Abstract
Characteristics of egg surfaces serve as recognition cues that allow avian hosts to detect and reject foreign eggs in brood parasitism. The blunt egg pole hypothesis suggests that the blunt egg pole is an essential signal in parasitic egg recognition. In the present study, eggs of Yellow-bellied Prinia (Prinia flaviventris) were painted with black spots at either the blunt or sharp egg pole to determine and compare the responses of different populations exposed to brood parasitism (parasitized population) or not (non-parasitized population). These results supported the above hypothesis and showed that the parasitized population displayed a higher rate of rejection of eggs with blunt pole-painted spots than with sharp pole-painted spots. In contrast, the non-parasitized population accepted all experimental eggs, which may be related to decreased egg recognition resulting from low exposure to brood parasitism.
Zusammenfassung
Entfernen der Eier von Brutparasiten durch Gelbbauchprinien: Die Bedeutung der Position von Pigmentflecken
Merkmale von Eioberflächen dienen als Erkennungssignale, die es Wirtsvögeln ermöglichen, im Fall von Brutparasitismus fremde Eier zu erkennen und zu entfernen. Die „stumpfer Eipol“ Hypothese schlägt vor, dass der stumpfe Eipol ein entscheidendes Signal für das Erkennen der Eier von Brutparasiten darstellt. In der vorliegenden Studie haben wir die Eier von Gelbbauchprinien (Prinia flaviventris) mit schwarzen Punkten bemalt, entweder am stumpfen oder am spitzen Eipol, um die Antworten der Vögel in parasitierten und nicht parasitierten Populationen zu ermitteln und vergleichen. Unsere Ergebnisse stützten die obengenannte Hypothese und zeigten, dass Gelbbauchprinien in der parasitierte Population Eier, bei denen die Flecken auf den stumpfen Eipol gemalt wurden, mit höherer Wahrscheinlichkeit entfernten als Eier mit am spitzen Eipol aufgemalten Flecken. Im Gegensatz dazu wurden in der nicht parasitierten Population alle experimentellen Eier akzeptiert, was mit verminderter Eierkennung infolge geringer Brutparasitismusgefahr zusammenhängen könnte.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The evolutionary arms race between hosts and avian brood parasites is a classic example of co-evolution (Dawkins and Krebs 1979; Davies 2000). Parasitism can have severe and negative impacts on host fitness, forcing hosts to evolve corresponding counter-adaptations against parasites (Davies 2000, 2011; Soler 2014; Feeney et al. 2014). Among the various anti-parasite strategies, one such adaptation is the ability to recognize and reject foreign eggs (Davies and deL Brooke 1989a, b; Sealy and Underwood 2012).
Visual modeling has revealed that avian hosts recognize foreign eggs using multiple signals as recognition cues, with this selective pressure facilitating the extent of egg mimicry by parasites (Spottiswoode and Stevens 2010). Therefore, variations in the characteristics of eggs across species can serve as signals for recognition and rejection of foreign eggs by hosts in avian brood parasitism (Kilner 2006). These variations are smaller within intra-clutches, but larger across different inter-clutches (Øien et al. 1995; Soler and Møller 1996; Stokke et al. 2002; Yang et al. 2015a). The host may discriminate and reject foreign eggs based on various egg traits, such as size (Rothstein 1982; Marchetti 2000), shape (Rothstein 1982; Underwood and Sealy 2006), background color (Stokke et al. 1999; Cherry et al. 2007; Honza et al. 2007; Moskát et al. 2008a; Yang et al. 2010), and the presence or absence of spots or stripes (Stokke et al. 2007; Moskát et al. 2008b; López-de-Hierro and Moreno-Rueda 2010; Liu et al. 2019). A recent study showed that Magpies (Pica pica) use olfactory cues to identify and reject foreign eggs (Soler et al. 2014). Furthermore, in addition to visible light, birds can detect ultraviolet (UV) light using a fourth cone cell type in the retina (Bennett et al. 1996; Bowmaker et al. 1997; Cuthill et al. 2000). Spectral analyses of brood parasitism have indicated that UV light plays an important role in avian egg recognition cues (Cherry and Bennett 2001; Avilés and Møller 2004; Avilés et al. 2006; Honza and Polaciková 2008; Yang et al. 2013).
Among the numerous recognition signals, egg shape does not appear to affect egg recognition by hosts commonly (Underwood and Sealy 2006), whereas background color and spots on eggs may play more important roles in host egg recognition (Moskát et al. 2008a, 2010). For example, López-de-Hierro and Moreno-Rueda (2010) conducted a study on House Sparrows (Passer domesticus) and showed that egg spots are a key factor influencing egg recognition (also see Yang et al. 2015b).
Generally, avian eggs can be divided into two parts: the blunt pole (BP) and sharp pole (SP) (Polačiková et al. 2010; Polačiková and Grim 2010). Spots on eggshells are usually equally distributed or concentrated at the BP, but seldom at the SP (Lack 1968; Kilner 2006). Regarding the importance of specific egg parts in egg recognition, the blunt egg pole hypothesis suggests that spots at the BP play a key role in both intraspecific and interspecific egg recognition (Polačiková et al. 2007, 2010; Polačiková and Grim 2010). This hypothesis has been tested in several species of birds subjected to interspecific parasitism (e.g., Great-Reed Warblers Acrocephalus arundinaceus and Reed Warblers A. scirpaceus) or intraspecific parasitism (e.g., Song Thrushes Turdus philomelos and Blackbirds T. merula), which indicated that egg recognition cues are concentrated at the BP (Polačiková et al. 2010; Polačiková and Grim 2010). The eggs of the above host species contain spots that are mainly concentrated at the BP. To date, however, no study has investigated egg recognition ability in host species that have no spots on their eggs, as the effect of background color on egg spots cannot be excluded (Polačiková et al. 2010; Polačiková and Grim 2010).
Populations of the same species differ in rates of parasitism, which may lead to differential selection of egg recognition/rejection skills (Polačiková et al. 2009; Yang et al. 2014). The Yellow-bellied Prinia (Prinia flaviventris) is a small passerine bird species widely distributed in southern mainland China and Taiwan. Their eggs are red, with no markings on the surface (Yang et al. 2014; Figs. 1, 2); thus, the background color is suitable for adding experimental spots on the egg surface. In Taiwan, the Yellow-bellied Prinia has been geographically separated from the Chinese mainland for 2–3 million years (Chen et al. 2000; Yeung et al. 2011). This species is the main host of the Oriental Cuckoo (Cuculus optatus), which is the only obligate brood parasite of the Taiwanese prinia population (Yang et al. 2012, 2014). In contrast, no cases of cuckoo parasitism have been reported for Yellow-bellied Prinia in mainland China (Yang et al. 2012, 2013, 2014). Taiwanese populations also show significantly higher rates of rejection toward non-mimetic eggs than mainland populations (Yang et al. 2013, 2014; Wang et al., 2016). Thus, the Yellow-bellied Prinia is an ideal species for investigating and comparing egg recognition ability in host-parasitized (Taiwan) and non-parasitized systems (mainland). Yellow-bellied Prinias can recognize foreign eggs using egg color and UV light perception (Yang et al. 2013, 2014), but other recognition cues need to be further tested.
In this study, the blunt egg pole hypothesis was investigated by painting black spots at either the BP or SP in two populations of Yellow-bellied Prinia that differed in parasitism pressure. It was predicted that (1) eggs painted at the BP would have higher rates of rejection than those painted at the SP, and (2) the population in Taiwan would show higher rates of rejection toward both BP- and SP-painted eggs than observed for the Chinese mainland population.
Materials and methods
Study areas and species
The study site in mainland China was located at the Nonggang National Nature Reserve, Guangxi, Southwest China (23°39′ N, 107°04′ E). The study period was from April to July 2012–2014. The Taiwan study site was located at Shoufong, Hualien County (23°51′ N, 121°31′ E). The study period was from April to July 2010–2011 (for study site details, see Yang et al. 2013, 2014). During the breeding season, both male and female Yellow-bellied Prinias participate in nesting in low bushes or small shrubs. Nests are built 1 m high above the ground and have an irregular spherical shape with a side opening in the upper part of the nest (Ding et al. 2008). In this study, Yellow-bellied Prinia hosts exploited by oriental cuckoos were noted only in Taiwan, with a parasitism rate of 0.91% (n = 110). No cases of parasitism were found in mainland China (n = 232) (Yang et al. 2014), although several species of prinias have been listed as potential hosts for several other cuckoo species in mainland China (Yang et al. 2012, 2014). Furthermore, the Taiwanese Yellow-bellied Prinia populations show a stronger response to foreign egg rejection and defense against dummy cuckoos than populations in mainland China (Yang et al. 2013, 2014).
Field procedures
The Yellow-bellied Prinia species shows considerably stronger egg recognition ability and refuses all foreign eggs, including conspecific eggs (Yang et al. 2014), as well as their own eggs treated with UV-blocking agent (to reduce UV spectral reflectance) (Yang et al. 2013). Therefore, prinia nests were artificially parasitized by painting one egg within a clutch (Fig. 1) rather than replacement with conspecific eggs from other prinia nests (e.g., Polačiková et al. 2010; Polačiková and Grim 2010), thus eliminating interference caused by conspecific eggs. Nests were searched and the BP or SP of one egg in each nest was painted using a waterproof black pen (pen point size: 1.0 mm) after hosts laid their last egg or during early incubation. Before painting, each experimental egg was divided into two parts of equal area across their diagonal axis (see Polačiková and Grim 2010). Spots were then applied in one-third region of two poles (BP or SP) of these eggs, but in the mid-region, all the spots of approximately 1 mm diameter at a distance of about 2 mm from each other were painted by the same manipulator to eliminate manipulation deviation (see Fig. 1). Nests with unpainted conspecific clutches were used as the control (n = 12 in each population). For all treatments, prinia nests were monitored daily for six consecutive days (following Moksnes et al. 1991). Responses of the Yellow-bellied Prinias were classified into rejection or acceptance. Rejection referred to the disappearance of the experimental egg or abandonment of the clutch within the experimental period. Acceptance referred to a situation when the experimental egg remained unharmed in the nest and the clutch was incubated (Yang et al. 2013). The latency to rejection (i.e., time interval from brood parasitism to rejection) was calculated daily (considering days as units).
Egg spectrophotometric measurements
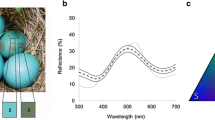
Conspecific eggs collected from deserted nests were used to replace the whole clutch and sent them back when measurement of egg reflectance was over. All eggs were measured and then one-eggpainted spots were selected on blunt or sharp pole to measure the spots. Because the painted color was from the same pen and had no color variation, only five eggs were selected and the spots were measured as representative reflectance spectra of this species (Polačiková and Grim 2010). Similarly, only 30 eggs from all samples were selected for mean egg reflectance. These treatments were used to show differences in painting spots and original surface egg color. Egg spectrophotometric measurements were performed using an Avantes-2048 spectrometer with a 10 W tungsten halogen light source (Avalight-Hal-S; Avantes, Apeldoorn, the Netherlands). Six spectral reflectance measurements were obtained per egg after the egg surface was divided into the three regions (i.e., blunt, middle, and sharp poles). Measurements covered a spot size of approximately 1 mm in diameter on the egg surface, so was the painting spots, and the reflection of the surface was measured at an angle of 45°, with the co-axial reflectance probe held 5 mm from the surface (FCR-7UV200-2-ME). The spectral data were analyzed using Ava-Soft 7.0 software (Avantes, Apeldoorn, the Netherlands) and interpolated at 1 nm steps in the 300–700 nm range.
Statistical analysis
The responses of Yellow-bellied Prinia to experimental eggs were compared using the generalized linear model (GLM) with binomial distribution and logit link function used to assess host responses to parasitic eggs (acceptance or rejection) according to (1) populations in Taiwan and mainland China (two groups), (2) painted spots (three experimental groups, i.e., control, blunt pole, sharp pole), and (3) interactions between populations and groups. One-way ANOVA was used to test egg rejection latency between painted BP and SP. Statistical analyses were performed using IBM SPSS 25.0 (IBM Inc., Armonk, NY, USA). Values are presented as mean ± standard deviation (SD) unless stated otherwise.
Results
The mean spectral reflectance of painted spots was close to zero in the investigated wavelength range of 300–700 nm (Fig. 2), with no differences in reflectance found between BP (36.11 ± 11.9) and SP (36.19 ± 12.81) (t test, t = − 0.019, df = 58, P = 0.985).
The rejection of eggs was explained by interaction between populations and experimental groups (Table 1). This interaction showed that while no rejection was observed in the control groups in either population, the rejection rate of experimental eggs varied between populations (GLM, F = 7.575, P < 0.001). Specifically, no experimental eggs were rejected by the mainland population, whereas experimental eggs painted at the BP were rejected at a higher rate than experimental eggs painted at the SP by the Taiwan population (GLM, SP vs. BP, P < 0.001, SP vs. control, P = 0.560, BP vs. control, P < 0.001, Fig. 3). In the Taiwan population, the egg rejection latency showed no significant differences between painted BP and SP (ANOVA, F = 1.507, P = 0.235), with eggs painted at the BP rejected within 3 days (n = 18), and those painted at the SP rejected after 4–5 days.
Discussion
These results confirmed the predictions that the island population of Yellow-bellied Prinia used the BP as a discrimination cue to recognize and reject foreign eggs. On the other hand, the mainland Chinese population, a non-parasitized system, accepted all experimental eggs, and the blunt pole hypothesis was not supported. Generally, hosts under high parasitic pressure show higher egg rejection rates than those under low parasitic pressure (Davies and deL Brooke 1989a; Moksnes et al. 1991; Lindholm and Thomas 2000; Stokke et al. 2008; Yang et al. 2013, 2014). The Taiwanese prinia population parasitized by cuckoos exhibits stronger foreign egg rejection ability and stronger defenses against invaders (Yang et al. 2014), suggesting that the Taiwanese population is under high parasitic pressure, unlike mainland populations. In this study, it was found that the Taiwanese population rejected eggs painted at the BP within 3 days, whereas those painted at the SP were rejected after 4 to 5 days. This suggests that the latency to rejection was smaller for BP-painted eggs than for SP-painted eggs. Thus, these findings are consistent with those of previous studies showing that the Taiwanese prinia populations have higher rejection rates than those in mainland China (Yang et al. 2014).
The mainland Chinese population accepted all experimental eggs, which was likely due to decreased recognition of foreign eggs by hosts. In previous study, the hosts in this population showed only a 50% rejection rate to non-mimetic eggs but accepted all conspecific eggs. On the other hand, the Taiwan population showed 100% rejection toward non-mimetic eggs and even partial rejection toward conspecific eggs (16.7%) (Yang et al. 2014; Table 2). Retreat of parasites may be one of the reasons for the reduced recognition ability in the mainland population. In addition, the surfaces of eggs within a clutch are highly uniform in the Taiwanese population, unlike that in the mainland population (Yang et al. 2014), which may help explain why the latter showed reduced recognition ability and further supports the hypothesis that parasitic interaction may be the agent promoting the co-evolution of egg appearance (Swynnerton 1916; Takasu 2003; Davies 2011).
Recognition cues such as egg color and markings affect host decisions against foreign eggs. Markings on eggs are considered as fingerprints by female individuals and, hence, play a decisive role in egg recognition (Davies and deL Brooke 1989a; Lahti and Lahti 2002; López-de-Hierro and Moreno-Rueda 2010). However, some studies indicate that egg color is a more important recognition cue (Moskát et al. 2008a, 2010). Both prinia populations showed higher rejection of non-mimetic model eggs (Yang et al. 2014) than of experimental eggs painted with spots (this study) (Table 2). In particular, for the mainland population, spots on eggs did not provoke detection and recognition by the parental birds for rejection, whereas half of the model eggs were rejected. These findings imply that, in contrast, eggshell color is a potentially more important recognition cue than egg spots in the Yellow-bellied Prinia.
In conclusion, this study provides further support for the blunt egg pole hypothesis, showing that the parasitized population displayed a higher rate of rejection of BP-painted eggs than of SP-painted eggs.
References
Avilés JM, Møller AP (2004) How is host egg mimicry maintained in the cuckoo (Cuculus canorus). Biol J Linn Soc 82:57–68
Avilés JM, Soler JJ, Pérez-Contreras T (2006) Dark nests and egg colour in birds: a possible functional role of ultraviolet reflectance in egg detectability. Proc R Soc Lond B 273:2821–2829
Bennett ATD, Cuthill IC, Partridge JC, Maier EJ (1996) Ultraviolet vision and mate choice in zebra finches. Nature 380:433–435
Bowmaker JK, Heath LA, Wilkie SE, Hunt DM (1997) Visual pigments and oil droplets from six classes of photoreceptor in the retinas of birds. Vis Res 37:2183–2194
Chen K, Liu L, Fan F (2000) The changes of Taiwan Island. J Tianjin Normal Univ 20:59–63
Cherry MI, Bennett ATD (2001) Egg colour matching in an African cuckoo, as revealed by ultraviolet visible reflectance spectrophotometry. Proc R Soc Lond B 268:565–571
Cherry MI, Bennett ATD, Moskát C (2007) Host intra-clutch variation, cuckoo egg matching and egg rejection by great reed warblers. Naturwissenschaften 94:441–447
Cuthill IC, Partridge JC, Bennett ATD, Church SC, Hart NS, Hunt S (2000) Ultraviolet vision in birds. Adv Study Behav 29:159–214
Davies NB (2000) Cuckoos, cowbirds and other cheats. T & A. D, Poyser, London
Davies NB (2011) Cuckoo adaptations: trickery and tuning. J Zool 284:1–14
Davies NB, deL Brooke M (1989a) An experimental study of co-evolution between the cuckoo Cuculus canorus, and its hosts. I. Host egg discrimination. J Anim Ecol 58:207–224
Davies NB, deL Brooke M (1989b) An experimental study of co-evolution between the cuckoo, Cuculus canorus, and its hosts. II. Host egg markings, chick discrimination and general discussion. J Anim Ecol 58:225–236
Dawkins R, Krebs JR (1979) Arms races between and within species. Proc R Soc Lond B 205:489–511
Ding Z, Yuan L, Zhu C, Huang J, Zheng X, Guo X, Lin J, Hu H, Tang S (2008) Nesting characteristics of yellow-bellied prinia, Prinia flaviventris. Zool Res 29:270–276
Feeney WE, Welbergen JA, Langmore NE (2014) Advances in the study of coevolution between avian brood parasites and their hosts. Annu Rev Ecol Evol System 45:227–246
Honza M, Polaciková L (2008) Experimental reduction of ultraviolet wavelength reflected from parasitic eggs affects rejection behaviour in the blackcap Sylvia atricapilla. J Exp Biol 211:2519–2523
Honza M, Polaciková L, Procházka P (2007) Ultraviolet and green parts of the colour spectrum affect egg rejection in the song thrush (Turdus philomelos). Biol J Linn Soc 92:269–276
Kilner RM (2006) The evolution of egg colour and patterning in birds. Biol Rev 81:383–406
Lack D (1968) Ecological adaptations for breeding in birds. Methuen, London
Lahti DC, Lahti AR (2002) How precise is egg discrimination in weaverbirds? Anim Behav 63:1135–1142
Lindholm AK, Thomas RJ (2000) Differences between populations of reed warblers in defences against brood parasitism. Behaviour 137:25–42
Liu J, Yang C, Yu J, Wang H, Liang W (2019) Egg recognition in cinereous tits (Parus cinereus): eggshell spots matter. Avian Res 10:37
López-de-Hierro MDG, Moreno-Rueda G (2010) Egg-spot pattern rather than egg colour affects conspecific egg rejection in the house sparrow (Passer domesticus). Behav Ecol Sociobiol 64:317–324
Marchetti K (2000) Egg rejection in a passerine bird: size does matter. Anim Behav 59:877–883
Moksnes A, Røskaft E, Braa AT (1991) Rejection behavior by common cuckoo hosts towards artificial brood parasite eggs. Auk 108:348–354
Moskát C, Avilés JM, Bán M, Hargitai R, Zölei A (2008a) Experimental support for the use of egg uniformity in parasite egg discrimination by cuckoo hosts. Behav Ecol Sociobiol 62:1885–1890
Moskát C, Székely T, Cuthill IC, Kisbenedek T (2008b) Hosts’ responses to parasitic eggs: which cues elicit hosts’ egg discrimination? Ethology 114:186–194
Moskát C, Bán M, Székely T (2010) Discordancy or template-based recognition? Dissecting the cognitive basis of the rejection of foreign eggs in hosts of avian brood parasites. J Exp Biol 213:1976–1983
Øien IJ, Moksnes A, Røskaft E (1995) Evolution of variation in egg color and marking pattern in European passerines: adaptations in a coevolutionary arms race with the cuckoo Cuculus canorus. Behav Ecol 6:166–174
Polačiková L, Grim T (2010) Blunt egg pole holds cues for alien egg discrimination: experimental evidence. J Avian Biol 41:111–116
Polačiková L, Honza M, Procházka P, Topercer J, Stokke BG (2007) Colour characteristics of the blunt egg pole: cues for recognition of parasitic eggs as revealed by reflectance spectrophotometry. Anim Behav 74:419–427
Polačiková L, Porcházka P, Cherry MI, Honza M (2009) Choosing suitable hosts: common cuckoos Cuculus canorus parasitize great reed warblers Acrocephalus arundinaceus of high quality. Evol Ecol 23:879–891
Polačiková L, Stokke BG, Procházka P, Honza M, Moksnes A, Røskaft E (2010) The role of blunt egg pole characteristics for recognition of eggs in the song thrush (Turdus philomelos). Behaviour 147:465–478
Rothstein SI (1982) Mechanisms of avian egg recognition: which egg parameters elicit responses by rejecter species? Behav Ecol Sociobiol 11:229–239
Sealy SG, Underwood TJ (2012) Egg discrimination by hosts and obligate brood parasites: a historical perspective and new synthesis. Chin Birds 3:274–294
Soler M (2014) Long-term coevolution between avian brood parasites and their hosts. Biol Rev 89:688–704
Soler JJ, Møller AP (1996) A comparative analysis of the evolution of variation in appearance of eggs of European passerines in relation to brood parasitism. Behav Ecol 7:89–94
Soler JJ, Pérez Contreras T, De Neve L, Macías Sánchez E, Møller AP, Soler M (2014) Recognizing odd smells and ejection of brood parasitic eggs: An experimental test in magpies of a novel defensive trait against brood parasitism. J Evol Biol 27:1265–1270
Spottiswoode CN, Stevens M (2010) Visual modeling shows that avian host parents use multiple visual cues in rejecting parasitic eggs. Proc Natl Acad Sci 107:8672–8676
Stokke BG, Moksnes A, Røskaft E, Rudolfsen S, Honza M (1999) Rejection of artificial cuckoo (Cuculus canorus) eggs in relation to variation in egg appearance among reed warblers (Acrocephalus scirpaceus). Proc R Soc Lond B 266:1483–1488
Stokke BG, Moksnes A, Røskaft E (2002) Obligate brood parasites as selective agents for evolution of egg appearance in passerine birds. Evolution 56:199–205
Stokke BG, Takasu F, Moksnes A, Røskaft E (2007) The importance of clutch characteristics and learning for anti-parasite adaptations in hosts of avian brood parasites. Evolution 61:2212–2228
Stokke BG, Hafstad I, Rudolfsen G, Moksnes A, Møller AP, Røskaft E, Soler M (2008) Predictors of resistance to brood parasitism within and among reed warbler populations. Behav Ecol 19:612–620
Swynnerton CFM (1916) On the coloration of the mouths and eggs of birds. II. On the coloration of eggs. Ibis 4:529–606
Takasu F (2003) Co-evolutionary dynamics of egg appearance in avian brood parasitism. Evol Ecol Res 5:345–362
Underwood TJ, Sealy SG (2006) Influence of shape on egg discrimination in American robins and gray catbirds. Ethology 112:164–173
Wang L, Liang W, Yang C, Cheng S-J, Hsu Y-C, Lu X (2016) Egg rejection and clutch phenotype variation in the plain prinia Prinia inornata. J Avian Biol 47:788–794
Yang C, Liang W, Cai Y, Shi S, Takasu F, Møller AP, Antonov A, Fossøy F, Moksnes A, Røskaft E, Stokke BG (2010) Coevolution in action: disruptive selection on egg colour in an avian brood parasite and its host. PLoS ONE 5:e10816
Yang C, Liang W, Antonov A, Cai Y, Stokke BG, Fossøy F, Moksnes A, Røskaft E (2012) Diversity of parasitic cuckoos and their hosts in China. Chin Bird 3:9–32
Yang C, Wang L, Hsu Y-C, Antonov A, Moksnes A, Røskaft E, Liang W, Stokke BG (2013) UV reflectance as a cue in egg discrimination in two prinia species exploited differently by brood parasites in Taiwan. Ibis 155:571–575
Yang C, Wang L, Cheng S-J, Hsu Y-C, Liang W, Møller AP (2014) Nest defenses and egg recognition of yellow-bellied prinia against cuckoo parasitism. Naturwissenschaften 101:727–734
Yang C, Hu Y, Ma M, Liang W, Møller AP (2015a) Absence of egg rejection in an Asian population of house sparrow (Passer domesticus), a conspecific brood parasite in Europe. Behav Ecol Sociobiol 69:723–727
Yang C, Liu Y, Liang W (2015b) Egg trait variation in a large hawk-cuckoo (Hierococcyx sparverioides) host population of Chinese babax (Babax lanceolatus). Int Zool 10:295–301
Yeung CK, Lin RC, Lei F, Robson C, Hung LM, Liang W, Zhou F, Han L, Li SH, Yang X (2011) Beyond a morphological paradox: complicated phylogenetic relationships of the parrotbills (Paradoxornithidae, Aves). Mol Phylogenet Evol 61:192–202
Acknowledgements
The authors would like to thank Canchao Yang for help with spectral data analysis, Christine Watts for improving the English proofreading, Ian Will from the University of California, Berkeley, USA, and Aiwu Jiang, Qiuli Huang, Demeng Jiang, and Yungao Hu for their assistance with fieldwork in Guangxi and Shun-Jen Cheng in Taiwan. This work was supported by the National Natural Science Foundation of China (nos. 31660617 and 31960105 to LW, 31772453 and 31970427 to WL), Initial Fund Key Laboratories of Guizhou Province (no. 2011-4005), and Joint Fund of the National Natural Science Foundation of China and Karst Science Research Center of Guizhou Province (no. U1812401).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards
The studies reported in this paper conformed to the laws in the countries in which they were performed. Experimental procedures were conducted in agreement with the Animal Research Ethics Committee of Hainan Provincial Education Centre for Ecology and Environment, Hainan Normal University (No. HNECEE-2011-005).
Conflict of interest
There are no competing conflicts to declare.
Additional information
Communicated by S. Bouwhuis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, L., Hsu, YC. & Liang, W. Rejection of parasitic eggs by Yellow-bellied Prinias: importance of egg spot location. J Ornithol 161, 987–994 (2020). https://doi.org/10.1007/s10336-020-01789-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-020-01789-2