Abstract
The potential productivity of calcareous soils is high when adequate nutrients can be available. However, these soils are frequently deficient in nitrogen (N) and phosphorus (P) and cannot meet plant nutrient requirements. The application of microbes with chemical fertilizers can be beneficial in fulfilling plant-nitrogen-phosphorus (NP) requirements. Given that, a field experiment was conducted to determine the effect of arbuscular mycorrhiza fungi (AMF) infected roots of maize and single super phosphate (SSP) in improving NP acquisition, yield, root infection activity, and AMF spore density in alkaline-calcareous soil. The experiment was composed of five levels of SSP (0, 30, 60, 90, 120 kg ha−1) and two levels of AMF (with and without) arranged in a two factorial randomized complete block design (RCBD) with three replications. Yield-related parameters were significantly (P ≤ 0.05) improved with the addition of AMF and SSP. Post-harvest soil total N, extractable soil P, plant N, and plant P concentration and its uptake by wheat plants were also significantly (P ≤ 0.05) improved by the combined application of AMF and SSP compared to sole SSP application. Very often, the performance of 90 kg ha−1 SSP with AMF was either similar to or better than 120 kg ha−1. Moreover, AMF inoculation was found effective in soil AMF spore density and root infection intensity leading to an increase of 18 to 48/20 g soil and 19 to 42% compared to no AMF inoculation. It was proved that wheat-NP requirements could be improved with AMF inoculation, and it has the potential to increase crop yield in alkaline calcareous soils.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plant growth and development are majorly dependent on the availability of nutrients and their level in the soil. An inappropriate, minimum or excessive amount of nutrients in the soil inhibits soil fertility, plant growth, and yield (Pan et al. 2022). Among those soil nutrients, phosphorus (P) is a macro-element and an essential requirement for plants and almost all life on earth (Lambers 2022). It plays a crucial role in many processes such as photosynthesis, energy transfer, and metabolism of nucleic acid (Tshibangu Kazadi et al. 2022). Besides its vital role in crop growth, it is challenging to manage adequate P levels in alkaline calcareous soil because of its narrow pH range of 6–6.5, which is desirable for plants to uptake P. In calcareous soils, calcium (Ca2+) and magnesium (Mg2+) are the dominant cations due to which P immobilizes, and plants are unable to uptake P (Tshibangu Kazadi et al. 2022). The role of P is yet vital for plant growth and crop production. It is supplied through various chemical fertilizers such as single superphosphate (SSP), rock phosphate (RP), and diammonium phosphate (DAP). The issues related to the frequent use of chemical fertilization are that it is costly, disturbs soil quality, and its utilization issues. It has been reported that only 25–30% of the applied P is available to plants, while the rest are converted into plant unusable forms (Pan et al. 2022; Penn and Camberato 2019). Therefore, the researchers adopted different management practices to increase plant‑P acquisition. The use of targeted P fertilizers, the development of sustained released P fertilizers, various manures, growing crops with low P requirements, and advanced cropping systems are some P‑management practices (An et al. 2021; Aziz et al. 2014; Penn and Camberato 2019). Due to some limitations of these strategies, researchers have adopted an alternative way, such as inoculation with beneficial microorganisms in the form of bio-fertilizer, to reduce dependency on chemical fertilization and improve crop‑P acquisition (Cozzolino et al. 2013; Higo et al. 2018; Khan et al. 2022).
In this regard, beneficial microbes such as arbuscular mycorrhiza fungi (AMF) are most pronounced in alkaline calcareous soils as they are eco-friendly and efficient. Around 90% of crops are mycorrhizal. They form a symbiosis in which both fungi and plants benefit, primarily arbuscular, such as glomus, gigaspora scuteusllospora, acaulospora, and sclerocystis. AMF is regarded as a natural fertilizer due to its potential to boost the availability of nutrients to plant roots, piquing the interest of most scientists worldwide (Ahanger et al. 2014; Toussaint et al. 2007). The creation of AMF symbiosis is beneficial to plants because it gives a natural remedy to plants when they thrive in nutrient-deficient circumstances with limited fertilizer supply (Wahid et al. 2020). AMF root colonization is accomplished by establishing a mycelial network of fungi that spreads beneath plant roots, allowing the intake of otherwise unapproachable nutrients to plants (Begum et al. 2019).
Previous research has found that inoculation of AMFs can improve plant development by increasing nutrients uptake, especially in low-nutrients soil (Meng et al. 2015; Tarraf et al. 2017). Inoculation of AMF improves nutrients uptake, particularly phosphorus (P), by secreting phosphate in the rhizosphere, which catalyzes and releases P for plant uptake (Erdinc et al. 2017), and plant tolerance to many types of stresses, influencing plant growth and reproduction (d Oliveira et al. 2022), and hence increase crop yield (Jan et al. 2014; Tshibangu Kazadi et al. 2020).
In Pakistan, the soils are usually alkaline and calcareous, with low organic matter content, N and P for plant growth (Ahanger et al. 2014; Rahim et al. 2020). Even though these soils have plenty of stores of total P; the bio-available fraction is frequently 100 times less than the total P due to its adsorption on the calcite surface and becomes unavailable to plants (Hinsinger et al. 2011; Izhar Shafi et al. 2020). Likewise, N concentration in these soils is sufficient but lost through ammonia volatilization and becomes deficient for plant growth (FAO 2022). AMF could be a productive biological approach to replenishing NP availability in calcareous soils. However, the effect of AMF in soils of calcareous nature and alkaline reactions has not been well-studied. Therefore, the current study was performed to determine the influence of AMF inoculation with different levels of SSP on the wheat-NP availability, yield, spore density, and root infection intensity in alkaline calcareous soils of the semiarid climate of Khyber Pakhtunkhwa, Pakistan.
Material and Methods
Experimental Setup
A field experiment was conducted during the winter of 2018 at a research farm, the University of Agriculture Peshawar Pakistan, located at 34.1o 21″ N, 71o 28′5″ E (Fig. 1), to investigate the impact of mycorrhiza inoculation along with different levels of SSP on wheat-N-P-acquisition, yield, and AMF spore density and root infection intensity in alkaline-calcareous soil of the semiarid region of Khyber Pakhtunkhwa, Pakistan. During the period of the field experiment, Peshawar city had a soil temperature of 10.09–33.12 ℃ with an average temperature of 22.64 oC, while the air temperature was within the range of 9.47–31.11 oC with an average temperature of 21.50 ℃. Monthly mean relative humidity and rainfall were 53.08% and 12.66 mm (Fig. 2). Wheat variety Pirsabaq-2013 was used as a test crop with a seed rate of 120 kg ha−1. Recommended levels of nitrogen (N) and potassium (K) @ 120 and 60 Kg ha−1 were applied uniformly as basal doses to all plots. K was applied at the sowing time in the form of potassium sulfate, while N was applied in the form of urea in two equal splits, i.e., with sowing and after 20 days of wheat germination at the time of irrigation. Phosphorus was applied in the form of single super phosphate (SSP) at different levels of 0 (control), 30, 60, 90, and 120 kg ha−1 with or without AMF inoculation (Table 1). The AMF inoculation was produced in the sterilized pots by growing maize crop inoculated with AMF spores isolated from the fresh growing berseem crop. The AMF infected maize root and soil were used uniformly as inoculum at 1.5 kg plot−1. A randomized complete block design (RCBD) was used with three replicates.
Geographical location of the study site (Agricultural Research Farm, The University of Agriculture, Peshawar, Pakistan). (Khan et al. 2022; CC BY 4.0, http://creativecommons.org/licenses/by/4.0/)
Production of the AMF Inoculum
The soil as a substrate was autoclaved at 121 ℃ for 30 min to ensure complete sterility. A total of 100 AMF spores per pot with 4 maize seeds were inoculated. The plants were harvested after 80 days, and the whole root system along with the soil was removed from each pot. The air-dried soil and roots were having an average of 55–60 spores per 10 g of soil with 43% AMF colonization. All roots were cut carefully and mixed with the soil and stored at 4 ℃ for field application as inoculum.
Soil Sampling, Processing, and Analysis
Soil samples were collected at a depth of 0–30 cm in a zigzag manner before the experiment (pre-harvest) to represent the experimental site and at the end of the experiment (post-harvest) to evaluate the effect of treatments in each experimental plot. Both soil samples (pre and post-harvest) were air-dried and were subjected to a sieve (< 2 mm) to analyze different soil parameters. Soil and plant total nitrogen content was determined by the method of (Bremner 1996), AB-DTPA extractable soil P by the method of (Soltanpour and Schwab 1977), while total P content in the plant was analyzed by the method of (Kuo et al. 1996). Pre-harvest soil analysis reveals that the experimental field was silt loam, alkaline in reaction, calcareous in nature, and deficient in organic matter, ABDTPA extractible P, and total N content (Table 2).
Morphological and Yield-related Parameters
Data regarding morphological and yield-related traits such as grain yield, straw yield, and thousand-grain weight were collected and analyzed using the appropriate procedure as suggested by (Akram et al. 2008). Grains were removed from the straw by threshing each plot separately to estimate grain yield. Grain yield was noted for a single plot and converted into grain yield kg ha−1. A thousand grains were collected from each treatment plot randomly and weighed with the help of electric balance. Mycorrhiza infection rates in the roots of wheat were determined by staining the mycorrhiza chitin with lactic-trypan blue according to the procedure described by (Koske and Gemma 1989; Phillips and Hayman 1970). Soil spores density of mycorrhiza in 20 g soil was calculated and analyzed by dispersion method as suggested by (Berta et al. 2014).
Statistical Analysis
Data obtained from various parameters were statistically analyzed using the one-way analysis of variance (ANOVA). A significant difference in the means of treatments at a 5% level of significance was parted using the least significant difference (LSD) test using the statistix 8.1 package tool.
Results
Wheat Yield Parameters
Data regarding yield-related parameters revealed that the sole applications of AMF and SSP levels significantly (P ≤ 0.05) affected the yield parameters of wheat (grain yield, straw yield, and thousand-grain weight); however, their interactive effect was non-significant (Table 3). SSP application at the rate of 90 kg ha−1 performed better and recorded a maximum grain yield of 3340.2 kg ha−1, a straw yield of 5010 kg ha−1, and a thousand-grain weight of 47 g as compared to 0, 30, 60, and 120 kg ha−1 SSP. Although SSP application at the rate of 90 kg ha−1 SSP recorded maximum yield, the differences b/w 90 & 120 kg ha−1 SSP are negligible. The data shows the trend of 90 ≥ 120 > 60 > 30 > 0 kg ha−1 SSP concerning wheat yield. In the case of mycorrhiza, AMF inoculation performed better and recorded a maximum grain yield of 3177 kg ha−1, a straw yield of 4765 kg ha−1, and a thousand-grain weight of 44 g compared to un-inoculated treatment.
Post-harvest Soil and Plant NP Concentration, and Its Uptake
Soil NP Concentration
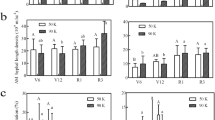
The data regarding post-harvest soil NP concentrations recorded contrasting effects as shown in Table 4. AMF resulted in a drastic increase in soil total nitrogen from 1550 mg kg−1 to 1555 mg kg−1, whereas SSP levels had no effect. Interestingly, AMF increase the concentration of soil P compared to control but the effect was non-significant statistically, while, SSP levels had a significant effect. Among SSP levels, 90 & 120 kg/ha−1 recorded the same concentration of P in soil i.e., 4.9 mg kg−1. The interactive effect of AMF with SSP levels on soil total N and extractable P was found non-significant.
Plant NP Concentration
Data regarding plant N reveals that P levels significantly affect plant N (Table 4). Maximum plant N concentration, i.e., 1.1%, was recorded with 90 kg ha−1 SSP and 120 kg ha−1 SSP. In the case of mycorrhiza inoculation, it had no significant effect on plant N concentration. However, the interaction effect of AMF inoculation and different P levels on plant N concentration was significant. The mean values indicate that a maximum plant N concentration of 1.15% was obtained from the plot inoculated with AMF and 90 kg ha−1 SSP. Interestingly the same results were also given by AMF inoculation with 120 kg ha−1 SSP. The lowest plant N concentration of 0.92% was recorded in the control. Data concerning plant P concentration indicates that the application of different levels of phosphorus increases plant P content. Maximum plant P content of 0.3% was recorded with the application of 90 kg ha−1. The application of 120 and 60 kg ha−1 SSP recorded the same results of plant P concentration (0.2%). A higher plant P concentration of 0.21% was recorded from AMF inoculation than from un-inoculated treatment (0.16%) between mycorrhiza inoculation and un-inoculated treatments. Moreover, the interaction effect of AMF inoculation and different P levels on plant P concentration was found non-significant.
Plant NP Uptake
The application of AMF and SSP levels significantly (P ≤ 0.05) enhanced the uptake of NP by wheat plants. However, their interactive effect on NP uptake was non-significant (Table 5). Specifically, the combined application of AMF and 90 kg ha−1 resulted in maximum N (98 kg ha−1) and P (22 kg ha−1) uptake compared to other SSP levels applied either in combination with AMF or not.
Effect On Soil Spores Density and Root Infection Intensity
SSP levels resulted in a linear increase in AMF spore density and root infection activity (Table 6). AMF spore densities were 30–36 per 20 g for all SSP levels, maximum those of control (28). The root infection activities were 31–35% for all SSP levels compared to control (23%). Mycorrhiza treatment also resulted in a notable increase in AMF spore density and root infection activity. Higher AMF spore density (47.5 spores per 20 g soil) was recorded with AMF inoculation than with un-inoculated treatment (18 spores per 20 g soil). Similarly, higher root infection intensity of 42% was recorded with AMF inoculation compared to un-inoculated treatment (19%). Furthermore, the interactive effect of AMF inoculation and P levels on both AMF spore density and root infection activity were found significant. Maximum mycorrhiza spore density of 53 spores per 20 g soil was obtained from the plots inoculated with AMF and 90 kg ha−1 SSP compared to control plots (15 spores per 20 g soil). Similarly, maximum root infection intensity of 47% was obtained from the plots inoculated with AMF and both 120 & 90 kg ha−1 SSP compared to control (15.6%).
Discussion
The availability of nutrients such as phosphorous and nitrogen in alkaline-calcareous soil is challenging worldwide (Karimi et al. 2020). The current trends toward less use of chemical fertilization and techniques based on microbes to improve crop yield in alkaline soils are increasing daily (Khan et al. 2022). Improvement in the availability and uptake of plant nutrients is an alternative to the higher application of chemical fertilization. The use of AMF is of particular importance for improving the efficiency of applied nutrients in alkaline soils (Bush 2008; Smith and Read 2010). Therefore, in the current study, we investigated the effect of AMF with SSP levels on wheat-NP availability, uptake, yield, root infection activity, and AMF spores density in nutrient deficient alkaline-calcareous soil.
Wheat yield were significantly (P ≤ 0.05) improved with the integrated use of AMF and SSP. The application of AMF with 90 kg ha−1 SSP level produced a significantly maximum grain yield of wheat (3399 kg ha−1), straw yield (5099 kg ha−1), and thousand-grain weight (49 g) compared to no AMF treatment, and other SSP levels including, control. However, the interaction of AMF and SSP was non-significant. These differences in wheat yield among treatments indicated that the yield of wheat was limited by N & P, confirming that the soil is deficient in N and plant-available P. The increase in wheat yield by AMF with SSP could be due to the stimulating effect of AMF inoculation and SSP levels on nutrient availability and uptake, resulting in desirable plant growth and production (Zhou et al. 2015; Zhu et al. 2018).
Our results also indicated that AMF with SSP at the rate of 90 kg ha−1 significantly enhance STN from 1529 to 1564 mg kg−1, extractable P from 3.5 to 5.2 mg kg−1, plant N concentration from 0.92 to 1.15%, plant P concentration from 0.1 to 0.3%, N uptake from 63 to 98 kg ha−1, and P uptake from 9 to 22 kg ha−1 compared to no AMF treatment, and other SSP levels including control. These results revealed that AMF stimulated the NP use efficiency and its uptake by wheat plants which ultimately increased the yield-related parameters of the wheat crop. This increase in P use efficiency and uptake in alkaline calcareous soils could be ascribed to the release of exudates and organic acids in the rhizosphere that neutralized the high pH of and increase the availability of P in the calcareous soil for plant growth (Samadi 2006; Wahid et al. 2020). In addition, the maximum uptake of NP in wheat and maize plants by the inoculation of AMF could be due to the fact that the fungi extended their hyphae beyond the plant rhizosphere and enhanced nutrients (NP) uptake (Pierre et al. 2014). In more detail, the arbuscular mycorrhizal (AM) symbiosis plays a key role in the nutrient uptake of more than 60% of land plants, including many important crop species such as wheat, maize, and soybean (Wang and Qiu 2006). There are various mechanisms that explain how AMF inoculation enhances P acquisition. Likewise, root extension mechanism, the extra radical mycelium (ERM) of the fungus acts as an extension of the root system and increases the uptake of phosphate (P) (Bolan 1991; Bücking and Kafle 2015), enhancing the adsorption from soil solution by lowering threshold concentration (Howeler et al. 1982), production of exudates which enhance the availability of P (Bethlenfalvay et al. 1997; Bolan 1991; Tawaraya and Saito 1994), changing the pH of rhizosphere due to an anion and cation adsorption by mycorrhizal plants making phosphorus readily available to plants (Lapeyrie et al. 1990), production of enzyme particularly phosphatase that solubilizes organic P enhance its availability (Tarafdar and Marschner 1994).
N uptake and nutrition can also be enhanced via AMF (McFarland et al. 2010; Meding and Zasoski 2008). The extra radical mycelium (ERM) fungal hyphae effectively acquire nitrate (NO3−) (Bago et al. 1996; Tobar et al. 1994), ammonium (NH4+) (Breuninger et al. 2004; Hawkins et al. 2000; Toussaint et al. 2004), and amino acids (AAs) from the external medium (Azcón et al. 2001; ShacharHill et al. 1997; Vázquez et al. 2001), and then transport them to the roots of the host plant. The ERM of AM fungi associated with the roots of the host plant absorbs various forms of N from the surroundings. Johansen et al. (1996) proved that G. intraradices grown in a medium containing 15NH4+ generated abundant free AA in the ERM, among which glutamate (Glu), glutamine (Gln), asparagine (Asn), aspartate, and alanine were predominantly produced and labeled with 15N. These results indicated that AM fungi can uptake and assimilate exogenous NH4+. When both NH4+ and NO3− were present in the medium, the AM fungi preferentially used the former (Toussaint et al. 2004), signifying that it is more energy-efficient for the ERM to assimilate NH4+ than NO3−. As well, these results suggested that a reduced‑N source, such as NH4+ or a downstream N metabolite, might repress NO3− influx at both the transcriptional and posttranslational levels, like in plants (Coruzzi and Zhou 2001). Nevertheless, NO3− is more mobile in soil and more readily accessible to the roots of the host plant.
The application of SSP of 90 kg ha−1 along with inoculation of AMF resulted in a 240% increase in spore density compared to control. Moreover, sole inoculation of AMF increased spore density by 166.66% compared to no AMF. This increase could be due to the strong regulation of AMF spore density in soils by seasonal climate (Silva-Flores et al. 2019). Additionally, according to the (Barea 2015), different soil microorganisms produce some chemicals that could improve the amount of root exudates, resulting in higher rates of mycorrhizal colonization. The application of SSP of 90 kg ha−1 along with inoculation of AMF resulted in a 193.75% increase in root infection intensity compared to control, while a 121.05% increase was recorded in the case of sole inoculation of AMF compared to no AMF. Our results are in line with Xie et al. (2021), who reported that AMF inoculation in the soil can improve above-ground plant growth and change root morphology.
Conclusions
AMF integrated with different levels of SSP improved wheat-NP acquisition, yield, root infection intensity, and AMF spore density in alkaline-calcareous soil compared to control. Generally, SSP at the rate of 90 kg ha−1 with AMF performed better than other SPP levels and no-inoculation treatments. Overall, we conclude that the sole application of SSP and AMF inoculation could be an excellent way to increase wheat-N‑P acquisition and yield of wheat in alkaline-calcareous soil of semi-arid areas. Further field-based research is suggested by using AMF and NPK compound fertilizers with variable rates on NPK availability, uptake, and crop performance under different agro-ecological conditions.
References
Ahanger MA, Hashem A, Abd-Allah EF, Ahmad P (2014) Arbuscular mycorrhiza in crop improvement under environmental stress. In: Emerging technologies and management of crop stress tolerance. Academic Press, pp 69–95 https://doi.org/10.1016/B978-0-12-800875-1.00003-X
Akram Z, Ajmal SU, Munir M (2008) Estimation of correlation coefficient among some yield parameters of wheat under rainfed conditions. Pak J Bot 40:1777–1781
An T, Cheng H, Qin Y, Su W, Deng H, Wu J, Liu Z, Guo X (2021) The dual mechanisms of composite biochar and biofilm towards sustainable nutrient release control of phosphate fertilizer: effect on phosphorus utilization and crop growth. J Clean Prod 311:127329. https://doi.org/10.1016/j.jclepro.2021.127329
Azcón R, Ruiz-Lozano J, Rodriguez R (2001) Differential contribution of arbuscular mycorrhizal fungi to plant nitrate uptake (15N) under increasing N supply to the soil. Can J Bot 79:1175–1180. https://doi.org/10.1139/b01-093
Aziz T, Sabir M, Farooq M, Aamer MM, Hamaad Raza A, Ahmad Warraich E (2014) Phosphorus deficiency in plants: responses, adaptive mechanisms, and signaling. In: Plant signaling: understanding the molecular crosstalk. Springer, pp 133–148 https://doi.org/10.1007/978-81-322-1542-4_7
Bago B, Vierheilig H, Piché Y, Azcón-Aguilar C (1996) Nitrate depletion and pH changes induced by the extraradical mycelium of the arbuscular mycorrhizal fungus Glomus intraradices grown in monoxenic culture. New Phytol 133:273–280. https://doi.org/10.1111/j.1469-8137.1996.tb01894.x
Barea J (2015) Future challenges and perspectives for applying microbial biotechnology in sustainable agriculture based on a better understanding of plant-microbiome interactions. J Soil Sci Plant Nutr 15:261–282. https://doi.org/10.4067/S0718-95162015005000021
Begum N, Qin C, Ahanger MA, Raza S, Khan MI, Ashraf M, Ahmed N, Zhang L (2019) Role of arbuscular mycorrhizal fungi in plant growth regulation: implications in abiotic stress tolerance. Front Plant Sci. https://doi.org/10.3389/fpls.2019.01068
Berta G, Copetta A, Gamalero E, Bona E, Cesaro P, Scarafoni A, D’Agostino G (2014) Maize development and grain quality are differentially affected by mycorrhizal fungi and a growth-promoting pseudomonad in the field. Mycorrhiza 24:161–170. https://doi.org/10.1007/s00572-013-0523-x
Bethlenfalvay GJ, Schreiner RP, Mihara KL (1997) Mycorrhizal fungi effects on nutrient composition and yield of soybean seeds. J Plant Nutr 20:581–591. https://doi.org/10.1080/01904169709365276
Bolan NS (1991) A critical review on the role of mycorrhizal fungi in the uptake of phosphorus by plants. Plant Soil 134:189–207. https://doi.org/10.1007/BF00012037
Bremner JM (1996) Nitrogen-total. In: Sparks DL, Page AL, Helmke PA, Loeppert RH, Soltanpour PN, Tabatabai MA, Johnston CT, Sumner ME (eds) Methods of soil analysis: part 3 chemical methods, 5.3, p 1085–1121. https://doi.org/10.2136/sssabookser5.3.c37
Breuninger M, Trujillo CG, Serrano E, Fischer R, Requena N (2004) Different nitrogen sources modulate activity but not expression of glutamine synthetase in arbuscular mycorrhizal fungi. Fungal Genet Biol 41:542–552. https://doi.org/10.1016/j.fgb.2004.01.003
Bücking H, Kafle A (2015) Role of arbuscular mycorrhizal fungi in the nitrogen uptake of plants: current knowledge and research gaps. Agron 5:587–612. https://doi.org/10.3390/agronomy5040587
Bush J (2008) The potential role of mycorrhizae in the growth and establishment of Juniperus seedlings. In: Western North American Juniperus Communities. Springer, pp 111–130 https://doi.org/10.1007/978-0-387-34003-6_6
Coruzzi GM, Zhou L (2001) Carbon and nitrogen sensing and signaling in plants: emerging ‘matrix effects. Curr Opin Plant Biol 4:247–253. https://doi.org/10.1016/S1369-5266(00)00168-0
Cozzolino V, Di Meo V, Piccolo A (2013) Impact of arbuscular mycorrhizal fungi applications on maize production and soil phosphorus availability. J Geochem Explor 129:40–44
Erdinc C, Durak ED, Ekincialp A, Şensoy S, Demir S (2017) Variations in response of determinate common bean (Phaseolus vulgaris L.) genotypes to arbuscular mycorrhizal fungi (AMF) inoculation. Turk J Agric For 41:1–9. https://doi.org/10.3906/tar-1609-68
FAO (2022) Management of calcareous soils. https://www.fao.org/soils-portal/soil-management/management-of-some-problem-soils/calcareous-soils/en/
Hawkins H‑J, Johansen A, George E (2000) Uptake and transport of organic and inorganic nitrogen by arbuscular mycorrhizal fungi. Plant Soil 226:275–285. https://doi.org/10.1023/A:1026500810385
Higo M, Takahashi Y, Gunji K, Isobe K (2018) How are arbuscular mycorrhizal associations related to maize growth performance during short–term cover crop rotation? J Sci Food Agric 98:1388–1396. https://doi.org/10.1002/jsfa.8606
Hinsinger P, Betencourt E, Bernard L, Brauman A, Plassard C, Shen J, Tang X, Zhang F (2011) P for two, sharing a scarce resource: soil phosphorus acquisition in the rhizosphere of intercropped species. Plant Physiol 156:1078–1086. https://doi.org/10.1104/pp.111.175331
Howeler RH, Asher C, Edwards D (1982) Establishment of an effective endomycorrhizal association on cassava in flowing solution culture and its effects on phosphorus nutrition. New Phytol 90:229–238. https://doi.org/10.1111/j.1469-8137.1982.tb03255.x
Izhar Shafi M, Adnan M, Fahad S, Wahid F, Khan A, Yue Z, Danish S, Zafar-ul-Hye M, Brtnicky M, Datta R (2020) Application of single superphosphate with humic acid improves the growth, yield and phosphorus uptake of wheat (Triticum aestivum L.) in calcareous soil. Agron 10:1224. https://doi.org/10.3390/agronomy10091224
Jan B, Ali A, Wahid F, Shah SNM, Khan A, Khan F (2014) Effect of arbuscular mycorrhiza fungal inoculation with compost on yield and phosphorous uptake of berseem in alkaline calcareous soil. American J Plant Sci 5:1359. https://doi.org/10.4236/ajps.2014.59150
Johansen A, Finlay RD, Olsson PA (1996) Nitrogen metabolism of external hyphae of the arbuscular mycorrhizal fungus Glornus intraradices. New Phytol 133:705–712. https://doi.org/10.1111/j.1469-8137.1996.tb01939.x
Karimi A, Moezzi A, Chorom M, Enayatizamir N (2020) Application of biochar changed the status of nutrients and biological activity in a calcareous soil. J Soil Sci Plant Nutr 20:450–459. https://doi.org/10.1007/s42729-019-00129-5
Khan H, Akbar WA, Shah Z, Rahim HU, Taj A, Alatalo JM (2022) Coupling phosphate-solubilizing bacteria (PSB) with inorganic phosphorus fertilizer improves mungbean (Vigna radiata) phosphorus acquisition, nitrogen fixation, and yield in alkaline-calcareous soil. Heliyon 8:e9081. https://doi.org/10.1016/j.heliyon.2022.e09081
Koske R, Gemma J (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486. https://doi.org/10.1016/S0953-7562(89)80195-9
Kuo S, Sparks D, Page A, Helmke P, Loeppert R (1996) Phosphorus. Methods of soil analysis. Part 3. Chemical methods. Soil Science Society of America, American Society of Agronomy, Madison
Lambers H (2022) Phosphorus acquisition and utilization in plants. Annu Rev Plant Biol 73:11–126. https://doi.org/10.1146/annurev-arplant-102720-125738
Lapeyrie F, Picatt C, Gerard J, Dexheimer J (1990) TEM study of intracellular and extracellular calcium oxalate accumulation by ectomycorrhizal fungi in pure culture or in association with Eucalyptus seedlings. Symbiosis 9:163–166
McFarland JW, Ruess RW, Kielland K, Pregitzer K, Hendrick R, Allen M (2010) Cross-ecosystem comparisons of in situ plant uptake of amino acid‑N and NH4. Ecosystems 13:177–193. https://doi.org/10.1007/s10021-009-9309-6
Meding S, Zasoski R (2008) Hyphal-mediated transfer of nitrate, arsenic, cesium, rubidium, and strontium between arbuscular mycorrhizal forbs and grasses from a California oak woodland. Soil Biol Biochem 40:126–134. https://doi.org/10.1016/j.soilbio.2007.07.019
Meng L, Zhang A, Wang F, Han X, Wang D, Li S (2015) Arbuscular mycorrhizal fungi and rhizobium facilitate nitrogen uptake and transfer in soybean/maize intercropping system. Front Plant Sci 6:339. https://doi.org/10.3389/fpls.2015.00339
d Oliveira JS, Ramos NP, Júnior JL, Xavier LP, Andrade EH, Mello AH, Setzer WN, Da Silva JKR (2022) Secondary metabolism and plant growth of piper divaricatum (Piperaceae) inoculated with arbuscular mycorrhizal fungi and phosphorus supplementation. Agron 12:596. https://doi.org/10.3390/agronomy12030596
Pan Y, Song Y, Zhao L, Chen P, Bu C, Liu P, Zhang D (2022) The genetic basis of phosphorus utilization efficiency in plants provide new insight into woody perennial plants improvement. Int J Mol Sci 23:2353. https://doi.org/10.3390/ijms23042353
Penn CJ, Camberato JJ (2019) A critical review on soil chemical processes that control how soil pH affects phosphorus availability to plants. Agric 9:120. https://doi.org/10.3390/agriculture9060120
Phillips JM, Hayman D (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158–IN18
Pierre MJ, Bhople BS, Kumar A, Erneste H, Emmanuel B, Singh YN (2014) Contribution of arbuscular mycorrhizal fungi (AM fungi) and rhizobium inoculation on crop growth and chemical properties of rhizospheric soils in high plants. IOSR JAVS 7:45–55
Rahim H, Mian IA, Arif M, Ahmad S, Khan Z (2020) Soil fertlity status as influenced by the carryover effect of biochar and summer legumes. Asian J Agric Biol 8:11–16. https://doi.org/10.35495/ajab.2019.05.198
Samadi A (2006) Contribution of inorganic phosphorus fractions to plant nutrition in alkaline-calcareous soils. J Agric Sci Technol 8:77–89
ShacharHill Y, Rolin D, Pfeffer P, Douds D (1997) Uptake and transfer to the host of nitrogen by an arbuscular mycorrhizal (AM) fungus. Plant Physiol 114:106–106
Silva-Flores P, Bueno CG, Neira J, Palfner G (2019) Factors affecting arbuscular mycorrhizal fungi spore density in the Chilean Mediterranean-type ecosystem. J Soil Sci Plant Nutr 19:42–50. https://doi.org/10.1007/s42729-018-0004-6
Smith SE, Read DJ (2010) Mycorrhizal symbiosis. Academic Press
Soltanpour P, Schwab A (1977) A new soil test for simultaneous extraction of macro-and micro-nutrients in alkaline soils. Commun Soil Sci Plant Anal 8:195–207. https://doi.org/10.1080/00103627709366714
Tarafdar JC, Marschner H (1994) Phosphatase activity in the rhizosphere and hyphosphere of VA mycorrhizal wheat supplied with inorganic and organic phosphorus. Soil Biol Biochem 26:387–395. https://doi.org/10.1016/0038-0717(94)90288-7
Tarraf W, Ruta C, Tagarelli A, De Cillis F, De Mastro G (2017) Influence of arbuscular mycorrhizae on plant growth, essential oil production and phosphorus uptake of Salvia officinalis L. Ind Crops Prod 102:144–153. https://doi.org/10.1016/j.indcrop.2017.03.010
Tawaraya K, Saito M (1994) Effect of vesicular-arbuscular mycorrhizal infection on amino acid composition in roots of onion and white clover. Soil Sci Plant Nutr 40:339–343. https://doi.org/10.1080/00380768.1994.10413308
Tobar R, Azcón R, Barea J (1994) Improved nitrogen uptake and transport from 15N-labelled nitrate by external hyphae of arbuscular mycorrhiza under water-stressed conditions. New Phytol 126:119–122. https://doi.org/10.1111/j.1469-8137.1994.tb07536.x
Toussaint J‑P, St-Arnaud M, Charest C (2004) Nitrogen transfer and assimilation between the arbuscular mycorrhizal fungus Glomus intraradices Schenck & Smith and Ri T‑DNA roots of Daucus carota L. in an in vitro compartmented system. Can J Microbiol 50:251–260. https://doi.org/10.1139/w04-009
Toussaint J‑P, Smith F, Smith S (2007) Arbuscular mycorrhizal fungi can induce the production of phytochemicals in sweet basil irrespective of phosphorus nutrition. Mycorrhiza 17:291–297. https://doi.org/10.1007/s00572-006-0104-3
Tshibangu Kazadi A, Baert G, Lwalaba wa Lwalaba J, Kirika Ansey B, Haesaert G, Mukobo Mundende R‑P (2020) Increasing of NPK fertilizer efficiency by arbuscular mycorrhiza in common bean (Phaseolus Vulgaris L.). Gesunde Pflanz 72:303–310. https://doi.org/10.1007/s10343-020-00513-7
Tshibangu Kazadi A, Lwalaba wa Lwalaba J, Kirika Ansey B, Mavungu Muzulukwau J, Manda Katabe G, Iband Karul M, Baert G, Haesaert G, Mukobo Mundende R‑P (2022) Effect of phosphorus and arbuscular mycorrhizal fungi (AMF) inoculation on growth and productivity of maize (Zea mays L.) in a tropical ferralsol. Gesunde Pflanz 74:159–165. https://doi.org/10.1007/s10343-021-00598-8
Vázquez M, Barea J, Azcón R (2001) Impact of soil nitrogen concentration on Glomus spp.-Sinorhizobium interactions as affecting growth, nitrate reductase activity and protein content of Medicago sativa. Biol Fertil Soils 34:57–63. https://doi.org/10.1007/s003740100373
Wahid F, Fahad S, Danish S, Adnan M, Yue Z, Saud S, Siddiqui MH, Brtnicky M, Hammerschmiedt T, Datta R (2020) Sustainable management with mycorrhizae and phosphate solubilizing bacteria for enhanced phosphorus uptake in calcareous soils. Agric 10:334. https://doi.org/10.3390/agriculture10080334
Wang B, Qiu Y‑L (2006) Phylogenetic distribution and evolution of mycorrhizas in land plants. Mycorrhiza 16:299–363. https://doi.org/10.1007/s00572-005-0033-6
Xie L, Bi Y, Ma S, Shang J, Hu Q, Christie P (2021) Combined inoculation with dark septate endophytes and arbuscular mycorrhizal fungi: synergistic or competitive growth effects on maize? BMC Plant Biol 21:1–11. https://doi.org/10.1186/s12870-021-03267-0
Zhou Q, Ravnskov S, Jiang D, Wollenweber B (2015) Changes in carbon and nitrogen allocation, growth and grain yield induced by arbuscular mycorrhizal fungi in wheat (Triticum aestivum L.) subjected to a period of water deficit. J Plant Growth Regul 75:751–760. https://doi.org/10.1007/s10725-014-9977-x
Zhu X, Song F, Liu S, Liu F, Li X (2018) Arbuscular mycorrhiza enhances nutrient accumulation in wheat exposed to elevated CO2 and soil salinity. J Plant Nutr Soil Sci 181:836–846. https://doi.org/10.1002/jpln.201700575
Acknowledgements
The authors are thankful to Agriculture Research Farm, and Department of Soil and Environmental Sciences, The University of Agriculture Peshawar Pakistan for providing financial and technical support during this research study. Moreover, we are thankful to Prof. Dr. Juha M. Alatalo for their technical comments during the write-up of this research paper.
Author information
Authors and Affiliations
Contributions
J. Ul Haq & M. Sharif: Methodology, Experiment execution, Data collection, Lab and statistical analysis, Supervision, Writing; W. A. Akbar: Writing, Statistical analysis, Discussion; I. Ahmad Mian, S. Ahmad, Z. Khan & M. Mudassir: Experiment execution; J. M. Alatalo: Review & editing; H. Ur Rahim: Conceptualization, Investigation, Statistical Analysis, Supervision, Discussion, Writing, Review & editing
Corresponding author
Ethics declarations
Conflict of interest
J. Ul Haq, M. Sharif, W.A. Akbar, H. Ur Rahim, I. Ahmad Mian, S. Ahmad, J. M. Alatalo, Z. Khan and M. Mudassir declare that they have no competing interests.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ul Haq, J., Sharif, M., Akbar, W.A. et al. Arbuscular Mycorrhiza Fungi Integrated with Single Super Phosphate Improve Wheat-nitrogen-phosphorus Acquisition, Yield, Root Infection Activity, and Spore Density in Alkaline-calcareous Soil. Gesunde Pflanzen 75, 539–548 (2023). https://doi.org/10.1007/s10343-022-00718-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-022-00718-y