Abstract
A factorial split-plot experiment (2015–17) was carried out in a randomized complete block design with three replications at Karaj, Iran, to investigate the quantitative and qualitative responses of rapeseed genotypes to the exogenous application of salicylic acid at various plant densities. Three plant densities (40, 50, and 60 plants m−2) and two levels of application of salicylic acid (non-application and foliar application) were factorial in the main plots and six rapeseed genotypes (HW118, WPN6, HL3721, L14, Tassilo, and Natali) were in sub-plots. The number of siliques per plant, number of seeds per silique, seed yield, and oil yield improved when rapeseed genotypes treated with salicylic acid. The oil quality of studied genotypes enhanced due to a reduction in erucic acid and glucosinolate contents under the foliar application of salicylic acid. On average, by foliar application of salicylic acid, the highest seed and oil yields at the density of 40 plants m−2 belonged to the L14 genotype, while the HW118 produced the maximum seed and oil yields at the densities of 50 and 60 plants m−2. Generally, rapeseed genotypes should be cultivated at the density of 40 plant m−2 and treated with foliar application of salicylic acid for achieving the maximum seed yield and oil quality.
Zusammenfassung
Ein faktorieller Split-Plot-Versuch (2015–17) wurde in einem randomisierten vollständigen Blockdesign mit drei Wiederholungen in Karaj, Iran, durchgeführt, um die quantitativen und qualitativen Reaktionen von Rapsgenotypen auf die exogene Anwendung von Salicylsäure bei verschiedenen Pflanzdichten zu untersuchen. Drei Pflanzdichten (40, 50 und 60 Pflanzen pro m2) und zwei Arten der Salicylsäureapplikation (keine Applikation und Blattapplikation) waren Faktoren in den Hauptplots, sechs Rapsgenotypen (HW118, WPN6, HL3721, L14, Tassilo und Natali) in den Subplots. Die Anzahl der Schoten pro Pflanze, die Anzahl der Samen pro Schote, der Samenertrag und der Ölertrag verbesserten sich, wenn die Rapsgenotypen mit Salicylsäure behandelt wurden. Die Ölqualität der untersuchten Genotypen verbesserte sich aufgrund einer Reduzierung des Erucasäure- und Glucosinolatgehalts unter der Blattapplikation von Salicylsäure. Im Durchschnitt erreichte bei einer Blattapplikation von Salicylsäure der Genotyp L14 bei einer Dichte von 40 Pflanzen pro m2 die höchsten Samen- und Ölerträge, während HW118 bei Dichten von 50 und 60 Pflanzen pro m2 die höchsten Samen- und Ölerträge lieferte. Generell sollten die Rapsgenotypen bei einer Dichte von 40 Pflanzen pro m2 angebaut und mit Salicylsäure behandelt werden, um den maximalen Samenertrag und die beste Ölqualität zu erreichen.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rapeseed (Brassica napus L.) has always been considered as one of the most important oilseed crops in the world (Confortin et al. 2019; Rahimi-Moghaddam et al. 2020) and it is the third major oil crop after palm and soybean. According to the latest report of the Food and Agriculture Organization (FAO) of the United Nations, total rapeseed production in the world and Iran is 75 and 0.14 million tons, respectively. This oilseed crop is mainly considered due to its high oil content (about 40–45%). Rapeseed oil is one of the most common edible oils because of low content of saturated fatty acids (SFA; 7%), high content of monounsaturated fatty acids (MUFA; 60%), and sufficient content of polyunsaturated fatty acids (PUFA; 8–12%) (Starner et al. 1999).
Identifying optimum crop-managing practices for oilseed crops such as the selection of high-yielding genotypes (Eyni-Nargeseh et al. 2020a), optimal planting date (Moradi-Aghdam et al. 2019), optimal plant density (Li et al. 2014), application of macro and micro elements (Bybordi 2016; Amiri-Darban et al. 2020), and application of plant growth regulators (PGRs) (Elhamahmy et al. 2016) can lead to greater seed yield and oil quality as well as expanding cultivated area. Plant density per unit area has the greatest effect on yield components and seed yield of individual plants (Diepenbrock 2000). Optimum plant density depends on the environment, production system, and genotype (Ozer 2003; Caliskan et al. 2009) and it should be determined by local researches. If the plant density is considered so high, it is not properly used from the production capacity of the crop, and also the competition between plants occurs which reduces total crop efficiency. Therefore, the maximum yield is obtained when all production factors are provided for optimal development and growth of the plant. Caliskan et al. (2009) in evaluating the effect of different plant densities (15, 20, 25, and 30 plants m−2) on yield and yield components of potato hybrids concluded that the optimal plant density varied between 25 and 30 plants m−2 in terms of total yield, while 20 and 25 plants m−2 were known as optimal plant densities with regard to the marketable yield depending on studied hybrids. Nasiri et al. (2017) evaluated the response of six canola cultivars (Ahmadi, Okapi, Opera, L72, Karaj1, and SW102) at three plant densities (40, 60, and 80 plants m−2) and reported that the highest seed yield was obtained at 40 plants m−2.
The exogenous application of PGRs is very useful to amend crop growth under different conditions (Elhamahmy et al. 2016; Ijaz et al. 2019). Salicylic acid (SA) or ortho-hydroxy benzoic acid as a PGR improves photosynthesis, growth, and different physiological and biochemical processes when its lower concentrations are applied within the range of 0.1 to 0.5 µM. It acts as a potent PGR that can effectively modulate different plant growth responses. Exogenous application of SA induces the systemic acquired resistance in plants, thereby provides considerable protection against different biotic stress (Hayat et al. 2010). The effects of application of salicylic acid on plant development and growth are controversial in various plant species under different environmental conditions (Janda et al. 2014). Total chlorophyll content and net photosynthesis could increase in Brassica family, leading to an increase in seed yield (Fariduddin et al. 2003; Ullah et al. 2012). Increased seed yield and oil quality of crops have been reported due to the foliar application of SA under different environmental and management conditions such as drought stress (Razmi et al. 2017), ionic toxicity stress (Metwally et al. 2018), biotic stress (Elhamahmy et al. 2016), and salt stress (Jini and Joseph 2017).
Considering the growing importance of rapeseed oil for edible and industrial purposes, its cultivated area could be increased if its seed and oil yields as well as oil characteristics are improved in quality and quantity with better management strategies. Accordingly, the current study aimed to elucidate the quantitative and qualitative responses of rapeseed genotypes to the exogenous application of salicylic acid at various plant densities.
Materials and Methods
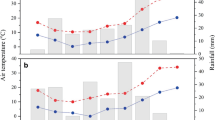
A factorial split-plot experiment (2015–16 and 2016–17) was carried out in a randomized complete block design (RCBD) with three replications at Karaj (Research Field of Seed and Plant Improvement Institute), Iran, to investigate the quantitative and qualitative responses of rapeseed genotypes to the exogenous application of salicylic acid at various plant densities. The geographical coordinates of the experimental site (Karaj) are 35°59′N latitude and 50°75′ E longitude at an altitude of 1321 m above sea level. According to long-term climatic data of 30-years (1985–2015), the average annual rainfall in the studied location is 243 mm. The meteorology data recorded over the experiment period in both years are presented in Table 1.
In this study, three plant densities of 40 (PD1), 50 (PD2), and 60 (PD3) plants m−2 and two levels of exogenous application of salicylic acid including non-application (SA0) and foliar application of 1 µM salicylic acid at two stages of six-leaf and stem elongation (SA1) were in the main plots and six rapeseed genotypes of HW118, WPN6, HL3721, L14, Tassilo, and Natali (Hybrid) were in sub-plots.
The experimental plots were 6 m long and consisted of 5 rows, with an inter-row space of 300 mm and the inter-plant spaces of 83, 67, and 55 mm on the rows to achieve the densities of 40, 50, and 60 plants m−2, respectively. A 2 m distance was kept between experimental plots. Soil samples were taken before sowing to determine physicochemical characteristics at two depths of 0–30 and 30–60 cm (Table 2). Soil texture was clay loam. Consumed fertilizers according to the recommendations and soil analysis were: (i) ammonium sulfate (150 kg ha−1), (ii) and ammonium phosphate (150 kg ha−1) at pre-plant, (iii) 350 kg ha−1 urea in three splits (100, 150, and 100 kg ha−1 in four-leaf, stem elongation, and budding stages, respectively). Irrigation was conducted equally after 80 mm evaporation from class A evaporation pan.
The final harvest was performed by harvesting four middle rows from each experimental plot at physiological maturity stage (humidity content of 14%) and weighing using a digital scale to measure the rapeseed seed yield. Number of siliques per plant and number of seeds per silique were counted from 50 randomly selected siliques. The 1000-seed weight was calculated by measuring the 10 random samples, each of which consisted of 100 seeds, from each experimental plot and multiplying it by 10, and finally expressed in 1000-seed weight.
To measure and determine the oil content of rapeseed seeds, five grams of seeds were chosen from each plot, and the oil content was estimated by Nuclear Magnetic Resonance (NMR) German Broker Brand minispec mq20 model (International standard ISO 1992). After determining the oil content of rapeseed seed, oil yield was obtained from multiplying the yield of seed by oil content.
In the present study, rapeseed seed oil was extracted according to the method described by Azadmard-Damirch et al. (2005) and Fathi-Achachlouei and Azadmard-Damirchi (2009). Briefly, rapeseed seed samples were processed with 30 ml hexane/isopropanol (3:2, v/v) at room temperature under vigorous shaking for one hour. To extract oil from 100 g of rapeseed seed, 10 tubes, each tube containing 10 g of rapeseed seed, were utilized. After shaking the samples for one hour, extracts were filtered through filter paper with a Buchner funnel under vacuum. The remaining defatted cake was washed twice with the same solution to extract the entire oil content. Then, 35 ml of sodium sulfate was added to the oil and was thoroughly mixed. After that, the organic solvent layers containing the oil were separated and evaporated at 35 °C under reduced pressure. Finally, the extracted oil was stored at −20 °C for subsequent analyzes.
Fatty acid methyl esters (FAMEs) were prepared from oil samples according to the method proposed by Savage et al. (1997). In summary, 2 ml of 0.01 M NaOH solution were added to a tube containing oil samples dissolved in 0.5 ml hexane and then were placed in a water bath at 60 °C for 10 min. After that, boron trifluoride solution in methanol (20% of BF3 in methanol) was added, and the samples were kept in a 60 °C water bath for 10 min. The sample was cooled under running water, and 2 ml of 20% sodium chloride and 1 ml of hexane were added. After complete mixing, the hexane layer containing fatty acid methyl esters was separated using centrifugation.
Fatty acid methyl esters were analyzed using gas chromatography (GC) based on the method described by Azadmard-Damirchi and Dutta (2006). The GC instrument was equipped with a flame ionization detector and a Split/Splitless injector. The injector and detector temperatures were 230 and 250 °C, respectively. The oven conditions were such that the temperature increased by 2 °C from 158 to 220 °C for over a minute and was maintained at each temperature for five minutes. Helium was applied as a carrier gas, and nitrogen was applied as an auxiliary gas at a flow rate of 30 ml/min. The FAMEs were identified through comparing their retention time with standard fatty acid methyl esters, and peak areas were reported as the percentage of total fatty acids. In the current study, palmitic, linoleic, linolenic, oleic, and erucic acid were measured.
Rapeseed seed oil glucosinolate was measured by a Varian Cary 100 Spectrophotometer equipped with a 50 m long CP-Sil 88 capillary, 0.25 mm inner diameter, and 0.2 μm static phase thickness (Harinder et al. 2007).
Finally, after verification of the test assumptions and the determination of the variances similarity in each year (based on Bartlett’s test), SAS software, version 9.2 was employed to do the combined variance analysis. It is worth noting that the year was considered a random factor. The comparison of means was conducted by the least significant difference (LSD) test at p < 0.05.
Results
The results indicated that the main effect of year was statistically significant on number of siliques per plant, 1000-seed weight, seed yield, oil yield, and palmitic acid. Accordingly, the number of siliques per plant, 1000-seed weight, seed yield, oil yield, and palmitic acid were 116.8, 3.47 g, 3343 kg ha−1, 1386 kg ha−1, and 4.06% in the first year, and were 140.6, 4.02 g, 3583 kg ha−1, 1526 kg ha−1, and 4.41% in the second year, respectively.
Combined analysis of variance showed that three-way interactions between plant density × foliar application of salicylic acid × genotype were statistically significant for all studied traits (P < 0.01) except for oleic acid (Table 3). Accordingly, the means comparison of all traits except for oleic acid were performed based on the three-way interactions of plant density × foliar application of salicylic acid × genotype. The main effect of plant density was significant on oleic acid (Table 3).
Seed Yield and Yield Components
Increasing plant density per m2 dramatically decreased rapeseed seed yield and yield components (number of siliques per plant, number of seeds per silique, and 1000-seed weight) (Table 4). On average by foliar application of salicylic acid and genotype treatments, the maximum seed yield, number of siliques per plant, number of seeds per silique, and 1000-seed weight were obtained when the rapeseed was planted at the PD1 treatment (40 plants m−2) and these traits were decreased by 33.6, 32.6, 25.7, and 25.0% at the PD2 treatment (50 plants m−2) and by 57.0, 56.2, 51.2, and 40.7% at the PD3 treatment (60 plants m−2), respectively (Table 4).
Averaged across plant density and genotype treatments, the highest seed yield, number of siliques per plant, number of seeds per silique, and 1000-seed weight were observed in the SA1 treatment (foliar application of salicylic acid) with the averages of 3577 kg ha−1, 132.0, 17.9, and 3.75 g, and decreased by 6.1, 5.3, 4.2, and 3.3% in the SA0 treatment (non-application of salicylic acid), respectively (Table 4).
There was a significant difference between studied genotypes in terms of seed yield and yield components. On average across plant density and foliar application of salicylic acid treatments, the HW118 genotype produced the highest seed yield, number of siliques per plant, number of seeds per silique, and 1000-seed weight with averages of 3941 kg ha−1, 145.8, 19.6, and 4.0 g, respectively (Table 4).
Oil Content and Yield
The results indicated that rapeseed genotypes responded differently to the plant density and foliar application of salicylic acid in terms of oil content and yield (Table 5). When averaged across foliar application of salicylic acid and genotype treatments, the highest oil content and yield were detected at the PD1 treatment with the averages of 42.9% and 2127 kg ha−1 and decreased by 2.8 and 33.5% at the PD2 treatment, and by 5.0 and 59.2% at the PD3 treatment, respectively (Table 5). The oil content and yield of rapeseed were boosted by 0.3 and 5.8% (on average across plant density and genotype) when the SA1 treatment was applied in comparison with the SA0 treatment (Table 5). The results of the mean comparison showed that the oil content and yield of rapeseed genotypes responded differently to plant density and foliar application of salicylic acid treatments so that the HW118 genotype (on average across plant density and foliar application of salicylic acid treatments) had the highest oil content and yield with the averages of 42.1% and 1667.7 kg ha−1, respectively (Table 5).
Oil Compositions
The amount of palmitic acid, linoleic acid, linolenic acid (Table 5), and oleic acid (Fig. 1) at the plant density treatments was significantly different. Mean comparison of simple effect of plant density showed that the highest oleic acid content was detected at the PD1 with an average of 62.8% and decreased by 3.02 and 5.66% at the PD2 and PD3 treatments, respectively (Fig. 1). On average, by foliar application of salicylic acid and genotype treatments, the palmitic acid and linoleic acid contents were decreased by 12.2 and 9.6% at the PD2, and by 24.7 and 17.6% at the PD3 in comparison with the PD1, respectively (Table 5). By contrast, the PD1 produced the lowest linolenic acid content (on average across foliar application of salicylic acid and genotype treatments) with an average of 4.43% and increased by 28.9 and 54.5% when rapeseeds were planted at the PD2 and PD2 treatments (Table 5).
The results showed that the rapeseed genotypes responded differently to the foliar application of salicylic acid at three plant densities in terms of oleic acid, linoleic acid, and linolenic acid contents (Table 5). When averaged across plant density and foliar application of salicylic acid treatments, HW118 genotype had the highest palmitic acid and linoleic acid contents with the averages of 4.43 and 19.75%, while the maximum linolenic acid content was observed in Tassilo genotype with an average of 5.97% (Table 5).
In comparison with PD1, the erucic acid and glucosinolate contents of rapeseed over foliar application of salicylic acid and genotype were enhanced on average by 45.4 and 40.5% at PD2, and by 82.3 and 75.6% at PD3, respectively (Table 5). A slight decrease was observed in terms of erucic acid and glucosinolate contents by those rapeseed plants treated with SA1 (0.33% and 12.2 μmolg−1, respectively) compared with SA0 treatment (0.35% and 12.6 μmolg−1, respectively) (Table 5). The response of studied genotypes was different in terms of erucic acid and glucosinolate contents. The results of means comparison indicated that HW118 genotype had the lowest erucic acid and glucosinolate contents with the averages of 0.30% and 11.24 μmolg−1 (on average across plant density and foliar application of salicylic acid treatments) (Table 5).
Discussion
The difference between two years (2015–16 and 2016–17) in terms of number of siliques per plant, 1000-seed weight, seed yield, oil yield, and palmitic acid might be attributed to the weather conditions including rainfall distribution and the mean temperature, especially at the end of the growing period (April, May, and June) in studied years (Table 2).
Seed Yield and Yield Components
Plant density is one of the most important factors affecting crops yield. Crop yields respond to increased plant density as a parabolic curve if other factors are supplied at an optimal level (Shirani Rad et al. 2011; Guo-zheng et al. 2014). Our findings clearly indicated that the amounts of seed yield and yield components substantially decreased when rapeseed genotypes were planted at higher plant densities. This might mainly be attributed to greater competition of rapeseed plants to use resources such as light, water, and nutrients at both the intra-specific and the inter-specific levels (Kuai et al. 2015). These results are similar to those of Nasiri et al. (2017) who reported that the seed yield significantly decreased when rapeseeds were planted at densities of 60 and 80 plants m−2 by 33.2 and 57.7% compared with 40 plants m−2, respectively. By contrast, Li et al. (2014) concluded that the rapeseed had a slight increase in seed yield when plant density increases from 15 to 45 plants m−2. In the current study, lodging also occurred when density was increased from 40 to 50 and 60 plants m−2, and lower seed yield and yield components were obtained under these conditions. In fact, reduction in light interception and photosynthesis in lodged plants might be possible reasons underlying rapeseed seed yield losses as well as yield components. This finding is corroborated by Khan et al. (2018) who reported that the lodging significantly decreased rapeseed seed yield, number of silique per plant, silique weight per plant, and seed weight per plant. They stated that in lodged plants, more yield loss was detected at higher densities (45 plants m−2 compared with 15 and 30 plants m−2).
The results of this research revealed that the exogenous application of salicylic acid improved the seed yield of rapeseed genotypes by increasing the number of silique per plant and the number of seeds per silique. It could be largely attributed to the beneficial effects of foliar application of salicylic acid, which promotes a wide range of metabolic and physiological functions, thereby contributing to enhanced plant productivity and growth (Askari and Ehsanzadeh 2015a). Yousefzadeh Najafabadi and Ehsanzadeh (2017a) reported that carotenoid concentration, ascorbate peroxidase activity, peroxidase activity, superoxide dismutase activity, and photosynthesis rate were increased when the sesame (Sesamum indicum L.) genotypes treated with salicylic acid, leading an increase in seed and oil yields. Moreover, the exogenous application of salicylic acid boosts crop yield by improving root properties (e.g. root length, root diameter, root dry mass, root length density, and root/shoot ratio) and yield components (Askari and Ehsanzadeh 2015b). Our findings are in agreement with those of Yousefzadeh Najafabadi and Ehsanzadeh (2017b) who found that the exogenous application of salicylic acid has a positive effect on development and growth of sesame genotypes and seed yield due mainly to increases in number of seed/m2, number of capsules/plant, and number of seeds/capsule when sesames treated with salicylic acid compared with control treatment.
Oil Content and Compositions
Although oil content and compositions are genetically determined, both are significantly influenced by environmental conditions such as water deficit stress (Eyni-Nargeseh et al. 2020b), fertilization (Amiri-Darban et al. 2020), plant density (Khan et al. 2018), exogenous application of PGRs (Ijaz et al. 2019; Estaji and Niknam 2020), and sowing date (Moradi Aghdam et al. 2019). In this study, oil content and yield negatively responded to the increasing plant density. Seed filling is a vital stage in rapeseed growth, and the oil quality, stability, composition, and content might be affected by increasing plant density as well as lodging at this stage. A tendency to decrease in rapeseed oil content may be linked with reduced silique photosynthesis (Khan et al. 2018). In fact, increasing plant density per unit area resulted in more lodging. When lodging occurs, the penetration of light in the lodged crop canopy decreases, the siliques receive low light levels, and ultimately the photosynthesis rate of siliques decreases (Khan et al. 2018). Momoh and Zhou (2001) concluded that the seed oil content of winter rapeseed was significantly decreased with the plant densities increased from 6.75 to 9.75 and 12.75 plants m−2 by 1.5 and 3.2%, respectively. By contrast, Zhang et al. (2012) reported that the oil content of rapeseed genotypes significantly enhanced with increasing plant density.
Oil quality is related with fatty acid compositions, mainly the oleic, linoleic, and linolenic acids. Photosynthetic assimilates such as sucrose are converted into different fatty acids over the lipid biosynthesis (Faraji 2014). Thus, changes in the photosynthesis function or converting carbohydrates into lipids negatively affect the fatty acids biosynthesis and accumulation (Khan et al. 2018). Our results indicated that the oleic acid, linoleic acid, and palmitic acid contents were decreased, while a slight increase were detected in linolenic acid when plant density was increased from 40 to 50 and 60 plants m−2. In this regard, Khan et al. (2018) found that erucic acid and arachidic acid contents were significantly reduced at high plant densities (30 and 45 plants m−2) compared with those at low plant densities (15 plants m−2). However, the stearic acid, palmitic acid, linoleic acid, and linolenic acid contents were not significantly affected by plant density. Ruuska et al. (2004) concluded that seeds produced by rapeseeds and soybeans grown under shaded conditions had a lower fatty acid content as compared with that in seeds of plants fully exposed to sunlight, and therefore, shading due to lodging may affect fatty acid composition. Although erucic acid and glucosinolate contents were increased at the PD2 and PD3 in comparison with the PD1, the values were within the safe limit. This finding is corroborated by Nasiri et al. (2017), who reported that the glucosinolate content was increased in rapeseed when an increase occurred at plant density.
There was a statistically significant difference between studied genotypes in terms of oil content and compositions, indicating different genetic makeup among them. Similar findings have been reported in rapeseed by Confortin et al. (2019) and Eyni-Nargeseh et al. (2020b). In the current study, the seed oil quality of treated genotypes with salicylic acid was improved through a slight reduction in erucic acid and glucosinolate contents. Genetic, management, and environmental factors could affect erucic acid and glucosinolate contents in Brassica family (Moradi Aghdam et al. 2019; Amiri-Darban et al. 2020; Ashkiani et al. 2020). In this study, erucic acid and glucosinolate contents were within the safe limit under foliar application and non-foliar application of salicylic acid.
Conclusion
When compared with the plant density of 40 plants m−2 (PD1), it was observed that the yield components, seed yield, and oil yield and content of rapeseed genotypes were considerably decreased at plant density of 50 and 60 plants m−1 (PD2 and PD3, respectively). Owing to the importance of oleic and linoleic fatty acids in improving the quality of rapeseed oil regarding edible uses high plant densities caused a reduction in oil quality by reducing these traits. A slight increase was detected in seed and oil yields of rapeseed genotypes when salicylic acid was applied in comparison with non-application treatment. Oil quality improved mainly due to a slight reduction in erucic acid and glucosinolate contents when rapeseed genotypes treated with foliar application of salicylic acid compared with non-application treatment. In general, rapeseed genotypes sown at the density of 40 plants m−2 and treated with foliar application of salicylic acid had the maximum seed and oil yields, linoleic acid, and oleic acid and the minimum erucic acid and glucosinolate contents; and this practice is recommended for achieving the maximum seed yield and oil quality.
References
Amiri-Darban N, Nourmohammadi GH, Shirani Rad AH, Mirhadi SMJ, Majidi Heravan I (2020) Potassium sulfate and ammonium sulfate affect quality and quantity of camelina oil grown with different irrigation regimes. Ind Crops Prod 148:112308
Ashkiani A, Sayfzadeh S, Shirani Rad AH, Valadabadi A, Hadidi Masouleh E (2020) Effects of foliar zinc application on yield and oil quality of rapeseed genotypes under drought stress. J Plant Nutr. https://doi.org/10.1080/01904167.2020.1739299
Askari E, Ehsanzadeh P (2015a) Drought stress mitigation by foliar application of salicylic acid and their interactive effects on physiological characteristics of fennel (Foeniculum vulgare Mill.) genotypes. Acta Physiol Plant 37:4–14
Askari E, Ehsanzadeh P (2015b) Effectiveness of exogenous salicylic acid on root and shoot growth attributes, productivity, and water use efficiency of water-deprived fennel genotypes. Hortic Environ Biotechnol 56:689–696. https://doi.org/10.1007/s13580-015-0038-9
Azadmard-Damirchi S, Dutta PC (2006) Novel solid-phase extraction method to separate 4 desmethyl-, 4‑monomethyl-, and 4, 40-dimethylsterols in vegetable oils. J Chromatogr 1108:183–187
Azadmard-Damirchi S, Savage GP, Dutta PC (2005) Sterol fractions in hazelnut and virgin olive oils and 4, 40-dimethylsterols as possible markers for detection of adulteration of virgin olive oil. J Am Oil Chem Soc 82:717–725
Bybordi A (2016) Influence of zeolite, selenium and silicon upon some agronomic and physiologic characteristics of canola grown under salinity. Comm Soil Sci Plant Anal 47:832–850
Caliskan ME, Kusman N, Caliskan S (2009) Effects of plant density on the yield and yield components of true potato seed (TPS) hybrids in early and main crop potato production systems. Field Crop Res 114:223–232
Confortin TC, Todero I, Luft L, Ugalde A, Mazutti MA et al (2019) Oil yields, protein contents, and cost of manufacturing of oil obtained from different hybrids and sowing dates of canola. J Environ Chem Eng 7(2):102972
Diepenbrock W (2000) Yield analysis of winter oilseed rape: a review. Field Crop Res 67:35–49
Elhamahmy MAM, Mahmoud MF, Bayoumi TY (2016) The effect of applying exogenous salicylic acid on aphid infection and its influence on histo-physiological traits and thermal imaging of canola. Cercet Agron Mold 166(2):67–85
Estaji A, Niknam F (2020) Foliar salicylic acid spraying effect’ on growth, seed oil content, and physiology of drought-stressed Silybum marianum L. plant. Agric Water Manag 234:106116
Eyni-Nargeseh H, Aghaalikhani M, Shirani Rad AH, Mokhtassi-Bidgoli A, Modares Sanavy SAM (2020a) Comparison of 17 rapeseed cultivars under terminal water deficit conditions using drought tolerance indices. J Agr Sci Tech 22(2):489–503
Eyni-Nargeseh H, Aghaalikhani M, Shirani Rad AH, Mokhtassi-Bidgoli A, Modares Sanavy SAM (2020b) Late season deficit irrigation for water-saving: Selection of rapeseed (Brassica napus) genotypes based on quantitative and qualitative features. Arch Agron Soil Sci 66(1):126–137
Faraji A (2014) Seed weight in canola as a function of assimilate supply and source-sink ratio during seed filling period. Int J Plant Prod 8:255–270
Fariduddin Q, Hayat S, Ahmad A (2003) Salicylic acid influences net photosynthetic rate, carboxylation efficiency, nitrate reductase activity, and seed yield in Brassica juncea. Photosynt 41:281–284
Fathi-Achachlouei F, Azadmard-Damirchi S (2009) Milk thistle seed oil constituents from different varieties grown in Iran. J Am Oil Chem Soc 86:643–649
Guo-zheng Y, Xue-jiao L, Yi-chum N, Xian-long Zh (2014) Effects of plant density on yield and canopy micro environment in hybrid cotton. J Integr Agric 13(10):2154–2163
Harinder P, Makkar S, Siddhuraju P, Becker K (2007) Plant secondary metabolites. Hummana Press, , pp 58–60
Hayat Q, Hayat S, Ifran M, Ahmad A (2010) Effect of exogenous salicylic acid under changing environment: a review. Environ Exp Bot 68:14–25
Ijaz M, Sher A, Sattar A, Shahid M, Nawaz A et al (2019) Response of canola (Brassica napus L.) to exogenous application of nitrogen, salicylic acid and gibberellic acid under an arid climate. Soil Environ 38(1):90–96
International standard ISO (1992) Oilseeds-determination of oil content-Method using continuous-wave low-resolution nuclear magnetic resonance spectrometry (Rapid method).
Janda T, Gondor OK, Yordanova R, Szalai G, Pal M (2014) Salicylic acid and photosynthesis: signalling and effects. Acta Physiol Plant 36:2537–2546. https://doi.org/10.1007/s11738-014-1620-y
Jini D, Joseph B (2017) Physiological mechanism of salicylic acid for alleviation of salt stress in rice. Rice Sci 24(2):97–108
Khan Sh, Anwar S, Kuai J, Noman A, Shahid M et al (2018) Alteration in yield and oil quality traits of winter rapeseed by lodging at different planting density and nitrogen rates. Sci Rep 8:634
Kuai J, Sun Y, Zuo Q, Huang H, Liao Q et al (2015) The yield of mechanically harvested rapeseed (Brassica napus L.) can be increased by optimum plant density and row spacing. Sci Rep 5:18835
Li YSH, Yu CHB, Zhu Sh, Xif LH, Hu XJ et al (2014) High planting density benefits to mechanized harvest and nitrogen application rates of oilseed rape (Brassica napus L.). Soil Sci Plant Nutr 60:384–392
Metwally AM, Radi AA, El-Shazoly R, Hamada AM (2018) The role of calcium, silicon and salicylic acid treatment in protection of canola plants against boron toxicity stress. J Plant Res 131(6):1015–1028
Momoh EJJ, Zhou W (2001) Growth and yield responses to plant density and stage of transplanting in winter oilseed rape (Brassica napus L.). J Agron Crop Sci 186:253–259
Moradi Aghdam A, Sayfzadeh S, Shirani Rad AH, Valadabadi SA, Zakerin HR (2019) The assessment of water stress and delay cropping on quantitative and qualitative traits of rapeseed genotypes. Ind Crop Prod 131:160–165
Nasiri A, Samdaliri M, Shirani Rad A, Shahsavari N, Mirkale AM, Jabbari H (2017) Effect of plant density on yield and physiological characteristics of six canola cultivars. J Sci Agric 1:249–253
Ozer H (2003) The effect of plant population densities on growth, yield and yield components of two spring rapeseed cultivars. Plant Soil Environ 49(9):422–426
Rahimi-Moghaddam S, Eyni-Nargeseh H, Kalantar Ahmadi SA, Azizi KH (2020) Towards withholding irrigation regimes and resistant genotypes as strategies to increase canola production in drought-prone environments: A modeling approach. Agric Water Manag 243:106487. https://doi.org/10.1016/j.agwat.2020.106487
Razmi N, Ebadi A, Daneshian J, Jahanbakhsh S (2017) Salicylic acid induced changes on antioxidant capacity, pigments and grain yield of soybean genotypes in water deficit condition. J Plant Interac 12(1):457–464
Ruuska SA, Schwender J, Ohlrogge JB (2004) Te capacity of green oilseeds to utilize photosynthesis to drive biosynthetic processes. Plant Physiol 136:2700–2709
Savage GP, Mcneil DL, Dutta PC (1997) Lipid composition and oxidative stability of oils in hazelnuts (Corylus avellana L.) grown in New Zealand. J Am Oil Chem Soc 74:755–759
Shirani Rad AH, Kavandi H, Bitarafan Z (2011) Plant density and foliar application of zn and fe effects on some quantitative and qualitative traits of german chamomile. Int J Sci Adv Tech 1(10):59–64
Starner DE, Hamama AA, Bhardwaj L (1999) Canola oil yield and quality as affected by production practices in Virginia. In: Janick J (ed) Perspectives on new crops and new uses. ASHS Press, Alexandria, pp 254–256
Ullah F, Bano A, Nosheen A (2012) Effects of plant growth regulators on growth and oil quality of canola (Brassica napus L.) under drought stress. Pak J Bot 44(6):1873–1880
Yousefzadeh Najafabadi M, Ehsanzadeh P (2017a) Photosynthetic and antioxidative upregulation in drought-stressed sesame (Sesamum indicum L.) subjected to foliar-applied salicylic acid. Photosynt 55:611–622. https://doi.org/10.1007/s11099-017-0673-8
Yousefzadeh Najafabadi M, Ehsanzadeh P (2017b) Salicylic acid effects on osmoregulation and seed yield in drought-stresses sesame. Agron J 9(4):1404–1422
Zhang Sh, Liao X, Zhang Ch, Xu H (2012) Influences of plant density on the seed yield and oil content of winter oilseed rape (Brassica napus L.). Ind Crops Prod 40:27–32
Acknowledgements
This study is a part of a Ph.D. dissertation. The authors gratefully acknowledge the support provided for this survey by the Islamic Azad University, Tehran, Iran, and Seed and Plant Improvement Institute (SPII), Karaj, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
F. Ganj-Abadi, A.H. Shirani Rad, B. Sani and H. Mozafari declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Ganj-Abadi, F., Shirani Rad, A.H., Sani, B. et al. Grain Yield and Qualitative of Rapeseed Genotypes Change in Response to Exogenous Application of Salicylic Acid and Planting Density. Gesunde Pflanzen 73, 335–344 (2021). https://doi.org/10.1007/s10343-021-00558-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-021-00558-2