Abstract
Intraguild predators can have behavioral mechanisms to maximize foraging and/or avoid predation. However, there is a lack of information about the influence of such prey-predator interactions on the daily activity of the species involved. Therefore, we investigated the daily activity of two intraguild predators, Tityus pusillus Pocock, 1893 and Ananteris mauryi Lourenço, 1982, in the presence and absence of each other. Animals were observed in three experimental conditions, containing individuals of T. pusillus (control 1), A. mauryi (control 2), and both species (treatment). In addition, we evaluated the correlation between the number of active individuals with air temperature and humidity. Our results showed that T. pusillus and A. mauryi have similar daily activity between 18:00 and 05:00 h. However, T. pusillus was more active and shifted from a sit-and-wait hunting mode to actively hunting when in the presence of A. mauryi. In contrast, under predation risk, A. mauryi did not change its level of activity but became more vigilant by reducing the frequency of rest, hydration, and mating attempts. Activity of A. mauryi was positively correlated with air humidity whereas activity of T. pusillus was negatively correlated. This work highlights the influence of intraguild predators in the behavioral decisions during daily activities of each other, indicating adaptive behaviors in both prey and predator.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prey-predator relationships have been a central issue in many behavioral studies focusing on how species may modify their behavioral decisions in order to increase their survival. Traditionally, many studies have focused on how animals can change their behavior and habitat exploitation activities to avoid predation, as well as to optimize prey encounter during foraging (e.g., Snyder and Wise 2000; Okuyama 2002; Schmidt et al. 2012, 2014; Wilkinson et al. 2015; Belgrad and Griffen 2016; Nisani et al. 2018). In many ecological systems, such interactions involve species acting as both predator and competitor for similar food resources, a phenomenon termed intraguild predation (IGP) (Holt and Polis 1997; Polis et al. 1989). The relationship between intraguild predators is particularly important because of the potential of IGP to affect community structure and diversity, regulate trophic cascades in food webs, and influence ecosystem productivity and population dynamics (Polis and McCormick 1986, 1987; Holt and Polis 1997; Kondoh 2008; Stouffer and Bascompte 2010; Ryan and Cantrell 2015). Furthermore, behavioral decisions to avoid predation between intraguild predators play a key role in the persistence of species in complex food webs (Okuyama 2002; Kondoh 2003, 2007; Urbani and Ramos-Jiliberto 2010). For instance, Okuyama (2002) found that the jumping spider Phidippus octopunctatus (Peckham & Peckham, 1883) reduced its foraging activity in the presence of its predator, Phidippus audax (Hentz, 1845), to avoid predation and consequently they reduced their predation rate on a shared prey, Drosophila melanogaster (Meigen, 1830).
As IGP generally occurs when small species are preyed upon by larger ones (Polis et al. 1981; Polis and McCormick 1987; Moreno-González and Hazzi 2012; Lira et al. 2017a, b), it is expected that the smaller species have behavioral mechanisms to coexist with their intraguild predators (Urbani and Ramos-Jiliberto 2010). For instance, smaller scorpions avoid the period of higher activity of larger scorpions (Polis and McCormick 1987) in addition to changing foraging sites to avoid predation risk (Sánchez-Piñero and Urbano-Tenorio 2016). Furthermore, the surface activity of arthropods can be influenced by microclimate variations; these animals can increase their activity at optimal values of air temperature and humidity to avoid desiccation risk (Cloudsley-Thompson 1962) or even to maximize their locomotor performance, such as other ectothermic animals (Carlson and Rowe 2009; Nime et al. 2013; Webber et al. 2015).
Thus, considering the potential of interactions between intraguild predators to influence the stability of food webs and population dynamics, we investigated the behavior of two intraguild predators in the presence of each other (a prey-predator system), the scorpions Tityus pusillus Pocock, 1893 and Ananteris mauryi Lourenço, 1982. Furthermore, we evaluated whether variation of air temperature and humidity along the nocturnal period would influence their surface activity. Tityus pusillus and A. mauryi are two sympatric scorpions co-occurring on leaf litter (Lira et al. 2018). However, A. mauryi are smaller scorpions (adult size, 13.6–24.1 mm compared with 24.1–34.9 mm of T. pusillus; Lira et al. 2018) which can be harmed in antagonistic interactions with T. pusillus (Lira et al. 2017a). Tityus pusillus is a sedentary scorpion hunting in upper layers of leaf litter and waiting for prey in ambush posture (Lira et al. 2013, 2018). Conversely, A. mauryi are more active spending time wandering and actively hunting their prey, moving around mainly through the bottom layers of leaf litter in the interstitial spaces between leaves (Lira et al. 2013, 2018). In such microhabitat, A. mauryi can find small arthropods to feed on and greater complexity of habitat that allows avoidance of predation (Schmidt and Rypstra 2010; Kalinkat et al. 2013). Therefore, we predict that A. mauryi will reduce their activity in the period of greater activity of T. pusillus. In addition, we predict that A. mauryi will change its behavioral acts to enhance the predator avoidance, whereas T. pusillus will perform more acts that optimize the predation. We think that T. pusillus will be more active at high air temperature and low humidity, whereas, in contrast, A. mauryi will be more active at low temperature and high humidity.
Methods
Sample collection and maintenance
Tityus pusillus and A. mauryi individuals were collected from Campo de Instrução Marechal Newton Cavalcante (07° 50′ 32.7″ S, 035° 06′ 33.2″ W), a semi-deciduous second-growth fragment of Atlantic Forest, northeast Brazil. Field collections were carried out by four researchers between April 2018 and June 2018 at 19:00 and 22:00 h using UV flashlights. Scorpions were identified by species, following procedure of Lourenço (2002), and separated by age (immature or adult) and sex. No juveniles or pregnant females were used in this experiment. The animals were kept individually in plastic terraria (9.5 cm of diameter and 7.5 cm of depth) containing shelter (cardboard), water supply, and fed weekly with cockroach nymphs, Nauphoeta cinerea (Olivier, 1789). Before the experiments, scorpions were kept under observation for a period of 30 days to assure that the individuals were healthy; they were then left without food for a week to stimulate foraging during the trials.
Experimental trials
Experiments were performed using rectangular arenas (47 × 31.5 × 15.5 cm) containing a 3-cm layer of sifted leaf litter (previously dried at 50 °C for 24 h) and ten leaves of Fabaceae (~ 12 cm in length) to serve as shelter. Two water-soaked cotton balls were available in the arena as an ad libitum water source. Immediately before the experiments, scorpions were marked with non-toxic paint on the dorsal surface of the mesosoma to facilitate identification during the behavioral observations. All experimental trials were conducted with arenas placed outdoors to have a gradual switch between light and dark periods.
We first determined the daily activity and behaviors of the scorpions so as to classify their behavioral acts. Tityus pusillus and A. mauryi (n = 12 per trial; sex ratio 1:1 each) were placed in different arenas and instantaneous scan samplings (Altmann 1974) were performed for 15 min every hour over a consecutive 48 h (12:12 light:dark period) with three replicates. Prior to experiments, animals were left in the arenas for 24 h to allow chemical cue dissemination, as these arachnids can be orientated by chemical stimuli (Abushama 1964; Brownell and Farley 1979; Nisani et al. 2018). Scorpions were considered active if they were not hiding below the leaves or cotton balls and could be seen by the observers performing any behavioral act except rest.
After characterization of the daily activity and scorpion behavioral acts, we evaluated the behavioral acts of the species in the absence and presence of each other. Trials consisted of conspecifics of T. pusillus (control 1), conspecifics of A. mauryi (control 2), and heterospecific pairs (treatment) to record behavioral changes under predation pressure; all trials had a sample size of 12 animals with a 1:1 sex ratio and were replicated six times. Treatment trials have 1:1 species ratio and 1:1 sex ratio and individuals of A. mauryi were initially placed for 24 h to expose their chemical cues on the substrate and then removed. After this period, T. pusillus were placed in the arena to allow for their chemical cue exposure for 24 h. Then, A. mauryi were reintroduced in the arena. Observations started 1 h after acclimation. During acclimation, the animals only search and took shelter. In these experiments, scan samplings of 15 min every hour (Altmann 1974) were performed during the dark period (18:00–5:00 h) by two experienced observers (Dionisio-da-Silva W and Lira AFA) using a red-light lamp (Machan 1968). Frequency of behavioral acts were quantified during all observation period in the three experimental conditions. Temperature and relative humidity during the active period were only recorded for conspecific trials to avoid interference of heterospecific interactions in the number of active individuals rather than response to microclimatic conditions, using a digital thermohygrometer (Incoterm–7663.02.0.00).
Data analysis
Data normality was evaluated through of Jarque-Bera tests. Welch’s t test was used to detect the effect of the heterospecific treatment on the proportion of active scorpions (T. pusillus and A. mauryi). Mann-Whitney’s tests and Welch’s t tests were performed to assess the differences in relative frequency of each behavioral act of T. pusillus and A. mauryi between treatments. Finally, Spearman’s correlations were used to evaluate the relationship between the variables (air temperature and humidity) and avoid covariance; and also, to correlate the number of active scorpions with the corresponding values of air temperature and humidity for every hour (we used the data of the control arenas to avoid effect of the scorpions’ competition). All analyses were carried out using normtest and Hmisc packages (Gavrilov and Pusev 2014; Harrell Jr. 2019) in R Studio v. 1.1.463 (RStudio Team 2019).
Results
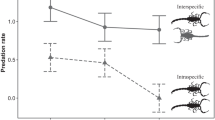
Similar daily activities and variability levels of overall activity were observed when comparing the daily activity pattern of T. pusillus and A. mauryi. Based on a 48-h observation, the active period was determined as 18:00–05:00 h where 30–50% of T. pusillus and 30–80% of A. mauryi were active per hour. Only one A. mauryi and two T. pusillus remained active up to 06:00 h and 07:00 h, respectively (Fig. 1). During the light period, all T. pusillus individuals rested below the leaves while A. mauryi rest below the wet cotton balls. All behavioral acts observed were classified into five categories, of which immobility and exploration were further subdivided into two (Table 1). Both species differed in the hunting mode, with T. pusillus mostly in the alert behavior (73.3%), while A. mauryi was in the foraging behavior (56.84%).
During the experiment period, humidity and temperature varied between 70 and 86% and 25.5 and 28.4 °C, respectively. We used only air humidity in posterior analysis because of its covariance with air temperature (Spearman rank correlation, rs = − 0.52; P = 0.0095). Nocturnal activity of both scorpion species exhibited a distinct relationship with air humidity. The number of active T. pusillus was negatively correlated with air humidity (rs = − 0.34, P = 0.0032). In contrast, A. mauryi was positively correlated with air humidity (rs = 0.33, P = 0.0047).
During the heterospecific trials, in contrast to the conspecific trials, T. pusillus was stimulated to increase their predation, and significantly increase their number of active individuals (72.7% of individuals were active; Welch’s t test, T14.84 = − 11.09; P < 0.0001) and their foraging activities on the substrate (T9.23 = − 2.37, P = 0.0498). Individuals spent 6.75 ± 2.73 h (mean ± SD) feeding on one A. mauryi. They usually eat the entire individual, excluding the pincers and telson. In contrast, the number of active A. mauryi was similar on the presence or absence of T. pusillus (Welch’s t test, T20.32 = − 1.37; P = 0.1862). However, A. mauryi modified their behavior in the presence of the predator, reducing rest (T8.06 = 3.44, P = 0.0086), hydration (Wilcoxon-Mann-Whitney test, W = 36; P = 0.0036) and mating attempts (W = 30, P = 0.0284; Table 1).
When the predators (T. pusillus) contacted the prey (A. mauryi) the latter immediately tried to walk away to avoid predation. Despite this, 55.56% of the A. mauryi individuals were captured by T. pusillus. Of those captured, 4.71% used metasoma autotomy by shedding their “tail” to escape from predation.
Discussion
Our results showed that, despite T. pusillus and A. mauryi scorpions cohabiting in the leaf litter, they clearly differ in their habits, in their relationship with air humidity, and in the frequency of behavioral acts when in the presence of heterospecifics. Overall, T. pusillus spent more time in the alert behavior with a sit-and-wait posture. However, in response to the presence of A. mauryi, this species increased their surface activity and the frequency of foraging (actively hunting). In contrast, when perceiving the presence of its predator, A. mauryi did not change their surface activity but changed behavioral acts that would reduce their predation risk; for example, they became more vigilant by reducing rest, hydration, and mating attempts.
In the experimental trials, the conspecific and heterospecific arenas had equal density of scorpions, which are visually weak animals (Hjelle 1990). Therefore, differences in the behavior are likely to result from response to chemical cues. Arthropods, including some scorpions, can use airborne and substrate chemical cues to detect conspecifics, competitors, and prey to optimize foraging or avoid predation and competition (e.g., Miller and Formanowicz 2011; Hettyey et al. 2015; Binz et al. 2016; Nisani et al. 2018; Pears et al. 2018). In the presence of A. mauryi, T. pusillus were much more active and increased the frequency of foraging (active hunting) rather than the alert behavior (sit-and-wait posture). The sit-and-wait hunting mode is efficient when prey are scarce because it has a low energy cost and increases the encounter rate when the predator cannot move faster than prey. However, when prey are abundant, have a clumped distribution, or there is interspecific competition, it is more efficient for the predator to shift to actively hunting (Huey and Pianka 1981; Fausch et al. 1997; Fulton and Bellwood 2002; Scharf et al. 2006). Furthermore, Santos et al. (2018) showed that A. mauryi populations have a clumped distribution. Therefore, T. pusillus could have increased their activity and foraging to increase the efficiency in predation of A. mauryi.
Because sit-and-wait predators are sedentary, they have a narrow extent of movement and their chemical cues on the substrate indicate a strong predation risk for their prey, which can then alter the level of activity or microhabitat use to avoid predation (Persons et al. 2001; Preisser et al. 2007; Schmitz 2008). However, A. mauryi did not change their level of activity in the presence or absence of T. pusillus, suggesting a distinct strategy of this species. Ananteris mauryi avoided behavioral acts that would increase their predation risk such as rest, hydration, and mating attempts. However, it is not clear whether A. mauryi can perceive T. pusillus presence through its airborne and substrate chemical cues or just touching him with pedipalps, which have resulted in fast avoidance of A. mauryi in our observations.
Interestingly, A. mauryi was more vigilant as indicated by decreasing rest but did not reduce foraging under predation risk by T. pusillus. To avoid predation in environments with high risk, animals can stay more vigilant by reducing their rest rather than their foraging (Sharpe and Van Horne 1998; Tchabovsky et al. 2001). In a motionless state, A. mauryi would be more vulnerable to capture by T. pusillus that forage more to find it. However, rest deprivation can be harmful to scorpions because it results in decreased alert behaviors and increased rest behaviors as a compensation, on the subsequent night (Tobler and Stalder 1988). Furthermore, A. mauryi also reduced hydration behaviors in the presence of the predator. Although scorpions withstand a greater dehydration level (ca. 40% of their body mass) when compared with other arthropods (Cloudsley-Thompson 1962; Hadley 1974; Riddle et al. 1976), Ananteris are humiculous scorpions that have preference for moist environments and are more vulnerable to desiccation risk than other species (Lourenço 2015; Dionisio-da-Silva et al. 2018). Because hydration and rest behaviors are performed when the animal is immobile and vulnerable to attacks from enemies, the avoidance of these behaviors can reduce the predation risk but will increase desiccation risk.
Although it has potential to cause chronic physiological stress as well as known for other arthropods (e.g., Persons et al. 2002; Stoks et al. 2006; Dievel et al. 2016), for instance, Persons et al. (2002) observed that wolf spider Pardosa milvina (Hentz, 1844) had a negative physiological effect of being under constant predation risk. These spiders when exposed to chemical cues of their predator, Hogna helluo (Walckenaer, 1837), ate fewer prey to avoid predation risk, and, consequently, lost weight more quickly and produced lighter egg sacs with fewer eggs. Yet, as predator avoidance, the P. milvina climbed the walls of the experimental containers significantly more when silk and excreta of H. helluo were on the substrate. Climbing behavior is often reported to avoid predation by ground-dwelling arthropods (Brown and O’Connell 2000; Persons et al. 2002; Sánchez-Piñero and Urbano-Tenorio 2016). In contrast, A. mauryi does not have difference in the frequency of climbing in the presence of T. pusillus. Ananteris mauryi are more active in the bottom layers of the leaf litter, where there is greater complexity of habitat that improves predator avoidance, whereas T. pusillus are more active in the upper layers (Schmidt and Rypstra 2010; Kalinkat et al. 2013; Lira et al. 2013, 2018). Therefore, when A. mauryi were under predation risk of T. pusillus, they may have two escape responses: (1) avoid climbing as a natural avoidance of the upper layers of the leaf litter that are inhabited by T. pusillus and other large predators, or (2) climb the vegetation to avoid predation such as other arthropods do.
The presence of T. pusillus was also determinant for the decline of mating attempts of A. mauryi. Mating is dangerous to perform under predation risk because the courtship of scorpions may last a long time (several minutes) (Stockmann 2015; Olivero et al. 2017). Although information about courtship of Ananteris is scarce (Mattoni et al. 2015), Dionisio-da-Silva et al. (2018) found that A. mauryi presents a non-overlapping reproductive period with T. pusillus, reproducing in the dry season, whereas A. mauryi reproduces in the rainy season. These authors speculate that such strategy may be a combination of predation avoidance by A. mauryi and the distinct moisture requirements of these species (A. mauryi: high humidity; T. pusillus: low humidity). Our findings support this assumption as A. mauryi reduced its mating behavior in the presence of T. pusillus and they also have a greater number of active individuals during high humidity, contrary to T. pusillus.
Small invertebrates avoid temperature and humidity conditions that cause water loss because desiccation is a major physiological risk for them (Cloudsley-Thompson 1962). Furthermore, the resistance to desiccation depends on their abundance of cuticular lipids and body surface area in proportion to their mass (Cloudsley-Thompson 1962; Toolson and Hadley 1977; Polis 1990; Lourenço 2012). Thus, small-sized animals are more susceptible to water loss, such as A. mauryi that have preference for moist environments and inhabits the humid bottom layers of the leaf litter (Cloudsley-Thompson 1962; Sampaio et al. 1993; Lourenço 2012; Lira et al. 2018). Therefore, under high air humidity, A. mauryi increased their activity in order to avoid water loss. Moreover, they also had a preference for resting in humid places (under the wet cotton) during the period of inactivity (light period). In contrast, for T. pusillus, there were a greater number of active scorpions when air humidity was low what suggests that they are more resistant to water loss than A. mauryi. It was probable that T. pusillus used the air humidity as an indicator of ecological condition rather than a physiological requirement. Tityus pusillus could avoid higher relative humidity to avoid surface activity during rainfall periods. Strong negative effects of diurnal rainfall were observed on the surface activity of the wandering spider Phoneutria reidyi (FO Pickard-Cambridge, 1897) (Queiroz and Gasnier 2017). Because the rainfall washes chemical cues of prey and predators, it makes difficult to perceive prey and risk of predation. Therefore, decreased activity in a humid environment would be beneficial to optimize foraging (Queiroz and Gasnier 2017).
Overall, our predictions were partially corroborated. Tityus pusillus and A. mauryi had a similar period of activity during the night; however, T. pusillus were more active in periods of low air humidity, whereas A. mauryi, which are more vulnerable to desiccation, have a preference for high humidity. Furthermore, both species had adaptive behavior in the presence of each other, but just T. pusillus increase the number of active individuals. Tityus pusillus optimize their foraging by increasing their activity and shifting from the sit-and-wait hunting mode to actively hunting. While A. mauryi did not change their activity level, they showed a strong antipredator behavior due to their several changes in their behavioral decisions to enhance their survival in the presence of a predator.
This work highlights the influence of an intraguild predator in the behavioral decisions during daily activity of an intraguild prey as well as the inverse. Yet, it elucidates how combination of response to microclimatic variation and adaptive behavior between prey and predator could be associated with non-overlapping reproductive periods and spatial partitioning of these animals recorded in previous studies.
References
Abushama FT (1964) On the behaviour and sensory physiology of the scorpion Leiurus quinquestriatus (H. & E.). Anim Behav 12(1):140–153
Altmann J (1974) Observational study of behavior: sampling methods. Behaviour 49(3):227–266
Belgrad BA, Griffen BD (2016) Predator–prey interactions mediated by prey personality and predator hunting mode. Proc R Soc B 283:20160408
Binz H, Kraft EF, Entling MH, Menzel F (2016) Behavioral response of a generalist predator to chemotactile cues of two taxonomically distinct prey species. Chemoecology 26(4):153–162
Brown CA, O’Connell DJ (2000) Plant climbing behavior in the scorpion Centruroides vittatus. Am Midl Nat 144(2):406–418
Brownell PH, Farley RD (1979) Prey localization behaviour of the nocturnal scorpion, Paruroctonus mesaensis: orientation to substrate vibrations. Anim Behav 27:185–193
Carlson BE, Rowe MP (2009) Temperature and desiccation effects on the antipredator behavior of Centruroides vittatus (Scorpiones: Buthidae). J Arachnol 37(3):321–330
Cloudsley-Thompson JL (1962) Microclimates and the distribution of terrestrial arthropods. Annu Rev Entomol 7(1):199–222
Dievel M, Janssens L, Stoks R (2016) Short-and long-term behavioural, physiological and stoichiometric responses to predation risk indicate chronic stress and compensatory mechanisms. Oecologia 181(2):347–357
Dionisio-da-Silva W, Lira AFA, Albuquerque CMR (2018) Distinct edge effects and reproductive periods of sympatric litter-dwelling scorpions (Arachnida: Scorpiones) in a Brazilian Atlantic forest. Zoology 129:17–24
Fausch KD, Nakano S, Kitano S (1997) Experimentally induced foraging mode shift by sympatric charrs in a Japanese mountain stream. Behav Ecol 8(4):414–420
Fulton CJ, Bellwood DR (2002) Patterns of foraging in labrid fishes. Mar Ecol Prog Ser 226:135–142
Gavrilov I, Pusev R (2014) normtest: tests for normality. R package version 1.1. Available from: <https://CRAN.R-project.org/package=normtest>. Accessed 01 Feb 2019
Hadley NF (1974) Adaptational biology of desert scorpions. J Arachnol 2(1):11–23
Harrell Jr. FE (2019) Hmisc: Harrell Miscellaneous. R package version 4.2-0. Available from: https://CRAN.R-project.org/package=Hmisc. Accessed: 02 April 2019
Hettyey A, Tóth Z, Thonhauser KE, Frommen JG, Penn DJ, Van Buskirk J (2015) The relative importance of prey-borne and predator-borne chemical cues for inducible antipredator responses in tadpoles. Oecologia 179(3):699–710
Hjelle TJ (1990) Anatomy and morphology. In: Polis GA (ed) The biology of scorpions, pp. 5-30
Holt RD, Polis GA (1997) A theoretical framework for intraguild predation. Am Nat 149(4):745–764
Huey RB, Pianka ER (1981) Ecological consequences of foraging mode. Ecology 62(4):991–999
Kalinkat G, Brose U, Rall BC (2013) Habitat structure alters top-down control in litter communities. Oecologia 172(3):877–887
Kondoh M (2003) Foraging adaptation and the relationship between food-web complexity and stability. Science 299(5611):1388–1391
Kondoh M (2007) Anti-predator defence and the complexity–stability relationship of food webs. P Roy Soc Lond B Bio 274(1618):1617–1624
Kondoh M (2008) Building trophic modules into a persistent food web. Proc Natl Acad Sci USA 105(43):16631–16635
Lira AFA, DeSouza AM, Silva Filho AAC, Albuquerque CMR (2013) Spatio-temporal microhabitat use by two co-occurring species of scorpions in Atlantic rainforest in Brazil. Zoology 116(3):182–185
Lira AFA, DeSouza AM, Albuquerque CMR (2017a) Report of intraguild predation and cannibalism in scorpions (Scorpiones: Buthidae) in the Brazilian Atlantic forest. North-West J Zool e167302
Lira AFA, Pordeus LM, Albuquerque CMR (2017b) A new species of Ananteris (Scorpiones: Buthidae) from Caatinga biome, Brazil. Acta Arachnol 66(1):9–15
Lira AFA, DeSouza AM, Albuquerque CMR (2018) Environmental variation and seasonal changes as determinants of the spatial distribution of scorpion (Arachnida: Scorpiones) in Neotropical forests. Can J Zool 96(9):963–972
Lourenço WR (2002) Scorpions of Brazil. Les editions de l’If, Paris
Lourenço WR (2012) Humiculous scorpions: on the genera Ananteris Thorell, 1891 and Microananteris Lourenço, 2004 (Scorpiones: Buthidae), with the description of a new species from French Guiana. C R Biol 335(8):555–561
Lourenço WR (2015) Comments on the Ananterinae Pocock, 1900 (Scorpiones: Buthidae) and description of a new remarkable species of Ananteris from Peru. C R Biol 338(2):134–139
Machan L (1968) Spectral sensitivity of scorpion eyes and the possible role of shielding pigment effect. J Exp Biol 49(1):95–105
Mattoni CI, García-Hernández S, Botero-Trujillo R, Ochoa JA, Ojanguren-Affilastro AA, Pinto-da-Rocha R, Prendini L (2015) Scorpion sheds ‘tail’ to escape: consequences and implications of autotomy in scorpions (Buthidae: Ananteris). PLoS One 10(1):e0116639
Miller AL, Formanowicz DR (2011) Friend or foe: behavioral responses to conspecifics in the northern scorpion, Paruroctonus boreus (Scorpionida: Vaejovidae). J Ethol 29(2):251–256
Moreno-González JA, Hazzi NA (2012) Intraguild predation case: Tityus forcipula Gervais, 1843 (Scorpiones, Buthidae) feeding on Chactas vanbenedeni Gervais, 1843 (Scorpiones, Chactidae) in Colombia. Rev Iber Aracnol 20:117–120
Nime MF, Casanoves F, Vrech DE, Mattoni CI (2013) Relationship between environmental variables and surface activity of scorpions in the Arid Chaco ecoregion of Argentina. Invertebr Biol 132(2):145–155
Nisani Z, Honaker A, Jenne V, Loya F, Moon H (2018) Evidence of airborne chemoreception in the scorpion Paruroctonus marksi (Scorpiones: Vaejovidae). J Arachnol 46(1):40–44
Okuyama T (2002) The role of antipredator behavior in an experimental community of jumping spiders with intraguild predation. Popul Ecol 44(2):121–125
Olivero PA, Mattoni CI, Peretti AV (2017) Differences in mating behavior between two allopatric populations of a Neotropical scorpion. Zoology 123:71–78
Pears JB, Emberts Z, Bateman PW (2018) The scent of danger: the impact of predator chemical cues on emergence from refuge and willingness to autotomize limbs in the house cricket (Acheta domesticus). J Insect Behav 31:416–426
Persons MH, Walker SE, Rypstra AL, Marshall SD (2001) Wolf spider predator avoidance tactics and survival in the presence of diet-associated predator cues (Araneae: Lycosidae). Anim Behav 61(1):43–51
Persons MH, Walker SE, Rypstra AL (2002) Fitness costs and benefits of antipredator behavior mediated by chemotactile cues in the wolf spider Pardosa milvina (Araneae: Lycosidae). Behav Ecol 13(3):386–392
Polis GA, McCormick SJ (1986) Patterns of resource use and age structure among species of desert scorpion. J Anim Ecol 55:59–73
Polis GA, McCormick SJ (1987) Intraguild predation and competition among desert scorpions. Ecology 68(2):332–343
Polis GA, Sissom WD, McCormick SJ (1981) Predators of scorpions: field data and a review. J Arid Environ 4:309–326
Polis GA, Myers CA, Holt RD (1989) The ecology and evolution of intraguild predation: potential competitors that eat each other. Annu Rev Ecol Syst 20(1):297–330
Preisser EL, Orrock JL, Schmitz OJ (2007) Predator hunting mode and habitat domain alter nonconsumptive effects in predator–prey interactions. Ecology 88(11):2744–2751
Queiroz M, Gasnier TR (2017) Strong negative effect of diurnal rainfall on nocturnal activity of a wandering spider in Central Amazonia. Rev Biol Trop 65(3):1152–1160
Riddle WA, Crawford CS, Zeitone AM (1976) Patterns of hemolymph osmoregulation in three desert arthropods. J Comp Physiol 112(3):295–305
RStudio Team (2019) RStudio: integrated development for R. RStudio, Inc., Boston. Available from: <http://www.rstudio.com/>. Accessed: 01 Feb 2019
Ryan D, Cantrell RS (2015) Avoidance behavior in intraguild predation communities: a cross-diffusion model. Discrete Cont Dyn S 35:1641–1663
Sampaio EVDSB, Dall’Olio A, Nunes KS, Lemos EEP (1993) A model of litterfall, litter layer losses and mass transfer in a humid tropical forest at Pernambuco, Brazil. J Trop Ecol 9(3):291–301
Sánchez-Piñero F, Urbano-Tenorio F (2016) Watch out for your neighbor: climbing onto shrubs is related to risk of cannibalism in the scorpion Buthus cf. occitanus. PloS One 11(9):e0161747
Santos GCSG, Dionisio-da-Silva W, Souza-Alves JP, Albuquerque CMR, Lira AFA (2018) Random or clumped: how litter dwelling scorpions are distributed in a fragment of Brazilian Atlantic forest. Eur J Entomol 115:445–449
Scharf I, Nulman E, Ovadia O, Bouskila A (2006) Efficiency evaluation of two competing foraging modes under different conditions. Am Nat 168(3):350–357
Schmidt JM, Rypstra AL (2010) Opportunistic predator prefers habitat complexity that exposes prey while reducing cannibalism and intraguild encounters. Oecologia 164(4):899–910
Schmidt JM, Sebastian P, Wilder SM, Rypstra AL (2012) The nutritional content of prey affects the foraging of a generalist arthropod predator. PLoS One 7(11):e49223
Schmidt JM, Crist TO, Wrinn K, Rypstra AL (2014) Predator interference alters foraging behavior of a generalist predatory arthropod. Oecologia 175(2):501–508
Schmitz OJ (2008) Effects of predator hunting mode on grassland ecosystem function. Science 319(5865):952–954
Sharpe PB, Van Horne B (1998) Influence of habitat on behavior of Townsend’s ground squirrels (Spermophilus townsendii). J Mammal 79(3):906–918
Snyder WE, Wise DH (2000) Antipredator behavior of spotted cucumber beetles (Coleoptera: Chrysomelidae) in response to predators that pose varying risks. Environ Entomol 29(1):35–42
Stockmann R (2015) Introduction to scorpion biology and ecology. In: Gopalakrishnakone P (ed), Scorpion Venoms, Springer Netherlands, pp.25–59
Stoks R, Block MD, Slos S, Doorslaer WV, Rolff J (2006) Time constraints mediate predator-induced plasticity in immune function, condition, and life history. Ecology 87(4):809–815
Stouffer DB, Bascompte J (2010) Understanding food-web persistence from local to global scales. Ecol Lett 13(2):154–161
Tchabovsky AV, Krasnov B, Khokhlova IS, Shenbrot GI (2001) The effect of vegetation cover on vigilance and foraging tactics in the fat sand rat Psammomys obesus. J Ethol 19(2):105–113
Tobler I, Stalder J (1988) Rest in the scorpion—a sleep-like state? J Comp Physiol A 163(2):227–235
Toolson EC, Hadley NF (1977) Cuticular permeability and epicuticular lipid composition in two Arizona vejovid scorpions. Physiol Zool 50(4):323–330
Urbani P, Ramos-Jiliberto R (2010) Adaptive prey behavior and the dynamics of intraguild predation systems. Ecol Model 221(22):2628–2633
Webber MM, Gibbs AG, Rodríguez-Robles JA (2015) Hot and not-so-hot females: reproductive state and thermal preferences of female Arizona Bark Scorpions (Centruroides sculpturatus). J Evol Biol 28(2):368–375
Wilkinson EB, Grabowski JH, Sherwood GD, Yund PO (2015) Influence of predator identity on the strength of predator avoidance responses in lobsters. J Exp Mar Biol Ecol 465:107–112
Acknowledgments
We are also very grateful to the military command of the CIMNC for permission to use the area for our study and to Divisão de Transportes da Universidade Federal de Pernambuco for all technical support.
Funding
This work was supported by the Fundação de Amparo à Ciência e Tecnologia de Pernambuco (FACEPE) (grant number: APQ-0.437-2.04/15) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for a financial support to CMR Albuquerque (grant number: 307759/2015-6). We are grateful to Fundação de Apoio à Pesquisa do Estado da Paraíba (FAPESQ) for granting a doctoral scholarship to W. Dionisio-da-Silva (grant number: 519/18) and to Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE) for postdoctoral scholarship (BFP-0010-2.05/19) to A.F.A. Lira.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article did not contain any studies with human participants and also did not involve endangered or protected species. It was approved by Sistema de Autorização e Informação em Biodiversidade/Instituto Chico Mendes de Conservação da Biodiversidade (SISBIO/ICMBIO no. 36336-1) and complied with Brazilian law. All procedures of this work were determined to assure the animals’ welfare. Animal collection and handling was conducted with the use of tweezers without harming the animals. The experiments were conducted with only healthy animals. Water supply and shelters were made available for the animals during the experimental trials, and food was also made available during their rearing. Application of non-toxic paint markings on the individuals during the experiments is a minimally invasive technique. The animals were returned to the wild after the study.
Informed consent
This article did not contain any studies with human participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dionisio-da-Silva, W., de Araujo Lira, A.F. & de Albuquerque, C.M.R. Prey-predator interactions between two intraguild predators modulate their behavioral decisions. acta ethol 22, 195–201 (2019). https://doi.org/10.1007/s10211-019-00326-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10211-019-00326-6