Abstract
The novel severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) causes major challenges to the healthcare system. SARS-CoV-2 infection leads to millions of deaths worldwide and the mortality rate is found to be greatly associated with pre-existing clinical conditions. The existing dataset strongly suggests that cardiometabolic diseases including hypertension, coronary artery disease, diabetes and obesity serve as strong comorbidities in coronavirus disease (COVID-19). Studies have also shown the poor outcome of COVID-19 in patients associated with angiotensin-converting enzyme-2 polymorphism, cancer chemotherapy, chronic kidney disease, thyroid disorder, or coagulation dysfunction. A severe complication of COVID-19 is mostly seen in people with compromised medical history. SARS-CoV-2 appears to attack the respiratory system causing pneumonia, acute respiratory distress syndrome, which lead to induction of severe systemic inflammation, multi-organ dysfunction, and death mostly in the patients who are associated with pre-existing comorbidity factors. In this article, we highlighted the key comorbidities and a variety of clinical complications associated with COVID-19 for a better understanding of the etiopathogenesis of COVID-19.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection is challenging due to the lack of immunity in humans and the generation of novel variants due to frequent mutations in receptor binding domain [1]. According to the World Health Organization (WHO) (as of March seventh, 2022), 445,096,612 confirmed positive cases and 5,998,301 deaths were reported worldwide [2]. COVID-19 patients with co-existing diseases (comorbidities) need special attention and counseling for disease management. The previous meta-analysis studies have shown that the COVID-19 patients who had comorbidities including diabetes, obesity, hypertension, respiratory complications, and acute kidney failure presented severe complications [3, 4]. The elderly cohort is reported to be at higher risk of SARS-CoV-2–mediated severe complications and death, largely due to the presence of age-related co-existing clinical conditions [5, 6]. Recent studies have shown several risk factors associated with poor prognosis of the disease which further increased the mortality rate particularly in elderly patients with pre-existing cardiovascular diseases (CVDs), thyroid disorder, diabetes, renal failure, hypertension, cancer, and respiratory diseases [7, 8]. This comprehensive review aims to provide an overview of comorbidities and clinical complications associated with the severity of SARS-CoV-2 infection. We have also briefly discussed the viral genome and, morbidity, mortality, and management of COVID-19 patients associated with comorbidity factors.
Diagnosis and pathophysiology of COVID-19
Coronavirus is a positive-sense single-stranded enveloped RNA virus, which belong to the family Coronaviridae that can infect many animal species and human beings [9]. COVID-19 is an acute respiratory disease caused by SARS-CoV-2 in which viral particles are released in respiratory secretions during coughing and sneezing. For diagnosis, nasal secretions, blood, sputum, and bronchoalveolar lavage are collected from suspected COVID-19 patients. The patients’ samples are subjected to serological and molecular tests specific for SARS-CoV-2 including real-time-polymerase chain reaction (RT-PCR), enzyme-linked immunosorbent assay (ELISA) and western blots. Viral antigens present in clinical samples are detected by direct immunofluorescence assay (IFA), antibody (or serology) tests, or lateral flow tests (LFTs) [10]. The mode of transmission of SARS-CoV-2 is through inhalation of infectious aerosols and close contact with an infected individual. The virus can affect people of any age, however, more severe conditions have mostly been reported in elderly people and immunocompromised patients [11]. In the elderly population, SARS-CoV-2 often infects the lower respiratory tract which often leads to fatal pneumonia with symptoms such as fever, cough, myalgia, dyspnea with or without diarrhea. The virus first infects the epithelial lining of the throat, trachea and lung, transforming these cells into a virus reservoir that produces huge amounts of viruses and infects more cells [12, 13]. In the second week of infection, it progresses to hypoxemia, difficulty in breathing and acute respiratory distress syndrome (ARDS) where patients require intensive care and are kept on the ventilator [14].
The viral genome
The Coronaviridae family comprises five genera which include alpha, beta, gamma, delta, and omicron coronaviruses [1] (Fig. 1A, B). The viral genome is encapsulated within membrane and envelope proteins, studded with spike glycoprotein enclosed within nucleocapsid protein. The coronavirus genome consists of ORF1 at 5’ end which encodes papain-like protease (PLpro) and 3C-like protease (3Clpro or Nsp5). After invading the host cell, the genome of the virus is translated into viral proteins which are further cleaved into effector proteins by protease 3CLpro and PLpro. PLpro deubiquitinates the interferon regulatory factor 3 (IRF3) and nuclear factor kappa-light chain-enhancer of activated B cells (NF-κB), thus resulting in the suppression of the host immune system. In addition, RNA-dependent RNA polymerase (RdRp) synthesizes a full-length negative-strand RNA template to make more viral genomic RNA [10]. The interaction between the S-protein of the virus and the angiotensin-converting enzyme 2 (ACE2) receptor of the host cell is the initiation point of viral infection. The viral coat of SARS-CoV-2 expresses the spike protein containing a receptor-binding domain which binds to the ACE2 receptor and facilitates its entry into the host cell. ACE2 receptors are present abundantly on the cell membranes of the heart, lung, kidney, testis, vasculatures and intestine [12].
(A) Schematic representation shows detailed structure of SARS-CoV-2 genome. ORF1a; Open Reading Frame (266–13,468 nucleotide base pair), ORF1b; Open Reading Frame (13,468–21,563 nucleotide base pair), PLpro; Papain-like protease (4955–5900 nucleotide base pair) 3CLpro; 3CL-protease (10,055–10,977 nucleotide base pair), RdRp; RNA dependent RNA polymerase (13,442–16,236 nucleotide base pair), nsp; nonstructural protein, Endoribonuclease (19,621–20,658 nucleotide base pair), Helicase (16,237–18,043 nucleotide base pair), S Protein; Spike Protein (21,563–25,384 nucleotide base pair), E Protein; Envelope Protein (26,245–26,472 nucleotide base pair), M Protein; Membrane Protein (26,523–27,191 nucleotide base pair), N Protein; Nucleocapsid Protein (28,274–29,533 nucleotide base pair). (B) The schematic presentation shows potentially key pathogenic mutations identified in the receptor binding domain (RBD) of different SARS-CoV-2 variants. RBD is the region located between amino acids 319–541
Impact of comorbidity factors on COVID-19 severity
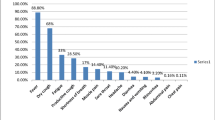
As the global pandemic has progressed, multiple research reports link several risk factors associated with the disease severity especially higher COVID-19 severity is mostly seen in the elderly population [15, 16]. To date, several research studies published related to comorbidities associated with COVID-19 infection [17]. The presence of comorbidities is associated with the severity of COVID-19 infection and the most common comorbidities include diabetes, CVD, hypertension, cancer, oxidative stress, vitamin deficiency, and kidney disease (Fig. 2). Several reports suggest that post-COVID-19 infection manifests varied clinical outcomes and a few of these are selectively summarized in Table 1.
Cardiovascular diseases
COVID-19 has a profound effect on the cardiovascular system (Fig. 3). CVDs are one of the most common causes of immature death and disability in humans and it is gradually rising worldwide. The most common types of CVDs are hypertension, stroke, ischemic heart disease, congestive cardiac failure, sickle cell disease and coagulation dysfunction. According to an epidemiological study, more than 90% of people are at risk of CVDs [28]. People with higher risk factors of cardiac diseases and other proven CVDs are more vulnerable to COVID-19 progression and seem to develop more severe disease with poorer clinical outcomes [29].
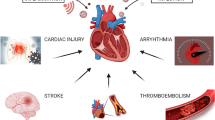
Cardiovascular manifestation and clinical complications associated with SARS-CoV-2 infection: The figure picturizes how SARS-CoV-2 infection triggers cardiovascular events upon the interaction with ACE2 receptor present on alveolar epithelial and endothelial linings. The binding of SARS-CoV-2 with ACE2 receptors induces an inflammatory reaction in alveoli which leads to compromised gaseous exchange due to fluid accumulation in interstitial space. Severe infection induces robust inflammatory reaction which produces cytokine storms, and resulting inflammatory mediators exert their adverse effects on several soft tissues including the heart and vasculatures. The SARS-CoV-2-mediated cytokine storms often cause myocarditis-induced cardiomyopathy. Moreover, SARS-CoV-2 infection is largely known to cause endothelial dysfunction that puts the patients, with pre-existing atherosclerotic plaques, at higher risk of acute coronary syndrome and myocardial infarction through destabilizing the plaques
The main clinical manifestation of COVID‐19 is the presence of respiratory symptoms; however, some patients develop severe cardiovascular and renal complications [30]. The public report released from Wuhan, Lombardy and New York supported that hypertension is the most common comorbidity among the COVID-19 patient admitted to the hospital [3, 4]. An observational study was conducted from different centers in different countries to determine the role of CVDs in COVID-19. In a study, over 3011 patients with an average age of 67 were included [31]. A total of 315 had presented cardiovascular complications like coronary artery disease (15.4%) and arrhythmia (15.1%). In the critical care unit, patients with younger age and comparatively low comorbidity factors presented fewer complications. A total of 349 patients with no co-morbidity during hospitalization, was later found to develop cardiovascular complications. Around 595 deaths were reported, and these patients were having several other complications. This study indicated that COVID-19 severity is often associated with comorbidities and the viral infection further increases the complications in cases with known CVD history [32]. Another study from China reported 44,672 confirmed cases of COVID-19 with a total of 1023 (2.3%) fatalities. Surprisingly, this fatality rate was increased to 6% in patients with hypertension. In this study, 50% of patients were of 70–79 years age group with hypertension and in this group, the fatality rate was 8% [12, 33]. The ACE2 receptor blockers has long been used to treat patients with hypertension and heart failure and ACE2 receptor emerged an important player in COVID-19 pathogenesis.
Role of ACE2 in SARS-CoV-2-induced cardiovascular complication
It is quite evident by now that SARS-CoV-2 entry is facilitated through the ACE2 receptor present on the host cell [34]. ACE2 is a fundamental enzymatic component of the renin–angiotensin–aldosterone system (RAAS). Inhibition of the RAAS plays a key role in reducing morbidity and mortality of patients with heart failure (HF) [33]. Renin cleaves hepatic peptide angiotensinogen to produce angiotensin-I (Ang-I). ACE2 hydrolyzes Ang-I and leads to the generation of biological active octapeptide Ang-II which binds with Ang-II type 1 and type 2 receptors (AT1R and AT2R). Ang-II promotes vasoconstriction, inflammation, salt and water reabsorption, and oxidative stress via the activation of AT1R [12]. The Ang-II is a part of RAAS and binds with AT1R and causes increase peripheral vascular resistance resulting in various types of cardiovascular complications such as hypertension, inflammation, coronary heart disease, myocardial fibrosis [35].
The SARS-CoV-2 virus enters the expressing cells such as alveolar cell, type I and type II pneumocytes, and in endothelial cells through the ACE2 receptor [34]. Another study shows that the binding affinity of spike glycoprotein of SAR-CoV-2 is 10–20 fold higher with ACE2 receptors [12]. These studies confirm that ACE2 is the main receptor that facilitates SARS-CoV-2 virus entry into the host cells. The RAAS plays an important role in pulmonary hypertension and pulmonary fibrosis which are generally seen in chronic obstructive pulmonary disease (COPD). The RAAS also regulates the other types of acute pulmonary disease like ARDS, which is frequently observed in severe COVID-19 patients [36]. ACE2 mediates activation of Ang-II which is a well-known factor for the pathogenesis of pulmonary hypertension. In vitro studies deciphers that the Ang-II directly causes growth/proliferation of pulmonary artery smooth muscle cells through the activation of AT1R [36]. The researchers randomly used different types of therapeutic approaches for maintaining respiratory distress in severe COVID-19. Roelie Asperem et al., investigated the effect of angiotensin receptor blockers (Losartan) with cyclic (c) Ang-II (1–7), a lanthionine-stabilized Ang-II (1–7) analog peptidase-resistant to metabolism [37]. cAng-II (1–7) is a promising clinical cardiovascular therapeutic agent which acts as a specific agonist for AT1–7/Mas receptors [38]. The lipopolysaccharides (LPS)-induced bronchoalveolar lavage fluid increased the ACE2 activity but administration of cAng-II (1–7), significantly reduced the ACE2 activity in animals. Moreover, Losartan also shows a similar effect and helps to decrease blood pressure. It maintains the PaO2 level in unexposed ventilated animals. The study indicates that LPS induced pulmonary hypertension was maintained by the losartan but not by cAng-II (1–7) [37]. Since the ACE2 receptor facilitates SARS-CoV-2 entry into the cell, it emerges as a potential target for drug development. Therefore, restoring the balance between the RAAS and the ACE2/angiotensin enzyme could help in limiting the soft organ damage and failure in COVID-19.
Coagulation dysfunction
ACE2 receptors are abundantly expressed in endothelial cells of vasculatures, and it is most likely the cause of increased coagulopathy in the COVID-19. Coagulation dysfunction, thrombosis and thromboembolism (blood clot dissolution), particularly deep vein thrombosis (DVT) are commonly seen in moderate to severe COVID-19 patients and significantly contribute to the severity of the disease [39]. The hospitalized COVID-19 patients showed increased levels of inflammatory and coagulation dysfunction markers such as C-reactive protein (CRP), ferritin, D-Dimer (DD), fibrinogen and other abnormalities such as anemia, lymphocytopenia, increased troponin I, myoglobin and lactate dehydrogenase (LDH) levels (Fig. 4) [40,41,42,43]. Platelets play critical roles in the pathogenesis of thromboembolism in severe COVID-19 [39]. These cell fragments have long been known to play only a pro-adhesive role via granule release and integrin activation [44]; however, later studies uncover the procoagulant roles of platelet in the pathogenesis of thromboembolic disorders [45, 46]. Most importantly, viral infection is often associated with platelet activation either through direct interaction with the virus or via viral-induced antibodies [47]. Platelets contribute to inflammatory reactions through interacting with inflammatory cells [39]. Collectively, these important functions put the platelet at the nexus of inflammation, coagulation dysfunction and thromboembolic complications in severe COVID-19.
The viral particles of SARS-CoV-2 infect the alveolar epithelial lining triggering a rapid immune response, which further leads to dysregulation of the immune system including elevation of a variety of inflammatory markers (cytokine storm) and coagulation dysfunction. The fibrinogen level in COVID-19 patients is increased by 2–3 folds due to cytokine storm, causing thrombus formation, particularly in severe cases [48]. Thrombotic events vary from DVT and pulmonary embolism (PE) to unusual thrombosis of central lines or arterial catheters, very early thromboses of extra-renal hemodialysis filters, and ECMO cannulas [49]. We recently reported that induction of soluble P-selectin, soluble CD40 ligand (sCD40L) at the early moderate stage of SARS-CoV-2 infection likely induces platelet activation and, severe COVID-19 patients are at a higher risk of venous, and arterial thromboembolic complications potentially due to acquired FXIII deficiency [50]. A clinical study reported an elevated level of DD in hospitalized COVID-19 patients presenting ARDS symptoms and an increased mortality rate [51]. Other cohort studies reported a correlation between increased levels of DD and COVID-19 severity [52, 53]. Moreover, studies reported the elevation in DD levels and DVT in lower limbs of COVID-19 patients [54, 55]. The radiological investigation revealed PE in severe COVID-19 patients with low oxygen saturation and elevated DD levels [56]. The venous thromboembolism (VTE) was reported to be 15% and 34% at day 7 and day 14 of infection and even higher (48%) in patients admitted to the intensive care unit (ICU) which strongly indicates VTE as a high death risk factor in COVID-19 [57]. Pulmonary CT angiography in patients with ARDS demonstrated that the PE was increased by 23% [58,59,60]. The VTE impacts not only lung, but it has broader adverse effects on different soft organs including kidney and heart. The PE, myocardial infarction (MI) and stroke are amongst the most serious consequences of thromboembolism which are often seen in severe COVID-19 patients [28]. Thus, in-depth studies of coagulation patterns in COVID-19 may help design therapeutic prophylaxis to limit coagulation dysfunction.
Sickle cell disease
Other than coagulation dysfunctions, sickle cell disease (SCD) is another blood disorder that increases the severity of COVID-19. SCD is an inherited hemoglobin disorder caused by a mutation in the beta-globin gene in which hemoglobin is prone to aggregate in hypoxic conditions [61]. Accumulating evidence suggests that patients with thalassemia and SCD are vulnerable to COVID-19 related severe complications [62]. A study on SCD patients with homozygous sickle mutation (SS) showed lymphopenia, thrombocytopenia and increased CRP levels, which were identified as poor prognostic markers in elderly COVID-19 patients [63]. In addition, another observation study reported the association of SCD trait with PE, VTE, chronic kidney disease (CKD), and exertional rhabdomyolysis [64]. Moreover, studies have suggested that SCD carriers can be considered to have an increased risk of hypercoagulability which may adversely affect clinical outcomes of SARS-CoV-2 infected patients [65]. Acute chest syndrome (ACS) is the major cause of morbidity and mortality in SCD patients and overlapping of COVID-19 and ACS may result in severe complications in SCD carriers [63, 66]. Consistent with this observation, a study has reported that irrespective of genotype or sex, COVID-19 patients with SCD (Age > 50 years) carrying increased levels of D-dimer, serum creatinine, and lactate dehydrogenase (LDH), are at higher risk of death [67]. These studies strongly suggest that people suffering from SCD are at higher risk of severe COVID-19 complications. Therefore, SCD patients infected with SARS-CoV-2 should be given special considerations and appropriate prophylaxis should be designed accordingly.
Myocarditis
Inflammation of heart muscles called myocarditis is caused due to infections, immune system activation, or exposure to certain medications [68]. Myocarditis leads to cardiomyopathy and triggers extensive scarring followed by left ventricular (LV) adverse remodeling and HF [69]. Viral infections stand out as one of the most common causes of myocardial inflammation. Clinical studies reported the association between SARS-CoV-2 and several complications leading to myocarditis [70, 71]. In a clinical assessment, the Centers for Disease Control and Prevention (CDC) reported the frequency and the risk of patients hospitalized with or without COVID-19 infection. A study on patients associated with myocarditis has reported an increased number of myocarditis cases in 2020 as compared to 2019. The same study also reported that the risk of myocarditis associated with COVID-19 infection was 16 times higher among children and elderly people [72]. Another study revealed that the myocardial muscles injury elevated the troponin I (cardiac injury marker) levels which correspond to an increased risk of mortality by 51.2% when compared to patients without cardiac muscles injury (4.5%).
Increased levels of leukocytes, DD, CRP, ferritin, and interleukin-6 (IL-6), hallmarks of myocarditis, pave the way to ARDS, kidney dysfunction, and coagulopathy [73]. Even though the precise mechanism for myocardial damage caused by SARS-CoV-2 infection is not completely elucidated, myocarditis in COVID-19 patients is an element for poor prognosis and is linked with increased death rate. Systemic inflammation post-SARS-CoV-2 infection promotes myocardium damage/injury [74]. Apart from myocarditis, pericarditis also plays a significant role in defining the cardiovascular comorbidities in SARS-CoV-2 infection. A study reported the severity of fulminant myocarditis (FM), a condition characterized by sudden, severe diffused cardiac inflammation post- SARS-CoV-2 infection. It leads to pericarditis which promotes excess fluid accumulation in the pericardial sac around the heart (pericardial effusion) ultimately leads to cardiac tamponade [75]. Accumulated evidences on myocarditis in COVID-19 demands more screening methods to diagnose and prevent the development of cardiac complications associated with the viral infection. Complimentary screening can be implemented in infected patients via. checking the troponin I levels, electrocardiogram, echocardiography etc. [76].
Diabetes
Diabetes is a metabolic disorder characterized by increased blood glucose levels due to insufficient insulin production from pancreatic β-cells [77]. Several studies reported that chronic diabetes increases the complications of other diseases such as respiratory tract infections [77, 78], cardiovascular complications [79], renal impairment [80], immunosuppressant [81], etc. Immunometabolism indicates the interaction between immunity and metabolism which play an important role in causing immune dysfunction and inflammation. The abnormal metabolic and immune system increases the risk of COVID-19 severity [82].
Hyperglycemia impedes the production of type-1 interferon (IFN) and IL-22. The type-1 IFN has antiviral activity [83]. Although IL-22 reduces chronic inflammation, activates anti-microbial immunity and improves insulin sensitivity. It causes non-enzymatic glycation of several proteins and involves the opsonization of pathogens [84]. Since the condition of diabetes mellitus interfere with the immune system, viral pathogens can easily replicate and survive in the diabetic host body [85]. Patients with SARS-CoV-2 infection along with diabetes mellitus are shown to have an increased mortality rate. Yang et al. conducted a single centered retrospective observational study to determine the complications of patients infected with SARS-CoV-2 who had pre-existing co-morbidities. In this study, a total of 9 COVID-19 patients were reported to have diabetes of which 2 (10%) survived and the other 7 (22%) died [86]. A meta-analysis was performed including18 clinical reports with a total of 15,558 COVID-19 patients. Among all the illustrated comorbidities, 11.5% were diabetic with a relatively increased mortality rate [87]. Another study included a total of 834 COVID-19 patients of which 64.6% were hypertensive, 28.1% were diabetic with an increased mortality rate [88]. These studies clearly indicate that diabetes is not only the major factor to increase the COVID-19 severity but it also increases the risk of death in COVID-19.
Thyroid disorder
Thyroid gland dysfunction leads to severe health consequences and is more commonly observed in females vs. males. Thyroid glands are responsible for human physiology, growth and homeostasis [89], and produce thyroxine (T4) and tri-iodothyronine (T3) under the control of thyroid-stimulating hormone (TSH). COVID-19 complications may involve metabolic events affected by the thyroid glands dysfunction which has emerged as a comorbid factor in COVID-19 complications [90]. Thyroid-related events occurring post-SARS-CoV-2 infection cause inflammation of thyroid glands (thyroiditis), or formation of antibodies that can damage the thyroid glands (Hashimoto’s disease), thyrotoxicosis, hypothyroidism, hyperthyroidism and Graves’ disease. The cytokine storm in COVID-19 activates immune-mediated thyroid disorder [91, 92] resulting in excessive inflammatory signals leading to cell apoptosis, increased vascular permeability which may aggravate multiple organ dysfunction [93, 94]. An amplified expression of Th1/Th17 immune responses and IL-17-related cytokine pathways are reported in COVID-19 [95]. In addition, studies reported the imbalance and hyperactivation of Th1/Th2 and Th1 and Th17, respectively, in patients with an autoimmune thyroid disorder. Increased serum levels of the Th1/Th17-related cytokines have also been reported in patients with COVID-19 [96]. Excessive cytokines circulation causes alteration in thyroid gland function leading to non-thyroidal illness (NTI) [97] which may typically decrease the serum T3, T4 and TSH levels and further reduce the enzymatic activity of 5′-monodeiodinase. In addition, type 3 iodothyronine deiodinase (D3) catalyzes the inactivation of thyroid hormones (T3 and T4). Another study reported that inflammatory cytokines such as IL-1β, IL-6, IFN-γ, and tumor necrosis factor-alpha (TNF-α) affect the functioning of the hypothalamus-pituitary thyroid (HPT) axis via. decreasing the TSH secretion. Subacute thyroiditis (SAT), an endocrine complication, is found to be associated with COVID-19 complications. Studies have reported the increased number of SAT patients associated either with COVID-19 complications [98, 99] or post COVID-19 symptoms [100,101,102]. Viremia is also reported in patients infected with SARS-CoV-2 as ACE2 receptors are highly expressed in thyroid follicles. Moreover, a study reports the expression of ACE2 receptor on thyroid epithelial cells, which could be a potential route of novel coronavirus entry [103]. These studies provide evidence that the SARS-CoV-2 virus causes thyroid inflammation upon interacting with thyroid cells through the ACE2 receptor.
Vitamin D deficiency
Vitamin D is a fat-soluble secosteroid that is produced by the epidermis of skin in response to UV exposure. Vitamin D deficiency has emerged as a cosmopolitan health condition that affects children, young adults and is more persistent in elderly population with an all-cause mortality rate [104]. Vitamin D plays some biological roles such as regulation of bone metabolism, immune system regulation, endocrine control of calcium and phosphorous homeostasis, cardiovascular system, and infectious diseases [15, 16, 105]. SARS-CoV-2 infection cause alveolar type-II pneumocyte dysfunction which increases the risk of ARDS. Vitamin D in pneumocytes type-II cells attenuates lung injury ultimately ARDS by decreasing the epithelial cell apoptosis and increasing the surfactant synthesis [106]. Nevertheless, patients with a high prevalence of vitamin D deficiency manifested organ failure and chronic renal failure [107, 108]. A study linked the prevalence of vitamin D deficiency with COVID-19 severity and suggested vitamin D deficiency likely increases the death risk in COVID-19 patients [109]. Moreover, vitamin D plays an immune system regulatory role as it suppresses adaptive immune responses in respiratory cells during viral infection [110]. As immune dysfunction is a prominent factor that promotes COVID-19 severity, hemostasis should be maintained to prevent cytokine storms to combat the severity of COVID-19. Since vitamin D prevents viral replication and downregulates the ACE2 receptors, it could be used as a dietary supplement by the COVID-19 patients to limit the progression of disease severity.
Respiratory disease
Respiratory complications directly commensurate the cardiovascular complications and become a perpetual reason of morbidity [111]. Asthma is the most common respiratory disease characterized by chronic airway inflammation [112]. The coexisting respiratory diseases like asthma, chronic obstructive pulmonary disease (COPD), pulmonary hypertension, cystic fibrosis, etc., increase the risks of COVID-19 severity [113]. Previous systemic review and meta-analysis have shown that bronchial asthma increases the severity of COVID-19 patients, and particularly, COPD increases the risk by four times [114, 115]. In chronic SARS-CoV-2 infection, many patients experience post-COVID respiratory complications; thus, the NHS published the guidelines for the patients recovered from COVID-19 to determine the respiratory symptoms and complications like pulmonary fibrosis, pulmonary edema, persistence cough, etc. [116]. The asthma-prone patients are more sensitive to viral infections, thus delaying the antiviral immune response and impaired IFN-λ secretion, which ultimately promotes disease severity [117]. In the same line, a systemic review observed that elderly asthmatic smokers have shown an increased risk of COVID-19 severity [118]. Even though COVID-19 is not directly related to asthma, many other diseases and respiratory complications are more likely to be associated with asthma. About 15–20% of COVID-19 patients experience hypoxia and require a ventilator. The cytokine storm, weak immunity, productive cough, inhalational corticosteroids, and structural destruction of pulmonary tissues develop COPD. The presence of the ACE2 receptor on pneumocytes, the target protein of the SARS-CoV-2 virus, likely increases COPD complications [17].
Oxidative stress exacerbates COVID-19 severity
The reactive oxygen species (ROS) induces oxidative stress that disrupts normal cellular function and promotes cell death [119]. ROS is seen in inflammatory disorders and other infectious diseases caused by different types of pathogens [120]. These are the key factors that connect the COVID-19 with oxidative stress. During infection, cytokine levels are increased and stimulate the immune response. Severe infection causes ARDS and a hypoxic condition that activates macrophages which leads to the induction of proinflammatory cytokines in the pulmonary microvessels [121]. The elevation of inflammatory mediators and hypoxic conditions lead to increased ROS generation and activates hypoxia-induced factor-1α (HIF-1α), IL-1β, IL-18, IL-6 and NF-κB and ultimately activates caspase-1 and caspase-2 [122, 123].
Immune disorder
The immune system is the first line of defense mechanism implemented by the human body against the invading foreign entity. Upon invasion, viral proteins trigger an immune response in the host which are implemented by host B and T cells, inducing antibodies formation. Therefore, increased levels of cytokines are released in host bodies [124]. The SARS-CoV-2 virus interacts with the immune system of the patient and causes a variety of clinical conditions followed by activation of inflammatory tissue repair response [125]. In addition, these responses may trigger cytokine storms which may severely damage host organs further worsening clinical manifest as discussed earlier [126, 127]. Different types of immune responses are linked to SARS-CoV-2 infection and details of which are listed below.
Innate immune response
An innate immune response is a kind of non-specific immune response that occurs naturally in an individual which involves physical, chemical and cellular defenses against pathogens. Viral invasion is recognized by pattern recognition receptors (PRRs), pathogen-associated molecular patterns (PAMPs), or damage-associated molecular patterns (DAMPs) such as toll-like receptor 7 (TLR7) and TLR8, and node-like receptor (NLR), by macrophages [128, 129]. Upon interaction with viral particle, PRRs triggers the activation of interferon regulatory factor (IRF), NF-κB, and AP-1 and other cells including polymorphonuclear leukocytes (PMNs), monocytes, natural killer (NK) and dendritic cells (DC) [130,131,132]. A comparative study of SARS-CoV-1 and SARS-CoV-2 reports the subsequent effects of virus infection on the immune system. It was demonstrated that both viruses infect type I and type II pneumocytes covering air sacs and modulate the levels of IL-6, MCP1, CXCL1, CXCL5, and CXCL10/IP10 cytokines [133]. In addition, another study reports the induction of proinflammatory chemokines, IL-1β, IL-6, TNF, and IL1RA in infected human lungs [129].
SARS-CoV-2 gains its entry through respiratory tracts, a mucosa-associated lymphoid tissue (MALT) that acts as a physical barrier to defend foreign entry. The level of mucosal-generated antibody, immunoglobulin A (IgA), increases during the first week of SARS-CoV-2 infection [134]. Autopsy data from virus-infected individuals demonstrated high infiltration of macrophages in hemopoietic organs such as spleen and lymph nodes where ACE2 receptor expressed, contributing to significantly increased levels of IL-6 which further exacerbates inflammation [135, 136]. Such hyperactivation of the immune system leads to cytokine storm which is now evident in severe COVID-19 cases [43]. These findings imply that the inflammatory response is potentially more harmful than the viral direct impact.
Adaptive immune response
If the innate immune system is unable to eradicate the foreign entity, the adaptive immune system takes over which comprises T lymphocytes, B lymphocytes and antibodies in the blood. Both cell-mediated and humoral immunity cell responses are associated with the COVID-19. It is in the limelight that viral infection declines the CD8 + T cells, memory CD4 + T cell and T regulatory cell count in lymph nodes and spleen [137, 138]. Similarly, studies also demonstrated the increased expression of CD94/NK group 2 member A (NKG2A) on natural killer cells (NK) and cytotoxic T cells (CTLs) in patients infected with the SARS-CoV-2 virus which was subsequently reduced in recovered individuals. Infected patients were found to express low cytokine levels such as CD107a, IFN-γ, IL-2, granzyme B, and TNF-α. Therefore, it is equitable that T cells memorized the functional activities after the infection [139]. Memory T cells, upon stimulation, triggers B cells and other immune cells to knock down the infected cells [140]. NK-like T cells express markers CD57 and killer like receptor G1(KLRG1) which induces inflammatory cytokine production such as IFN-γ and latter controls T cell receptor (TCR) signaling cascade [141,142,143]. These findings uncovered the facts of abnormal antibodies production in aging individuals suffering from COVID-19. A study reports that human CD26 binds with S protein of SARS-CoV-2 and generates ineffective T cells [144]. CD147 protein is involved in T cell activation and binds to S1 domain of S protein, facilitating its entry into the host cell [145]. The binding of SARS-CoV-2 proteins to CD147 and CD26 forms activation-induced cell death (AICD), which may lead to abnormal T cell function [146]. However, a similar study reports that the T cell apoptosis induces in MERS-CoV and the same was observed in COVID-19 patients [139, 147]. These findings may gather anomalous facts of T-cell functioning in SARS-CoV-2 infected individuals.
Humoral immune response (antibody response)
Once the virus invades the host body, the host immune system recognizes and elicits innate, adaptive or humoral responses. Antibody immune response is characterized by IgG and IgM antibody production within the host body. During the early stage of SARS-CoV-1, B-cells response against nucleocapsid (N) protein is detected, whereas response against spike protein was identified at 4–8 days after infection [148, 149]. Even though N protein is smaller than S protein, it induces antibody production upon infection [150]. Similarly, coronavirus-specific antibodies IgA, IgG and IgM were identified at the beginning of infection where IgG retain for a longer period and IgM declined after 3 months [151, 152]. A study conducted on 16 COVID-19 patients identified serum anti-S-RBD IgG antibodies. Among these patients, 15 were detected with anti-N IgG and anti-S-RBD IgM and 14 patients with serum anti-N IgM only [153]. An ELISA-based time kinetics study demonstrated that IgM and IgA antibodies were detected 5 days after the onset of initial symptoms, whereas IgG was detected after 14 days [154, 155]. In addition, a clinical study from 6 children reported protective serum IgG and IgM antibodies response towards N and S-RBD proteins of SARS-CoV-2 [156]. Such ELISA-based early diagnosis along with qRT-PCR can be employed to detect the severity of COVID-19. Secretory IgA (SIgA) protects respiratory mucosa against viral entry. SIgA neutralizes respiratory tracts of COVID-19 infected individuals where neutralizing IgA was detected in bronchoalveolar lavages of patients [157]. Another study reported the presence of neutralizing antibodies in multisystem inflammatory syndrome in children (MIS-C) which can be utilized to treat COVID-19 as these antibodies stimulate immune cells and ILs such as monocytes, lymphocytes, NK cells, myeloid chemotaxis, and IL-18 or IL-16 [158]. These data provide evidence that antibody titers specific to SARS-CoV-2 can be a source of diagnosis and therapeutic.
SARS-CoV-2 in cardio-oncology
Cancer is one of the most prevalent diseases across the globe. The disease, the treatment and the post-cancer conditions are found to affect several internal organs and hamper the efficiency of the immune system to actively fight infections. The patients with active cancers and those undergoing therapies are more exacerbated with a negative prognosis of viral infection [159, 160]. The magnitude of the problem may also vary with the type of cancer, chemotherapies/ radiotherapy, intake of immunosuppressant and other preexisting conditions like diabetes or any metabolic syndrome [160, 161]. A study on a Chinese cohort demonstrated that patients with pre-existing comorbidities had a higher risk of death of which 5.6% accounts for cancer [162]. On the bright side, the emergence of recent therapeutic strategies improved the survival rate of cancer patients; the darker side discusses the cardiotoxic side effects which in turn affects the vulnerability to the viral infection [8]. The high risk of SARS-CoV-2 infection and poor prognosis are attributed to the following categories of patients [163]:
-
Patients undergoing active chemotherapy
-
Patients administering protein kinase inhibitors or poly ADP ribose polymerase (PARP) inhibitors; patients treated with transplants of bone marrow or stem cells (in the last 6 months), or under treatment with immunosuppression drugs.
-
Patients with leukemia, lymphoma or myeloma
-
Patients undergoing radiotherapy for lung cancer
Damage mediated by SARS-CoV-2 infection is conciliated through inflammation, mainly via IL-6, low albumin level, increase in neutrophils as well as lymphopenia [164]. Lymphopenia is highly related to acute cases of SARS-CoV-2 infection. A cytokine storm prevails largely in COVID-19 patients showing a reduction in the immune cells like CD4 + T cells, CD8 + T cells, and NK cells [165]. Inflammation once elicited, it persists throughout and alters the stromal microenvironment. A loss of balance in the RAAS system combined with the loss of ACE2 in patients with COVID-19 are further contributing factors to tissue and systemic inflammation [166]. Such weakened immune systems are mainly exhibited by cancer patients too; with active cancers or the ones receiving chemo or immunotherapies. SARS-CoV-2 is one such opportunistic pathogen that takes advantage of such hosts and accounts for the major cause of mortality in cancer-related COVID-19 patients [167].
Cancer associated cardiac complications and treatment regimens
Zordoky alluringly described three possible scenarios for increased cardiovascular complications of COVID-19 in cancer survivors. The first one pinpoints the pro-inflammatory, pro-apoptotic and pro-oxidative effect which synergizes myocardial damages with anticancer therapies along with viral infection. Second, the harmful cardiotoxic effect as an outcome of cancer therapies which renders the patients more vulnerable to SARS-CoV-2 infection. The third scenario emphasizes on the cumulative risk factors due to COVID-19 in cancer patients which further complicates the cardiovascular outcome in such patients [168]. In such situations, certain cancer therapeutics which falls under anthracyclines, anti-growth factors (eg: HER2 blocking antibodies) and inhibitors (tyrosine kinase inhibitors, protease inhibitors, checkpoint inhibitors etc.); which potentially increase apoptosis, oxidative stress or necrosis may affect myocardial cells and compromises the cardiac health leading to cardiomyopathies. A controversial fact exists for the use of checkpoint inhibitors, a drug that is a boon not only for cancer patients but also occasionally for COVID-19 patients [169, 170]. Checkpoint inhibitors are found to stimulate macrophage infiltration and lymphocytes which exerts immune-stimulating response but also activates survival pathways through the inhibition of CTLA-4 and PDL-1 [171, 172]. The combination therapies used with immune checkpoint inhibitors increase the prevalence of myocarditis in cancer patients when compared to monotherapies. For example, Nivolumab treated in combination with Ipilimumab increased the rate of myocarditis in patients from 0.6 to 0.27% when compared to Nivolumab monotherapy [173, 174]. Moreover in lung cancer patients, Nivolumab causes acute lymphocytic myocarditis [175]. Therefore, to compare the clinical benefits of immune checkpoint inhibitor treatment in cancer and COVID-19 patients with respect to the possible after effect such as myocarditis are subjected to further validation using retrospective studies.
Certain types of cancer like lung, brain, gastric, pancreatic, genito-urinal and some metastatic cancers aggravate conditions like bleeding and VTE [176]. Platinum-based anticancer drugs like cisplatin, hormonal therapies like tamoxifen, anti-VEGF therapies like Bevacizumab, immunomodulators like Thalidomide, Bcr-Abl kinase inhibitors like Nilotinib, proteasome inhibitors like Carfilzomib increases the risk factors in cancer patients for elevated coagulation and inhibition of fibrinolysis during SARS-CoV-2 infection [177]. The hyperactivation of T-cytotoxic lymphocytes and NK cells as a secondary inflammatory condition in COVID-19 patients induces the risk for myocarditis and VTE which may lead to death accounted with ARDS. A high level of fibrin degradation was reported in death cases of SARS-CoV-2 infection, especially those cases which were reported to express disseminated intravascular coagulation (DIC) [178, 179]. It is comprehensive that anticancer therapies could aggravate the damages due to intravascular coagulation in COVID-19 patients. In such situations, low molecular weight heparins are best suggested for the treatment of critically ill patients along with a proper thromboprophylax is regimen [51, 180]. A self-contradiction of increased thrombotic risk parallel to increased bleeding risks is associated with cancer patients. Khorana score is a tool used to address the prophylactic score of VTE in cancer patients [129]. Once records of Khorana score are applied with COVID-19 patients, the scenario will appear clearer in the context of COVID-19 related thromboembolism in cancer patients.
Myocarditis, type II MI, acute coronary syndrome and stress-related cardiomyopathy are widely categorized into the etiology of myocardial injury in cancer patients [181]. It was reported that cancer patients exhibiting high levels of troponin and possible acute coronary disease are found to have type II MI [182]. This situation can highly be in tune with the fact that COVID-19 patients; (reported from China) who expired without any CVD history; either had elevated cardiac troponin I levels or a cardiac arrest during hospitalization [183]. Thereafter, medical societies across the world recommend the monitoring of troponin I level for patients presented with cancer/cancer history with viral infection having any significant cardiac involvement. Stress cardiomyopathy aka Takutsubo syndrome is well associated with cancer patients with adverse clinical outcomes like thromboembolism, shock and arrhythmias [184, 185]. Such patients are usually given respiratory support. The reason for Takutsubo syndrome mainly being physical and emotional stress is also very much accredited with SARS-CoV-2 infection [186]. Even though the combination of the two- cancer and SARS-CoV-2 infection will increase the incidence of Takutsubo syndrome, a retrospective study is recommended to confirm the occurrence.
Drugs like hydroxychloroquine, azithromycin etc. used against emergencies in COVID-19 situations earlier are known to cause QT prolongation [187]. Similarly, certain anti-cancer drugs like tyrosine kinase inhibitors, HDAC inhibitors and antidepressants are also known to promote QT prolongation [188, 189]. It will be important to learn how COVID-19 treatments in cancer patients are affecting the QT prolongation due to which patients are at increased risk of fatal arrhythmia. Since chloroquine drugs are not in much use for COVID-19 treatment now and drugs like remdesivir are less likely to cause QT prolongation, no arrhythmias were actually reported from COVID-19 cases [190]. However, cancer therapies can potentially be risky and render the patients with viral infection more prone to QT prolongation-induced arrhythmias. Apart from the irregular heartbeat conditions like arrhythmia, certain possibilities are also likely to happen with COVID-19 related cardiac events in cancer patients. One such is a condition affecting the tissues around the heart called as pericarditis as well as a condition of fluid retention around the heart called as pericardial tamponade [191]. It is postulated that SARS-CoV-2 infection can impart myopericarditis in patients with the severity of the disease [74, 192]. Similarly, certain cancers, as well as anti-cancer therapeutics, are also reported to cause myopericarditis which may sometimes be presented with pericardial tamponade [75]. In those patients with active cancer or anti-cancer treatments undergoing SARS-CoV-2 infection, it will be difficult to predict the cause or occurrence of pericarditis and its aftermath. It is advised to follow the NSAID category of drugs and immediate critical care if a patient is presented with pericarditis upon viral infection.
CVDs stand as the most important cause of mortality in COVID-19 patients. It is absolutely paramount in this situation to increase cardiovascular screening or surveillance in cancer patients. Those patients who are either immunocompromised or have a history of any cardiovascular events or metabolic diseases or are of higher age should be provided with prudent cardiovascular screening. Strict precautions and preventive measures should be adopted to keep cancer patients or post-cancer care patients against contact with the SARS-CoV-2 infection [193]. Once a cancer patient is suspected to come in contact with the virus, immediate medical attention should be given with timely screening of the organ functions. During the SARS-CoV-2 infection, the delayed occurrence of acute MI and subsequent complicated events leading to death has been reported. Even with the end of the third wave of the SARS-CoV-2 infection globally, information is still lacking to deeply excavate the real correlation between COVID-19 and cardio-oncology and more retrospective studies in larger cohorts are in high demand to tailor the treatment strategies (including both supportive and targeted therapies) for COVID-19-cardio-oncology axis.
Alzheimer’s disease
Alzheimer’s disease (AD), a progressive neurological disorder, has been reported as a clinical complication and, comorbidity in elderly COVID-19 patients [194]. AD is a common cause of dementia where a person loses cognitive function. Accumulating evidence suggests that SARS-CoV-2 enters the brain and makes morphological changes which is the likely cause of cognitive impairment in some COVID-19 patients [195]. A recent study has shown that SARS-CoV-2 infection leads to Alzheimer’s-like changes in the brain of some COVID-19 patients [196]. In addition, the central nervous system (CNS) and the peripheral nervous system (PNS) are both damaged by SARS-CoV-2 and also show lengthy impairment [197]. A retrospective cohort study in Spain reported that around 29.1% of COVID-19 patients with AD exhibited further cognitive impairment which significantly increased the morbidity rate in elderly people [198]. Several studies have reported that AD is a common predictive factor to mortality in COVID-19. AD patients infected with SARS-CoV-2 have shorter survival than the COVID-19 patients without AD. Around 54.5% death rates were observed in COVID-19 patients with AD [199]. Another cohort study reports that 25.2% of COVID-19 patients with AD, mainly elderly, died despite active treatment [200]. COVID-19 infection may lead to neurological symptoms as an observational study from China reported that 14.8% of hospitalized COVID-19 patients suffered from neurological manifestations including impaired consciousness [201]. In addition, similar observations were reported from the European cohort where critical COVID-19 patients showed working memory dysfunction. Around 65% of COVID-19 patients admitted to the ICU showed confusion [202, 203]. ACE2 receptors are highly expressed in different brain regions which provide an initial target for SARS-CoV-2 invasion. A study reported that the pons and medulla oblongata in the human brainstem were more susceptible to virus entry and subsequently caused neurodegenerative disorder [204]. Along with AD, CNS damage results in Parkinson’s disease (PD) and is associated with risk factors such as aging, family history, environment and chemical exposure [205]. A study reported that PD is not a risk factor for COVID-19 [206]. This study suggested that some PD patients with COVID-19 present with worsening parkinsonian symptoms, requiring increased anti-PD therapy with worse outcomes. These studies suggest that COVID-19 patients need long-term clinical follow-up to avoid further COVID-19 complications.
Conclusions
Treating COVID-19 patients is a crucial task for healthcare providers despite few anti-viral medications and, vaccines being available for prevention. COVID-19 causes an endless loop of mortality and morbidity in patients suffering from CVD, diabetes, hypertension, thyroid disorder. These complications ultimately lead to multi-organ failure eventually leading to death. Comorbidities such as CVD, diabetes, thyroid, respiratory, vitamin D deficiency and hypertension (Fig. 5) have been reported as high-risk factors in COVID-19 patients and special consideration must be provided to such patients. Since the pre-existing comorbidity is the leading cause of mortality in COVID-19 patients, a thorough clinical assessment is required during hospitalization. Patients with comorbidity factors should take preventive care to safeguard their survival entity and should be prioritized for the vaccination.
References
Khateeb J, Li Y, Zhang H. Emerging SARS-CoV-2 variants of concern and potential intervention approaches. Crit Care. 2021;25:244.
WHO. Coronavirus disease (COVID-19) pandemic, 2021.
Wang Z, Deng H, Ou C, et al. Clinical symptoms, comorbidities and complications in severe and non-severe patients with COVID-19: a systematic review and meta-analysis without cases duplication. Medicine (Baltimore). 2020;99:e23327.
Yang J, Zheng Y, Gou X, et al. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis. Int J Infect Dis. 2020;94:91–5.
Eskandari A, Brojakowska A, Bisserier M, et al. Retrospective analysis of demographic factors in COVID-19 patients entering the Mount Sinai Health System. PLoS ONE. 2021;16:e0254707.
Pepe M, Maroun-Eid C, Romero R, et al. Clinical presentation, therapeutic approach, and outcome of young patients admitted for COVID-19, with respect to the elderly counterpart. Clin Exp Med. 2021;21:249–68.
Hebbard C, Lee B, Katare R, Garikipati VNS. Diabetes, heart failure, and COVID-19: an update. Front Physiol. 2021;12:706185.
Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the chinese center for disease control and prevention. JAMA. 2020;323:1239–42.
V’Kovski P, Kratzel A, Steiner S, Stalder H, Thiel V. Coronavirus biology and replication: implications for SARS-CoV-2. Nat Rev Microbiol. 2021;19:155–70.
Hariharan A, Hakeem AR, Radhakrishnan S, Reddy MS, Rela M. The role and therapeutic potential of NF-kappa-B pathway in severe COVID-19 patients. Inflammopharmacology. 2021;29:91–100.
Liu K, Chen Y, Lin R, Han K. Clinical features of COVID-19 in elderly patients: a comparison with young and middle-aged patients. J Infect. 2020;80:e14–8.
Patel VB, Zhong JC, Grant MB, Oudit GY. Role of the ACE2/Angiotensin 1–7 axis of the renin-angiotensin system in heart failure. Circ Res. 2016;118:1313–26.
South AM, Diz DI, Chappell MC. COVID-19, ACE2, and the cardiovascular consequences. Am J Physiol Heart Circ Physiol. 2020;318:H1084–90.
Gibson PG, Qin L, Puah SH. COVID-19 acute respiratory distress syndrome (ARDS): clinical features and differences from typical pre-COVID-19 ARDS. Med J Aust. 2020;213:54-6e1.
Valderas JM, Starfield B, Sibbald B, Salisbury C, Roland M. Defining comorbidity: implications for understanding health and health services. Ann Fam Med. 2009;7:357–63.
Ording AG, Sorensen HT. Concepts of comorbidities, multiple morbidities, complications, and their clinical epidemiologic analogs. Clin Epidemiol. 2013;5:199–203.
Ejaz H, Alsrhani A, Zafar A, et al. COVID-19 and comorbidities: deleterious impact on infected patients. J Infect Public Health. 2020;13:1833–9.
Lee SC, Son KJ, Han CH, Jung JY, Park SC. Impact of comorbid asthma on severity of coronavirus disease (COVID-19). Sci Rep. 2020;10:21805.
Zhang N, Wang C, Zhu F, et al. Risk factors for poor outcomes of diabetes patients with COVID-19: a single-center, retrospective study in early outbreak in China. Front Endocrinol (Lausanne). 2020;11:571037.
Gaztanaga J, Ramasamy R, Schmidt AM, et al. A pilot open-label study of aldose reductase inhibition with AT-001 (caficrestat) in patients hospitalized for COVID-19 infection: results from a registry-based matched-control analysis. Diabetes Metab Syndr. 2021;15:102328.
Ghanem M, Homps-Legrand M, Garnier M, et al. Blood fibrocytes are associated with severity and prognosis in COVID-19 pneumonia. Am J Physiol Lung Cell Mol Physiol. 2021;321:L847–58.
Gibertoni D, Reno C, Rucci P, et al. COVID-19 incidence and mortality in non-dialysis chronic kidney disease patients. PLoS ONE. 2021;16:e0254525.
Kantroo V, Kanwar MS, Goyal P, et al. Mortality and clinical outcomes among patients with COVID-19 and diabetes. Med Sci (Basel) 2021; 9.
Coca A, Burballa C, Centellas-Perez FJ, et al. Outcomes of COVID-19 among hospitalized patients with non-dialysis CKD. Front Med (Lausanne). 2020;7:615312.
Abayomi A, Osibogun A, Kanma-Okafor O, et al. Morbidity and mortality outcomes of COVID-19 patients with and without hypertension in Lagos, Nigeria: a retrospective cohort study. Glob Health Res Policy. 2021;6:26.
Espiritu AI, Chiu HHC, Sy MCC, Anlacan VMM, Jamora RDG. The outcomes of patients with diabetes mellitus in The Philippine CORONA Study. Sci Rep. 2021;11:24436.
van Gerwen M, Alsen M, Little C, et al. Outcomes of patients with hypothyroidism and COVID-19: a retrospective cohort study. Front Endocrinol (Lausanne). 2020;11:565.
Flora GD, Nayak MK. A brief review of cardiovascular diseases, associated risk factors and current treatment regimes. Curr Pharm Des. 2019;25:4063–84.
Brojakowska A, Eskandari A, Bisserier M, et al. Comorbidities, sequelae, blood biomarkers and their associated clinical outcomes in the Mount Sinai Health System COVID-19 patients. PLoS ONE. 2021;16:e0253660.
Lazaridis C, Vlachogiannis NI, Bakogiannis C, et al. Involvement of cardiovascular system as the critical point in coronavirus disease 2019 (COVID-19) prognosis and recovery. Hellenic J Cardiol. 2020;61:381–95.
Giri M, Puri A, Wang T, Guo S. Clinical features, comorbidities, complications and treatment options in severe and non-severe COVID-19 patients: a systemic review and meta-analysis. Nurs Open. 2021;8:1077–88.
Linschoten M, Peters S, van Smeden M, et al. Cardiac complications in patients hospitalised with COVID-19. Eur Heart J Acute Cardiovasc Care. 2020;9:817–23.
Zhou P, Yang XL, Wang XG, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–3.
Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(271–80):e8.
Petrovic V, Radenkovic D, Radenkovic G, Djordjevic V, Banach M. Pathophysiology of cardiovascular complications in COVID-19. Front Physiol. 2020;11:575600.
Kuba K, Imai Y, Penninger JM. Angiotensin-converting enzyme 2 in lung diseases. Curr Opin Pharmacol. 2006;6:271–6.
Wosten-van Asperen RM, Lutter R, Specht PA, et al. Acute respiratory distress syndrome leads to reduced ratio of ACE/ACE2 activities and is prevented by angiotensin-(1–7) or an angiotensin II receptor antagonist. J Pathol. 2011;225:618–27.
Seva Pessoa B, Becher PM, Van Veghel R, et al. Effect of a stable angiotensin-(1–7) analogue on progenitor cell recruitment and cardiovascular function post myocardial infarction. J Am Heart Assoc 2015; 4.
Ahmad F, Kannan M, Ansari AW. Role of SARS-CoV-2 -induced cytokines and growth factors in coagulopathy and thromboembolism. Cytokine Growth Factor Rev. 2022;63:58–68.
Al-Tamimi AO, Yusuf AM, Jayakumar MN, et al. Induction of soluble P-selectin and CD40 ligand and, FXIII deficiency promote aberrant coagulation and thromboembolism in severe COVID-19. Circ Res. 2021;129:AP357.
Bergamaschi G, Borrelli de Andreis F, Aronico N, et al. Anemia in patients with Covid-19: pathogenesis and clinical significance. Clin Exp Med. 2021;21:239–46.
Goyal P, Choi JJ, Pinheiro LC, et al. Clinical characteristics of Covid-19 in New York City. N Engl J Med. 2020;382:2372–4.
Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. China Lancet. 2020;395:497–506.
Kannan M, Ahmad F, Saxena R. Platelet activation markers in evaluation of thrombotic risk factors in various clinical settings. Blood Rev. 2019;37:100583.
Ahmad F, Boulaftali Y, Greene TK, et al. Relative contributions of stromal interaction molecule 1 and CalDAG-GEFI to calcium-dependent platelet activation and thrombosis. J Thromb Haemost. 2011;9:2077–86.
Reddy EC, Rand ML. Procoagulant phosphatidylserine-exposing platelets in vitro and in vivo. Front Cardiovasc Med. 2020;7:15.
Assinger A. Platelets and infection - an emerging role of platelets in viral infection. Front Immunol. 2014;5:649.
Al-Samkari H, Karp Leaf RS, Dzik WH, et al. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136:489–500.
Mezalek ZT, Khibri H, Ammouri W, et al. COVID-19 associated coagulopathy and thrombotic complications. Clin Appl Thromb Hemost. 2020;26:1076029620948137.
Al-Tamimi AO, Yusuf AM, Jayakumar MN, et al. SARS-CoV-2 infection induces soluble platelet activation markers and PAI-1 in the early moderate stage of COVID-19. Int J Lab Hematol 2022.
Tang N, Bai H, Chen X, et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18:1094–9.
Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan. China JAMA Intern Med. 2020;180:934–43.
Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–62.
Cui S, Chen S, Li X, Liu S, Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18:1421–4.
Ren B, Yan F, Deng Z, et al. Extremely high incidence of lower extremity deep venous thrombosis in 48 patients with severe COVID-19 in Wuhan. Circulation. 2020;142:181–3.
Helms J, Tacquard C, Severac F, et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intens Care Med. 2020;46:1089–98.
Middeldorp S, Coppens M, van Haaps TF, et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. 2020;18:1995–2002.
Leonard-Lorant I, Delabranche X, Severac F, et al. Acute pulmonary embolism in patients with COVID-19 at CT angiography and relationship to d-dimer levels. Radiology. 2020;296:E189–91.
Poyiadji N, Cormier P, Patel PY, et al. Acute pulmonary embolism and COVID-19. Radiology. 2020;297:E335–8.
Grillet F, Behr J, Calame P, Aubry S, Delabrousse E. Acute pulmonary embolism associated with COVID-19 pneumonia detected with pulmonary CT angiography. Radiology. 2020;296:E186–8.
Hassell KL. Population estimates of sickle cell disease in the U.S. Am J Prev Med. 2010;38:S512–21.
Mucalo L, Brandow AM, Dasgupta M, et al. Comorbidities are risk factors for hospitalization and serious COVID-19 illness in children and adults with sickle cell disease. Blood Adv. 2021;5:2717–24.
Hussain FA, Njoku FU, Saraf SL, et al. COVID-19 infection in patients with sickle cell disease. Br J Haematol. 2020;189:851–2.
Naik RP, Smith-Whitley K, Hassell KL, et al. Clinical outcomes associated with sickle cell trait: a systematic review. Ann Intern Med. 2018;169:619–27.
Kehinde TA, Osundiji MA. Sickle cell trait and the potential risk of severe coronavirus disease 2019-A mini-review. Eur J Haematol. 2020;105:519–23.
Singh A, Brandow AM, Panepinto JA. COVID-19 in individuals with sickle cell disease/trait compared with other Black individuals. Blood Adv. 2021;5:1915–21.
Minniti CP, Zaidi AU, Nouraie M, et al. Clinical predictors of poor outcomes in patients with sickle cell disease and COVID-19 infection. Blood Adv. 2021;5:207–15.
Caforio AL, Pankuweit S, Arbustini E, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34(2636–48):48a-d.
Towbin JA, Lowe AM, Colan SD, et al. Incidence, causes, and outcomes of dilated cardiomyopathy in children. JAMA. 2006;296:1867–76.
Murk W, Gierada M, Fralick M, et al. Diagnosis-wide analysis of COVID-19 complications: an exposure-crossover study. CMAJ. 2021;193:E10-8.
Barda N, Dagan N, Ben-Shlomo Y, et al. Safety of the BNT162b2 mRNA Covid-19 vaccine in a nationwide setting. N Engl J Med. 2021;385:1078–90.
Boehmer TK, Kompaniyets L, Lavery AM, et al. Association between COVID-19 and Myocarditis using hospital-based administrative data - United States, March 2020-January 2021. MMWR Morb Mortal Wkly Rep. 2021;70:1228–32.
Shi S, Qin M, Shen B, et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020;5:802–10.
Inciardi RM, Lupi L, Zaccone G, et al. Cardiac involvement in a patient with coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020;5:819–24.
Hua A, O’Gallagher K, Sado D, Byrne J. Life-threatening cardiac tamponade complicating myo-pericarditis in COVID-19. Eur Heart J. 2020;41:2130.
Azevedo RB, Botelho BG, Hollanda JVG, et al. Covid-19 and the cardiovascular system: a comprehensive review. J Hum Hypertens. 2021;35:4–11.
Berbudi A, Rahmadika N, Tjahjadi AI, Ruslami R. Type 2 diabetes and its impact on the immune system. Curr Diabetes Rev. 2020;16:442–9.
Sharma B, Khanal VK, Jha N, Pyakurel P, Gurung GN. Study of the magnitude of diabetes and its associated risk factors among the tuberculosis patients of Morang. Eastern Nepal BMC public health. 2019;19:1545.
Shah AD, Langenberg C, Rapsomaniki E, et al. Type 2 diabetes and incidence of cardiovascular diseases: a cohort study in 1.9 million people. Lancet Diabetes Endocrinol. 2015;3:105–13.
Jitraknatee J, Ruengorn C, Nochaiwong S. Prevalence and risk factors of chronic kidney disease among type 2 diabetes patients: a cross-sectional study in primary care practice. Sci Rep. 2020;10:1–10.
Ferlita S, Yegiazaryan A, Noori N, et al. Type 2 Diabetes mellitus and altered immune system leading to susceptibility to pathogens, especially mycobacterium tuberculosis. J Clin Med 2019; 8.
Daryabor G, Atashzar MR, Kabelitz D, Meri S, Kalantar K. The effects of type 2 diabetes mellitus on organ metabolism and the immune system. Front Immunol. 2020;11:1582.
Lin JY, Kuo RL, Huang HI. Activation of type I interferon antiviral response in human neural stem cells. Stem Cell Res Ther. 2019;10:387.
Toniolo A, Cassani G, Puggioni A, et al. The diabetes pandemic and associated infections: suggestions for clinical microbiology. Rev Med Microbiol. 2019;30:1–17.
Rouse BT, Sehrawat S. Immunity and immunopathology to viruses: What decides the outcome? Nat Rev Immunol. 2010;10:514–26.
Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8:475–81.
Singh AK, Gillies CL, Singh R, et al. Prevalence of co-morbidities and their association with mortality in patients with COVID-19: a systematic review and meta-analysis. Diabetes Obes Metab. 2020;22:1915–24.
Posso M, Comas M, Roman M, et al. Comorbidities and mortality in patients with COVID-19 aged 60 years and older in a university hospital in Spain. Arch Bronconeumol (Engl Ed). 2020;56:756–8.
Kumari K, Chainy GBN, Subudhi U. Prospective role of thyroid disorders in monitoring COVID-19 pandemic. Heliyon. 2020;6:e05712.
Ruggeri RM, Campenni A, Deandreis D, et al. SARS-COV-2-related immune-inflammatory thyroid disorders: facts and perspectives. Expert Rev Clin Immunol. 2021;17:737–59.
Picchianti Diamanti A, Rosado MM, Pioli C, Sesti G, Lagana B. Cytokine release syndrome in COVID-19 patients, a new scenario for an old concern: the fragile balance between infections and autoimmunity. Int J Mol Sci 2020; 21.
Moore JB, June CH. Cytokine release syndrome in severe COVID-19. Science. 2020;368:473–4.
Gavriatopoulou M, Korompoki E, Fotiou D, et al. Organ-specific manifestations of COVID-19 infection. Clin Exp Med. 2020;20:493–506.
Berlin DA, Gulick RM, Martinez FJ. Severe Covid-19. N Engl J Med. 2020;383:2451–60.
De Biasi S, Meschiari M, Gibellini L, et al. Marked T cell activation, senescence, exhaustion and skewing towards TH17 in patients with COVID-19 pneumonia. Nat Commun. 2020;11:3434.
Pavel AB, Glickman JW, Michels JR, et al. Th2/Th1 cytokine imbalance is associated with higher COVID-19 risk mortality. Front Genet. 2021;12:706902.
Moutchia J, Pokharel P, Kerri A, et al. Clinical laboratory parameters associated with severe or critical novel coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis. PLoS ONE. 2020;15:e0239802.
Asfuroglu Kalkan E, Ates I. A case of subacute thyroiditis associated with Covid-19 infection. J Endocrinol Invest. 2020;43:1173–4.
Brancatella A, Ricci D, Viola N, et al. Subacute thyroiditis after sars-COV-2 infection. J Clin Endocrinol Metab 2020; 105.
Ruggeri RM, Campenni A, Siracusa M, Frazzetto G, Gullo D. Subacute thyroiditis in a patient infected with SARS-COV-2: an endocrine complication linked to the COVID-19 pandemic. Hormones (Athens). 2021;20:219–21.
Khatri A, Charlap E, Kim A. Subacute thyroiditis from COVID-19 infection: a case report and review of literature. Eur Thyroid J. 2021;9:324–8.
Dworakowska D, Morley S, Mulholland N, Grossman AB. COVID-19-related thyroiditis: A novel disease entity? Clin Endocrinol (Oxf). 2021;95:369–77.
Rotondi M, Coperchini F, Ricci G, et al. Detection of SARS-COV-2 receptor ACE-2 mRNA in thyroid cells: a clue for COVID-19-related subacute thyroiditis. J Endocrinol Invest. 2021;44:1085–90.
Holick MF. The vitamin D deficiency pandemic: approaches for diagnosis, treatment and prevention. Rev Endocr Metab Disord. 2017;18:153–65.
Grasselli G, Zangrillo A, Zanella A, et al. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region. Italy JAMA. 2020;323:1574–81.
Clark CE, McDonagh STJ, McManus RJ, Martin U. COVID-19 and hypertension: risks and management. a scientific statement on behalf of the British and Irish Hypertension Society. J Hum Hypertens. 2021;35:304–7.
Courbebaisse M, Alberti C, Colas S, et al. VITamin D supplementation in renAL transplant recipients (VITALE): a prospective, multicentre, double-blind, randomized trial of vitamin D estimating the benefit and safety of vitamin D3 treatment at a dose of 100,000 UI compared with a dose of 12,000 UI in renal transplant recipients: study protocol for a double-blind, randomized, controlled trial. Trials. 2014;15:430.
Zhou Q, Li L, Chen Y, et al. Vitamin D supplementation could reduce the risk of acute cellular rejection and infection in vitamin D deficient liver allograft recipients. Int Immunopharmacol. 2019;75:105811.
Grant WB, Lahore H, McDonnell SL, et al. Evidence that Vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020; 12.
Greiller CL, Martineau AR. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients. 2015;7:4240–70.
Chowdhuri S, Crook ED, Taylor HA Jr, Badr MS. Cardiovascular complications of respiratory diseases. Am J Med Sci. 2007;334:361–80.
Papi A, Brightling C, Pedersen SE, Reddel HK. Asthma. Lancet. 2018;391:783–800.
Beltramo G, Cottenet J, Mariet AS, et al. Chronic respiratory diseases are predictors of severe outcome in COVID-19 hospitalised patients: a nationwide study. Eur Respir J 2021; 58.
Sanchez-Ramirez DC, Mackey D. Underlying respiratory diseases, specifically COPD, and smoking are associated with severe COVID-19 outcomes: a systematic review and meta-analysis. Respir Med. 2020;171:106096.
Aveyard P, Gao M, Lindson N, et al. Association between pre-existing respiratory disease and its treatment, and severe COVID-19: a population cohort study. Lancet Respir Med. 2021;9:909–23.
Fraser E. Long term respiratory complications of covid-19. BMJ. 2020;370:m3001.
Contoli M, Message SD, Laza-Stanca V, et al. Role of deficient type III interferon-lambda production in asthma exacerbations. Nat Med. 2006;12:1023–6.
Zhao Q, Meng M, Kumar R, et al. The impact of COPD and smoking history on the severity of COVID-19: a systemic review and meta-analysis. J Med Virol. 2020;92:1915–21.
Forcados GE, Muhammad A, Oladipo OO, Makama S, Meseko CA. Metabolic implications of oxidative stress and inflammatory process in SARS-CoV-2 pathogenesis: therapeutic potential of natural antioxidants. Front Cell Infect Microbiol. 2021;11:654813.
Maltepe E, Saugstad OD. Oxygen in health and disease: regulation of oxygen homeostasis–clinical implications. Pediatr Res. 2009;65:261–8.
Cecchini R, Cecchini AL. SARS-CoV-2 infection pathogenesis is related to oxidative stress as a response to aggression. Med Hypotheses. 2020;143:110102.
Alwazeer D, Liu FF, Wu XY, LeBaron TW. Combating oxidative stress and inflammation in COVID-19 by molecular hydrogen therapy: mechanisms and perspectives. Oxid Med Cell Longev. 2021;2021:5513868.
Wang C, Xie J, Zhao L, et al. Alveolar macrophage dysfunction and cytokine storm in the pathogenesis of two severe COVID-19 patients. EBioMedicine. 2020;57:102833.
Hughes MM, O’Neill LAJ. Metabolic regulation of NLRP3. Immunol Rev. 2018;281:88–98.
Spihlman AP, Gadi N, Wu SC, Moulton VR. COVID-19 and systemic lupus erythematosus: focus on immune response and therapeutics. Front Immunol. 2020;11:589474.
Garcia LF. Immune Response, inflammation, and the clinical spectrum of COVID-19. Front Immunol. 2020;11:1441.
Tay MZ, Poh CM, Renia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. 2020;20:363–74.
Fung TS, Liu DX. Human coronavirus: host-pathogen interaction. Annu Rev Microbiol. 2019;73:529–57.
Barnes GD, Burnett A, Allen A, et al. Thromboembolism and anticoagulant therapy during the COVID-19 pandemic: interim clinical guidance from the anticoagulation forum. J Thromb Thrombolysis. 2020;50:72–81.
Hur S, Double-Stranded RNA. Sensors and modulators in innate immunity. Annu Rev Immunol. 2019;37:349–75.
Perlman S, Dandekar AA. Immunopathogenesis of coronavirus infections: implications for SARS. Nat Rev Immunol. 2005;5:917–27.
Chen J, Subbarao K. The immunobiology of SARS*. Annu Rev Immunol. 2007;25:443–72.
Blanco-Melo D, Nilsson-Payant BE, Liu WC, et al. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020;181(1036–45):e9.
Padoan A, Sciacovelli L, Basso D, et al. IgA-Ab response to spike glycoprotein of SARS-CoV-2 in patients with COVID-19: a longitudinal study. Clin Chim Acta. 2020;507:164–6.
Barton LM, Duval EJ, Stroberg E, Ghosh S, Mukhopadhyay S. COVID-19 Autopsies, Oklahoma, USA. Am J Clin Pathol. 2020;153:725–33.
Park MD. Macrophages: a Trojan horse in COVID-19? Nat Rev Immunol. 2020;20:351.
Prompetchara E, Ketloy C, Palaga T. Immune responses in COVID-19 and potential vaccines: lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol. 2020;38:1–9.
Russell B, Moss C, George G, et al. Associations between immune-suppressive and stimulating drugs and novel COVID-19-a systematic review of current evidence. ecancermedicalscience. 2020;14:1022.
Zheng M, Gao Y, Wang G, et al. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol Immunol. 2020;17:533–5.
Stockinger B, Bourgeois C, Kassiotis G. CD4+ memory T cells: functional differentiation and homeostasis. Immunol Rev. 2006;211:39–48.
Michel JJ, Griffin P, Vallejo AN. Functionally diverse NK-like T cells are effectors and predictors of successful aging. Front Immunol. 2016;7:530.
Henson SM, Franzese O, Macaulay R, et al. KLRG1 signaling induces defective Akt (ser473) phosphorylation and proliferative dysfunction of highly differentiated CD8+ T cells. Blood. 2009;113:6619–28.
Brenchley JM, Karandikar NJ, Betts MR, et al. Expression of CD57 defines replicative senescence and antigen-induced apoptotic death of CD8+ T cells. Blood. 2003;101:2711–20.
Bailey SR, Nelson MH, Majchrzak K, et al. Human CD26(high) T cells elicit tumor immunity against multiple malignancies via enhanced migration and persistence. Nat Commun. 2017;8:1961.
Wang K, Chen W, Zhang Z, et al. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal Transduct Target Ther. 2020;5:283.
Huang IC, Bailey CC, Weyer JL, et al. Distinct patterns of IFITM-mediated restriction of filoviruses, SARS coronavirus, and influenza A virus. PLoS Pathog. 2011;7:e1001258.
Ying T, Li W, Dimitrov DS. Discovery of T-cell infection and apoptosis by middle east respiratory syndrome coronavirus. J Infect Dis. 2016;213:877–9.
Tan YJ, Goh PY, Fielding BC, et al. Profiles of antibody responses against severe acute respiratory syndrome coronavirus recombinant proteins and their potential use as diagnostic markers. Clin Diagn Lab Immunol. 2004;11:362–71.
Wu HS, Hsieh YC, Su IJ, et al. Early detection of antibodies against various structural proteins of the SARS-associated coronavirus in SARS patients. J Biomed Sci. 2004;11:117–26.
Meyer B, Drosten C, Muller MA. Serological assays for emerging coronaviruses: challenges and pitfalls. Virus Res. 2014;194:175–83.
Li G, Chen X, Xu A. Profile of specific antibodies to the SARS-associated coronavirus. N Engl J Med. 2003;349:508–9.
Li CK, Wu H, Yan H, et al. T cell responses to whole SARS coronavirus in humans. J Immunol. 2008;181:5490–500.
To KK, Tsang OT, Leung WS, et al. Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2: an observational cohort study. Lancet Infect Dis. 2020;20:565–74.
Guo L, Ren L, Yang S, et al. Profiling Early Humoral Response to Diagnose Novel Coronavirus Disease (COVID-19). Clin Infect Dis. 2020;71:778–85.
Tan W, Lu Y, Zhang J, et al. Viral kinetics and antibody responses in patients with COVID-19. MedRxiv 2020.
Zhang Y, Xu J, Jia R, et al. Protective humoral immunity in SARS-CoV-2 infected pediatric patients. Cell Mol Immunol. 2020;17:768–70.
Sterlin D, Mathian A, Miyara M, et al. IgA dominates the early neutralizing antibody response to SARS-CoV-2. Sci Transl Med 2021; 13.
Wang C, Li W, Drabek D, et al. A human monoclonal antibody blocking SARS-CoV-2 infection. Nat Commun. 2020;11:2251.
Kamboj M, Sepkowitz KA. Nosocomial infections in patients with cancer. Lancet Oncol. 2009;10:589–97.
Hijano DR, Maron G, Hayden RT. Respiratory viral infections in patients with cancer or undergoing hematopoietic cell transplant. Front Microbiol. 2018;9:3097.
Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335–7.
Yu J, Ouyang W, Chua MLK, Xie C. SARS-CoV-2 transmission in patients with cancer at a tertiary care hospital in Wuhan. China JAMA Oncol. 2020;6:1108–10.
Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA. 2020;323:2052–9.
Zhang L, Zhu F, Xie L, et al. Clinical characteristics of COVID-19-infected cancer patients: a retrospective case study in three hospitals within Wuhan. China Ann Oncol. 2020;31:894–901.
Fathi N, Rezaei N. Lymphopenia in COVID-19: therapeutic opportunities. Cell Biol Int. 2020;44:1792–7.
Santos RA, Simoes e Silva AC, Maric C, et al. Angiotensin-(1–7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc Natl Acad Sci U S A. 2003;100:8258–63.
Zaim S, Chong JH, Sankaranarayanan V, Harky A. COVID-19 and multiorgan response. Curr Probl Cardiol. 2020;45:100618.
Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected Pneumonia in Wuhan. China JAMA. 2020;323:1061–9.
Lozahic C, Maddock H, Sandhu H. Anti-cancer therapy leads to increased cardiovascular susceptibility to COVID-19. Front Cardiovasc Med. 2021;8:634291.
Vivarelli S, Falzone L, Grillo CM, et al. Cancer management during COVID-19 pandemic: Is immune checkpoint inhibitors-based immunotherapy harmful or beneficial? Cancers (Basel) 2020; 12.
Sullivan RJ, Johnson DB, Rini BI, et al. COVID-19 and immune checkpoint inhibitors: initial considerations. J Immunother Cancer 2020; 8.
Palaskas N, Lopez-Mattei J, Durand JB, Iliescu C, Deswal A. Immune checkpoint inhibitor myocarditis: pathophysiological characteristics, diagnosis, and treatment. J Am Heart Assoc. 2020;9:e013757.
Darvin P, Toor SM, Sasidharan Nair V, Elkord E. Immune checkpoint inhibitors: recent progress and potential biomarkers. Exp Mol Med. 2018;50:1–11.
Johnson DB, Balko JM, Compton ML, et al. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med. 2016;375:1749–55.
Gurdogan M, Yalta K. Myocarditis associated with immune checkpoint inhibitors: practical considerations in diagnosis and management. Anatol J Cardiol. 2020;24:68–75.
Sakai T, Yahagi K, Hoshino T, et al. Nivolumab-induced myocardial necrosis in a patient with lung cancer: a case report. Respir Med Case Rep. 2019;27:100839.
Farge D, Frere C, Connors JM, et al. 2019 international clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. Lancet Oncol. 2019;20:e566-81.
Aronson D, Brenner B. Arterial thrombosis and cancer. Thromb Res. 2018;164(Suppl 1):S23-8.
Danzi GB, Loffi M, Galeazzi G, Gherbesi E. Acute pulmonary embolism and COVID-19 pneumonia: A random association? Eur Heart J. 2020;41:1858.
Welt FGP, Shah PB, Aronow HD, et al. Catheterization laboratory considerations during the coronavirus (COVID-19) pandemic: from the ACC’s interventional council and SCAI. J Am Coll Cardiol. 2020;75:2372–5.
Khorana AA, Kuderer NM, Culakova E, Lyman GH, Francis CW. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902–7.
Iliescu CA, Grines CL, Herrmann J, et al. SCAI Expert consensus statement: evaluation, management, and special considerations of cardio-oncology patients in the cardiac catheterization laboratory (endorsed by the cardiological society of india, and sociedad Latino Americana de Cardiologia intervencionista). Catheter Cardiovasc Interv. 2016;87:E202-23.
Iliescu C, Balanescu DV, Donisan T, et al. Safety of diagnostic and therapeutic cardiac catheterization in cancer patients with acute coronary syndrome and chronic thrombocytopenia. Am J Cardiol. 2018;122:1465–70.
Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420–2.
Duan J, Tao J, Zhai M, et al. Anticancer drugs-related QTc prolongation, torsade de pointes and sudden death: current evidence and future research perspectives. Oncotarget. 2018;9:25738–49.
Cammann VL, Sarcon A, Ding KJ, et al. Clinical features and outcomes of patients with malignancy and Takotsubo Syndrome: observations from the international Takotsubo registry. J Am Heart Assoc. 2019;8:e010881.
Jabri A, Kalra A, Kumar A, et al. Incidence of stress cardiomyopathy during the coronavirus disease 2019 Pandemic. JAMA Netw Open. 2020;3:e2014780.
Mercuro NJ, Yen CF, Shim DJ, et al. Risk of QT interval prolongation associated with use of hydroxychloroquine with or without concomitant azithromycin among hospitalized patients testing positive for coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020;5:1036–41.
Roden DM. Drug-induced prolongation of the QT interval. N Engl J Med. 2004;350:1013–22.
Boulware DR, Pullen MF, Bangdiwala AS, et al. A randomized trial of hydroxychloroquine as postexposure prophylaxis for covid-19. N Engl J Med. 2020;383:517–25.
Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of covid-19 - final report. N Engl J Med. 2020;383:1813–26.
Dabbagh MF, Aurora L, D’Souza P, et al. Cardiac tamponade secondary to COVID-19. JACC Case Rep. 2020;2:1326–30.
El Haddad D, Iliescu C, Yusuf SW, et al. Outcomes of cancer patients undergoing percutaneous pericardiocentesis for pericardial effusion. J Am Coll Cardiol. 2015;66:1119–28.
Fotuhi M, Mian A, Meysami S, Raji CA. Neurobiology of COVID-19. J Alzheimers Dis. 2020;76:3–19.
Meinhardt J, Radke J, Dittmayer C, et al. Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat Neurosci. 2021;24:168–75.
Reiken S, Sittenfeld L, Dridi H, et al. Alzheimer's-like signaling in brains of COVID-19 patients. Alzheimers Dement 2022.
Singal CMS, Jaiswal P, Seth P. SARS-CoV-2, more than a respiratory virus: its potential role in neuropathogenesis. ACS Chem Neurosci. 2020;11:1887–99.
Martin-Jimenez P, Munoz-Garcia MI, Seoane D, et al. Cognitive impairment is a common comorbidity in deceased COVID-19 patients: a hospital-based retrospective cohort study. J Alzheimers Dis. 2020;78:1367–72.
Matias-Guiu JA, Pytel V, Matias-Guiu J. Death rate due to COVID-19 in Alzheimer’s disease and frontotemporal dementia. J Alzheimers Dis. 2020;78:537–41.
Hwang JM, Kim JH, Park JS, Chang MC, Park D. Neurological diseases as mortality predictive factors for patients with COVID-19: a retrospective cohort study. Neurol Sci. 2020;41:2317–24.
Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683–90.
Varatharaj A, Thomas N, Ellul MA, et al. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: a UK-wide surveillance study. Lancet Psychiatry. 2020;7:875–82.
Helms J, Kremer S, Merdji H, et al. Neurologic features in severe SARS-CoV-2 infection. N Engl J Med. 2020;382:2268–70.
Lukiw WJ, Pogue A, Hill JM. SARS-CoV-2 infectivity and neurological targets in the brain. Cell Mol Neurobiol. 2022;42:217–24.
Beitz JM. Parkinson’s disease: a review. Front Biosci (Schol Ed). 2014;6:65–74.
Fearon C, Fasano A. Parkinson’s disease and the COVID-19 pandemic. J Parkinsons Dis. 2021;11:431–44.
Funding
Firdos Ahmad is supported by Competitive (1901090162-P), Targeted (1801090144-P) and COVID-19 (CoV-0302) research grants from the University of Sharjah.
Author information
Authors and Affiliations
Contributions
AG contributed to writing, figures preparation, HM contributed to writing, and FA contributed to the conceptualization, writing, revising and proofreading of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gupta, A., Marzook, H. & Ahmad, F. Comorbidities and clinical complications associated with SARS-CoV-2 infection: an overview. Clin Exp Med 23, 313–331 (2023). https://doi.org/10.1007/s10238-022-00821-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-022-00821-4