Abstract
The brown marmorated stink bug, Halyomorpha halys, is considered a serious invasive species capable of causing significant agricultural and nuisance problems. Researchers in the USA recently identified the two-component H. halys aggregation pheromone from populations in North America and found that when it is combined with the pheromone synergist methyl decatrienoate (MDT), it resulted in reliable, season-long captures of H. halys throughout the USA. However, no study has assessed whether H. halys populations in the native range are attracted to and can also be reliably captured by the combination of these stimuli. In this study, our goal was to evaluate the response of H. halys adults, nymphs, and non-target organisms to traps baited with the aggregation pheromone, MDT alone, or in combination at three locations in the native range. Traps baited with combined stimuli reliably captured the greatest numbers of H. halys adults and nymphs, and traps baited with either lure treatment caught a unique community of non-target organisms. In addition, Plautia stali was cross-attracted to the H. halys aggregation pheromone. Overall, we demonstrate that the pheromone-based tools developed in the USA may be used for worldwide detection and surveillance programs for H. halys and P. stali.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key messages
-
It is unknown whether the aggregation pheromone identified from the USA is effective in the native range of the brown marmorated stink bug, Halyomorpha halys.
-
Our study is the first to demonstrate attraction by H. halys adults and nymphs in the native Asian range to pheromone-baited traps.
-
This suggests that pheromone-based technology developed in the USA may be applicable in other parts of the world for the surveillance and management of this invasive species.
Introduction
The invasive brown marmorated stink bug, Halyomorpha halys (Stal) (Hemiptera: Pentatomidae), is a growing threat to worldwide agricultural production (Rice et al. 2014; Haye et al. 2015; Lee 2015). This species is both extremely polyphagous, feeding on over 150 economically important crops (www.stopbmsb.org), and also highly damaging, causing $37 million in damage to apples in the mid-Atlantic region of the USA alone in 2010 (Leskey et al. 2012b). Where H. halys has been introduced, it has led to the disruption of established integrated pest management (IPM) programs for many agricultural crops, and in some cases, as much as a four-fold increase in insecticide usage (Leskey et al. 2012b).
Halyomorpha halys is originally from Asia, specifically the Republic of Korea, Japan, Taiwan, and China (Hoebeke and Carter 2003). However, it is now established in the USA (Leskey et al. 2012a), Canada (Gariepy et al. 2014), and Europe (Wermelinger et al. 2008). When exotic species are introduced to new areas, often only a small propagule is transported or survives such that genetic drift is a large factor in founding populations (Baker and Moeed 1987; Dlugosch and Parker 2008). This has similarly been revealed as the case for the H. halys populations in the USA, which originates from Beijing, China (Xu et al. 2013). Invasive species that establish populations in new regions often experience different abiotic and biotic conditions such as reduced natural enemies (Vilà et al. 2005), leading to rapid life history change (Blossey and Notzold 1995; Zangerl and Berenbaum 2005; Phillips et al. 2010). Moreover, the strong effects of genetic drift on a small population may result in altered chemical perception of the environment, and differing responses to the same semiochemicals, as has been the case in the red imported fire ant, Solenopsis invicta (Hymenoptera: Formicidae), and the Argentine ant, Linepithema humile (Hymenoptera: Formicidae), which both lost the ability to chemically discriminate queens and other nestmates in their introduced range (Porter and Savignano 1990; Tsutsui and Case 2001). In fact, these founding events have contributed to their invasiveness in their introduced range compared to their native range. This suggests that populations in an introduced range may have different responses to behavioral stimuli than populations in the native range and that this may actually facilitate the invasion process.
In the USA, researchers have been characterizing the baseline biology and ecology of H. halys in the introduced range (e.g., Ioannidis et al. 2014; Jones et al. 2014; Lee and Leskey 2015; Rice et al. 2014), as well as developing effective monitoring (Leskey et al. 2012c, 2015a, b, c; Morrison et al. 2015; Nielsen et al. 2013) and management tactics (e.g., Blaauw et al. 2015; Lara et al. 2016; Leskey et al. 2014; Morrison et al. 2016b, d ; Short et al. 2016). In particular, there has been a large amount of work examining the chemical ecology of H. halys, including the identification of the two-component aggregation pheromone, a 3.5:1 ratio of (3S,6S,7R,10S)-10,11-epoxy-1-bisabolen-3-ol and (3R,6S,7R,10S)-10,11-epoxy-1-bisabolen-3-ol (Khrimian et al. 2014). An investigation of the remaining stereoisomeric mixtures of the parent compound that exists in 16 stereoisomeric forms found that it was not critical to have a highly purified mixture of pheromone in order for lures to be effective in attracting H. halys to traps (Leskey et al. 2015b), which contrasts with the case for many lepidopteran sex pheromones (e.g., Adati and Tatsuki 1999). Subsequently, it has been determined that combining the H. halys aggregation pheromone with methyl (E,E,Z)-2,4,6-decatrienoate (MDT) has a synergistic effect on trap captures (Weber et al. 2014a). Throughout the USA, H. halys exhibits season-long attraction to traps baited with the aggregation pheromone in combination with MDT (Leskey et al. 2015a). More recently, these semiochemicals have been used in a targeted attract-and-kill approach for managing H. halys populations (Morrison et al. 2016b) and developing effective thresholds in apple orchards (Short et al. 2016).
Interestingly, little information exists regarding the chemical ecology of H. halys in its native range. In Asia, H. halys responds to the cross-attractive MDT, which is the male-produced aggregation pheromone of Plautia stali Scott (Sugie et al. 1996). The compound MDT has frequently been employed to attract H. halys to traps in its native (e.g., Adachi et al. 2007; Park et al. 2010) and introduced range (Aldrich et al. 2007; Khrimian et al. 2008). In these studies, captures of H. halys occur primarily between July and September (Funayama 2003, 2008). Importantly, though, it is unknown whether H. halys in Asia responds similarly to the aggregation pheromone, which was identified from individuals in the introduced range, or whether there is a synergistic response to traps baited with both its aggregation pheromone and MDT. If H. halys responds similarly across its introduced and native range, pheromone traps may easily be transferrable to other localities, thereby having worldwide implications for attempts at detection and exclusion of this pest.
One common concern with pheromone-based trapping is the abundance of non-target organisms captured in addition to the targeted taxon. Elevated amounts of non-target organisms may be problematic, as it can increase the time it takes to process samples (e.g., Kelly et al. 2013). Moreover, stink bugs are well-known for generally exhibiting cross-attraction to the pheromones of heterospecifics. For example, Piezodorus hybnei (Gmelin) (Hemiptera: Pentatomidae) is cross-attracted to the pheromone of its main competitor, Riptortus pedestris (Endo et al. 2010), which is itself a large pest in the Republic of Korea (Son et al. 2000). Finally, it is known that tachinid flies are attracted to certain stink bug-produced pheromones, including methyl (2E,4Z)-decadienoate (Aldrich et al. 1991) and MDT (Aldrich et al. 2007). For instance, Trichopoda pennipes (Diptera: Tachinidae) is known to parasitize adult and late instar H. halys nymphs (Rice et al. 2014). The goals of the current study were to evaluate the response of H. halys adults, H. halys nymphs, and non-target insects to the aggregation pheromone and MDT synergist throughout the growing season in Republic of Korea using standard black pyramid traps (Morrison et al. 2015).
Materials and methods
Trapping study
The following experiments were performed in the Laboratory of Insect Pests and Natural Enemies at the National Institute of Agricultural Sciences in the Republic of Korea. Large black pyramid traps (Rural Development Administration, Wansan-gu, Jeonju, Republic of Korea) were used for this study to investigate the efficacy of semiochemical stimuli on H. halys across the Republic of Korea. Similar pyramid traps reliably capture H. halys adults and nymphs over the entire growing season across the USA (Leskey et al. 2015a). Traps were modified and made based on Leskey et al. (2012). The trap base consisted of two 1-cm-thick black wooden base triangular panels 1.07 m (height) × 52 cm (width at base) × 8.2 cm (width at top). These panels served as the base for the trap. At the top of the base, each trap had an inverted plastic collection jar (16 cm × 10 cm height × diameter) with a funnel inserted at the bottom of the jar top, containing an internal cone opening of 1.6 cm, and vented on all four sides with a 3-cm opening covered with vinyl-coated polyester screen (mesh size: 3 mm × 3 mm). Each jar top contained a lure treatment (see below) and an insecticide-infused kill strip containing 10% dimethyl 2,2-dichlorovinyl phosphate (DDVP) (Hot Shot No-Pest Strip, Spectrum Group, St. Louis, MO, USA), which was changed every 2 weeks in 2014 and every 4 weeks in 2015 to prevent captured insects from escaping. Traps were set in three representative sites in Republic of Korea: Paju-si, Gongju-si, and Jinju-si, from April 24 until October 28, 2014 and from May 29 to Oct 29, 2015, respectively (Table 1). Every 2 weeks, the number of H. halys adults and nymphs captured in a trap was brought back to the laboratory for identification. Non-target insects captured in traps were also identified and counted, and these taxa included: P. stali Scott (Hemiptera: Pentatomidae), Riptortus pedestris (Fabricius) (Hemiptera: Alydidae), Reduviidae (Hemiptera), Ichneumonidae (Hymenoptera), Tachinidae (Diptera), and all other non-target true bugs. Treatments were re-randomized after each sampling date in 2014, but not in 2015.
Lure comparisons
In 2014, three lure treatments were deployed in pyramid traps. These treatments included lures formulated with 2.67 mg of the 2-component H. halys aggregation pheromone alone on gray septa (PHER; details of formulation in Morrison et al. 2015), 66 mg of methyl (E,E,Z)-2,4,5-decatrienoate alone in a green sachet (MDT; AgBio, Inc., Westminster, CO, USA), and an unbaited control. To prevent the escape of adults, each trap also contained a DDVP strip (Hot Shot No-Pest Strip, Spectrum Group, St. Louis, MO, USA) which was changed every 2 weeks in 2014 and every 4 weeks in 2015. Traps were placed in a >50-m transect containing each treatment, and spaced at least 25 m apart to reduce trap interference, and re-randomized every 2 weeks in 2014. There were a total of 4 or 6 replicate transects at the three sites (Table 1). The PHER lure was changed every 2 weeks, while the MDT was changed every 4 weeks, as prior research has shown these lures optimally dispense pheromone over a month (Joseph et al. 2013).
In 2015, four lure treatments were investigated: (1) 10 mg murgantiol lure formulated with the two-component H. halys aggregation pheromone alone (IP060, AgBio, Inc.); (2) 132 mg of MDT alone (2x lure, AgBio, Inc.); (3) both lures deployed together; or (4) an unbaited control. Again, lures were deployed in collection jars of pyramid traps arranged in a >50-m transect, with each trap spaced at least 25 m away from each other to minimize trap interference. Each transect was replicated 3 times per site at three sites (Table 1), for a total of 36 traps. In both years, transects were separated by 767 m ± 198.3 (SEM).
Statistical analysis
In 2014, the season was split up into three sampling periods: early (April 24 to June 15, 2014), mid (June 16 to August 15, 2014), and late (August 16 to November 1, 2014). Because sampling started later in 2015, the season was split up into two time periods instead of three: mid (June 16 to August 15, 2015) and late (August 16 to November 1, 2015).
Four generalized linear mixed models were used to analyze trap captures of H. halys. Adult and nymphs were analyzed separately. Different lures were tested during 2014 and 2015, and thus years were analyzed separately. In 2014, the models used the abundance of each life stage as the response and used the lure treatment (H. halys aggregation pheromone (PHER), MDT and unbaited control), and sampling period (early, mid, and late) as explanatory variables. In addition, the interaction between sampling period and lure treatment was included, and sampling site was used as a random blocking variable for the analysis. The model employed an autoregressive variance/covariance matrix using sampling date as the repeated measure with an underlying negative binomial distribution and log-link function to account for overdispersion in the dataset. To calculate the significance of the fixed effects, likelihood ratio tests were conducted in which the full model above was compared with a model that was missing the fixed effect term being evaluated for significance. A P value was calculated based on a Chi-square distribution. Upon a significant result for a given fixed effect in the model, Tukey’s HSD test was used for pairwise comparisons. In order to evaluate whether the position of the lure in a transect affected the resulting captures of adults and nymphs due to lack of re-randomization, a preliminary ANOVA was performed for adults and nymphs. The position of the lure within each transect was nested within site and used as the explanatory variable. After eliminating possible positional effects, an identical full model to 2014 was used for the 2015 dataset, except the lure treatments included H. halys aggregation pheromone (PHER), MDT, both lures combined (PHER + MDT) and an unbaited control. All statistical tests were carried out in R Software (R Core Development Team 2015), with α = 0.05.
To understand how lure treatment affected captures of non-target insect community in 2014 (non-target insects were not recorded in 2015), non-metric multi-dimensional (NMDS) ordination was employed to visualize the data. Season-long mean trap captures were calculated for each replicate trap. Subsequently, Bray-Curtis index values were calculated in a pairwise fashion among the replicates within and among treatments. Next, the data matrix was visualized using NMDS via the metaMDS function in the R package Vegan (Dixon 2003), using 35 random starts to find the most stable solution. Stress was calculated in the final solution, with stress values less than 0.13 typically being considered reliable and interpretable (Clarke 1993). To assess the significance of group differences in the ordination, an analysis of similarity (ANOSIM) was employed, with N = 10,000 permutations. This test also computes an R value, which represents a measure of similarity/dissimilarity, and the value for this ranges from 0 (identical) to −1 or 1 (completely different) (Anderson 2008).
In addition, non-target insect trap capture among lure treatments was compared using a multivariate analysis of variance (MANOVA) to test whether abundance of specific taxa varied. The abundance of each taxon (Plautia stali, Riptortus pedestris, Reduviidae, Ichneumonidae, Tachinidae, and all other bugs) was aggregated, and a model was created to explain their abundance by the lure treatment (unbaited control, MDT, and PHER), sampling period (early, mid, and late), and their interaction. Roy’s maximum root approximation for the F-statistic was used. Upon a significant result of the MANOVA, individual univariate ANOVAs were performed for each taxon. After a significant result from the ANOVA, pairwise comparisons were performed using Tukey’s HSD test.
Finally, to assess whether there is synergism between the combined stimuli in 2015, the following procedure was applied to adult and nymphal captures in each time period. Firstly, the trap captures with the H. halys aggregation alone and MDT alone were averaged across the time period for each life stage and then added together to create a theoretical threshold above which captures could be considered synergistic, and below which, they could be simply be considered additive. Secondly, a two-tailed t-test was performed between the average captures in traps with single stimuli and traps with combined stimuli. Lastly, if both the captures in traps with combined stimuli were above the threshold established for synergism, and these were significantly greater than captures in traps with single stimuli, then this was taken as evidence for synergism.
Results
2014 Trapping experiment
Adult captures
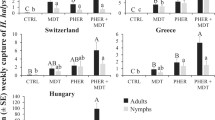
There were a total of 317 adults captured by traps, with our overall model explaining a significant amount of the variation in the data (likelihood ratio test: χ 2 = 99.1; df = 10, 445; P < 0.0001; Fig. 1). Lure treatment significantly affected adult captures (χ 2 = 60.1; df = 6, 445; P < 0.0001). For example, traps baited with MDT or the aggregation pheromone captured approximately 20 and 7 times more adults compared to unbaited controls, respectively. Moreover, trap captures varied with sampling period (χ 2 = 47.8; df = 6, 445; P < 0.0001), with 2 and 10 times more H. halys adults captured in the mid- and late-season, respectively, compared with the early season. There was also a significant interaction between sampling period and the lure treatment (χ 2 = 21.8; df = 4, 445; P < 0.0003). Importantly, traps with the aggregation pheromone showed greater sensitivity for capturing adults earlier in the season than with MDT (Fig. 2).
Captures of H. halys (± SE) adults (black bars) or nymphs (gray bars) in black pyramid traps checked once every 2 weeks in 2014 at three locations in Republic of Korea. The control remained unbaited, while MDT refers to lures containing the synergist methyl (2E,4E,6Z)-decatrienoate, and PHER indicates lures containing the H. halys aggregation pheromone. Bars with shared letters are not significantly different from one another (Tukey’s HSD, α = 0.05)
Season-long mean captures of H. halys adults (right column) and nymphs (left column) in traps checked once every 2 weeks during 2014 in the early (top panel), mid-season (middle), and late season (bottom) at three locations in Republic of Korea. The control remained unbaited, while MDT refers to lures containing the synergist methyl (2E,4E,6Z)-decatrienoate, and PHER indicates lures containing the H. halys aggregation pheromone. The standard error bars have been removed from the line for the sake of clarity of presentation
Nymphal captures
A total of 245 H. halys nymphs were captured, and the overall model described 61% of the variation in trap capture (likelihood ratio test: χ 2 = 85.3; df = 10, 445; P < 0.0001). The lure treatment in the trap significantly affected the number of nymphs captured (χ 2 = 25.4; df = 6, 445; P < 0.0003; Fig. 1). In particular, over 10 and 6 times more nymphs were captured in traps baited with MDT and the aggregation pheromone alone, respectively, compared with unbaited controls. Additionally, the sampling period also significantly affected trap captures (χ 2 = 53.8; df = 6, 445; P < 0.0001), with almost 7 times more H. halys nymphs captured in the late season compared with the mid- or early season. The interaction between the sampling period and lure treatment was not significant (χ 2 = 1.71; df = 4, 445; P = 0.789). Overall, MDT-baited traps caught more nymphs than traps with the aggregation pheromone alone throughout the entire season (Fig. 2). This is likely due to the 6-times higher loading rate of MDT in the lure compared to aggregation pheromone.
2015 Trapping experiment
Adult captures
In total, 1012 adult H. halys were captured, with trap position not having a significant effect on the captures of H. halys (ANOVA: F = 1.71; df = 8, 361; P = 0.094). The overall full model significantly explained over 50% of the variation in trap capture (likelihood ratio test: χ 2 = 184.7; df = 13, 359; P < 0.0001). The lure treatment affected adult captures (χ 2 = 129.8; df = 9, 359; P < 0.0001), with over 13 and 36 times greater captures in traps containing either the aggregation pheromone alone or in combination with MDT, respectively, compared with traps baited with MDT alone (Fig. 3). During the course of the entire season, unbaited traps captured no adults. The abundance of adults caught in traps was affected by sampling date (χ 2 = 64.2; df = 8, 359; P < 0.0001). Traps captured 24 times more adults in the late season compared to the mid-season. The interaction between the sampling period and the lure treatment was not significant (χ 2 = 4.91; df = 6, 359; P = 0.0.556). Traps baited with a combination of the aggregation pheromone +MDT captured more adults than any of the other lure treatment (Fig. 4). Finally, traps with the combined stimuli resulted in a synergistic effect on attraction of adults in both the mid- (t = 3.60; df = 106; P < 0.001) and late-season (t = 4.42; df = 160; P < 0.0001), capturing on average 2.38 and 2.51 times more individuals than one would expect if it were only an additive process.
Captures of H. halys (± SE) adults (black bars) or nymphs (gray bars) in black pyramid traps checked once every 2 weeks in 2015 at three locations in Republic of Korea. The control remained unbaited, while MDT refers to lures containing the synergist methyl (2E,4E,6Z)-decatrienoate, and PHER indicates lures containing the H. halys aggregation pheromone. Bars with shared letters are not significantly different from one another (Tukey’s HSD, α = 0.05). Because of later deployment, the early season has been omitted from the figure
Season-long mean captures of H. halys adults and nymphs in traps checked once every 2 weeks during 2015 in the mid-season (top panel), and late season (bottom) at three locations in Republic of Korea. The control remained unbaited, while MDT refers to lures containing the synergist methyl (2E,4E,6Z)-decatrienoate, and PHER indicates lures containing the H. halys aggregation pheromone. The early season was not included in this graph because only one sampling date is represented due to delayed deployment of traps. The standard error bars have been removed from the line for the sake of clarity of presentation. Because of later deployment, the early season has been omitted from the figure
Nymphal captures
A total of 1817 nymphs were captured in traps, with the position of the trap not significantly influencing trap captures (ANOVA: F = 1.56; df = 8, 361; P = 0.121). The full model significantly explained a significant amount of the variation in the trap capture (likelihood ratio tests: χ 2 = 236.7; df = 13, 359; P < 0.0001; Fig. 3). The lure treatment significantly affected trap captures (χ 2 = 125.6; df = 9, 359; P < 0.0001), with the aggregation pheromone- and aggregation pheromone + MDT-baited traps capturing 91 and 72 times more nymphs than traps with MDT alone, respectively (Fig. 3, Tukey’s HSD). Furthermore, trap captures varied significantly among sampling periods (χ 2 = 120.8; df = 8, 359; P < 0.0001). Over 1200 times more nymphs were captured during the late season compared with the mid-season. There was a significant interaction between the sampling period and lure treatment (χ 2 = 1.25; df = 6, 359; P = 0.974). Finally, traps with the combined stimuli resulted in an additive effect on attraction of nymphs in both the mid- (t = 1.41; df = 106; P = 0.158) and late-season (t = 0.845; df = 160; P = 0.399), capturing on average 1.27 times fewer individuals than one would expect if it were a synergistic process. However, traps baited with aggregation pheromone + MDT captured the most nymphs on each date throughout the season (Fig. 4).
Non-target insects in traps
In 2014, there were a total of 62 P. stali, 95 R. pedestris, 14 reduviids, 35 tachinids, and 66 other insects captured in pyramid traps. Lure treatment captured a unique community of insect taxa in traps (ANOSIM: R = 0.202; P < 0.047; Fig. 5). The overall model explaining the abundance of non-target insects was significant (MANOVA: Roy’s approx. F = 2.86; df = 6, 449; P < 0.01). However, only captures of adult P. stali were significantly affected by the lure treatment (F = 4.95; df = 2, 453; P < 0.007), with MDT-baited traps capturing approximately 12 and 2 times more individuals than either unbaited traps or traps baited with the H. halys aggregation pheromone alone, respectively (Fig. 6). Traps baited with H. halys aggregation pheromone consistently captured more adult P. stali than unbaited traps. Sampling period also affected P. stali captures with 2 and 4 times more adults captured in the early season compared with the mid- or late-season, respectively (Fig. 6). However, there was no significant interaction between sampling period and lure treatment (F = 0.8416; df = 2, 453; P = 0.499). The other non-target insects did not significantly vary with lure treatment in traps (range: F = 0.789–1.46; df = 2, 453; P = 0.234–0.455; Table 1).
Non-metric multi-dimensional scaling ordination plot based on Bray–Curtis similarities between the non-target insect communities in large pyramid traps deployed in the Republic of Korea at three sites for traps baited with methyl (2E,4E,6Z)-decatrienoate (red), H. halys aggregation pheromone (green), or left unbaited (black) in 2014. Each dot represents the insect community in a replicate for each treatment. Stress = 0.09. (Colour figure online)
Mean trap capture (± SE) of Plautia stali adults once every 2 weeks during the growing season at three sites in the Republic of Korea in 2014 with traps baited with methyl (2E,4E,6Z)-decatrienoate (MDT), the H. halys aggregation pheromone (PHER), or left unbaited (Control). Bars with shared letters are not significantly different from each other (Tukey’s HSD, α = 0.05)
Discussion
This is the first study to assess the response of H. halys adults and nymphs to its aggregation pheromone in its Asian native range. In the Republic of Korea, H. halys are most attracted to traps baited with a combination of the aggregation pheromone and MDT, which provided a synergistic influence on attraction for adults and an additive effect for nymphs. In 2014, traps baited with MDT were not attractive to H. halys in the early season, but were very attractive in the late-season. Conversely, H. halys adults were attracted to traps baited with the aggregation pheromone in the early and mid-season, but less so in the late season. Traps baited with MDT captured more nymphs and adults in the late-season compared with traps baited with the pheromone alone likely because of the difference in loading rates, leading to a dose-dependent response in captures. In the USA, MDT is generally not attractive to adults in the early season, but becomes more attractive in the late-season, while it is attractive to nymphs season-long (Leskey et al. 2015a, Weber et al. 2014a). While this study was not able to assess the combined stimuli in the early season, the pattern of captures for MDT and the aggregation pheromone alone in all parts of the season was similar to the responses found in the introduced range in the USA, and the combined stimuli also performed similarly in the native range in the mid- and late-season compared to the introduced range. In other words, the population-level pattern observed in the introduced range of the USA is essentially the same as that in the native range of H. halys in the Republic of Korea (Table 2).
In 2015, the attraction of H. halys adults and nymphs to traps baited with MDT alone was not as robust in the mid- and late-season compared with 2014. However, there was still enhanced attraction when traps contained the MDT combined with the H. halys aggregation pheromone on adult captures. Prior research suggests that only a small amount of MDT is required to create a synergistic effect on attraction (Leskey et al. 2015a; Weber et al. 2014a). Moreover, there are several factors that may have influenced the overall attractiveness of MDT lures in 2015. For example, the longevity of field-aged MDT lures used in a prior study showed significant attenuation in attraction of H. halys adults and nymphs to traps over a 7-week period deployment period. (Joseph et al. 2013). Specifically, 3-week-old MDT lures started to capture fewer H. halys than fresh lures, and lures that were 4–7 weeks old captured statistically equivalent numbers of adults to traps without an MDT lure. Other factors such as dose (Mason et al. 1990), air velocity and temperature (Van der Kraan and Ebbers 1990), and the underlying genetics of the behavioral perception of the pheromone (Roelofs et al. 1987) may affect lure performance.
Importantly, traps baited with a combination of the H. halys aggregation pheromone and MDT pheromone synergist should be useful for monitoring, and detecting H. halys worldwide. Adult captures indicated a statistically significant increase, i.e., synergism, when stimuli were combined, while nymphal captures indicated an additive response only, though nymphal captures may be less reliable for measuring synergy based on their dispersal capacity compared with adults and their tendency not to be evenly distributed, but instead to be aggregated or clumped on select host plants. Subsequently, nymphal captures are more dependent on sampling location and the specific native hosts surrounding crop production areas, which may have resulted in the premature depletion of local nymphal populations within the distance of attraction for H. halys to these semiochemicals. However, with demonstrated effectiveness in attraction both in the USA and now in the Republic of Korea, there is strong evidence that the pheromone-based monitoring and management developed in the USA could be applicable globally. This is a salient point, because in Asia, H. halys is one of the top five most serious stink bug pests, undergoing periodic outbreaks that are mostly managed through applications of pyrethroids and neonicotinoids (Lee et al. 2013). Production systems in Asia may benefit from the use of pheromone and MDT synergists in threshold-based (Short et al. 2016) or perimeter-based (Blaauw et al. 2015; Morrison et al. 2016b) management for H. halys. For other countries, such as New Zealand (MacLellan 2013) or Australia (Australian Government 2015), sensitive pheromone traps for H. halys will likely be important in exclusion/detection programs. This may also include more convenient trap styles to increase ease of monitoring and surveillance (Morrison et al. 2015). This is important as New Zealand has declared H. halys one of their top six pests species of concern (MacLellan 2013). One question yet to be addressed, however, is when diapausing H. halys begin to respond to traps baited with pheromonal stimuli. Most hitchhiking H. halys are believed to be in diapause (MacLellan 2013), and it is unknown when these individuals will begin to respond to pheromonal stimuli when inadvertently moved to regions with abiotic conditions, including day length and temperature, that are conducive to diapause termination (Lee et al. 2013). However, because H. halys adults have been captured in traps baited with the aggregation pheromone alone (Khrimian et al. 2014) or in combination with MDT (Leskey et al. 2015a) in the early season, it appears that as adults become active after diapause termination, they also become responsive to these stimuli.
The population of H. halys in the eastern USA has been traced back to Beijing, China (Haye et al. 2015); however, its native range includes the Republic of Korea (Lee et al. 2013). In this study, we have evaluated field-level responses of the population in the Republic of Korea, which are similar to the data collected on the field-level responses of the population in the USA (Leskey et al. 2015a, b, c). While we did not analyze the underlying genetics in the Asian population, it is generally observed that the genetic diversity in the native range is higher than in an introduced range for an invasive species (reviewed in Lee 2002), and at least some portions of the haplotypes present in Asian population appear to be responding to the H. halys aggregation pheromone and MDT. Moreover, we documented reliable mid- and late-season captures of H. halys with the combined stimuli. Taken together with prior work showing that even in Europe (part of the introduced range), there is significant variability in the haplotypes present, we suggest the high likelihood that these combined stimuli will be effective at trapping and monitoring H. halys in other areas of its introduced and native range. Further work investigating the underlying genetic architecture of H. halys in Asia may help shed light on which specific haplotypes are responding, but this was beyond the scope of the current work.
The findings of H. halys are different from those of other species, which may sometimes exhibit host races that respond differently to the same semiochemicals. For example, in the historically invasive European corn borer, Ostrinia nubilalis (Lepidoptera: Pyralidae), there are three pheromone races with one responding primarily to Z11-14:OAc, E11-14:OAc, or a mix of each (Linn et al. 1997). Other species, such as the apple maggot fly, Rhagoletis pomonella (Diptera: Tephritidae), exhibit regional differences in their response to other olfactory cues, for example with populations responding to the feeding cue ammonium carbonate on the west coast (Yee et al. 2014), but not in the eastern USA (Morrison et al. 2016a). In the current study, we found no evidence of pheromone races or regional differentiation for H. halys.
In terms of non-target insect captures, there were less than 300 individuals spread across 36 traps that were sampled 13 times during 2014. Non-target captures are always a consideration when conducting pheromone-based trapping, because it adds time to the counting process for the target insect taxon (e.g., Kelly et al. 2013). However, our study suggests that non-target organisms will not impede monitoring of H. halys, nor result in excessive mortality of other insects. Notably, different lures caught a unique community of arthropods, though MDT and the H. halys aggregation pheromone seemed to capture more similar communities of non-target insects compared to the unbaited control (Fig. 5).
In addition to being attracted to its own aggregation pheromone, P. stali exhibited cross-attraction to the H. halys aggregation pheromone. This is similar to the cross-attraction of H. halys to P. stali’s aggregation pheromone, MDT (Weber et al. 2014a). In fact, cross-attraction to aggregation pheromones from heterospecifics is a common phenomenon among Pentatomidae (e.g., Tillman et al. 2010, Weber et al. 2014b; reviewed in Wertheim et al. 2005) and may allow other species to locate favorable resources. For example, there is cross-attraction between the pheromone of Piezodorus hybnei (Gmelin) (Hemiptera: Pentatomidae) and that of its main competitor R. pedestris (Endo et al. 2010). Interestingly, R. pedestris, one of the main stink bug pests in Republic of Korea (Son et al. 2000), did not exhibit any cross-attraction to either MDT or the H. halys aggregation pheromone in our study, despite catching 1.5 times more individuals in traps overall compared to Plautia stali.
Natural enemies often utilize volatile signals given off by their prey or the plants that their prey is associated with (reviewed in Vet and Dicke 1992). Now that attraction of H. halys to the combination of its aggregation pheromone and synergist MDT has been established in its introduced and native range, and it is important to understand whether we can manipulate the H. halys natural enemy community using semiochemicals. In other stink bug species, such as Euschistus heros, the pheromone for the species also acted as a kairomone for its egg parasitoid (Borges et al. 2011). In the USA, MDT acts as a kairomone for the tachinid parasitoid Euclytia flava (Townsend) (Aldrich et al. 2007) and the predatory digger wasp, Astata occidentalis (Hymenoptera: Sphecidae) (Cottrell et al. 2014; Millar et al. 2001). With the spread of the main Asian egg parasitoid of H. halys, Trissolcus japonicus (Hymenoptera: Platygastridae), in the introduced range in the USA (Herlihly et al. 2016; Talamas et al. 2015), it is increasingly important to understand whether the H. halys aggregation pheromone can also be used as a kairomone to aid pest management. Moreover, it is a possibility that some of the native predators that have been observed attacking H. halys in the USA (e.g., Morrison et al. 2016c) may also use kairomones or other herbivore-induced plant volatiles to locate egg masses. The identification of kairomones and other semiochemicals that may be involved in recruiting the natural enemy community of H. halys should be elucidated in the future.
Overall, we have established the attractiveness of the H. halys aggregation pheromone and pheromone synergist in the native range of H. halys. This suggests that much of the work being done around the world with pheromone-based tools may be transferrable across borders. In particular, these pheromone-based tools may be useful for worldwide exclusion and detection measures for H. halys. However, there are still outstanding topics that require attention, including when H. halys begin to respond to these stimuli as they break diapause, how the natural enemy community responds to these stimuli in its introduced range, and whether we can use that knowledge to help increase pest management for this destructive invasive species.
Authors’ contribution
TL, DL, and YP conceived and designed the study. CP and BS carried out the study and checked traps. WM analyzed the data and wrote the manuscript. All authors revised, read, and approved the manuscript.
References
Adachi I, Uchino K, Mochizuki F (2007) Development of a pyramidal trap for monitoring fruit-piercing stink bugs baited with Plautia crossota stali (Hemiptera: Pentatomidae) aggregation pheromone. Appl Entomol Zool 42:425–431
Adati T, Tatsuki S (1999) Identification of female sex pheromone of the legume pod borer, Maruca vitrata and antagonistic effects of geometrical isomers. J Chem Ecol 25:105–115
Aldrich JR, Hoffmann MP, Kochansky JP, Lusby WR, Eger JE, Payne JA (1991) Identification and attractiveness of the major pheromone component for Nearctic Euschistus spp. stink bugs (Heteroptera: Pentatomidae). Environ Entomol 20:477–483
Aldrich JR, Khrimian A, Camp M (2007) Methyl 2,4,6-decatrienoates attract stink bugs and tachinid parasitoids. J Chem Ecol 33:801–815
Anderson MJ (2008) ControlChart: a FORTRAN computer program for calculating control charts for multivariate response data through time, based on a chosen resemblance measure. University of Auckland, Auckland
Australian Government (2015) Brown marmorated stink bug: emergency measures for break bulk and containerised vehicles, machinery, automotive parts, and tyres. Department of Agriculture and Water Resources, Australian Government, Canberry City, Australia
Baker AJ, Moeed A (1987) Rapid genetic differentiation and founder effect in colonizing populations of common mynas (Acridotheres tristis). Evolution 41:525
Blaauw BR, Polk D, Nielsen AL (2015) IPM-CPR for peaches: incorporating behaviorally-based methods to manage Halyomorpha halys and key pests in peach. Pest Manag Sci 71:1513–1522
Blossey B, Notzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83(5):887
Borges M, Moraes MCB, Peixoto MF, Pires CSS, Sujii ER, Laumann RA (2011) Monitoring the Neotropical brown stink bug Euschistus heros (F.) (Hemiptera: Pentatomidae) with pheromone-baited traps in soybean fields. J Appl Entomol 135:68–80
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Cottrell TE, Landolt PJ, Zhang Q-H, Zack RS (2014) A chemical lure for stink bugs (Hemiptera: Pentatomidae) is used as a kairomone by Astata occidentalis (Hymenoptera: Sphecidae). Fla Entomol 97:233–237
Dixon P (2003) VEGAN, a package of R functions for community ecology. J Veg Sci 14:927–930
Dlugosch KM, Parker IM (2008) Founding events in species invasions: genetic variation, adaptive evolution, and the role of multiple introductions. Mol Ecol 17:431–449
Endo N, Sasaki R, Muto S (2010) Pheromonal cross-attraction in true bugs (Heteroptera): Attraction of Piezodorus hybneri (Pentatomidae) to its pheromone versus the pheromone of Riptortus pedestris (Alydidae). Environ Entomol 39:1973–1979
Funayama K (2003) Outbreak and control of stink bugs in apple orchards. Jpn Agric Tech 47:35–39
Funayama K (2008) Seasonal fluctuations and physiological status of Halyomorpha halys (Stal) adults captured in traps baited with synthetic aggregation pheromone of Plautia crossota stali Scott. Jpn J Appl Entomol Z 52:69–75
Gariepy TD, Fraser H, Scott-Dupree CD (2014) Brown marmorated stink bug (Hemiptera: Pentatomidae) in Canada: recent establishment, occurrence, and pest status in southern Ontario. Can Entomol 146:579–582
Haye T, Gariepy T, Hoelmer K, Rossi J-P, Streito J-C, Tassus X, Desneux N (2015) Range expansion of the invasive brown marmorated stinkbug, Halyomorpha halys: an increasing threat to field, fruit and vegetable crops worldwide. J Pest Sci 88:665–673
Herlihly MV, Talamas EJ, Weber DC (2016) Attack and success of native and exotic parasitoids on eggs of Halymorpha halys in three Maryland habitats. PLoS ONE 11:e0150275
Hoebeke ER, Carter ME (2003) Halyomorpha halys (Stål) (Heteroptera: Pentatomidae): a polyphagous plant pest from Asia newly detected in North America. Proc Entomol Soc Wash 105:225–237
Ioannidis P et al (2014) Rapid transcriptome sequencing of an invasive pest, the brown marmorated stink bug, Halyomorpha halys. BMC Genom 15:738–760
Jones AL, Jennings DE, Hooks CRR, Shrewsbury PM (2014) Sentinel eggs underestimate rates of parasitism of the exotic brown marmorated stink bug, Halyomorpha halys. Biol Control 78:61–66
Joseph SV, Bergh CJ, Wright SE, Leskey TC (2013) Factors affecting captures of brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae), in baited pyramid traps. J Entomol Sci 48:43–51
Kelly JA, Avery TS, Stewart DT, Cutler CG, Gaul SO, MacKenzie KE, Hillier NK (2013) Non-target Gelechiidae and Noctuidae attraction to Aroga trialbamaculella (Lepidoptera: Gelechiidae) pheromone-based trapping systems. Can Entomol 145:48–52
Khrimian A, Shearer PW, Zhang A, Hamilton GC, Aldrich JR (2008) Field trapping of the invasive brown marmorated stink bug, Halyomorpha halys, with geometric isomers of methyl-2,4,6-decatrienoate. J Agric Food Chem 56:197–203
Khrimian A et al (2014) Discovery of the aggregation pheromone of the brown marmorated stink bug (Halyomorpha halys) through the creation of stereoisomeric libraries of 1-bisabolen-3-ols. J Nat Prod 77:1708–1717
Lara J et al (2016) Biological control program is being developed for brown marmorated stink bug. Calif Agric 70:15–23
Lee CE (2002) Evolutionary genetics of invasive species. Trends Ecol Evol 17:386–391
Lee D-H (2015) Current status of research progress on the biology and management of Halyomorpha halys (Hemiptera: Pentatomidae) as an invasive species. Appl Entomol 50:277–290
Lee DH, Leskey TC (2015) Flight behavior of foraging and overwintering brown marmorated stink bug, Halyomorpha halys (Hemiptera: Pentatomidae). Bull Entomol Res 105:566–573
Lee DH, Short BD, Joseph SV, Bergh JC, Leskey TC (2013) Review of the biology, ecology, and management of Halyomorpha halys (Hemiptera: Pentatomidae) in China, Japan, and the Republic of Korea. Environ Entomol 42:627–641
Leskey TC et al (2012a) Pest status of the brown marmorated stink bug, Halyomorpha halys in the USA. Outlooks Pest Manag 23:218–226
Leskey TC, Short BD, Butler BR, Wright SE (2012b) Impact of the invasive brown marmorated stink bug, Halyomorpha halys (Stål), in mid-Atlantic tree fruit orchards in the United States: Case studies of commercial management. Psyche 2012:1–14
Leskey TC, Wright SE, Short BD, Khrimian A (2012c) Development of behaviorally-based monitoring tools for the brown marmorated stink bug (Heteroptera: Pentatomidae) in commercial tree fruit orchards. J Entomol Sci 47:76–85
Leskey TC, Short BD, Lee DH (2014) Efficacy of insecticide residues on adult Halyomorpha halys (Stål) (Hemiptera: Pentatomidae) mortality and injury in apple and peach orchards. Pest Manag Sci 70:1097–1104
Leskey TC et al (2015a) Attraction of the invasive Halyomorpha halys (Hemiptera: Pentatomidae) to traps baited with semiochemical stimuli across the United States. Environ Entomol 44:746–756
Leskey TC, Khrimian A, Weber DC, Aldrich JR, Short BD, Lee DH, Morrison WR (2015b) Behavioral responses of the invasive Halyomorpha halys (Stal) to traps baited with stereoisomeric mixtures of 10,11-epoxy-1-bisabolen-3-ol. J Chem Ecol 41:418–429
Leskey TC, Lee DH, Glenn DM, Morrison WR (2015c) Behavioral responses of the invasive Halyomorpha halys (Stal) (Hemiptera: Pentatomidae) to light-based stimuli in the laboratory and field. J Insect Behav 28:674–692
Linn CE, Young MS, Gendle M, Glover TJ, Roelofs WL (1997) Sex pheromone blend discrimination in two races and hybrids of the European corn borer moth, Ostrinia nubilalis. Physiol Entomol 22:212–223
MacLellan R (2013) Brown marmorated stink bug: a potential risk to New Zealand. Surveillance 40:34–36
Mason LJ, Jansson RK, Heath RR (1990) Sampling range of male sweetpotato weevils (Cylas formicarius elegantulus) (Summers) (Coleoptera: Curculionidae) to pheromone traps: influence of pheromone dosage and lure age. J Chem Ecol 16:2493–2502
Millar JG, Rice RE, Steffan SA, Daane KM, Cullen E, Zalom FG (2001) Attraction of female digger wasps, Astata occidentalis Cresson (Hymneoptera: Sphecidae) to the sex pheromone of the bug Thyanta pallidovirens (Hemiptera: Pentatomidae). Pan Pac Entomol 77:244–248
Morrison WR, Cullum JP, Leskey TC (2015) Evaluation of trap designs and deployment strategies for capturing Halyomorpha halys (Hemiptera: Pentatomidae). J Econ Entomol 108:1683–1692
Morrison WR, Lee D-H, Reissig WH, Combs D, Leahy K, Poling B, Leskey TC (2016a) Inclusion of specialist and generalist stimuli in attract-and-kill programs: their relative efficacy in apple maggot fly (Diptera: Tephritidae) pest management. Environ Entomol 45:974–982
Morrison WR, Lee DH, Short BD, Khrimian A, Leskey TC (2016b) Establishing the behavioral basis for an attract-and-kill strategy to manage the invasive Halyomorpha halys in apple orchards. J Pest Sci 89:81–96
Morrison WR, Mathews CR, Leskey TC (2016c) Frequency, efficiency, and physical characteristics of predation by generalist predators of brown marmorated stink bug (Hemiptera: Pentatomidae) eggs. Biol Control 97:120–130
Morrison WR, Poling B, Leskey TC (2016d) The consequences of sublethal exposure to insecticide on the survivorship and mobility of Halyomorpha halys (Hemiptera: Pentatomidae). Pest Manag Sci. doi:10.1002/ps.4322
Nielsen AL, Holmstrom K, Hamilton GC, Cambridge J, Ingerson-Mahar J (2013) Use of black light traps to monitor the abundance, spread, and flight behavior of Halyomorpha halys (Hemiptera: Pentatomidae). J Econ Entomol 106:1495–1502
Park G-M, Jang S-A, Choi S-H, Park C-G (2010) Attraction of Plautia stali (Hemiptera: Pentatomidae) to different amounts of its aggregation pheromone and the effect of different dispensers. Korean J Appl Entomol 49:123–127
Phillips BL, Brown GP, Shine R (2010) Life-history evolution in range-shifting populations. Ecology 91:1617–1627
Porter SD, Savignano DA (1990) Invasion of polygyne fire ants decimates native ants and disrupts arthropod community. Ecology 71:2095–2106
Rice KB et al (2014) Biology, ecology, and management of brown marmorated stink bug (Hemiptera: Pentatomidae). J Integr Pest Manag 5:1–13
Roelofs WL et al (1987) Sex pheromone production and perception in European corn borer moths is determine by both autosomal and sex-linked genes. Proc Natl Acad Sci USA 84:7585–7589
Short BD, Khrimian A, Leskey TC (2016) Pheromone-based decision support tools for management of Halyomorpha halys in apple orchards: development of a trap-based treatment threshold. J Pest Sci. doi:10.1007/s10340-016-0812-1
Son C-K, Park SG, Hwang Y-H, Choi B-S (2000) Field occurrence of stink bug and its damage in soybean. Korean J Crop Sci 45:405–410
Sugie H et al (1996) Identification of the aggregation pheromone of the brown-winged green bug, Plautia stali Scott (Heteroptera: Pentatomidae). Appl Entomol Zool 31:427–431
Talamas EJ, Herlihly MV, Dieckhoff C, Hoelmer K, Buffington M, Bon M-C, Weber DC (2015) Trissolcus japonicus (Ashmead) (Hymenoptera: Scelionidae) emerges in North America. J Hymenopt Res 43:119–128
R Core Development Team (2015) R: a Language and environment for statistical computing. http://www.R-project.org/
Tillman PG, Aldrich JR, Khrimian A, Cottrell TE (2010) Pheromone attraction and cross-attraction of Nezara, Ascosternum, and Euschistus spp. stink bugs (Heteroptera: Pentatomidae) in the field. Environ Entomol 39:610–617
Tsutsui ND, Case TJ (2001) Population genetics and colony structure of the argentine ant (Linepithema humile) in its native and introduced ranges. Evolution 55:976
Van der Kraan C, Ebbers A (1990) Release rates of tetradecen-1-ol acetates from polymeric formulations in relation to temperature and air velocity. J Chem Ecol 16:1041–1058
Vet LEM, Dicke M (1992) Ecology of infochemical use by natural enemies in a tritrophic context. Annu Rev Entomol 37:141–172
Vilà M, Maron JL, Marco L (2005) Evidence for the enemy release hypothesis in Hypericum perforatum. Oecologia 142:474–479
Weber DC, Leskey TC, Walsh GC, Khrimian A (2014a) Synergy of aggregation pheromone with methyl (E, E, Z)-2,4,6-decatrienoate in attraction of Halyomorpha halys (Hemiptera: Pentatomidae). J Econ Entomol 107:1061–1068
Weber DC, Walsh GC, DiMeglio AS, Athanas MM, Leskey TC, Khrimian A (2014b) Attractiveness of harlequin bug, Murgantia histrionica, aggregation pheromone: field response to isomers, ratios, and dose. J Chem Ecol 40:1251–1259
Wermelinger B, Wyniger D, Forster B (2008) First records of an invasive bug in Europe: Halyomorpha halys Stal (Heteroptera: Pentatomidae), a new pest on woody ornamentals and fruit trees? Mitt Schweiz Entomol Ges 81:1–8
Wertheim B, van Baalen E-J, Dicke M, Vet LEM (2005) Pheromone-mediated aggregation in nonsocial arthropods: an evolutionary ecological perspective. Annu Rev Entomol 50:321–346
Xu J, Fonseca DM, Hamilton GC, Hoelmer KA, Nielsen AL (2013) Tracing the origin of US brown marmorated stink bugs, Halyomorpha halys. Biol Invasions 16:153–166
Yee WL, Nash MJ, Goughnour RB, Cha DH, Linn CE, Feder JL (2014) Ammonium carbonate is more attractive than apple and hawthorn fruit volatile lures to Rhagoletis pomonella (Diptera: Tephritidae) in Washington State. Environ Entomol 43:957–968
Zangerl AR, Berenbaum MR (2005) Increase in toxicity of an invasive weed after reassociation with its coevolved herbivore. Proc Natl Acad Sci 102:15529–15532
Acknowledgements
We would like to thank J.J. Ahn (West Virginia University), J. Cullum (USDA), J.T. Goldner, Dr. Sang-Ku Lee, and M.M. McKinney for their excellent technical assistance. This work was funded by a Cooperative Research Program for between Agricultural Science and the Technology Development and the Rural Development Administration, Republic of Korea (Project No. PJ008946), Cooperative Research Agreement between the Rural Development Administration of the Republic of Korea and West Virginia University, USA (Project No. 10016999) and National Research Foundation of Korea (Grant No. NRF-2014R1A1A2056993). This research was also funded, in part, by a USDA-NIFA SCRI CAP Grant#2011-51181-30937. Mention of trade names or commercial products in this publication is solely for the purpose of providing scientific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest in publishing this work.
Human and animal rights
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants by any of the authors.
Additional information
Communicated by T. Haye.
Special Issue: The brown marmorated stink bug Halyomorpha halys - an emerging pest of global concern
Rights and permissions
About this article
Cite this article
Morrison III, W.R., Park, CG., Seo, B.Y. et al. Attraction of the invasive Halyomorpha halys in its native Asian range to traps baited with semiochemical stimuli. J Pest Sci 90, 1205–1217 (2017). https://doi.org/10.1007/s10340-016-0816-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-016-0816-x