget for all wintering waterfowls under study. However, feeding by geese and dabbling ducks comprised less than 10% of the time budget. Paulus (1988) also published a time-activity budget for nonbreeding Anatids. He recorded that for many Anatinae, time spent on feeding usually decreased when ambient temperature declined. He also recorded that many Anatidae spend much of their time resting diurnally and feeding and resting at night. Ali et al. (2016) also recorded that several wintering ducks mostly foraged at night. Such predominant nocturnal feeding habit of wintering waterbirds could lead to lower feeding time in the diurnal hours.
Resting was the most dominant daytime activity in nine out of the twelve waterbirds studied. Our study corroborated with several previous reports. Khan et al. (1998) studied the diurnal activity budget of wintering COPO in Turkey and commented that this species spent most of the time (> 75%) resting, probably due to warmer temp. Muzaffar (2004) quantified diurnal time-activity budgets for wintering FEDU and reported that individuals of this species allotted the highest time for resting (60%) in winter months. Feeding (17%), preening (14%), and swimming (9%) were less prevalent diurnal activities. Ali et al. (2016) reported sleeping as the major diurnal activity of FEDU, COPO, and TUDU. However, as reported by Saker et al. (2016) and Ali et al. (2016) feeding is the dominant daytime activity of wintering EUWI and EUCO, GADW, and EUWI. Higher foraging than other daytime activities was possibly due to the accessibility of varied foraging habitats and copious food items throughout the wintering season. Contrary to these reports, we recorded resting followed by swimming were the major daytime activities of EUCO, GADW, and EUWI. Diurnal resting and other comfort activities in Anatidae represented one of the best ways to preserve energy because of migratory preparedness for wintering populations (Green et al. 1999; Draidi et al. 2019).
We recorded that swimming was the dominant activity of GCGR, LIGR, and CPGO, while for the rest nine waterbirds (namely, RCPO, FEDU, TUDU, GADW, EUWI, EUCO, COPO, NOPI, and LWDU), swimming was the second most important diurnal activity. Such differences were well depicted in the cluster analysis. On average the birds in our study spent 31% swimming and 16% feeding in their diurnal activities. In conformity with our findings, Abdellioui et al. (2015) also reported that for both GCGR and LIGR, swimming was the main diurnal activity in winter. Mason et al. (2013) reported that GADW invested nearly 50% of their daytime activities in swimming. However, Paulus (1984) recorded that GADW spent 64% in their daytime feeding and spent more time resting during the night than day. Time allotted for searching for food corresponded to one-fourth of the total diurnal hours of the wintering FEDU (Draidi et al. 2019). Kaminski and Prince (1981) recorded that foraging and swimming were negatively correlated in dabblers and that response might have been linked to nutrient constraints. However, no difference in diurnal time activity budget between dabblers and divers was significant. Interestingly, migrant and resident waterbird species in this study showed significant differences in diurnal time activity budget. Swimming, rather the locomotion, was an indispensable activity for waterbirds as it was associated with searching for food and suitable foraging and roosting habitats, often rewarded with small snacks. Wintering waterbirds, especially the duck species, spent long diurnal hours loafing, sleeping, and executing rudimentary maintenance and comfort activities (Johnson et al. 2016). Under diverse weather conditions and at different diel hours, waterbirds can capitalize on their energy savings by moving amid various loafing and roosting sites. Karasov (1990) claimed that birds can adaptively control the efficiency of food utilization and thereby undergo pre-migratory fattening without increases in energy intake or decreases in energy expenditure. Waterbirds using foraging habitats regularly adjusted their activity and foraging strategies to meet energy demands while splitting foraging effort among habitat types (Daniels et al. 2019).
GCGR, a migrant species, and two residents, namely LIGR and CPGO, had feeding activity as the second-highest daytime activity in our study. CPGO, the tropical resident species fed mostly in the first and last hours of the daylight (Upadhyay and Saikia 2010); however, we observed a more or less similar trend, and CPGO in this study were largely busy in foraging throughout the diurnal hours. Fox (1994) studied the feeding ecology of wintering LIGR and recorded that LIGR foraged throughout the daylight hours. He recorded a notable decline in the time allotted for resting in the daytime during the winter months with comparatively shorter day lengths and also suggested that LIGR needed to forage for 7–9 h per day during the winter months. Our study amply supported the records of Fox (1994). In our study, both LIGR and GCGR were engaged in feeding almost throughout the diurnal period. Gagliardi et al. (2006) reported that the distribution of feeding efforts of LIGR and GCGR, two predominantly clear surface divers, was correlated to augmenting foraging success in the milieu of energetic costs experienced in diving for food. Ali and Ripley (1987) mentioned that FEDU and LWDU were preferably nocturnal foragers; the present study recorded higher feeding activity of these two species during the late hours of the day. The diurnal feeding maxima at the end of the day, i.e., from the afternoon onwards, was probably the commencement of the higher nocturnal foraging activity that compensated the increase of energy needs spent in thermoregulation (Draidi et al. 2019). Feeding was recorded as the third important diurnal activity for seven out of eight winter migrants in this study. Foraging in diving species like FEDU, TUDU, COPO, and RCPO and dabbling species like EUWI, GADW, and NOPI took third place in the total budget of diurnal activities. Numerous factors like types of food, temperature, and both inter- and intraspecific competitions determine the importance of diurnal foraging (De Leeuw et al. 1999). Feeding rates in tropical areas were lower than the colder northern latitudes (Draidi et al. 2019) which could be explained by upper energy needs for colder temperature.
Stopover areas during the wintering period were considered significant habitats for migratory waterbirds. From an ecological perspective, detailed research on the importance of specific stopover grounds was deficient (Beatty et al. 2013). Wetland degradation or changes in land use patterns might lead to a transformation in waterbird habitat selection and food resource choice (Begam et al. 2021). Time-activity budgets described dominance relationships, foraging strategies, and responses of wintering waterbirds to the environment and habitat conditions (Bergan et al. 1989). However, a few studies have examined behavioral strategies of wintering coexisting species of resident and migrant waterbirds. Besides, the behavioral ecology of wintering diving and dabbling ducks has received minimal attention. We extended our observations on diurnal feeding time-activity budgets of both resident and migratory waterbirds to examine potential interspecific differences in niche sharing and guild structure. The richness of foraging resources and accessibility to that wealth often greatly influenced habitat use by waterbirds (Bolduc and Afton 2004). Intrinsic (neck length, tarsus length, and body size, etc.) and extrinsic factors (depth of a wetland, aquatic macrophyte density, etc.) of food accessibility varied among waterbird species and groups, and particular species of waterbirds largely fed in particular waterbody with features that optimized the copiousness and accessibility of their foods (Taft and Haig 2003). Waterbirds were assigned into a specific guild primarily depending on several dimensions like the type of foraging habitat used by the species, techniques used for foraging, types of food items consumed, time of day allocated for maximum foraging activity, and depth of water frequently used for foraging (MacNally 1994). Resource partitioning in the guilds could depend on other niche dimensions like prey size and depth of water body (Pearse et al. 2012). Thus, the compositions of waterbird guilds differed among various study sites and were interrelated with mainly physical habitat attributes and foraging techniques in resource exploitation (Liordos 2010; Peŕez-Crespo et al. 2013; Pöysä and Väänänen 2014; Pöysä et al. 2016; Chatterjee et al. 2020b). Guillemain and Fritz (2002) also reported that foraging resources were commonly dispersed heterogeneously on both temporal and spatial scales and foraging waterbirds showed supple responses to this heterogeneity.
We recorded that diverse foraging habitats were occupied by diverse species with different feeding techniques to exploit resources differently in a particular microhabitat. Niche breadth and niche overlap of species in this study emphasized that waterbirds were specialists in foraging techniques while plastic, so far as feeding habitats were concerned. In the present study, the waterbird species of the same guild showed a higher niche overlap than other waterbirds in the community. Niche overlap was the situation in which co-occurring species shared their niche space. High niche overlap might lead to conflicting interactions (such as competition and exclusion) for some species (Pascual-Rico et al. 2020). However, higher niche overlap between co-occurring species in a guild indicated strong interactions in one niche dimension, with coexistence permitted by only weak interactions in others (Cloyed and Eason 2017; Tsafack et al. 2021). Higher niche overlaps of the waterbirds in a guild in the present study would suggest the absence of strong competition. Pérez-Crespo et al. (2013) reported that niche overlaps of species positioned in the same guild were relatively higher but, at the same time, observed habitat partitioning to evade intense competition. The greater degree of specialization within waterbird groups was the primary reason for the small niche overlap between species of several guilds. Liordos (2010), (Liordos and Kontsiotis 2020) reported a comparable outline of niche overlap among waterbirds placed in different foraging guilds. Processes like morphological variances, diverse abundances of species, resource vacillations, migration, and clumped resources could elucidate the observed patterns (Pérez-Crespo et al. 2013). Niche breadths (uni- and bi-dimensional) evaluated the resemblance in resource exploitation among waterbird species. The bi-dimensional niche breadth structured the twelve waterbird species under study into four foraging guilds. Pöysä (1983) commented that changes in resource availability in the case of narrow niches could affect the composition of the community and the reverse was true for the wider niches. Guild 1 (GADW and LWDU) having members with exceedingly high niche breadth in both dimensions were generalist dabblers and, thereby, would be less affected by changes in resource availability. Contrarily, Guild 4 (GCGR and LIGR) was formed by two clear surface divers who showed the narrowest niche breadth and thereby any change in habitat, especially the water depth or food resources at the depth would adversely affect the abundance of these waterbirds. Ali et al. (2016) stated that members of the guild with a specialized single feeding technique considered specialists. Both GCGR and LIGR used more than 90% of foraging techniques in diving. We observed that the specialist guild members showed a notably lower value of bi-dimensional niche breadth (mean value 1.11), whereas the generalist guild associates displayed much greater values of niche breadths that ranged from 2.56 to 8.63. Guild 2 (EUWI, CPGO, and NOPI) and Guild 3 (COPO, FEDU, TUDU, RCPO, and EUCO) had intermediate bi-dimensional niche breadths between breadths those defined Guild 1 and Guild 4. Guild 2 and Guild 3 had broader niche breadths considering both feeding habitats and feeding techniques, therefore, would exhibit less consequence of habitat perturbations. The aptitude of these guild members to consume resources using varied foraging techniques at several microhabitats of the wetlands permitted segregation, thus decreasing probable inter-and intraspecific competitions. Similar situations prevailed in the works of Liordos (2010) and Pérez-Crespo et al. (2013). Interestingly, in our study, we find each of the four foraging guilds had one resident waterbird species along with other migratory species (Guild 1-LWDU, Guild 2-CPGO, Guild 3-EUCO, Guild 4-LIGR). Wiens (1989) reported birds with similar foraging habitats and feeding techniques utilized similar food types and formed guilds. Therefore, both migratory and resident species effectively partitioned foraging resources, and they might have depended on other niche dimensions like prey size, time of foraging, depth of water body, and physical advantages.
Meerhoff et al. (2003) reported that the waterbirds preferred wetlands with diverse foraging resources in different water depths. Varied water depths supported a higher diversity of waterbirds like dabblers, divers, and waders. Our study suggested that feeding habitats, diverse feeding techniques, and temporal variations in foraging activities accommodated resident and winter migrant waterbirds in feeding guilds. Present findings and dataset on time-activity budget and foraging behavior indicate sustainable habitat heterogeneity and foraging resources of wintering sites of eastern India. Therefore, such information would be crucially important for the conservation and effective management strategies of these wetland habitats on EAAF and CAF that shelter residents alongside winter migratory waterbird species during the tropical wintering period at staging and wintering sites.
Conclusion
Initiation of the region-specific acquaintance with communities of waterbirds for effective wetland management is the need of the hour. We need regular monitoring of the richness and abundance of species composition and foraging preferences of the waterbirds, especially in the wetlands that are staging and wintering sites on the EAAF and CAF. Time activity budgets, alongside habitat studies, are instrumental in framing suitable conservation strategies (Das et al. 2011). This work records resource partitioning in waterbird and their guild structures during the wintering period. The time apportioned for different activities is studied to understand the demands for specific resources and the use of habitats in wintering grounds. Foraging habitat requirements and foraging techniques vary between species of the resident and migratory waterbirds or specifically between foraging guilds occupied by these wetland birds. Temporal variations in diurnal activities and the variations in niche utilization, depending on varied foraging resources and foraging techniques, explain the co-occurrence of resident and migratory waterbirds at the wintering/staging grounds.
Availability of data and material
All data produced from this study are provided in this manuscript. However, anything more if needed, the same will be produced.
Code availability
STATISTICA 8.0 software, PAST 4.03 were used.
References
Abdellioui S, Bensouilah T, Houhamdi M (2015) Abundance and diurnal activity budget of sympatric Podicipedidae species at a Ramsar site in north-east Algeria. Zool Ecol. https://doi.org/10.1080/21658005.2015.1074434
Ali E, Ismahan H, Moussa H (2016) Time budget patterns and complementary use of a Mediterranean wetland (Tonga, North-east Algeria) by migrant and resident waterbirds. Riv Ital Ornitol – Res Ornithol 86:19–28
Ali S, Ripley SD (1968) Handbook of the Birds of India and Pakistan together with those of Nepal, Sikkim, Bhutan, Bangladesh and Srilanka. Divers to Hawks (1sted.). Bombay: Oxford University Press
Ali S, Ripley SD (1987) Compact handbook of the Birds of India and Pakistan together with those of Nepal, Sikkim, Bhutan, Bangladesh and Srilanka, 2nd edn. Oxford University Press, Delhi
Altmann J (1974) Observational study of behaviour: sampling methods. Behaviour 49:227–267
Beatty WS, Kesler DC, Webb EB, Raedeke AH, Naylor LW, Humburg DD (2013) Quantitative and qualitative approaches to identifying migration chronology in a continental migrant. PLoS ONE 8(10):e75673. https://doi.org/10.1371/journal.pone.0075673
Begam M, Pal S, Mitra N, Chatterjee A, Mukherjee A, Mukhopadhyay SK (2021) A GIS based approach to determine the changes of water hyacinth (Eichhornia crassipes) cover and its relation with Lesser Whistling Teal (Dendrocygna javanica) assemblage at Santragachi Wetland. West Bengal Res Ecol 3(1):52–58. https://doi.org/10.30564/re.v3i1.2905
Bergan JF, Smith LM, Mayer JJ (1989) Time-activity budgets of diving ducks wintering in South Carolina. J Wildl Manag 53(3):769
Bolduc F, Afton AD (2004) Relationships between wintering waterbirds and invertebrates, sediments, and hydrology of coastal marsh ponds. Waterbirds 27:333–341
Chatterjee A, Adhikari S, Pal S, Mukhopadhyay SK (2020a) Community structure of migratory waterbirds in two important wintering sites at sub-Himalayan forest tract of West Bengal, India. Ring 43. https://doi.org/10.1515/ring-2020a-0002
Chatterjee A, Adhikari S, Pal S, Mukhopadhyay SK (2020b) Foraging guild structure and niche characteristics of waterbirds wintering in selected sub-Himalayan wetlands of India. Ecol Indic 108:105693
Cloyed CS, Eason PK (2017) Niche partitioning and the role of intraspecific niche variation in structuring a guild of generalist anurans. Royal Society Open Science 4(3): 170060. https://doi.org/10.1098/rsos.170060
Daniels BI, Ward DH, Black JM (2019) Activity budgets, daily energy expenditure and energetic model of Black Brant (Branta bernicla nigricans) during winter and spring along the Lower Alaska Peninsula. Wildfowl 69:134–159
Das J, Deka H, Saikia PK (2011) Diurnal activity budgeting of Large Whistling Teal (Dendrocygna bicolor) (Vieillot 1816 (Anseriformes: Anatidae) in Deepor Beel wetlands, Assam. India J Threat Taxa 3(12):2263–2267
De Leeuw JJ, van Eerden MR, Visser GH (1999) Wintering tufted ducks (Aythya fuligula) diving for Zebra mussels Dreissena polymorpha balance feeding costs within narrow margins of their energy budget. J Avian Biol 30(2):182
Dhanjal-Adams KL, Klaassen M, Nicol S, Possingham HP, Chadès I, Fuller RA (2017) Setting conservation priorities for migratory networks under uncertainty. Conserv Biol 31(3):646–656
Draidi K, Bakhouche B, Lahlah N, Djemadi I, Bensouilah M (2019) Diurnal feeding strategies of the ferruginous duck (Aythya nyroca) in Lake Tonga (Northeastern Algeria). Ornis Hung 27(1):85–98
Eyres A, Böhning-Gaese K, Orme CDL, Rahbek C, Fritz SA (2020) A Tale of Two Seasons: the Link between Seasonal Migration and Climatic Niches in Passerine Birds. https://doi.org/10.1002/ece3.6729
Fernández M, Hamilton H (2015) Ecological niche transferability using invasive species as a case study. PLoS ONE 10(3):e0119891. https://doi.org/10.1371/journal.pone.0119891
Fowler J, Cohen L (1990) Practical statistics for field biology (1st ed.). Philadelphia
Fox AD (1994) Estuarine winter-feeding patterns of little grebes (Tachybaptus ruficollis) in central Wales. Bird Stud 41:15–24
Gagliardi A, Martinoli A, Preatoni D, Wauters LA, Tosi G (2006) Behavioural responses of wintering great crested grebes to dissuasion experiments: implications for management. Waterbirds 29:105–114
Gopal B, Sah M (1995) Inventory and Classification of Wetlands in India. Vegetatio 118(1/2):39–48
Green AJ (1998) Habitat selection by the Marbled Teal Marmaronetta angustirostris, ferruginous duck Aythya nyroca and other ducks in the Goksu Delta, Turkey, in summer. Revue D’ecologie (terre Vie) 53:225–243
Green AJ, Fox AD, Hughes B, Hilton GM (1999) Time–activity budgets and site selection of White-headed Ducks (Oxyura leucocephala) at Burdur Lake, Turkey in late winter. Bird Stud 46(1):62–73. https://doi.org/10.1080/00063659909461115
Grimmett R, Inskipp C, Inskipp T, Allen R (2011) Birds of the Indian Subcontinent, 1st edn. Oxford University Press, London
Guillemain M, Fritz H (2002) Temporal variation in feeding tactics: exploring the role of competition and predators in wintering dabbling ducks. Wildl Biol 8(1):81–90
Hanski I, Gilpin M (1997) Metapopulation biology: ecology, genetics and evolution, 1st edn. Academic Press, USA
Johnson F, David C, Humburg D (2016) Learning and adaptation in waterfowl conservation: By chance or by design? Adaptation in Waterfowl Conservation. Wildl Soc Bull 40. https://doi.org/10.1002/wsb.682
Kabasakal B, Poláček M, Aslan A, Hoi H, Erdoğan G, Griggio M (2017) Sexual and non-sexual social preferences in male and female white-eyed bulbuls. Sci Rep. https://doi.org/10.1038/s41598-017-06239-3
Kaminski RM, Prince HH (1981) Dabbling duck activity and foraging responses to aquatic macroinvertebrates. Auk 98:115–126
Karasov WH (1990) Digestion in birds: chemical and physiological determinants and ecological implications. Stud Avian Biol 13:391–415
Kazmierczak K, van Perlo B (2000) A field guide to the birds of India, 1st edn. Om Book Service, India, Delhi
Khan AA, Bilgin C, Kence A, Khan KR (1998) Diurnal activity budget analysis of nonbreeding Pochards (Aythya Ferina) with reference to the local environmental variables at Sarp Lake, Sultan Marshes, Turkey. Pak J Ornithol 2:11–23
Khan TN, Sinha A, Hazra P (2016) Population trends and community composition of migratory waterbirds in three emerging wetlands of global significance in southwestern Bengal. India J Threat Taxa 8(3):8541–8555
Levins R (1968) Evolution in changing environments, 1st edn. Princeton University Press, Princeton, NJ
Li WD, Bloem A, Delany S, Martakis G, Quintero JO (2009) Status of waterbirds in Asia - results of the Asian Waterbird Census: 1987–2007. Wetlands International, Kuala Lumpur, Malaysia
Liordos V (2010) Foraging guilds of waterbirds in a Mediterranean coastal wetland. Zool Stud 49:311–323
Liordos V, Kontsiotis VJ (2020) Identifying important habitats for waterbird conservation at a Greek Regional Nature Park. Avian Res 11:39. https://doi.org/10.1186/s40657-020-00224-7
Losito MP, Mirarchi E, Baldassarre GA (1989) New techniques for time activity studies of avian flocks in view-restricted habitats. J Field Ornithol 60:388–396
MacNally R (1994) Habitat-specific guild structure of forest birds in south-eastern Australia: a regional scale perspective. J Anim Ecol 68:988–1001
Mandal SK, Mukherjee A (2017) Documentation of some rare species of macrophytes associated with wetlands in Purulia district. West Bengal Ind J Sci Res 16(1):73–82
Martin P, Bateson P (1993) Recording methods. In Measuring Behaviour: An introductory guide, 1st edn. Cambridge University Press, Cambridge
Mason C, Whiting R, Conway W (2013) Time-activity budgets of waterfowl wintering on livestock ponds in Northeast Texas. Southeast Nat 12:757–768. https://doi.org/10.1656/058.012.0423
Meerhoff M, Néstor M, Brian M, Lorena RG (2003) The structuring role of free-floating versus submerged plants in a subtropical shallow lake. Aquat Ecol 37(4):377–391
Ministry of Environment, Forests and Climate Change (MoEFCC), Government of India (2018) India’s national action plan for conservation of migratory birds and their habitats along Central Asian Flyway (2018–2023)
Mondal K, Patra A (2015) Ichthyofaunal diversity of Purulia district, W.B. India J Global Biosci 4:2590–2593
Morrison ML (1984) Influences of sample size and sampling design on analysis of avian foraging behaviour. Condor 86:146–150
Mukherjee A, Pal S, Mukhopadhyay SK (2020) Diurnal time-activity budget and foraging techniques of red-crested pochards (Netta rufina) wintering at the wetlands of West Bengal. India Turk J Zool 44(5):424–439
Mukherjee A, Pal S, Adhikari S, Mukhopadhyay SK (2022) Physical habitat attributes influence diversity and turnover of waterbirds wintering at wetlands on Central Asian and East Asian-Australasian Flyways in Eastern India. Wetlands 42:50. https://doi.org/10.1007/s131157-022-01559-1
Muzaffar SB (2004) Diurnal time-activity budgets in wintering ferruginous pochard (Aythya nyroca) in Tanguar Haor, Bangladesh. Forktail 20:17–19
Nandi NC, Bhuinya S, Das SR (2004) Notes on mid-winter waterbird population of some selected wetlands of Bankura and Puruliya districts, West Bengal. Rec Zool Surv Ind 102:47–51
Nandi NC, Venkataram K, Das SR, Das SK (2007) Wetland faunal resources of West Bengal 5. Bankura and Puruliya districts. Rec Zool Surv Ind 107(2):61–91
Pascual-Rico R, Sánchez-Zapata JA, Navarro J, Eguía S, Anadón JD, Botella F (2020) Ecological niche overlap between cooccurring native and exotic ungulates: insights for a conservation conflict. Biol Invasions 22:2497–2508
Paulus SL (1984) Activity budgets of nonbreeding gadwalls in Louisiana. J Wildl Manag 48(2):371–380
Paulus SL (1988) Time-activity budgets of non-breeding Anatidae: a review. In: Weller MW (ed) Waterfowl in Winter. University of Minnesota Press, Minneapolis, pp 135–152
Pearse AT, Kaminski R, Reinecke KJ, Dinsmore S (2012) Local and landscape associations between wintering dabbling ducks and wetland complexes in Mississippi. Wetlands 32:859–869
Pérez-Crespo MJ, Fonseca J, Pineda-López R, Palacios E, Lara C (2013) Foraging guild structure and niche characteristics of waterbirds in an epicontinental lake in Mexico. Zool Stud 52:54–58
Perez-Garcia J, Sebestian-Gonzalez E, Alexander KL, Sanchez-Zapata JA, Botella F (2014) Effect of landscape configuration and habitat quality on the community structure of the waterbirds using a man-made habitat. Eur J Wildl Res 60:875–883. https://doi.org/10.1007/s10344-014-0854-8
Pianka ER (1974) Niche overlap and diffuse competition. Proc Nat Acad Sci 71:2141–2145
Polla WM, Pasquale VD, Rasuk M, Barberis I, Romano M, Manzo RA et al (2018) Diet and feeding selectivity of the Andean Flamingo (Phoenicoparrus andinus) and Chilean Flamingo (Phoenicopterus chilensis) in lowland wintering areas. Wildfowl 68:3–29
Poysa H (1983) Morphology-mediated niche organization in a guild of dabbling ducks. Ornis Scand 14:317–326
Pöysä H, Väänänen VM (2014) Drivers of breeding numbers in a long-distance migrant, the Garganey (Anas querquedula): effects of climate and hunting pressure. J Ornithol 155:679–687
Pöysä H, Rintala J, Johnson DH, Kauppinen J, Lammi E, Nudds TD, Väänänen VM (2016) Environmental variability and population dynamics: do European and North American ducks play by the same rules? Ecol Evol 6:7004–7014
Pyle P, DeSante DF (2003) Four-letter and six-letter alpha codes for birds recorded from the American Ornithologists’ Union Check-list Area. North Am Bird Bander 28(2):64–79
Root RB (1967) The niche exploitation patterns of the blue-grey gnatcatcher. Ecol Monogr 37:317–350
Roy C, Vass K, Patra BC, Sanyal AK (2013) Fish diversity in two south-western districts of West Bengal – Bankura and Purulia. Rec Zool Surv Ind 113(4):167–179
Saker H, Rouaiguia M, Talai-Harbi S, Mouslim B, Bouslama Z, Houhamdi M (2016) Diurnal time budget of the Eurasian Wigeon (Anas penelope) at Lac des Oiseaux (northeast of Algeria). J Entomol Zool Stud 4(4):248–251
Sao SB (2016) Aquatic plant diversity measurement and ecological importance of Saheb Bandh in Purulia district, West Bengal. IOSR J Environ Sci Toxicol Food Technol 10(11):66–69
Simberloff D, Dayan T (1991) The guild concept and the structure of ecological communities. Annu Rev Ecol Evol Syst 22:115–143
Soriano-Redondo A, Gutiérrez JS, Hodgson D et al (2020) Migrant birds and mammals live faster than residents. Nat Commun 11:5719. https://doi.org/10.1038/s41467-020-19256-0
Taft OW, Haig SM (2003) Historical wetlands in Oregon’s Willamette valley: implications for restoration of winter waterbird habitat. Wetlands 23:51–64
Thomson BC, Tabor JE, Turner CL (1988) Diurnal Behavior Patterns of Waterfowl Wintering on the Columbia River, Oregon and Washington. In: Weller MW (ed) Waterfowl in Winter. University of Minnesota Press, Minneapolis, pp 153–167
Tsafack N, Wang X, Xie Y, Fattorini S (2021) Niche overlap and species co-occurrence patterns in carabid communities of the northern Chinese steppes. In: Spence J, Casale A, Assmann T, Liebherr J, Penev L (Eds) Systematic Zoology and biodiversity science: a tribute to Terry Erwin (1940–2020). ZooKeys 1044:929–949. https://doi.org/10.3897/zookeys.1044.62478
Upadhyaya S, Saikia PK (2010) Habitat use and activity budgets in cotton pygmy-goose in Kadamani wetland, Assam (India). NeBIO 1(2):290–294
Weller MW (1999) Wetland birds: habitat resources and conservation implications (1sted.). Cambridge, UK: Cambridge University Press
Wiens JA (1989) Spatial Scaling in Ecology. Funct Ecol 3(4):385–397
Winer BJ (1971) Statistical principles in experimental design, 2nd edn. McGraw-Hill, New York
Acknowledgements
Authors are thankful to Prof. Sanjoy Chakraborty, Principal and Dr. Anjan Biswas of Government College of Engineering and Leather Technology, Kolkata, and Dr. Asitava Chatterjee, DFO, Kangsabati South Division, Purulia, for cooperation and necessary infrastructural and laboratory support. The first author is thankful to the University Grants Commission (UGC), Government of India, for the Junior Research Fellowship [16-6(DEC.2017)/2018(NET/CSIR] that helped in completing this work. The last author is thankful to the UGC for awarding the Emeritus Fellowship (F.6-6/2016- 17/EMERITUS-2015-17-GEN-5244/SA-II).
Funding
AM is thankful to the University Grants Commission (UGC), Government of India, for Junior Research Fellowship [16–6(DEC.2017)/2018(NET/CSIR] that helped in completing this work. SKM is thankful to the UGC for awarding Emeritus Fellowship (F.6–6/2016–17/EMERITUS-2015–17-GEN-5244/SA-II).
Author information
Authors and Affiliations
Contributions
AM: Conceptualization; data curation; writing, original draft preparation. SP: Methodology; software, statistical analysis; writing, reviewing and editing; investigation. PD: Validation, supervision. SKM: Conceptualization, methodology, visualization, investigation, validation, supervision.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
All patients included in this study have given written informed consent to participate in this research. Whenever, it needed, the same will be produced.
Consent for publication
All patients included in this study have given written informed consent to participate in this research. Whenever, it needed, the same will be produced.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix. Description of the study sites
Appendix. Description of the study sites
Study sites | Description | Major aquatic vegetation | Major macroinvertebrates | Major fish species |
|---|---|---|---|---|
Adra Sahebbandh (Site 1: 23°28′N, 86°42′E) | Shore length: 7.98 km; area: 76 ha; altitude: 166 msl; mean depth: 3.5 m. It is a natural wetland in Purulia district and surrounded by “Kang” forest. Timely water hyacinth management and proper protection by Indian Railway Department make this habitat suitable for winter avian fauna | Eichhornia crassipes, Nymphaea pubescens, Nymphoides hydrophylla, Hydrilla verticillata, Myriophyllum spicatum, Alternanthera philoxeroides | Macrobrachium sp., Gerris sp., Anisops sp., Limnogonus sp., Ranatra filiformis, Bellamya dissimilis, Corbicula sp., Gyraulus labiatus | Labeo rohita, Cirrhinus cirrhosus, Cyprinus carpio carpio, Labeo fimbriatus, Labeo gonius, Barilius barna, Puntius terio, Puntius ticto, Aplocheilus panchax, Garra lamta |
Purulia Sahebbandh (Site 2: 23°33′N, 86°35′E) | Shore length: 2.87 km; area: 31.3 ha; altitude: 250 msl; mean depth: 2.5 m. It is a man-made lake located in the middle of Purulia town and governed by Purulia Municipality. Wastewater input, plastic and thermocol pollution load, different anthropogenic activities, tourism and land use change (urbanization) in the surrounding area are major reasons for habitat degradation in this site | Eichhornia crassipes, Wolffia globosa, Nelumbo nucifera, Hydrilla verticillata, Ceratophyllum demersum, Marsilea minuta | Macrobrachium sp, Gerris sp., Anisops sp., Diplonychus sp., Micronecta sp., Bithynia pulchella, Bellamya bengalensis, Gyraulus convexiusculus | Catla catla, Cirrhinus cirrhosus, Ctenopharyngodon idella, Cyprinus carpio carpio, Labeo bata, Puntius sophore, Puntius ticto, Oreochromis mossambicus, Notopterus notopterus, Wallago attu |
Kadamdeuli dam (Site 3: 23°10′N, 86°85′E) | Shore length: 5.26 km; area: 38 ha; altitude: 116 msl; mean depth: 4.5 m. Located in Bankura District and it is a dam area of Shilabati river where a canal from Mukutmanipur-Kangsabati dam meets. The area is mainly surrounded by bushes, patch forest; however, barren lateritic land also presents. Anthropogenic pressure is less in this habitat due to its remote location | Pistia stratiotes, Utricularia gibba, Ipomea aquatica, Nymphoides hydrophylla, Hydrilla verticillata, Typha angustifolia | Macrobrachium sp., Palemon sp., Gerris sp., Limnogonus sp., Canthydrus sp., Indoplanorbis exustus, Gyraulus convexiusculus, Lymnaea acuminata | Labeo rohita, Catla catla, Hypophthalmicthys molitrix, Ctenopharyngodon idella, Labeo boga, Labeo pangusia, Barilius vagra, Amblypharyngodon mola, Glossogobius giuris, Mastacembelus armatus |
Gangdoa dam (Site 4: 23°40′N, 87°08′E) | Shore length: 9.24 km; area: 92.7 ha; altitude: 108 msl; mean depth: 7.5 m. It is a reservoir of Shali river located in Bankura district. This area is mostly surrounded by agricultural land, but fishing and illegal hunting creates pressure on this wetland | Eichhornia crassipes, Ipomea aquatica, Nymphoides indica, Hydrilla verticillata, Myriophyllum spicatum, Typha angustifolia, Polygonum barbatum | Macrobrachium sp., Gerris sp., Anisops sp., Lethocerus indicus, Corixa sp., Bellamya bengalensis, Pila globosa, Lymnaea acuminata | Labeo rohita, Catla catla, Cyprinus carpio carpio, Labeo calbasu, Labeo boga, Glossogobius giuris, Puntius sophore, Puntius sarana, Brachygobius nunus, Oreochromis mossambicus |
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mukherjee, A., Pal, S., Das, P. et al. Time activity budget and foraging behavior: important determinants of resource sharing and guild structure in wintering waterbirds. Eur J Wildl Res 69, 30 (2023). https://doi.org/10.1007/s10344-023-01648-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-023-01648-4