Abstract
Spinal hemangioblastomas constitute 1.6–5.8% of all spinal cord tumors. Microsurgical excision of these tumors is challenging. The purpose of this study is to analyze the neurological improvement and long-term functional outcome of spinal hemangioblastomas. This retrospective study included 15 patients who underwent surgery for intramedullary spinal hemangioblastoma at the Department of Neurosurgery of Sri Chitra Tirunal Institute for Medical Sciences and Technology from January 2001 to June 2014. Eight patients (53%) were diagnosed to have von Hippel-Lindau (vHL) disease. Eight (53%) of them were females, and seven were males (47%). Mean age was 33.8 years (16–55 years). Duration of illness ranged from 2 weeks to 4 years, and average duration was 10.5 months. Most common symptom was motor weakness followed by sensory disturbances, pain, and bladder incontinence. Six (85.7%) sporadic spinal hemangioblastomas were in McCormick grade I; whereas, 7 (87.5%) of vHL spinal hemangioblastomas were in grade II or above. In the immediate postoperative period, three patients noticed improvement in their motor weakness. Six patients (40%) experienced deterioration of preoperative neurological status in the immediate postoperative period. Three of them were sporadic tumors, and others had vHL syndrome. Favorable long-term outcome was achieved in 80% of cases. Though neurological deterioration is common after surgical resection of spinal hemangioblastomas, majority of them are reversible. Long-term functional outcome is favorable with minimal postoperative morbidities. Both sporadic- and vHL-associated tumors share common clinical and radiological features, and neurological outcome is equally good in both.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spinal hemangioblastomas are very rare and constitute 1.6–5.8% of all spinal cord tumors and 1–2.5% of all tumors of the central nervous system [4, 23]. They can present as sporadic lesions or in association with von Hippel-Lindau syndrome (vHL syndrome). Sporadic tumors constitute for majority of the cases, up to 70–80% [13]. Microsurgical excision has long been established as the standard of care for spinal hemangioblastomas [15]. Though surgical outcome of spinal hemangioblastomas has been improved as a result of advances microsurgical techniques, postoperative neurological deterioration is still common and reported in 50–80% of cases with permanent neurological deficits up to 10% [13,14,15,16,17,18]. Few studies directly compared the sporadic- and vHL-associated spinal hemangioblastomas in terms of clinical presentation, tumor characters, and neurological outcome. Management of asymptomatic tumors associated with vHL syndrome is debatable. The natural history of asymptomatic spinal tumors is not clearly understood because of the rarity of these tumors. The purpose of this study is to analyze the clinical presentation, surgical management, and long-term functional outcome of patients with sporadic- and vHL-associated spinal intramedullary hemangioblastomas.
Materials and methods
This retrospective study included 15 patients who underwent surgery for intramedullary spinal hemangioblastoma at the Department of Neurosurgery of Sri Chitra Tirunal Institute for Medical Sciences and Technology from January 2001 to June 2014. Symptoms at presentation, neurological deficits, neuroradiological features, intraoperative findings, and postoperative course were recorded prospectively. Preoperatively, all the patients underwent contrast-enhanced MRI. Spinal angiogram was done in selected patients. They also underwent screening for other manifestations of vHL syndrome including intracranial tumors, retinal angiomas, pancreatic and renal masses, and pheochromocytoma.
Clinical criteria proposed by Melmon have been used to identify patients with vHL syndrome [20]. Neurological function of the patients was graded according to the clinical scale described by McCormick [17]. Functional grade was assessed in the pre- and immediate postoperative periods, and at follow-up. Functional outcome was compared between the sporadic- and vHL-associated cases.
Results
Clinical presentation
Among 15 patients, eight (53%) of them were females, and seven were males (47%). Mean age was 33.8 years (16–55 years). Mean age of patients with vHL disease at the time of presentation was 32.5 years. Sporadic tumor was more common in female (71%); whereas, vHL tumors were more common in males (63%). Duration of illness ranged from 2 weeks to 4 years, and average duration was 10.5 months. Thirteen patients (87%) presented motor weakness in the form of hemiparesis, quadriparesis, or paraparesis at the time of presentation. Nine patients (60%) present with sensory disturbances. Pain was the presenting symptom in 4 patients (27%). Bladder disturbance was present in 3 patients (20%). Table 1 shows the clinical presentation of all the patients.
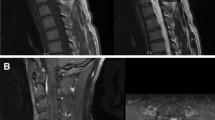
Fifteen patients had 23 spinal tumors. Thirteen tumors (57%) present in the thoracic spinal cord, and 9 (39%) in the cervical cord. One (4%) was present at the conus. Spinal hemangioblastomas had predilection for thoracic region in vHL subjects (6/8, 75%); whereas, only 2 lesions (28.6%) were found in thoracic spine in sporadic hemangioblastoma group (Fig 1).
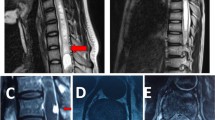
Five patients (33%) had cerebellar hemangioblastoma associated with spinal cord tumor. Seven patients (47%) had associated renal and pancreatic cysts. Two patients (13%) had retinal disease. Based on the clinical criteria for vHL disease, 8 patients (53%) were diagnosed to have vHL disease. Of the 8, one had positive family history. Syrinx was present in 10 patients (67%). Eight tumors were asymptomatic and coexisted with the symptomatic tumors. Preoperative angiogram was done in 11 patients. Preoperatively, two patients underwent embolization to reduce the blood loss during surgery. One had tumor in the conus, and the other had tumor in the dorsal cord (Fig 2).
McCormick functional grade was assessed before surgery, at the time of discharge, and at follow-up. Six (85.7%) cases of sporadic spinal hemangioblastomas were in grade I; whereas, 7 (87.5%) of vHL spinal hemangioblastomas were in grade II or above. Table 2 summarizes the preoperative functional grades in all patients.
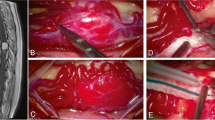
Total of 15 patients underwent 19 surgeries, of which 16 for spinal hemangioblastomas and 3 for cerebellar hemangioblastomas. One patient was operated twice for cerebellar hemangioblastoma on either side, and one patient underwent surgery for right cerebellar hemangioblastoma. One patient had simultaneous resection of both cerebellar and cervical hemangioblastomas. All the patients underwent posterior spinal laminectomy and posterior midline approach for the resection of spinal tumors. Laminectomy was limited to the level of the solid portion of the tumor. All patients underwent complete microscopic resection of the tumor. None of the patients required instrumentation.
Postoperative course
Fifteen patients underwent 16 surgeries for spinal cord tumor including reoperation for a recurrent tumor. In the immediate postoperative period, three patients noticed improvement in their motor weakness. Among them, 2 had vHL syndrome, and one had sporadic tumor. Six were stable neurologically in the immediate postoperative period. Six patients (40%) experienced deterioration of preoperative neurological status in the immediate postoperative period. Three of them were sporadic tumors, and others had vHL syndrome. Three of them had tumor in the cervical cord, and three in the dorsal cord. Two patients were grade II, and the remaining four were grade I preoperatively. In 5 patients, neurological deterioration was transient and regained the preoperative functional grade at the time of discharge, and remaining one with vHL did not show any improvement.
Sensory symptoms in the remaining patients improved postoperatively. They also noticed improvement in the spasticity and motor weakness. All patients had improvement in pain symptom. Non-neurological complications were rare postoperatively. None of the patients had wound infection in the postoperative period. Only one patient had CSF leak from the wound in the postoperative period. It was managed by resuturing the wound. We had one mortality (6.7%) in the postoperative period (patient no 4). He developed massive intraventricular bleed due to malignant hypertension in the postoperative period.
Long-term follow-up
Mean follow-up was 5 years, ranging from 6 months to 11 years. Seven patients had more than 5 years of follow-up. Minimum follow-up was 6 months. During follow-up, 12 patients (80%) experienced improvement in their preoperative neurological deficit among the surviving 14 patients. Eleven patients (73%) were functionally grade I, and one was grade II. All patients with sporadic tumors (100%) noticed improvement in functional grade. Among patients with vHL syndrome, five (71%) of them noticed improvement in functional grade, and two (29%) had poor functional outcome.
Among the remaining 2 patients, one did not show improvement in functional grade. The other patient experienced deterioration in functional grade after 3 years (grade I1 to grade III) due to recurrence at the operative site though she had improvement in preoperative neurological deficit after the initial surgery. Postoperatively, she had further deterioration in her neurological deficit. Both of them had tumors in thoracic cord and were associated with vHL syndrome. These two patients had some improvement in their lower limb power following surgery, but they remained functionally dependent (McCormick grade III). Both of them had improvement in their bladder function and became free of indwelling urinary catheter.
Recurrence of the tumor was noticed at the operative site in one patient (6.7%) at 3 years of follow-up, although follow-up MRI was negative for residual lesion. She also developed another new lesion apart from the operative site and pancreatic cysts. She underwent re-exploration and excision of the recurrent tumor. Radical tumor excision was achieved, but she developed paraplegia postoperatively (preoperative functional grade II). Syrinx either resolved completely or decreased in size in the follow-up MRI in all patients who had syrinx preoperatively. None of our patient required further intervention for syrinx.
Discussion
Clinical features
Hemangioblastomas are benign and highly vascular tumors. Spinal hemangioblastoma represents 2 to 15% of primary intramedullary spinal cord tumors [24, 12]. Sporadic hemangioblastomas are always solitary. Those associated with vHL disease are often multiple and associated with tumors in other locations especially cerebellum, brainstem, and retina [26]. Majority of these tumors are intramedullary reaching the pial surface dorsally. They are often seen in association with dorsal rootlets. They can have intra- and extramedullary components. They can also be intradural extramedullary or even extradural in location.
Spinal hemangioblastomas are more common in males with male to female ratio of 1.6:1 to 5.5:1 [26]. In our series, overall incidence is slightly higher in females, but vHL-associated spinal tumors are more common in males as compared to sporadic tumor. These tumors commonly occur in the third or fourth decades, and vHL syndrome-related tumors occur in second or third decades of life. Though vHL-related spinal tumor is commonly reported in younger age as compared to sporadic tumors, symptomatic tumors can occur between 12 to 66 years of life [5]. In our series, majority of vHL cases occurred in middle age (> 35 years) as compared to sporadic tumors.
The clinical presentation depends on the location of the tumor and presence of syrinx, cyst, or edema. More than 95% of patients who are symptomatic have syrinx, edema, or cyst along with the tumor [3, 19]. Common symptoms include sensory or motor deficits, pain, dysesthesias, and bowel and urinary dysfunctions. Previous studies have reported sensory dysfunction as the most prevalent first clinical symptom [12, 14, 19]. This has been explained by the dorsal location of the tumor close to posterior column and dorsal rootlets in the spinal cord. In some series, pain was the most common symptom [9, 27]. In our series, motor weakness was the predominant symptom (86%). Apart from the direct mass effect of the tumor, presence of pseudo cyst, edema, or syrinx also contributes to the development of symptoms. This may explain the predominant motor symptoms in our series. The duration of symptoms ranges from 2 weeks to 4 years.
Contrast-enhanced MRI is the diagnostic modality of choice for spinal hemangioblastoma. Radiologically, both sporadic- and vHL-associated tumors share similar features. The most common location of hemangioblastoma in the spinal cord is cervical, thoracic, and lumbar region in the descending order of frequency in most of the studies [11, 18]. In our series, the most common site of the tumor was thoracic spinal cord (57%) followed by cervical cord (39%). Tumors associated with vHL syndrome had tendency to occur in thoracic region in our study. Kanno H et al. [13] also found that the thoracic cord was the more frequent site of tumors in vHL patients, but larger series have not shown such predilection [13, 15, 19, 21]. Incidence of syrinx and cyst component in the tumor has been reported to be between 50 and 70% [2, 6, 7, 25]. In our series, tumor-associated syrinx was seen in 66.7% of cases.
Angiography is not necessary for diagnosis and rarely required preoperatively. Preoperative embolization of feeding vessel reduces the tumor vascularity. It helps in reducing the intraoperative blood loss and thereby to achieve safe removal [11, 29]. Though preoperative embolization is not necessary for complete tumor removal, it can be considered as an adjunct to microsurgery especially in patients with large tumors and tumors in critical location like conus.
In agreement with literature, we did not find significant difference in clinical and radiological features between sporadic cases and vHL syndrome [9, 17, 30]. Though many patients with vHL syndrome in our series had significantly lower functional grade at the time of presentation as compared to sporadic cases, previous studies have not reported such a differential distribution of functional grade among vHL and sporadic cases [9, 13, 15, 17, 19, 21].
Neurological outcome
Several authors have shown good neurological outcome after surgical resection of hemangioblastoma [8, 11, 12, 14, 19, 22, 23, 27, 30]. Previous studies reported deterioration in the neurological status as high as 60% in the immediate postoperative period [12, 19]. But these are usually transient and reversible over a period of few weeks to 6 months. The incidence of permanent neurological worsening is rather low (< 10%) [13,14,15,16,17,18]. Although we had deterioration in neurological status postoperatively in 6 patients (40%), many of them were transient and showed significant improvement in the follow-up. Overall, good neurological outcome was achieved in 80% patients. All of the sporadic cases had good functional outcome. Among the patients with vHL disease, favorable long-term functional outcome was achieved in 71%.
Microsurgery for spinal hemangioblastoma has been associated with low morbidity. The long-term surgical outcome is favorable with stable or improvement in neurological status. Studies have shown favorable outcome in 68–96% following microsurgery [19, 27]. Our results are consistent with other series. In our series, patients showed improvement in preoperative functional grade by one or two grades. With timely surgery, even patients with poor functional grade noticed improvement in neurological deficit and could become functionally independent.
Neurological outcome of sporadic and vHL syndrome-related spinal tumors has also been compared in previous studies. Many studies showed surgical outcome of vHL-related spinal tumors was not different from that of sporadic tumors [30]. In contrary, Siller et al. [28] concluded vHL disease itself is a risk factor for a worse long-term prognosis. Though patients with vHL syndrome had and lower functional grade preoperatively in our study, long-term neurological outcome is equally good in both vHL and sporadic tumors. Three of the four patients with McCormick’s grade III had shown good surgical outcome. Only one patient with grade III had poor outcome. Presence of multiple tumors in spinal cord did not affect the long-term outcome. Only multiple resections of tumors at the same site in vHL syndrome may result in poor neurological outcome as in our patient who had recurrence at the previously operated site.
Several studies have tried to find out the factors which could predict the neurological outcome of spinal hemangioblastomas, but the results were not consistent in all studies [15, 19, 30]. Recently in a large study with 108 patients, Mehta and colleagues found out that tumor location and resection of completely intramedullary tumor correlated with postoperative neurological outcome using multivariate analysis [19]. Other factors like tumor volume, preoperative functional grade, presence of syrinx, and peritumoral edema did not predict the outcome.
Patients with vHL syndrome usually have multiple hemangioblastomas in the spinal cord. These can also be found in cerebellum or brainstem along with spinal tumors. All the tumors do not warrant surgical excision. The indication for surgical removal of asymptomatic tumors is still debated. Hemangioblastomas show a period of growth followed by a phase of quiescence [1]. There are no reliable threshold tumor size and threshold growth rate that could predict the evolution of symptoms or cyst [31]. In their study on 19 patients, Ammerman et al. [1] advised against the surgical resection of asymptomatic spinal cord tumor based only on radiographic progression. Such an approach could result in additional procedures which may not be required at all during follow-up. In our series, eight patients with vHL syndrome had 16 spinal cord tumors. Eight tumors remained asymptomatic and did not show radiological progression. None of them required surgical resection during follow-up.
Recurrence of hemangioblastoma is rare and usually seen in patients with vHL syndrome as in our study [10]. vHL patients should undergo screening for associated systemic tumors. Screening should include both radiological and biochemical evaluation. Urinary catecholamines should be assessed in all patients preoperatively, though pheochromocytoma associated with vHL is less likely to have symptoms or biochemical evidence of catecholamine production than sporadic cases [3]. Undiagnosed pheochromocytoma may result in grave complications in intraoperative as well as postoperative period as in our study.
Conclusion
Microsurgical resection of spinal hemangioblastomas is associated with good neurological outcome irrespective of preoperative functional grade. Radical tumor resection should be the surgical goal as most of the immediate postoperative neurological deteriorations are reversible. Age, sex, duration of illness, tumor location, volume, and preoperative functional grade do not predict the postoperative neurological improvement. There is no difference in the long-term neurological outcome between sporadic- and vHL-associated spinal hemangioblastomas. Asymptomatic spinal tumors in vHL syndrome can be safely followed without additional intervention. Appearance of symptoms should be the indication for surgical removal of asymptomatic spinal hemangioblastomas.
References
Ammerman JM, Lonser RR, Dambrosia J, Butman JA, Oldfield EH (2006) Long-term natural history of hemangioblastomas in patients with von Hippel–Lindau disease: implications for treatment. J Neurosurg 105:248–255
Baker KB, Moran CJ, Wippold FJ 2nd, Smirniotopoulos JG, Rodriguez FJ, Meyers SP, Siegal TL (2000) MR imaging of spinal hemangioblastoma. Am J Roentgenol 174:377–382
Beitner MM, Winship I, Drummond KJ (2011) Neurosurgical consideration in von Hippel Lindau disease. J Clin Neurosci 18:171–180
Browne TR, Adams RD, Robertson GH (1976) Hemangioblastoma of the spinal cord. Arch Neurol 33:435–441
Butman AJ, Linehan WM, Lonser RR (2008) Neurologic manifestations of von Hippel Lindau’s disease. JAMA 300:1334–1342
Choyke PL, Glenn GM, Walther MM, Patronas NJ, Linehan WM, Zbar B (1995) Von Hippel–Lindau disease: genetic, clinical, and imaging features. Radiology 194:629–642
Chu BC, Terae S, Hida K, Furukawa M, Abe S, Miyasaka K (2001) MR findings in spinal hemangioblastoma: correlation with symptoms and with angiographic and surgical findings. Am J Neuroradiol 22:206–217
Conway JE, Chou D, Clatterbuck RE, Brem H, Long DM, Rigamonti D (2001) Hemangioblastomas of the central nervous system in von Hippel–Lindau syndrome and sporadic disease. Neurosurgery 48:55–62
Cristante L, Herrmann HD (1999) Surgical management of intramedullary hemangioblastoma of the spinal cord. Acta Neurochir 141:333–440
de la Monte SM, Horowitz SA (1989) Hemangioblastomas: clinical and histopathological factors correlated with recurrence. Neurosurgery 25:695–698
Eskridge JM, McAuliffe W, Harris B, Kim DK, Scott J, Winn HR (1996) Preoperative endovascular embolisation of craniospinal hemangioblastomas. Am J Neuroradiol 17:525–531
Jia D, Tang B, Shi Y, Wang J, Sun Z, Chen Z, Zhang L, Xia K, Jiang H (2013) A deletion mutation of the VHL gene associated with a patient with sporadic von Hippel-Lindau disease. J Clin Neurosci 20:842–847
Kanno H, Yamamoto I, Nishikawa R, Matsutani M, Wakabayashi T, Yoshida J, Shitara N, Yamasaki I, Shuin T (2009) Spinal cord hemangioblastomas in von Hippel-Lindau disease. Spinal Cord 47:447–452
Lonser RR, Oldfield EH (2005) Microsurgical resection of spinal cord hemangioblastomas. Neurosurgery 57:372–376
Lonser RR, Weil RJ, Wanebo JE, DeVroom HL, Oldfield EH (2003) Surgical management of spinal cord hemangioblastomas in patients with von Hippel-Lindau disease. J Neurosurg 98:106–116
Malis LI (2002) Atraumatic bloodless removal of intramedullary hemangioblastomas of the spinal cord. J Neurosurg 97:1–6
Mandigo CE, Ogden AT, Angevine PD, McCormick PC (2009) Operative management of spinal hemangioblastoma. Neurosurgery 65:1166–1177
McCormick PC, Torres R, Post KD, Stein BM (1990) Intramedullary ependymoma of the spinal cord. J Neurosurg 72:523–532
Mehta GU, Asthagiri AR, Bakhtian KD, Auh S, Oldfield EH, Lonser RR (2010) Functional outcome after resection of spinal cord hemangioblastomas associated with von Hippel–Lindau disease. J Neurosurg Spine 12:233–242
Melmon K, Rosen S (1964) Lindau’s disease. Review of the literature and study of a large kindred. Am J Med 36:595–617
Neumann HP, Eggert HR, Weigel K, Friedburg H, Wiestler OD, Schollmeyer P (1989) Hemangioblastomas of the central nervous system: a 10-year study with special reference to von Hippel-Lindau syndrome. J Neurosurg 70:24–30
Niemelä M, Lemeta S, Summanen P, Böhling T, Sainio M, Kere J, Poussa K, Sankila R, Haapasalo H, Kääriäinen H, Pukkala E, Jääskeläinen J (1999) Long-term prognosis of haemangioblastoma of the CNS: impact of von Hippel–Lindau disease. Acta Neurochir 141:1147–1156
Pietila TA, Stendel R, Schilling A, Krznaric I, Brock M (2000) Surgical treatment of spinal hemangioblastomas. Acta Neurochir 142:879–886
Plate KH, Vortmeyer AO, Zagzag D, Neumann HPH (2007) Von Hippel-Lindau disease and haemangioblastoma. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (eds) WHO Classification of Tumours of the Central Nervous System, 4th edn. IARC, Lyon, pp 215–217
Qi-Wu X, Wei-Min B, Ren- Ling M, Guo-Yuan Y (1994) Magnetic resonance imaging and microsurgical treatment of intramedullary hemangioblastoma of the spinal cord. Neurosurgery 35(4):671–676
Rachinger J, Buslei R, Prell J, Strauss C (2009) Solid hemangioblastomas of the CNS. A review of 17 consecutive cases. Neurosurg Rev 32:37–48
Roonprapunt C, Silvera VM, Setton A (2001) Surgical management of isolated hemangioblastomas of the spinal cord. Neurosurgery 49:321–327
Siller S, Szelényi A, Herlitz L, Tonn JC, Zausinger S (2017) Spinal cord hemangioblastomas: significance of intraoperative neurophysiological monitoring for resection and long-term outcome. J Neurosurg Spine 26:483–493
Tampieri D, Leblanc R, TerBrugge K (1993) Preoperative embolisation of brain and spinal hemangioblastomas. Neurosurgery 33:502–505
Van Velthoven V, Reinacher PC, Klisch J, Neumann HP, Gläsker S (2003) Treatment of intramedullary hemangioblastomas, with special attention to von Hippel-Lindau disease. Neurosurgery 53:1306–1314
Wanebo JE, Lonser RR, Glenn GM, Oldfield EH (2003) The natural history of central nervous system hemangioblastomas in patients with von Hippel-Lindau disease. J Neurosurg 98:82–94
Acknowledgements
The authors are thankful to Dr. Sahul Bharti and his team for helping us in statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the institute ethical committee. This article does not contain any studies with human participants by any of the authors. The data was collected retrospectively from case files.
Informed consent
This is a retrospective study. Patient identifying information is not included in this article. For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Sadashivam, S., Abraham, M., Kesavapisharady, K. et al. Long-term outcome and prognostic factors of intramedullary spinal hemangioblastomas. Neurosurg Rev 43, 169–175 (2020). https://doi.org/10.1007/s10143-018-1025-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-018-1025-2