Abstract
Objective
The recommended durations of treatment for acute focal bacterial nephritis (AFBN) and acute pyelonephritis (APN) are different. This study aimed to clarify the sonographic findings used to differentiate AFBN from APN during diagnosis and to compare these findings with those obtained using computed tomography (CT).
Methods
Eleven children with urinary tract infection who underwent contrast-enhanced CT and ultrasound examinations within a 24-h period were included. Diagnoses of AFBN and APN were established using CT data as the gold standard; viz., a focal area of poor enhancement is observed in AFBN but not in APN. The following ultrasound findings were evaluated: focal loss of corticomedullary differentiation (one/multiple), focal hyperechogenicity, abscess formation, and diffuse nephromegaly. Fisher’s exact test was used for statistical analysis.
Results
Of the 11 patients, 8 had AFBN and 3 had APN. The two groups differed significantly in the incidence of a focal loss of corticomedullary differentiation (present/absent, 8/8 vs. 0/3; p = 0.01) but not in the incidence of focal hyperechogenicity, abscess formation, and diffuse nephromegaly (present/absent, 2/8 vs. 0/3, p > 0.99; 1/8 vs. 0/3, p > 0.99; and 5/8 vs. 3/3, p = 0.49, respectively). The poorly enhanced area used to diagnose AFBN on CT images appeared as a focal loss of corticomedullary differentiation in ultrasound examinations. CT revealed multiple lesions in two cases in which ultrasound revealed only single lesions.
Conclusion
In our small cohort, ultrasound could be adequately used to diagnose AFBN based on the presence of a focal loss of corticomedullary differentiation. CT may not be required to differentiate AFBN from APN.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pediatric urinary tract infections are a common problem encountered in emergency departments. Febrile urinary tract infection usually manifests as acute focal bacterial nephritis (AFBN), which is also known as acute lobar nephroma, or as acute pyelonephritis (APN) [1, 2]. The differentiation of AFBN and APN during the acute phase is important because the recommended treatment durations differ for these conditions [3]. A 3-week course of intravenous and oral antibiotics therapy, rather than 2 weeks, is recommended for pediatric patients with AFBN [3].
AFBN presents as a localized, non-liquefactive, inflammatory renal bacterial infection that typically involves one or more lobes [1, 2, 4, 5]. In recent studies, AFBN diagnoses were based on imaging findings obtained using computed tomography (CT) [3, 6, 7]. Pediatric and infection control physicians assign a diagnosis of AFBN if the patient exhibits nephromegaly on ultrasound and positive CT findings and APN if nephromegaly is absent or present without lesions on CT [8, 9]. In contrast, radiologists use very similar definitions for AFBN and APN, which are both characterized by poorly enhanced areas in the kidney [1, 3, 4, 6, 7, 10,11,12]. This discrepancy in definitions might cause confusion among radiologists and physicians with respect to the differentiation of AFBN from APN.
In the emergency department, ultrasound is usually the first modality used to evaluate urinary tract infections in pediatric patients [10, 13, 14]. The characteristic sonographic findings of AFBN involve variable echogenicity [2, 5, 15, 16]. Although only one report has suggested that ultrasound is as effective as CT for diagnosing AFBN and that systematic study focused on nephromegaly rather than echogenicity, CT was performed more than 72 h after ultrasound examination [9]. The present study aimed to clarify the sonographic findings that could be used to diagnose and differentiate AFBN from APN and compare the sonographic findings with CT findings obtained within 24 h.
Patients and groups
The ethics committee of our institution approved this retrospective study and waived the requirement of informed consent from the patients. The medical records of 14 children with suspected urinary tract disease who underwent contrast-enhanced CT and ultrasound within 24 h between May 2014 and September 2019 were reviewed. The exclusion criteria were as follows: (1) no clinical diagnosis of urinary tract infection (n = 1) and (2) clinical diagnosis of chronic- rather than acute-phase disease (n = 2). The remaining patients in our cohort were classified into two groups: AFBN and APN.
Diagnosis of AFBN and APN
Diagnoses of AFBN and APN were established according to previous reports, using CT findings as the gold standard. Specifically, AFBN was indicated by positive CT findings such as a poorly enhanced area in the kidney (Figs. 1–3), while APN was diagnosed clinically if nephromegaly was absent or present without focal CT lesions (Fig. 4) [8, 9].
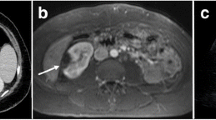
Case 2. A 1-year-old male pediatric patient with acute focal bacterial nephritis. a A sagittal sonogram shows focal hyperechogenicity in the upper pole of the right kidney (arrow). In this lesion, a focal loss of corticomedullary differentiation is shown relative to other areas. Ultrasound shows only one lesion. The right renal size is 76 mm, and the left kidney is atrophied. The size of the right kidney was greater than that in a previous report (estimate, 57.65 ± 11.35 mm) [18]. b An axial CT image shows a poorly enhanced area in the upper pole (arrow). This location shows focal nephromegaly. c An axial CT image at the lower level of (b) depicts another lesion at the mid-pole level (arrow) and does not show focal nephromegaly
Case 4. A 2-year-old female pediatric patient with acute focal bacterial nephritis. a A sagittal sonogram shows a focal loss of corticomedullary differentiation without hyperechogenicity (arrows). Multiple lesions are visible (arrows). A liquefactive change with low echogenicity was revealed, and abscess formation was diagnosed. The renal size is 124 mm. The size of the right kidney is greater than that in a previous report (estimate, 89.13 ± 11.35 mm) [18], indicating the presence of nephromegaly. b Axial sonogram shows a focal loss of corticomedullary differentiation without hyperechogenicity (arrowheads). A liquefactive change with low echogenicity was revealed, and abscess formation was diagnosed (arrows). c A coronal reconstructed CT image shows an area of poor or no enhancement indicative of abscess formation (arrow). Focal nephromegaly is also visible. d A coronal reconstructed CT image shows multiple areas of poor enhancement and an area of no enhancement indicative of abscess formation (arrow). The left kidney is atrophied
Case 7. An 8-year-old male pediatric patient with acute focal bacterial nephritis. a A sagittal sonogram shows a focal loss of corticomedullary differentiation without hyperechogenicity (arrows). This wedge-shaped area of decreased echogenicity involves the upper pole of the kidney. The right and left renal sizes are 87 and 93 mm, respectively. The sizes of the right and left kidneys are not greater than those in a previous report (estimates: 80.1 ± 11.35 and 83.4 ± 11.17 mm, respectively) [18] and therefore do not indicate diffuse nephromegaly. b A coronal reconstructed CT image reveals a poorly enhanced area without focal nephromegaly (arrow)
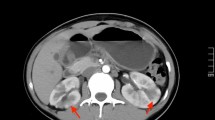
Case 9. A 2-year-old male pediatric patient with acute pyelonephritis. a A sagittal sonogram shows diffuse nephromegaly without a focal loss of corticomedullary differentiation in the left kidney. The size of the left right kidney is greater than that in a previous report (estimate: 63.22 ± 11.17 mm) [18]. b A sagittal sonogram shows a normally sized right kidney without abnormal sonographic findings. The renal size is 62 mm. The left kidney is larger than the right kidney, suggesting the presence of nephromegaly. c A coronal reconstructed CT image shows diffuse nephromegaly in the left kidney (arrows) relative to the right kidney (arrowheads). A focal area of poor enhancement is not visible
CT
All CT investigations were performed using 64-detector CT scanners (Definition AS+, Siemens Healthcare, Erlangen, Germany, from 2014 to 2017 and Definition Wedge, Siemens Healthcare, Erlangen, Germany, from 2017 to 2019). All examinations were performed using a low-dose technique with either a weight-based table or automated tube current modulation to determine the tube current (in mA) and a voltage of 80–120 kV, which was automatically adjusted according to the patient’s physique. The standard section thickness was 3 mm. Contrast material (300 mg iodine/mL) was administered at a dose of 2 mL/kg, with a maximum volume of 100 mL. The contrast material was injected either via automated or manual methods. CT examinations were performed in the portal or delayed phase, and not in the arterial phase. The CT dose index was 2.61 ± 2.00 mGy (range, 0.81–6.92 mGy; 32-cm phantom).
Ultrasound
All sonograms were obtained using a 9–15-MHz linear transducer (LOGIQ 7, E9, E10 and S8; GE Healthcare, Waukesha, WI, USA). The sonographic examinations were conducted by four pediatric radiologists with 7, 10, 15, and 20 years of clinical experience in pediatric ultrasound.
Evaluation of imaging findings
In accordance with previous studies [1, 2, 4, 7,8,9, 11, 12, 17], the number of focal lesions (null, single, or multiple) was recorded, and the imaging findings from CT were evaluated as follows: focal poor enhancement (Figs. 1, 2, and 3), focal nephromegaly (Fig. 1), abscess formation (Fig. 2), and diffuse nephromegaly (Fig. 4). The number of focal lesions (null single or multiple) and the following findings were also evaluated on ultrasound images: focal loss of corticomedullary differentiation (Figs. 1, 2, and 3), focal hyperechogenicity (Fig. 1), abscess formation (Fig. 2), and diffuse nephromegaly (Fig. 4). Diffuse nephromegaly was defined as a renal size greater than the previously reported normal size (>1 cm) [18]. Focal nephromegaly on CT images was defined as a focal area of bulking around the renal shape. The diagnostic criteria for an abscess were an area of hypoattenuation and an absence of enhancement in the renal parenchyma on CT [5, 8, 11, 12, 19] and a focal anechoic area in the renal parenchyma on ultrasound [11, 20]. The CT findings from cases exhibiting abnormal sonographic findings were reviewed. For patients exhibiting multiple lesions, the findings for the largest lesion were compared in this study.
Review process
The presence or absence of the abovementioned image findings was evaluated. All CT data were transferred to multi-planar reconstructions (MPRs); in some patients, sections with a slice thickness of 1 mm were reviewed. Two radiologists with 10 and 15 years of clinical experience reviewed all images on a 1600 × 1200 picture archiving and communication system monitor (PACS; GE Healthcare). Any discrepancies were resolved through consensus. During the review process, the radiologists were unaware of the physical or other imaging findings.
Statistical analysis
Data are presented as medians and ranges. Fisher’s exact test was used to compare the sex distribution and presence or absence of previous sonographic findings between pediatric patients with AFBN and APN. The significance level for all tests (two-sided) was set at 5%. All data were analyzed using a commercially available software program (SPSS version 24; IBM, Armonk, NY, USA).
Results
This study included 11 children (9 boys and 2 girls). The patients’ characteristics are summarized in Table 1. The mean age was 6.2 ± 6.0 years (range, 0–19 years). Of the 11 patients, eight were diagnosed with AFBN and three were diagnosed with APN. No significant difference in the sex ratio was observed between pediatric patients with AFBN and APN (sex [male/female], 6/2 vs. 3/0, p > 0.99).
Number of focal lesions
CT identified multiple lesions in six cases, while ultrasound identified multiple lesions in four cases.
Sonographic findings
The sonographic findings in the AFBN and APN groups are summarized in Table 2. A CT finding of a poorly enhanced area in the kidney was required for a diagnosis of AFBN. Locations corresponding to this CT finding were detected as focal losses of corticomedullary differentiation on ultrasound in all cases with AFBN, but no cases with APN.
Focal loss of corticomedullary differentiation
The presence of a focal loss of corticomedullary differentiation differed significantly between patients with AFBN and those with APN (present/absent, 8/8 vs. 0/3; p = 0.01; Figs. 1, 2, and 3).
Focal hyperechogenicity
The presence of focal hyperechogenicity differed significantly between patients with AFBN and those with APN (present/absent, 2/8 vs. 0/3; p > 0.99; Fig. 1).
Abscess formation
No significant difference was observed in the presence of abscess formation between patients with AFBN and those with APN (present/absent, 1/8 vs. 0/3, p > 0.99; Fig. 2).
Diffuse nephromegaly
No significant difference was observed in the presence of nephromegaly between pediatric patients with AFBN and those with APN (present/absent, 5/8 vs. 3/3, p = 0.49; Fig. 4).
Comparison of sonographic and CT findings
Table 3 summarizes the comparison between sonographic and CT findings at each location. The locations exhibiting focal hyperechogenicity on ultrasound appeared both as focal areas of nephromegaly and poor enhancement on CT. Locations exhibiting a focal loss of corticomedullary differentiation on ultrasound appeared as focal areas of poor enhancement on CT. Some of these locations also exhibited focal nephromegaly or abscess formation. The same locations corresponding to abscess formation were identified via ultrasound and CT. However, CT detected multiple lesions in two cases in which ultrasound identified only one lesion.
Discussion
We demonstrated that in our small cohort, ultrasound could adequately diagnose AFBN on the basis of sonographic findings such as a focal loss of corticomedullary differentiation. CT may not be required to differentiate AFBN from APN. However, CT might be superior to ultrasound for the detection of multiple lesions. When faced with recommendations by pediatric and infection control physicians to use CT as a diagnostic modality for AFBN, radiologists may respond that CT might not provide additional value over ultrasound in terms of decisions regarding the treatment duration.
Various sonographic findings were observed in patients with AFBN. In our small cohort, two of the three locations that appeared as focal nephromegaly on CT also exhibited hyperechogenicity on ultrasound. The third case exhibited abscess formation, which was detected as a lesion with low echogenicity, consistent with previous reports [21, 22]. None of the cases exhibiting poor focal enhancement and no focal nephromegaly on CT exhibited focal hyperechogenicity on ultrasound. We speculate that the severity and phase of inflammation influences the variability observed in sonographic findings. Focal nephromegaly on CT and hyperechogenicity on ultrasound may be detected in cases presenting with a more acute or severe inflammatory condition than in cases without these findings [2, 5, 20]. These findings may indicate a hemorrhagic or edematous change in the renal parenchyma [11, 12]. In contrast, the absence of hyperechogenicity on ultrasound might indicate the non-acute phase of AFBN and may be attributable to recovery or evolution to a renal abscess [1, 5, 8, 11, 15, 16, 20]. One case report described a hyperechoic lesion that changed to a hypoechoic lesion after 3 days [5]. Therefore, lesions that do not appear as focal nephromegaly on CT or hyperechogenicity on ultrasound may correspond to a more acute phase that occurs a few days after the onset of urinary tract infection.
A common radiological finding of APN is a striated nephrogram on a CT image, wherein alternating linear bands of high and low attenuation appear in a radial pattern that extends to the corticomedullary layers of the kidney [23]. In our small cohort, however, we did not observe any cases with only imaging findings of a striated nephrogram, and this finding was only detected in limited focal areas of kidney and was accompanied by multiple lesions. This finding was not clearly defined in a previous study that focused on the duration of treatment provided to patients with AFBN and APN [3]. This discrepancy between studies might have led to confusion regarding the definitions used by radiologists and physicians to differentiate AFBN from APN.
Abscess formation is an important factor in decisions regarding the treatment method [1, 8, 11, 17, 20]. In our study, only one case exhibited a renal abscess, which was detected by both ultrasound and CT. Ultrasound may be a useful modality for evaluating renal abscess.
Previous studies have reported that urinary tract infection is more common in female than in male patients [24, 25]. Our result was not consistent with those findings. We note that the inclusion of patients who underwent CT led to the selection of patients with relatively severe urinary tract infection. Our cohort also included some infants, and previous studies have demonstrated a male predominance among young infants with urinary tract infection [25]. These factors might have affected the sex ratio in our cohort.
The number of focal lesions was correctly diagnosed by CT. Although CT involves radiation exposure and contrast medium injection, this technique allows the evaluation of the entire kidney, without any blind areas. Although the number of focal lesions does not affect the treatment duration, CT may be recommended when a patient responds poorly to an initial 72-h course of antibiotic treatment prescribed based on a sonographic diagnosis, when an ultrasound assessment does not yield a diagnosis, or when clinical and laboratory findings strongly suggest renal involvement, given the dependency of ultrasound results on the operator’s skill level [3, 6, 8, 9, 17]. Contrast-enhanced ultrasound was reported to be useful for evaluating small renal abscesses or focal pyelonephritis [26,27,28]. This technique requires neither irradiation nor sedation and may thus be more acceptable for children. Although contrast-enhanced ultrasound may be difficult to perform in an emergency department, it is useful in cases involving a suspicion of renal abscess or when the patient does not respond as expected to treatment.
This study had some limitations. First, it included a small number of patients with APN or AFBN and was retrospective in nature. Particularly, the number of patients with APN was very small. Clinically, most patients with suspected APN are prescribed with antibiotic therapy based on the sonographic findings, and CT is not usually performed. Therefore, additional prospective studies with larger patient populations are needed to confirm our preliminary findings. Second, we included only cases in which both CT and ultrasound were performed within 24 h. This inclusion criterion may have led to inclusion bias toward patients with more severe urinary tract infection. Moreover, patients who exhibited only striated nephrogram on imaging findings may have been excluded. Third, some cases involved multiple lesions and therefore might have represented various phases of AFBN (acute or chronic). Because it was difficult to differentiate these phases, we focused solely on the largest lesion in each case.
Conclusion
In our small cohort, ultrasound was adequate for the diagnosis of AFBN, based on sonographic findings such as a focal loss of corticomedullary differentiation. Our results suggest that CT may not be needed to differentiate AFBN from APN when determining the appropriate treatment method.
References
Bitsori M, Raissaki M, Maraki S, Galanakis E (2015) Acute focal bacterial nephritis, pyonephrosis and renal abscess in children. Pediatr Nephrol 30(11):1987–1993. https://doi.org/10.1007/s00467-015-3141-3
Rosenfield AT, Glickman MG, Taylor KJ, Crade M, Hodson J (1979) Acute focal bacterial nephritis (acute lobar nephronia). Radiology 132(3):553–561. https://doi.org/10.1148/132.3.553
Cheng CH, Tsau YK, Lin TY (2006) Effective duration of antimicrobial therapy for the treatment of acute lobar nephronia. Pediatrics 117(1):e84–e89. https://doi.org/10.1542/peds.2005-0917
Morello W, La Scola C, Alberici I, Montini G (2016) Acute pyelonephritis in children. Pediatr Nephrol 31(8):1253–1265. https://doi.org/10.1007/s00467-015-3168-5
Rathore MH, Barton LL, Luisiri A (1991) Acute lobar nephronia: a review. Pediatrics 87(5):728–734
Klar A, Hurvitz H, Berkun Y, Nadjari M, Blinder G, Israeli T, Halamish A, Katz A, Shazberg G, Branski D (1996) Focal bacterial nephritis (lobar nephronia) in children. J Pediatr 128(6):850–853. https://doi.org/10.1016/s0022-3476(96)70340-2
Kline MW, Kaplan SL, Baker CJ (1988) Acute focal bacterial nephritis: diverse clinical presentations in pediatric patients. Pediatr Infect Dis J 7(5):346–349. https://doi.org/10.1097/00006454-198805000-00012
Cheng CH, Tsau YK, Lin TY (2010) Is acute lobar nephronia the midpoint in the spectrum of upper urinary tract infections between acute pyelonephritis and renal abscess? J Pediatr 156(1):82–86. https://doi.org/10.1016/j.jpeds.2009.07.010
Cheng CH, Tsau YK, Hsu SY, Lee TL (2004) Effective ultrasonographic predictor for the diagnosis of acute lobar nephronia. Pediatr Infect Dis J 23(1):11–14. https://doi.org/10.1097/01.inf.0000105202.57991.3e
Dacher JN, Pfister C, Monroc M, Eurin D, LeDosseur P (1996) Power Doppler sonographic pattern of acute pyelonephritis in children: comparison with CT. AJR Am J Roentgenol 166(6):1451–1455. https://doi.org/10.2214/ajr.166.6.8633462
Stunell H, Buckley O, Feeney J, Geoghegan T, Browne RF, Torreggiani WC (2007) Imaging of acute pyelonephritis in the adult. Eur Radiol 17(7):1820–1828. https://doi.org/10.1007/s00330-006-0366-3
Craig WD, Wagner BJ, Travis MD (2008) Pyelonephritis: radiologic-pathologic review. Radiographics 28(1):255–277; quiz 327-258. https://doi.org/10.1148/rg.281075171
Hosokawa T, Yamada Y, Tanami Y, Sato Y, Ishimaru T, Tanaka Y, Kawashima H, Oguma E (2019) Comparison of diagnostic accuracy for fistulae at ultrasound and voiding cystourethrogram in neonates with anorectal malformation. Pediatr Radiol 49(5):609–616. https://doi.org/10.1007/s00247-018-04339-4
Lavocat MP, Granjon D, Allard D, Gay C, Freycon MT, Dubois F (1997) Imaging of pyelonephritis. Pediatr Radiol 27(2):159–165. https://doi.org/10.1007/s002470050091
Mizutani M, Hasegawa S, Matsushige T, Ohta N, Kittaka S, Hoshide M, Kusuda T, Takahashi K, Ichihara K, Ohga S (2017) Distinctive inflammatory profile between acute focal bacterial nephritis and acute pyelonephritis in children. Cytokine 99:24–29. https://doi.org/10.1016/j.cyto.2017.06.012
Farmer KD, Gellett LR, Dubbins PA (2002) The sonographic appearance of acute focal pyelonephritis 8 years experience. Clin Radiol 57(6):483–487. https://doi.org/10.1053/crad.2002.0935
Soulen MC, Fishman EK, Goldman SM, Gatewood OM (1989) Bacterial renal infection: role of CT. Radiology 171(3):703–707. https://doi.org/10.1148/radiology.171.3.2655002
Kim JH, Kim MJ, Lim SH, Kim J, Lee MJ (2013) Length and volume of morphologically normal kidneys in Korean children: ultrasound measurement and estimation using body size. Korean J Radiol 14(4):677–682. https://doi.org/10.3348/kjr.2013.14.4.677
Hosokawa T, Yamada Y, Tanami Y, Sato Y, Ko Y, Nomura K, Oguma E (2020) Computed tomography findings of mediastinitis after cardiovascular surgery. Pediatr Int 62(2):206–213. https://doi.org/10.1111/ped.14101
Shimizu M, Katayama K, Kato E, Miyayama S, Sugata T, Ohta K (2005) Evolution of acute focal bacterial nephritis into a renal abscess. Pediatr Nephrol 20(1):93–95. https://doi.org/10.1007/s00467-004-1646-2
Hosokawa T, Suzuki S, Tanami Y, Sato Y, Ko Y, Nomura K, Hosokawa M, Oguma E, Yamada Y (2019) Ultrasound evaluation of complications after cardiovascular surgery in pediatric patients: a case series. Med Ultrason Epub ahead of print. Doi:https://doi.org/10.11152/mu-1982
Hosokawa T, Yamada Y, Tanami Y, Sato Y, Ishimaru T, Kawashima H, Oguma E (2019) Associations between sonographic findings and operative time of transumbilical laparoscopic-assisted appendectomy for acute appendicitis in children. AJR Am J Roentgenol:1–9. doi:https://doi.org/10.2214/ajr.18.20937
Saunders HS, Dyer RB, Shifrin RY, Scharling ES, Bechtold RE, Zagoria RJ (1995) The CT nephrogram: implications for evaluation of urinary tract disease. Radiographics 15(5):1069–1085; discussion 1086-1068. https://doi.org/10.1148/radiographics.15.5.7501851
He Y, Zhang W, Sun N, Feng G, Ni X, Song H (2019) Experience of pediatric urogenital tract inserted objects: 10-year single-center study. J Pediatr Urol 15(5):554.e551–554.e558. https://doi.org/10.1016/j.jpurol.2019.05.038
Albracht CD, Hreha TN, Hunstad DA (2020) Sex effects in pyelonephritis. Pediatr Nephrol:1–9. https://doi.org/10.1007/s00467-020-04492-9
Seitz K, Bernatik T, Strobel D, Blank W, Friedrich-Rust M, Strunk H, Greis C, Kratzer W, Schuler A (2010) Contrast-enhanced ultrasound (CEUS) for the characterization of focal liver lesions in clinical practice (DEGUM multicenter trial): CEUS vs. MRI--a prospective comparison in 269 patients. Ultraschall Med 31(5):492–499. https://doi.org/10.1055/s-0029-1245591
Fontanilla T, Minaya J, Cortes C, Hernando CG, Aranguena RP, Arriaga J, Carmona MS, Alcolado A (2012) Acute complicated pyelonephritis: contrast-enhanced ultrasound. Abdom Imaging 37(4):639–646. https://doi.org/10.1007/s00261-011-9781-2
Hashimoto M, Ohkuma K, Akita H, Yamada Y, Nakatsuka S, Mizuno R, Oya M, Jinzaki M (2019) Usefulness of contrast-enhanced ultrasonography for diagnosis of renal cell carcinoma in dialysis patients: comparison with computed tomography. Medicine (Baltimore) 98(47):e18053. https://doi.org/10.1097/md.0000000000018053
Author information
Authors and Affiliations
Contributions
T.H. and Y.T designed the study; Y.T and Y.S collected and analyzed data; T.H and Y.T
wrote the manuscript; T.H and Y.T performed the statistical analysis and drafted the manuscript; and E.O critically reviewed the manuscript and supervised the whole study process.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human subjects
This research was performed in accordance with the tenets of the Declaration of Helsinki.
Informed consent
This retrospective study was approved by the ethics committee of our institution, and informed consent was waived.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hosokawa, T., Tanami, Y., Sato, Y. et al. Comparison of imaging findings between acute focal bacterial nephritis (acute lobar nephronia) and acute pyelonephritis: a preliminary evaluation of the sufficiency of ultrasound for the diagnosis of acute focal bacterial nephritis. Emerg Radiol 27, 405–412 (2020). https://doi.org/10.1007/s10140-020-01771-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10140-020-01771-8