Abstract
In this study, the effects of different maturity stages and different fruit parts on some quality parameters of two plum cultivars were investigated. For this purpose, ‘Stanley’ and ‘Sugar Plum’ cultivars, which are extensively cultivated, were used, and the fruits were harvested in three different periods. Firstly, width, length and weight as well as flesh firmness measurements were performed on fruits harvested. Later on, samples were taken separately from the flesh and peel of the fruits collected at three different maturity stages and color, chemical properties (total soluble solids [TSS], titratable acidity [TA]) and phytochemical properties (total phenolics, total anthocyanins and antioxidant activity) of these parts were determined. Height and weight increased, while firmness values decreased with the progress of maturity. Fruit color development increased with the progress of maturity and the fruits gained a brighter appearance. Similarly, in both cultivars, TSS values of fruit peel and flesh increased and TA decreased with the progress of maturity. Although varied with the maturity stages, it was determined that fruit peels had higher TSS and TA values than the fruit flesh. Total phenolics and antioxidant activity increased in both the fruit peel and flesh with the progress of maturity, and such increases were more prominent in the fruit peel. Antioxidant activity of fruit peel was found to be significantly higher than the fruit flesh at the Stage 3 maturity level. In both plum cultivars, it was determined that the fruit peel contained greater total anthocyanins than the fruit flesh in three different maturity stages. Throughout the maturity process, there was a slight increase in the total anthocyanins of the fruit flesh of the ‘Stanley’ cultivar, while there was no significant change in the fruit flesh total anthocyanins of the ‘Sugar Plum’ cultivar. On the other hand, in both cultivars, total anthocyanins of the fruit peel significantly increased with the progress of maturity. As a result, fruit weight, fruit appeal, fruit flavor, aroma and phytochemical traits increased and fruit firmness decreased with the progress of maturity. Phytochemicals were mostly encountered in fruit peels. It was concluded based on present findings that consuming mature fruits with their peels will contribute positively to human health and nutrition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plum (Prunus domestica L.) grows in temperate climate regions of the world. Plums are rich in pectin (Lin et al. 2018), phenolic compounds, anthocyanins (Gunduz and Saraçoğlu 2012), flavonoids and vitamins (Mahmoudi et al. 2022), thus constitute an indispensable component of daily diets of consumers. In recent years, consumers desire to consume fruits richer in nutritional components. However, nutritional content of the fruits may differ depending on several factors, including the maturity stage. Therefore, it is very important to determine the maturity stage where the nutrient content is the richest, as well as the appeal that affects consumer preference (Yarılgaç et al. 2019; Bobasa et al. 2020). However, quality losses may occur in plum fruits after harvest (Adiletta et al. 2022; Xu et al. 2022). There are various techniques to be applied before the harvest (Avcı et al. 2022; Bi et al. 2022; Sinha et al. 2022) and after the harvest (Li et al. 2021; Arabia et al. 2022) to prevent postharvest losses. It was stated that the optimal harvest time was also effective in prevention of post-harvest losses (Meng et al. 2021). As it was in many other fruit species, Alvarez-Herrera et al. (2021) reported a close relationship between harvest maturity and post-harvest life of plum fruits. It was indicated that semi-ripe fruits at harvest were preserved for longer durations, but the flavor characteristics of these fruits were lower than those harvested at the optimal maturity stage (Guerra and Casquero 2008).
Color, texture, soluble solids content (SSC) and fruit size are important criteria for determining the harvest time and fruit quality (Ndou et al. 2019; Salazar et al. 2020; Tena et al. 2020; Piccolo et al. 2021; Qiu et al. 2021). Along with the changes in consumer demands, some compounds such as anthocyanins, phenolic substances and antioxidants (Phan et al. 2021; Bahrin et al. 2022), which are stated to have positive effects on human health, have started to take place among important fruit quality criteria (Ndou et al. 2019). It was stated in previous studies that the harvest date directly affected the antioxidant capacity, phenolics and anthocyanins of the fruit (Smanalieva et al. 2019; Kodagoda et al. 2021). Besides maturity level, fruit phytochemicals vary in different parts of the fruit (Ozturk et al. 2013; Redondo et al. 2021; Traore et al. 2021). In general, total sugar content is high in fruit flesh (Lebaka et al. 2021), while total anthocyanin and antioxidant activities are high in the peel of plum, peach and apple (Drogoudi et al. 2008; Remorini et al. 2008; Usenik et al. 2013; Beyazit et al. 2021; Redondo et al. 2021; Ranganath 2022).
In this study, the effects of different ripening stages and fruit parts of plum fruits on some quality characteristics, which are stated to have positive effects for human health, especially antioxidants, phenolic substances and anthocyanins, were investigated.
Materials and Methods
Plant Materials and Experimental Design
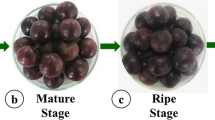
In this research, ‘Sugar Plum’ and ‘Stanley’ varieties, which are commonly cultivated plum cultivars (Murathan et al. 2020; Neshev et al. 2021), from the European plum (Prunus domestica) group were used as the plant material. The research was carried out in a commercial orchard in central district of Tokat province (40o21′00″ N latitude, 36o33′30″ E longitude, 775 m altitude), Turkey. Plum trees that are 9 years old and planted at 5.0 × 5.0 m spacings were used in this study. Standard cultural practices (pruning, fertilization and irrigation) were applied during the trials. The study was designed with three replications and 100 fruits in each replication. Fruit samples were harvested at three different maturities (Stage 1 [early harvest], Stage 2 [commercial maturity] and Stage 3 [full harvest]) on coloration status. Harvested samples were placed into 8‑kg capacity plastic boxes and brought to the laboratory where the analyses are conducted. Fruits were divided into two groups: the first group was used as whole fruit and the other group was used as peel and flesh (Table 1).
Physical Quality Traits
A total of 20 fruits were used for physical measurements on harvested fruits. Fruit weight of the samples was measured with a digital balance (±0.01 g) (Weightlab, Turkey). Fruit sizes (width, length) were determined by digital caliper (±0.01 mm) (Absolute, Germany). Firmness was measured with a penetrometer as the maximum force (Newton, N) required to penetrate 20 mm vertically into the fruit (Zwick Z 0.5 Universal). In the measurement, test speed was selected as 30 mm/min and a 1.8-mm diameter stainless steel head was used.
Chemical Quality Traits
For chemical analyses, 40 fruits from each replication were used. After removing the seeds of the fruits for the analysis, 20 fruits were cut as whole fruits and the other 20 fruits were cut by separating the skin and pulp parts for each replication. Whole fruit and fruit pulp were blended without adding water, and the peel part was blended with water. The dilution factor was considered in later measurements. SSC values were measured with the help of a digital refractometer (HI 96801, Hanna, Italy). The juice was mixed with distilled water at a ratio of 1:4 for titratable acidity (TA). The resultant mixture was then titrated with 0.1 N NaOH (sodium hydroxide) until pH of 8.1 and the amount of NaOH consumed in the titration was expressed as g malic acid 100 g−1.
Phytochemical Properties
In each measurement period, 40 fruits from each replication were homogenized with a blender. About 30 ml of homogenate was taken and placed into a 50-ml falcon tube. The samples were centrifuged and used to determine the phytochemicals. Spectrophotometric measurements were made in an ultraviolet-visible (UV-VIS) spectrophotometer (Model T60U, PG Instruments) to determine total phenolics, antioxidant capacity and total anthocyanins. The total phenolic content of the fruits was determined according to the method recommended by Singleton and Rossi (1965) and expressed as µg GAE (gallic acid equivalent) g−1 fresh weight (fw). Antioxidant activity was determined in accordance with two different procedures as of trolox equivalent antioxidant capacity (TEAC) (Özgen et al. 2006) and ferric reducing antioxidant power (FRAP) (Benzie and Strain 1999) and the results expressed in µmol/TE g mmol Trolox Equivalent (TE) kg−1 fw.
Total Antioxidant Activity
Total antioxidant activity (TAA) was estimated by two standard procedures, FRAP and TEAC assays. Benzie and Strain method was used in FRAP analysis (Benzie and Strain 1999). Accordingly, for the stock solution, 0.1 mol/L acetate (pH 3.6), 10 mmol/L 2,4,6-Tris(2-pyridyl)-s-triazine (TPTZ) and 20 mmol/L ferric chloride solutions were mixed in 10:1:1 ratios. For analysis, 20 µL of fruit extract and 2.97 mL of prepared solution were added. After waiting for 30 min at room temperature, its absorbance was measured at 593 nm wavelength in a spectrophotometer. Absorbance values were calculated with the use of Trolox (10–100 µmol/L) standard curves and the results were expressed in µmol Trolox equivalent/g fw.
TEAC analysis was performed according to Özgen et al. (2006). The 7 nm ABTS (2,2′-Azino-bis 3‑ethylbenzothiazoline-6-sulfonic acid) was mixed with 2.45 mM potassium bisulfate and kept in the dark for 12–16 h. Afterwards, this solution was diluted with 20 mM sodium acetate (pH4.5) buffer as to have an absorbance of 0.700 ± 0.01 at 734 nm wavelength of a spectrophotometer. About 20 µL of extract was mixed with 2.98 mL of copper and the absorbance was measured after 10 min at a wavelength of 734 nm in a spectrophotometer.
Total Monomeric Anthocyanins (TMA)
Total anthocyanin analysis was performed with the use of the pH difference (pH 1.0 and 4.5) method (Wrolstad et al. 2005). Extracts were supplemented with 300 µL and 2700 µl pH 1.0 solution and measurements were made at 520 and 700 nm wavelengths. The same process was done for pH 4.5 solution. Total anthocyanin quantity (molar extinction coefficient of 28000 cyanidin 3‑glycoside) was calculated from the absorbances ([A520–A700] pH 1.0-[A520–A700] pH 4.5) and results were expressed in µg anthocyanin/g fw.
Statistical Analyses
Data were analyzed through analysis of variance using the general linear model (GLM) procedure. Means were separated using the least significant difference (LSD) method at 0.05 significance level. All statistical analyses were performed with SAS Version 9.3 (SAS Institute Inc., Cary, NC, USA).
Results and Discussion
Fruit Physical Properties
Regular increases were seen in fruit widths of both cultivars with increasing maturity levels. However, a distinctive change was not encountered in fruit lengths with the progress of maturity. Increasing fruit weights were seen in both cultivars with the progress of maturity. Among the physical properties, fruit firmness was negatively influenced by the maturity levels (Table 2). Average fruit weights were reported as between 31.40–75.16 g for fully ripened fruits (Altuntaş et al. 2019; Butac et al. 2019). Fruit weights obtained in previous studies are similar to our study.
In general, there was a negative relationship between maturity change and fruit weight and fruit firmness. In the ‘Stanley’ cultivar, 34.95% increase in fruit weight and 38.46% decrease in fruit firmness occurred with the progression of maturation from Stage 1 to Stage 3. In the cultivar ‘Sugar Plum’, on the other hand, with the progress of maturation from Stage 1 to Stage 3, an increase of 64.05% in fruit weight and a decrease in fruit firmness of 56.33% occurred. The data revealed that there was an inverse relationship between flesh firmness and maturity and fruit flesh softened with the progress of maturity. The increase in fruit weight and the decrease in firmness values during ripening have also been demonstrated in previous studies (Altuntaş et al. 2020; Khalili et al. 2020; Mesa et al. 2021).
Fruit Color
Fruit color greatly influences consumer preferences and significantly changes with the progress of maturity. In present study, fruit color was determined as L*, chroma and hue angle (Table 3). In terms of fruit color parameters, significant differences were seen between the maturity levels and fruit parts. Fruit flesh L values increased with the progress of maturity, but differences in peel L values were not found to be significant. Fruit flesh chroma (C*) values increased with the progress of maturity in both ‘Stanley’ and ‘Sugar Plum’ cultivars, but such an increase in fruit peel was observed only in the ‘Sugar Plum’ cultivar. While the hue angle value of the ‘Stanley’ cultivar decreased in fruit flesh and peel with the progress of maturity, it increased in fruit flesh of the ‘Sugar Plum’ cultivar. No significant change was observed in the color values of the fruit peel.
In previous studies, researchers found that the fruit flesh and skin L* values of different fruit species increased (Özdemir 2008; Avcı et al. 2022) and decreased (Ndou et al. 2019; Guo et al. 2022) with the progress of maturity. Differences in research findings were mainly attributed to color differences of the fruit flesh. In present study, the greatest C* values were seen in Stage 3 maturity level (32.66 and 30.82) (Table 3). It was indicated in previous studies that there was an increase in chroma values of plum fruits with the progress of maturity (Kucuker et al. 2015; Özturk et al. 2015). In the ‘Stanley’ cultivar, H value decreased in both fruit flesh and peel with the progress of maturity, while it increased in fruit flesh of the ‘Sugar Plum’ cultivar, but there was no significant change in fruit peal. In previous studies, researchers stated hue angle values varied with the maturity levels depending on the species and cultivars (Ndou et al. 2019; Kodagoda et al. 2021; Winkler and Knoche 2021; Avcı et al. 2022; Farcuh et al. 2022; Li et al. 2022).
Chemical Properties
The total soluble solids (TSS) content and TA values were investigated in the samples taken from the whole fruit, fruit flesh and peel of the ‘Stanley’ and ‘Sugar Plum’ plum cultivars harvested at different maturity levels (Table 4). While the TSS values of both cultivars increased with the progress of maturity, TA decreased. When the fruit and its parts were compared, the greatest TSS was seen in fruit peel of both cultivars and there was no significant difference in TSS of fruit flesh and whole fruit. The highest TA values were measured in fruit peels.
In previous similar studies conducted on different fruit species, researchers reported increasing soluble solids content and decreasing TA values with the progress of ripening (Montaya et al. 2010; Lin and Lay 2013; Álvarez-Herrera et al. 2021). High SSC and TA especially in fruit peel are thought to be important criteria for the taste of fruits consumed without peeling.
Phytochemical Properties
In the ‘Stanley’ cultivar, total phenolics (TP) increased with the progress of maturity and average value was measured as 685.28 µg GAE/g fw in the whole fruit, 1140.94 28 µg GAE/g fw in the fruit peel and 528.86 µg GAE/g fw in the fruit flesh. In the ‘Sugar Plum’ cultivar, total phenolics also increased with the progress of maturity and average values was identified as 867.53 µg GAE/g in the whole fruit, 1244.70 µg GAE/g in the fruit peel and 547.87 µg GAE/g in the fruit flesh (Table 5). The greatest total phenolics level was observed in fruit peels. While the total phenolic content of fruit peel was 1030.89 µg GAE/g at the first maturity stage, it increased 1.3 times and reached to 1415.53 µg GAE/g in mature fruits. Researchers have determined with previous studies that phenolic substances are found in higher amounts in the peel of the fruit and with increasing maturity. Khalili et al. (2020) stated that the total phenol content increased with the progress of maturity. Redondo et al. (2021) indicated that fruit peels had 2.5–3.0 times greater total phenolics than fruit flesh. Drogoudi et al. (2008) reported 3.3 times greater total phenolics for apple peels than for fruit flesh.
In terms of antioxidant activity with the use of FRAP method, the greatest values were measured in fruit peels at Stage 3 maturity level (10.34 µmol TE/g fw in the ‘Stanley’ cultivar and 11.33 µmol TE/g fw in the ‘Sugar Plum’ cultivar). When the cultivars were compared, the average value of fruit peel of the ‘Stanley’ cultivar was 6.59 µmol TE/g fw, while this value was found to be 1.4 times (9.29 µmol TE/g fw) greater in the fruit peel of the ‘Sugar Plum’ cultivar. FRAP antioxidant activity in fruit flesh and whole fruit increased in both cultivars with the progress of maturity, but there was no significant difference between them.
In the ‘Stanley’ cultivar, the greatest TEAC antioxidant activity was measured in fruit peels. The antioxidant activity of 4.07 µmol TE/g fw at Stage 1 maturity increased to 10.34 µmol TE/g fw at Stage 3 maturity level. While there was no significant difference between whole fruit and fruit flesh, significantly higher antioxidant activity values were determined in the fruit peel. In the ‘Sugar Plum’ cultivar, the highest antioxidant activity was found in the peel at the Stage 3 maturity level (8.91 µmol TE/g fw). Average TEAC antioxidant activity was measured as 7.56 µmol TE/g fw in the fruit peel, 4.13 µmol/TE g fw in the fruit flesh and 4.45 µmol TE/g fw in the whole fruit. There was no significant difference between whole fruit and fruit flesh, but significant differences were seen in fruit peels.
Maturity has positive effects on the phytochemical properties of the fruit. In previous studies examining the antioxidant activities of Prunus domestica fruits, Michalska et al. (2019) reported FRAP antioxidant activity as 4.5 mmol Trolox∙100 g−1 dm and TEAC antioxidant activity as 4.6 mmol Trolox∙100 g−1 dm; Polatci (2021) reported FRAP activity of the ‘Stanley’ cultivar as 2.36 µmol TE/g dw and TEAC activity as 2.26 µmol TE/g, FRAP activity of the ‘Sugar Plum’ cultivar as 1.15 µmol TE/g dw and TEAC activity as 1.15 µmol TE/g dw; Mocan et al. (2018) reported TEAC activity as 30.19 mg TE/g. In different fruit species (cherry and cranberry), an increase in antioxidant capacity was observed with fruit ripening (Serrano et al. 2005; Çelik et al. 2008). In another study, Guo et al. (2003) investigated the effects of maturity levels on phytochemical properties of different fruit parts of plum fruits and reported FRAP antioxidant activity as 0.71 mmol/100g fw for fruit flesh and 8.09 mmol/100g fw for fruit peel. Redondo et al. stated that fruit peels had a high antioxidant activity. Drogoudi et al. (2008) stated that apple peels had about 1.5 times greater antioxidant activity than the fruit flesh.
In the ‘Stanley’ cultivar, the total anthocyanins increased with the progress of maturity and the highest values were seen at Stage 3 maturity level. In terms of total anthocyanins of different fruit parts, the highest value was seen in fruit peel (26.20 µg cy-3-glu/g fw), followed by the whole fruit (21.65 µg cy-3-glu/g fw) and fruit flesh (3.14 µg cy-3-glu/g fw). Similar results were observed in the ‘Sugar Plum’ cultivar. Total anthocyanins increased with the progress of maturity and the greatest value was seen at Stage 3 maturity level (17.05 µg cy-3-glu/g fw). Considering the average total anthocyanins of different fruit parts, the highest value was measured in fruit peel (7.27 µg cy-3-glu/g fw), followed by the whole fruit (5.90 µg cy-3-glu/g fw) and the fruit flesh (0.51 µg cy-3-glu/g fw). Peels of the ‘Stanley’ cultivar had 1.6 times greater total anthocyanins than peels of the ‘Sugar Plum’ cultivar. In studies conducted on European plums, total anthocyanins were reported as between 17.03 and 32.98 µg cy-3-glu/g (Michalska et al. 2016) and as 14.2 µg cy-3-glu/g (Michalska et al. 2019). Researchers stated that maturity of different species such as strawberry (Pineli et al. 2011), blackberry and mulberry (Montaya et al. 2010) increased total anthocyanins.
It was reported that total anthocyanins and antioxidant activities in fruit peels of plum (Usenik et al. 2013) and peach (Remorini et al. 2008) species increased with the progress of maturity. Researchers have stated that the total phenolics and antioxidant capacity of some fruits were much higher in the peel (Vieira et al. 2011; Manzoor et al. 2012), thus it would be more beneficial for human health to consume fruits with their peels. Present findings comply with the results of previous studies since total phenolics, antioxidant activity and total anthocyanins all increased with the progress of maturity. As it was in previous studies, all these phytochemical substances were found more in the peel of the fruit.
Total phenolics, total anthocyanins and antioxidant activity increase with the progress of maturity. Antioxidants in fruits and vegetables destroy free radicals that negatively affect human health (Isabelle et al. 2010) and prevent abnormal proliferation of cells (Kasnak and Palamutoğlu 2015). Consumption of some fruits prevents the formation of some diseases that cause cancer, cardiovascular diseases and premature death with the effect of antioxidant substances (Özgen and Scheerens 2006; Miraliakbari and Shahidi 2008; Isabelle et al. 2010). It is clearly understood from the studies that the consumption of the fruits of the ‘Stanley’ and ‘Sugar Plum’ cultivars at full maturity had a positive effect on human health.
Conclusion
In this study, quality parameters of the plum fruit in different maturity stages and different parts of the fruit were determined and the suitable harvest period was identified for commercial purposes and human health benefits. In both cultivars, fruit width, fruit length and fruit weight, fruit flavor and aroma increased and fruit firmness decreased with the progress of maturity. It was determined that maturity also resulted in differences in chemical properties of fruit parts. TSS and TA values were higher in fruit peel, thus fruits were recommended to be consumed with the peels. With the maturity, the color brightness of the fruit increased and the color reached a darker tone, thus the allure of the fruit increased. Total phenolics, total anthocyanins and antioxidant activity of the fruits increased with the progress of maturity and the highest values were reached in the peel of mature fruit. On the other hand, maturity has a negative effect on fruit flesh firmness and may create a problem in storage and marketing. As a result, fruit weight, fruit appeal, fruit flavor, aroma and phytochemical traits increased and fruit firmness decreased with the progress of maturity. Phytochemicals were mostly encountered in fruit peels. It was concluded based on present findings that consuming mature fruits with their peels will contribute positively to human health and nutrition.
References
Adiletta G, Gliottone G, Di Matteo M, Petriccione M (2022) Response of qualitative traits and antioxidant systems to chitosan postharvest treatment in ‘Black Golden’Japanese plum. Foods 11(6):853
Altuntaş E, Ozturk B, Saracoglu O (2019) Effect of AVG (Aminoethoxyvinylglycine) treatment and maturity stages on physico-mechanical and chemical properties of plum (cv. Giant) fruit. Adv Agric Sci 7(2):88–99
Altuntaş E, Ozturk B, Saracoglu O (2020) Physico-mechanical and chemical properties of president plum affected by amino ethoxy vinyl glycine (AVG) treatment and maturity stages. Agric Eng Int CIRG J 22(3):241–249
Alvarez-Herrera JG, Deaquiz YA, Rozo-Romero X (2021) Effect of storage temperature and maturity stage on the postharvest period of ’Horvin’ plums (Prunus domestica L.). Ingeniería Investig 41(2):e82530–e82530
Arabia A, Munné-Bosch S, Muñoz P (2022) Melatonin triggers tissue-specific changes in anthocyanin and hormonal contents during postharvest decay of Angeleno plums. Plant Sci 320:111287
Avcı V, Islam A, Ozturk B, Aglar E (2022) Effects of aloe vera gel and modified atmosphere packaging treatments on quality properties and bioactive compounds of plum (Prunus salicina L.) fruit throughout cold storage and shelf life. Erwerbs-Obstbau. https://doi.org/10.1007/s10341-022-00694-7
Bahrin AA, Moshawih S, Dhaliwal JS, Kanakal MM, Khan A, Le KS, Ming LC (2022) Cancer protective effects of plums: a systematic review. Biomed Pharmacother 146:112568
Benzie IF, Strain JJ (1999) Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Meth Enzymol 299:15–27
Beyazit S, Çalişkan O, Kiliç D (2021) Akdeniz Bölgesi’nde Örtüalti Meyve Yetiştiriciliği. Bahçe 50(1):59–70
Bi X, Dai Y, Zhou Z, Xing Y, Che Z (2022) Combining natamycin and 1‑methylcyclopropene with modified atmosphere packaging to evaluate plum (Prunus salicina cv.‘Cuihongli’) quality. Postharvest Biol Technol 183:111749
Bobasa EM, Phan ADT, Manolis C, Netzel M, Smyth H, Cozzolino D, Sultanbawa Y (2020) Effect of sample presentation on the near infrared spectra of wild harvest Kakadu plum fruits (Terminalia ferdinandiana). Infrared Phys Technol 111:103560
Butac M, Militaru M, Chitu E, Plopa C, Sumedrea M, Sumedrea D (2019) Differences and similarities between some European and Japanese plum varieties. Acta Hortic 1260:129–136
Çelik H, Özgen M, Serçe S, Kaya C (2008) Phytochemical accumulation andantioxidant capacity at fourmaturity stages of cranberry fruit. Sci Hortic 117(4):345–348
Drogoudi PD, Michailidis Z, Pantelidis G (2008) Peel and flesh antioxidant contentand harvest quality characteristics of seven apple cultivars. Sci Hortic 115(2):149–153
Farcuh M, Tajima H, Lerno LA, Blumwald E (2022) Changes in ethylene and sugar metabolism regulate flavonoid composition in climacteric and non-climacteric plums during postharvest storage. Food Chem Mol Sci 4:100075
Guerra M, Casquero PA (2008) Effect of harvest date on cold storage and postharvest quality of plum cv. Green Gage. Postharvest Biol Technol 47(3):325–332
Gunduz K, Saraçoğlu O (2012) Variation in total phenolic content and antioxidant activity of Prunus cerasifera Ehrh. selections from Mediterranean region of Turkey. Sci Hortic 134:88–92
Guo C, Yang J, Wei J, Li Y, Xu J, Jiang Y (2003) Antioxidant activities of peel, pulp and seed fractions of common fruits as determined by FRAP assay. Nutr Res 23:1719–1726
Guo H, Yan F, Li P, Li M (2022) Determination of storage period of harvested plums by near-infrared spectroscopy and quality attributes. J Food Process Preserv. https://doi.org/10.1111/jfpp.16504
Isabelle M, Lee BL, Lim MT, Koh WP, Huang D, Ong CN (2010) Antioxidant activity and profiles of common fruits in Singapore. Food Chem 123(1):77–84
Kasnak C, Palamutoğlu R (2015) Doğal antioksidanların sınıflandırılması ve insan sağlığına etkileri. Türk Tarımgıda Bilim Teknol Derg 3(5):226–234
Khalili H, Hajilou J, Dehghan G, Bakhshi D (2020) Assessing quality characteristics of green gage (Prunus domestica L.) genotypes at different harvest times. Int J Fruit Sci 20(4):667–681
Kodagoda G, Hong HT, O’Hare TJ, Sultanbawa Y, Topp B, Netzel ME (2021) Effect of storage on the nutritional quality of queen garnet plum. Foods 10(2):352
Kucuker E, Ozturk B, Yildiz K, Ozkan Y (2015) Effect of aminoethoxyvinylglycine (avg) on the quality of japanese plum (Prunus salicina Lindell cv.‘Fortune’) fruits. Acta Sci Pol Hortorum Cultus 14:3–17
Lebaka VR, Wee YJ, Ye W, Korivi M (2021) Nutritional composition and bioactive compounds in three different parts of mango fruit. Int J Environ Res Public Health 18(2):741
Li G, Li X, Li J, Lv Z, Wang H, Gao R, Luo L (2021) Effect of natural green coating materials on the postharvest quality of Prunus salicina Lindl. cv.‘Shazikongxinli fruit’: a sustainable approach to prevent economic and food loss. Sustain Chem Pharm 24:100516
Li H, Huang Z, Addo KA, Yu Y (2022) Evaluation of postharvest quality of plum (Prunus salicina L. cv.‘French’) treated with layer-by-layer edible coating during storage. Sci Hortic 304:111310
Lin CY, Lay HL (2013) Characteristics of fruitgrowth, component analysis and antioxidant activity of mulberry (Morusspp.). Sci Hortic 162:285–292
Lin Y, Lin Y, Lin H, Lin M, Li H, Yuan F, Xiao J (2018) Effects of paper containing 1‑MCP postharvest treatment on the disassembly of cell wall polysaccharides and softening in Younai plum fruit during storage. Food Chem 264:1–8
Mahmoudi R, Razavi F, Rabiei V, Gohari G, Palou L (2022) Application of glycine betaine coated chitosan nanoparticles alleviate chilling injury and maintain quality of plum (Prunus domestica L.) fruit. Int J Biol Macromol 207:965–977
Manzoor M, Anwar F, Mahmood Z, Rashid U, Ashraf M (2012) Variation in minerals, phenolicsand antioxidant activity of peeland pulpof different varieties of peach (Prunus persica L.) fruitfrom Pakistan. Molecules 17:6491–6506
Meng Q, Shang J, Huang R, Zhang Y (2021) Determination of soluble solids content and firmness in plum using hyperspectral imaging and chemometric algorithms. J Food Process Eng 44(1):13597
Mesa K, Guerrero C, Contador L, Manríquez D, Reginato G (2021) Preliminary results of the determination of harvest indexes for the fresh consumption of’d’Agen’plum in Chile. In: XII international symposium on plum and prune genetics, breeding and pomology, vol 1322, pp 327–334
Michalska A, Honke J, Łysiak G, Andlauer W (2016) Effect of drying parameters on the formation of early and intermediate stage products of the Maillard reaction in different plum (Prunus domestica L.) cultivars. LWT Food Sci Technol 65:932–938
Michalska A, Wojdyło A, Majerska J, Lech K, Brzezowska J (2019) Qualitative and quantitative evaluation of heat-induced changes in polyphenols and antioxidant capacity in Prunus domestica L. by-products. Molecules 24(16):3008
Miraliakbari H, Shahidi F (2008) Antioxidant activity of minor components of tree nut oils. Food Chem 111(2):421–427
Mocan A, Diuzheva A, Carradori S, Andruch V, Massafra C, Moldovan C, Locatelli M (2018) Development of novel techniques to extract phenolic compounds from Romanian cultivars of Prunus domestica L. and their biological properties. Food Chem Toxicol 119:189–198
Montaya O, Vaillant F, Cozzano S, Mertz C, Perez A, Castro M (2010) Phenolic content andantioxiant capacity of tropical highland blackberry (Rubus adenotrichus Schltdl.) during tree edible maturity stages. Food Chem 119:1497–1501
Murathan ZT, Arslan M, Erbil N (2020) Analyzing biological properties of some plum genotypes grown in Turkey. Int J Fruit Sci 20(sup3):S1729–S1740
Ndou A, Tinyani PP, Slabbert RM, Sultanbawa Y, Sivakumar D (2019) An integrated approach for harvesting Natal plum (Carissa macrocarpa) for quality and functional compounds related to maturity stages. Food Chem 293:499–510
Neshev N, Nesheva M, Akova V (2021) Agronomic and fruit quality characteristics of the Bulgarian plum cultivar ’Pagane’. In: XII international symposium on plum and prune genetics, breeding and pomology, vol 1322, pp 33–40
Özdemir (2008) Venüs Nektarinlerinin Meyve Büyümesi Sırasında Kalite Parametrelerindeki Değişimlerin Derim Olumuyla İlişkilendirilmesi. Tarım Bilimi Araştırma Derg 1(1):19–24
Özgen M, Reese RN, Tulio AZ, Miller AR, Scheerens JC (2006) Modified 2,2-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2’-Diphenyl-1-picrylhydrazyl (DPPH) methods. J Agric Food Chem 54(4):1151–1157
Ozturk B, Altuntas E, Yildiz K, Ozkan Y, Saracoglu O (2013) Effect of methyl jasmonate treatments on the bioactive compounds and physicochemical quality of ‘Fuji’ apples. Cien Investig Agrar 40(1):201–211
Özturk B, Kucuker E, Yıldız K, Celik SM (2015) Changes of bioactive compounds and ethylene production of Japanese plums treated with pre-harvest aminoethoxyvinylglycine. Int J Food Prop 18(10):2165–2186
Phan ADT, Damyeh MS, Chaliha M, Akter S, Fyfe S, Netzel ME, Sultanbawa Y (2021) The effect of maturity and season on health-related bioactive compounds in wild harvested fruit of Terminalia ferdinandiana (Exell). Int J of Food Sci Tech 56(12):6431–6442
Piccolo EL, Araniti F, Landi M, Massai R, Guidi L, Abenavoli MR, Remorini D (2021) Girdling stimulates anthocyanin accumulation and promotes sugar, organic acid, amino acid level and antioxidant activity in red plum: an overview of skin and pulp metabolomics. Sci Hortic 280:109907
Pineli LDLDO, Moretti CL, Santos MS, Campos AB, Brasileiro AV, Córdova AC, Chiarello MD (2011) Antioxidants and other chemical and physical characteristics of two strawberry cultivars at different ripeness stages. J Food Compos Anal 24(1):11–16
Polatci H (2021) Determination of quality attributes of european plums (Prunus Domestica L.) cultivars under different drying conditions. Pak J Bot 53(2):579–584
Qiu X, Zhang H, Zhang H, Duan C, Xiong B, Wang Z (2021) Fruit textural characteristics of 23 plum (Prunus salicina Lindl) cultivars: evaluation and cluster analysis. HortScience 56(7):816–823
Ranganath KG (2022) Pigments that colour our fruits: an overview. Erwerbs-Obstbau. https://doi.org/10.1007/s10341-022-00698-3
Redondo D, Gimeno D, Calvo H, Venturini ME, Oria R, Arias E (2021) Antioxidant activity and phenol content in different tissues of stone fruits at thinning and at commercial maturity stages. Waste Biomass Valorization 12(4):1861–1875
Remorini D, Tavarini S, Degl’Innocenti E, Loreti F, Massai R, Guidi L (2008) Effect of rootstocks and harvesting time on the nutritional quality of peel and flesh of peach fruits. Food Chem 110:361–367
Salazar JA, Pacheco I, Zapata P, Shinya P, Ruiz D, Martínez-Gómez P, Infante R (2020) Identification of loci controlling phenology, fruit quality and post-harvest quantitative parameters in Japanese plum (Prunus salicina Lindl.). Postharvest Biol Technol 169:111292
Serrano M, Guillén F, Martínez-Romero D, Castillo S, Valero D (2005) Chemical constituents and antioxidant activity of sweet cherry at different ripening stages. J Agric Food Chem 53(7):2741–2745
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16(3):144–158
Sinha A, Jawandha SK, Gill PPS, Kaur N, Arora A (2022) Pre-harvest application of calcium maintained fruit quality in cold stored plum. Indian J Ecol 49(2):491–495
Smanalieva J, Iskakova J, Oskonbaeva Z, Wichern F, Darr D (2019) Determination of physicochemical parameters, phenolic content, and antioxidant capacity of wild cherry plum (Prunus divaricata Ledeb.) from the walnut-fruit forests of Kyrgyzstan. Eur Food Res Technol 245(10):2293–2301
Tena N, Martín J, Asuero AG (2020) State of the art of anthocyanins: antioxidant activity, sources, bioavailability, and therapeutic effect in human health. Antioxidants 9(5):451
Traore KF, Kone KY, Ahi AP, Soro D, Assidjo NE, Fauconnier ML, Sindic M (2021) Phenolic compounds characterisation and antioxidant activity of black plum (Vitex doniana) fruit pulp and peel from Côte d’Ivoire. J Food Meas Charact 15(2):1281–1293
Usenik V, Stampar F, Kastelec D (2013) Phytochemicals Infruits of two prunus domestica L. plum cultivars during ripening. J Sci Food Agric 93:681–692
Vieira FGK, Borges GDSC, Copetti C, DiPietro PF, da Costa Nunes E, Fett R (2011) Phenolic compound sand antioxidant activity of the apple flesh and peel of eleven cultivars grown in Brazil. Sci Hortic 128(3):261–266
Winkler A, Knoche M (2021) Xylem, phloem and transpiration flows in developing European plums. PLoS ONE 16(5):e252085
Wrolstad RE, Durst RW, Lee J (2005) Tracking color and pigment changes in anthocyanin products. Trends Food Sci Technol 16(9):423–428
Xu R, Wang L, Li K, Cao J, Zhao Z (2022) Integrative transcriptomic and metabolomic alterations unravel the effect of melatonin on mitigating postharvest chilling injury upon plum (cv. Friar) fruit. Postharvest Biol Technol 186:111819
Yarılgaç T, Kadim H, Ozturk B (2019) Role of maturity stages and modified-atmosphere packaging on the quality attributes of cornelian cherry fruits (Cornus mas L.) throughout shelf life. J Sci Food Agric 99(1):421–428
Funding
No financial support was received for this study.
Author information
Authors and Affiliations
Contributions
Berna Ergun Cetin: participated in data collection, analysis, description, and draft of the manuscript. Onur Saraçoğlu: participated in supervision of the work starting from the proposal up to final draft, edited and revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
B.E. Cetin and O. Saraçoğlu declare that they have no competing interests.
Ethical standards
For this article no studies with human participants or animals were performed by any of the authors. All studies mentioned were in accordance with the ethical standards indicated in each case. Consent for publication: Not applicable.
Additional information
This study is derived from a part of Berna Ergun Cetin’s master thesis.
Data availability
Not applicable.
Rights and permissions
Springer Nature oder sein Lizenzgeber (z.B. eine Gesellschaft oder ein*e andere*r Vertragspartner*in) hält die ausschließlichen Nutzungsrechte an diesem Artikel kraft eines Verlagsvertrags mit dem/den Autor*in(nen) oder anderen Rechteinhaber*in(nen); die Selbstarchivierung der akzeptierten Manuskriptversion dieses Artikels durch Autor*in(nen) unterliegt ausschließlich den Bedingungen dieses Verlagsvertrags und dem geltenden Recht.
About this article
Cite this article
Cetin, B.E., Saraçoğlu, O. Effects of Different Maturity Stages and Fruit Parts on Quality Traits of Plum (Prunus domestica) Fruits. Erwerbs-Obstbau 65, 1069–1077 (2023). https://doi.org/10.1007/s10341-022-00795-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-022-00795-3