Abstract
Thousands of marine animals are injured and killed each year across Europe. Wildlife rehabilitation centres play an important role to rehabilitate sick and injured animals and return them to the wild. This study aims to assess seabird rehabilitation outcomes and causes of mortality of stranded seabirds in the central Portuguese coast. During a 7-year period (2010–2016) a total of 2042 admissions were registered, including 1135 live and 907 dead seabirds. The main causes for live admissions were trauma (30%) and toxicity (29.5%) and the main cause of all admissions (dead and alive) was entanglement/bycatch representing 42.5% of all seabirds. Large gulls, auks, gannets, and shearwaters and petrels were the most admitted seabird groups. A total of 445 seabirds (39%) were released to the wild following rehabilitation, 346 (31%) died during the rehabilitation process and 344 (30%) were considered untreatable after diagnosis and were euthanised. Considering the rehabilitation success rate and the high number of individuals admitted due to causes associated with human activities (entanglement/bycatch, trauma), marine animal rehabilitation centres and stranding networks are important to seabird conservation. These data also contribute to monitor the impacts of human activities on seabird populations outside their breeding areas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The most common anthropogenic causes of bird mortality include poisoning, collisions, commercial fishing, predation by introduced species and hunting (Harden 2002; Rodríguez et al. 2010). Seabird populations are threatened by incidental catch by fisheries (Moore et al. 2009; Good et al. 2009; Hamel et al. 2009; Brothers et al. 2010; Zydelis et al. 2013) and oil spills (Lovvorm 2007; Humple et al. 2007; Camphuysen 2013; Troisi et al. 2016) or similar.
A large number of wild animals are taken annually to rehabilitation centres for treatment, care and release (e.g. Anon 2009; Mazaris et al. 2008; Kalpakis et al. 2009; Wimberger and Downs 2010; Balmori 2019; Garcês et al. 2019a, b; Garcês et al. 2020). Apart from rehabilitation centres, stranding networks also allow collecting both dead and alive-beached marine animals, providing standardized data and adequate animal handling, which may determine subsequent treatment and outcomes, while contributing to estimates of minimal mortality and to the establishment of cause of death (Moore et al. 2018).
Although analyses of admittance records in rehabilitation centres may provide valuable insight into disease emergence, threats and impacts to wildlife, a systematic collection of information is usually lacking (Wimberger and Downs 2010; Willette et al. 2013). The analyses of the admission records (animals found dead and alive) may provide information on the variety of species and the number of individuals that are vulnerable in a local area or region (Harden et al. 2006), particularly as a result of human activities (Harden 2002; Rodríguez et al. 2010).
Common species living in close association with humans are those most frequently admitted to rehabilitation centres (Deem et al. 1998) because of the increased probabilities of injury and subsequent detection (du Toit 1999; Reeve and Huijser 1999; Barnett and Westcott 2001; Wimberger and Downs 2010). At the same time, since we have access only to animals stranded on the coast, this can significantly underestimate the magnitude of the problem, as we can only account for animals that were able to return to shore and/or survived long enough to reach the coast.
In the present study, we aim to assess seabird rehabilitation outcomes and causes of mortality of stranded seabirds in the study area.
Methods
The Marine Animal Rehabilitation Centre was initially located in Figueira da Foz (CRAMQ, 2012 PT 07/CR) and later transferred to Ílhavo (CRAM-Ecomare, 2017 PT 11/CR), both locations occurring in the centre of Portugal. The rehabilitation centre is managed by a non-government organisation—SPVS (The Portuguese Wildlife Society), in cooperation with the University of Aveiro. CRAM–Ecomare integrates the National Network of Fauna rehabilitation centres (legal ordinance no. 1112/2009, 28 September). Both rehabilitation centre and stranding network are regulated by the Instituto para a Conservação da Natureza e Florestas (ICNF). Permission is annually issued by the National Authority (ICNF) to rehabilitation and stranding network technicians to collect wild animals and wildlife samples according to laws n.140/99, n.49/2005, n.156-A/2013 and n.316/89.
The present study focuses on seabirds collected from 2010 to 2016 along a 300-km coastline, between the cities of Esposende and Torres Vedras. The marine area in this region is part of the Natura 2000 network, including Site Aveiro/Nazaré (PTZPE0060) and Site Ria de Aveiro (PTZPE0004), designated in accordance with the Birds Directive 2009/147/EC.
Animals were reported to the rehabilitation centre or to the stranding network by the general public, local authorities and different organisations. Upon arrival at the rehabilitation centre, all live animals were identified and fully examined by the veterinary and/or veterinary nurses. Age was categorized as <1 year or juvenile and >1 year, or adult, according to the European Union for Bird Ringing (Euring 1994). Upon diagnoses, injured/diseased birds were either kept for rehabilitation or euthanised. Once rehabilitated, seabirds were marked with an individually numbered aluminium ring and released back to the wild. Dead seabirds (including euthanised seabirds and birds deceased during collection or rehabilitation processes) were frozen until necropsy. Complete post mortem analyses were performed according to standard protocols (Work 2000) in order to determine a more detailed cause of death.
Seabirds were subdivided into 10 groups: auks, cormorants, gannets, large gulls, loons, shearwaters and petrels, small gulls, seaducks, terns and skuas (Table 1). The large gulls group included a subset of unidentified large gulls of juvenile and immature individuals of Larus michahellis and Larus fuscus, where species determination was not possible due to their similarities (Olsen and Larsson 2004; Catry et al. 2010).
In the present study, all collected seabirds (both dead an alive) were considered admissions, which were classified into eight categories: (1) trauma, (2) toxicity, (3) disease, (4) emaciation, (5) entanglement/bycatch, (6) plumage damage, (7) orphaned and (8) unknown. The trauma category included all birds presenting fractured bones, articular dislocations and open wounds of unknown origin. The toxicity category included animals presenting symptoms that were compatible with biotoxin poisoning, which include incapacity to fly, diarrhoea, paresis, dyspnoea, stiffness of neck and dehydration. The entanglement/bycatch category included all birds presenting fishing related material (hooks, lines, nets and cables) and other marine debris such as non-fishing lines or ropes. Diseased birds presented parasites or lesions and signs of infection, which causative agents were not considered in the present study. Emaciated birds presented low body mass, wasted muscles and no body fat (observed during necropsies), along with no other symptoms included in the other admission categories. The plumage damage category included all waterlogged birds, lacking feather permeability, resulting in hypothermia, along with no other symptoms included in other admission categories.
For all live seabird admissions, based on the study of Molina-López et al. (2013), the following parameters were calculated: euthanised birds (Er—euthanasia rate, birds with negative prognosis), birds that died during the rehabilitation process (Mr—mortality rate) and birds released into the wild (Rr—release rate). For each cause of admission, it was possible to calculate time until death (Td—number of days between admission and death) for birds that were euthanised or that died during the rehabilitation process, and time until release (Tr—number of days between admission and release) for all fully rehabilitated individuals.
To assess differences in cause of admission by season, the following periods were considered: Spring (March to May), Summer (June to August), Fall (September to November) and Winter (December to February).
Statistical analyses were performed with IBM SPSS v24.0 software (IBM 2016). The χ2 test was used to compare proportions among admission cause categories, seasons and years. Median, percentile 10 (P10) and percentile 90 (P90) for variables Td and Tr were calculated.
The present study was conducted in accordance with all applicable laws and rules dictated by the Portuguese Government and all licenses, including individual licenses for technicians (to collect, handle and transport wild fauna), were granted by the ICNF. Samples from dead seabirds were archived in the Marine Animal Tissue bank from SPVS (13PT0124/S) recognised by the ICNF with CITES permit code PT009 to maintain samples.
Results
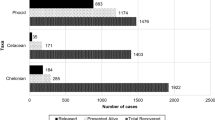
Between 2010 and 2016, 2042 seabird admissions were registered belonging to 26 species. The most common groups were large gulls (940 individuals), auks (702 individuals), gannets (272 individuals) and shearwaters and petrels (74 individuals) (Table 1).
Seabird admissions
Of the 2042 seabird admissions, 1135 corresponded to live seabirds (55.6%) and 907 corresponded to dead seabirds (44.4%). The majority of seabirds were juveniles corresponding to 54.3% (n = 1108) of all cases, whereas adults corresponded to 37.7% (n = 769) of all cases. No age class was identified in 8% (n = 165) of the cases. Sex was undetermined in 76.9% of the cases (considering the number of live seabirds and juveniles), so this parameter was not taken into any further consideration. The main cause of admission (dead and alive animals) was entanglement/bycatch (42.5%, n = 867), followed by trauma (17.0%, n = 347) and toxicity (16.8%, n = 344). The highest number of individuals was included in the category entanglement (867 individuals, χ2 = 2112.4, p < 0.001).
The main causes of admission for live seabirds were trauma (30%, n = 341) and toxicity (29.5%, n = 335), followed by emaciation (14.4%, n = 163) and plumage damage (141 individuals—12.4%).
Regarding age class, juveniles represented more than 50% of the individuals in the following causes of admission: disease (54.5%), emaciation (59.3%), entanglement/bycatch (61.0%), orphaned (100%) and trauma (68.6%). In other causes of admission, there was a higher number of adult seabirds: toxicity (57.6%), plumage damage (55.2%) and unknown (41.4%).
More birds were admitted during fall (709 individuals) and winter (695 individuals) (χ2 = 335.8, p < 0.001) than in the other seasons. Also, there were more admissions during fall due to emaciation (χ2 = 74.1, p < 0.001, 50%), toxicity (χ2 = 279.0, p < 0.001, 62%), trauma (χ2 = 142.5, p < 0.001, 48%) and unknown causes (χ2 = 153.1, p < 0.001, 70%). In the summer, there were more orphaned seabirds admitted (χ2 = 16.2, p < 0.001, 95%) and more entangled birds admitted in winter (χ2 = 742.7, p < 0.001, 65%) (Fig. 1). No significant differences were detected between seasons for the category disease (χ2 = 5.09, p = 0.08) (Fig. 2). More seabirds were admitted in 2012 than in the other study years (556 individuals, χ2 = 497.7, p < 0.001) (Fig. 1).
Final dispositions
Among the live seabirds initially admitted, 445 individuals (39.2%) were successfully released to the wild following rehabilitation, 346 (30.5%) died during rehabilitation and 344 (30.3%) were considered untreatable after diagnosis and were either euthanised or transferred to other centres where they are maintained in captivity for education and conservation purposes (Table 2). The highest success rates for released seabirds were obtained for the categories orphaned (90%), plumage damage (68.8%) and toxicity (52.8%). The lowest success rate was found for diseased birds since 77.8% (n = 7) of the diseased seabirds died during rehabilitation and 22.2% (n = 2) were euthanised. The highest percentage of euthanised individuals was recorded for the trauma category (77.1%, n = 263) (Table 2).
A median treatment time of 20.0 days (P10–P90 = 3–51.5 days) was obtained for released seabirds, whereas a median treatment time of 1.0 day (P10–P90 = 0–16.5 days) was obtained for birds that died during rehabilitation and a median treatment time of 0.0 days (P10–P90 = 0–8.6 days) was obtained for euthanised birds (Table 2).
Most representative groups
Taking into consideration the species groups with more than 50 seabird admissions, significant annual and seasonal differences were detected. The highest number of auk admissions occurred in 2012 (χ2 = 858.9, p < 0.001) during winter (χ2 = 1211.4, p < 0.001) (Fig. 3). The highest number of gannet admissions also occurred in 2012 (χ2 = 31.3, p < 0.001), during summer and fall (χ2 = 173.0, p < 0.001) (Fig. 3). The highest number of large gull admissions occurred in 2011 (χ2 = 356.4, p < 0.001) during fall (χ2 = 596.7, p < 0.001) (Fig. 3). The highest number of shearwater and petrel admissions occurred in 2010 (χ2 = 26.5, p < 0.001) during summer (χ2 = 82.2, p < 0.001) (Fig. 3).
Large gulls were more affected by toxicity (34%, n = 321) and trauma (29%, n = 270), auks as well as shearwaters and petrels were more affected by entanglement/bycatch (97%, n = 678 and 65%, n = 48, respectively) and gannets were more affected by emaciation (29%, n = 79), entanglement/bycatch (27%, n = 73) and trauma (25%, n = 67) (Fig. 4). Considering only live seabird admissions, large gulls presented the highest percentage of release (42.9%) and also the highest percentage of euthanised birds (35.5%). Shearwaters and petrels presented the highest percentage of deaths during rehabilitation (69.2%).
Discussion
This study is the first attempt to jointly assess the main causes of seabird admittances to a rehabilitation centre and seabird mortality causes based on the collection and analysis of dead seabirds of coastal Portugal. Entanglement/bycatch, trauma and toxicity are the main causes of concern for seabirds in coastal Portugal. The number of admissions caused by trauma and entanglement/bycatch were most likely associated with fishing activities, while toxicity was probably related to warmer waters and increasing algal toxins (Wells et al. 2015).
Considering the total number of individuals included in this study (dead and live admissions), most cases were related with the bycatch/entanglement category (42.5%, n = 867), mainly among the auk group. Fisheries are one of the most important economic activities in Portugal, with close to 8000 fishing vessels and the largest Economic Exclusion Zone in the European Union (INE 2017), thus explaining the high entanglement/bycatch rates. The auk group was mainly constituted by razorbills, and this pursuit-diver species exhibit high annual mortality rates due to bycatch (Artyukhin and Burkanov 2000; Österblom et al. 2002; Benjamins et al. 2008; Davoren 2007; Hamel et al. 2009; Zydelis et al. 2013; Costa et al. 2019). The impacts of several fishing types, including professional and recreational fishing, and also illegal, unreported and unregulated (IUU) fishing, together with fishing-related litter (discarded lines, weights, lures and hooks; see Costa et al. 2020), should be addressed and mitigated in order to decrease the importance of this cause of mortality. However, in the bycatch/entanglement category, only 12.7% (n = 110) of the cases correspond to live animals whereas 87.3% (n = 757) correspond to animals collected already dead, emphasising the importance of measures that may help prevent the occurrence of fishery-related mortality.
Trauma and toxicity represented the second and third major causes of admission, and the majority of these cases were diagnosed in the large gull group. This is probably as a result of the constant and increasing presence of large gulls in urban habitats (Rock 2005). Due to their opportunistic behaviour, these gulls are able to feed at landfill sites and fishing harbours and they also consume fishing discards and unwanted food in coastal towns (Garthe and Scherp 2003; Rock 2005). The trauma category included a high number of gulls presenting wing bone fractures. As described in other studies (Rodríguez et al. 2010; Garcês et al. 2019a), most trauma cases were most likely related with human-related collisions (cars, boats), in their pursuit for food in fishing harbours and urbanised areas. Toxicity presumptive affected 34% of individuals in the large gull group. However, the associated symptoms observed in these birds fit several types of possible toxicity sources. The inability to fly, diarrhoea, paresis, dyspnoea, stiffness of neck and dehydration may result from natural toxins (Shumway et al. 2003), heavy metals (Lewis and Schweitzer 2000; Rodríguez et al. 2010; Pikula et al. 2013; Soares 2014), pesticides (Stone et al. 1984; Glaser 1999; Kwon et al. 2004; Whitney 2004; Martínez-Haro et al. 2007; Soares 2014) or deliberate poisoning for population control or similar (Seamans and Belant 1999; Thompson 2013). The large amount of admissions within the fall season, especially in 2011 when more than 100 gulls exhibiting the same symptoms were recorded in a 3-month period, indicates that algal toxins are probably the main toxicity source. No specific analyses were made on the affected seabirds to confirm algal toxins as the main toxicity source. However, the presence of toxins produced by Dinophysis sp. (leading to diarrhetic shellfish poisoning, DSP) and by Alexandrium sp. (leading to paralytic shellfish poisoning, PSP) were detected along the study area during the study period by the Portuguese Institute for the Sea and Atmosphere (IPMA). IPMA defines interdiction areas for bivalve harvesting (www.ipma.pt/pt/bivalves/index.jsp) based on the regulatory screening of indicator mollusc species in compliance with European regulations EC 853/2004 and 854/2004.
The emaciation category was the fourth cause of admission and affected highly the gannet group. The northern gannet breeds in the North Atlantic and feeds on shoaling pelagic fish usually by plunge diving from large heights (del Hoyo et al. 1992). Emaciation, most likely due to starvation, may arise during periods of severe weather conditions, which hamper prey capture. Also, the high energy demands necessary for migration may not be met, especially for first year juveniles, since they receive no parental help and have to learn an efficient method of catching fish (Nelson 2002).
The categories plumage damage, unknown, orphaned and disease had lower numbers of admission (less than 7%) and were spread out through the species groups.
Juveniles had higher number of admissions compared to adults and they are probably less experienced in foraging and in avoiding debris (Riotte-Lambert and Weimerskirch 2013; Montesdeoca et al. 2017), which makes them more vulnerable than adults.
The significant differences detected in the seasonal admissions analysis and in the cause of admission analysis within the four large seabird groups were clearly related to the specific breeding/wintering seasons of the studied species included in each group. The auk group, mainly constituted by razorbills, is common in the Portuguese continental shelf between November and March (Beja 1989; Catry et al. 2010); the northern gannet is a common wintering species in the Portuguese coast (Meirinho et al. 2014). The large gull group is particularly constituted by lesser black-backed gulls, a common wintering species, and yellow-legged gulls, a resident species in continental Portugal (Catry et al. 2010). There were more admissions of these four groups in the winter and fall seasons. As for the shearwater and petrels, there were more admissions in the summer. The Balearic shearwater in particular spends around a quarter of the year (generally, late summer) on migration off north-eastern Atlantic coasts, from Portugal to France (Guilford et al. 2012).
Considering only the live seabird admissions, an average release rate of 39.2% was obtained. Comparing to other similar studies, Molina-López et al. (2017) reported a lower release rate for marine birds (27.5%) and other studies reported higher rehabilitation success rates for birds in rehabilitation centres (Rodríguez et al. 2010; Molina-López et al. 2013; Montesdeoca et al. 2017). These higher rates may however be related with the most important causes of admission in each study. Notice that Montesdeoca et al. (2017) report light pollution (25.81%) as the most important cause of admission, which is much more easily resolved than trauma cases, which constitute the main cause of admission in the present study, followed by toxicity.
In the present study, the high number of severe trauma cases and corresponding high rates of euthanasia decreases substantially the release rate. Disregarding the number of euthanised birds due to trauma, the release rate increases to 51%, leading us to believe that bird group and cause of admission are therefore important determinants of rehabilitation success. Due to the location of the rehabilitation facilities, bird admissions may originate from several cities and, more importantly, from two major fishing and industrial harbours (Aveiro and Figueira da Foz harbours). The large coastal area covered by the stranding network and the rehabilitation centre also increases the probability of a high number of trauma and entanglement/bycatch cases. Although the results of our study present a high number of directly human-induced seabird injuries, our study population of rescued individuals does not necessarily represent the wild population. As pointed out by Dau et al. (2009), this type of data must be interpreted in light of the fact that proportions of injuries are being compared with overall admissions to rehabilitation facilities, rather than with the wild population as a whole.
Time spent at the rehabilitation facilities should be as short as possible to reduce the risk of exposure to secondary bacterial/fungal infections of seabirds (Steele et al. 2005). Therefore, seabirds should be immediately released once they are fully recovered and presenting a high probability of survival. In the present study, the median time until release was 20 days. Montesdeoca et al. (2017) reported a much lower valued (zero days) since the main admission cause generally had a very positive prognosis (light pollution leading to “fallout”). The severity of the different admission causes must be taken into consideration when comparing rehabilitation parameters between studies.
Survival rate is also an important parameter since the actual rehabilitation success can only be assessed when data on post-release survival are available. Presently, data on post-release survival rate are being compiled for large gulls using a colour ringing scheme (www.cr-birding.org/node/2331) and 45.5% (n = 134) of all ringed seagulls were already re-sighted (further data will be the scope of another publication).
Further efforts should be put into citizen science (Pellissier et al. 2019; Nadal et al. 2020) to increase information on bird occurrence and ultimately increase reports to strandings networks and to rehabilitation centres. Unveiling bird population patterns related with climate change or phenological shifts (Almpanidou et al. 2020; Lehikoinen et al. 2019) may also capitalise on the data provided by rehabilitation centres and stranding networks.
Conclusion
This study highlights the importance of wildlife rehabilitation centres for the conservation of seabirds and the high number of seabird admissions due to human activity–associated causes (entanglement/bycatch, trauma). Further efforts to prevent negative interactions between fishing activities and seabirds are urgent and further studies would be necessary to know the post-release survival rate of rehabilitated seabirds and to understand the effects of rehabilitation on the conservation of seabird populations in Portugal.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
References
Almpanidou V, Tsapalou V, Tsavdaridou AI, Mazris A (2020) The dark side of raptors’ distribution ranges under climate change. Landscape Ecol 35:1435–1443
Anon (2009) BWRC-Wildlife Casualty Recording Scheme. British Wildlife Rehabilitation Council, UK http://wildlife1.widllifeinformation.org/S/00Ref/miscellaneouscontents/BWRC-CasualtyRecordingScheme/Titlepage.htm
Artyukhin YB, Burkanov VN (2000) Incidental mortality of seabirds in the drift net salmon fishery by Japanese vessels in the Russian Exclusive Economic Zone, 1993–97. In: Kondratyev AY, Litvinenko NM, Kaiser GW (eds) Seabirds of the Russian Far East. Canadian Wildlife Service, Ottawa, ON, pp 105–115
Balmori A (2019) Endangered bird mortality by gunshots: still a current problem. Biodivers Conserv 28:2555–2564
Barnett J, Westcott S (2001) Distribution, demographics and survivorship of grey seal pups (Halichoerus grypus) rehabilitated in southwest England. Mammalia 65(3):349–361
Beja (1989) Beja PR (1989) A note on the diet of Razorbills Alca torda wintering off Portugal. Seabird 12:11–13
Benjamins S, Kulka DW, Lawson J (2008) Incidental catch of seabirds in Newfoundland and Labrador gillnet fisheries, 2001–2003. Endang Species Res 5:149–160
Brothers N, Duckworth AR, Safina C, Gilman EL (2010) Seabird bycatch in pelagic longline fisheries is grossly underestimated when using only haul data. PLoS ONE 5(8):e12491. https://doi.org/10.1371/journal.pone.0012491
Camphuysen CJ (2013) Monitoring and assessment of the proportion of oiled Common Guillemots from beached bird surveys in The Netherlands: update winter 2012/13. Report commissioned by the Ministry of Infrastructure and the Environment (I&M), RWS Water, Traffic and Living Environment (RWS-WVL) and DG Mobility and Transport (DGB). Royal Netherlands Institute for Sea Research, Texel.
Catry P, Costa H, Elias G, Matias R (2010) Aves de Portugal. Ornitologia do território continental. Assírio & Alvim, Lisboa
Costa RA, Pereira AT, Costa E, Henriques AC, Miodonski J, Ferreira M, Vingada JV, Eira C (2019) Razorbill Alca torda mortality in the Portuguese west coast. Eur J Wildlife Res 65:3
Costa RA, Sá S, Pereira AT, Ângelo AR, Vaqueiro J, Ferreira M, Eira C (2020) Prevalence of entanglements of seabirds in marine debris in the central Portuguese coast. Marine Pollution Bulletin 161:111746
Dau BK, Gilardi KVK, Gulland FM, Higgins A, Holcomb JB, St. Leger J, Ziccardi MH (2009) Fishing gear–related injury in California marine wildlife. J Wildl Dis 45(2):355–362
Davoren GK (2007) Effects of gill-net fishing on marine birds in a biological hotspot in the Northwest Atlantic. Conserv Biol 21:1032–1045
Deem SL, Terrell SP, Forrester DJ (1998) A retrospective study of morbidity and mortality of raptors in Florida: 1988–1994. J Zoo Wildl Med 29(2):160–164
del Hoyo J, Elliot A, Sargatal J (1992) Handbook of the birds of the world, vol 1: ostrich to ducks. Lynx Edicions, Barcelona, Spain
Du Toit J (1999) A helping hand. Africa Birds and Birding 4(3):60–63
EURING (1994) El anillamiento de aves: herramienta científica y de gestión ambiental. Dirección General de Conservación de la Naturaleza-SEO/BirdLife, Madrid
Garcês A, Pires I, Pacheco FAL, Fernandes LFS, Soeiro V, Lóio S, Prada J, Cortes R, Queiroga FL (2019a) Preservation of wild bird species in northern Portugal – effects of anthropogenic pressures in wild bird populations (2008–2017). Sci Total Environ 650:2996–3006
Garcês A, Pires I, Pacheco F, Fernandes LS, Soeiro V, Lóio S, Prada J, Cortes R, Queiroga F (2019b) Natural and anthropogenic causes of mortality in wild birds in a wildlife rehabilitation centre in Northern Portugal: a ten-year study. Bird Study 66(4):484–493
Garcês A, Queiroga F, Prada J, Pires I (2020) A review of the mortality of wild fauna in Europe in the last century: the consequences of human activity. Journal of Wildlife and Biodiversity 4(2):34–55
Garthe S, Scherp B (2003) Utilization of discards and offal from commercial fisheries by seabirds in the Baltic Sea. Ices J Mar Sci 60(5):980–989
Glaser LC (1999) Organophosphorus and carbamate pesticides. In: Friend M, Franson JC (eds) Field manual of wildlife diseases: general field procedures and diseases of birds. U.S. Geological Survey, Washington DC, pp 287–293
Good TP, June JA, Etnier MA, Broadhurst G (2009) Ghosts of the Salish Sea: threats to marine birds in Puget Sound and the Northwest Straits from derelict fishing gear. Mar Ornithol 37:67–76
Guilford T, Wynn R, McMinn M, Rodríguez A, Fayet A, Maurice L, Jones A, Meier R (2012) Geolocators reveal migration and pre-breeding behaviour of the critically endangered Balearic shearwater Puffinus mauretanicus. PLoS ONE 7(3):e33753. https://doi.org/10.1371/journal.pone.0033753
Hamel NJ, Burger AE, Charleton K, Davidson P, Lee S, Bertram DF, Parrish JK (2009) Bycatch and beached birds: assessing mortality impacts in coastal net fisheries using marine bird strandings. Mar Ornithol 37:41–60
Harden J (2002) An overview of anthropogenic causes of avian mortality. J Wildlife Rehabil 25:4–11
Harden J, Dickerman RW, Elliston EP (2006) Collection, value, and use of wildlife rehabilitation data. J Wildlife Rehabil 28(1):10–28
Humple D, Abraham C, Ramos A, Dopico M, Fernández-Boán M, García L, Bao R, Heubeck M, Camphuysen CJ (2007) Age and sex structure of auk (Alcidae) mortality during the Prestige oil spill in Galicia, NW Spain, November 2002–April 2003. In: Conference Proceedings of the Ninth International Effects of Oil on Wildlife Conference, Monterey, California
IBM Corp. Released (2016) IBM SPSS Statistics for Macintosh, version 24.0. IBM Corp, Armonk, NY
INE (2017) Estatísticas da Pesca 2017. Instituto Nacional de Estatística, I.P. Lisboa, Portugal.
Kalpakis S, Mazaris AD, Mamakis Y, Poulopoulos Y (2009) A retrospective study of mortality and morbidity factors for common buzzards Buteo buteo and long-legged buzzards Buteo rufinus in Greece: 1996–2005. Bird Conserv Int 19(1):15–21
Kwon Y-K, Wee S-H, Kim J-H (2004) Pesticide poisoning in wild birds in Korea from 1998 to 2002. Short communications. J Wildl Dis 40(4):737–740
Lehikoinen A, Lindén A, Karlsson M, Andersson A, Crewe TL, Dunn EH, Gregory G, Karlsson L, Kristiansen V, Mackenzie S, Newman S, Røer JE, Sharpe C, Sokolov LV, Steinholtz A, Stervander M, Tirri I, Tjørnløv RS (2019) Phenology of the avian spring migratory passage in Europe and North America: asymmetric advancement in time and increase in duration. Ecol Indic 10:985–991
Lewis L, Schweitzer SH (2000) Lead poisoning in a Northern bobwhite in Georgia. J Wildl Dis 36:180–183
Lovvorm JR (2007) Modeling oil spill impacts on seabirds, with special reference to Cassin’s auklets. In: Conference Proceedings of the Ninth International Effects of Oil on Wildlife Conference, Monterey, California
Martínez-Haro M, Vinuela J, Mateo R (2007) Exposure of birds to cholinesterase-inhibiting pesticides following a forest application for tick control. Environ Toxicol Pharmacol 23(3):347–349
Mazaris A, Mamakis Y, Kalpakis S, Poulopoulos Y, Matsinos Y (2008) Evaluating potential threats to birds in Greece: an analysis of a 10-year data set from a rehabilitation centre. Fauna and Flora International, Oryx 42(3):408–414
Meirinho A, Barros N, Oliveira N, Catry P, Lecoq M, Paiva V, Geraldes P, Granadeiro JP, Ramírez I, Andrade J (2014) Atlas das Aves Marinhas de Portugal. Sociedade Portuguesa para o Estudo das Aves, Lisboa
Molina-López RA, Casal J, Darwich L (2013) Final disposition and quality auditing of the rehabilitation process in wild raptors admitted to a wildlife rehabilitation centre in Catalonia, Spain, during a twelve year period (1995–2007). PLoS ONE 8(4):e60242. https://doi.org/10.1371/journal.pone.0060242
Molina-López RA, Mañosa S, Torres-Riera A, Pomarol M, Darwich L (2017) Morbidity, outcomes and cost–benefit analysis of wildlife rehabilitation in Catalonia (Spain). PLoS ONE 12(7):e0181331. https://doi.org/10.1371/journal.pone.0181331
Montesdeoca N, Calabuig P, Corbera JA, Orós J (2017) A long-term retrospective study on rehabilitation of seabirds in Gran Canaria Island, Spain (2003–2013). PLoS ONE 12(5):e0177366
Moore JE, Wallace BP, Lewison RL, Zydelis R, Cox TM, Crowder LB (2009) A review of marine mammal, sea turtle and seabird bycatch in USA fisheries. Mar Policy 33(3):435–451
Moore KM, Simeone CA, Brownell L Jr (2018) Strandings. In: Würsig B, Thewissen JGM, Kovacs KM (eds) Encyclopedia of marine mammals (third edition). Academic Press, pp 945–951
Nadal J, Ponz C, Margalida A, Pennisi L (2020) Ecological markers to monitor migratory bird populations: integrating citizen science and transboundary management for conservation purposes. J Environ Manage 255:109875
Nelson B (2002) The Atlantic gannet, 2nd edn. Fenix Books, Norfolk
Olsen KM, Larsson H (2004) Gulls of North America, Europe, and Asia, 2nd edn. Princeton University Press
Österblom H, Fransson T, Olsson O (2002) Bycatches of Common Guillemot (Uria aalge) in the Baltic Sea gillnet fishery. Biol Conserv 105:309–319
Pellissier V, Schmucki R, Pe’er G, Aunins A, Brereton TM, Brotons L, Carnicer J, Chodkiewicz T, Chylarecki P, del Moral JC, Escandell V, Evans D, Foppen R, Harpke A, Heliola J, Herrando S, Kuussaari M, Kuhn E, Lehikoinen A, Lindstrom A, Moshøj CM, Musche M, Noble D, Oliver TH, Reif J, Richard D, Roy DB, Schweiger O, Settele J, Stefanescu C, Teufelbauer N, Touroult J, Trautmann S, van Strien AJ, van Swaay CAM, van Turnhout C, Vermouzek Z, Vorısek P, Jiguet F, Julliard R (2019) Effects of Natura 2000 on nontarget bird and butterfly species based on citizen science data. Conserv Biol 34(3):666–676
Pikula J, Hajkova P, Bandouchova H, Bednarova I, Adam V, Bemklova M, Kral J, Ondracek K, Osickova J, Pohanka M, Sedlackova J, Skochova H, Sobotka J, Treml F, Kizek R (2013) Lead toxicosis of captive vultures: case description and responses to chelation therapy. BMC Vet Res 9(11):1–10
Reeve NJ, Huijser MP (1999) Mortality factors affecting wild hedgehogs: a study of records from wildlife rescue centers. Lutra 42:6–24
Riotte-Lambert L, Weimerskirch H (2013) Do naive juvenile seabirds forage differently from adults? Proc. R. Soc. B 280:20131434
Rock P (2005) Urban gulls: problems and solutions. British birds 98:338–355
Rodríguez B, Rodríguez A, Siverio F, Siverio M (2010) Causes of raptor admissions to a wildlife rehabilitation center in Tenerife (Canary Islands). J Raptor Res 44(1):30–39
Seamans TW, Belant JL (1999) Comparison of DRC-1339 and alpha-chloralose to reduce herring gull populations. Wildl Soc Bull 27(3):729–733
Shumway SE, Allen SM, Dee Boersma P (2003) Marine birds and harmful algal blooms: sporadic victims or under-reported events? Harmful Algae 2:1–17
Soares SPV (2014) Paretic syndrome in gulls (Laridae) in the south of Portugal. M.Sc. thesis, Faculdade de Medicina Veterinária, Lisbon University.
Steele CM, Brown RN, Botzler RG (2005) Prevalences of zoonotic bacteria among seabirds in rehabilitation centers along the Pacific Coast of California and Washington, USA. J Wildl Dis 41(4):735–744
Stone WB, Overmann SR, Okoniewski JC (1984) Intentional poisoning of birds with parathion. The Condor 86(3):333–336
Thompson RP (2013) Factors influencing the admission of urban nesting Herring Gulls Larus argentatus into a rehabilitation centre and post release survival in comparison with wild counterparts. University of Sussex DPhil thesis.
Troisi G, Barton S, Bexton S (2016) Impacts of oil spills on seabirds: unsustainable impacts of non-renewable energy. International Journal of Hydrogen Energy 41(37):16549–16555
Wells ML, Trainer VL, Smayda TJ, Karlson BSO, Trick CG, Kudela RM, Ishikawa A, Bernard S, Wulff A, Anderson DM, Cochlan WP (2015) Harmful algal blooms and climate change: learning from the past and present to forecast the future. Harmful Algae 49:68–93
Whitney H (2004) Wildlife diseases fact sheet: Pesticide poisoning of seagulls. Government of Newfoundland and Labrador – Department of Natural Resources, AP051
Willette M, Ponder J, McRuer DL, Clark EE Jr (2013) Wildlife health monitoring systems in North America: from sentinel species to public policy. In: Aguirre A (ed) Conservation Medicine: Applied Cases of Ecological Health
Wimberger K, Downs CT (2010) Annual intake trends of a large urban animal rehabilitation centre in South Africa: a case study. Anim Welfare 19:501–513
Zydelis R, Small C, French G (2013) The incidental catch of seabirds in gillnet fisheries: a global review. Biol Conserv 162:76–88
Acknowledgements
The authors wish to thank everyone who reported and collected seabirds, including the Maritime Authority and the Republican National Guard (GNR-SEPNA) and all the staff and volunteers of the stranding network and rehabilitation centres CRAM-Q and CRAM-Ecomare.
Funding
This study was partly supported by the Portuguese Foundation for Science and Technology (FCT) with Grants attributed to R.A.C. (SFRH/BPD/99394/2013), to S.S. (PD/BD/127920/2016), to A.T.P. (SFRH/BD/122890/2016) and to M.F. (SFRH/BD/30240/2006). R.A.C. is funded by national funds (OE), through FCT—Fundação para a Ciência e a Tecnologia, I.P., in the scope of the framework contract foreseen in the numbers 4, 5 and 6 of article 23, of the Decree-Law 57/2016, of August 29, changed by Law 57/2017, of July 19. C.E. is supported at CESAM (UID/AMB/50017) by FCT/MCTES, through national funds and co-funding by FEDER, within the PT2020 Partnership Agreement and Compete 2020 (POCI-01-0145-FEDER-007638). Thanks are due for the financial support to CESAM (UIDP/50017/2020 + UIDB/50017/2020) through national funds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was conducted in accordance with all applicable laws and rules dictated by the Portuguese Government and all licenses granted by Institute for Nature and Forest Conservation (ICNF). Samples from stranded birds are archived in the Marine Animal Tissue bank (13PT0124/S) recognised by the ICNF with CITES permit code PT009 to maintain samples.
Consent for publication
All authors read and approved the final manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
J. V. Vingada Deceased 19 March 2019
Rights and permissions
About this article
Cite this article
Costa, R.A., Sá, S., Pereira, A.T. et al. Threats to seabirds in Portugal: integrating data from a rehabilitation centre and stranding network. Eur J Wildl Res 67, 41 (2021). https://doi.org/10.1007/s10344-021-01483-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-021-01483-5