Abstract
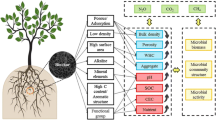
Protection and appropriate management of forests is one of the key instruments for climate change adaptation. Soil amendments with biochar have shown to be promising in achieving this goal; however, the evaluation of its long-term effects on forest soils has largely been neglected. To assess the advantages and drawbacks of biochar in forest soils, data from relict charcoal hearths (RCH) can be a potent tool as they show changes in soil properties after up to several hundred years. RCHs can be found in places of former metallurgical hot spots and their presence leaves characteristic formations identifiable on a large scale using laser detection technologies. Forest soils with biochar amendment show an increase in base cations, shift towards more alkaline pH, smaller bulk density and seem to be especially beneficial to hostile environments. Sites with favourable conditions may show little to no improvement or may even be adversely affected. Still, with proper investigation, areas with affordable feedstock materials and poor forest soils—such as spruce monocultures of Central Europe—may benefit from biochar amendments and continue to do so in the long term.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
When it comes to the development and rise of human society, there are only a few areas of the environment that can compete in terms of importance with forests. Forests have played a role as a crucial resource, providing pasture for animals, building materials, energy, food but also as places of recreational opportunities (Mayer et al. 2006; Hillring 2006). Europe has experienced both deforestation and reforestation throughout its history and compared to the Americas, where the first major deforestation event occurred in the nineteenth century (Flinn and Vellend 2005), a significant exploitation of European forests that dates several centuries before the industrial era (Kaplan et al. 2009). At current point, almost all of European forests have been to some extent affected by human activities (European Environmental Agency 2016).
To counter the degradation and decline of forests and their soils, many different approaches have been explored—afforestation, watershed management, continuous pest control and soil amendments (Fanta 1997; Wenhua 2004; Mezei et al. 2017). In the last twenty years, biochar application has gained spotlight among soil treatments, as it appears to be an affordable and relatively simple option for improving soil quality and facilitating carbon sequestration (Ennis et al. 2012). Using the definition of the International biochar initiative (IBI), biochar can be described as “a solid material obtained from the thermochemical conversion of biomass in an oxygen-limited environment” (IBI 2013). Materials used specifically for the purpose of soil application will therefore be referred to as biochars in this paper. Other materials with similar characteristics, which were not originally intended as soil amendments, will be referred to as charcoal or charcoal remains.
Rapid changes in the environment and the rise of global temperatures (Mann et al. 2016) are calling for action in reducing the carbon footprint but also in mitigating the changes already in place. Biochar is currently considered an inexpensive, safe and effective instrument for carbon sequestration (Roobroeck et al. 2019), reduction of greenhouse gases (GHG) (Senbayram et al. 2019) and improvement of soil properties (Yu et al. 2019). The longevity of biochar, however, makes it difficult for the evaluation of long-term effects on soils and natural environment, since they can remain in soils for thousands of years (Grossman et al. 2010). While many studies have been published on the topic of biochar amendments in agricultural soils, the effects on forest are much less explored. The purpose of this review is to summarize and evaluate current knowledge about the effects of biochar application on forest soils and vegetation from experiments as well as from relict charcoal hearths (RCH) and other sites historically affected by charcoal.
Charcoal hearths: past, present and future
Soils heavily influenced by human activity are by no means a modern-day phenomenon, and anthropogenic soils have even been suggested to act as markers for anthropocene (Certini and Scalenghe 2011). While contamination and degradation of soils are more prevalent and easier to identify (e.g. metal pollution from Roman times (De Vleeschouwer et al. 2007), saline water irrigation in Mesopotamia (Jacobsen & Adams 1958), etc.), positive effects on soil characteristics have also been observed. Slash and burn agriculture has been practised since early Holocene (Gerlach et al. 2012), the preservation of chernozems in Germany has been attributed to human activity (Lorz & Saile 2011) and unique plant communities have been found on soils originating from human-made aquatic shell middens (Erlandson 2013).
Still, the most prominent example of soil improvement by humans is the South American terra pretas, where relatively infertile ferralsols have been converted into nutrient and stable organic matter-rich soils suitable for agriculture. The creation of such soils is mainly attributed to the deliberate introduction of charcoal from household hearths, manure and animal remains (Sombroek et al. 2002). While terra pretas offer an important insight into the properties and effects of biochar in the long term, data obtained from them may not correlate with biochar effects on soils in other regions and climatic zones. Nevertheless, similarly promising study areas can be found on relict charcoal hearths.
Among trades connected with forests, charcoal burning holds a prominent position. In the past, charcoal was a crucial resource needed for the production of iron and it still serves as fuel in households, particularly in developing countries (Harris 1999; Bolognesi et al. 2015). Numerous historical charcoal kilns have been studied in the northeast of New England (Raab et al. 2017; Witharana et al. 2018) and other areas of the USA (Hart et al. 2008; Carter 2019). In Europe, charcoal was burned especially in the eighteenth-century metallurgical hot spots (Ludemann et al. 2004; Groenewoudt 2007) in structures known as hearths and their remains may provide information on biochar application in European conditions. Several types of hearths are known to be used, such as pits (Church et al. 2007; Deforce et al. 2018), brick or stone kilns (Straka 2017) or mounds of various size (Schindler 1872; Dragoun et al. 2006). Historic charcoal burning has been recorded throughout Europe in Italy (Carrari et al. 2016), UK (Hayman 2008), Norway (Risbøl et al. 2013), Czech Republic (Dragoun et al. 2006) and many others (Deforce et al. 2013; Hardy et al. 2016). Locations of specific interest for the study of charcoal hearths include Germany with several well-studied areas with high density of hearths in mountain areas (Knapp et al. 2015) and the Mała Panew area in Poland, where the incidence could be as high as 184 hearths per kilometre square (Rutkiewicz et al. 2019). A comprehensive study on the location and structures of charcoal hearths in central Europe has been published by Hirsch et al. (2020).
Hearths can be found as solitary landscape features or aggregated together. Permanent hearths were set up on a large, cleared area of land where several hearths could be maintained at once and which had to be easily accessible by horse carts. Temporary hearths on the other hand were smaller, located deeper in the forests and used smaller amounts of wood that was being brought from their immediate vicinity (Krištuf 2007; Raab et al. 2017). Relict charcoal hearths can be easily detected on a large scale with LiDAR (light detection and ranging) (Schmidt et al. 2016), as they form a distinctive platform with raised edges that mark the substrate used to cover the mound. Macroscopic charcoal fragments can also be observed both in the spoil material and in the soil (Carrari et al., 2016; Hirsch et al. 2017). Soils on these sites are characterized by a black layer with accumulated charcoal (see Fig. 1) which is usually distinctly separated from the lower positioned mineral horizons. The charcoal layer can range in thickness, from 10 up to almost 50 cm (Carrari et al. 2016; Mastrolonardo et al. 2018).
Comparison of RCH soil and control soil from Upper Palatinate forest in the Czech Republic with their respective horizons. The sampling site at the RCH and the control were 25 m apart. L—litter, F—fragmented, H—humus, A—organic horizon, U—anthropic (charcoal) layer, RED—compact, heat-affected layer, Bvs AND B/C—mineral horizons
Chemistry of biochars
Biochar has many characteristic chemical properties which allow it to interact with soil components and which play a role in soil improvement. These characteristics are not uniform—they change based on properties of the feedstock, burning temperature, production method, time and environmental conditions (Zhao et al. 2013; Ahmed et al. 2016). One of the most important properties of biochar is its high organic carbon content, primarily concentrated in highly stable aromatic compounds resistant to microbial decay (Xu et al. 2012). This property serves as a base for proposing biochar as a suitable carbon sequestration medium. Soils on charcoal hearths have been observed to have increased concentrations of black carbon, and larger quantities of condensed aromatic structures were also present in soils with charcoal remnants compared to reference soils which displayed long, unsaturated aliphatic chains, signalling greater ratio of fresh plant material (Abdelrahman et al. 2018).
Primary factors influencing the properties of biochar are the temperature and feedstock. Higher production temperature generally results in higher product pH, increased electrical conductivity (EC) and greater specific surface across a variety of feedstocks. Elemental composition varies greatly by individual elements—higher-temperature biochars contain higher percentage of C while O and H decrease with increasing temperature (Pariyar et al. 2020; Sun et al. 2014). N, K and P contents also vary with temperature. Data compilation by Ippolito et al. (2015) shows rise in contents of these elements up to around 500 °C before their subsequent decline at higher temperatures. Biochar characteristics are closely tied to the initial properties of the parent material and often influence the same aspects as temperature (EC, cation exchange capacity—CEC, pH, etc.). Wood biochars tend to be more stable with high C contents but low on nutrients, while biochars from manure display a reverse trend (Singh et al. 2010). The choice of material and production method are crucial for creating the right product for the desired effect during application (Weber & Quicker 2018).
Biochar’s ability to positively influence soil pH is among the reasons for employing it as a soil amendment—biochar has already been shown to have strong liming properties (Van Zwieten et al. 2010). Changes in soil pH depend strongly on the pH of used biochar, which in turn depends on the type of feedstock and to some extent also on the production temperature. While biochars are mostly pH neutral or alkaline, some types of biochar (hardwood) can be acidic, reaching values down to pH 4 (Enders et al. 2012; Mukome et al. 2013). Both active and exchangeable pH have been higher in hearth soils than control plots (Hardy et al. 2016), with the pH difference on hearth and control soils generally being more pronounced in more acidic environment (Faghih et al. 2019).
pH is inseparably linked to another crucial aspect of biochar—its CEC. High CEC of biochars is part of the motivation to use this material for experimental soil amendments (Liang et al. 2006; Chintala et al. 2014). The CEC of biochar largely depends on production methods, feedstock or temperature. Fast pyrolysis produces biochars with greater CEC than biochars created through gasification (Lee et al. 2010), and different values of CEC have also been obtained not only from biochars produced from different materials, but also from different parts of the same plant, such as with the bark and wood of Douglas fir (Pseudotsuga menziesii) (Suliman et al. 2016). Applying biochar to forest soils has a beneficial effect and increases the soil CEC significantly (Robertson et al. 2012), with relict hearth soils demonstrating it having a long-term effect (Hesson 2016; Mastrolonardo et al. 2019). In terms of pyrolysis conditions, CEC usually decreases with increasing temperature as carbonylic and carboxylic functional groups on the surface of the biochar are reduced during the pyrolysis process (Song & Guo 2012; Kloss et al. 2012).
Cation exchange capacity tends to be low freshly after burning, but increases with age (150 + years) to the point where it exceeds the CEC of non-hearth soils. On the other hand, exchangeable K+ and available P are depleted from hearth soils and may be a limitation for the establishment of plant life. This depletion is, however, only visible in the topsoil and disappears in deeper layers of the soil (Hardy et al. 2016). Criscuoli (2014) shows that these changes in P and K+ are time dependent, as fresh charcoal fragments contain significantly more P and K+ than old fragments. Mastrolonardo et al. (2019) reported a doubling in soil nitrogen contents in topsoils on relict hearths compared to reference soils as well as a significant increase in the concentration of bases in both topsoil and subsoil. CEC was higher in RCH soils. Mg concentrations were greater in the subsoil of RCHs. In a study conducted by Faghih et al. (2019), charcoal hearths had 75% increase in Ca2+ and 65% increase in Mg compared to controls. Na+ and K+ also displayed an increase, though not to the extent of Mg and Ca2+. We have observed base cation increase on RCHs in Czech Republic as shown in Fig. 2.
Hazardous compounds in biochar
While the beneficial effects and qualities of biochar are often discussed in the literature, concerns about ecotoxic characteristics of this intriguing material have not received as much spotlight. Nevertheless, biochar may aggravate the state of contamination in soils or may itself be a source of a wide range of pollutants which must be taken into account in both short- and long-term applications.
The rise of inorganic pollutants in soils—such as As, Cd, Cu, Pb and Zn—are considered a major effect of human activity on the environment. Polluted feedstock material may result in biochar unsuitable for soil application as the production process leads to increased concentration of risk elements in the final product (Hilber et al. 2017a). This can be particularly problematic with biochars originating from sewage sludges, which can contain high levels of these elements and may thus prove toxic to plants (Jamali et al. 2009). Still, despite increased total concentration of risk elements, the bioavailable fraction does not always increase and even biochar from contaminated feedstocks can still be used for application to soils (Liu et al. 2014; Figueiredo et al. 2019). As a matter of fact, biochars are commonly used for remediation of polluted soils precisely for their ability to immobilize toxic elements (Herath et al. 2015; Shen et al. 2016; Venegas et al. 2016). To ensure safety of used materials, IBI (2013) has proposed limits for risk elements in biochars.
Polycyclic aromatic hydrocarbons (PAH) are common compounds originating from the production of biochars. Just like many other biochar characteristics, the concentration of PAHs depends on feedstock and pyrolysis temperature but also on pyrolysis rate—slow pyrolysis produces biochar with lower PAH contents than fast pyrolysis (Quilliam et al. 2013). Biochars produced in controlled environments have also shown more favourable PAH concentrations than charcoal produced by more unstable methods (such as traditional kilns) (Hale et al. 2012). Hilber et al. (2017b) analysed PAH contents in various types of biochar, finding that the majority of them had very small bioavailable fraction of PAHs. Nevertheless, Schellekens et al. (2017) reported a significant increase in PAH contents in terra pretas compared to reference soils and similar findings were found in other charcoal-rich soils (Kaal et al. 2008; Marques et al. 2015). Since many biochar characteristics have been shown to change over time—sometimes quite significantly—it is safe to assume such changes might affect even biochar’s potential toxicity and contaminant retention ability, putting even greater pressure on proper choice of feedstock material and production processes.
Another hazard stemming from biochar is the presence of volatile organic compounds (VOC). In a fashion similar to PAHs, VOCs often condense on biochar surface or in the pores and are associated with specific production methods (Buss et al. 2015). Unlike PAHs, the effect of feedstock on the generation of VOCs is very variable and may not show consistent results even within the same material under the same conditions (Spokas et al. 2011). The proportions of VOCs, however, depend significantly on production temperature and are generally considered labile—this may provide an explanation as to why biochars often display negative initial effects on soils and plant life, but improve in the long term (Spokas et al. 2011; Buss et al. 2015).
Biochars with low concentrations of PAHs (conforming to biochar standards set by IBI) pose low risks to soil as the bioavailable fraction released into the environment was not statistically significant during a four-year field test. However, elevated contents of PAHs in applied biochar may result in increased concentrations in soil and pose danger to soil health (Sigmund et al. 2017). As for VOCs, low-temperature pyrolysis may result in biochar high in VOCs and may inhibit the germination of plants in amended soils (Buss and Mašek 2014). Biochar presence may also stimulate formation of VOCs through microbial decomposition of organic matter as seen in a seven-year experiment on vineyard soils (Giagnoni et al. 2019). There is considerable literature on the topic of biochar toxicity in the short term; however, long-term experiments on biochar contaminants in forests or data from RCHs are still largely absent.
Physical characteristics
While chemistry of soil tends to be the primary studied objective of soils on relict charcoal hearths, their presence also affects important physical properties. Biochar is characterized by a large amount of both micropores (< 2 nm) and macropores (> 2 nm) which are defined as the percentage of biochar volume not filled up by solids (Brewer et al. 2014). The high porosity of biochar is caused by the decomposition of lignin in the production process and by a quick release of gases, namely CH4 and H2 (Zhao et al. 2017). Biochars made from wood with lower densities tend to have greater water holding capacity and provide more available water to plants—their high-porosity stems from thinner cell walls, which upon pyrolysis create space for water intake (Werdin et al. 2020).
Many of biochar’s favourable properties come from its specific surface area (SSA), which has a direct effect on the sorption and water holding capacity in soils. Biochar applications to sandy soils have positively influenced the water regime in sandy and loam soils (Laird et al. 2010; Baiamonte et al. 2019). While fresh biochars tend to have high SSA, it is expected to be lower in naturally aged biochars due to blocking of pores with soil particles and the breakdown of natural structures (Mia et al. 2017).
Soils on RCH have an altered temperature regime compared to unaffected soils in the same conditions—in warm months, RCH soils warm up faster and reach higher temperatures in upper layers of the soil profile; however, lower layers are generally colder than their control counterparts. In cold months, this trend is reversed, with RCH soils reaching lower temperatures in upper layers and higher ones with increasing depth (Schneider et al. 2019).
The charcoal soil layers on RCHs tend to have low bulk densities; however, in contrast to control plots, deeper layers of soil may be more compacted, likely due to human activity at the site (Borchard et al. 2014; Schneider et al. 2018). Charcoal layers tend to be very heterogeneous and impact the water regimes in soil, showing preferential infiltration patterns and low infiltration rates after periods of drought, leaving some areas of soil with insufficient moisture to support proper plant growth (Schneider et al. 2018).
Plant growth and biological diversity
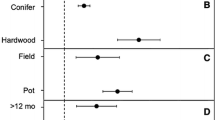
Carrari et al. (2018) observed that soils from Italian relict hearths affected survival and prosperity of tree seedlings, with higher seedling mortality on charred soils, but higher growth rate in older trees, probably due to greater nutrient (nitrogen especially) content. Tree and overstory diversity is reported to be lower at hearth sites while understory does not show significant differences after sixty years since the hearth platform was abandoned—this may be caused by slow growth and maturation of species present. Cleared areas may be welcoming for fast-growing, pioneer species (Young et al. 1996). In an analysis of tree growth by Mastrolonardo et al. (2019), two species of trees (silver birch (Betula pendula), European beech (Fagus sylvatica)) have shown smaller width of tree rings in trees growing on charcoal hearths, indicating their slower growth despite greater availability of nutrients.
According to our original findings in Upper Palatinate forest, we declare significantly greater long-term resilience of spruces from historical hearths (150 + years) towards climatic influence compared to the trees from control plots which experienced more uneven annual growth (Fig. 3). At the same time, no effect on the overall life span growth was found indicating stabilizing effect of charcoal remains on tree growth.
Buras et al. (2020) measured the diameter at breast height and total height of Scot’s pine (Pinus sylvestris) on charcoal hearths in Germany, which were significantly smaller than those of trees of the same age growing on unaffected control soils. Hearth-grown trees also displayed lower tree ring density and smaller basal area increment than their unaffected counterparts. Element counts on the other hand were reported to be significantly higher in hearth trees, namely Fe, Ca, K and Mn. The lower increase in aboveground biomass is thus mostly attributed to the aforementioned changes in water regimes in RCH soils. Unfortunately, there have been very few studies done on tree growth on RCHs.
The application of biochar in boreal areas on the other hand had a significant positive effect on Scot’s pine growth. Palviainen et al. (2020) conducted a study on nutrient poor soils in Finland, concluding that trees growing on biochar amended soils had up to 25% greater diameter increase annually compared to control soils. Similar results were achieved in a seven-year experiment at a Monterey pine (Pinus radiata) stand in Spain (Omil et al. 2013), where biochar amendments resulted in an increased tree growth in both height and diameter, with more pronounced improvements in stands with poor soil quality.
Hearths as indicators of past species composition
Human influence on the state of forests is often also reflected in species composition in a given location. Changes in tree species have already been successfully reconstructed through palynological research (Carter et al. 2017; Szabó et al. 2017), but charcoal hearths can also offer an insight into the state of vegetation in the recent past through anthracological analyses (Ludemann et al. 2004). Charcoal remains in remote areas or areas with otherwise limited wood supply represent the state of vegetation in the times of charcoal production quite accurately as the entire production depends on wood availability—with scarce resources, any wood is good enough (Ludemann 2010).
Problems may arise in areas where certain wood type is preferred over others—comparison with pollen analysis at hearths and tar pits from Białowieża primeval forest showed clear preference for the use of pine in tar production and avoidance of spruce in charcoal burning (Samojlik et al. 2013). Preference of certain tree species may result in an initial overexploitation of the favoured (usually hardwood) species and subsequent shift towards species which grow faster and are more sustainable for charcoal production (Rutkiewicz and Malik 2019). In this manner, whole landscapes can be transformed, as is the case of the Harz mountains, where what was assumed to be a primal spruce forest was originally likely a mixed forest altered by intense iron industry (Knapp et al. 2015).
Biochar and microbial activity
Biochar provides a new environment for colonization by bacteria and other soil biota. Soil microbial respiration is a factor that may aid in evaluation of biochar addition to forest soils, as it has been frequently used to assess microbial activity. Studies such as those conducted by Sackett et al. (2015) or Noyce et al. (2015) have shown that the rate of microbial respiration may not be significantly changed after biochar application in the short term, but may be more dependent on soil pH. Other studies have recorded increase in soil respiration, connected to the increase of soil water and soil temperature as a result of biochar addition (Lu et al. 2014; Zhang et al. 2019). In a meta-analysis of Chinese soils by Zhou et al. (2017), microbial respiration was significantly greater for biochar-treated temperate forests, but forests in the subtropical zone were not significantly affected. Experiments on soil respiration on relict charcoal hearths in Iran have shown no significant difference between control and hearth soils (Faghih et al. 2019), and Hardy et al. (2019) on the other hand did not dispute aged charcoal influence of soil respiration, but did not find evidence for major effect on RCH soils.
Lasota et al. (2021) investigated the effect of RCHs on microbial diversity under a Scot’s pine cover in Poland. Their research showed increase in both bacterial and fungal diversity on RCHs compared to control soils, with species richness calculated at 238 and 218 in RCHs and 150 in controls. The activity of enzymes contributing to the processing of P, C and N was also higher in RCHs. Similar results were reported by Zhou et al. (2020) in experimental application of biochar from the wood and leaves of Chinese fir (C. lanceolata) to soil collected from a plantation of trees of the same species. Soil microbial diversity was higher in soils with added biochar, and the increase in abundance of P-solubilizing species of bacteria was observed as well. Mitchell et al. (2015) further showed the change towards gram-positive dominated communities in biochar amended forest soils compared to more gram-negative communities in controls. The authors suggest a shift in microbial communities towards species with greater potential in the decomposition of labile organic matter.
In a study by Gießelmann et al. (2019), a significantly lower amount of nematodes was observed in the litter of RCH plots compared to controls; however, RCHs had a greater number of fungivorous nematodes which implies greater promotion of fungal growth on charcoal. Similar shift towards fungi-dominated communities has also been observed in the short term on grassland soils amended by biochar (Gao and DeLuca 2020). Forest soils on RCHs examined by Hardy et al. (2019) have displayed significantly altered communities of gram-positive bacteria, general bacteria and actinomycetes, attributing this change mostly to biochar-related pH changes, nutrient availability and colonization of biochar pore space. Microbial carbon use efficiency was observed to be significantly higher in soils amended by biochar than in soils without its application. This, however, has only been true for artificially aged (15% H2O2) biochar which also decreased the biomass turnover time (Pei et al. 2020).
The activity and microbial communities of biochar amended soils are a complicated issue which shows great variability with treatment, age and geographical location and would ideally require a proper study of its own.
Future for European forests
Maintaining healthy, diverse forests can be an arduous task, especially since many natural European forests have been replaced with spruce monocultures (Klimo et al. 2000; Felton et al. 2010). Longevity of European forest capitalization put a large proportion of continental forests into an uncertain position when coping with current climate–environmental change (Lindner et al. 2008). Internally caused factors such as preference of fast growing species has led into the significant decrease in the forest diversity and resilience which in turn leads to carbon source scenario (DeSoto et al. 2020). Although the ultimate goal of European forest management is defined as sustainable use of resources, accomplishment of this paradigm has failed in the context of large-scale waste of trees. On the other hand, knowledge gained within centuries of managing European forests, extensive European forest landcover as well as increasing public support for forest cultivation may facilitate the effective, relatively inexpensive and safe transition into the future where forests are part of the solution.
In order to achieve such transformation, both adaptation and mitigation potential must be well understood and incentivized. Among the means which have the potential to tackle both, the application of biochar into forest soils stands out (Stavi 2013). Historical motivations for biochar production mostly disappeared; nonetheless, there is still a vast amount of forest residue biomass which can be converted into biochar without significantly compromising traditional forest production functions (Lehmann et al. 2006). Moreover, in the current context when the logistic costs of harvesting are close to timber market price, the resources of European feedstock become immense. Carbon capture initiatives are a part of green policies globally, many of the studied technologies still facing the industrial, legislative, societal or ethical barriers (Bäckstrand et al. 2011). This is not the case for biochar production and application which is believed to be an inexpensive and safe way to capture carbon and promote future resilient forest growth, water retention and biodiversity at the same time (Thomas and Gale 2015).
Conclusion
The provided evidence suggests that while biochar amendment is not always a universal solution for a healthy forest, it is undoubtedly beneficial to trees growing on poor soils and in harsh environments. This can be especially valuable for production forests where biochar application could not only stabilize plots with unfavourable conditions but also protect them from environmental changes in the future. Selecting the right material and production method for biochar should also be taken into account from the perspective of both economy and safety to ensure optimal solution for every target region.
Despite the growing amount of information gathered from biochar experiments, our knowledge about application of biochar to forest soils has barely scratched the surface. Current data suggests that biochar effects depend on many variables, stemming both from the properties of the biochar and from the soil conditions in studied areas. While RCHs provide a valuable insight into the long-term effects of this amendment, there are not many studies to allow for definite conclusions and proper comparison between sites. An in-depth exploration into this topic could lead to healthier practices in forest management and help create a stable environment despite the ever-present threat of global climatic change.
References
Abdelrahman H, Hofmann D, Berns AE, Meyer N, Bol R, Borchard N (2018) Historical charcoal additions alter water extractable, particulate and bulk soil C composition and stabilization. J Plant Nutr Soil Sci 181:809–817. https://doi.org/10.1002/jpln.201800261
Ahmed MB, Zhou JL, Ngo HH, Guo W (2016) Insight into biochar properties and its cost analysis. Biomass Bioenergy 84:76–86. https://doi.org/10.1016/j.biombioe.2015.11.002
Bäckstrand K, Meadowcroft J, Oppenheimer M (2011) The politics and policy of carbon capture and storage: framing an emergent technology. Glob Environ Change 21:275–281. https://doi.org/10.1016/j.gloenvcha.2011.03.008
Baiamonte G, Crescimanno G, Parrino F, De Pasquale C (2019) Effect of biochar on the physical and structural properties of a sandy soil. CATENA 175:294–303. https://doi.org/10.1016/j.catena.2018.12.019
Bolognesi M, Vrieling A, Rembold F, Gadain H (2015) Rapid mapping and impact estimation of illegal charcoal production in southern Somalia based on WorldView-1 imagery. Energy Sustain Dev 25:40–49. https://doi.org/10.1016/j.esd.2014.12.008
Borchard N, Ladd B, Eschemann S, Hegenberg D, Möseler BM, Amelung W (2014) Black carbon and soil properties at historical charcoal production sites in Germany. Geoderma 232–234:236–242. https://doi.org/10.1016/j.geoderma.2014.05.007
Brewer CE, Chuang VJ, Masiello CA, Gonnermann H, Gao X, Dugan B, Driver LE, Panzacchi P, Zygourakis K, Davies CA (2014) New approaches to measuring biochar density and porosity. Biomass Bioenergy 66:176–185. https://doi.org/10.1016/j.biombioe.2014.03.059
Buras A, Hirsch F, Schneider A, Scharnweber T, van der Maaten E, Cruz-García R, Raab T, Wilmking M (2020) Reduced above-ground growth and wood density but increased wood chemical concentrations of Scots pine on relict charcoal hearths. Sci Total Environ 717:137189. https://doi.org/10.1016/j.scitotenv.2020.137189
Buss W, Mašek O (2014) Mobile organic compounds in biochar-a potential source of contamination-phytotoxic effects on cress seed (Lepidium sativum) germination. J Environ Manage 137:111–119. https://doi.org/10.1016/j.jenvman.2014.01.045
Buss W, Mašek O, Graham M, Wüst D (2015) Inherent organic compounds in biochar–their content, composition and potential toxic effects. J Environ Manage 156:150–157. https://doi.org/10.1016/j.jenvman.2015.03.035
Carrari E, Ampoorter E, Verheyen K, Coppi A, Selvi F (2016) Former charcoal platforms in Mediterranean forest areas a hostile microhabitat for the recolonization by woody species. iFor Biogeosci for 10:136. https://doi.org/10.3832/ifor1701-009
Carrari E, Ampoorter E, Bussotti F, Coppi A, Garcia Nogales A, Pollastrini M, Verheyen K, Selvi F (2018) Effects of charcoal hearth soil on forest regeneration: evidence from a two-year experiment on tree seedlings. For Ecol Manage 427:37–44. https://doi.org/10.1016/j.foreco.2018.05.038
Carter BP (2019) Identifying landscape modification using open data and tools: the charcoal hearths of the Blue Mountain, Pennsylvania. Hist Archaeol 53:432–443. https://doi.org/10.1007/s41636-019-00171-1
Carter VA, Chiverrell RC, Clear JL, Kuosmanen N, Moravcová A, Svoboda M, Svobodová-Svitavská H, van Leeuwen JFN, van der Knaap WO, Kuneš P (2017) Quantitative palynology informing conservation ecology in the Bohemian/Bavarian Forests of Central Europe. Front Plant Sci 8:2268. https://doi.org/10.3389/fpls.2017.02268
Certini G, Scalenghe R (2011) Anthropogenic soils are the golden spikes for the Anthropocene. Holocene 21:1269–1274. https://doi.org/10.1177/0959683611408454
Chintala R, Mollinedo J, Schumacher TE, Malo DD, Julson JL (2014) Effect of biochar on chemical properties of acidic soil. Arch Agron Soil Sci 60:393–404. https://doi.org/10.1080/03650340.2013.789870
Church MJ, Dugmore AJ, Mairs KA, Millard AR, Cook GT, Sveinbjarnardóttir G, Ascough PA, Roucoux KH (2007) Charcoal production during the norse and early medieval periods in Eyjafjallahreppur, Southern Iceland. Radiocarbon 49:659–672. https://doi.org/10.1017/S0033822200042557
Criscuoli I, Alberti G, Baronti S, Favilli F, Martinez C, Calzolari C, Pusceddu E, Rumpel C, Viola R, Miglietta F (2014) Carbon sequestration and fertility after centennial time scale incorporation of charcoal into soil. PLoS ONE 9:e91114. https://doi.org/10.1371/journal.pone.0091114
de Figueiredo CC, Chagas JKM, da Silva J, Paz-Ferreiro J (2019) Short-term effects of a sewage sludge biochar amendment on total and available heavy metal content of a tropical soil. Geoderma 344:31–39. https://doi.org/10.1016/j.geoderma.2019.01.052
De Vleeschouwer F, Gérard L, Goormaghtigh C, Mattielli N, Le Roux G, Fagel N (2007) Atmospheric lead and heavy metal pollution records from a Belgian peat bog spanning the last two millenia: human impact on a regional to global scale. Sci Total Environ 377:282–295. https://doi.org/10.1016/j.scitotenv.2007.02.017
Deforce K, Boeren I, Adriaenssens S, Bastiaens J, De Keersmaeker L, Haneca K, Tys D, Vandekerkhove K (2013) Selective woodland exploitation for charcoal production. A detailed analysis of charcoal kiln remains (ca. 1300–1900 AD) from Zoersel (northern Belgium). J Archaeol Sci 40:681–689. https://doi.org/10.1016/j.jas.2012.07.009
Deforce K, Vanmontfort B, Vandekerkhove K (2018) Early and high medieval (c. 650 AD–1250 AD) charcoal production and its impact on woodland composition in the Northwest-European Lowland: a study of charcoal pit Kilns from Sterrebeek (Central Belgium). Environ Archaeol. https://doi.org/10.1080/14614103.2018.1538087
DeSoto L, Cailleret M, Sterck F, Jansen S, Kramer K, Robert EMR, Aakala T, Amoroso MM, Bigler C, Camarero JJ, Čufar K, Gea-Izquierdo G, Gillner S, Haavik LJ, Hereş A-M, Kane JM, Kharuk VI, Kitzberger T, Klein T, Levanič T, Linares JC, Mäkinen H, Oberhuber W, Papadopoulos A, Rohner B, Sangüesa-Barreda G, Stojanovic DB, Suárez ML, Villalba R, Martínez-Vilalta J (2020) Low growth resilience to drought is related to future mortality risk in trees. Nat Commun 11:545. https://doi.org/10.1038/s41467-020-14300-5
Dragoun B, Matoušek V, Woitsch J (2006) A contribution towards learning about “Forest Industries.” EuroREA 3:83–87
Enders A, Hanley K, Whitman T, Joseph S, Lehmann J (2012) Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour Technol 114:644–653. https://doi.org/10.1016/j.biortech.2012.03.022
Ennis CJ, Garry Evans A, Islam M, Komang Ralebitso-Senior T, Senior E (2012) Biochar: carbon sequestration, land remediation, and impacts on soil microbiology. Crit Rev Environ Sci Technol 42:2311–2364
Erlandson JM (2013) Shell middens and other anthropogenic soils as global stratigraphic signatures of the Anthropocene. Anthropocene 4:24–32. https://doi.org/10.1016/j.ancene.2013.12.001
European Environmental Agency (2016) European forest ecosystems: state and trends. Publications Office of the European Union, Luxembourg
Faghih F, Emadi M, Sadegh-Zadeh F, Bahmanyar MA (2019) Long-term charcoal-induced changes to soil properties in temperate regions of northern Iran. Res J for 30:1063–1071. https://doi.org/10.1007/s11676-018-0641-6
Fanta J (1997) Rehabilitating degraded forests in Central Europe into self–sustaining forest ecosystems. Ecol Eng 8:289–297. https://doi.org/10.1016/S0925-8574(97)00024-4
Felton A, Lindbladh M, Brunet J, Fritz Ö (2010) Replacing coniferous monocultures with mixed-species production stands: an assessment of the potential benefits for forest biodiversity in northern Europe. For Ecol Manage 260:939–947. https://doi.org/10.1016/j.foreco.2010.06.011
Flinn KM, Vellend M (2005) Recovery of forest plant communities in post-agricultural landscapes. Front Ecol Environ. https://doi.org/10.1890/1540-9295(2005)003[0243:ROFPCI]2.0.CO;2
Gao S, DeLuca TH (2020) Biochar alters nitrogen and phosphorus dynamics in a western rangeland ecosystem. Soil Biol Biochem 148:107868. https://doi.org/10.1016/j.soilbio.2020.107868
Gerlach R, Fischer P, Eckmeier E, Hilgers A (2012) Buried dark soil horizons and archaeological features in the Neolithic settlement region of the Lower Rhine area, NW Germany: formation, geochemistry and chronostratigraphy. Quat Int 265:191–204. https://doi.org/10.1016/j.quaint.2011.10.007
Giagnoni L, Maienza A, Baronti S, Vaccari FP, Genesio L, Taiti C, Martellini T, Scodellini R, Cincinelli A, Costa C, Mancuso S, Renella G (2019) Long-term soil biological fertility, volatile organic compounds and chemical properties in a vineyard soil after biochar amendment. Geoderma 344:127–136. https://doi.org/10.1016/j.geoderma.2019.03.011
Gießelmann UC, Borchard N, Traunspurger W, Witte K (2019) Long-term effects of charcoal on nematodes and other soil meso- and microfaunal groups at historical kiln-sites–a pilot study. Eur J Soil Biol 93:103095. https://doi.org/10.1016/j.ejsobi.2019.103095
Groenewoudt B (2007) Charcoal burning and landscape dynamics in the early medieval Netherlands. In: Klápšte J, Sommer P (eds) Arts and Crafts in Medieval Rural Environment. Brepols Publishers, pp 327–337
Grossman JM, O’Neill BE, Tsai SM, Liang B, Neves E, Lehmann J, Thies JE (2010) Amazonian anthrosols support similar microbial communities that differ distinctly from those extant in adjacent, unmodified soils of the same mineralogy. Microb Ecol 60:192–205
Hale SE, Lehmann J, Rutherford D, Zimmerman AR, Bachmann RT, Shitumbanuma V, O’Toole A, Sundqvist KL, Arp HPH, Cornelissen G (2012) Quantifying the total and bioavailable polycyclic aromatic hydrocarbons and dioxins in biochars. Environ Sci Technol 46:2830–2838. https://doi.org/10.1021/es203984k
Hardy B, Cornelis J-T, Houben D, Lambert R, Dufey JE (2016) The effect of pre-industrial charcoal kilns on chemical properties of forest soil of Wallonia, Belgium. Eur J Soil Sci 67:206–216
Hardy B, Sleutel S, Dufey JE, Cornelis J-T (2019) The long-term effect of biochar on soil microbial abundance, activity and community structure is overwritten by land management. Front Environ Sci Eng China 7:110. https://doi.org/10.3389/fenvs.2019.00110
Harris P (1999) On charcoal. Interdisc Sci Rev 24:301–306
Hart JL, Van de Gevel SL, Mann DF, Clatterbuck WK (2008) Legacy of charcoaling in a western highland rim forest in Tennessee. Am Midland Nat 159:238–250. https://doi.org/10.1674/0003-0031(2008)159[238:LOCIAW]2.0.CO;2
Hayman R (2008) Charcoal ironmaking in nineteenth-century Shropshire. Econ Hist Rev 61:80–98. https://doi.org/10.1111/j.1468-0289.2007.00387.x
Herath I, Kumarathilaka P, Navaratne A, Rajakaruna N, Vithanage M (2015) Immobilization and phytotoxicity reduction of heavy metals in serpentine soil using biochar. J Soils Sediments 15:126–138. https://doi.org/10.1007/s11368-014-0967-4
Hesson K (2016) Charcoal hearth soils: remnants of the iron industry in the Northeastern U.S. Natl Sci Educ. https://doi.org/10.4195/nse2016.02.0771
Hilber I, Bastos AC, Loureiro S, Soja G, Marsz A, Cornelissen G, Bucheli TD (2017a) The different faces of biochar: contamination risk versus remediation tool. J Environ Eng Landsc Manage 25:86–104. https://doi.org/10.3846/16486897.2016.1254089
Hilber I, Mayer P, Gouliarmou V, Hale SE, Cornelissen G, Schmidt H-P, Bucheli TD (2017b) Bioavailability and bioaccessibility of polycyclic aromatic hydrocarbons from (post-pyrolytically treated) biochars. Chemosphere 174:700–707. https://doi.org/10.1016/j.chemosphere.2017.02.014
Hillring B (2006) World trade in forest products and wood fuel. Biomass Bioenergy 30:815–825. https://doi.org/10.1016/j.biombioe.2006.04.002
Hirsch F, Raab T, Ouimet W, Dethier D, Schneider A, Raab A (2017) Soils on historic charcoal hearths: terminology and chemical properties. Soil Sci Soc Am J 81:1427–1435
Hirsch F, Schneider A, Bonhage A, Raab A, Drohan PJ, Raab T (2020) An initiative for a morphologic-genetic catalog of relict charcoal hearths from Central Europe. Geoarchaeology. https://doi.org/10.1002/gea.21799
IBI (2013) Standardized product definition and product testing guidelines for biochar that is used in soil: product definition and specification standards
Ippolito JA, Spokas KA, Novak JM, Lentz RD, Cantrell KB (2015) Biochar elemental composition and factors infl uencing nutrient retention. In: Biochar for environmental management. Routledge, pp 171–196
Jacobsen T, Adams RM (1958) Salt and silt in ancient mesopotamian agriculture. Science 128:1251–1258
Jamali MK, Kazi TG, Arain MB, Afridi HI, Jalbani N, Kandhro GA, Shah AQ, Baig JA (2009) Heavy metal accumulation in different varieties of wheat (Triticum aestivum L.) grown in soil amended with domestic sewage sludge. J Hazard Mater 164:1386–1391. https://doi.org/10.1016/j.jhazmat.2008.09.056
Kaal J, Martínez-Cortizas A, Buurman P, Boado FC (2008) 8000 yr of black carbon accumulation in a colluvial soil from NW Spain. Quat Res 69:56–61. https://doi.org/10.1016/j.yqres.2007.10.005
Kaplan JO, Krumhardt KM, Zimmermann N (2009) The prehistoric and preindustrial deforestation of Europe. Quat Sci Rev 28:3016–3034. https://doi.org/10.1016/j.quascirev.2009.09.028
Klimo E, Hager H, Kulhavý J (2000) Spruce Monocultures in Central Europe: Problems and Prospects. https://www.researchgate.net/profile/Heinrich-Spiecker/publication/285638409_Growth_of_Norway_spruce_Picea_abies_L_Karst_under_changing_environmental_conditions_in_Europe/links/5e5279cb92851c7f7f512e20/Growth-of-Norway-spruce-Picea-abies-L-Karst-under-changing-environmental-conditions-in-Europe.pdf. Accessed 3 Feb 2021
Kloss S, Zehetner F, Dellantonio A, Hamid R, Ottner F, Liedtke V, Schwanninger M, Gerzabek MH, Soja G (2012) Characterization of slow pyrolysis biochars: effects of feedstocks and pyrolysis temperature on biochar properties. J Environ Qual 41:990–1000. https://doi.org/10.2134/jeq2011.0070
Knapp H, Nelle O, Kirleis W (2015) Charcoal usage in medieval and modern times in the Harz Mountains Area, Central Germany: wood selection and fast overexploitation of the woodlands. Quat Int 366:51–69. https://doi.org/10.1016/j.quaint.2015.01.053
Krištuf P (2007) Průzkum Křivoklátské vrchoviny. Opomíjená archeologie 2005-2006
Laird DA, Fleming P, Davis DD, Horton R, Wang B, Karlen DL (2010) Impact of biochar amendments on the quality of a typical Midwestern agricultural soil. Geoderma 158:443–449. https://doi.org/10.1016/j.geoderma.2010.05.013
Lasota J, Błońska E, Babiak T, Piaszczyk W, Stępniewska H, Jankowiak R, Boroń P, Lenart-Boroń A (2021) Effect of charcoal on the properties, enzyme activities and microbial diversity of temperate pine forest soils. For Trees Livelihoods 12:1488. https://doi.org/10.3390/f12111488
Lee JW, Kidder M, Evans BR, Paik S, Buchanan AC 3rd, Garten CT, Brown RC (2010) Characterization of biochars produced from cornstovers for soil amendment. Environ Sci Technol 44:7970–7974. https://doi.org/10.1021/es101337x
Lehmann J, Gaunt J, Rondon M (2006) Bio-char sequestration in terrestrial ecosystems–a review. Mitig Adapt Strat Glob Change 11:403–427. https://doi.org/10.1007/s11027-005-9006-5
Liang B, Lehmann J, Solomon D, Kinyangi J, Grossman J, O’Neill B, Skjemstad JO, Thies J, Luizão FJ, Petersen J, Neves EG (2006) Black carbon increases cation exchange capacity in soils. Soil Sci Soc Am J 70:1719–1730. https://doi.org/10.2136/sssaj2005.0383
Lindner M, Garcia-Gonzalo J, Kolstrom M, Green T, Reguera R, Maroschek M, Seidl R, Lexer MJ, Netherer S, Schopf A, Others (2008) Impacts of climate change on European forests and options for adaptation
Liu T, Liu B, Zhang W (2014) Nutrients and heavy metals in biochar produced by sewage sludge pyrolysis: its application in soil amendment. Pol J Environ Stud 23:271–275
Lorz C, Saile T (2011) Anthropogenic pedogenesis of Chernozems in Germany?–a critical review. Quat Int 243:273–279. https://doi.org/10.1016/j.quaint.2010.11.022
Lu N, Liu X-R, Du Z-L, Wang Y-D, Zhang Q-Z (2014) Effect of biochar on soil respiration in the maize growing season after 5 years of consecutive application. Soil Res 52:505–512. https://doi.org/10.1071/SR13239
Ludemann T (2010) Past fuel wood exploitation and natural forest vegetation in the Black Forest, the Vosges and neighbouring regions in western Central Europe. Palaeogeogr Palaeoclimatol Palaeoecol 291:154–165. https://doi.org/10.1016/j.palaeo.2009.09.013
Ludemann T, Michiels H-G, Nölken W (2004) Spatial patterns of past wood exploitation, natural wood supply and growth conditions: indications of natural tree species distribution by anthracological studies of charcoal-burning remains. Eur J for Res 123:283–292. https://doi.org/10.1007/s10342-004-0049-z
Mann ME, Rahmstorf S, Steinman BA, Tingley M, Miller SK (2016) The likelihood of recent record warmth. Sci Rep 6:19831. https://doi.org/10.1038/srep19831
Marques FA, Buurman P, Schellekens J, Vidal-Torrado P (2015) Molecular chemistry in humic Ferralsols from Brazilian Cerrado and forest biomes indicates a major contribution from black carbon in the subsoil. J Anal Appl Pyrolysis 113:518–528. https://doi.org/10.1016/j.jaap.2015.03.016
Mastrolonardo G, Francioso O, Certini G (2018) Relic charcoal hearth soils: a neglected carbon reservoir. Case study at Marsiliana forest, Central Italy. Geoderma 315:88–95. https://doi.org/10.1016/j.geoderma.2017.11.036
Mastrolonardo G, Calderaro C, Cocozza C, Hardy B, Dufey J, Cornelis J-T (2019) Long-term effect of charcoal accumulation in hearth soils on tree growth and nutrient cycling. Front Environ Sci Eng China 7:51. https://doi.org/10.3389/fenvs.2019.00051
Mayer AC, Stöckli V, Konold W, Kreuzer M (2006) Influence of cattle stocking rate on browsing of norway spruce in subalpine wood pastures. Agrofor Syst 66:143–149. https://doi.org/10.1007/s10457-005-5460-z
Mezei P, Blaženec M, Grodzki W, Škvarenina J, Jakuš R (2017) Influence of different forest protection strategies on spruce tree mortality during a bark beetle outbreak. Ann for Sci 74:65. https://doi.org/10.1007/s13595-017-0663-9
Mia S, Dijkstra FA, Singh B (2017) Chapter one-long-term aging of biochar: a molecular understanding with agricultural and environmental implications. In: Sparks DL (ed) Advances in Agronomy. Academic Press, pp 1–51
Mitchell PJ, Simpson AJ, Soong R, Simpson MJ (2015) Shifts in microbial community and water-extractable organic matter composition with biochar amendment in a temperate forest soil. Soil Biol Biochem 81:244–254. https://doi.org/10.1016/j.soilbio.2014.11.017
Mukome FND, Zhang X, Silva LCR, Six J, Parikh SJ (2013) Use of chemical and physical characteristics to investigate trends in biochar feedstocks. J Agric Food Chem 61:2196–2204. https://doi.org/10.1021/jf3049142
Noyce GL, Basiliko N, Fulthorpe R, Sackett TE, Thomas SC (2015) Soil microbial responses over 2 years following biochar addition to a north temperate forest. Biol Fertil Soils 51:649–659. https://doi.org/10.1007/s00374-015-1010-7
Omil B, Piñeiro V, Merino A (2013) Soil and tree responses to the application of wood ash containing charcoal in two soils with contrasting properties. For Ecol Manage 295:199–212. https://doi.org/10.1016/j.foreco.2013.01.024
Palviainen M, Aaltonen H, Laurén A, Köster K, Berninger F, Ojala A, Pumpanen J (2020) Biochar amendment increases tree growth in nutrient-poor, young Scots pine stands in Finland. For Ecol Manage 474:118362. https://doi.org/10.1016/j.foreco.2020.118362
Pariyar P, Kumari K, Jain MK, Jadhao PS (2020) Evaluation of change in biochar properties derived from different feedstock and pyrolysis temperature for environmental and agricultural application. Sci Total Environ 713:136433. https://doi.org/10.1016/j.scitotenv.2019.136433
Pei J, Li J, Mia S, Singh B, Wu J, Dijkstra FA (2020) Biochar aging increased microbial carbon use efficiency but decreased biomass turnover time. Geoderma 382:114710. https://doi.org/10.1016/j.geoderma.2020.114710
Quilliam RS, Rangecroft S, Emmett BA, Deluca TH, Jones DL (2013) Is biochar a source or sink for polycyclic aromatic hydrocarbon (PAH) compounds in agricultural soils? Glob Change Biol Bioenergy 5:96–103. https://doi.org/10.1111/gcbb.12007
Raab T, Hirsch F, Ouimet W, Johnson KM, Dethier D, Raab A (2017) Architecture of relict charcoal hearths in northwestern Connecticut, USA. Geoarchaeology 32:502–510. https://doi.org/10.1002/gea.21614
Risbøl O, Bollandsås OM, Nesbakken A, Ørka HO, Næsset E, Gobakken T (2013) Interpreting cultural remains in airborne laser scanning generated digital terrain models: effects of size and shape on detection success rates. J Archaeol Sci 40:4688–4700. https://doi.org/10.1016/j.jas.2013.07.002
Robertson SJ, Rutherford PM, López-Gutiérrez JC, Massicotte HB (2012) Biochar enhances seedling growth and alters root symbioses and properties of sub-boreal forest soils. Can J Soil Sci 92:329–340. https://doi.org/10.4141/cjss2011-066
Roobroeck D, Hood-Nowotny R, Nakubulwa D, Tumuhairwe J, Mwanjalolo MJG, Ndawula I, Vanlauwe B (2019) Biophysical potential of crop residues for biochar carbon sequestration, and co-benefits, in Uganda. Ecol Appl 29:1. https://doi.org/10.1002/eap.1984
Rutkiewicz P, Malik I (2019) Environmental effects of historical charcoal burning associated with water-powered ferrous metallurgy (Mała Panew basin, southern Poland). Geol Geophys Environ. https://doi.org/10.7494/geol.2019.45.3.231
Rutkiewicz P, Malik I, Wistuba M, Osika A (2019) High concentration of charcoal hearth remains as legacy of historical ferrous metallurgy in southern Poland. Quat Int 512:133–143. https://doi.org/10.1016/j.quaint.2019.04.015
Sackett TE, Basiliko N, Noyce GL, Winsborough C, Schurman J, Ikeda C, Thomas SC (2015) Soil and greenhouse gas responses to biochar additions in a temperate hardwood forest. Glob Change Biol Bioenergy 7:1062–1074. https://doi.org/10.1111/gcbb.12211
Samojlik T, Jedrzejewska B, Michniewicz M, Krasnodebski D, Dulinicz M, Olczak H, Karczewski A, Rotherham ID (2013) Tree species used for low-intensity production of charcoal and wood-tar in the 18th-century Biaowieza Primeval forest, Poland. Phytocoenologia 43:1–12. https://doi.org/10.1127/0340-269X/2013/0043-0511
Schellekens J, Almeida-Santos T, Macedo RS, Buurman P, Kuyper TW, Vidal-Torrado P (2017) Molecular composition of several soil organic matter fractions from anthropogenic black soils (Terra Preta de Índio) in Amazonia—a pyrolysis-GC/MS study. Geoderma 288:154–165. https://doi.org/10.1016/j.geoderma.2016.11.001
Schindler K (1872) O uhlířství. Háj: Časopis pro lesníka, myslivce a přítele přírody
Schmidt M, Mölder A, Schönfelder E, Engel F, Fortmann-Valtink W (2016) Charcoal kiln sites, associated landscape attributes and historic forest conditions: DTM-based investigations in Hesse (Germany). For Ecosyst 3:8. https://doi.org/10.1186/s40663-016-0067-6
Schneider A, Hirsch F, Raab A, Raab T (2018) Dye tracer visualization of infiltration patterns in soils on relict charcoal hearths. Front Environ Sci Eng China 6:143. https://doi.org/10.3389/fenvs.2018.00143
Schneider A, Hirsch F, Raab A, Raab T (2019) Temperature regime of a charcoal-enriched land use legacy soil. Soil Sci Soc Am J 83:565–574
Senbayram M, Saygan EP, Chen R, Aydemir S, Kaya C, Wu D, Bladogatskaya E (2019) Effect of biochar origin and soil type on the greenhouse gas emission and the bacterial community structure in N fertilised acidic sandy and alkaline clay soil. Sci Total Environ 660:69–79. https://doi.org/10.1016/j.scitotenv.2018.12.300
Shen X, Huang D-Y, Ren X-F, Zhu H-H, Wang S, Xu C, He Y-B, Luo Z-C, Zhu Q-H (2016) Phytoavailability of Cd and Pb in crop straw biochar-amended soil is related to the heavy metal content of both biochar and soil. J Environ Manage 168:245–251. https://doi.org/10.1016/j.jenvman.2015.12.019
Sigmund G, Bucheli TD, Hilber I, Micić V, Kah M, Hofmann T (2017) Effect of ageing on the properties and polycyclic aromatic hydrocarbon composition of biochar. Environ Sci Process Impacts 19:768–774. https://doi.org/10.1039/c7em00116a
Singh B, Singh BP, Cowie AL (2010) Characterisation and evaluation of biochars for their application as a soil amendment. Soil Res 48:516–525. https://doi.org/10.1071/SR10058
Sombroek W, Kern D, Rodrigues TE (2002) Terra preta and terra mulata: pre-columbian Amazon Kitchen middens and agricultural fields, their sustainability and their replication. Oriental-Artigo
Song W, Guo M (2012) Quality variations of poultry litter biochar generated at different pyrolysis temperatures. J Anal Appl Pyrolysis 94:138–145. https://doi.org/10.1016/j.jaap.2011.11.018
Spokas KA, Novak JM, Stewart CE, Cantrell KB, Uchimiya M, Dusaire MG, Ro KS (2011) Qualitative analysis of volatile organic compounds on biochar. Chemosphere 85:869–882. https://doi.org/10.1016/j.chemosphere.2011.06.108
Stavi I (2013) Biochar use in forestry and tree-based agro-ecosystems for increasing climate change mitigation and adaptation. Int J Sustain Dev World Ecol 20:166–181. https://doi.org/10.1080/13504509.2013.773466
Straka TJ (2017) Charcoal as a fuel in the ironmaking and smelting industries. Adv Hist Stud 6:56. https://doi.org/10.4236/ahs.2017.61004
Suliman W, Harsh JB, Abu-Lail NI, Fortuna A-M, Dallmeyer I, Garcia-Perez M (2016) Influence of feedstock source and pyrolysis temperature on biochar bulk and surface properties. Biomass Bioenergy 84:37–48. https://doi.org/10.1016/j.biombioe.2015.11.010
Sun Y, Gao B, Yao Y, Fang J, Zhang M, Zhou Y, Chen H, Yang L (2014) Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem Eng J 240:574–578. https://doi.org/10.1016/j.cej.2013.10.081
Szabó P, Kuneš P, Svobodová-Svitavská H, Švarcová MG, Křížová L, Suchánková S, Müllerová J, Hédl R (2017) Using historical ecology to reassess the conservation status of coniferous forests in Central Europe. Conserv Biol 31:150–160. https://doi.org/10.1111/cobi.12763
Thomas SC, Gale N (2015) Biochar and forest restoration: a review and meta-analysis of tree growth responses. New for 46:931–946. https://doi.org/10.1007/s11056-015-9491-7
Van Zwieten L, Kimber S, Morris S, Chan KY, Downie A, Rust J, Joseph S, Cowie A (2010) Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 327:235–246. https://doi.org/10.1007/s11104-009-0050-x
Venegas A, Rigol A, Vidal M (2016) Changes in heavy metal extractability from contaminated soils remediated with organic waste or biochar. Geoderma 279:132–140. https://doi.org/10.1016/j.geoderma.2016.06.010
Weber K, Quicker P (2018) Properties of biochar. Fuel 217:240–261. https://doi.org/10.1016/j.fuel.2017.12.054
Wenhua L (2004) Degradation and restoration of forest ecosystems in China. For Ecol Manage 201:33–41. https://doi.org/10.1016/j.foreco.2004.06.010
Werdin J, Fletcher TD, Rayner JP, Williams NSG, Farrell C (2020) Biochar made from low density wood has greater plant available water than biochar made from high density wood. Sci Total Environ 705:135856. https://doi.org/10.1016/j.scitotenv.2019.135856
Witharana C, Ouimet WB, Johnson KM (2018) Using LiDAR and GEOBIA for automated extraction of eighteenth–late nineteenth century relict charcoal hearths in southern New England. Gisci Remote Sens 55:183–204. https://doi.org/10.1080/15481603.2018.1431356
Xu G, Lv Y, Sun J, Shao H, Wei L (2012) Recent advances in biochar applications in agricultural soils: benefits and environmental implications. Clean: Soil, Air, Water 40:1093–1098. https://doi.org/10.1002/clen.201100738
Young MJ, Johnson JE, Abrams MD (1996) Vegetative and edaphic characteristics on relic charcoal hearths in the Appalachian mountains. Vegetatio 125:43–50. https://doi.org/10.1007/BF00045203
Yu H, Zou W, Chen J, Chen H, Yu Z, Huang J, Tang H, Wei X, Gao B (2019) Biochar amendment improves crop production in problem soils: a review. J Environ Manage 232:8–21. https://doi.org/10.1016/j.jenvman.2018.10.117
Zhang J, Li Q, Wu J, Song X (2019) Effects of nitrogen deposition and biochar amendment on soil respiration in a Torreya grandis orchard. Geoderma 355:113918. https://doi.org/10.1016/j.geoderma.2019.113918
Zhao L, Cao X, Mašek O, Zimmerman A (2013) Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. J Hazard Mater 256–257:1–9. https://doi.org/10.1016/j.jhazmat.2013.04.015
Zhao S-X, Ta N, Wang X-D (2017) Effect of temperature on the structural and physicochemical properties of biochar with apple tree branches as feedstock material. Energies 10:1293. https://doi.org/10.3390/en10091293
Zhou G, Zhou X, Zhang T, Du Z, He Y, Wang X, Shao J, Cao Y, Xue S, Wang H, Xu C (2017) Biochar increased soil respiration in temperate forests but had no effects in subtropical forests. For Ecol Manage 405:339–349. https://doi.org/10.1016/j.foreco.2017.09.038
Zhou C, Heal K, Tigabu M, Xia L, Hu H, Yin D, Ma X (2020) Biochar addition to forest plantation soil enhances phosphorus availability and soil bacterial community diversity. For Ecol Manage 455:117635. https://doi.org/10.1016/j.foreco.2019.117635
Acknowledgements
We would like to thank Dr. Ondřej Drábek for his analysis of exchangeable elements in soils from RCHs.
Funding
The research was supported by the Technology Agency of the Czech Republic (project no. TL02000160).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could influence the work reported in this paper.
Additional information
Communicated by Agustín Merino.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Johanis, H., Lehejček, J. & Tejnecký, V. An insight into long-term effects of biochar application on forest soils. Eur J Forest Res 141, 213–224 (2022). https://doi.org/10.1007/s10342-022-01440-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-022-01440-0