Abstract
The mosquito larvicidal activity of Zanthoxylum monophyllum leaf essential oil (EO) and its major chemical constituents was tested against the three mosquito vectors Anopheles subpictus, Aedes albopictus and Culex tritaeniorhynchus. In the EO of Z. monophyllum, it contains 36 compounds with the two major compounds being Germacrene D-4-ol (19.40 %) and α-Cadinol (12.30). The larvicidal activity of the essential oil against An. subpictus, Ae. albopictus and Cx. tritaeniorhynchus was determined and LC50 values were estimated at 41.50, 45.35 and 49.01 µg/mL, respectively. The two major compounds Germacrene D-4-ol and α-Cadinol were tested for acute toxicity against larvae of the three mosquito vectors. Germacrene D-4-ol showed a significantly higher efficacy compared to α-Cadinol. While LC50 for Germacrene D-4-ol ranged from 6.12 to 7.26 µg/mL, LC50 values for α-Cadinol were estimated in the range from 10.27 to 12.28 µg/mL. The EO, Germacrene D-4-ol and α-Cadinol were found safer to the non-target organism Gambusia affinis (LC50 = 4234.07, 414.05 and 635.12 µg/mL, respectively), which was manifested in the high suitability of the index/predator safety factor value, ranging from 86.36 for the least sensitive larvae of Cx. tritaeniorhynchus to 102.02 for the most sensitive larvae of An. subpictus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Key message
-
Germacrene D-4-ol and α-Cadinol were the major chemical compounds of the essential oil obtained from Zanthoxylum monophyllum.
-
The larvicidal activity of the essential oil against An. subpictus, Ae. albopictus and Cx. tritaeniorhynchus was determined.
-
LC50 values against the three mosquito vectors ranged for essential oil from 41.5 to 49.01 µg/mL.
-
Germacrene D-4-ol showed a significantly higher efficacy compared to α-Cadinol. While LC50 for Germacrene D-4-ol ranged from 6.12 to 7.26 µg/mL, LC50 values for α-Cadinol were estimated in the range from 10.27 to 12.28 µg/mL.
-
The essential oil, Germacrene D-4-ol and α-Cadinol were found safer for the non-target organism—fish Gambusia affinis.
Introduction
The negative impact of some pesticides on the environment and non-target organisms, including humans, as well as problems with the development of mosquito populations resistant to active insecticidal substances (Hemingway and Ranson 2000; Soderlund et al. 2002; McCaffery and Nauen 2006), are currently forcing us to seek new alternatives for protection against vectors (Govindarajan et al. 2013; Benelli 2015a, b; Benelli et al. 2015). Alternatives include the use of secondary plant metabolites such as active substances in so-called botanical insecticides (Regnault-Roger et al. 2012; Pavela 2015a, b).
Some plant metabolites have been shown to offer the possibility of being used as repellents, adulticides and larvicides (Isman 2006; Regnault-Roger et al. 2012). Suitably selected plant extracts provide an advantage because of the minimum toxicity for non-target organisms, rapid and natural degradation of the residues (Regnault-Roger et al. 2012; Isman and Grieneisen 2014).
The group of essential oils (EOs) is also among highly efficient plant substances. EOs are defined as volatile oils that have strong aromatic components and give a distinctive odour, flavour or scent to an aromatic plant. EOs are produced by more than 17,500 aromatic plant species commonly belonging to many angiospermic families, e.g. Lamiaceae, Rutaceae, Myrtaceae, Zingiberaceae and Asteraceae (Regnault-Roger et al. 2012). As shown by many studies, EOs exhibit repellent, antiovipositional and insecticidal effects against many mosquito species (Seyoum et al. 2003; Isman 2006; Pavela 2011; Govindarajan 2011, 2012, 2016). Currently, EOs are used primarily as repellents (Nerio et al. 2010). However, within the framework of the development of new botanical insecticides, EOs are also generally considered to be highly suitable for the development of botanical larvicides (Govindarajan 2010; Dias and Moraes 2014; Benelli 2015a; Govindarajan et al. 2016). Application of larvicides has been shown to represent a very important strategy that can result in an efficient reduction in the population density of adult mosquitoes in the treated area, resulting in a considerable reduction in infectious pressure (Walker and Lynch 2007; WHO 2012). Although EOs are generally considered to be safe for the environment (Regnault-Roger et al. 2012, Isman and Grieneisen 2014), only a few studies exist that have focused on their effects concerning both target and non-target organisms.
The Zanthoxylum genus (Rutaceae Juss.) is made up of about 250 species of trees and shrubs in the world’s tropical and temperate regions (Pirani 1993). It is economically very important as a source of edible fruits and raw materials for both the cosmetics and perfume industries, as well as culinary applications. Z. monophyllum is a shrub or tree that grows 4–12 m in some countries of the Americas at elevations of 300–1000 m (Quesada and Fernández 2005). Previous chemical studies of this plant (bark, stems, wood and leaves) have indicated the isolation of several components, including coumarins, alkaloids, benzenoids, triterpenes, sterols and lignans (Cuca et al. 1998; De García et al. 1989; Adesina 2005). Z. monophyllum has been used popularly as a colourant (De García et al. 1989) and has been used in Venezuelan traditional medicine to treat runny nose, jaundice and ophthalmia, in addition to being used as an anaesthetic (Gómez et al. 2007; Gilani et al. 2010; Barkatullah Ibrar et al. 2013).
Our study is therefore focused on the efficacy of an EO obtained from Zanthoxylum monophyllum (Lam.) P. Wilson (syn. Fagara furfuracea (Tul.) Engl.; F. monophylla Lam.) against the larvae of three important vectors and one representative of non-target organisms. We report the larvicidal activity of the essential oil of the Z. monophyllum leaf, grown in India, and its major chemical constituents against three important mosquito species, the malaria vectors Anopheles subpictus Grassi, the dengue vector Aedes albopictus (Skuse) and the Japanese encephalitis vector Culex tritaeniorhynchus Giles. A representative of the larvivorous fish of the Poeciliidae family—Gambusia affinis (S. F. Baird & Girard) was chosen as the natural predator of the mosquito larvae (Shahi et al. 2015).
Materials and methods
Plant material and extraction of essential oil
Z. monophyllum was gathered in Nilgiris, Western Ghats, Tamil Nadu, India, and authenticated at the Department of Botany at Annamalai University. The herbarium of the Plant Phytochemistry Division at the Department of Zoology, Annamalai University, contains voucher specimens on deposit. The EO was extracted by hydro-distilling 3 kg of fresh leaves in a Clevenger apparatus for 8 h. Using a separating funnel, the oil layer was separated from the aqueous phase. The resulting essential oil produced was dried over anhydrous sodium sulphate and stored in the dark at 4 °C until the testing phase.
Gas chromatography
A Varian gas chromatograph equipped with a flame ionization detector and BPI (100 % dimethyl polysiloxane) capillary column was used for the gas chromatography (GC). The carrier gas used was helium, at a flow rate of 1.0 mL/min and 8 psi inlet pressure. The temperature was programmed from 60 to 220 °C at 5 °C/min and had a final hold time of 6 min. Temperatures for the injector and detector were kept at 250 and 300 °C, respectively. The sample (0.2 μL) was injected using a split ratio of 1:20.
Gas chromatography–mass spectrometry
Gas chromatography–mass spectrometry (GC–MS) was carried out using an Agilent 6890 GC that was also equipped with a 5973 N mass selective detector and an HP-5 (5 % phenyl methylpolysiloxane) capillary column. The temperature of the oven was programmed from 50 to 280 °C at a rate of 4 °C/min and held at this temperature for 5 min. The temperatures of the inlet and interface were 250 and 280 °C, respectively. Helium, at a flow rate of 1.0mL/min (constant flow), was used as the carrier gas. The sample (0.2 μL) was injected using a split of 20:1. Electron impact mass spectrometry was performed at 70 eV. The temperatures for the ion source and quadrupole were maintained at 230 and 150 °C, respectively. The compounds were identified by comparing their retention indices and mass spectra with those found in the NIST 98.1 and Mass Finder 3.1 commercial libraries. The integration area of the chromatographer was used to calculate the concentration of each component of the essential oil.
Mosquitoes
The test organism, Aedes albopictus, Anopheles subpictus and Culex tritaeniorhynchus, was reared in a laboratory (Vector Control Laboratory at the Department of Zoology, Annamalai University, India). The larvae were fed with dog biscuits and yeast powder at a 3:1 ratio. Adults were provided with a 10 % sucrose solution and a 1-week-old chick for blood feeding. The mosquitoes were held at 28 ± 2 °C and 70–85 % relative humidity, with a photo period of 12-h light and 12-h dark.
Acute toxicity against mosquito larvae
Following (WHO 2005), the larvicidal activity of the Z. monophyllum EO and its major compounds, Germacrene D-4-ol and α-Cadinol, was evaluated. Germacrene D-4-ol and α-Cadinol were purchased from Sigma-Aldrich (Germany). The EO was tested at 20, 40, 60, 80 and 100 µg/mL. Furthermore, every compound was tested at different concentrations within the range of 3–25 µg/mL. To obtain each of the desired concentrations, the EO and/or individual compounds were dissolved in 1 mL DMSO and then diluted in 249 mL of filtered tap water. The control was prepared using 1 mL of DMSO in 249 mL of water. Each solution received twenty early third-instar larvae, and five replicates were performed for each concentration. No food was given to the larvae during the first 24 h following exposure, and after this time larval mortality was recorded.
Biotoxicity on non-target organisms
The method of Sivagnaname and Kalyanasundaram (2004) was used to assess the effects on non-target organisms. The effects of the EO and its major compounds, Germacrene D-4-ol and α-Cadinol, were tested against the non-target organism Gambusia affinis. The species were field collected and kept in separate concrete tanks, 85 cm in diameter and 30 cm deep, which contained water at 27 ± 3 °C. Fish were fed according to the needs of the mosquito larvae. The relative humidity in the experimental room was 80 ± 5 %. Young, immature fish around 1–2 cm in size were selected for the experiment.
A concentration of 50 times higher than the LC50 dose for mosquito larvae was used when evaluating the EO and its major compounds, Germacrene D-4-ol and α-Cadinol. Ten fish were separated (to avoid cannibalism) in 500 mL glass containers with 250 mL of tap water that had been sitting for a while. Ten replicates were performed for each concentration as well as four replicates for the untreated controls. After 48 h of exposure, the non-target organisms were inspected for mortality and other abnormalities such as lethargy as well as reduced swimming activity. Furthermore, in order to understand the extract’s post-treatment effect on survival and swimming activity, the exposed non-target organisms were kept under continuous observation for 10 days. No food was given to the fish for the first 48 h. Subsequently, the fish were fed with mosquito larvae ad libitum every other day until the end of the observation.
Data analysis
Experimental tests demonstrated that more than 20 % of the controlled mortality was discharged and repeated. When the controlled mortality reached 1–20 %, the observed mortality was corrected by Abbott’s formula (Abbott 1925) and an LC50, LC90 regression equation, and a 95 % confidence limit (CI95) was calculated using probit analysis (Finney 1971).
In experiments evaluating biotoxicity on non-target organisms, the Suitability Index (SI) for each non-target species was calculated using the following formula (Deo et al. 1988):
Results
Essential oil
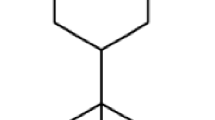
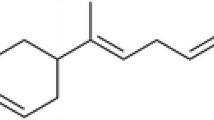
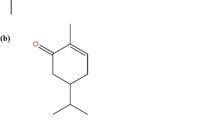
The yield of the Z. monophyllum leaf’s essential oil was 16.4 mL/kg (=1.64 %) of fresh weight. The results of EO chemical analysis are shown in Table 1. A total of 36 compounds representing 96.2 % of the essential oil were identified, with the major compounds being (see Fig. 1) Germacrene D-4-ol (19.4 %) and α-Cadinol (12.3 %). The percentage of compositions concerning the remaining 34 compounds ranged from 0.7 to 3.2 %.
Larvicidal activity
The larvicidal activity of the essential oil and its two major compounds against the mosquito vectors An. subpictus, Ae. albopictus and Cx. tritaeniorhynchus is presented in Tables 2, 3 and 4.
The comparison of LC50 and LC90 values revealed different sensitivities of individual mosquito species. The highest sensitivity to the EO was shown by the larvae of An. subpictus, for which the LC50 and LC90 values were estimated at 41.50 and 82.19 µg/mL, respectively, while the least sensitivity was shown by the larvae of Cx. tritaeniorhynchus, for which the LC50 and LC90 values were estimated at 49.01 and 92.08 µg/mL, respectively (Table 2). However, the difference in larval sensitivity to the EO among individual mosquito species was not significant (P = 0.05) because the CI95 values overlap.
Upon comparison of the estimated confidence intervals, we can claim that the two major compounds extracted from the Z. monophyllum EO were tested individually against the three mosquito vector larval populations. Germacrene D-4-ol showed a significantly higher efficacy (see Table 3) compared to α-Cadinol (see Table 4). While LC50 for Germacrene D-4-ol ranged from 6.12 to 7.26 µg/mL, LC50 values for α-Cadinol were estimated to be in the range from 10.27 to 12.28 µg/mL. Different sensitivities of the larvae of individual mosquito species were also determined for both tested major compounds. The larvae of An. subpictus showed the highest sensitivity again, with their LD50 estimated as 6.12 and 10.27 µg/mL for Germacrene D-4-ol and α-Cadinol, respectively, while the least sensitivity was shown by the larvae of Cx. tritaeniorhynchus with their LD50 estimated as 7.26 and 12.28 µg/mL for Germacrene D-4-ol and α-Cadinol, respectively. However, given the overlapping confidence limits we must note that the determined differences in larval sensitivity were not significant (P ≤ 0.05).
Although the determined interspecies differences in larval mortality were not significant, based on the repeated trend the tested mosquito species can be ordered according to their sensitivity from the most sensitive to the least sensitive larvae as follows: An. subpictus < Ae. albopictus < Cx. tritaeniorhynchus.
Effect against a non-target organism
The results of the effect on a non-target organism revealed that the EO and its major chemical compounds are safe for the predatory fish, Gambusia affinis. Given that the LC50 for G. affinis was estimated at 4234.07 µg/mL (Table 5), the EO can be considered completely safe for this fish species, which was manifested in a high suitability index/predator safety factor, ranging from 86.39 for the least sensitive larvae of Cx. tritaeniorhynchus to 102.02 for the most sensitive larvae of An. subpictus (Table 6).
Compared to the EO, the fish was significantly more sensitive to both major substances, Germacrene D-4-ol and α-Cadinol (Table 5). Where LC50 was estimated at 414.05 and 635.12 µg/mL, these substances can be considered only mildly toxic for fish as the index/predator safety factor was higher than 50.
The EO as well as both tested substances caused acute toxicity in the fish in very high doses. Surviving fish were monitored for 10 days; no further mortality or other abnormalities in the bodies of the G. affinis fish were observed throughout this period.
Discussion
In our study, we explored the efficacy of an EO obtained from the fresh leaves of Zanthoxylum monophyllum in terms of acute toxicity for the larvae of three mosquito species as the target carriers of serious diseases and for their natural predator, the Gambusia affinis fish.
Although the obtained essential oil contained a complex mixture of 36 substances, only two sesquiterpenoid alcohols, Germacrene D-4-ol (19.4 %) and α-Cadinol (12.3 %), showed a major share. Although it is known that these substances can be contained in the EOs of other plant species of this genus, such high shares of these sesquiterpenes have not yet been seen, based on available literature (Setzer et al. 2005; Tiwary et al. 2007; Pitasawat et al. 2007; Prieto et al. 2011; Rodríguez-Guzmán et al. 2011; Borah et al. 2012; Liu et al. 2014). Nevertheless, it should be noted that only a few studies have focused in detail on the composition of an EO obtained from the leaves of Z. monophyllum. The different composition of the EOs may be due to various influences such as the time of harvest, method of obtaining the EO, plant chemotype, course of climatic and pedological conditions, and many other factors (Burt 2004; Bakkali et al. 2008; Pavela et al. 2009).
The EO that we studied was highly toxic for the larvae of all three mosquito species, where LC50 was lower than 50 ppm and LC90 was <100 ppm. This is an excellent efficacy compared to other EOs studied for their larvicidal activity. The larvicidal efficacy of EOs is very well summarized in a study by Pavela (2015a), which provides us with a summary of present-day knowledge of larvicidal efficacy of EOs based on two criteria: (i) LC50 ≤ 100 ppm and (ii) their chemical composition is known. Although many studies have been published, it is clear from this study that only the EOs obtained from 122 plant species of 26 families showed LC50 ≤ 100 ppm, and among these, only 77 showed LC50 ≤ 50 ppm. These plants also included several species of the genus Zanthoxylum (Tiwary et al. 2007; Liu et al. 2014; Pitasawat et al. 2007; Borah et al. 2012). Although our estimated lethal concentrations of the EOs were more than 100 times higher than, for example, that of organophosphates or pyrethroids (Shahi et al. 2015), the complex mixture of active substances suggests that various mechanisms of action which, through their co-action, may prevent the development of resistance when repeated (Bakkali et al. 2008).
Larvicidal efficacy has been known to depend primarily on EO composition. In particular, where individual chemotypes of the plant species can synthesize various secondary metabolites (Pavela et al. 2009), the chemical composition of the EOs has to be known. The EO that we tested was a complex mixture of many substances that showed excellent larvicidal efficacy along with a very low toxicity for the representative of non-target organisms—G. affinis, where the achieved suitability index reached almost 100. Substances with such a high SI are generally considered completely safe for non-target organisms, in this case, for fish (Deo et al. 1988). Other authors have studied the toxicity of an EO obtained from Z. alatum Roxb. in an oral administration on mice, calculated to be 6124 μL/kg body weight during a safety profile assessment (Prakash et al. 2012). This provides evidence of the very low toxicity of this EO for mammals.
The pure major substances that we tested showed not only significantly higher toxicity for the larvae but also a higher toxicity for the fish as well. The significantly higher efficacy of pure substances than of the EO can probably be explained by the fact that although Germacrene D-4-ol and α-Cadinol were detected as the major substances, their contents in the EO reached only 19.4 and 12.3 %, respectively. This also demonstrates that these major substances had a major share in the insecticidal efficacy of the EO obtained from Z. monophyllum. Most likely, there was no significant synergistic effect between the substances that would increase the efficacy of the complex mixture of the tested EO. Nevertheless, this phenomenon was not the subject of this study. Mutual synergistic or antagonistic relationships among major substances will be the focus of our further studies given that, as has been proven, a suitable mutual ratio of some aromatic substances may significantly increase their insecticidal efficacy (Pavela 2014b, 2015b). Both tested compounds can still be considered relatively safe for the fish, given that a concentration of 20 ppm, causing 100 % mortality of the mosquito larvae, resulted in no mortality of the G. affinis, which is often used as a model non-target organism due to its potential use as predators of mosquito larvae (Lawrence et al. 2016). Although it is difficult (with respect to the different compositions of the EOs) to compare the efficacy on fish achieved by us with other authors, it can be noted that as found also by other authors, EOs and their active substances show only a weak toxicity for fish. For example, Stroh et al. (1998) presented the results of toxicological tests on juvenile salmonids where they estimated LC50 for thyme oil, α-terpineol and eugenol as 21.1, 6.5 and 67.6 mg/mL, respectively. As already discovered, some EOs may be relatively friendly to non-target organisms, including some representatives of aquatic plankton, provided that they are used properly (Pavela 2014a), although some EOs may also have a negative effect on aquatic plankton as shown in some studies (Conti et al. 2014).
However, we are aware that further toxicological tests on non-target organisms will be needed to ensure that potential botanical larvicides based on the EO from Z. monophyllum are completely safe for the environment. In addition, the effect of lethal and sublethal doses on target and non-target organisms will be a topic of our further study, given that some EOs have shown the ability to reduce the fecundity, fertility and vitality of insects (Pavela 2007). This knowledge could be used to determine the optimal application dose of potential larvicides based on this EO. Similarly, it will be important to study the mutual synergistic and antagonistic relationships of substances contained in the EO, given that the mutual ratios of individual substances contained in EOs could lead to either a significant reduction or rise in biological efficacy (Pavela 2014b, 2015b).
Conclusion
The promising results of this study indicate that the EO obtained from the fresh leaves of Zanthoxylum monophyllum, with the sesquiterpenoid alcohols Germacrene D-4-ol and α-Cadinol constituting major shares, can be advantageous as an active substance for the development of botanical larvicides against malaria, dengue, and the Japanese encephalitis mosquito vectors. As shown in our tests, this EO provides excellent larvicidal effects and is non-toxic for fish; moreover, it was found that the EO has a relatively high yield (more than 16 mL from 1 kg of fresh leaves). This plant is also available in endemic areas of the vectors.
Author contribution
MG conceived and designed research. RP wrote the manuscript. All authors read and approved the manuscript.
References
Abbott WS (1925) A method of computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Adesina SK (2005) The Nigerian Zanthoxylum; chemical and biological values. Afr J Tradit Complement Altern Med 2:282–301
Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils—a review. Food Chem Toxicol 46:446–475
Barkatullah Ibrar M, Muhammad N, Ur-Rehman I, Rehman MU, Khan A (2013) Chemical composition and biological screening of essential oils of Zanthoxylum armatum DC leaves. J Clin Toxicol 3:172. doi:10.4172/2161-0495.1000172
Benelli G (2015a) Research in mosquito control: current challenges for a brighter future. Parasitol Res 114:2801–2805
Benelli G (2015b) Plant-borne ovicides in the fight against mosquito vectors of medical and veterinary importance: a systematic review. Parasitol Res 114:3201–3212
Benelli G, Murugan K, Panneerselvam C, Madhiyazhagan P, Conti B, Nicoletti M (2015) Old ingredients for a new recipe? Neem cake, a low-cost botanical by-product in the fight against mosquito-borne diseases. Parasitol Res 114:391–397
Borah R, Saikia K, Talukdar AK, Kalita MC (2012) Chemical composition and biological activity of the leaf essential oil of Zanthoxylum oxyphyllum. Planta Med 78:P_100
Burt S (2004) Essential oils: their antibacterial properties and potential applications in foods—a review. Int J Food Microbiol 94:223–253
Conti B, Flamini G, Cioni PL, Ceccarini L, Leonardi MM, Benelli G (2014) Mosquitocidal essential oils: are they safe against non-target aquatic organisms? Parasitol Res 113:251–259
Cuca LE, Martinez JC, Monache FD (1998) Constituyentes químicos de Zanthoxylum monophyllum. Rev Colomb Quim 27:17–27
De García LA, Calle J, Reguero MT, Nathan PJ (1989) Phytochemical study of Zanthoxylum monophyllum. Fitoterapia 60:447–448
Deo PG, Hasan SB, Majumdar SK (1988) Toxicity and suitability of some insecticides for household use. Int Pest Control 30:118–129
Dias N, Moraes DFC (2014) Essential oils and their compounds as Aedes aegypti L. (Diptera: Culicidae) larvicides: review. Parasitol Res 113:565–592
Finney DJ (1971) Probit analysis. Cambridge University Press, London
Gilani SN, Khan AU, Gilani AH (2010) Pharmacological basis for the medicinal use of Zanthoxylum armatum in gut, airways and cardiovascular disorders. Phytother Res 24:553–558
Gómez Y, Gil K, González E, Farías LM (2007) Actividad antifúngica de extractos orgánicos del árbol Fagara monophylla (Rutaceae) en Venezuela. Int J Trop Biol 55:767–775
Govindarajan M (2010) Chemical composition and larvicidal activity of leaf essential oil from Clausena anisata (willd.) Hook. F. Benth (Rutaceae) against three mosquito species. Asian Pac J Trop Med 3:874–877
Govindarajan M (2011) Larvicidal and repellent properties of some essential oils against Culex tritaeniorhynchus Giles and Anopheles subpictus Grassi (Diptera: Culicidae). Asian Pac J Trop Med 4:106–111
Govindarajan M, Sivakumar R, Rajeswari M, Yogalakshmi K (2012) Chemical composition and larvicidal activity of essential oil from Mentha spicata (Linn.) against three mosquito species. Parasitol Res 110:2023–2032
Govindarajan M, Sivakumar R, Rajeswary M, Yogalakshmi K (2013) Chemical composition and larvicidal activity of essential oil from Ocimum basilicum (L.) against Culex tritaeniorhynchus, Aedes albopictus and Anopheles subpictus (Diptera: Culicidae). Exp Parasitol 134:7–11
Govindarajan M, Rajeswary M, Hoti SL, Benelli G (2016) Larvicidal potential of carvacrol and terpinen-4-ol from the essential oil of origanum vulgare (lamiaceae) against Anopheles stephensi, Anopheles subpictus, Culex quinquefasciatus and culex tritaeniorhynchus (Diptera: Culicidae). Res Vet Sci. doi:10.1016/j.rvsc.2015.11.011
Hemingway J, Ranson H (2000) Insecticide resistance in insect vectors of human disease. Ann Rev Entomol 45:371–391
Isman MB (2006) Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu Rev Entomol 51:46–66
Isman MB, Grieneisen ML (2014) Botanical insecticide research: many publications, limited useful data. Trends Plant Sci 19:140–145
Lawrence C, Rutherford N, Hamilton R, Meredith D (2016) Experimental evidence indicates that native freshwater fish outperform introduced Gambusia in mosquito suppression when water temperature is below 25°C. Hydrobiologia 766:357–364
Liu XC, Liu QY, Zhou L, Liu QR, Liu ZL (2014) Chemical composition of Zanthoxylum avicennae essential oil and its larvicidal activity on Aedes albopictus Skuse. Trop J Pharm Res 13:399–404
McCaffery A, Nauen R (2006) The Insecticide Resistance Action Committee (IRAC): public responsibility and enlightened industrial self interest. Outlook Pest Manag 17:11–14
Nerio LS, Olivero-Verbela J, Stashenko E (2010) Repellent activity of essential oils: a review. Bioresour Technol 101:372–378
Pavela R (2007) Lethal and sublethal effects of thyme oil (Thymus vulgaris L.) on the house fly (Musca domestica Lin.). J Essent Oil Bear Pl 10:346–356
Pavela R (2011) Insecticidal properties of phenols on Culex quinquefasciatus Say and Musca domestica L. Parasitol Res 190:1547–1553
Pavela R (2014a) Insecticidal properties of Pimpinella anisum essential oils against the Culex quinquefasciatus and the non-target organism Daphnia magna. J Asia Pac Entomol 17:287–293
Pavela R (2014b) Acute, synergistic and antagonistic effects of some aromatic compounds on the Spodoptera littoralis Boisd. (Lep., Noctuidae) larvae. Ind Crop Prod 60:247–258
Pavela R (2015a) Essential oils for the development of eco-friendly mosquito larvicides: A review. Ind Crops Prod 76:174–187
Pavela R (2015b) Acute toxicity and synergistic and antagonistic effects of the aromatic compounds of some essential oils against Culex quinquefasciatus Say larvae. Parasitol Res 114:3835–3853
Pavela R, Vrchotova N, Triska J (2009) Mosquitocidal activities of thyme oils (Thymus vulgaris L.) against Culex quinquefasciatus (Diptera: Culicidae). Parasitol Res 105:1365–1370
Pirani JRA (1993) New species and new combination in Zanthoxylum (Rutaceae) from Brazil. Brittonia 45:154–158
Pitasawat B, Champakaew D, Choochote W (2007) Aromatic plant-derived essential oil: an alternative larvicide for mosquito control. Fitoterapia 78:205–210
Prakash B, Singh P, Mishra PK, Dubey NK (2012) Safety assessment of Zanthoxylum alatum Roxb. essential oil, its antifungal, antiaflatoxin, antioxidant activity and efficacy as antimicrobial in preservation of Piper nigrum L. fruits. Int J Food Microbiol 153:183–191
Prieto JA, Patiño OJ, Delgado WA, Moreno JP, Cuca LE (2011) Chemical composition, insecticidal, and antifungal activities of fruit essentials oils of free Colombian Zanthoxylum species. Chil J Agric Res 71:73–82
Quesada RM, Fernández JV (2005) Actualización de listado de especies arbóreas de uso forestal y otros usos en Costa Rica. Kurú: Revista Forestal (Costa Rica) 2:1–45
Regnault-Roger C, Vincent C, Arnason JT (2012) Essential oils in insect control: low-risk products in a high-stakes world. Ann Rev Entomol 57:405–424
Rodríguez-Guzmán R, Johansmann Fulks LC, Radwan MM, Burandt CL, Ross SA (2011) Chemical constituents, antimicrobial and antimalarial activities of Zanthoxylum monophyllum. Plant Med 77:1542–1544
Setzer WN, Noletto JA, Lawton RO, Haber WA (2005) Leaf essential oil composition of five Zanthoxylum species from Monteverde, Costa Rica. Mol Divers 9:3–13
Seyoum A, Killeen GF, Kabiru EW, Knols BGJ, Hassanali A (2003) Field efficacy of thermally expelled or live potted repellent plants against African malaria vectors in western Kenya. Tropic Med Int Health 8:1005–1011
Shahi M, Kamrani E, Salehi M, Habibi R, Hanafi-Bojd AA (2015) Native Larvivorous Fish in an endemic malarious area of Southern Iran, a biological alternative factor for chemical larvicides in malaria control program. Iran J Publ Health 44:1544–1549
Sivagnaname N, Kalyanasundaram M (2004) Laboratory evaluation of methanolic extract of Atlantia monophylla (Family: Rutaceae) against immature stages of mosquitoes and non-target organisms. Mem Inst Oswaldo Cruz 99:115–118
Soderlund DM, Clark JM, Sheets LP, Mullin LS, Piccirillo VJ, Sargent D, Stevens JT, Weiner ML (2002) Mechanisms of pyrethroid neurotoxicity: implications for cumulative risk assessment. Toxicol 171:3–59
Stroh J, Wan MT, Isman MB, Moul DL (1998) Evaluation of the acute toxicity to juvenile pacific coho salmon and rainbow trout of some plant essential oils, a formulated product, and the carrier. Bull Environ Contam Toxicol 60:923–930
Tiwary M, Naik SN, Tewary DK, Mittal PK, Yadav S (2007) Chemical composition and larvicidal activities of the essential oil of Zanthoxylum armatum DC (Rutaceae) against three mosquito vectors. J Vector Borne Dis 44:198–204
Walker K, Lynch M (2007) Contributions of Anopheles larval control to malaria suppression in tropical Africa: review of achievements and potential. Med Vet Entomol 21:2–21
WHO (World Health Organization) (2005) Guidelines for laboratory and field testing of mosquito larvicides. WHO/CDS/WHOPES/GCDPP/2005.13
WHO (World Health Organization) (2012) Global plan for insecticide resistance management in malaria vectors (GPIRM). http://whqlibdoc.who.int/Publications/2012/9789241564472_eng.pdf. Accessed 15 Apr 2012
Acknowledgments
The authors would like to thank Dr. R. Karuppasamy, Professor and Head of the Department of Zoology at Annamalai University for the laboratory facilities provided. We also acknowledge the cooperation of the staff members from the VCRC (ICMR), Pondicherry. Dr. Pavela, as co-author, would like to thank the Ministry of Agriculture of the Czech Republic for its financial support concerning botanical pesticide research (Project No. MZE RO0415).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
All applicable international and national guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards and practices of the institution at which the studies were conducted.
Additional information
Communicated by M. B. Isman.
Rights and permissions
About this article
Cite this article
Pavela, R., Govindarajan, M. The essential oil from Zanthoxylum monophyllum a potential mosquito larvicide with low toxicity to the non-target fish Gambusia affinis . J Pest Sci 90, 369–378 (2017). https://doi.org/10.1007/s10340-016-0763-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-016-0763-6