Abstract
Predation is the leading cause of avian reproductive failure and theory predicts clutch size reduction under high nest predation risk. However, the impact of nest predation on population dynamics is debated, and spatiotemporal variation in reproductive output is poorly understood due to the predominance of short-term, single-site studies. To this end, we investigated how island geography (size, forest cover, isolation), predation risk (White-tailed Eagle Haliaeetus albicilla abundance), climate (migration phenology, winter severity) and time trends affected early breeding success (inverse of early predation rate) and clutch size of endangered Baltic Common Eiders (Somateria mollissima). The study was conducted at two close sites (Tvärminne, Velkua) during 1991–2020. Local breeding numbers showed a similar annual decline. Interestingly, fecundity components were negatively correlated across sites: early breeding success was higher in Velkua and clutch size in Tvärminne. Island forest cover and exposure were the best determinants of early breeding success and clutch size (with only weak explanatory power for clutch size), but their effects were mediated by differential susceptibility to eagle predation risk. Clutch size increased in Tvärminne but decreased in Velkua. The former finding likely reflects increased breeder phenotypic quality due to selective disappearance at this high-predation site, whereas the latter finding may indicate increasingly compromised food intake. Only at the high-risk site did clutch size show the expected decline with increasing eagle predation risk, and the expected increase with increasing overhead protection (forest cover). We identify the least exposed forested islands, associated with the highest productivity, as prime targets of conservation.
Zuammenfassung
Prädationsrisiko und Eigenschaften Landschaft prägen die Fortpflanzungsrate einer gefährdeten Meeresente: zwei Teilpopulationen mit unterschiedlichen Nesträuber-Risiken
Prädation ist die Hauptursache für Misserfolge in der Fortpflanzung von Vögeln, und eine Theorie sagt, dass unter starker Nestprädation die Gelege immer kleiner werden. Die Auswirkungen der Nestprädation auf die Populationsdynamik sind jedoch umstritten, und die räumlich-zeitlichen Schwankungen der Fortpflanzungsleistung sind wegen der überwiegenden Zahl von Kurzzeitstudien an nur einzelnen Standorten kaum bekannt. Deshalb untersuchten wir, wie sich die geographische Lage einer Insel (Größe, Bewaldung, Isolation), das Prädationsrisiko (Vorhandensein des Seeadlers Haliaeetus albicilla), das Klima (Zugphänologie, Strenge des Winters) und die zeitlichen Abläufe auf den frühen Bruterfolg (invers zu früher Prädationsrate) und die Gelegegröße der in der Ostsee gefährdeten Eiderente (Somateria mollissima) auswirken. Die Studie wurde zwischen 1991 und 2020 an zwei nahe gelegenen Standorten (Tvärminne, Velkua; Finnland) durchgeführt. Die örtlichen Brutzahlen zeigten einen ähnlichen jährlichen Rückgang. Interessanterweise waren einzelne Faktoren des Fortpflanzungserfolgs zwischen den Standorten negativ korreliert: der frühe Bruterfolg war in Velkua höher und die Gelegegröße in Tvärminne. Die Bewaldung der Insel und ihre Exponiertheit waren die besten Bestimmungsgrößen für den frühen Bruterfolg und die Gelegegröße (mit nur schwacher Erklärungskraft für die Gelegegröße), aber ihre Auswirkungen wurden durch die unterschiedliche Gefährdung durch Seeadler beeinflusst. Die Gelegegröße nahm in Tvärminne zu, in Velkua dagegen ab. Der erste Befund spiegelt wahrscheinlich die erhöhte phänotypische Qualität der Brutvögel auf Grund der selektiven Auslese an diesem Ort mit hohem Prädationsrisiko wider, während der zweite Befund auf eine zunehmend schlechtere Nahrungsaufnahme hinweisen könnte. Nur am Hochrisikostandort zeigte sich der erwartete Rückgang der Gelegegröße mit zunehmendem Prädationsrisiko durch die Seeadler und der erwartete Anstieg der Gelegegrößen mit zunehmendem Schutz nach oben hin durch Bäume. Wir sehen die am wenigsten exponierten, bewaldeten Inseln mit der höchsten Produktivität als vorrangige Ziele für den Schutz.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nest predation is the most important factor causing reproductive failure in birds (Ricklefs 1969). Particularly, the effects of nest concealment (Götmark et al. 1995) and nest location (e.g. edge effects; Donovan et al. 1997) on nest predation rates have been extensively studied. Nevertheless, a long-held view is that spatiotemporal variation in nest predation has only limited impact on population dynamics in iteroparous animals, because adult survival is considered to have the greatest impact on population growth rate (Saether and Bakke 2000). However, some studies have challenged this view by showing that experimental removal of egg predators—or their risk cues—may have large effects on population growth (Zanette et al. 2011; Hanssen et al. 2013). Thus, nest predation may directly and indirectly have long-term consequences on future fecundity (Doligez and Clobert 2003; Zanette et al. 2011), breeding propensity (Öst et al. 2018), and lead to adaptive avoidance of the dangerous breeding site (Hanssen and Erikstad 2013).
Nest predation shows large variation both spatially (Ricklefs 1969; Martin 1995) and temporally (Thompson 2007; Cox et al. 2012), due to the combined influence of multiple ecological factors, such as variation in protective vegetation and predator community structure, which themselves may show synchrony among sites. As a broad generalization, variation in the macro- and microhabitat of nests tends to be associated with spatial variation in nest predation risk (Hollander et al. 2015). In fact, a substantial proportion (even exceeding 60%; Bulla et al. 2019) of the variation in nest predation may be explained by study site alone. In contrast, factors such as variation in predator abundance and community structure (Hoset and Husby 2018) and climate forcing (Ims et al. 2019) are more likely to induce temporal fluctuations in nest predation risk. However, these generalizations ignore the complex ways in which the breeding habitat may interact with predator–prey dynamics. For example, the habitat preferences of egg and adult predators may differ, which may force parents to trade off their own safety against that of their offspring when selecting nest sites (Götmark et al. 1995; Öst and Steele 2010). This may lead to a situation of elevated nest predation risk in the preferred nesting habitat—consistent with an ecological trap (Hollander et al. 2015).
Nest predation is closely linked to the evolution of clutch size, as life-history theory predicts that increased predation risk should reduce the value of current reproduction and thus favour reduced clutch size (Skutch 1949; Dillon and Conway 2018). However, adaptive predation risk-induced adjustments of clutch size may be confounded by undetected partial clutch predation (Öst et al. 2008), and predators of adults and eggs may further vary in their ability to inflict such partial loss. Given this complexity, a better understanding of the drivers of spatiotemporal variation in nest predation risk and the association between nest predation and clutch size requires long-term data collected from multiple breeding sites, but such data are rare.
Heeding this call, we investigate spatiotemporal variation in early breeding success and clutch size in the Common Eider (hereafter, Eider) (Somateria mollissima). The long-lived Eider is an excellent study species, because incubating ground-nesting female Eiders strongly rely on crypsis rather than flight, rendering them more susceptible to predation during breeding (e.g. Ramula et al. 2018). Furthermore, the focal Eider population in the northern Baltic Sea is currently facing intensified predation pressure from the White-tailed Eagle (Haliaeetus albicilla), the main native predator on adults, as well as from invasive mammalian predators, the American Mink (Neovison vison) and the Raccoon Dog (Nyctereutes procyonoides) (Öst et al. 2018; Jaatinen et al. 2022). Although predation is believed to be one of the main causes of the population decline of Eiders in the northern Baltic Sea (Tjørnløv et al. 2020; Öst et al. 2022), the significance of this factor has been debated (Sonne et al. 2012; Mörner et al. 2017). Variable fecundity may also constitute a major driver of population dynamics (Hario et al. 2009). Furthermore, because there is considerable variation in predation risk across the Baltic Sea (Tjørnløv et al. 2020), the drivers of decline may differ even between adjacent regions (Öst et al. 2016), particularly because colony-specific factors may largely determine local population dynamics (Jónsson et al. 2013). Adding interesting complexity is the fact that Eiders inhabit diverse breeding habitats, from small open skerries to slightly larger forested islands, which are interspersed close to each other (Kilpi and Lindström 1997; Öst et al. 2008). Not only do these habitats differ in average predation rates on nesting females and eggs (Ekroos et al. 2012), but they also attract different types of predators (Öst and Steele 2010). Open islands are preferred hunting areas for visually oriented eagles due to reduced opportunities for nest concealment, whereas forest cover may not afford significant protection from mammalian predators primarily hunting by scent.
We investigated the factors affecting early breeding success and clutch size, their spatiotemporal trends and synchrony in two close, but geographically separated (ca 120 km apart) subpopulations of Eiders (Tvärminne and Velkua) over the past 30 years (1991–2020). We further assessed long-term trends in breeding numbers at both sites, to explore whether these trends mirrored the local long-term variation in the observed fecundity components. Individual islands were used as the sampling unit, and we set out to explore the effects of habitat characteristics (island size, forest cover and geographical isolation) and predation risk (annual abundance of White-tailed Eagles) on the fecundity components, while also examining the presence of temporal trends. We also explored other possible drivers of nest predation and fecundity, i.e. annual spring migration phenology and previous winter severity. We expected a general increase in nest predation and a concomitant decrease in clutch size over time, the magnitude of which, however, should be site-specific and depend on the abundance of local predators.
Materials and methods
Study area and predator community composition
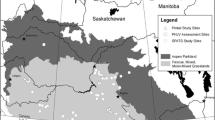
Fieldwork was conducted in two subpopulations in the south-western Finnish archipelago during 1991–2020 (Fig. 1). However, nest census data were missing from the year 1992. The study areas are located in the Velkua area (60.45°N, 21.60°E) in the Archipelago Sea, and outside Tvärminne (59.50°N, 23.15°E), western Gulf of Finland, ca. 120 km south-east of the Velkua area. The study sites in Velkua and Tvärminne consist of 27 and 35 islands, respectively, of varying size (Velkua: range 0.04–9.00 ha, mean = 1.30 ha ± 2.00 (SD), Tvärminne range 0.13–10.22 ha, mean = 1.70 ha ± 2.40 (SD)) and forest cover (Velkua range 0–65%, mean = 17.5% ± 22 (SD), Tvärminne range 0–53%, mean = 16% ± 20.20 (SD)). Both areas contain 13 skerries with no forest cover. A notable difference with respect to anthropogenic disturbance is that the islands in Tvärminne are protected as a nature conservation area, with access only allowed to researchers. In contrast, four of the islands in Velkua have summer cottages, although these are rarely visited during the Eider breeding season.
Eiders on the Finnish coast suffer from egg predation, primarily by Hooded Crows (Corvus corone cornix) and Ravens (Corvus corax). Adult Eiders are depredated by White-tailed Eagles, Eagle Owls (Bubo bubo), Minks and Raccoon Dogs (Öst et al. 2018), the last two of which also eat eggs. Minks and Raccoon Dogs have been removed from the Tvärminne study site within an invasive predator control scheme since 2011 (Jaatinen et al. 2022).
Island geographical properties
Island area was measured in ArcMap, using a vector-based standard shoreline map from the National Land Survey. Island forest cover was measured using Google Earth satellite maps (version 7.3.3). The geographical isolation of the breeding habitat from the surrounding islands (i.e. exposure) can affect predation pressure on Eiders, because more exposed islands may limit access to terrestrial predators (Kurvinen et al. 2016). To quantify island exposure, we used the Buffer Wizzard tool in ArcMap (version 10.8) to create buffer zones with 0.5, 1 and 2 km radius around each island (von Numers 2015) and measured the percentage of sea inside each buffer area. An examination of the means and variances of these island geographical characteristics revealed only two statistically significant differences between study sites. Thus, island exposure within a 0.5 km radius was significantly smaller in Tvärminne compared to Velkua (independent samples test: t60 = 2.4, P = 0.02), and exposure within a 2 km radius showed higher between-island variability in Velkua compared to Tvärminne (Levene’s test: P = 0.004). Neither the mean island size nor forest cover differed between localities.
Monitoring nest success and fecundity
Field work was conducted in both study areas from early May to mid-June, during the incubation period of female Eiders. The annual median date of monitoring was significantly later in Velkua (23 May) than in Tvärminne (18 May) (Wilcoxon signed-ranks test: z30 = − 3.51, p < 0.001). Island visits were timed to the latter half of incubation, as the tendency to abandon the nest decreases towards the end of incubation (Bolduc and Guillemette 2003). Each of the islands were searched systematically for all Eider nests. The clutch sizes in the active nests and the nest fate of non-active nests were recorded (Velkua: Ntotal = 6240 nesting attempts, range = 167–320, mean ± SD = 223 ± 54.7, Tvärminne: Ntotal = 13,631 nesting attempts, range = 237–638, mean ± SD = 470 ± 103.0). Active nests at the time of the census contained an incubating female or hatched eggshells, characterised by a leathery membrane. Depredated nests were characterized by the presence of shells broken into pieces with a thin bloody membrane or the complete absence of eggshells and hatched membranes (Öst and Steele 2010). The annual island-specific nest predation pressure was quantified by dividing the number of depredated nests on an island with the total number of nesting attempts (active, hatched, abandoned and depredated nests) on that island in each year (Öst et al. 2011), yielding the proportion of depredated nests at the time of census. Correspondingly, annual island-specific breeding success at the time of census (‘early breeding success’) was calculated as (1—annual island-specific nest predation risk) to be used in further analysis. Please note that we use the qualifier ‘early’ merely to distinguish it from final nest fate at hatching, which was unavailable in our study.
Incubation stage was estimated using an egg floatation test (Kilpi and Lindström 1997) in Tvärminne, but this method has not been used at Velkua. Instead, status as a full-laid clutch has been assigned based on presence of down in the nest. To verify that the presence of down is a reliable indicator of a completed clutch, egg floatation was carried out also at Velkua in 2016. Based on this pilot study, incubation in downy nests had progressed further (mean ± SD = 13.66 ± 5.75 days) than in nests lacking protective down (4.23 ± 6.10 days) (t187 = − 5.88, P < 0.001). Because Eiders complete their clutches within 5 days of starting incubation (Andersson and Waldeck 2006), it is thus legitimate to assume that downy nests represent completed clutches. In Tvärminne clutches were considered full-laid when the incubation stage was at least 8 days. This conservative threshold was applied to avoid any errors in clutch completion status even for the largest clutches (Öst et al. 2008). Because nests containing more than seven eggs (1.12% of all nests) necessarily contain parasitic eggs (Waldeck et al. 2004), such nests were excluded from the clutch size analysis.
Site-specific annual eagle abundance
For each study site, the abundance of White-tailed Eagles was estimated in each year. For Tvärminne, we used an eagle index based on the average number of eagles observed per day at Hanko Bird Observatory (HALIAS, ca 20 km west of Tvärminne) during the Eider breeding season, i.e. 1 April to 15 June (Jaatinen et al. 2011; Öst et al. 2022). The benefit of this index is that it considers the presence of all eagles irrespective of their age or breeding status. For the Velkua area, such an index was not available. Instead, we used the annual number of active eagle nests inside a 20 km radius around the study area (20 km radius measured around all the study islands and merged together), with nest locations provided by courtesy of the working group in support of the White-tailed Eagle in Finland (Stjernberg 1981). The annual eagle indices were standardized within each site (mean of 0, SD of 1), which reduces potential methodological bias caused by the use of different methods. Within-site standardization allowed us to investigate the effects of relative site-specific changes in eagle abundance, expressed on a common scale (standard deviations), on local nest success and fecundity of Eiders.
Climate indices and migration phenology
Winter climate has been shown to affect the migration, body condition and breeding phenology of Eiders (Lehikoinen et al. 2006). We used the extended annual PC-based winter (December–March) North Atlantic Oscillation (NAO) index (Hurrell 1995) and the maximum sea ice cover of the Baltic Sea (Seinä and Palosuo 1996) as integrated measures of winter climate. Annual Eider migration phenology was measured at HALIAS, where daily observations were conducted by 1–6 observers during a 4 h period starting at sunrise (Lehikoinen et al. 2008). The dates when 5% and 50% of the annual cumulative number of Eiders had migrated past HALIAS in spring were defined as the early and median phase of migration, respectively (Lehikoinen et al. 2006). Early annual migration phenology may correlate with early breeding phenology, which in turn is associated with larger clutches (Öst et al. 2022), but annual phenology may also be related to the level of nest predation risk (Öst and Jaatinen 2015).
Statistical analysis
First, we analysed the temporal trend in breeding numbers in both subpopulations, using a linear regression of the log-transformed total number of nesting attempts (response variable) as a function of year, calculated separately for each island at both sites. An island-by-island approach was necessitated by the fact that total annual breeding numbers contained gaps for individual islands and island-specific trends in breeding numbers show large variability within sites. Log-transformation permitted interpretation of the regression coefficient as an average annual percentage change. The absolute change in breeding numbers is tightly linked to the initial absolute numbers of breeders, and thus inappropriate for assessing temporal trends in breeding numbers.
Next, we used correlation analysis to explore the between-site temporal correlation in the mean annual early breeding success (for definition, see above) and mean annual clutch size. To test if any potential correlations between sites were a consequence of the study sites sharing a common time trend rather than truly matching the interannual variability in the variables of interest, we performed a partial correlation test, where mean annual early breeding success and clutch size were detrended by controlling for year.
To investigate the impact of island geography, eagle predation risk, winter climate and annual migration phenology on spatiotemporal variation in early breeding success and clutch size, we used generalized linear mixed models (GLMMs) and linear mixed models (LMMs), respectively. The random effect of island identity was added to models to account for repeated samples from the same islands. All explanatory variables were standardized (mean = 0, SD = 1) to facilitate comparison of effect sizes and model convergence; however, eagle abundance indices were standardized within study sites (see above). We then tested the explanatory variables used in each model for multicollinearity by calculating variance inflation factors (VIFs). We found four pairs of collinear variables: island size and forest cover, the winter NAO index and ice cover, the different levels of exposure and the two migration variables (VIF > 2). We therefore chose the variables that had the lower Akaike information criterion corrected for small sample size (AICc), when comparing the fit of univariate models. The following variables were discarded at this point: exposure within 1 km (both breeding success and clutch size analysis), exposure within 2 km for breeding success analysis, exposure within 0.5 km for clutch size analysis, island size, early migration (5% of the eiders) and NAO index. Model selection was then performed with the ‘dredge’ function in the R package MuMIn (Bartoń 2020), using AICc to find the most parsimonious model among the set of candidate models. If several equally plausible top-ranked models (ΔAICc < 2; Burnham and Anderson 2002) were identified, the one with the smallest number of parameters was chosen to avoid overfitting, given the known leniency of the AIC based approach in retaining model terms (Aho et al. 2014). The full model included the following candidate fixed effects: subpopulation, year, forest cover, exposure within 0.5 km (early breeding success analysis) and 2 km (clutch size analysis), eagle abundance, nest predation pressure, median laying date and a priori selected two-way interactions terms (Table S1). Potential subpopulation-specific three-way interactions were considered only if visual illustration of the final model led us to suspect such additional complexity; if so, such models were compared against the top-ranked model with lower order terms. We calculated the marginal R2 (R2m, the variance explained by fixed effects) and conditional R2 (R2c, the variance explained by both fixed and random effects) using the ‘r.squaredGLMM’ function in MuMIn. The variance explained by the random effect was calculated using the ‘var.random’ function (insight package; (Lüdecke 2019).
To analyse the influence of candidate explanatory variables on spatiotemporal variation in early breeding success, we used GLMMs with a binomial error distribution and Laplace estimation. The GLMM was fitted using maximum likelihood. The dependent variable comprised the total number of undepredated and depredated nests on each island during the nest census, combined with the ‘cbind’ function in R. The fixed effects for the model selection included subpopulation, year, forest cover, exposure at 0.5 km level, eagle abundance and the a priori selected two-way interactions. To elucidate the factors affecting spatiotemporal variation in clutch size at the two study sites, we constructed LMMs, assuming a normal error distribution and restricted maximum likelihood estimation. The following candidate explanatory variables were included in the full model: subpopulation, year, forest cover, exposure at 2 km level, eagle abundance, and median laying date, as well as the a priori selected two-way interactions. Early island-specific nest predation risk was included as an additional explanatory variable. This was done to account for any within-colony correlation between complete and partial clutch predation (e.g. Ackerman and Eadie 2003), the latter phenomenon of which is intricately connected to documented clutch sizes (Öst et al. 2008). All statistical analyses were performed using R 4.0.3 (R Core Team 2020).
Results
Subpopulation-specific trends
The mean annual island-specific trend in breeding numbers was significantly negative in both Tvärminne (b = − 0.032 ± 0.0098 (SE); one-sample t-test: t34 = −3.22, P = 0.003) and Velkua (b = − 0.014 ± 0.0066 (SE); t26 = − 2.16, P = 0.04). The mean annual trend in breeding numbers was numerically, although not statistically significantly, more negative in Tvärminne compared to Velkua (two-sample t-test: t60 = − 1.38, P = 0.17). Expressed as percentage change, the mean annual island-specific decline in Tvärminne was 3.11% [Bootstrap BCa 95% CI (− 5.11%, − 1.32%)], and 1.4% in Velkua [Bootstrap BCa 95% CI (− 2.60%, − 0.20%)].
Island-specific early breeding success
The annual proportion of early successful nests showed a significantly positive correlation between study sites (r29 = 0.54, P < 0.01). After controlling for year, the positive correlation between sites was no longer significant (rp29 = 0.18, P = 0.36). This indicates that a shared temporal trend in nest depredation risk is the most likely explanation for the positive correlation in the proportion of early successful nests between sites.
The final top-ranked model explaining spatiotemporal variation in early breeding success accounted for a high proportion of the variation (marginal R2m = 0.53 and R2c = 0.64). Early breeding success was explained by significant two-way interactions between subpopulation and forest cover, year and forest cover, year and exposure, forest cover and eagle abundance, as well as exposure and eagle abundance, respectively (Table 1). The random effect of island identity explained 5.7% of the variation in early breeding success. Early breeding success was generally lower in Tvärminne than in Velkua, but this difference tended to level off at high levels of forest cover (subpopulation × forest cover interaction; Fig. 2; Table 1). While early breeding success decreased over the years in both subpopulations, the decrease was strongest on islands with high forest cover (year × forest cover interaction; Fig. 3a, 3b; Table 1) and low exposure (year × island exposure interaction; Fig. 3c, 3d; Table 1). The relationship between annual eagle abundance and early breeding success was generally negative, but positive for islands with the highest forest cover (eagle abundance × forest cover interaction; Fig. 4a, 4b; Table 1) and lowest exposure (eagle abundance × island exposure interaction; Fig. 4c, 4d; Table 1).
Clutch size
The annual mean clutch sizes between the two study sites were not significantly correlated (r29 = 0.09, P = 0.63), with this correlation remaining non-significant after controlling for year (rp29 = 0.08, P = 0.68), indicating a lack of temporal trend between sites.
The final top-ranked model explained a small fraction of the variation in clutch size (R2m = 0.047 and R2c = 0.069). The final model included the main effects of early nest depredation risk and median laying date, and the significant two-way interactions between subpopulation and eagle abundance, exposure and eagle abundance, subpopulation and year, exposure and year and subpopulation and forest cover, respectively (Table 1). The random effect of island identity explained 6.4% of the variation in clutch size.
Smaller clutch sizes were associated with elevated early nest depredation risk (Fig. 5a, 5b; Table 1) and later breeding phenology (Fig. 5c, 5d; Table 1). Clutch size declined with eagle abundance in Tvärminne, but increased in Velkua (subpopulation × eagle abundance interaction; Fig. 6a; Table 1). Subpopulation-specific visual inspection revealed a poor fit of the two-way interactive effect of island exposure and eagle abundance on clutch size (Fig. 6b, 6c; Table 1). Thus, clutch size tended to decline with eagle abundance at Tvärminne, and more so for islands with higher exposure. Clutch size at Velkua was, however, positively associated with eagle abundance, particularly on islands with low exposure. Indeed, adding a three-way interaction (subpopulation × eagle abundance × island exposure, P < 0.05) to the original best-performing model significantly improved model fit (ΔAICc = 2).
Clutch size declined with eagle abundance in Tvärminne (TVZ), but increased in Velkua (VELK) (panel a). The decline in TVZ was most pronounced on islands with the highest exposure (panel b), while the positive association between clutch size and eagle abundance in VELK was most pronounced on low-exposure islands (panel c). Blue low, yellow medium, orange high (color figure online)
Clutch size slightly increased over time in Tvärminne, while clearly decreasing in Velkua (subpopulation × year interaction; Fig. 7a; Table 1). Furthermore, graphical inspection suggested a subpopulation-specific response of island exposure and year on clutch size (Fig. 7b, c; Table 1). Indeed, adding a three-way interaction (subpopulation × year × island exposure, P < 0.05) to the final lower order model improved model fit (ΔAICc = 3.4), and adding both three-way interaction terms (see above) further improved model fit (ΔAICc = 2.1). Clutch size in Tvärminne showed an increasing trend, exept for the lowest exposure islands (Fig. 7b). In contrast, clutch sizes decreased over time in Velkua, particularly on low-exposure islands (Fig. 7c). Finally, clutch size was unrelated to forest cover in Velkua, but showed a clear positive association with increasing forest cover in Tvärminne (subpopulation × forest cover interaction; Fig. 7d; Table 1).
Clutch size showed a slight increase over time in Tvärminne (TVZ), but decreased noticeably in Velkua (VELK) (panel a). Clutch size increased especially on the most exposed islands in TVZ (panel b), whereas the decrease in clutch size in VELK was most evident on low-exposure islands (panel c). Clutch size was unrelated to forest cover in Velkua (VELK), but showed a positive relationship with forest cover in Tvärminne (TVZ) (panel d). Blue low, yellow medium, orange high (color figure online)
Discussion
The nest predation hypothesis postulates clutch size reduction under high nest predation risk (Law 1979; Slagsvold 1982), and therefore clutch size should be inversely related to rates of nest predation. However, we show here that fecundity components may show negative rather than positive correlations across geographically proximate sites, as island-specific early nest predation risk was generally lower in Velkua, yet clutch size was higher in Tvärminne. The lower nest predation risk at Velkua is a conservative and hence robust finding, because the later timing of nest monitoring at Velkua (see Materials and methods) implies longer exposure to predation prior to monitoring. Nevertheless, exceptions to the nest-predation hypothesis are common, but the reason for the inconsistent relationship between clutch size and nest predation risk is unclear and appears species- and context-specific (Samaš et al. 2013). Our study shows that the expected negative correlation between fecundity and predation risk may be difficult or impossible to detect when extending the spatial scale beyond a single study site. Another noteworthy finding was the general lack of effect of preceding winter climate on breeding success and clutch size. This finding agrees with the conclusions of Öst et al. (2022), who suggest a three-decade-long shift from climate forcing to a system dominated by direct and indirect predation effects on vital rates of Baltic eiders.
Interestingly, differential subpopulation-specific early breeding success and fecundity translated into similar mean annual declines in local breeding numbers. The conventional explanation for this is that the population trend in long-lived species may primarily depend on adult survival rather than fecundity (Goodman 1974; Lebreton and Clobert 1991; Saether and Bakke 2000; Ekroos et al. 2012; Öst et al. 2016). Alternatively, however, the contrasting site-specific variation in fecundity components may serve to equalize trends in breeding numbers across sites. Although the Tvärminne subpopulation should experience a more negative trend in breeding numbers due to higher predation-induced nest failure (Figs. 1–3), this trend may at least in part be compensated for by increased fecundity (Fig. 7). Consequently, the net effect may be a smoothing out of differences in population growth between sites, as observed here.
Predictive performance of models of breeding success and clutch size
Our set of explanatory variables captured a substantial proportion (> 50%) of the variation in early breeding success, but only a minor fraction (ca. 5%) of the variation in clutch size. This disparity is consistent with empirical evidence suggesting that the habitat in the broad sense, that is, considering predation risk as a habitat component, often explains the majority of the variation in nest predation rates (Hollander et al. 2015; Bulla et al. 2019). The low explanatory power for predicting clutch size variation may reflect the presence of confounding factors associated with predation risk, but not accounted for. Such factors include the occurrence of breeding larids and corvids. The presence of breeding larids on islands positively affects breeding numbers of Eiders (Erwin 1979; Burger 1984; von Numers 1995; Väänänen 2000; Kurvinen et al. 2016), by offering protection from nest predation (Gerell 1985), and may thereby positively influence clutch size. Most gull species have shown decreasing population trends over the course of the study (EIONET 2020). In contrast, a large local Hooded Crow population should negatively affect clutch sizes, as crows are potent predators of unguarded clutches (e.g. Stien 2008), and partial clutch predation often eludes detection by human observers (Öst et al. 2008). However, the Hooded Crow has shown no change in annual nest numbers (log-transformed) over the study period in Velkua (linear regression: b = − 0.003, bootstrap BCa 95% CI [− 0.012, 0.007], t = − 0.58, P = 0.57), and a similar lack of a population trend may also apply to Tvärminne (pers. comm.). We therefore believe that more likely explanations include the influence of intrinsic life-history traits on clutch size, as well as factors affecting food availability of breeding females (see ‘Variation in clutch size’ below). Older mothers (Baillie and Milne 1982; Nisbet et al. 1984; Öst and Steele 2010) and those in good body condition (Daan et al. 1988; Erikstad et al. 1993) produce larger clutches, and clutch size is often individually repeatable (Batt and Prince 1979; Svensson 1997), indicating a link between individual quality and clutch size (Yoccoz et al. 2002). Furthermore, breeders are often non-randomly distributed in space: older individuals may prefer more concealed nest sites (Hatchwell et al. 1999; Öst and Steele 2010) that tend to offer higher offspring survivorship (Figs. 4a, b, Fig. 7d; Baillie and Milne 1982). However, selecting a concealed nest site—an adaptive life-history strategy if ageing mothers respond adaptively to their diminishing residual reproductive value (Stearns 1992; Roff 1992)—may incur a higher risk to the incubating parent once detected by the predator (Götmark et al. 1995; Wiebe and Martin 1998). Such state-dependent individual heterogeneity in clutch size decisions suggests that external environmental factors, such as those studied here, may only explain a limited portion of the variance in clutch size.
Variation in early breeding success
Island forest cover and exposure were the main factors affecting the variation in early breeding success. However, these habitat features did not affect breeding success directly (i.e. as main effects); rather their influence was mediated by differential susceptibility to eagle-induced predation risk (Fig. 4). Furthermore, as Tvärminne is an area of high predation risk (Figs. 2–4; Ekroos et al. 2012; Tjørnløv et al. 2020), also the finding that the differences in early breeding success between sites levelled off at the highest forest cover (Fig. 2), is likely to be linked to predator-avoidance. Thus, mean proportional nest cover has increased over time in Tvärminne (Mohring et al. 2022), presumably as an adaptive response to the increased predation threat by visually hunting eagles.
Intriguingly, while the relationship between annual eagle abundance and early breeding success was generally negative, as expected, it was positive on islands with the densest forest cover (Fig. 4a, b) and the lowest exposure (Fig. 4c, d). This seemingly counterintuitive finding clearly warrants further investigation. At this point, we may speculate that this result may arise as a by-product of variable nesting density. It is conceivable that high annual eagle predation risk may strengthen the tendency of female Eiders to increasingly nest on larger forested islands—also characterized by low exposure—as such a gradual large-scale change in nest-site selection patterns has been ongoing for the past decades (Ekroos et al. 2012). If so, per capita early nest depredation risk may be reduced simply due to the risk dilution afforded by breeding in denser aggregations (Inman and Krebs 1987). Current breeding densities are relatively low in a historical context, but it is noteworthy that island-specific nest success in Eiders tends to increase from low to intermediate nesting density (Åhlén and Andersson 1970; Öst et al. 2011; Pratte et al. 2016).
Variation in clutch size
The two sites showed strikingly different clutch size trends, with clutch size decreasing over time in Velkua but increasing in Tvärminne (Fig. 7a). Apart from potential predator-induced selective disappearance effects (see below), these differences may reflect different food availability at the breeding sites prior to breeding. In contrast to the conventional view that capital-breeding Eiders rely almost exclusively on stored reserves acquired prior to spring migration for producing their eggs (Meijer and Drent 1999), female Eiders utilize a considerable amount of local food resources to produce eggs, both in the northern Baltic (Hobson et al. 2015; Jaatinen et al. 2016) and in Arctic Canada (Sénéchal et al. 2011). Furthermore, this reliance on local food resources is likely to have become ever more important under the prevailing intense predation risk regime. This is because the delay between spring arrival and onset of breeding has increased over the past decades, a shift best explained by the increasing abundance of eagles (Öst et al. 2022). Velkua in the Archipelago Sea is increasingly affected by large-scale eutrophication. This promotes periodic hypoxia and accumulation of drifting algal mats, with negative repercussions on benthic fauna (e.g. Vahteri et al. 2000; Karlson et al. 2002), including blue mussels (Mytilus trossulus), the staple food of Eiders. Consequently, pre-breeding food conditions for Eiders in Velkua may have deteriorated during the study, whereas Tvärminne still supports thriving populations of blue mussels (Jaatinen et al. 2021), potentially contributing to the divergent temporal trends in clutch size between study areas.
Nevertheless, the decline in clutch size at the low predation site Velkua has been so dramatic (Figs. 7a, 7c) as to suggest the operation of additional unknown constraints. One potentially important but poorly understood factor is the recent invasion of the Mud Crab (Rhithropanopeus harrisii), which may negatively affect the availability of blue mussels for Eiders. The Mud Crab has strongly increased in the whole Velkua area beginning from ca. 2010 (Fowler et al. 2013). In contrast, the crab has not invaded the Tvärminne area during the study period (Mats Westerbom, pers. comm.). The Mud Crab is a potent predator of Blue mussels (Turoboyski 1973), potentially reducing the food base for breeding Eiders at Velkua (Forsström et al. 2015).
Increasing predation risk is expected to decrease investment in current reproduction (Zanette et al. 2011), but we only observed a negative effect of increasing eagle abundance on clutch size in Tvärminne (Fig. 5). However, it is noteworthy that we modelled the effects of relative within-site abundance of eagles on fecundity of Eiders. It is highly likely that absolute predation pressure by eagles on Eiders differs considerably between areas. The survival of female Eiders from Tvärminne is exceptionally low, compared to Eiders from the Baltic Sea/Wadden Sea flyway population (Tjørnløv et al. 2020) as well as in global comparison (Ekroos et al. 2012). In contrast, we still rarely find killed incubating females during nest monitoring in Velkua (M. Numers, pers. obs.). At Tvärminne, plumage characteristics indicate that non-breeding eagles may be responsible for the bulk of predation pressure on Eiders (pers. comm.). We may hypothesize that a threshold density of eagles exists, at which negative effects on fecundity become apparent, and that the density of resident breeding eagles alone may not be sufficient for reaching such a hypothetical threshold.
How can we reconcile the increasing predation pressure by eagles on Eiders in Tvärminne with the fact that clutch size here showed a slightly increasing trend over time (Fig. 6a)? An alternative theoretical possibility is that reduced life expectancy may favour increased reproductive effort (Roff 1992; Stearns 1992). However, the evidence supporting this view mainly derives from semelparous and/or short-lived species (Javoiš and Tammaru 2004; Kersch-Becker and Thaler 2015), whereas the bulk of empirical evidence in long-lived iteroparous organisms supports a negative relationship between predation risk and reproductive effort (Zanette et al. 2011 and references herein). However, several proximate mechanisms may be important in driving these patterns. One explanation could be that the proportion of females nesting on forested islands has gradually increased over time, due to selection imposed by eagle predation (Ekroos et al. 2012). Island forest cover, in turn, had a strong positive association with clutch size (Fig. 7d), as also shown previously in this subpopulation (Kilpi and Lindström 1997; Öst et al. 2008). However, this fact alone is unlikely to provide a satisfactory explanation. First, we statistically controlled for the effect of forest cover on clutch size, and island forest cover did not differ between sites. Second, the trend towards breeding closer to mainland and on larger islands has also occurred elsewhere in the Finnish archipelago (Kurvinen et al. 2016), yet clutch size showed a decreasing time trend in Velkua (Fig. 7a). We can also dismiss the possibility that the increasing clutch size in Tvärminne would be an artefact of predation-induced increase in conspecific brood parasitism. Conspecific brood parasitism is rare in this subpopulation (Waldeck et al. 2004), and based on empirical grounds, rates of conspecific brood parasitism should decrease, rather than increase, with progressively decreasing densities of breeders over time (e.g. Hoi et al. 2010), an effect also observed in Eiders (Robertson et al. 1992; Bjørn and Erikstad 1994; Waldeck et al. 2004).
Instead, the most plausible explanation for reconciling the dilemma of how progressively increasing eagle predation in Tvärminne (Öst et al. 2022) can reduce clutch sizes (Fig. 6a) concomitant with a temporal increase in clutch size (Fig. 7a), is strong selective (dis)appearance effects under high-predation danger. First, female annual mean body condition has increased over time in Tvärminne (Öst et al. 2018, 2022). This may reflect that only good-condition females opt to breeding under current predation levels (Öst et al. 2018), and/or that poor-condition females are more likely to fail early, prior to our clutch size monitoring (Lehikoinen et al. 2010; Mohring et al. 2021). Second, the proportion of first-time breeders has gradually declined over the past 3 decades (Öst et al. 2022). Taken together, and all else being equal, these long-term changes—an increase in breeder mean body condition and age—should translate into a larger clutch size (Baillie and Milne 1982; Erikstad et al. 1993), evident at the subpopulation level.
Conclusions and future directions
Early breeding success showed a common decreasing trend, and among our candidate predictors, island forest cover and exposure were the most important factors affecting breeding success and clutch size, albeit with low explanatory power for clutch size. However, the effects of island geography were indirect and mediated by habitat-specific susceptibility to eagle predation risk. Intriguingly, we found an opposite time trend in clutch size between sites, potentially reflecting differences in local food resources available to laying females and a progressive increase in the phenotypic quality of breeders under increased predation risk. Furthermore, several expected relationships between predation risk and fecundity components were only supported in the high-risk subpopulation. Thus, only in Tvärminne did clutch size decline with increasing eagle abundance, and, likewise, only there did breeding success and clutch size increase with forest cover, given that greater nest concealment should reduce nest discovery by visually hunting predators (Lima 2009).
Mitigating the impacts of increasing predation is particularly urgent in this endangered population (Ekroos et al. 2012; BirdLife International 2015). The ongoing increase in predation risk on both adults and offspring (Öst et al. 2022) is likely to continue in the near future. Our findings help to identify habitat characteristics correlated with high breeding success, which should therefore be targeted for future conservation. Thus, we advocate conservation measures particularly on the least exposed forested islands, associated with the highest early breeding success and clutch size in Tvärminne, the subpopulation subject to the highest predation pressure. Alarmingly, breeding success has shown the steepest temporal decline on islands with dense forest cover and low exposure (Figs. 3a-d), suggesting that predators are increasingly adjusting their distribution in response to changes in the distribution of their prey. This fact points to the urgency of conservation measures. Since such habitats occur close to mainland, inhabited by thriving populations of invasive mammalian predators, control of these alien predators may be the most cost-effective way to counter further population loss (Jaatinen et al. 2022).
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ackerman JT, Eadie JMA (2003) Current versus future reproduction: an experimental test of parental investment decisions using nest desertion by Mallards (Anas platyrhynchos). Behav Ecol Sociobiol 54:264–273. https://doi.org/10.1007/s00265-003-0628-x
Åhlén I, Andersson Å (1970) Breeding ecology of an Eider population on Spitsbergen. Ornis Scand 1:83–106. https://doi.org/10.2307/3676027
Aho K, Derryberry D, Peterson T (2014) Model selection for ecologists: the worldviews of AIC and BIC. Ecology 95:631–636. https://doi.org/10.1890/13-1452.1
Andersson M, Waldeck P (2006) Reproductive tactics under severe egg predation: an Eider’s dilemma. Oecologia 148:350–355. https://doi.org/10.1007/s00442-006-0374-7
Baillie SR, Milne H (1982) The influence of female age on breeding in the Eider Somateria mollissima. Bird Study 29:55–66. https://doi.org/10.1080/00063658209476738
Bartoń K (2020) MuMIn: multi-model inference. R Package Version 1(43):17
Batt BDJ, Prince HH (1979) Laying dates, clutch size and egg weight of captive Mallards. Condor 81:35–41. https://doi.org/10.2307/1367853
BirdLife International (2015) European Red List of birds. Office for Official Publications of the European Communities, Luxembourg
Bjørn TH, Erikstad KE (1994) Patterns of intraspecific nest parasitism in the high Arctic Common Eider (Somateria mollissima borealis). Can J Zool 72:1027–1034. https://doi.org/10.1139/z94-139
Bolduc F, Guillemette M (2003) Human disturbance and nesting success of Common Eiders: interaction between visitors and gulls. Biol Conserv 110:77–83. https://doi.org/10.1016/S0006-3207(02)00178-7
Bulla M, Reneerkens J, Weiser EL, et al. (2019) Comment on “Global pattern of nest predation is disrupted by climate change in shorebirds.” Science 364:eaaw8529. https://doi.org/10.1126/science.aaw8529
Burger J (1984) Grebes nesting in gull colonies: protective associations and early warning. Am Nat 123:327–337. https://doi.org/10.1086/284207
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Cox WA, Thompson FR III, Faaborg J (2012) Species and temporal factors affect predator-specific rates of nest predation for forest songbirds in the Midwest. Auk 129:147–155. https://doi.org/10.1525/auk.2012.11169
Daan S, Dijkstra C, Drent R, Meijer T (1988) Food supply and the annual timing of avian reproduction. Proc Int Ornithol Congr 19:392–407
Dillon KG, Conway CJ (2018) Nest predation risk explains variation in avian clutch size. Behav Ecol 29:301–311. https://doi.org/10.1093/beheco/arx130
Doligez B, Clobert J (2003) Clutch size reduction as a response to increased nest predation rate in the Collared Flycatcher. Ecology 84:2582–2588. https://doi.org/10.1890/02-3116
Donovan TM, Jones PW, Annand EM, Thompson Iii FR (1997) Variation in local-scale edge effects: mechanisms and landscape context. Ecology 78:2064–2075. https://doi.org/10.1890/0012-9658(1997)078[2064:VILSEE]2.0.CO;2
EIONET (2020) Population status and trends at the EU and member state levels. Article 12 web tool.
Ekroos J, Öst M, Karell P, Jaatinen K, Kilpi M (2012) Philopatric predisposition to predation-induced ecological traps: habitat-dependent mortality of breeding Eiders. Oecologia 170:979–986. https://doi.org/10.1007/s00442-012-2378-9
Erikstad KE, Bustnes JO, Moum T (1993) Clutch size determination in precocial birds: a study of the Common Eider. Auk 110:623–628. https://doi.org/10.2307/4088426
Erwin RM (1979) Species interactions in a mixed colony of Common Terns (Sterna hirundo) and Black Skimmers (Rynchops niger). Anim Behav 27:1054–1062. https://doi.org/10.1016/0003-3472(79)90053-8
Forsström T, Fowler AE, Manninen I, Vesakoski O (2015) An introduced species meets the local fauna: predatory behavior of the Crab Rhithropanopeus harrisii in the Northern Baltic Sea. Biol Invasions 17:2729–2741. https://doi.org/10.3391/ai.2013.8.1.10
Fowler AE, Forsström T, von Numers M, Vesakoski O (2013) The North American Mud Crab Rhithropanopeus harrisii (Gould, 1841) in newly colonized Northern Baltic Sea: distribution and ecology. Aquat Invasions 8:89–96. https://doi.org/10.3391/ai.2013.8.1.10
Goodman D (1974) Natural selection and a cost ceiling on reproductive effort. Am Nat 108:247–268. https://doi.org/10.1086/282906
Götmark F, Blomqvist D, Johansson OC, Bergkvist J (1995) Nest site selection: a trade-off between concealment and view of the surroundings? J Avian Biol 26:305–312. https://doi.org/10.2307/3677045
Hanssen SA, Erikstad KE (2013) The long-term consequences of egg predation. Behav Ecol 24:564–569. https://doi.org/10.1093/beheco/ars198
Hanssen SA, Moe B, Bårdsen BJ, Hanssen F, Gabrielsen GW (2013) A natural antipredation experiment: predator control and reduced sea ice increases colony size in a long-lived duck. Ecol Evol 3:3554–3564. https://doi.org/10.1002/ece3.735
Hario M, Mazerolle MJ, Saurola P (2009) Survival of female Common Eiders Somateria m. mollissima in a declining population of the northern Baltic Sea. Oecologia 159:747–756. https://doi.org/10.1007/s00442-008-1265-x
Hatchwell BJ, Russell AF, Fowlie MK, Ross DJ (1999) Reproductive success and nest-site selection in a cooperative breeder: effect of experience and a direct benefit of helping. Auk 116:355–363. https://doi.org/10.2307/4089370
Hobson KA, Jaatinen K, Öst M (2015) Differential contributions of endogenous and exogenous nutrients to egg components in wild Baltic Common Eiders (Somateria mollissima): A test of alternative stable isotope approaches. Auk 132:624–633. https://doi.org/10.1642/AUK-14-294.1
Hoi H, Darolová A, Krištofík J (2010) Conspecific brood parasitism and anti-parasite strategies in relation to breeding density in female Bearded Tits. Behaviour. https://doi.org/10.1163/000579510X511060
Hollander FA, van Dyck H, San Martin G, Titeux N (2015) Nest predation deviates from nest predator abundance in an ecologically trapped bird. PLoS ONE 10:e0144098. https://doi.org/10.1371/journal.pone.0144098
Hoset KS, Husby M (2018) Small between-year variations in nest predation rates are not related with between-year differences in predator identity. Ecoscience 25:199–208. https://doi.org/10.1080/11956860.2018.1427309
Hurrell JW (1995) Decadal trends in the North Atlantic oscillation: regional temperatures and precipitation. Science 269:676–679. https://doi.org/10.1126/science.269.5224.676
Ims RA, Henden J-A, Strømeng MA, Thingnes AV, Garmo MJ, Jepsen JU (2019) Arctic greening and bird nest predation risk across tundra ecotones. Nat Clim Change 9:607–610. https://doi.org/10.1038/s41558-019-0514-9
Inman AJ, Krebs J (1987) Predation and group living. Trends Ecol Evol 2:31–32. https://doi.org/10.1016/0169-5347(87)90093-0
Jaatinen K, Öst M, Lehikoinen A (2011) Adult predation risk drives shifts in parental care strategies: a long-term study. J Anim Ecol 80:49–56. https://doi.org/10.1111/j.1365-2656.2010.01757.x
Jaatinen K, Öst M, Hobson KA (2016) State-dependent capital and income breeding: a novel approach to evaluating individual strategies with stable isotopes. Front Zool 13:24. https://doi.org/10.1186/s12983-016-0157-x
Jaatinen K, Westerbom M, Norkko A, Mustonen O, Koons DN (2021) Detrimental impacts of climate change may be exacerbated by density-dependent population regulation in blue mussels. J Anim Ecol 90:562–573. https://doi.org/10.1111/1365-2656.13377
Jaatinen K, Hermansson I, Mohring B, Steele BB, Öst M (2022) Mitigating impacts of invasive alien predators on an endangered sea duck amidst high native predation pressure. Oecologia 198:543–552. https://doi.org/10.1007/s00442-021-05101-8
Javoiš J, Tammaru T (2004) Reproductive decisions are sensitive to cues of life expectancy: the case of a moth. Anim Behav 68:249–255. https://doi.org/10.1016/j.anbehav.2003.10.022
Jónsson JE, Gardarsson A, Gill JA, Pétursdóttir UK, Petersen A, Gunnarsson TG (2013) Relationships between long-term demography and weather in a sub-Arctic population of Common Eider. PLoS ONE 8:e67093. https://doi.org/10.1371/journal.pone.0067093
Karlson K, Rosenberg R, Bonsdorff E (2002) Temporal and spatial large-scale effects of eutrophication and oxygen deficiency on benthic fauna in Scandinavian and Baltic waters: a review. Oceanogr Mar Biol 40:427–489. https://doi.org/10.1201/9780203180594.ch8
Kersch-Becker MF, Thaler JS (2015) Plant resistance reduces the strength of consumptive and non-consumptive effects of predators on aphids. J Anim Ecol 84:1222–1232. https://doi.org/10.1111/1365-2656.12371
Kilpi M, Lindström K (1997) Habitat-specific clutch size and cost of incubation in Common Eiders, Somateria mollissima. Oecologia 111:297–301. https://doi.org/10.1007/s004420050238
Kurvinen L, Kilpi M, Nordström M, Öst M (2016) Drivers of decline and changed nest-site preference of the Baltic Eider: an island-level analysis from south-western Finland. Ornis Fenn 93:55–66
Law R (1979) Optimal life histories under age-specific predation. Am Nat 114:399–417. https://doi.org/10.1086/283488
Lebreton J-D, Clobert J (1991) Bird population dynamics, management and conservation: the role of mathematical modelling. In: Perrins CM, Lebreton J-D, Hirons GJM (eds) Bird population studies. University Press, Oxford, pp 105–125
Lehikoinen A, Kilpi M, Öst M (2006) Winter climate affects subsequent breeding success of Common Eiders. Glob Change Biol 12:1355–1365. https://doi.org/10.1111/j.1365-2486.2006.01162.x
Lehikoinen A, Christensen TK, Öst M, Kilpi M, Saurola P, Vattulainen A (2008) Large-scale change in the sex ratio of a declining Eider Somateria mollissima population. Wildl Biol 14:288–301. https://doi.org/10.2981/0909-6396(2008)14[288:LCITSR]2.0.CO;2
Lehikoinen A, Jaatinen K, Öst M (2010) Do female ornaments indicate quality in Eider ducks? Biol Lett 6:225–228. https://doi.org/10.1098/rsbl.2009.0744
Lima SL (2009) Predators and the breeding bird: Behavioral and reproductive flexibility under the risk of predation. Biol Rev 84:485–513. https://doi.org/10.1111/j.1469-185X.2009.00085.x
Lüdecke D, Makowski D (2019) insight: Easy Access to Model Information for Various Model Objects. R package version 0.2.0.
Martin TE (1995) Avian life history evolution in relation to nest sites, nest predation, and food. Ecol Monogr 65:101–127. https://doi.org/10.2307/2937160
Meijer T, Drent R (1999) Re-examination of the capital and income dichotomy in breeding birds. Ibis 141:399–414. https://doi.org/10.1111/j.1474-919X.1999.tb04409.x
Mohring B, Angelier F, Jaatinen K, Parenteau C, Öst M (2021) Parental investment under predation threat in incubating Common Eiders (Somateria mollissima): a hormonal perspective. Front Ecol Evol 9:637561. https://doi.org/10.3389/fevo.2021.637561
Mohring B, Angelier F, Jaatinen K, Steele BB, Lönnberg E, Öst M (2022) Drivers of within- and among-individual variation in risk-taking behaviour during reproduction in a long-lived bird. Proc R Soc B-Biol Sci 289:20221338. https://doi.org/10.1098/rspb.2022.1338
Mörner T, Hansson T, Carlsson L, Berg AL, Ruiz Muños Y, Gustavsson H, Mattson R, Balk L (2017) Thiamine deficiency impairs Common Eider (Somateria mollissima) reproduction in the field. Sci Rep 7:14451. https://doi.org/10.1038/s41598-017-13884-1
Nisbet ICT, Winchell JM, Heise AE (1984) Influence of age on the breeding biology of Common Terns. Colon Waterbirds 7:117–126. https://doi.org/10.2307/1521090
von Numers M (1995) Distribution, numbers and ecological gradients of birds breeding on small islands in the Archipelago Sea, SW Finland. Acta Zoologica Fennica 197:1–127
von Numers M (2015) Changes in distributions of selected vascular plants in a Baltic archipelago. Ann Bot Fenn 52:101–119. https://doi.org/10.5735/085.052.0212
Öst M, Jaatinen K (2015) Smart and safe? Antipredator behavior and breeding success are related to head size in a wild bird. Behav Ecol 26:1371–1378. https://doi.org/10.1093/beheco/arv093
Öst M, Steele BB (2010) Age-specific nest-site preference and success in Eiders. Oecologia 162:59–69. https://doi.org/10.1007/s00442-009-1444-4
Öst M, Wickman M, Matulionis E, Steele B (2008) Habitat-specific clutch size and cost of incubation in Eiders reconsidered. Oecologia 158:205–216. https://doi.org/10.1007/s00442-008-1139-2
Öst M, Lehikoinen A, Jaatinen K, Kilpi M (2011) Causes and consequences of fine-scale breeding dispersal in a female-philopatric species. Oecologia 166:327–336. https://doi.org/10.1007/s00442-010-1855-2
Öst M, Ramula S, Lindén A, Karell P, Kilpi M (2016) Small-scale spatial and temporal variation in the demographic processes underlying the large-scale decline of Eiders in the Baltic Sea. Popul Ecol 58:121–133. https://doi.org/10.1007/s10144-015-0517-y
Öst M, Lindén A, Karell P, Ramula S, Kilpi M (2018) To breed or not to breed: drivers of intermittent breeding in a seabird under increasing predation risk and male bias. Oecologia 188:129–138. https://doi.org/10.1007/s00442-018-4176-5
Öst M, Lehikoinen A, Jaatinen K (2022) Top–down effects override climate forcing on reproductive success in a declining sea duck. Oikos 2022:e08762. https://doi.org/10.1111/oik.08762
Pratte I, Davis SE, Maftei M, Mallory ML (2016) Aggressive neighbors and dense nesting: nest site choice and success in high-arctic Common Eiders. Polar Biol 39:1597–1604. https://doi.org/10.1007/s00300-015-1884-8
R Core Team (2020) A language and environment for statistical computing. R Found Stat Comput
Ramula S, Öst M, Lindén A, Karell P, Kilpi M (2018) Increased male bias in Eider ducks can be explained by sex-specific survival of prime-age breeders. PLoS ONE 13:e0195415. https://doi.org/10.1371/journal.pone.0195415
Ricklefs RE (1969) An analysis of nesting mortality in birds. Smithson Contrib Zool 9:1–48. https://doi.org/10.5479/si.00810282.9
Robertson GJ, Watson MD, Cooke F (1992) Frequency, timing and costs of intraspecific nest parasitism in the Common Eider. Condor 94:871–879. https://doi.org/10.2307/1369284
Roff D (1992) Evolution of life histories: theory and analysis. Chapman & Hall, New York
Saether B-E, Bakke O (2000) Avian life history variation and contribution of demographic traits to the population growth rate. Ecology 81:642–653. https://doi.org/10.1890/0012-9658(2000)081[0642:ALHVAC]2.0.CO;2
Samaš P, Grim T, Hauber ME, Cassey P, Weidinger K, Evans KL (2013) Ecological predictors of reduced avian reproductive investment in the southern hemisphere. Ecography 36:809–818. https://doi.org/10.2307/177366
Seinä A, Palosuo E (1996) The classification of the maximum annual extent of ice cover in the Baltic Sea 1720–1995. Meri 27:79–91
Sénéchal É, Bêty J, Gilchrist HG, Hobson KA, Jamieson SE (2011) Do purely capital layers exist among flying birds? Evidence of exogenous contribution to arctic-nesting Common Eider eggs. Oecologia 165:593–604. https://doi.org/10.1007/s00442-010-1853-4
Skutch. AF (1949) Do tropical birds rear as many young as they can nourish? Ibis 91:430–455. https://doi.org/10.1111/j.1474-919X.1949.tb02293.x
Slagsvold T (1982) Clutch size variation in passerine birds: the nest predation hypothesis. Oecologia 54:159–169. https://doi.org/10.1007/BF00378388
Sonne C, Alstrup AKO, Therkildsen OR (2012) Reply to Tillitt et al. 2012: Thiamine deficiency: a viable hypothesis for paralytic syndrome in Baltic birds. Sci Tot Env 433:563–564. https://doi.org/10.1016/j.scitotenv.2012.06.081
Stearns SC (1992) The evolution of life histories. Oxford University Press, Oxford
Stien J (2008) The role of the Hooded Crow (Corvus corone) in the nesting success of the Common Eider (Somateria mollissima) at two colonies in Troms county, Northern Norway. Msc Thesis. University of Tromsø, Tromsø
Stjernberg T (1981) Projekt havsörn i Finland [White-tailed Eagle project in Finland]. In: The White-tailed Eagle projects in Finland and Sweden. Proceedings from a sea-eagle symposium 8–9.1.1979, Tvärminne zoological station, Finland.
Svensson E (1997) Natural selection on avian breeding time: causality, fecundity-dependent, and fecundity-independent selection. Evolution 51:1276–1283. https://doi.org/10.1111/j.1558-5646.1997.tb03974.x
Thompson III FR (2007) Factors affecting nest predation on forest songbirds in North America. Ibis 149:98–109. https://doi.org/10.1111/j.1474-919X.2007.00697.x
Tjørnløv RS, Ens BJ, Öst M et al (2020) Drivers of spatiotemporal variation in survival in a flyway population: a multi-colony study. Front Ecol Evol 8:341. https://doi.org/10.3389/fevo.2020.566154
Turoboyski K (1973) Biology and ecology of the Crab Rhithropanopeus harrisii ssp. tridentatus. Mar Biol 23:303–313. https://doi.org/10.1007/BF00389338
Väänänen V-M (2000) Predation risk associated with nesting in gull colonies by two Aythya species: observations and an experimental test. J Avian Biol 31:31–35. https://doi.org/10.1034/j.1600-048X.2000.310105.x
Vahteri P, Mäkinen A, Salovius S, Vuorinen I (2000) Are drifting algal mats conquering the bottom of the Archipelago Sea, SW Finland? Ambio 29:338–343. https://doi.org/10.1579/0044-7447-29.6.338
Waldeck P, Kilpi M, Öst M, Andersson M (2004) Brood parasitism in a population of common eider (Somateria mollissima). Behaviour 141:725. https://doi.org/10.1163/1568539042245132
Wiebe KL, Martin K (1998) Costs and benefits of nest cover for Ptarmigan: changes within and between years. Anim Behav 56:1137–1144. https://doi.org/10.1006/anbe.1998.0862
Yoccoz NG, Erikstad KE, Bustnes JO, Hanssen SA, Tveraa T (2002) Costs of reproduction in Common Eiders (Somateria mollissima): an assessment of relationships between reproductive effort and future survival and reproduction based on observational and experimental studies. J Appl Stat 29:57–64. https://doi.org/10.1080/02664760120108458
Zanette LY, White AF, Allen MC, Clinchy M (2011) Perceived predation risk reduces the number of offspring songbirds produce per year. Science 334:1398–1401. https://doi.org/10.1126/science.1210908
Acknowledgements
We thank all the field assistants throughout the years, especially Heikki Eriksson, Niilo Aro, Ben Steele, Sara Alfthan and Bertille Mohring. We thank Tvärminne Zoological Station for providing fieldwork facilities, and HALIAS and the WWF Finland’s White-tailed Eagle working group, especially Jouko Högmander and Heikki Lokki, for data. This study was supported by Walter and Andrée de Nottbeck Foundation (IH), the Finnish Foundation for Nature Conservation (IH), Waldemar von Frenckells Foundation (IH), Nordenskiöld-Samfundet (IH), the Swedish Cultural Foundation in Finland (MÖ), Sophie von Julin Foundation (KJ) and the Finnish Ministry of Agriculture and Forestry (KJ). The experiments comply with the current laws of Finland.
Funding
Open access funding provided by Abo Akademi University (ABO).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Barbraud.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hermansson, I., von Numers, M., Jaatinen, K. et al. Predation risk and landscape properties shape reproductive output of an endangered sea duck from two subpopulations with contrasting predation risk. J Ornithol 164, 311–326 (2023). https://doi.org/10.1007/s10336-022-02036-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-022-02036-6