Abstract
Thermostable phosphotriesterase-like lactonases (PLLs) from extremophile archaea, like SsoPox from Sulfolobus solfataricus, are attractive biotechnological tools with industrial applications as organophosphate decontaminants, but their manufacturing still remains an unresolved issue because of the high costs and the low production yields. In this paper, for the first time, an efficient biotechnological process for the production and purification of a recombinant, engineered PLL, SsoPox W263F, expressed in E. coli, has been set up by studying new induction strategies, by designing high cell density cultivations and a new membrane-based downstream process. In fed batches, the enzyme production was boosted of 69-fold up to 4660.0 U L−1 using galactose as inducer in the replacement of IPTG; the process was scalable from 2.5 up to 150 L. By coupling a single thermo-precipitation step and an ultrafiltration process, a total enzyme recovery of 77% with a purity grade of almost 80% was reached.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Organophosphates (OPs) are highly toxic compounds massively used worldwide as pesticides and insecticides in intensive agriculture and which today represents the major environmental pollutants [15]. Their diffusion in the environment on edible cultures and in potable water constitutes a risk for animal and human safety, because their toxic action is due to the ability to inactivate the acetylcholinesterase, a key enzyme for the proper functioning of nerve cells [33]. Parathion, paraoxon, chlorpyrifos and their derivates are the mostly diffused OPs [14] and their employment is estimated to cause 3 million intoxications yearly [8, 40]. Because of their high toxicity, some OPs (like sarin or soman) have been also used as dangerous nerve agents in war conflicts or in terroristic attacks (e.g., in Matsumoto and Tokyo subway in 1994 and 1995) and, although they are officially banned as chemical weapon agents [9, 31], recent news has reported their employment in warfares at the present time (e.g., in 2015 and 2016 in Syria). Thus in the last years, it has become an urgent ecological and safety necessity to find new bioremediation strategies for OP large-scale environmental-friendly decontamination [14]. It is also of scientific and industrial interest to design innovative biomedical countermeasures for the quick sequestration of OPs or for their inactivation in case of human exposure to nerve agent poisoning [14, 25]. Microbial hydrolases could represent valid exogenous bioscavengers and economic bioremediation tools to degrade and detoxify OPs [3, 14, 23, 25], but their employment in large-scale decontamination processes depends on their catalytic efficiency, versatility and stability in the usage conditions as well as on the possibility to easily manufacture these enzymes in high yields [14, 17]. Phosphotriesterase enzymes (PTEs; EC 3.1.8.1) able to degrade OPs were found in mesophilic soil microorganisms as Flavobacterium sp. ATCC 27551, Brevundimonas diminuta or Pseudomonas diminuta and Agrobacterium radiobacter P230, and they have been widely structurally characterized [13, 39]. An extensive literature in the last decades has reported diverse engineering strategies to enhance their catalytic activity up to several orders of magnitude as well as the stereoselectivity and enantiomeric preferences [14, 30, 41, 43]. Some PTEs were also expressed as recombinant proteins in E. coli cells to facilitate their production by fermentation processes [5, 6, 29], but so far very low concentrations and production yields have been reported in the range from 4 to 30 mg L−1 [5, 26]. Thus, large-scale manufacturing and preparation processes of these mesophilic enzymes still remain a key issue [14], while their use in large-scale degradation process has been always limited by the high instability of their free forms, their sensitivity to harsh process conditions and their low thermal stability at temperatures higher than 25 °C. Immobilization of these mesophilic enzymes has been frequently applied to overpass these constraints [14, 17, 25]. More recently, phosphotriesterase-like lactonase enzymes (PLLs), isolated from extremophile archaea, have attracted attention as being valid alternatives to PTEs [14, 25], because they are highly thermostable and usable in a wide pH range and in different buffer conditions [14] and much more adaptable to resist outdoor conditions than the mesophilic enzymes as well as to long-term storage at room temperature [14, 25]. Among them, the SsoPox PLL, isolated from Sulfolobus solfataricus [27], has been identified as the most promising enzyme [11, 14, 27], having a great thermostability and being active at temperature and pH ranges from 10 to 100 °C and from 5.0 to 9.0, respectively [11, 14, 28]. In a previous paper, a new single mutant (SsoPox W263F) of the SsoPox enzyme was obtained using an engineering strategy. This enzyme showed higher hydrolytic activity, a 16-fold enhanced kcat and sixfold higher specificity constant, and it has proved to be a valid bioscavenger and biocontaminant agent against both pesticides, like paraoxon, and nerve gases, such as cyclosarin, in small-scale tests [11, 28]. However, so far the perspectives of an SsoPox W263F industrial application have been limited by a very low enzyme production concentration (5 mg L−1), obtained in 7-L shake flask growth on LB medium with IPTG induction [27, 28], and by a long following purification process that included three thermo-precipitation steps and two different chromatographic phases by using affinity, size exclusion and/or anion exchange resins [32]. These techniques could not be used in an industrial perspective of enzyme production because of the their high costs and the low recovery of the product. Taking into consideration the necessity to prepare the enzyme in high yields on large scales, in this experimental work we have aimed to set up and optimize, for the first time, a new biotechnological process up to pre-industrial scales for the production and purification of SsoPox W263F, as expressed by a recombinant E. coli BL21 strain. According to our knowledge, a similar study concerning the production of thermostable phosphotriesterase enzyme has never been reported so far. To achieve an economical and reliable process, a wise manufacturing design should investigate protein induction/expression conditions and to develop high cell density cultivations to reach high production yields. Besides, it is also necessary to scale up the fermentation process and to imagine an industrial reliable purification train. IPTG is generally used as inducer for the expression of recombinant proteins and it is added to the culture in the late exponential growth phase at high biomass values. But IPTG concentrations could not be higher than 2.0 mM; otherwise, toxic effects on the bacterial growth could occur [7]. Different induction approaches based on the use of lactose or of galactose, in addition to IPTG or as replacements of it, have been successful in boosting the protein production by at least ten folds [4, 34]. Suitable fed-batch fermentation strategies have to be coupled with a good induction system to reach high enzyme production yields. The design of appropriate feeding and aeration profiles could support the growth and avoid any overflow metabolism and/or anaerobic conditions that drive the production of growth-inhibiting by-product formation, such as acetic acid [10, 21, 22, 35]. For this reason, glycerol is commonly preferred as a carbon source, instead of glucose, because it is more slowly uptaken by the bacterial cells [1, 18, 19]. In the process design, the study of an efficient downstream purification protocol is also essential to assure a high recovery of the product at the desired purity and at the lowest possible economic costs. In the case of recombinant protein purification processes, different factors have to be taken into account like the intra- or extracellular expression of the protein and its thermal stability, charge and molecular weight. The design of a purification protocol for SsoPox W263F started from the consideration that the enzyme is thermostable, but intracellularly expressed. Efficient mechanical cell disruption methods are commonly reported in literature for the extraction of intracellular proteins from biomass as the first step of a purification scheme [12]. In the case of thermostable enzymes, the following phase generally employs diverse thermal precipitation steps of the crude extract, taking advantage of the elevated stability of the recombinant proteins. In this procedure, factors like the temperature used, the number of steps of thermal precipitation, the buffer composition, the presence of cofactors or the initial total protein concentration are critical and have to be wisely set up to obtain high recovery values and good purification folds. After that, a following ultrafiltration membrane-based purification could be an efficient alternative to the chromatographic procedure, because it assures high recovery and selectivity in the separation of biological molecules. Moreover, it is also very economic and easy to be scaled up, thus being more appropriate to be applied in industrial processes [36–38]. To set up a new biotechnological process for the SsoPox W263F production and purification, in this research new alternative induction strategies for enzyme expression were exploited, high cell density fed-batch fermentation processes were set up and scaled up from 2.5 to 150.0 L and a new downstream purification protocol was studied, coupling thermal treatments with membrane-based ultrafiltration steps.
Materials and methods
Chemicals, microorganism and media
All the media components and the chemicals used in the shake flask and fermentation experiments or in the preparation of the biomass extraction buffer were from Sigma-Aldrich (Milan, Italy), except the tryptone that was furnished by OXOID (UK). IPTG was from Gold Biotechnology (Olivette, MO, USA). LB broth powder was purchased from BD Biosciences (New Jersey, USA). Chemicals, standards and solvents used in the purification procedure, in the enzyme assays, analytical procedures, SDS-PAGE and western blot analyses were from Sigma-Aldrich (Milan, Italy) as well, unless differently specified. The E. coli BL21 (DE3)-Ssopox strain was obtained by Prof. Manco’s group by genetic engineering through insertion of extra copies of the Ssopox gene isolated from S. solfataricus and then mutated at the amino acidic position 263, according to an already described procedure [28]. The strain was propagated on LB medium and stored at −80 °C in 20% (v/v) glycerol stock solutions. The inoculum and the different growths were performed on a semi-defined medium containing glycerol (20 g L−1), as the main carbon source, yeast extract (2.4 g L−1) and tryptone (1.2 g L−1) as nitrogen sources, and several salts and trace elements [4.3 g L−1 KH2PO4, 17.4 g L−1 K2HPO4, 1.0 g L−1 (NH4)2SO4, 1.25 g L−1 MgCl2, 3.0 g L−1 citric acid, 0.3 g L−1 thiamine, 1.25 g L−1 MgCl2·6H2O, 12.9 mg L−1 MnSO4·H2O and 10.0 ml L−1 of a trace metal solution consisting of 0.02 g L−1 FeSO4·3H2O, 0.02 g L−1 CaCl2·H2O, 8.7 mg L−1 ZnSO4·7H2O, 3.2 mg L−1 CuCl2·2H2O, 6.45 mg L−1 CoCl2·2H2O and 2.7 g L−1 Na2MoO4·2H2O]. In a shake flask and fermentation experiments, the medium was sterilized in autoclave or in situ, respectively, at 121 °C for 20 min; K2HPO4 was prepared as a 388.0 g L−1 concentrated stock solution, sterilized by filtration through 0.22 µm membranes (Millipore, France) and added to the medium to have a final concentration of 17.4 g L−1. 100.0 µg L−1 of ampicillin was added to the media after sterilization.
Microorganism cultivation
Shake flask experiments
Shake flask experiments were performed in triplicate to evaluate the effect of different inducers (IPTG, galactose or lactose) on the microbial growth and on the phosphotriesterase production. E. coli-Ssopox was grown in 0.2 L of medium in 1.0 L of baffled shake flasks at 37 °C and 200 rpm in a rotary air shaker incubator (Infors HT Incubator, Switzerland). A 10 ml of pre-culture, seeded with 100 µl of the cell stock solution and incubated overnight in 50-ml tubes was used as inoculum for each shake flask. During the experiments, 2 ml of culture was withdrawn each hour to determine the bacterial growth by measuring the absorbance at 600 nm (Spectrophotometer DU800, Beckman Coulter, USA). Induction was performed at around 1.0 Abs6oonm with IPTG (in a concentration range from 1.0 to 2.0 mM) or with galactose (from 1.0 to 5.0 mM) or with lactose (from 1.0 to 10.0 mM) and by adding 0.5 mM of CoCl2 used as cofactor for the enzyme. In all the experiments the growth rate (µ) was calculated before and after induction according to the following formula: µ = (lnAbs600 nm2 − lnAbs600 nm1)/(T 2 − T 1), in which T 2 > T 1; the µ values of three replicates were then averaged. At 5 h post-induction, the cultures were centrifuged at 4 °C and at 6000 rpm for 30 min (Avanti J-20XP, Beckman Coulter, USA) and the biomasses were recovered and frozen at −20 °C. The shake flask final biomasses were then extracted, the crude extracts were thermo-precipitated and the protein content and the phosphotriesterase activity were determined according to procedures described in the following paragraphs.
Fermentation experiments
Fermentation experiments were performed on 2.5, 22.0 and 150-L fermenters (Biostat CT, Biostat C and Biostat D, Braun Biotech International, Sartorius Group, Germany) having working volume of 2.0, 15.0 and 100.0 L, respectively. All of them were sterilizable in situ, equipped with pH, temperature and pO2 probes and four peristaltic pumps for the addition of alkali, acid, antifoam solutions and, eventually, for the feeding solution. The pO2 electrodes (Mettler Toledo, Switzerland) were calibrated using a pure oxygen flow at 100% value, before the experiment started. During all the processes the temperature was kept at 37 °C and the pH value 7.0 by adding to the culture 30% (v/v) NH4OH and/or 30% (v/v) H2SO4. The pO2 value inside the vessel was always kept higher than 20% by modulating the stirring in the range from 400 to 600 rpm, the airflow between 0.75 and 1.0 vvm and eventually by insufflating pure oxygen according to the microorganism metabolism request. In the course of all the fermentations, the process parameters were remotely controlled and all the data were collected by a Digital Control Unit (DCU), using an MFCS-win software (Braun Biotech International, Sartorius Group, Germany). In particular, batch experiments were run for 24 h in a 2.5-L vessel using an overnight-grown shaking flask culture as inoculum. Induction was performed at 6.0 Abs6oonm, at about the 6th hours of growth, by adding 1.0 mM IPTG or 5.0 mM galactose plus 0.5 mM CoCl2 used as cofactor. Fed-batch experiments, instead, were carried for 48–49 h in 2.5-, 22.0- or 150.0-L vessels. In all the experiments, after 8–9 h of batch fermentation, the fed-batch phase was started by feeding the culture with a concentrated medium solution (160.0 g L−1 of glycerol, 19.2 g L−1 of yeast extract and 9.6 g L−1 tryptone) supplemented according to a step profile (1.0 g L−1 h −1 of glycerol from 8 to 14 h of growth, 1.5 g L−1 h −1 from 14 to 20 h and 2.0 g L−1 h −1 from 20 h up to the end). Induction in fed-batch experiments was performed at 38–42Abs6oonm, at about the 24th hours of growth, by adding 1.0 mM of IPTG or 5.0 mM galactose plus 0.5 mM CoCl2 in the case of 2.5-L experiments, or 5.0 mM galactose plus 0.5 mM CoCl2 in the other scales. During all the batch and fed-batch fermentations, the bacterial growth was checked by measuring the absorbance at 600 nm of the broth samples, withdrawn regularly at different time points, or by weighing the wet biomass of 1 ml of broth sample (gcww L−1-grams of cell wet weight per volume) after centrifugation at 4 °C and at 6000 rpm for 15 min (Avanti J-20XP, Beckman Coulter, USA). The supernatants of the centrifuged broth samples at the different time points were collected to determine the concentration of glycerol, acetic acid and of inducers . Before and after induction 30-ml samples were withdrawn and centrifuged as previously described; the collected biomasses were then extracted to quantify the phosphotriesterase enzyme production at different time points. The batch and fed-batch final biomass was harvested at 24 h post-induction by centrifugation of the broth at 4 °C and 6000 rpm for 40 min (Avanti J-20XP, Beckman Coulter, USA), or by continuous centrifugation at 4 °C and 9600 rpm (Clara20, Alfa Laval, Italy) using a feed flow of 1.0 L min −1, a discharge time of 12 min and by applying a back pressure of 1.8 bar. The recovered biomass was then frozen at −20 °C.
Downstream purification process
Extraction of the enzyme from biomass
The enzyme was extracted from the shake flask final biomasses or from the different time point pellets of the fermentation experiments by sonication (Sonicator 3000, Misomix, USA) for 10 min (30 s on and 30 s off; power level of 3.5) and re-suspending the cells in an extraction buffer (20 mM hepes, 0.2 mM CoCl2, 0.1% (w/v) Triton X at pH 8.5) in a ratio of 1–3 (w/v). A high pressure cell homogenizer (Emulsiflex C3, Avestin, Germany) was used to break the final fermentation biomass (200 gcww) by applying a pressure of 15.000–20.000 psi (100.000–150.000 kPa). After biomass disruption, cell debris were removed by centrifugation at 6500 rpm and at 4 °C for 50 min (Avanti J-20XP, Beckman Coulter, USA). The crude extract was recovered and a protease inhibitor solution was added to it (100 µl L−1) (Complete Mini -EDTA free, Roche Diagnostic, USA). Thermo-precipitation of the crude extracts of shake flask and fermentation time point samples was performed at 70 °C and at 1200 rpm for 25 min by using a thermomixer (Eppendorf, Germany), after having diluted the samples with the buffer solution to have a total protein concentration of 4 g L−1. In the case of large-scale purification, the thermo-precipitation was performed in a 5-L glass jacked bio-reactor (Steroglass, Italy), equipped with a Rushton pale stirring system, always at 70 °C for 25 min. In all cases, the precipitated proteins were separated from the solution by centrifugation at 6500 rpm and 4 °C, for 50 min (Avanti J-20XP, Beckman Coulter, USA). In case of large-scale purification, the collected supernatants were then further purified by ultrafiltration/diafiltration on membranes. The crude extract samples and the thermo-precipitated ones were assayed to determine the total protein content and the enzyme activity.
Ultrafiltration/diafiltration membrane-based purification
The supernatants obtained by thermo-precipitation of the crude extracts of the final fermentation biomasses were ultrafiltered using an automatic tangential flow filtration system (TFF Uniflux 10 system, GE Healthcare, USA), equipped with level and pressure sensors, temperature and pH probes, a UV detector, an inlet transfer pump and a feeding and a permeate pump able to generate a back pressure for fine transmembrane pressure (TMP) regulation. This system was connected to a software (UNICORN, GE Healthcare, USA) to monitor and control the process parameters. The samples were first ultrafiltered on 100 kDa cutoff membranes, having a total filtering area of 0.1 m2, (Sartorius Group, Germany), by applying a feed flow of 1.2 L min−1 to obtain an outlet flow of 36 L m−1 h−1 with an initial inlet pressure of 0.6 bar. After permeating 95% of the sample, a diafiltration was performed with 3 or 5 volumes of a 20.0 mM hepes and 0.2 mM CoCl2 solution. The permeated and the diafiltered solution that contained the enzyme was concentrated on 5 kDa membranes and then diafiltered with 2 volumes of the buffer. The concentrated solution was then freeze-dried (Epsilon 2–6 D, Christ, Germany). During the process, samples were withdrawn at each step to perform analyses of the total protein content and the enzyme activity.
Analytical methods
Assays
The total protein concentration of the crude extracts and of the samples obtained at the different steps of the purification process was determined by the spectroscopic assay [2] using bovine serum albumin (BSA) as standard (Biorad, USA). The SsoPox W263F enzyme is able to hydrolyze the paraoxon, converting it into p-nitro-phenol that absorbs at 405 nm. Thus, the phosphotriesterase activity and the enzyme units in each sample were determined at 70 °C by adding 20 µg of enzyme in a mixture of 20.0 mM hepes at pH 8.5, containing 1.0 mM paraoxon as substrate. The kinetic of the hydrolysis reaction was followed by recoding the absorbance increase at 405 nm in a 2 min run as previously reported [28]. One enzyme unit was defined as the quantity of enzyme necessary to transform 1 µmol of paraoxon into p-nitro-phenol in 1 min (ε = 21 m Abs−1). Thus, the concentration of the enzyme in terms of U L−1 was calculated from the absorbance determined by the assay. Dividing this concentration value (U L−1) by the concentration of the biomass (gcww L−1), the enzyme yield was calculated as U g −1cww , while dividing it by the hours of run the productivity in terms of U L−1 h−1 was calculated. The enzyme concentration in terms of mg L−1 was determined by dividing the value of U L−1 for the specific activity of the pure enzyme (U mg−1) previously determined [28].
High-performance chromatography analyses
The broth samples withdrawn at different time points during the different fermentation experiments were centrifuged at 4 °C and at 4500 rpm for 30 min (Avanti J-20XP, Beckman Coulter, USA) to remove the bacterial biomasses and to recover the supernatants. 1 ml of these supernatants was ultrafiltered on 10 kDa centrifugal filter devices (Amicon, USA) at 4 °C and 11,000 rpm (Centrifuge Z216 MK, Hermle Labortechnik GmbH, Germany). The filtered solutions were then analyzed by high-performance anion exchange chromatography (HPAE-PAD) (ICS-3000, Dionex, USA) to determine the residual glycerol and the inducer concentrations in the medium during the growth and also by HPLC (Ultimate 3000, Dionex, USA) to determine the organic acid concentrations produced by the bacteria, according to methods previously described [34, 35].
SDS-PAGE and western blot analyses
To evaluate the enzyme production and the protein pattern in the different induction conditions in the shake flask growths, the biomass pellets were disrupted using a cell cracking method [24] and the extracts were analyzed by SDS-PAGE as described by Laemmli [20]. Analyses were also performed to investigate the protein pattern at the different steps of the downstream process: 20 μg of total protein content for each sample was loaded on a 12.5% polyacrylamide gel and then run as already described [20]. In the two cases, protein standards in the molecular weight range from 11 to 75 or from 17 to 38 kDa (Prestained Standard, Invitrogen, USA) were used as ladders. After the run, the gels were stained with coomassie (Coomassie Brilliant Blue R–250, BIORAD, USA) for 7–10 min and then de-stained for 4–8 h. In case of shake flask experiments, the gels were also acquired by using a densitometry system (GEL DOCTM EZ System, BIORAD, USA); the relative percentage of the SsoPox W263F enzyme, compared to the total protein content, was calculated by using the software IMAGE LABTM (BIORAD, USA). Western blot analyses of the purified enzyme samples obtained after the downstream purification process were performed by using a polyclonal rabbit antibody against SsoPox wt, produced by Prof. Manco’s group. After the SDS-PAGE analyses, the proteins (a total amount of 100 ng) was transferred on PVDF sheets at 50 V for 1.5 h at 4 °C that were then treated for 1.5 h with the blocking solution (1× PBS, 0.1% tween, 5% milk). Incubation with primary rabbit polyclonal antibody was performed for 2 h at room temperature in the same solution. The blots were then washed several times with a PBS solution (1× PBS, 0.1% tween) and the antibody binding was then detected by using horseradish peroxidase (HRP)-conjugated secondary anti-rabbit antibody after incubation for 1 h at room temperature. The HRP reaction was revealed using a kit for chemiluminescence (kit ECL Western Blotting Substrate, Abcam, UK) and immunopositive signals were detected on X-ray films.
Data analyses
Data reported in the figures and text are referred to averages of three independent experiments. Their averaged values with the standard deviations were calculated by using the Microsoft Office Excel 2007 program (Microsoft, USA). The yield and productivity were calculated from the experimental concentrations of biomass and enzyme as averaged values as well. Statistical comparisons between group of data, for instance on the diverse shake flasks experiments, were accomplished using the t Student test, and the data were considered to be significantly different if p values were <0.05.
Results
Shake flask experiments
Experiments in shake flasks were performed to study the bacterial growth and the optimal induction conditions using different types and concentrations of inducers. In all cases, the addition of the inducer to the cell culture caused a slowdown of the bacterial growth and a diminishing of the final biomass compared to the not-induced experiment. However, the reduction was different according to the type of inducer employed (Fig. 1a). For example, induction with 1.0 mM IPTG greatly inhibited the bacterial growth causing a decrease of the rate from 0.89 ± 0.02 h−1 before induction to 0.38 ± 0.02 h−1 in the first following hour; consequently, also the final biomass value was almost 2.3 times lower than the not-induced one. A similar effect was noted also in the experiments with higher IPTG concentrations, like 1.5 and 2.0 mM, in which there was a reduction of the growth rate of 50%. Instead, when galactose or lactose was used as inducers, a minor growth rate-inhibiting effect was observed (the µ values were between 0.66 ± 0.01 and 0.56 ± 0.01 h−1, respectively, at the first hour post-induction) and higher final biomass values were also reached (Fig. 1a). In all cases, the enzyme was active, but the production varied according to the different inducers and concentrations used (Fig. 1b): a basal phosphotriesterase expression was determined also in not-induced shake flasks (0.96 ± 0.27 U g −1cww , corresponding to 1.88 ± 0.30 U L−1). This production increased by inducing with IPTG, although it was not proportional to the concentration of the inducer used (1.67 ± 0.16 U g −1cww with 1.0 mM IPTG versus 1.13 ± 0.24 and 1.26 ± 0.31 U g −1cww in case of 1.5 and 2.0 mM, respectively). (IPTG concentrations in the range from 0.1 to 0.5 mM caused an enzyme production similar to the basal one in the range from 0.88 to 0.90 ± 0.20 U g −1cww ). Both galactose and lactose induction, instead, stimulated a higher enzyme expression (Fig. 1b). The best result was obtained by inducing with 5.0 mM of galactose: a production of 4.69 ± 0.53 U g −1cww was obtained, equivalent to 6.46 ± 0.20 U L−1, a value 4.88- and 2.80-fold higher than the non-induced experiments and than the 1.0 mM IPTG-induced ones, respectively (Fig. 1b) (no further increase was noted using higher concentrations of galactose up to 20 mM; data not shown). SDS-PAGE gel densitometry analyses of shake flask crude extracts established that the SsoPox W263F band was 8.5% of the total of protein bands produced in case of the not-induced shake flask, 13.9% in the experiment with 1.0 mM IPTG, 33.6% with 5.0 mM galactose and 22.9% with 5.0 mM lactose. These data confirmed an increase of expression values similarly to the ones reported in the enzyme assays (Fig. 1c). Because the induction with 5.0 mM galactose had low toxic effects on the bacterial growth and it more efficiently boosted the enzyme production, this new induction approach was used for further investigations, as alternative to the IPTG one, in batch and fed-batch experiments.
Shake flask experiments of E. coli-Ssopox performed in triplicate and induced with different molecules at 1.0 Abs600nm, as indicated by the arrow, in comparison to the not-induced experiment: a growth curves and b enzyme production reported in terms of units on wet biomass weight and units per liter (*p < 0.05 compared to the not-induced one; **p < 0.05 compared to the IPTG one). Data are averaged values with standard deviations; c SDS-PAGE analysis of the protein pattern profiles of shake flask experiments, as extracted from biomasses (lane 1 ladder; lane 2 not-induced shake flask; lane 3 1.0 mM IPTG induced one; lane 4 5.0 mM galactose one; lane 5 5.0 mM lactose one. The SsoPoxW263F bands are indicated by the square and their relative percentage values are reported below the gel)
Batch experiments
Batch fermentations (2.5 L) were performed inducing with 1.0 mM IPTG or 5.0 mM galactose during the exponential phase (at 6.0 Abs600nm). As noted in case of shake flask experiments, the induction with IPTG slowed down the growth and the final biomass was 17.7 ± 0.2 gcdw L−1 (Fig. 2a, b). More than 50% of the IPTG concentration was uptaken by the bacteria in the first 5 h post-induction, but in that time the bacterial growth rate also diminished of 52% (Fig. 2a, b). Enzyme production reflected this physiological behavior and a maximum of 5.28 ± 0.26 U g −1cww was determined at 5 h post-induction, corresponding to 152.0 ± 7.6 U L−1 with a productivity value of 15.2 ± 0.8 U L−1 h−1 (Fig. 2c). The uptake of the residual IPTG in the following hours did not cause any further inductive effect on the culture, the growth slowed down and the glycerol was completely consumed in 12 h of growth (Fig. 2a, b). The induction with 5.0 mM galactose, instead, allowed to better sustain the culture whose growth rate diminished by only 37% at 5 h post-induction; thus, the final biomass reached the value of 36.3 ± 0.5 gcww L−1 (Fig. 2d, e). The glycerol uptake was quicker, compared to the one observed after IPTG induction and the batch phase finished at 8 h post-induction (Fig. 2d, e). Galactose was completely consumed in 5 h after the induction, but its inductive effect lasted longer than the IPTG one, resulting in a 1.9-fold increased enzyme production up to 9.24 ± 0.46 U g −1cww , corresponding to 521.0 ± 25.0 U L−1 with a productivity value of 18.0 ± 0.9 U L−1 h−1 at 24 h post-induction (Fig. 2f).
Batch experiments (2.5 L) of E. coli-Ssopox performed in triplicate and induced at 6.0 Abs600nm with 1.0 mM IPTG (a–c) or 5.0 mM galactose (d–f), as indicated by the arrows: growth curves, glycerol consumption, IPTG or galactose uptake and acetic acid concentration (a or d, respectively); stirring profiles, dissolved oxygen percentages and bacteria growth rate in the two different conditions (b or e), SsoPox W263F enzyme production reported in terms of units on wet biomass weight or as units on liter (c or f). All the data are averaged values with standard deviations
Fed-batch experiments
In fed-batch experiments (2.5 L), a step feeding profile was supplied to the cultures at the end of the batch phase that in absence of induction was at 8 h (Fig. 3a–d); the growth was prolonged without accumulation of the carbon source, thus perfectly controlling the acetate formation (Fig. 3a, c). The cultures were induced with 1.0 mM IPTG or 5.0 mM galactose at 24 h, at biomass values higher than in batch experiments (at around 40.0 ± 3.0 Abs600nm) (Fig. 3a, c). After adding 1.0 mM IPTG, the growth slowed down as previously observed in batch experiments and the final biomass at 48 h of growth was 98.8 ± 1.0 gcww L−1 (Fig. 3a). The kinetic of the IPTG uptake was similar to the one previously observed in batch experiments as well as the trend of enzyme production. But the induction at higher biomass values determined a 2.3-fold increase of the production with a maximum of 12.2 ± 0.61 U g −1cww at 5 h post-induction corresponding to 1231.0 ± 60.0 U L−1 with a productivity value of 25.6 ± 1.3 U L−1 h−1 (Fig. 3b). After induction with galactose, instead, the growth reached a final biomass value of 108.1 ± 1.0 gcww L−1 (Fig. 3c), thus allowing the bacteria to produce a maximum enzyme concentration of 37.5 ± 1.8 U g −1cww at 24 h post-induction corresponding to 3865.0 ± 180.0 U L−1 with a productivity value of 80.5 ± 4.0 U L−1 h−1 (Fig. 3d). The increase of production was 7.4-fold higher than the batch one and 3.2-fold higher than the one obtained in fed batch with IPTG induction. Taking into consideration the good results obtained with the galactose induction, further investigations were performed to scale the process up to 22.0 and 150.0 L (Fig. 4; Table 1). Similar kinetics of growth were noted in the two scales up to 30 h, while a slightly higher final biomass was reached at the 150.0 L scale at the end of the growth (Fig. 4; Table 1). An equivalent kinetic of enzyme production was determined in both scales with production concentration, yield and productivity values even better than the ones obtained in the 2.5-L vessel (Table 1).
Fed-batch experiments (2.5 L) of E. coli-Ssopox performed in triplicate and induced at about 40.0 Abs600nm with 1.0 mM IPTG (a, b) or 5.0 mM galactose (c, d), as indicated by the arrows: growth curves, glycerol consumption, IPTG or galactose uptake, acetic acid concentration and feeding profile (a or c, respectively). Correspondingly, SsoPox W263F enzyme production reported in terms of units on wet biomass weight or units on liter (b or d, respectively). All the data are averaged values with standard deviations
Comparison of 22.0- and 150.0-L fed-batch experiments of E. coli-Ssopox performed in triplicate and induced at around 40 Abs600nm with 5.0 mM galactose, as indicated by the arrows: growth curves and correspondingly SsoPox W263F enzyme production as units on wet biomass weight. All the data are averaged values with standard deviations
Enzyme extraction and membrane-based purification
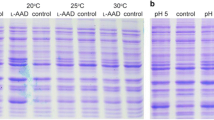
The purification process of SsoPox W263F was designed taking into consideration that the enzyme is a thermostable recombinant protein, intracellularly expressed (Fig. 5a; Table 2). Studies were performed on 200 g of the fed-batch final biomass that was mechanically disrupted by a homogenizer. In the crude extract, a total protein content of 6000 ± 500 mg was determined and a total enzyme amount of about 7800 ± 600 units (39.1 ± 1.0 U g −1cww ) was obtained (Table 2). Taking advantage of the thermal stability properties of the enzyme, a following thermo-precipitation process was designed. Investigations established that a unique step at 70 °C for 25 min, starting from a total protein concentration of 4 mg ml−1, was sufficient to obtain a good removal of the undesired proteins (total protein content was reduced to 60%) without losing the enzyme whose recovery was 96.1 ± 2.0%, with a purification fold of 2.4 ± 0.5 (Table 2). An ultrafiltration/diafiltration process on membranes of 100 KDa first and then on 5 KDa ones was performed to further remove proteins with a molecular weight higher than the SsoPox W263F one and to recover the enzyme in a concentrated solution. Ultrafiltration on 100 KDa membranes was followed by diafiltration with 2 volumes of hepes buffer and allowed to recover 68.0 ± 2.0% of the initial enzyme amount in the permeate. By using 5 volumes during the diafiltration, instead, a higher enzyme recovery of 81.8 ± 2.0% was reached, further increasing the purification fold up to 6.75 ± 0.6 (Table 2). The permeate was then concentrated on 5 KDa membranes and diafiltered with 2 volumes of buffer; the protein was mainly found in the retentate (93.0 ± 2.0% of recovery was reached in this step). The final enzyme recovery in the whole process was 76.5 ± 3.0% with a purification fold of 8.5 ± 0.6 and an increase of specific activity up to 10.4 ± 0.2 U mg−1 (Table 2). SDS-PAGE and western blot analyses confirmed the identity of the enzyme and determined a purification grade of 71.0 ± 2.0%, while by enzymatic assays the enzyme was active and pure at 77.3 ± 2.0% (Fig. 5b, c).
Scheme of the different steps of the downstream purification process of the SsoPox W263F enzyme as recovered from E. coli-Ssopox biomass of fed-batch fermentation induced with 5.0 mM galactose (a): extraction from biomass by cell disruption, thermo-precipitation at 70 °C, ultrafiltration/diafiltration on 100 kDa and then on 5 kDa membranes. SDS-PAGE (b) and western blot (c) analyses of the samples coming from the different steps of the downstream purification process: lane 1 ladder, lane 2 crude extract, lane 3 thermo-precipitated sample, lane 4 retentate on 100 kDa, lane 5 permeate on 100 kDa, lane 6 retentate on 5 kDa; lane 6′ immunoblotting of retentate on 5 kDa with specific anti-SsoPox antibody
Discussion
The recombinant thermostable enzyme SsoPox W263F, a mutant of the archaeal PLL first isolated from S. solfataricus and then expressed in E. coli BL21 (DE3), is able to degrade OPs and for this reason it is of industrial interest being potentially employable as an environmental decontaminating agent or as a degrading tool in case of nerve gas terroristic attacks [28]. However, so far its application has been limited by the very low concentrations obtained during the production process and by an economically unreliable purification [27, 28]. In this paper, for the first time, we have set up a new biotechnological process for the manufacturing of this enzyme on large scale. In this research, new strategies of induction were investigated and a new approach based on the use of galactose proved to be highly and more effective than the IPTG one, resulting in a 4.9-fold enzyme increase since the first shake flask experiments compared to the not-induced ones. Galactose has been already demonstrated to be a valid alternative as an inducer for the recombinant protein expression [4, 34, 42]; in our study, it resulted in much less growth inhibition than IPTG, allowing to obtain a higher and more prolonged expression of the enzyme during the fermentation, as visible in batch experiments. In 2.5-L fed batches, the optimized aeration and feeding profiles avoided the formation of acetate at inhibiting concentrations and allowed to reach high cell density values before induction. Thus, coupling these growth conditions with the galactose induction approach, a further 7.4-fold production increase was obtained up to 4660.0 U L−1, a value 69 times higher than the previously reported data [27, 28] and similar to the data reported for the mesophilic hydrolase from Pseudomonas diminuta [16]. Besides, this productive strategy was robust and easy to be scaled up to 150.0-L vessels. In the perspective of an industrial-scale production process, an efficient downstream purification scheme was also designed starting with the extraction of the enzyme from the biomass and taking advantage of its thermal stability. So far, three steps of thermal treatments at different temperatures have been employed to purify the enzyme on small scale [27, 28]. In this study, instead, we have obtained a very high recovery (>95%) of the sulfolobal enzyme from the crude extract (with a purification fold of 2.4) by using only one thermal precipitation step at 70 °C, and thus reducing the costs of the whole purification process to one-third in this phase. The size exclusion and affinity chromatography methods, used in previous studies to further purify the enzyme [27, 28], were substituted by a membrane-based ultrafiltration process that is easy to be industrially scaled up. The sound chromatographic outcomes reported at the laboratory scale are not scalable for pilot and then manufacturing plant size. Generally, enzymes are produced in fermentation vessels from 20,000 to 50,000-L capacity. In this respect, ultrafiltration is certainly more applicable than chromatography. Affinity or size exclusion chromatography-based purification are reported to be used at industrial level only for antibodies refining that in fact have a productive scale generally lower than 100 L. Besides, the time length of the downstream processing is also strongly reduced when moving from chromatography to ultra-filtration, and thus also the costs. Fluxes in membrane-based purification processes are up to five times higher than in chromatography; for example by using only one membrane, 100 L of protein solution can be treated at a time 25-fold lower than the time employed using a chromatographic column working at a flow rate of 2 ml min−1. In our process, the two-step membrane process allowed also to further increase the purification fold to 3.5 times (up to 8.5) after the thermal precipitation and to reach a sound final enzyme recovery and pureness good enough to be used as a decontaminating agent. The quantity of enzyme obtained in only one 150.0-L fed-batch process and in the following purification would be sufficient to decontaminate from paraoxon a surface 10,000-fold wider and to neutralize a nerve gas cyclosarin amount 9500-fold higher than the quantities that have been degraded so far [28].
In conclusion, all these results have demonstrated that a new reliable biotechnological process for the production of the recombinant SsoPox W263F enzyme has been set up to the pre-industrial scale by coupling high cell density fed-batch fermentation strategies with an alternative galactose induction scheme. An efficient purification and enzyme recovery was obtained by coupling one step of a thermal treatment procedure with an ultrafiltration membrane-based process.
References
Babaeipour V, Shojaosadati SA, Khalilzadeh R, Maghsoudi N, Tabandeh F (2008) A proposed feeding strategy for the overproduction of recombinant proteins in Escherichia coli. Biotechnol Appl Biochem 49:141–147. doi:10.1042/BA20070089
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Carullo P, Cetrangolo PG, Mandrich L, Manco G, Febbraio F (2015) Fluorescence spectroscopy approaches for the development of a real-time organophosphate detection system using an enzymatic sensor. Sensors 15:3932–3951. doi:10.3390/s150203932
Carvalho RJ, Cabrera-Crespo J, Tanizaki MM, Gonçalves VM (2011) Development of production and purification processes of recombinant fragment of pneumococcal surface protein A in Escherichia coli using different carbon sources and chromatography sequences. Appl Microbiol Biotechnol 94:683–694. doi:10.1007/s00253-011-3649-9
Cha HJ, Wu CF, Valdes JJ, Rao G, Bentley WE (2000) Observations of green fluorescent protein as a fusion partner in genetically engineered Escherichia coli: monitoring protein expression and solubility. Biotechnol Bioeng 67:565–574
Cheng TC, Harvey SP, Chen GL (1996) Cloning and expression of a gene encoding a bacterial enzyme for decontamination of organophosphorus nerve agents and nucleotide sequence of the enzyme. Appl Environ Microbiol 62:1636–1641
Donovan RS, Robinson CW, Glick BR (1996) Optimizing inducer and culture conditions for expression of foreign proteins under the control of the lac promoter. J Ind Microbiol 16:145–154
Gunnell D, Eddleston M, Phillips MR, Konradsen F (2007) The global distribution of fatal pesticide self-poisoning: systematic review. BMC Public Health 7:357. doi:10.1186/1471-2458-7-357
Gupta RC (2009) Global impact of chemical warfare agents used before and after 1945. In: Gupta RC (ed) Handbook of toxicology of chemical warfare agents, 2nd edn. Accademic Press/Elsevier, New York, pp 23–24
Han L, Enfors SO, Häggström L (2003) Escherichia coli high-cell-density culture: carbon mass balances and release of outer membrane components. Bioprocess Biosyst Eng 25:205–212. doi:10.1007/s00449-002-0300-2
Hiblot J, Gotthard G, Chabriere E, Elias M (2012) Characterisation of the organophosphate hydrolase catalytic activity of SsoPox. Sci rep 2:779. doi:10.1038/srep00779
Hopkins TR (1991) Physical and chemical cell disruption for the recovery of intracellular protein. In: Seetharam R, Sharma SK (eds) Purification and analysis of recombinant proteins. Macel Dekker, New York, pp 57–84
Horne I, Qiu X, Russell RJ, Oakeshott JG (2003) The phosphotriesterase gene opdA in Agrobacterium radiobacter P230 is transposable. FEMS Microbiol Lett 222:1–8. doi:10.1016/S0378-1097(03)00211-8
Jacquet P, Daudé D, Bzdrenga J, Masson P, Elias M, Chabrière E (2016) Current and emerging strategies for organophosphate decontamination: special focus on hyperstable enzymes. Environ Sci Pollut Res. doi:10.1007/s11356-016-6143-1
Jaipieam S, Visuthismajsrn P, Sutheravut P, Siriwong W, Thoumsang S, Borjam M, Robson M (2009) Organophosphate pesticides residues in drinking water from artesian wells and health risk assessment of agricultural communities, Thailand. Hum Ecol Risk Assess Int J 15:1304–1316
Jin LH, Kwun K, Lee J (2008) Intra- and extra-cellular organophosphorus hydrolase production with recombinant E. coli using fed-batch fermentation. World J Microbiol Biotechnol 24:1657–1662. doi:10.1007/s11274-008-9679-5
Kapoor M, Rajagopal R (2011) Enzymatic bioremediation of organophosphorus insecticides by recombinant organophosphorous hydrolase. Int Biodeterior Biodegradation 65:896–901. doi:10.1016/j.ibiod.2010.12.017
Knoll A, Bartsch S, Husemann B, Engel P, Schroer K, Ribeiro B, Stöckmann C, Seletzky J, Büchs J (2007) High cell density cultivation of recombinant yeasts and bacteria under non-pressurized and pressurized conditions in stirred tank bioreactors. J Biotechnol 132:167–179. doi:10.1016/j.jbiotec.2007.06.010
Korz DJ, Rinas U, Hellmuth K, Sanders EA, Deckwer WD (1995) Simple fed-batch technique for high cell density cultivation of Escherichia coli. J Biotechnol 39:59–65
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lara AR, Caspeta L, Gosset G, Bolívar F, Ramírez OT (2008) Utility of an Escherichia coli strain engineered in the substrate uptake system for improved culture performance at high glucose and cell concentrations: an alternative to fed-batch cultures. Biotechnol Bioeng 99:893–901. doi:10.1002/bit.21664
Lee SY, Choi J, Wong HH (1999) Recent advances in polyhydroxyalkanoate production by bacterial fermentation: mini-review. Int J Biol Macromol 25:31–36
Manco G, Nucci R, Febbraio F (2009) Use of esterase activities for the detection of chemical neurotoxic agents. Protein Pept Lett 16:1225–1234. doi:10.2174/092986609789071252
Martin TF (1989) Cell cracking: permeabilizing cells to macromolecular probes. Methods Enzymol 168:225–233
Masson P, Lushchekina SV (2016) Emergence of catalytic bioscavengers against organophosphorus agents. Chemico-Biological Interaction. doi:10.1016/j.cbi.2016.02.010
McLoughlin SY, Jackson C, Liu JW, Ollis DL (2004) Growth of Escherichia coli coexpressing phosphotriesterase and glycerophosphodiester phosphodiesterase, using paraoxon as the sole phosphorus source. Appl Environ Microbiol 70:404–412
Merone L, Mandrich L, Rossi M, Manco G (2005) A thermostable phosphotriesterase from the archaeon Sulfolobus solfataricus: cloning, overexpression and properties. Extremophiles 9:297–305
Merone L, Mandrich L, Rossi M, Porzio E, Rossi M, Müller S, Reiter G, Worek F, Manco G (2010) Improving the promiscuous nerve agent hydrolase activity of a thermostable archaeal lactonase. Bioresour Technol 101:9204–9212. doi:10.1016/j.biortech.2010.06.102
Mulchandani A, Kaneva I, Chen W (1999) Detoxification of organophosphate nerve agents by immobilized Escherichia coli with surface-expressed organophosphorus hydrolase. Biotechnol Bioeng 63:216–223. doi:10.1002/(SICI)1097-0290(19990420)63:2<216
Ningfeng W, Minjie D, Guoyi L, Xiaoyu C, Bin Y, Yunliu F (2004) Cloning and expression of ophc2, a new organophosphorus hydrolase gene. Chin Sci Bull 49:1245–1249. doi:10.1360/04wc0146
Arun V (2005) Convention on the prohibition of the development, production, stockpiling and use of chemical weapons and on their destruction, 3rd edn. The Technical Secretariat of the Organisation for the Prohibition of Chemical Weapons, Hague
Porzio E, Merone L, Mandrich L, Rossi M, Manco G (2007) A new phosphotriesterase from Sulfolobus acidocaldarius and its comparison with the homologue from Sulfolobus solfataricus. Biochimie 89:625–636
Ragnarsdottir KV (2000) Environmental fate and toxicology of organophosphate pesticides. J Geol Society 157:859–876. doi:10.1144/jgs.157.4.859
Restaino OF, Baskar U, Paul P, Li L, De Rosa M, Dordick JS, Linhardt RJ (2013) High cell density cultivation of a recombinant E. coli strain expressing a key enzyme in bioengineered heparin production. Appl Microbiol Biotechnol 97:3893–3900. doi:10.1007/s00253-012-4682-z
Restaino OF, Cimini D, De Rosa M, Catapano A, De Rosa M, Schiraldi C (2011) High cell density cultivation of Escherichia coli K4 in a microfiltration bioreactor: a step towards improvement of chondroitin precursor production. Microb Cell Fact 10:10. doi:10.1186/1475-2859-10-10
Saraswat M, Musante L, Ravidá A, Shortt B, Byrne B, Holthofer H (2013) Preparative purification of recombinant proteins: current status and future trends. BioMed Res Int. doi:10.1155/2013/312709
Schiraldi C, Carcarino IL, Alfano A, Restaino OF, Panariello A, De Rosa Mario (2011) Purification of chondroitin precursor from Escherichia coli K4 fermentation broth using membrane processing. Biotechnol J 6(4):410–419. doi:10.1002/biot.201000266
Schiraldi C, Alfano A, Cimini D, De Rosa M, Panariello A, Restaino OF, De Rosa M (2012) Application of a 22L scale membrane bioreactor and cross-flow ultrafiltration to obtain purified chondroitin. Biotechnol Prog 28:1012–1018. doi:10.1002/btpr
Singh BK (2009) Organophosphorus-degrading bacteria: ecology and industrial applications. Nat Rev Microbiol 7:156–164. doi:10.1038/nrmicro2050
Sinha SN, Vasudev K, Vishnu Vardhana Rao M, Odetokun M (2011) Quantification of organophosphate insecticides in drinking water in urban areas using lyophilisation and high-performance liquid chromatography-electrospray ionization-mass spectrometry techniques. Int J Mass Spectrom 300(1):12–20
Wu CF, Valdes JJ, Rao G, Bentley WE (2001) Enhancement of organophosphorous hydrolase yield in Escherichia coli using multiple gene fusions. Biotechnol Bioeng 75:100–103
Xu J, Banerjee A, Pan S, Jian Li Z (2012) Galactose can be an inducer for production of therapeutic proteins by auto-induction using E. coli BL21 strains. Protein Expr Purif 83:30–36. doi:10.1016/j.pep.2012.02.014
Yang H, Carr PD, Yu McLoughlin S, Liu JW, Horne I, Qiu X, Jeffries CMJ, Russell RJ, Oakeshott JG, Ollis DL (2003) Evolution of an organophosphate-degrading enzyme: a comparison of natural and directed evolution. Protein Eng 16:135–145. doi:10.1093/proeng/gzg015
Acknowledgements
This work was supported by the Italian Ministry of the University and Research (MIUR) and by the European Union Commission (project PON_01585 to Giuseppe Manco). All the authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Restaino, O.F., Borzacchiello, M.G., Scognamiglio, I. et al. Boosted large-scale production and purification of a thermostable archaeal phosphotriesterase-like lactonase for organophosphate decontamination. J Ind Microbiol Biotechnol 44, 363–375 (2017). https://doi.org/10.1007/s10295-016-1892-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-016-1892-x