Abstract
Background
For right colon surgery, there is an increasing body of literature comparing the safety of robotic right colectomy (RRC) with laparoscopic right colectomy (LRC). The aim of the present systematic review and meta-analysis is to assess the safety and efficacy of RRC versus LRC, including homogeneous subgroup analyses for extracorporeal anastomosis (EA) and intracorporeal anastomosis (IA).
Methods
PubMed, Web of Science, Embase, and Cochrane Library databases were searched for studies published between January 2000 and January 2022. Length of hospital stay, operation time, rate of conversion to laparotomy, time to first flatus, number of harvested lymph nodes, estimated blood loss, rate of overall complication, ileus, anastomotic leakage, wound infection, and total costs were measured.
Results
Forty-two studies (RRC: 2772 patients; LRC: 12,469 patients) were evaluated. Regardless of the type of anastomosis, RRC showed shorter length of hospital stay, lower rate of conversion to laparotomy, shorter time to first flatus, lower rate of overall complications, and a higher number of harvested lymph nodes compared with LRC, but longer operative time and higher total costs. In the IA subgroup, RRC had a shorter length of hospital stay, longer operative time, and lower rate of conversion to laparotomy compared with LRC, with no difference for the remaining outcomes. In the EA subgroup, RRC had a longer operative time, lower estimated blood loss, lower rate of overall complications, and higher total costs compared with LRC, with the other outcomes being similar.
Conclusion
The safety and efficacy of RRC is superior to LRC, especially when an intracorporeal anastomosis is performed. Most included articles were retrospective, offering low-quality evidence and limited conclusions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The first laparoscopic right colectomy (LRC) was performed in 1990 [1], and since then it has been increasingly used for benign and malignant disease. This minimally invasive (MI) approach offers faster recovery with earlier return to normal bowel function, less pain, shorter length of hospital stay, and lower postoperative complications compared with open surgery, with similar oncological outcomes [2,3,4,5]. The steep learning curve, the ability to provide only a two-dimensional surgical field of view, lens instability due to hand fatigue, and susceptibility to human muscle tremors all limit further development of laparoscopy.
Since 2000, robot-assisted systems have been widely used in various surgical fields [6]. As an emerging technology, the advantages of robots are that they can provide 3D local magnified vision, hand tremor filtering, more flexible and accurate multi-angle moving mechanical arms using Endowrist technology, more stable four arm operation, and their ergonomic design provides surgeons with a better surgical experience [7, 8]. These designs can effectively overcome some of the limitations of laparoscopic surgery.
Since Weber et al.. first reported using robotic colectomy surgery in 2002 [9], some centers have experimented with the technique with satisfactory results. However, these studies are based on single-center experience and non-randomized controlled, the results obtained are insufficient evidence. In addition, the advantages of the robotic surgery system in colectomy are still controversial [10,11,12,13, 29, 41]. The learning curve may be shorter with robotic technology compared with laparoscopy [22], and it may facilitate some procedural steps, such as the construction of an intracorporeal anastomosis during right colectomy [10]. Indeed, there is still a debate about the superiority of intracorporeal over extracorporeal anastomosis. Hence, it is of great significance to analyze the safety and effectiveness of robotic right colectomy (RRC), including a comprehensive, systematic, and intuitive homogeneous subgroup analyses for extracorporeal anastomosis (EA) and intracorporeal anastomosis (IA). We conducted this meta-analysis of published literature to compare the length of hospital stay, operation time, rate of conversion to laparotomy, time to first flatus, number of harvested lymph nodes, estimated blood loss, rate of overall complication, ileus, anastomotic leakage, wound infection, and total costs, to assess the safety and efficacy of RRC versus LRC including a homogeneous subgroup analyses for extracorporeal anastomosis (EA) and intracorporeal anastomosis (IA).
Materials and methods
Search strategy
The study was registered on PROSPERO (CRD42022324624), and the findings have been reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and Assessing the Methodological Quality of Systematic Reviews (AMSTAR) guidelines. The search terms “right colectomy,” “robotic,” and “laparoscopic” were used. Two authors independently searched the PubMed, Web of Science, Embase, and Cochrane Library databases for relevant studies published between January 2000 and January 2022. The search also included all of the Mesh terms. No language restriction was applied, and the search was limited to human studies. The reference lists of all retrieved articles were reviewed for further identification of potentially relevant studies.
Inclusion and exclusion criteria
Any studies that met the following criteria were considered: (1) compared RRC with LRC of intracorporeal anastomosis (IA) and (or) extracorporeal anastomosis (EA) and (2) compared RRC versus LRC on at least one short-term outcome, including operative and postoperative. If there were two or more articles by the same authors or research institutions, the most recent publication was selected. We also manually searched the abstracts published at major international conferences. A manual search of the bibliographies of relevant articles was also carried out to identify trials for possible inclusion. Reference lists of all retrieved articles were manually searched for additional studies.
Articles that met the following criteria were not considered: (1) studies that reported on patients with colorectal surgery but did not contain a independent group of right colectomy, (2) studies in which data could not be extracted from the published results, and (3) articles including abstracts, letters, expert opinions, reviews without raw data, case reports, or studies without control groups.
Data extraction
The data extraction and input of the included literature were completed by two reviewers independently. If there was any disagreement during the selection process, the decision would be made by a third reviewers. The compiled information included author name, geographical region, year of publication, study period, type of study, surgical type, surgical technique, sample size, mean age, gender, body mass index (BMI), length of hospital stay, operation time, rate of conversion to laparotomy, time to first flatus, number of harvested lymph nodes, estimated blood loss, rate of overall complication, ileus, anastomotic leakage, wound infection, and total costs.
Statistical analysis
All data were analyzed using RevMan 5.4 and Stata 17.0. Heterogeneity was detected by the chi-square test. A P-value > 0.10 was considered as homogeneous, otherwise as heterogeneous. Moreover, the I2 index was used to assess heterogeneity, and I2 > 50% was considered as statistically significant. For homogeneous affirmation, the fixed effects model was selected; otherwise, a random effects model was adopted. The odds ratio (OR) and mean difference (MD) were calculated, and publication bias was assessed by Egger’s test. In some publications, the mean values and standard deviation values were unavailable. The methods used to determine these values based on the available median and range data have been described previously by Hozo et al.. As not all patients had colon cancer, the mode of laparoscopic right hemicolectomy, which mainly included D3 and complete mesocolic excision robotically with standard D2/D1 right hemicolectomy, was performed with high heterogeneity in operative time. All costs reported in Euros or CNY were converted into US dollars. The pooled estimates of the mean difference (MD) and 95% CI were calculated using a random effects model because of the expected heterogeneity among the included studies.
Results
Selected studies
The study selection process is summarized in Fig. 1. According to the retrieval strategy and data collection method, a total of 675 related studies were retrieved. From reading the titles and abstracts, we excluded 400 studies and initially included 275 studies. A total of 228 studies were eliminated due to the lack of a right colon resection trial, while the remaining 47 studies met the criteria. After reading the full text according to the criteria and filtering for data integrity, 42 studies [14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55] with a total of 15,241 patients (RRC group, 2772 patients; LRC group, 12,469 patients) were eventually included in this meta-analysis.
Study characteristic
The characteristics of the individual trials are presented in Table 1. A total of 2772 patients were included in the RRC group, while 12,469 patients were included in the LRC group. The studies were from Australia (1 study), Italy (14 studies), the UK (1 study), the USA (14 studies), Korea (3 studies), France (2 studies), Denmark (2 studies), Turkey (1 study), Spain (1 study), Singapore (1 study), and China (2 studies).
Study quality
The Newcastle–Ottawa Scale (NOS) was used to assess the quality of the studies. The total NOS score was 9, and papers with a score ≥ 6 were classified as methodologically sound studies. All articles included in this study rated between 6 and 9, indicating that the study quality was sufficient (Table 2).
Comparison between RRC versus LRC
Overall, 42 retrospective studies compared RRC versus LRC on at least the primary outcome or one secondary outcome. Pooled data analysis showed that robotic surgery has a significantly longer operative time and higher total costs but shorter length of hospital stay, lower rate of conversion to laparotomy, shorter time to first flatus, lower rate of overall complication, and higher number of harvested lymph nodes (Fig. 2). All other operative and pathological outcomes, including estimated blood loss, ileus, wound infection, and anastomotic leakage were similar for RRC and LRC (Fig. 3).
Comparison between RRC-IA versus LRC-IA
Overall, ten retrospective studies compared RRC-EA versus LRC-EA on at least the primary outcome or one secondary outcome. Overall, 1534 patients were included: 809 (52.7%) undergoing robotic and 725 (47.3%) undergoing laparoscopic surgery. Pooled data analysis showed a shorter length of hospital stay for RRC-IA than for LRC-IA. Conversely, the overall operative time was longer and the rate of conversion to laparotomy was lower for RRC-IA. All other operative and pathological outcomes were similar between RRC-IA and LRC-IA (Fig. 4).
Comparison between RRC-EA versus LRC-EA
Overall, nine retrospective studies compared RRC-EA versus LRC-EA on at least the primary outcome or one secondary outcome. Overall, 669 patients were included: 211 (31.5%) undergoing robotic and 458 (68.5%) undergoing laparoscopy surgery. Pooled data analysis showed the overall operative time was longer, the estimated blood loss was lower, the rate of overall complication was lower, the total costs was higher for RRC-EA than for LRC-EA. For all other comparable outcomes, there was no difference between the two surgical groups. The results of this subgroup analysis are represented in Fig. 5.
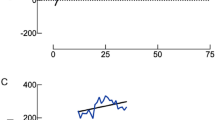
Publication bias
A funnel plot was used to assess the presence of publication bias. The absence of asymmetry in the funnel plot indicated no evidence of publication bias. No noticeable publication bias was observed according to the results of Egger’s test (P = 0.06; Fig. 6).
Discussion
Since the first report of laparoscopy surgery for a colorectal operation in 1991, the scope of its influence has gradually expanded and it has become the standard treatment in colorectal operation [1, 56]. However, laparoscopy techniques have inherent limitations, including poor stereoscopic vision and depth perception due to two-dimensional (2D) imaging (despite the rise of 3D platforms), lens instability, poor ergonomics, direct fixation of surgical instrument tips, and operational tremors associated with magnification [57].
With the advent of robotic surgery systems, these problems that plagued surgical operations have been solved, and they have become increasingly popular in various disciplines, especially colorectal operation. The design of robotic surgery systems overcomes some limitations of laparoscopic surgery, such as the stability of the control by the surgeon’s 3D view, with seven degrees of freedom and 540° rotating wrist instruments, the remarkable human body engineering design, and tremor filtering functions. Of course, the system also has limitations, such as the loss of haptic feedback, the limited range of movement of the robotic arm, and the fact that it is time consuming and costly [58,59,60].
There is evidence that robotic right colectomy is a feasible and safe procedure [57, 61,62,63,64,65,66], with less trauma and faster postoperative recovery, but there are few indicators, with limited literature and a lack of long-term prognostic indicators. In this meta-analysis, 42 studies involving 15,241 patients were included. We conducted this meta-analysis of published literature to compare the operation related indicators (operation time, estimated blood loss, rate of conversion to laparotomy, and number of harvested lymph nodes) and perioperative related indicators (length of hospital stay, time to first flatus, rate of overall complication, ileus, anastomotic leakage, wound infection, and total costs) to evaluate the safety and efficacy of robotic right colectomy (RRC) (including only homogeneous surgical subgroups).
In terms of operation time, we observed that there was statistically significant difference between the two groups (included subgroups: RRC-IA versus LRC-IA and RRC-EA versus LRC-EA). The operation time in the RRC group was longer than that in the LRC group, which may be related to the unskilled operation of the robotic surgical system in the early stage of surgery, the lack of experience in trocar placement, the long time to install the robot, and the lack of tacit cooperation between doctors and nurses [27]. de’Angelis [22] divided 30 cases of robotic surgery and 50 cases of laparoscopy surgery into three groups according to the operation sequence. Before the two groups of ten cases, LRC group operation time is superior to the RRC group, but after 20 cases, the RRC group is shorter. It should be emphasized that the type of anastomosis in this study was extracorporeal anastomoses (RRC-EA). Therefore, after the training of a certain number of surgical cases, the advantages of the robotic surgery systems accurate and flexible operation can be fully demonstrated, and the operation time can be significantly shortened. The shortening of time is also related to the fact that the robotic surgical system can filter out the natural tremor of the human hand while presenting a magnified 3D field of view, which is more convenient for intraoperative tissue positioning and grasping. It is believed that with the popularization and application of robotic surgery systems, the operation time of robots (not only on RRC-EA but also on RRC-IA) will be significantly reduced, even better than laparoscopic surgery in the future. There was no significant difference between the two groups in terms of estimated blood loss. Although robotic surgery is thought to potentially reduce intraoperative bleeding, the chance of intraoperative mishandling is higher relative to laparoscopy surgery due to its lack of force feedback. At the same time, with the popularization of complete mesocolic excision (CME) for tumors of the right colon, the intraoperative bleeding of laparoscopic surgery is reduced, resulting in no obvious advantage of robotic surgery in the control of intraoperative bleeding. However, estimated blood loss was demonstrated to be lower in RRC-EA compared with LRC-EA, possibly because extracorporeal anastomosis is more thorough in hemostasis.
The rate of conversion to laparotomy was significantly lower in the RRC group compared with the LRC group. The robotic surgical system with one lens arm and three robotic arms that can rotate 540°, can perform fine operations in small spaces, improve the ability to deal with more serious adhesions, and reduce the rate of conversion to laparotomy. However, there was no significant difference between RRC-EA and LRC-EA in terms of rate of conversion to laparotomy. This may be due to the lack of included data, and further large prospective series are needed. The number of harvested lymph nodes is an important index to evaluate the radical effect of surgery. This study showed that the number of harvested lymph nodes was higher in the RRC group compared with the LRC group. This demonstrates that robotic methods, which can perform similar radical resection compared with laparoscopic techniques, have potential advantages for tissue dissection.
In terms of perioperative indicators, the length of hospital stay and the time to first flatus in the RRC group were significantly shorter, indicating that the robot had less trauma and faster recovery compared with laparoscopy, which was in line with the minimally invasive concept in the current surgical field. Still, differences in the length of hospital stay were significant only when intracorporeal anastomosis was performed. This confirmed the superiority of intracorporeal anastomosis in improving the recovery of the bowel function. The RRC group showed lower rates of overall complications. There are two possible reasons for this: the average age of patients in the LRC group was older, or the robot itself caused less tissue loss. However, there was no significant difference in ileus, anastomotic leakage, or wound infection between the two groups (including the subgroup analysis). The total cost of robotic surgery is higher than that of laparoscopy surgery, which is mainly related to the use of robotic surgery systems and the high cost of related consumables. As a new type of equipment, the robotic surgery system currently has only one manufacturer, and therefore, the cost is bound to be high. The faster recovery of intestinal functions and shorter hospital stay are counterbalanced by the high general costs of robotic platform use and maintenance. It is believed that with the widespread use of robotic surgery systems and the advent of other robotic surgery systems, the cost of surgical equipment will inevitably drop significantly.
Several limitations exist in the present meta-analysis. Firstly, almost all the articles included in this meta-analysis were retrospective, which limited the strength of the final conclusions. Therefore, the risk of important bias is relevant. Secondly, it is impossible to match patient characteristics in most of the studies, increasing heterogeneity between the two groups. Thirdly, most of the literature presented an intergroup difference in terms of the technique used to perform the anastomosis, which could have significantly biased the comparison between the groups. Due to the differences in equipment resources and the technical level of the implementer among different research institutions, the meta-analysis results have certain heterogeneity. For example, the high heterogeneity in operation time results may be because the robot assistance system is an emerging technology; therefore, each surgical group is affected by experience and surgical level, resulting in the learning curve stage is not consistent, thus having an impact on the surgical results. On the other hand, different definitions of operative time in different surgical groups may also lead to heterogeneity. Finally, data on surgery-related costs are still based on a limited number of surgeries performed in different health systems from different countries, and need to be confirmed by further cost–benefit analyses.
Conclusion
This meta-analysis shows that RRC is superior to LRC in terms of the length of hospital stay. This could be explained by the lower overall complication rate and the shorter time to flatus of the robotic group. This may be due to the fact that intracorporeal anastomosis is more frequently performed in the robotic group than in the laparoscopic group. When an extracorporeal anastomosis is performed, the advantages provided by robotic surgery disappear. Almost all the articles included in this meta-analysis were retrospective, which limited the final conclusions. Therefore, a higher level of evidence achieved by further randomized clinical trials is required.
References
Jacobs M, Verdeja JC, Goldstein HS (1991) Minimally invasive colon resection (laparoscopic colectomy) [J]. Surg Laparosc Endosc 1(3):144–150
Bonjer HJ, Hop WC, Nelson H et al. (2007) Laparoscopically assisted vs open colectomy for colon cancer: a meta-analysis [J]. Arch Surg 142(3):298–303
Fleshman J, Sargent DJ, Green E et al. (2007) Laparoscopic colectomy for cancer is not inferior to open surgery based on 5-year data from the COST Study Group trial [J]. Ann Surg 246(4):655–662
Buunen M, Veldkamp R, Hop WC et al. (2009) Survival after laparoscopic surgery versus open surgery for colon cancer: long-term outcome of a randomised clinical trial [J]. Lancet Oncol 10(1):44–52
Jayne DG, Thorpe HC, Copeland J et al. (2007) Five-year follow up of the Medical Research Council CLASICC trial of laparoscopically assisted versus open surgery for colorectal cancer [J]. Br J Surg 97(11):1638–1645
Maeso S, Reza M, Mayol JA et al. (2010) Efficacy of the Da Vinci surgical system in abdominal surgery compared with that of laparoscopy: a systematic review and meta-analysis[J]. Ann Surg 252(2):254–262
Fransgaard T, Pinar I, Thygesen LC et al. (2018) Association between robot-assisted surgery and resection quality in patients with colorectal cancer [J]. Surg Oncol 27(2):177–184
Lim DR, Min BS, Kim MS et al. (2013) Robotic versus laparoscopic anterior resection of sigmoid colon cancer: comparative study of long-term oncologic outcomes [J]. Surg Endosc 27(4):1379–1385
Weber PA, Merola S, Wasielewski A et al. (2002) Telerobotic-assisted laparoscopic right and sigmoid colectomies for benign disease [J]. Dis Colon Rectum 45(12):1689–1694
Johnson CS, Kassir A, Marx DS et al. (2019) Performance of da Vinci Stapler during robotic-assisted right colectomy with intracorporeal anastomosis [J]. J Robot Surg 13(1):115–119
Helvind NM, Eriksen JR, Mogensen A et al. (2013) No differences in short-term morbidity and mortality after robot-assisted laparoscopic versus laparoscopic resection for colonic cancer: a case-control study of 263 patients [J]. Surg Endosc 27(7):2575–2580
Lee HJ, Choi GS, Park JS et al. (2018) A novel robotic right colectomy for colon cancer via the suprapubic approach using the da Vinci Xi system: initial clinical experience [J]. Ann Surg Treat Res 94(2):83–87
Ioannidis A, Kontzoglou K, Kouraklis G et al. (2018) Short-term outcomes in patients with colon cancer treated with robotic right colectomy [J]. J BUON 23(2):317–321
Ahmadi N, Mor I, Warner R (2022) Comparison of outcome and costs of robotic and laparoscopic right hemicolectomies [J]. J Robot Surg 16(2):429–436
Blumberg D (2019) Robotic colectomy with intracorporeal anastomosis is feasible with no operative conversions during the learning curve for an experienced laparoscopic surgeon developing a robotics program [J]. J Robot Surg 13(4):545–555
Cardinali L, Belfiori G, Ghiselli R et al. (2016) Robotic versus laparoscopic right colectomy for cancer: short-term outcomes and influence of body mass index on conversion rate [J]. Minerva Chir 71(4):217–222
Casillas MA, Leichtle SW, Wahl WL et al. (2014) Improved perioperative and short-term outcomes of robotic versus conventional laparoscopic colorectal operations [J]. Am J Surg 208(1):33–40
Ceccarelli G, Costa G, Ferraro V et al. (2021) Robotic or three-dimensional (3D) laparoscopy for right colectomy with complete mesocolic excision (CME) and intracorporeal anastomosis? A propensity score-matching study comparison [J]. Surg Endosc 35(5):2039–2048
Davis BR, Yoo AC, Moore M et al. (2014) Robotic-assisted versus laparoscopic colectomy: cost and clinical outcomes [J]. JSLS 18(2):211–224
deSouza AL, Prasad LM, Park JJ et al. (2010) Robotic assistance in right hemicolectomy: is there a role [J]? Dis Colon Rectum 53(7):1000–1006
Deutsch GB, Sathyanarayana SA, Gunabushanam V et al. (2012) Robotic vs. laparoscopic colorectal surgery: an institutional experience [J]. Surg Endosc 26(4):956–963
de’Angelis N, Lizzi V, Azoulay D, et al. (2016) Robotic versus laparoscopic right colectomy for colon cancer: analysis of the initial simultaneous learning curve of a surgical fellow [J]. J Laparoendosc Adv Surg Tech A, 26(11):882–892
Dohrn N, Klein MF, Gögenur I (2021) Robotic versus laparoscopic right colectomy for colon cancer: a nationwide cohort study [J]. Int J Colorectal Dis 36(10):2147–2158
Dolejs SC, Waters JA, Ceppa EP et al. (2017) Laparoscopic versus robotic colectomy: a national surgical quality improvement project analysis [J]. Surg Endosc 31(6):2387–2396
Ferrara F, Piagnerelli R, Scheiterle M et al. (2016) Laparoscopy versus robotic surgery for colorectal cancer: a single-center initial experience [J]. Surg Innov 23(4):374–380
Ferri V, Quijano Y, Nuñez J et al. (2021) Robotic-assisted right colectomy versus laparoscopic approach: case-matched study and cost-effectiveness analysis [J]. J Robot Surg 15(1):115–123
Gerbaud F, Valverde A, Danoussou D, et al. (2019) Experience with transitioning from laparoscopic to robotic right colectomy [J]. JSLS 23(4): e2019.00044.
Guerrieri M, Campagnacci R, Sperti P et al. (2015) Totally robotic vs 3D laparoscopic colectomy: a single centers preliminary experience [J]. World J Gastroenterol 21(46):13152–13159
Haskins IN, Ju T, Skancke M et al. (2018) Right colon resection for colon cancer: does surgical approach matter [J]? J Laparoendosc Adv Surg Tech A 28(10):1202–1206
Kang J, Park YA, Baik SH et al. (2016) A comparison of open, laparoscopic, and robotic surgery in the treatment of right-sided colon cancer [J]. Surg Laparosc Endosc Percutan Tech 26(6):497–502
Kelley SR, Duchalais E, Larson DW (2018) Short-term outcomes with robotic right colectomy [J]. Am Surg 84(11):1768–1773
Khan JS, Ahmad A, Odermatt M, et al. (2021) Robotic complete mesocolic excision with central vascular ligation for right colonic tumours—a propensity score-matching study comparing with standard laparoscopy [J]. BJS Open 5(2): zrab016.
Lujan HJ, Maciel VH, Romero R et al. (2013) Laparoscopic versus robotic right colectomy: a single surgeon’s experience [J]. J Robot Surg 7(2):95–102
Lujan HJ, Plasencia G, Rivera BX et al. (2018) Advantages of robotic right colectomy with intracorporeal anastomosis [J]. Surg Laparosc Endosc Percutan Tech 28(1):36–41
Mégevand JL, Amboldi M, Lillo E, et al. (2019) Right colectomy: consecutive 100 patients treated with laparoscopic and robotic technique for malignancy. Cumulative experience in a single centre [J]. Updat Surg 71(1):151–156.
Merola G, Sciuto A, Pirozzi F et al. (2020) Is robotic right colectomy economically sustainable? A multicentre retrospective comparative study and cost analysis [J]. Surg Endosc 34(9):4041–4047
Migliore M, Giuffrida MC, Marano A et al. (2021) Robotic versus laparoscopic right colectomy within a systematic ERAS protocol: a propensity-weighted analysis [J]. Updates Surg 73(3):1057–1064
Morpurgo E, Contardo T, Molaro R et al. (2013) Robotic-assisted intracorporeal anastomosis versus extracorporeal anastomosis in laparoscopic right hemicolectomy for cancer: a case control study [J]. J Laparoendosc Adv Surg Tech A 23(5):414–417
Ngu JC, Ng YY (2018) Robotics confers an advantage in right hemicolectomy with intracorporeal anastomosis when matched against conventional laparoscopy [J]. J Robot Surg 12(4):647–653
Nolan HR, Smith BE, Honaker MD (2018) Operative time and length of stay is similar between robotic assisted and laparoscopic colon and rectal resections [J]. J Robot Surg 12(4):659–664
Park JS, Kang H, Park SY et al. (2019) Long-term oncologic after robotic versus laparoscopic right colectomy: a prospective randomized study [J]. Surg Endosc 33(9):2975–2981
Rattenborg S, Bundgaard L, Andersen J, et al. (2021) Intracorporeal anastomosis in right hemicolectomy for colon cancer: short‑term outcomes with the DaVinci Xi robot. Journal of Robotic Surgery [J]. J Robot Surg 15(6):915–922.
Rawlings AL, Woodland JH, Vegunta RK et al. (2007) Robotic versus laparoscopic colectomy [J]. Surg Endosc 21(10):1701–1708
Scotton G, Contardo T, Zerbinati A et al. (2018) From laparoscopic right colectomy with extracorporeal anastomosis to robot-assisted intracorporeal anastomosis to totally robotic right colectomy for cancer: the evolution of robotic multiquadrant abdominal surgery [J]. J Laparoendosc Adv Surg Tech A 28(10):1216–1222
Shi XL, Zhu XL, Wang H et al. (2020) Robotic versus laparoscopic colectomy for right-sided colon cancer: short-term outcomes from a single center [J]. Chin J Robot Surg 1(5):307–316
Shin JY (2012) Comparison of short-term surgical outcomes between a robotic colectomy and a laparoscopic colectomy during early experience [J]. J Korean Soc Coloproctol 28(1):19–26
Solaini L, Cavaliere D, Pecchini F et al. (2019) Robotic versus laparoscopic right colectomy with intracorporeal anastomosis: a multicenter comparative analysis on short-term outcomes [J]. Surg Endosc 33(6):1898–1902
Sorgato N, Mammano E, Contardo T, et al. (2021) Right colectomy with intracorporeal anastomosis for cancer: a prospective comparison between robotics and laparoscopy [J]. J Robot Surg. https://doi.org/10.1007/s11701-021-01290-9.
Spinoglio G, Bianchi PP, Marano A et al. (2018) Robotic versus laparoscopic right colectomy with complete mesocolic excision for the treatment of colon cancer: perioperative outcomes and 5-year survival in a consecutive series of 202 patients [J]. Ann Surg Oncol 25(12):3580–3586
Tagliabue F, Burati M, Chiarelli M et al. (2020) Robotic vs laparoscopic right colectomy—the burden of age and comorbidity in perioperative outcomes: an observational study [J]. World J Gastrointest Surg 12(6):287–297
Trastulli S, Coratti A, Guarino S et al. (2015) Robotic right colectomy with intracorporeal anastomosis compared with laparoscopic right colectomy with extracorporeal and intracorporeal anastomosis: a retrospective multicentre study [J]. Surg Endosc 29(6):1512–1521
Widmar M, Keskin M, Beltran P et al. (2016) Incisional hernias after laparoscopic and robotic right colectomy [J]. Hernia 20(5):723–728
WidmarM, Keskin M, Strombom P, et al. (2017) Lymph node yield in right colectomy for cancer: a comparison of open, laparoscopic and robotic approaches [J]. Colorectal Dis 19(10):888–894.
Yozgatli TK, Aytac E, Ozben V et al. (2019) Robotic complete mesocolic excision versus conventional laparoscopic hemicolectomy for right-sided colon cancer [J]. J Laparoendosc Adv Surg Tech A 29(5):671–676
Zeng YJ, Wang GH, Liu Y et al. (2020) The ‘“Micro Hand S”’ robot-assisted versus conventional laparoscopic right colectomy: short-term outcomes at a single center [J]. J Laparoendosc Adv Surg Tech A 30(4):363–368
Araujo SE, Seid VE, Klajner S (2014) Robotic surgery for rectal cancer: current immediate clinical and oncological outcomes [J]. World J Gastroenterol 20(39):14359–14370
Mak TW, Lee JF, Futaba K et al. (2014) Robotic surgery for rectal cancer: a systematic review of current practice [J]. World J Gastrointest Oncol 6(6):184–193
Woeste G, Bechstein WO, Wullstein C (2005) Does telerobotic assistance improve laparoscopic colorectal surgery [J]? Int J Colorectal Dis 20(3):253–257
Balch GC (2009) Emerging role of laparoscopic and robotic surgery for rectal cancers [J]. Ann Surg Oncol 16(6):1451–1453
Ahmed K, Khan MS, Vats A et al. (2009) Current status of robotic assisted pelvic surgery and future developments [J]. Int J Surg 7(5):431–440
Trinh BB, Jackson NR, Hauch AT, et al. (2014) Robotic versus laparoscopic colorectal surgery [J]. JSLS 18(4).
Zarak A, Castillo A, Kichler K et al. (2015) Robotic versus laparoscopic surgery for colonic disease: a meta-analysis of postoperative variables [J]. Surg Endosc 29(6):1341–1347
Papanikolaou IG (2014) Robotic surgery for colorectal cancer: systematic review of the literature [J]. Surg Laparosc Endosc Percutan Tech 24(6):478–483
Petrucciani N, Sirimarco D, Nigri G et al. (2015) Robotic right colectomy: a worthwhile procedure? Results of a meta-analysis of trials comparing robotic versus laparoscopic right colectomy [J]. J Minim Access Surg 11(1):22–28
Tandogdu Z, Vale L, Fraser C et al. (2015) A systematic review of economic evaluations of the use of robotic assisted laparoscopy in surgery compared with open or laparoscopic surgery [J]. Appl Health Econ Health Policy 13(5):457–467
Waters PS, Cheung FP, Peacock O et al. (2020) Successful patient-oriented surgical outcomes in robotic vs laparoscopic right hemicolectomy for cancer-a systematic review [J]. Colorectal Dis 22(5):488–499
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Our study is a systematic review and meta-analysis, so there was no ethical approval required.
Research registration number
The name of the registry: RROSPERO; Research Registration Unique Identifying Number. (UIN): CRD42022324624.
Informed consent
Our study is a systematic review and meta-analysis, so there was no informed consent required.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, J., Zhao, S., Chen, W. et al. Comparison of robotic right colectomy and laparoscopic right colectomy: a systematic review and meta-analysis. Tech Coloproctol 27, 521–535 (2023). https://doi.org/10.1007/s10151-023-02821-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10151-023-02821-2