Abstract
Background
Oncologic benefits of laparoscopic radical nephroureterectomy (LNU) are unclear. We aimed to evaluate the impact of surgical approach for radical nephroureterectomy on oncologic outcomes in patients with locally advanced upper tract urothelial carcinoma (UTUC).
Methods
Of 426 patients who underwent radical nephroureterectomy at five medical centers between February 1995 and February 2017, we retrospectively investigated oncological outcomes in 229 with locally advanced UTUC (stages cT3-4 and/or cN+). The surgical approach was classified as open nephroureterectomy (ONU) or LNU, and oncologic outcomes, including intravesical recurrence-free survival (RFS), visceral RFS, cancer-specific survival (CSS), and overall survival (OS), were compared between the groups. The inverse probability of treatment weighting (IPTW)-adjusted Cox-regression analyses was performed to evaluate the impact of LNU on the prognosis.
Results
Of the 229 patients, 48 (21%) underwent LNU. There were significant differences in patient backgrounds, including preoperative renal function, lymph-node involvement, lymphovascular invasion, and surgical margins, between the groups. Before the background adjustment, intravesical RFS, visceral RFS, CSS, and OS were significantly inferior in the ONU group than in the LNU group. However, in the IPTW-adjusted Cox-regression analysis, no significant differences were observed in intravesical RFS (hazard ratio [HR], 0.65; P = 0.476), visceral RFS (HR, 0.46; P = 0.109), CSS (HR, 0.48; P = 0.233), and OS (HR, 0.40; P = 0.147).
Conclusion
Surgical approaches were not independently associated with prognosis in patients with locally advanced UTUC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Upper tract urothelial carcinoma (UTUC) is a relatively rare and heterogeneous disease accounting for approximately 5–10% of all urothelial tumors [1]. Open radical nephroureterectomy (ONU) with bladder-cuff excision remains the standard treatment modality for UTUC [2]. However, laparoscopic radical nephroureterectomy (LNU) has become popular as a minimally invasive surgical alternative. Several studies have compared the oncologic outcomes between ONU and LNU in patients with UTUC and have demonstrated comparative outcomes between these [2,3,4,5,6,7,8]. However, other studies have shown an increased risk of recurrence with LNU [9, 10]. Due to limitations of the heterogeneity of study population, sample size, and surgical techniques, the oncologic efficacy of LNU–ONU has remained undetermined. In addition, several guidelines have recommended laparoscopic procedures for localized (stage cT2 or less) disease [1, 11], but their use remains unclear for locally advanced (stages cT3-4 and/or cN+) disease. In this study, we compared the oncologic outcomes between ONU and LNU in a multi-institutional cohort of patients with locally advanced UTUC using inverse probability of treatment weighting (IPTW)-adjusted Cox-regression analyses.
Patients and methods
Design and ethics statement
The present retrospective, multicenter study was performed in accordance with the ethical standards of the Declaration of Helsinki and approved by the ethics review board of Hirosaki University School of Medicine (authorization numbers; 2017-089) and all the other participating hospitals.
Patient selection
Between February 1995 and February 2017, 426 adults underwent radical nephroureterectomy with bladder-cuff excision at the Hirosaki University Hospital, Aomori Rosai Hospital, Mutsu General Hospital, Tsugaru General Hospital, and Aomori Prefectural Central Hospital. We identified 229 (54%) patients with locally advanced disease (stages cT3-4 and/or cN+) and stratified the patients into two groups according to the surgical approach (ONU and LNU).
Evaluation of variables
The variables analyzed were age, sex, Eastern Cooperative Oncology Group performance status (ECOG-PS), smoking, clinical stage, renal function before radical nephroureterectomy, history of hypertension (HTN), cardiovascular disease (CVD), diabetes mellitus (DM), previous/concomitant bladder cancer, hydronephrosis, and tumor grade and location (renal pelvis, ureter, or multiple). Renal function was evaluated by estimated glomerular filtration rate (eGFR) before radical nephroureterectomy using a modified version of the abbreviated Modification of Diet in Renal Disease Study formula for Japanese patients [12]. Chronic kidney disease (CKD) was defined as a preoperative eGFR of < 60 mL/min/1.73 m2. Tumor stage and grade were assigned according to the 2009 Tumor, Nodes, and Metastasis (TNM) classification of the Union of International Cancer Control [13]. We used key findings of (1) irregularity of peri-ureteral/renal-pelvic fat or (2) presence of hydronephrosis to distinguish between cT2 and cT3 [14, 15]. Postoperative complications were evaluated using the Clavien–Dindo classification [16].
Neoadjuvant chemotherapy (NAC)
Since September 2006, we performed two-to-four courses of NAC for the treatment of locally advanced UTUC (stages cT3-4 and/or cN+) in selected patients. NAC comprised a platinum-based combination regimen using either gemcitabine plus cisplatin; gemcitabine plus carboplatin; or methotrexate, vinblastine, adriamycin, and cisplatin. Regimens were selected based on guidelines regarding the eligibility for the proper use of cisplatin [17] and on the patients’ overall status.
Surgical procedure
The schemas of incisions are shown in Fig. 1. ONU, which involved the removal of kidney, ureter, and ipsilateral bladder cuff, was performed via a retroperitoneal or transperitoneal approach [2] (Fig. 1a). All LNU procedures were performed via a retroperitoneal approach that was similar to the standard retroperitoneal laparoscopic nephroureterectomy with bladder-cuff resection [18]. Surgical indication for LNU was a nontumor invasion to the hilum. Four ports were applied in the procedure (Fig. 1b). Once a large retroperitoneal working space was available, the renal vessels were isolated and ligated and the kidney was completely mobilized. The ureter was dissected as distal as possible. Ureteral ligation was not performed. The incision was closed in two layers with interrupted stitches, and the patient was then placed in the supine position. A 6–8-cm oblique incision was made in the lower abdomen. The distal ureter was managed via the extravesical approach. The en bloc specimen was finally removed through the lower abdominal incision. A regional lymph-node dissection was not performed routinely during ONU and LNU. It was indicated only when an obvious lesion with imaging study was existed. We did not administer early (within 48 h) intravesical chemotherapy after nephroureterectomy.
Surgical approaches for upper tract urothelial carcinoma. ONU involved the removal of kidney, ureter, and ipsilateral bladder cuff and was performed via a transperitoneal (*) or retroperitoneal (**) approach (a). All LNU procedures were performed by a standard retroperitoneal laparoscopic nephroureterectomy with bladder-cuff resection. The surgical indication for LNU was nontumor invasion to the hilum. Four ports are applied in the procedure. After the renal vessels are isolated and ligated and the kidney is completely mobilized, the incision is closed in two layers with interrupted stitches. Thereafter, the patient is placed in the supine position, and a 6–8-cm oblique incision is made in the lower abdomen. The distal ureter is managed via the extravesical approach, and the en bloc specimen is finally removed through the lower abdominal incision (b). A regional lymph-node dissection was performed during ONU and LNU depending on the tumor stage
Patient follow-up
Oncologic follow-up after radical nephroureterectomy was performed according to the European Association of Urology guidelines [1] and the Japanese guidelines for UTUC [11] and bladder cancer [19]. Our follow-up protocol comprised complete blood counts, serum chemistry screenings, urine cytology, cystoscopy, ultrasound imaging of the abdomen, computed tomography, and chest radiography every 3–6 months (based on pathological findings) for at least 5 years [20, 21]. Adjuvant chemotherapy was not routinely administered. Salvage therapy was introduced when indicated by imaging.
Outcome evaluations
We evaluated pathological T and N stages, lymphovascular invasion (LVI), and surgical margins in the ONU and LNU groups. Oncologic outcomes, including intravesical recurrence-free survival (RFS), visceral RFS, cancer-specific survival (CSS), and overall survival (OS), were investigated for both the groups using the Kaplan–Meier method and compared using the log-rank test. Multivariate Cox-regression analysis was performed for independent predictors of intravesical RFS, visceral RFS, CSS, and OS. IPTW-adjusted Cox-proportional hazard regression analyses were performed to evaluate the impact of LNU on prognosis.
Statistical analysis
Statistical analyses of data were performed using SPSS version 24.0 (SPSS, Inc., Chicago, IL, USA), GraphPad Prism 5.03 (GraphPad Software, San Diego, CA, USA), and R 3.3.2 (The R Foundation for Statistical Computing, Vienna, Austria). Categorical variables were compared using the Fisher’s exact test or χ2 test. Quantitative variables were expressed as mean ± standard deviation or median with interquartile range. Differences between groups were statistically compared using the Student’s t test for data with a normal distribution or the Mann–Whitney U test for data with a non-normal distribution. P values of < 0.05 were considered statistically significant.
IPTW-adjusted Cox-proportional hazards regression analysis for patients with locally advanced disease (n = 229) and all patients (n = 426) were used to evaluate the impact of LNU on prognosis, which performs the reweighting of affected and unaffected groups to emulate a propensity score-matched population [22]. Hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated after controlling for potential confounders, including patient demographics and preoperative tumor variables. Variables included in the IPTW analysis were age, sex, ECOG- PS, HTN, CVD, DM, smoking, hydronephrosis, preoperative eGFR, cT and cN stage, tumor location, previous bladder cancer, NAC, and surgical margin.
Results
Baseline characteristics
Of the 426 patients, 351 (82%) and 75 (18%) underwent ONU and LNU, respectively. The median follow-up period was significantly shorter in the LNU group than in the ONU group (35 vs. 41 months, respectively; P = 0.001). There were significant differences in CVD, renal function, hydronephrosis, NAC, and surgical margins between the two groups (Table 1).
Of the 229 patients with locally advanced UTUC, 48 (21%) underwent LNU. The median follow-up period was not significantly different between the ONU and LNU groups (30 vs. 29 months, respectively; P = 0.199). However, there were significant differences in the backgrounds, including preoperative renal function, hydronephrosis, NAC, lymph-node involvement, LVI, and surgical margins, between the groups (Table 1). The number of locally advanced UTUC patients in each group who underwent LN dissection were 27/181 (15%) and 2/48 (4.2%), respectively. Only six (3.3%) and zero (0%) patients in the ONU and LNU groups, respectively, received adjuvant chemotherapy in our cohort.
Unadjusted oncologic outcomes
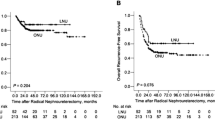
Before background adjustment, there were significant differences in intravesical RFS, visceral RFS, CSS, and OS between the ONU and LNU groups (n = 351 and 75, respectively) among all patients (Fig. 2) and between the groups (n = 181 and 48, respectively) among patients with locally advanced cancer (Fig. 3).
IPTW-adjusted Cox-regression analyses for intravesical RFS, visceral RFS, CSS, and OS
No significant differences were observed between the ONU and LNU groups in intravesical RFS (HR: 0.65, P = 0.476), visceral RFS (HR: 0.46, P = 0.109), CSS (HR: 0.48, P = 0.233), and OS (HR: 0.40, P = 0.147) in the IPTW-adjusted Cox-regression analyses for patients with locally advanced cancer (n = 229; Table 2, upper row) and in intravesical RFS (HR: 2.20, P = 0.119), visceral RFS (HR: 0.50, P = 0.157), CSS (HR: 0.32, P = 0.149), and OS (HR: 0.29, P = 0.080) for all patients (n = 426; Table 2, lower row).
Discussion
Our results demonstrate that the laparoscopic approach is used in 21% of the patients with locally advanced UTUC and the surgical approach is not independently associated with intravesical RFS, visceral RFS, CSS, and OS in patients with locally advanced UTUC using IPTW-adjusted Cox-regression analyses. These results are consistent with those of the previous reports [2,3,4,5,6,7,8]. In addition, background-adjusted IPTW analyses for all patients (n = 426) demonstrated that the laparoscopic approach was not a significant factor for intravesical RFS, visceral RFS, CSS, and OS. These results suggest that the impact of the laparoscopic approach was not independently associated with prognosis, but there might be a trend toward an association between the laparoscopic approach and prolonged OS.
Previous studies support our finding for oncologic benefits of LNU. In a meta-analysis of 21 observational studies of patients with UTUC who underwent ONU or LNU, LNU was reported to provide not different prognostic effects for UTUC and was associated with a better oncologic control of extravesical RFS and CSS than ONU [8]. The investigators reported pooled HRs of 1.05 (95% CI, 0.92–1.18; P = 0.134) for intravesical RFS between the LNU (n = 1959) and ONU (n = 4281) groups, 0.80 (95% CI, 0.64–0.96; P = 0.859) for extravesical RFS (n = 836 and 4315, respectively), 0.79 (95% CI, 0.68–0.91; P = 0.186) for CSS (n = 2518 and 8342, respectively), and 0.91 (95% CI, 0.66–1.17; P = 0.091) for OS (n = 1442 and 3119, respectively). A recent study from the Multi-institutional National Database of the Japanese Urological Association including 749 patients with stage pT2 ≥ cNxM0 disease who underwent LNU (n = 222) or ONU (n = 527) has suggested no significant differences between the two groups in RFS, CSS, and OS. In addition, OS was not significantly different between the two groups even when the patients were stratified by stage pT3/pT4 and/or pN + disease (P = 0.2876). They concluded that there is no evidence that oncologic outcomes of LNU are inferior to those of ONU in muscle-invasive UTUC when appropriate patients are selected [2]. In contrast, a multicenter analysis of 849 patients with UTUC who underwent LNU (n = 446, 53%) or ONU (n = 403, 47%) at the Canadian Upper Tract Collaboration demonstrated that the surgical approach is not independently associated with OS (HR, 0.89; 95% CI, 0.63–1.27; P = 0.52) and disease-specific survival (HR, 0.90; 95% CI, 0.60–1.37; P = 0.64), but there is a trend toward an independent association between LNU and poor RFS (HR, 1.24; 95% CI, 0.98–1.57, P = 0.08) [7]. A single center study from Seoul National University has suggested that LNU is independently associated with worse 5-year CSS (66.1% vs. 80.2%; P = 0.015) and OS (59.1% vs. 75.2%; P = 0.027) rates than those of ONU [10]. Multivariate analyses have shown that LNU is significantly associated with poor CSS (HR, 2.50; P = 0.005) and OS (HR, 2.59; P = 0.001) in patients with stage pT3/pT4 disease [10]. However, considering these studies with the intrinsic bias of retrospective study design, the results should be interpreted with caution. In the first randomized prospective study, progression-free survival and CSS were superior in the ONU group (n = 9) than in the LNU group (n = 7) among patients with stage pT3 tumors, whereas prognosis in all patients (n = 80) was not significantly different between the groups (n = 40 and 40, respectively) [4]. However, the number of patients in the stage pT3 subgroup was too small to conclude the impact of LNU on prognosis, even if it was conducted as a randomized prospective study. Taken together, as large multicenter studies [2,3,4,5,6,7,8, 23] and our results suggest, LNU could provide not different prognostic effects for UTUC as does ONU.
Regarding survival outcomes in all patients (n = 426), our data showed that LNU and had comparable intravesical RFS, visceral RFS, CSS, and OS. Trend of OS improvement (P = 0.080) in LNU may be due to selection bias, such as for small, low-grade, noninvasive tumors, particularly for the primary experience of LNU. The number of patients with stage cT3-4 disease who underwent LNU was significantly different between the years 2006 and 2008 (2/13; 15%) and the years 2009 and 2017 (46/62; 74%; P < 0.001), because LNU for UTUC was started in April 2006. This result suggests the unavoidable clinical trend toward an association between locally advanced UTUC and an open approach. In addition, the number of patients with NAC was significantly higher in the LNU group, because NAC for locally advanced UTUC was started in March 2008. Our previous study suggested an oncologic benefit of NAC for locally advanced UTUC [24, 25]. Therefore, we could not exclude selection bias between LNU and ONU even if we used matching methods for adjustment, and the result should be interpreted with caution.
The influence of NAC use for selection of less invasive surgery might be not ignored. In the present study, the number of locally advanced UTUC patients who underwent NAC and laparoscopic surgery were significantly higher (n = 31/69; 31%) than those without NAC (n = 17/129; 13%) (P = 0.001). Our results might suggest the clinical benefit of laparoscopic surgery in combination with NAC for locally advanced UTUC. However, strong selection biases are existing in this retrospective study, and further studies are necessary to address this issue.
Several limitations of the present study must be acknowledged. First, the use of data from multiple centers and the retrospective study design prevented us from making definitive conclusions regarding the impact of LNU on prognosis. We could not address median numbers of the resected LNs in each approach due to the lack of data, and the differences in surgical procedures including approaches (transperitoneal or retroperitoneal) and positions (supine or lateral position). In addition, positive surgical margin was significantly more frequent in the ONU group than in LNU group because of selection biases for surgical approaches. Despite the use of an IPTW method, which is an attractive method for estimating treatment effects using observational data, we were unable to control for selection bias and other unmeasurable confounders of retrospective studies. Despite these limitations, we evaluated the direct impact of LNU on oncologic outcomes in patients with UTUC using IPTW-adjusted Cox-regression analyses. Because only a single prospective study was available to compare the influence of surgical approach on prognosis, our results support the rationale that the laparoscopic approach provides a not different value on the prognosis for locally advanced UTUC.
In conclusion, a surgical approach was not independently associated with intravesical RFS, visceral RFS, CSS, and OS in patients with locally advanced UTUC. Although we could not exclude selection bias, the laparoscopic approach might result in a non-inferior prognosis compared with the open approach in patients with UTUC. More large-scale prospective randomized controlled study is required to assess the impact of the laparoscopic approach on the prognosis of UTUC.
Abbreviations
- UTUC:
-
Upper tract urothelial carcinoma
- ONU:
-
Open radical nephroureterectomy
- LNU:
-
Laparoscopic radical nephroureterectomy
- IPTW:
-
Inverse probability of treatment weighting
- ECOG-PS:
-
Eastern Cooperative Oncology Group performance status
- HTN:
-
Hypertension
- CVD:
-
Cardiovascular disease
- DM:
-
Diabetes mellitus
- eGFR:
-
Estimated glomerular filtration rate
- CKD:
-
Chronic kidney disease
- NAC:
-
Neoadjuvant chemotherapy
- LVI:
-
Lymphovascular invasion
- RFS:
-
Recurrence-free survival
- CSS:
-
Cancer-specific survival
- OS:
-
Overall survival
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
References
Roupret M, Babjuk M, Comperat E et al (2015) European Association of Urology Guidelines on Upper Urinary Tract Urothelial Cell Carcinoma: 2015 Update. Eur Urol 68(5):868–879. https://doi.org/10.1016/j.eururo.2015.06.044
Miyazaki J, Nishiyama H, Fujimoto H et al (2016) Laparoscopic versus open nephroureterectomy in muscle-invasive upper tract urothelial carcinoma: subanalysis of the Multi-Institutional National Database of the Japanese Urological Association. J Endourol 30(5):520–525. https://doi.org/10.1089/end.2015.0757
Capitanio U, Shariat SF, Isbarn H et al (2009) Comparison of oncologic outcomes for open and laparoscopic nephroureterectomy: a multi-institutional analysis of 1249 cases. Eur Urol 56(1):1–9. https://doi.org/10.1016/j.eururo.2009.03.072
Simone G, Papalia R, Guaglianone S et al (2009) Laparoscopic versus open nephroureterectomy: perioperative and oncologic outcomes from a randomised prospective study. Eur Urol 56(3):520–526. https://doi.org/10.1016/j.eururo.2009.06.013
Ariane MM, Colin P, Ouzzane A et al (2012) Assessment of oncologic control obtained after open versus laparoscopic nephroureterectomy for upper urinary tract urothelial carcinomas (UUT-UCs): results from a large French multicenter collaborative study. Ann Surg Oncol 19(1):301–308. https://doi.org/10.1245/s10434-011-1841-x
Ni S, Tao W, Chen Q et al (2012) Laparoscopic versus open nephroureterectomy for the treatment of upper urinary tract urothelial carcinoma: a systematic review and cumulative analysis of comparative studies. Eur Urol 61(6):1142–1153. https://doi.org/10.1016/j.eururo.2012.02.019
Fairey AS, Kassouf W, Estey E et al (2013) Comparison of oncological outcomes for open and laparoscopic radical nephroureterectomy: results from the Canadian Upper Tract Collaboration. BJU Int 112(6):791–797. https://doi.org/10.1111/j.1464-410X.2012.11474.x
Zhang S, Luo Y, Wang C et al (2016) Long-term oncologic outcomes of laparoscopic nephroureterectomy versus open nephroureterectomy for upper tract urothelial carcinoma: a systematic review and meta-analysis. PeerJ 4:e2063. https://doi.org/10.7717/peerj.2063
Kamihira O, Hattori R, Yamaguchi A et al (2009) Laparoscopic radical nephroureterectomy: a multicenter analysis in Japan. Eur Urol 55(6):1397–1407. https://doi.org/10.1016/j.eururo.2009.03.003
Kim HS, Ku JH, Jeong CW et al (2016) Laparoscopic radical nephroureterectomy is associated with worse survival outcomes than open radical nephroureterectomy in patients with locally advanced upper tract urothelial carcinoma. World J Urol 34(6):859–869. https://doi.org/10.1007/s00345-015-1712-3
Oya M, Kikuchi E (2015) Evidenced-based clinical practice guideline for upper tract urothelial carcinoma (summary–Japanese Urological Association, 2014 edition). Int J Urol 22(1):3–13. https://doi.org/10.1111/iju.12630
Matsuo S, Imai E, Horio M et al (2009) Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis 53(6):982–992. https://doi.org/10.1053/j.ajkd.2008.12.034
Sobin LH, Gospodarowicz MK, Wittekind C, International Union against Cancer., ebrary Inc. (2009) TNM classification of malignant tumours. 7th edn. Wiley-Blackwell, Chichester, West Sussex, UK; Hoboken, NJ
Messer JC, Terrell JD, Herman MP et al (2013) Multi-institutional validation of the ability of preoperative hydronephrosis to predict advanced pathologic tumor stage in upper-tract urothelial carcinoma. Urol Oncol 31(6):904–908. https://doi.org/10.1016/j.urolonc.2011.07.011
Ito Y, Kikuchi E, Tanaka N et al (2011) Preoperative hydronephrosis grade independently predicts worse pathological outcomes in patients undergoing nephroureterectomy for upper tract urothelial carcinoma. J Urol 185(5):1621–1626. https://doi.org/10.1016/j.juro.2010.12.035
Clavien PA, Barkun J, de Oliveira ML et al (2009) The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 250(2):187–196. https://doi.org/10.1097/SLA.0b013e3181b13ca2
Galsky MD, Hahn NM, Rosenberg J et al (2011) A consensus definition of patients with metastatic urothelial carcinoma who are unfit for cisplatin-based chemotherapy. Lancet Oncol 12(3):211–214. https://doi.org/10.1016/S1470-2045(10)70275-8
Fang Z, Li L, Wang X et al (2014) Total retroperitoneal laparoscopic nephroureterectomy with bladder-cuff resection for upper urinary tract transitional cell carcinoma. J Invest Surg 27(6):354–359. https://doi.org/10.3109/08941939.2014.930214
Kubota Y, Nakaigawa N, Committee for Establishment of the Clinical Practice Guideline for the Management of Bladder C, the Japanese Urological A (2016) Essential content of evidence-based clinical practice guidelines for bladder cancer: the Japanese Urological Association 2015 update. Int J Urol 23(8):640–645. https://doi.org/10.1111/iju.13141
Kusaka A, Hatakeyama S, Hosogoe S et al (2017) Risk-stratified surveillance and cost effectiveness of follow-up after radical cystectomy in patients with muscle-invasive bladder cancer. Oncotarget. https://doi.org/10.18632/oncotarget.19043
Kusaka A, Hatakeyama S, Hosogoe S et al (2017) Detecting asymptomatic recurrence after radical cystectomy contributes to better prognosis in patients with muscle-invasive bladder cancer. Med Oncol 34(5):90. https://doi.org/10.1007/s12032-017-0955-9
Austin PC, Stuart EA (2015) Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat Med 34(28):3661–3679. https://doi.org/10.1002/sim.6607
Walton TJ, Novara G, Matsumoto K et al (2011) Oncological outcomes after laparoscopic and open radical nephroureterectomy: results from an international cohort. BJU Int 108(3):406–412. https://doi.org/10.1111/j.1464-410X.2010.09826.x
Hosogoe S, Hatakeyama S, Kusaka A et al (2017) Platinum-based Neoadjuvant Chemotherapy Improves Oncological Outcomes in Patients with Locally Advanced Upper Tract Urothelial Carcinoma. Eur Urol Focus. https://doi.org/10.1016/j.euf.2017.03.013
Kubota Y, Hatakeyama S, Tanaka T et al (2017) Oncological outcomes of neoadjuvant chemotherapy in patients with locally advanced upper tract urothelial carcinoma: a multicenter study. Oncotarget. https://doi.org/10.18632/oncotarget.21551
Acknowledgements
The authors would like to thank Hiroyuki Ito, Kazuaki Yoshikawa, Atsushi Sasaki, Hiromichi Iwamura, Ken Fukushi, Jotaro Mikami, Takuma Narita, Toshikazu Tanaka, Itsuto Hamano, Yuki Fujita, Yukie Nishizawa, Satomi Sakamoto, and Yuriko Tanabe for their invaluable help with data collection. The authors would also like to thank Enago (www.enago.jp) for the English language review.
Author information
Authors and Affiliations
Contributions
Conception and design: Shingo Hatakeyama. Acquisition of data: Koichi Kido, Naoki Fujita, Shingo Hatakeyama, Takahiro Yoneyama, Yasuhiro Hashimoto, Takuya Koie, Ikuya Iwabuchi, Masaru Ogasawara, Toshiaki Kawaguchi. Analysis and interpretation of data: Shingo Hatakeyama, Yuki Tobisawa, Tohru Yoneyama. Drafting of the manuscript: Koichi Kido, Shingo Hatakeyama. Critical revision of the manuscript: Naoki Fujita, Takuya Koie, Chikara Ohyama. Statistical analysis: Shingo Hatakeyama, Tohru Yoneyama. Obtaining funding: Shingo Hatakeyama, Takuya Koie, Chikara Ohyama. Administrative, technical, or material support: Yuki Tobisawa, Tohru Yoneyama
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflicts of interest.
Financial disclosure
This work was supported by a Grant-in-Aid for Scientific Research (No. 15H02563, 15K15579, 17K11118, 17K11119, 17K16768, 17K16770, and 17K16771) from the Japan Society for the Promotion of Science.
Ethical standards
This study was performed in accordance with the ethical standards of the Declaration of Helsinki and approved by an ethics review board of Hirosaki University School of Medicine (authorization numbers; 2017–089) and all other hospitals.
Informed consent
For this type of study, formal written consent is not required. Pursuant to the provisions of the ethics committee and the ethic guideline in Japan, written consent was not required in exchange for public disclosure of study information in the case of retrospective and/or observational study using a material such as the existing documentation. The study information was open for the public consumption at http://www.med.hirosaki-u.ac.jp/~uro/html/IRB/IRBdoc.html.
About this article
Cite this article
Kido, K., Hatakeyama, S., Fujita, N. et al. Oncologic outcomes for open and laparoscopic radical nephroureterectomy in patients with upper tract urothelial carcinoma. Int J Clin Oncol 23, 726–733 (2018). https://doi.org/10.1007/s10147-018-1248-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-018-1248-9