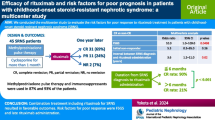
Abstract
Background
Rituximab is widely used in patients with steroid-dependent nephrotic syndrome. However, information on the effect of long-term rituximab treatment is limited. This study examined the efficacy of rituximab during and after treatment in adult patients with steroid-dependent nephrotic syndrome.
Methods
This retrospective cohort study included 30 patients with steroid-dependent nephrotic syndrome. Patients received regular single-dose rituximab (500 mg) intravenously every 6 months. Discontinuation of rituximab was considered after four to six doses if there was no recurrence of nephrotic syndrome. Glucocorticoid discontinuation with remission, first relapse after rituximab initiation, and relapse after regular rituximab treatment discontinuation were evaluated.
Results
The median age was 38 (range 18–67) years. Of 30 patients, 13 and 17 were men and women, respectively. Prior to rituximab treatment, the median number of nephrotic syndrome relapses in the patients was 5 (range 2– > 20). The 1 year discontinuation rate of glucocorticoids with remission was 83%. All patients discontinued glucocorticoid treatment at least once until 3 years and 7 months. The 1 and 2 year relapse rates after initiation of rituximab treatment were 0% and 3%, respectively. 25 patients discontinued regular rituximab treatment after a median number of six (4–12) doses. Six patients relapsed after discontinuing rituximab, and the 1 and 2 year relapse rates after the last regular rituximab treatment were 9% and 25%, respectively.
Conclusion
All patients with steroid-dependent nephrotic syndrome who received rituximab could discontinue glucocorticoid treatment with remission, and three-fourths of the patients remained in remission for > 2 years after discontinuing rituximab treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nephrotic syndrome (NS) is characterized by large amounts of proteinuria and hypoproteinaemia due to increased protein permeability caused by damage to the renal glomerular basement membrane. Primary NS has no evident causative disease. This condition comprises three main disorders, which are as follows: minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS), and membranous nephropathy [1]. Patients with Minimal change nephrotic syndrome (MCNS) and immune-mediated FSGS, which are disorders of the podocytes, commonly respond to immunosuppressive treatment with glucocorticoids (GCs). Moreover, they go into remission [2]. However, if the GC dose is tapered or GC treatment is discontinued, more than half of patients with steroid-sensitive NS can relapse, resulting in frequently relapsing NS (FRNS) or steroid-dependent NS (SDNS) [3].
Rituximab (RTX) is a chimeric IgG1 monoclonal antibody that specifically targets CD20, a surface antigen expressed in B cells, and induces B-cell apoptosis, complement-mediated lysis, and antibody-dependent cytotoxicity [4]. Via these mechanisms, RTX suppresses humoral immunity, and it is effective against several autoimmune diseases, including FRNS and SDNS. A multicentre randomized controlled trial was conducted on patients with childhood-onset FRNS and SDNS. Results showed that RTX could lead to the discontinuation of GC treatment and could significantly prolong the relapse-free period [5]. In adult patients with FRNS and SDNS, regular single-dose RTX treatment every 6 months suppresses NS relapse and reduces GC doses [6].
However, the duration of regular RTX treatment in patients with long-term NS remission and the frequency of NS relapse after discontinuing regular RTX treatment are unknown. In addition, there is limited information on the long-term efficacy and safety of RTX in adult patients with primary NS [7, 8].
This study aimed to determine the efficacy of regular RTX treatment during and after therapy in adult patients with SDNS including FRNS [9] based on GC discontinuation with remission, first relapse after rituximab initiation, and relapse after regular rituximab treatment discontinuation.
Materials and methods
Study design and participants
This retrospective cohort study was conducted at Iwate Prefectural Central Hospital, an acute care general hospital in Japan. Patients with primary NS who started regular RTX treatment at our department between December 2014 and March 2021 were screened. The inclusion criteria were as follows: (1) patients aged 18 years or older at RTX initiation, (2) those who achieved NS remission with immunosuppressive treatment including GC, and (3) those receiving GC due to relapse at least twice after the GC dose was tapered or GC treatment was discontinued. The exclusion criteria were as follows: (1) patients with idiopathic membranous nephropathy and (2) those who were monitored for < 12 months. Patients were followed-up until November 2023.
Treatment protocol
In principle, patients received regular single-dose RTX treatment (500 mg) intravenously every 6 months. All patients were treated with methylprednisolone at a dose of 125 mg and chlorpheniramine maleate at a dose of 5 mg intravenously before RTX infusion to prevent infusion reactions. Some patients received oral acetaminophen at a dose of 500 mg. Based on the discretion of the attending physician, regular RTX was discontinued after four to six doses if there was no NS recurrence.
GC treatment was discontinued at approximately 6 months and immunosuppressive agents at approximately 12 months, based on a report of adult patients with MCD from Tokyo Women’s Medical University [6]. The treatments for NS were as follows: GC, immunosuppressive agents, and intravenous immunoglobulin. Data on the date of treatment initiation, drug dose and frequency, and total number of regular RTX treatments were recorded.
Clinical parameters upon RTX initiation
Data on the characteristics of the patients and laboratory parameters were extracted from the electronic medical records. These included age, sex, height, weight, renal biopsy findings; total number of relapses prior to RTX; serum creatinine, hemoglobin, serum albumin, serum IgG, and C-reactive protein levels; urinalysis findings; and urinary protein levels.
Primary and secondary endpoints
The primary endpoints were defined as GC discontinuation with NS remission and first relapse after RTX initiation. The secondary endpoints were defined as the number of relapses within 24 months before and after RTX initiation and relapse after regular RTX discontinuation. Remission and relapse of NS were defined as a urinary protein level of ≤ 0.3 and ≥ 1.0 g/gCr, respectively [9]. NS relapse within 6 months after the last dose of RTX was considered to have occurred during regular RTX treatment.
Statistical analysis
The clinical parameters were presented as median (range, min–max) or percentage, as appropriate. Survival time analysis was performed using the Kaplan–Meier survival curves and the Cox proportional hazards model. The number of relapses within 24 months before and after RTX initiation was compared using the paired t-test. Coefficients were calculated using Pearson correlation methods. Statistical analyses were performed using STATA version 17.0 (Stata Corp., College Station, TX). R > 0.20 and P-value < 0.05 were considered statistically significant.
Results
Characteristics of the patients upon RTX initiation
This study included 30 patients of 41 patients with primary NS who were initially treated with regular RTX treatment at our department, 2 patients with idiopathic membranous nephropathy, 3 with an observation period of < 12 months, 4 aged < 18 years, and 2 who only had one relapse were excluded. The median age at RTX treatment initiation was 38 (range 18–67) years. In total, 13 and 17 patients were men and women, respectively (Table 1). The median age at NS onset was 27 (2–64) years. The median period from NS onset to RTX initiation was 112 (10–507) months. The median number of NS relapses prior to RTX was 5 (2– > 20). Of 30 patients, 27 (90%) underwent renal biopsy, and 24 (80%) had MCD. Three (10%) patients presented with FSGS. All patients were steroid-dependent, and they met the SDNS criteria. Five (17%) patients also met the FRNS criteria because they had two or more relapses within 6 months prior to RTX treatment [9]. The median serum creatinine level upon RTX initiation was 0.62 (0.46–1.75) mg/dL, and the median hemoglobin level was 13.8 (11.0–16.9) g/dL. The median serum albumin level was 3.9 (1.4–5.0) g/dL. The median urinary protein-to-creatinine ratio was 0.21 (0.00–9.00) g/gCr, and only four (13%) patients had a ratio of > 3.5 g/gCr. RTX treatment had been initiated in most patients in or near NS remission, and all patients had achieved NS remission until 3 months after initiation of RTX treatment.
Treatment before, upon, and after RTX initiation
All patients received oral prednisolone prior to RTX treatment. Ten (33%) patients received prednisolone alone, and 19 (63%) patients received prednisolone with cyclosporine. In addition, one (3%) patient received prednisolone with mizoribine. The median oral prednisolone dosage was 18.75 (range 5–60) mg/day upon RTX initiation (Table 1). Of 30 patients, in 25 (83%) patients who experienced long-term remission of NS without GC or immunosuppressants, regular RTX treatment was discontinued after a median of six (4–12) doses (Table 2). Patients with a higher number of NS relapses prior to RTX initiation received more RTX doses before discontinuation, which showed a positive correlation (R = 0.703). In remaining five patients, prognosis was unknown due to relocation in two patients; death in one patient; regular RTX treatment was interrupted due to infusion reaction and delayed urticaria in one patient; and regular RTX treatment (a total of nine doses) has been continuing in the remaining patient. The median time from the initiation to the last regular RTX treatment was 886 days. One-fourth of the patients discontinued RTX within 2 years (four doses) and two-thirds within 3 years (up to six doses) (Fig. 1a). The median follow-up of 30 patients after RTX initiation was 51 (18–105) months.
a Discontinuation rate of regular rituximab The median time from initiation to the last regular rituximab treatment was 886 days. Two-thirds of patients discontinued regular rituximab treatment within 3 years (up to six doses) under long-term remission after glucocorticoid discontinuation. b The discontinuation rate of glucocorticoids with remission of nephrotic syndrome. The 6-month and 1-year discontinuation rates were 30% and 83%, respectively. All patients achieved glucocorticoid discontinuation at least once until 3 years and 7 months
GC discontinuation with NS remission
Based on the survival time analysis with GC discontinuation with NS remission as the endpoint, the 6-month and 1-year achievement rates were 30% and 83%, respectively (Fig. 1b). The median time to achievement was 216 days. At 3 years and 7 months, all patients achieved GC discontinuation with NS remission at least once.
First relapse after RTX initiation
Seven (23%) patients experienced NS recurrence after RTX initiation. However, the sole recurrence during regular RTX treatment occurred in a teenage girl who relapsed 13 months after RTX initiation. Notably, her third RTX dose at 10 months was interrupted midway due to an injection reaction. The 1 and 2 year relapse rates were 0% and 3%, respectively. The relapse rate increased starting on the third year, coinciding with gradually discontinuation of RTX treatment, and the 5 year relapse rate reached 35% (Fig. 2a). Despite using the Cox proportional hazards model, no association was found between NS relapse and the characteristics of patients such as age, sex, number of previous relapses, and RTX doses.
a Relapse-free survival rate after rituximab treatment. The 1-year and 2-year relapse rates were 0% and 3%, respectively. The relapse rate increased starting on the third year when rituximab treatment was gradually discontinued. The 5-year relapse rate was 35%. b Relapse-free survival rate after the last regular rituximab treatment. Of 25 patients who discontinued regular rituximab treatment, 6 relapsed, with the shortest relapse occurring at 7 months. The 1 and 2 year relapse rates were 9% and 25%, respectively
Number of relapses within 24 months before and after RTX initiation in 29 patients who were monitored
The mean number of relapses was 1.48 with a median of 1 (0–3) within 24 months before RTX treatment. Then, it decreased to 0.03 with a median of 0 (0–1) within 24 months after RTX treatment (P < 0.01, paired t-test). The total number of relapses in 29 patients decreased from 43 to 1.
Relapse after the discontinuation of regular RTX treatment
Of 25 patients who discontinued regular RTX treatment because of long-term remission of NS, 6 (24%) patients relapsed, with the shortest relapse occurring at 7 months after the last regular RTX treatment. The 1 and 2 year relapse rates after the last regular RTX treatment were 9% and 25%, respectively (Fig. 2b). Six patients who relapsed after regular RTX treatment discontinuation achieved remission after resuming regular RTX treatment.
Changes in prednisolone and immunosuppressants after RTX initiation
In all patients, prednisolone was rapidly tapered, and immunosuppressants were gradually discontinued after the initiation of RTX (Fig. 3 and Supplementary Table). While prednisolone was discontinued within 12 months in 25 patients (83%), 4 patients (13%) receiving prednisolone at 24 months. One patient restarted prednisolone (PSL) after experiencing a relapse at 13 months. The patient, who eventually discontinued PSL at 3 years and 7 months, had difficulty discontinuing prednisolone due to steroid withdrawal symptoms, including general malaise and polyarthralgia.
Serum IgG levels and B-cell counts
Serum IgG levels were measured in 17 patients (57%) at RTX initiation and in approximately one-third from 6 to 24 months. Only one patient received intravenous immunoglobulin immediately after RTX initiation. B-cell counts were repeatedly measured in the first seven patients, but not in subsequent patients.
Adverse events
Several patients developed infusion reactions particularly pharyngeal and laryngeal discomfort. In all but one patient who had fever and arthralgia during the third dose of RTX, treatment with RTX was continued by slowing down infusion rate of RTX and adding 125 mg of methylprednisolone and 5 mg of chlorpheniramine. Oral acetaminophen before RTX administration was effective in reducing these symptoms. Two patients developed delayed urticaria after the second dose. In one case, the same patient interrupted RTX treatment because of an infusion reaction. The subsequent regular RTX administration was also discontinued. Another patient continued RTX treatment along with an oral compounding agent (betamethasone and d-chlorpheniramine maleate) for 2 weeks after RTX infusion. A male patient in his 40 s committed suicide 18 months after starting RTX, which was not directly related to the treatment. Nevertheless, serious adverse events associated with RTX, including severe infections, were not observed.
Discussion
In this study, all patients with SDNS achieved GC discontinuation with NS remission at least once after receiving regular single-dose RTX treatment. Further, remission was maintained during regular RTX in all patients except one teenage girl who had interrupted RTX treatment at the third dose due to an infusion reaction. NS relapse occurred gradually after the discontinuation of regular RTX treatment, with a 5-year relapse rate of 35%. Meanwhile, three-fourths of patients remained in remission for > 2 years after their last regular dose of rituximab, without any treatment for NS. Regarding safety, one patient discontinued rituximab treatment due to an infusion reaction. However, serious adverse events associated with RTX, including severe infections, were not observed. The safety of RTX in adult patients with SDNS was higher than originally expected based on one report in pediatric patients [5].
In this study, patient characteristics, such as age, primary NS disease, and number of relapses prior to RTX did not significantly differ from those in previous studies using RTX in patients with FRNS or SNDS [7, 8]. Age was not associated with the efficacy and safety of RTX in this study and RTX could be used in elderly patients with SDNS, as in the previous studies, although this study included only one patient over the age of 65 years [7]. All patients in this study received prednisolone upon RTX initiation. Cyclosporine, which is commonly used in MCD and immune-mediated FSGS, was administered to more than half of the patients. RTX was initiated patients in or near remission in this study and several previous ones [5, 10]. However, previous research has revealed that RTX is effective in untreated NS [11]. In this study, RTX was initiated in four patients with nephrotic proteinuria and was found to be safe under the nephrotic state.
In this study, the regular RTX treatment, with a single dose of 500 mg administered every 6 months, was based on a previous report on adult patients with SDNS [6, 12]. To date, previous studies have reported a wide range of RTX regimens for primary NS [8, 10, 13]. The induction dose of RTX may vary based on disease state and concomitant immunosuppressive treatment. The findings of this study suggested that a regular single-dose of RTX (500 mg) administered every 6 months was extremely effective in patients with SDNS and should be recommended for this patient group. Interestingly, patients with a higher number of NS relapses before RTX initiation received a high number of RTX doses in this study. Although neither the number of relapses before RTX initiation nor the number of RTX doses was associated with NS relapse after RTX discontinuation, the number of NS relapses before RTX initiation likely influenced the determination of the RTX dosing schedule by the attending physician.
Based on a previous meta-analysis, the remission rates after RTX treatment were 91.6% in adult patients with MCD and 43% in those with FSGS [7]. The relapse rate after RTX treatment was 27.4% in patients with MCD and FSGS. However, in this study, all patients achieved GC discontinuation with NS remission by continuing treatment with regular single-dose RTX. The 2-year relapse rate was extremely low at 3%. The difference can be attributed to the fact that previous studies with low remission rates in this meta-analysis included patients with poor renal function and those not receiving RTX maintenance therapy [10, 11, 14, 15].
In children with idiopathic NS, RTX-associated hypogammaglobulinemia is a serious problem that may lead to severe infections [16, 17]. Fortunately, there were no serious infections in this study, but it is well known that hypogammaglobulinemia occurs relatively frequently after a long period of repeated administration, even with a single-dose RTX regimen, and serum IgG levels should be checked regularly over a long time.
Relapse developed simultaneously with the recovery of B-cell counts after RTX administration in children with SDNS [18]. It is also known that the frequency of infection is higher during the B cell depletion period than after the B cell recovery [5]. Therefore, although the B cell count was not routinely measured in this cohort study, it is recommended that the B cell count be measured as regularly as possible when using rituximab. By contrast, in idiopathic membranous nephropathy, which is more common in adults, re-positivity or high levels of anti-phospholipase A2 receptor (PLA2R) antibodies are associated with NS recurrence while on RTX treatment [19]. Novel autoantibodies have recently been identified in a subset of patients with MCD, in whom autoantibodies had never been detected before [20, 21]. Thus, in the future, autoantibody titers can be more important than B-cell counts in determining RTX discontinuation in these diseases.
Current studies have reported that patients with FRNS or SDNS who received regular single-dose RTX treatment have a reduced relapse rate [7, 8]. However, no prospective studies have evaluated the discontinuation of regular RTX treatment in patients who maintained NS remission. Although this was a retrospective study, most patients discontinued regular RTX treatment after long-term remission. In addition, three-fourths of the patients remained in remission for > 2 years after their last regular dose of rituximab without any treatment for NS. One possible mechanism is that RTX restores the balance between autoreactive T cells and regulatory T cells by reducing antigen-presenting B cells [22]. Therefore, discontinuation of regular RTX treatment should be considered when patients have been in long-term remission without GC or immunosuppressants for > 1 year.
This study had several limitations. First, it was a retrospective study. Thus, patient treatment was based on the discretion of each attending physician. Further, the schedule of regular RTX was not completely defined. Second, all patients belonged to the RTX group, which was not compared with the control group without RTX. Third, the number of patients was extremely low. Hence, the factors that might cause NS relapse after the discontinuation of regular RTX could not be determined. Lastly, safety issues have not been adequately evaluated in this study. However, this study also had some strengths. The median follow-up period from regular RTX treatment initiation was 51 months, which was longer than that in other previous studies. This study could identify the long-term outcomes after the discontinuation of regular RTX treatment. Therefore, this study can have a high generalizability.
In conclusion, all patients with relapsing SDNS achieved GC discontinuation with NS remission by receiving regular single-dose RTX treatment. In addition, three-fourths of the patients remained in remission for > 2 years after their last regular dose of rituximab without any treatment for NS. Nevertheless, prospective controlled studies should be performed to determine the appropriate duration of regular RTX treatment. However, this study revealed that the discontinuation of RTX treatment should be considered in patients who maintained long-term remission with regular RTX treatment.
Data availability
Partial restrictions to the data and/or materials apply: The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Rovin BH, Adler SG, Barratt J, Bridoux F, Burdge KA, Chan TM, et al. Executive summary of the KDIGO 2021 guideline for the management of glomerular diseases. Kidney Int. 2021;100:753–79. https://doi.org/10.1016/j.kint.2021.05.015.
Campbell RE, Thurman JM. The immune system and idiopathic nephrotic syndrome. Clin J Am Soc Nephrol. 2022;17:1823–34. https://doi.org/10.2215/cjn.07180622.
Nakayama M, Katafuchi R, Yanase T, Ikeda K, Tanaka H, Fujimi S. Steroid responsiveness and frequency of relapse in adult-onset minimal change nephrotic syndrome. Am J Kidney Dis. 2002;39:503–12. https://doi.org/10.1053/ajkd.2002.31400.
Cartron G, Watier H, Golay J, Solal-Celigny P. From the bench to the bedside: ways to improve rituximab efficacy. Blood. 2004;104:2635–42. https://doi.org/10.1182/blood-2004-03-1110.
Iijima K, Sako M, Nozu K, Mori R, Tuchida N, Kamei K, et al. Rituximab for childhood-onset, complicated, frequently relapsing nephrotic syndrome or steroid-dependent nephrotic syndrome: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2014;384:1273–81. https://doi.org/10.1016/s0140-6736(14)60541-9.
Iwabuchi Y, Takei T, Moriyama T, Itabashi M, Nitta K. Long-term prognosis of adult patients with steroid-dependent minimal change nephrotic syndrome following rituximab treatment. Medicine (Baltimore). 2014;93: e300. https://doi.org/10.1097/md.0000000000000300.
Xue C, Yang B, Xu J, Zhou C, Zhang L, Gao X, et al. Efficacy and safety of rituximab in adult frequent-relapsing or steroid-dependent minimal change disease or focal segmental glomerulosclerosis: a systematic review and meta-analysis. Clin Kidney J. 2021;14:1042–54. https://doi.org/10.1093/ckj/sfaa191.
Aslam A, Koirala A. Review of the role of rituximab in the management of adult minimal change disease and immune-mediated focal and segmental glomerulosclerosis. Glomerular Dis. 2023;3:211–9. https://doi.org/10.1159/000533695.
Wada T, Ishimoto T, Nakaya I, Kawaguchi T, Sofue T, Shimizu S, et al. A digest of the evidence-based clinical practice guideline for nephrotic syndrome 2020. Clin Exp Nephrol. 2021;25:1277–85. https://doi.org/10.1007/s10157-021-02098-5.
Guitard J, Hebral AL, Fakhouri F, Joly D, Daugas E, Rivalan J, et al. Rituximab for minimal-change nephrotic syndrome in adulthood: predictive factors for response, long-term outcomes and tolerance. Nephrol Dial Transplant. 2014;29:2084–91. https://doi.org/10.1093/ndt/gfu209.
Fenoglio R, Sciascia S, Beltrame G, Mesiano P, Ferro M, Quattrocchio G, et al. Rituximab as a front-line therapy for adult-onset minimal change disease with nephrotic syndrome. Oncotarget. 2018;9:28799–804. https://doi.org/10.18632/oncotarget.25612.
Takei T, Itabashi M, Moriyama T, Kojima C, Shiohira S, Shimizu A, et al. Effect of single-dose rituximab on steroid-dependent minimal-change nephrotic syndrome in adults. Nephrol Dial Transplant. 2013;28:1225–32. https://doi.org/10.1093/ndt/gfs515.
Fujimoto K, Kagaya Y, Kumano S, Fujii A, Tsuruyama Y, Matsuura T, et al. Retrospective single-arm cohort study of steroid-dependent minimal change nephrotic syndrome treated with very low-dose rituximab. Clin Nephrol. 2021;95:29–36. https://doi.org/10.5414/cn110245.
Bruchfeld A, Benedek S, Hilderman M, Medin C, Snaedal-Jonsdottir S, Korkeila M. Rituximab for minimal change disease in adults: long-term follow-up. Nephrol Dial Transplant. 2014;29:851–6. https://doi.org/10.1093/ndt/gft312.
Roccatello D, Sciascia S, Rossi D, Alpa M, Naretto C, Radin M, et al. High-dose rituximab ineffective for focal segmental glomerulosclerosis: a long-term observation study. Am J Nephrol. 2017;46:108–13. https://doi.org/10.1159/000477944.
Inoki Y, Kamei K, Nishi K, Sato M, Ogura M, Ishiguro A. Incidence and risk factors of rituximab-associated hypogammaglobulinemia in patients with complicated nephrotic syndrome. Pediatr Nephrol. 2022;37:1057–66. https://doi.org/10.1007/s00467-021-05304-4.
Zurowska A, Drozynska-Duklas M, Topaloglu R, Bouts A, Boyer O, Shenoy M, et al. Rituximab-associated hypogammaglobulinemia in children with idiopathic nephrotic syndrome: results of an ESPN survey. Pediatr Nephrol. 2023;38:3035–42. https://doi.org/10.1007/s00467-023-05913-1.
Kamei K, Ito S, Nozu K, Fujinaga S, Nakayama M, Sako M, et al. Single dose of rituximab for refractory steroid-dependent nephrotic syndrome in children. Pediatr Nephrol. 2009;24:1321–8. https://doi.org/10.1007/s00467-009-1191-0.
Ruggenenti P, Debiec H, Ruggiero B, Chianca A, Pellé T, Gaspari F, et al. Anti-phospholipase a2 receptor antibody titer predicts post-rituximab outcome of membranous nephropathy. J Am Soc Nephrol. 2015;26:2545–58. https://doi.org/10.1681/asn.2014070640.
Watts AJB, Keller KH, Lerner G, Rosales I, Collins AB, Sekulic M, et al. Discovery of autoantibodies targeting nephrin in minimal change disease supports a novel autoimmune etiology. J Am Soc Nephrol. 2022;33:238–42. https://doi.org/10.1681/asn.2021060794.
Jamin A, Berthelot L, Couderc A, Chemouny JM, Boedec E, Dehoux L, et al. Autoantibodies against podocytic UCHL1 are associated with idiopathic nephrotic syndrome relapses and induce proteinuria in mice. J Autoimmun. 2018;89:149–61. https://doi.org/10.1016/j.jaut.2017.12.014.
Datta SK. Anti-CD20 antibody is an efficient therapeutic tool for the selective removal of autoreactive T cells. Nat Clin Pract Rheumatol. 2009;5:80–2. https://doi.org/10.1038/ncprheum0983.
Acknowledgements
This work originated from the Iwate Prefectural Central Hospital.
Funding
This study was not supported by any grant or sponsor.
Author information
Authors and Affiliations
Contributions
Conceptualization: Izaya Nakaya. Data curation: Eiichiro Saito, Izaya Nakaya. Formal analysis: Eiichiro Saito, Izaya Nakaya. Investigation: Eiichiro Saito, Izaya Nakaya. Methodology: Eiichiro Saito, Izaya Nakaya. Project administration: Eiichiro Saito, Izaya Nakaya. Resources: Jun Soma, Izaya Nakaya. Visualization: Eiichiro Saito, Izaya Nakaya. Writing-original draft: Eiichiro Saito, Izaya Nakaya. Writing-review and editing: Atsushi Oura, Tetsuya Kyo, Shun Ishigaki, Hitomi Kamei, Yuki Nakamura, Jun Soma.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
This study was approved by the Ethics Committee of Iwate Prefectural Central Hospital and conducted in accordance with the ethical principles of the Declaration of Helsinki.
Informed consent
A written informed consent was not obtained from the patients because the ethical guidelines for epidemiological research in Japan do not require informed consent for a retrospective cohort study using only existing medical records.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Saito, E., Oura, A., Kyo, T. et al. Relapse during and after regular single-dose rituximab treatment in adult patients with steroid-dependent nephrotic syndrome. Clin Exp Nephrol (2024). https://doi.org/10.1007/s10157-024-02508-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10157-024-02508-4