Abstract
Purpose
Data on short courses of antibiotic therapy for Enterobacterales bacteremia in high-risk neutropenic patients are limited. The aim of the study was to describe and compare the frequency of bacteremia relapse, 30-day overall and infection-related mortality, Clostridiodes difficile infection and length of hospital stay since bacteremia among those who received antibiotic therapy for 7 or 14 days.
Methods
This is a multicenter, prospective, observational cohort study in adult high-risk neutropenic patients with hematologic malignancies or hematopoietic stem cell transplant and monomicrobial Enterobacterales bacteremia. They received appropriate empirical antibiotic therapy, had a clinical response within 7 days, and infection source control. Clinical, epidemiological and outcomes variables were compared based on 7 or 14 days of AT.
Results
Two hundred patients were included (100, 7-day antibiotic therapy; 100, 14-day antibiotic therapy). Escherichia coli was the pathogen most frequently isolated (47.5%), followed by Klebsiella sp. (40.5%). Among those patients that received 7-day vs. 14-day antibiotic course, a clinical source of bacteremia was found in 54% vs. 57% (p = 0.66), multidrug-resistant Enterobacterales isolates in 28% vs. 30% (p = 0.75), and 40% vs. 47% (p = 0.31) received combined empirical antibiotic therapy. Overall mortality was 3% vs. 1% (p = 0.62), in no case related to infection; bacteremia relapse was 7% vs. 2% (p = 0.17), and length of hospital stay since bacteremia had a median of 9 days (IQR: 7–15) vs. 14 days (IQR: 13–22) (p = < 0.001).
Conclusions
These data suggest that seven-day antibiotic therapy might be adequate for patients with high-risk neutropenia and Enterobacterales bacteremia, who receive appropriate empirical therapy, with clinical response and infection source control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bacteremia is the most important infectious complication of high-risk neutropenic patients, leading to an increase in mortality, length of hospital stay, and healthcare costs [1,2,3]. In recent decades, an epidemiological change has been observed in the etiology of bacteremia in this population, with a predominance of gram-negative bacilli (GNB) as the leading cause, and Enterobacterales as the etiological agents most frequently involved [4, 5]. Likewise, growing antibiotic resistance is a major concern worldwide, especially in Latin America, where the most frequent resistance mechanism is the production of extended-spectrum beta-lactamases (ESBL) [6]. In addition, carbapenemase-producing Enterobacterales are on the rise [7, 8]. Several factors could be involved, with misuse and overuse of antibiotic therapy (AT) being the most significant. In this regard, receiving AT ≥ 10 days proved to be a risk factor for carbapenemase-producing Enterobacterales bacteremia in cancer and hematopoietic stem cell transplant (HSCT) patients [9]. Therefore, shortening the duration of AT should be an important measure in antimicrobial stewardship programs in this population.
Several guidelines on AT in oncohematological and neutropenic patients have been published [10,11,12,13,14]. Some of them suggest that antibiotic therapy should be continued for at least 7 days, until the infection is microbiologically eradicated and all signs of infection are resolved, with the patient afebrile for at least 4 days [13, 14]. However, this remains a controversial issue given the limited available data on high-risk neutropenic patients.
The efficacy and safety of short course AT have been evaluated in three randomized controlled studies in patients with uncomplicated GNB bacteremia. The rates of clinical cure, bacteremia relapse, and 30-day mortality of all those patients treated for 7 days were similar to those who received 14 days of AT [15,16,17]. Recently, a meta-analysis including the previously mentioned studies evaluated the subgroup of immunocompromised patients and found no differences between those who received AT for 7 days vs. 14 days. However, bacteremia was mainly isolated from urinary source and was observed in hemodynamically stable patients. Moreover, high-risk neutropenic patients and those with carbapenemase-producing Enterobacterales bacteremia were excluded [18].
This study was designed to describe and compare the clinical, epidemiological, and outcome characteristics, such as bacteremia relapse, Clostridiodes difficile infection, 30-day mortality and length of hospital stay, of high-risk neutropenic patients with Enterobacterales bacteremia based on 7 or 14 days of AT.
Material and methods
Setting, patients and study design
A prospective, observational, multicenter study was performed in 8 referral teaching centers (5 private and 3 public) specialized in the management of oncohematological and HSCT patients in Argentina.
All the episodes of monomicrobial Enterobacterales bacteremia in adult patients (≥ 18 years of age) managed as inpatients from May 2014 to March 2023 were included, provided that the following criteria were met: (a) patients presenting with hematologic malignancies (HM) or autologous and allogeneic HSCT; (b) high-risk febrile neutropenia; (c) appropriate empirical AT; (d) clinical response within 7 days; and (f) a total of 7 or 14 days of AT.
Patients with polymicrobial or recurrent bacteremia and those receiving palliative care were excluded from the analysis, as well as those with a source of bacteremia involving prolonged treatment (endocarditis/endovascular infections, severe skin and soft-tissue infections, central nervous system infections and osteomyelitis), or with a clinical source that required surgery.
Patients who received 14 days of antibiotic therapy were included since the beginning of the study, as this was the usual duration of AT for GNB bacteremia in this population [10]. In comparison, those who received 7 days were recruited since 2018, when short AT for Enterobacterales bacteremia was first carried out in selected patients by participating centers, in accordance with the ECIL-4 guidelines [13].
Patients were identified as a result of a positive blood culture and were then followed up prospectively. Data were obtained from medical records and direct patient care, with a double check made with microbiological records from the laboratory. Clinical, microbiological, treatment, and outcome variables were evaluated. Empirical individualized AT was initiated based on the patient’s clinical and epidemiological features, pursuant to each center institutional guidelines and IDSA and ECIL recommendations [10, 12, 13]. The study investigator chose definitive therapy (DT) based on Enterobacterales isolates and their antibiotic resistance profile.
Patients were followed for 30 days after the episode (by direct patient care in hospitalized cases, or by phone calls to discharged patients), or until the patient’s death, provided that it happened before (assessed by direct patient care in those still hospitalized, or by a local healthcare database in each center).
Definitions
Neutropenia was defined as an absolute neutrophil count < 500 cells/mm3. High-risk febrile neutropenia was defined according to the Multinational Association for Supportive Care in Cancer (MASCC) as a score < 21 and one or more clinical criteria [10]. The clinical source of bacteremia was determined based on the isolation of Enterobacterales from the suspected source and/or the associated clinical signs and symptoms. It was classified pursuant to the US CDC criteria [19]. Neutropenic enterocolitis was defined as fever with abdominal symptoms and bowel wall thickening > 4 mm [20].
Complicated bacteremia was defined as an episode presenting with hypotension, septic shock, or pneumonia as the clinical source [10, 12].
High doses of corticosteroids were defined as having received prednisone (or equivalent) at doses ≥ 20 mg/day for a period ≥ 2 weeks prior to bacteremia, and the use of biological agents and/or anti-lymphocyte therapies, having received these drugs within six months prior to bacteremia.
Patients presenting one of the abovementioned factors were considered as severely immunosuppressed.
Carbapenemase-producing Enterobacterales colonization was defined as “previous” when it occurred within six months prior to hospital stay, and “recent” when detected within the week prior to the episode of bacteremia.
Bacteremia was classified as nosocomial, healthcare-associated, or community-acquired according to Friedman et al. [21]. Hypotension was defined as systolic blood pressure < 90 mm Hg on the day of the positive blood culture [22]. Septic shock was defined as the need for vasopressors to maintain mean arterial pressure ≥ 65 mmHg and serum lactate level > 18 mg/dl [23]. Infection severity and mortality probability were defined using Pitt and APACHE-II scores. Relapse of bacteremia was defined as a new episode of bacteremia within 30 days of AT completion, with the same isolated Enterobacteral. EAT was considered appropriate provided that it was initiated after blood cultures were drawn, and one or more antibiotics used were active in vitro against the isolated bacteria. In patients with ESBL-producing Enterobacterales, empirical AT with piperacillin/tazobactam or cefepime monotherapy was considered inappropriate [24]. In patients with isolation of any Enterobacterales species, tigecycline as monotherapy was also deemed unsuitable. Clinical response on day 7 of AT was defined as absence of fever for at least four days, source control of bacteremia, absence of hypotension, and clinical resolution of all signs and symptoms of infection. In catheter-related bacteremia, catheters were removed on the day of diagnosis. Mortality was related to infection provided that there was microbiological, histological, or clinical evidence of active infection.
Microbiology
Bacteremia was defined as the isolation of a pathogenic bacteria in at least one bottle of blood culture (BD BACTEC F Aerobic and Anaerobic, analyzed with BACTEC FX BD, BacTALERT 3D BioMérieux depending on the method available at each center) for a minimum incubation period of five days. MDR-GNB were defined as Enterobacterales resistant to three or more of the following categories of antibiotics: carbapenems, piperacillin/tazobactam, third and fourth generation cephalosporins, aztreonam, fluoroquinolones, or aminoglycosides [25, 26]. Microbiological identification and susceptibility testing were done with manual biochemical and microbiological methods, disk diffusion (according to the CLSI recommendations), and/or Etest, VITEK II Compact (BioMérieux), PHOENIX 100 BD (Becton Dickinson), VITEK MS (BioMérieux) and MALDI-TOF (Microflex from Bruker). ESBL production was determined by disk diffusion method using both ceftazidime and cefotaxime, alone and combined with clavulanic acid. Carbapenemase production was investigated in carbapenem-resistant bacteria using the modified Hodge method, disk synergy tests with a carbapenem disk placed close to the boronic acid disk test for KPC, and the EDTA disk for identification of metallo-β-lactamases. The presence of genes coding for blaKPC and blaOXA-48 was investigated by monoplex or multiplex polymerase chain reaction (PCR) using specific primers depending on the method available at each center. Multiplex PCR for blaVIM, blaNDM, blaIMP, blaKPC and blaOXA-48 was used to investigate carbapenemase-producing Enterobacterales isolates at the National Reference Laboratory of Microbiology (ANLIS-Malbrán) [27]. In order to detect colonization with KPC-producing Enterobacterales, rectal swabs were routinely collected (once a week and in every pre-transplant evaluation) using chromogenic methods and/or PCR.
Clostridioides difficile was investigated by immunochromatography in every patient with diarrhea in order to determine the presence of glutamate dehydrogenase (GDH) antigen and toxins A and B. Those samples with positive GDH and negative toxins were analyzed by real-time PCR available in 7 centers [28, 29].
Statistical analysis
The study population was characterized by descriptive statistics. For continuous variables, centrality (median) and dispersion (IQR) measures were used according to the distribution of variables. Categorical variables were analyzed using absolute frequency and percentage. Groups were compared using the U Mann–Whitney test for continuous variables and the Fisher exact test or the chi-square test for categorical variables. For all tests, a 95% level of statistical significance was used. The analyses were performed with the SPSS (Statistics for Windows, Version 22.0. Armonk, NY, USA) software packages.
Results
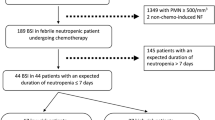
A total of 1,037 patients with HM and HSCT who presented Enterobacterales bacteremia were evaluated, and 837 were excluded because they did not meet the eligibility criteria (Fig. 1). Thus, the study population consisted of 200 patients, 100 in each treatment group. The baseline and epidemiological characteristics of bacteremia episodes that received AT for 7 and 14 days are described in Table 1. Most patients had acute leukemia as the underlying disease (127, 63.5%), followed by lymphoma (42, 21%). Sixty-five patients (32.5%) received HSCT (32, 46.1% allogeneic). HM was active in 144 (72%) patients; 171 (85.5%) had received chemotherapy one month prior to bacteremia episode, 49 (24.5%) and 40 (20%) had received high-dose corticosteroids or biological agents and/or anti-lymphocyte therapies, respectively. One hundred and nine (54%) patients had previously received antibiotics, with piperacillin-tazobactam being the most common (60, 30%). Recent and previous colonization by KPC-carbapenemase producing Enterobacterales were detected in 24 (12%) and 21 (11.5%), respectively. The Enterobacterales most frequently isolated were Escherichia coli (95, 47.5%) and Klebsiella sp. (81, 40.5%). Fifty-eight (29%) were MDR-GNB, with ESBL producers (44, 22%) and carbapenemase-producing Enterobacterales (12, 6%) being the most frequent, with similar distribution between both groups. Regarding resistance profile, more than 20% of the isolates were resistant to piperacillin-tazobactam and cefepime, and 6% to meropenem. Resistance to fluoroquinolones was 35%, being significantly higher in the group of patients treated for 14 days, and resistance to the other antibiotics was low in both groups. The microbiological characteristics and resistance profiles of bacteremia episodes are described in Figs. 2, 3, and 4.
Comparative frequency of multidrug-resistant gram-negative bacilli and resistance mechanisms detected between the 7- and 14-day antibiotic therapy groups. Abbreviations: MDR-GNB, multidrug-resistant gram-negative bacilli; ESBL, extended spectrum beta-lactamase; KPC, Klebsiella pneumoniae serin-carbapenemase; NDM, New Delhi metallo-beta-lactamase; AmpC; AmpC beta-lactamase. *p-value obtained by chi-square or Fisher exact tests
Comparative antibiotic resistance profile of gram-negative bacilli bacteremia between the 7- and 14-day antibiotic therapy groups. Abbreviation: CIP, Ciprofloxacin; CRO, Ceftriaxone; FEP, Cefepime; PTZ, Piperacillin-tazobactam; MER, Meropenem; AMK, Amikacin; COL, Colistin; TIG, Tigecycline; FOS, Fosfomycin; CZA, Ceftazidime-avibactam. ¶: Colistin resistance available in 176 isolates. ¥: Tigecycline and Fosfomycin resistance available in 169 isolates. § Ceftazidime-avibactam resistance available in 85 isolates. *p-value obtained by chi-square or Fisher exact tests
Table 2 shows the clinical characteristics, AT, and outcome of patients with Enterobacterales bacteremia according to the treatment group. Of 111 (55.5%) episodes of bacteremia with a clinical source, abdominal –evidenced as colitis (50, 25%), and catheter-related bacteremia (32, 16%) were the most frequent. EAT was combined in 88 (44%) cases, with carbapenems (57%) being the most commonly used antibiotics. DT as monotherapy was significantly higher in patients who received 7 days of AT (99% vs. 92%, p = 0.03), and carbapenems (33.5%) and piperacillin-tazobactam (28.2%) were the antibiotics most frequently prescribed. Forty-two patients (21%) received AT in prolonged infusion, which was largely used in patients with 7-day treatment (33% vs. 9%, p < 0.001). AT discontinuation before neutrophil recovery was mainly carried out in patients under 7-day treatment (46% vs. 26%, p = 0.003). No patients returned to antibiotic prophylaxis.
Bacteremia episodes were complicated in 76 (38%) patients with no differences between groups. Septic shock at onset occurred in 25 (12.5%) patients, and was more frequently observed in those who received 14 days of treatment (18% vs. 7%, p = 0.01). Clostridioides difficile infection and bacteremia relapse occurred in 9 (4.5%) patients. Thirty-day mortality was 2%, in no case related to infection, with no differences between both groups. Length of hospital stay since bacteremia had a median of 13 days (IQR: 8–20), being significantly lower in the group that received 7 days of treatment. The differences observed in treatment and outcome between the two groups are highlighted in Table 2.
Discussion
This study evaluated a total of 7-day AT as a feasible strategy for high-risk neutropenic patients with Enterobacterales bacteremia who received adequate empirical AT with clinical response. The cohort included several patients with complicated bacteremia, an identifiable clinical source of infection, and even septic shock. Moreover, many of them presented bacteremia due to MDR-GNB, including carbapenemase-producing Enterobacterales. Both groups had similar baseline, microbiological, clinical, and epidemiological characteristics. A low rate of bacteremia relapse and mortality was observed, which was similar in both groups. However, length of hospital stay since bacteremia diagnosis was significantly reduced in patients who received 7 days of AT.
Short courses of AT in hematological and neutropenic patients with GNB bacteremia have been previously evaluated in three single-center studies.
The first one is a retrospective cohort study that compared the efficacy of short (median 6 days, IQR: 6–7) versus prolonged (median 11 days, IQR: 9.5–14) AT for bacteremia in patients with acute myeloid leukemia and febrile neutropenia [30]. A total of 71 patients presenting 104 episodes of bacteremia were included, and 46% received a short course of AT. In the total population, 65% of bacteremias were primary, and 51% were caused by GNB, mostly Enterobacterales. Of them, 23% were ESBL producers, but carbapenemase-producing Enterobacterales bacteremias were not included. Bacteremia relapse and mortality were similar between both groups. This study largely differs from ours. Although the population was homogeneous regarding the underlying disease, patients were not stratified according to the risk of neutropenia; bacteremias were mainly primary, and not all of them were caused by GNB. The implemented AT was not described, the variables usually associated with mortality were not considered, and more than one episode per patient were included. These issues may cause bias in the interpretation of the study results.
The second one is a retrospective study in which three groups of neutropenic patients with GNB bacteremia were compared based on AT duration: ≤ 10 days, 11–14 days, and ≥ 15 days. The primary objective was to evaluate mortality and bacteremia relapse at day 90 [31]. Although most patients had HM and HSCT, subjects with different immunosuppressive diseases were also included. As in our study, more than 50% of bacteremias had a clinical source, with abdominal and central venous catheters being the most frequent. However, their etiology was heterogeneous since in the short treatment group, of 67 bacteremias, 13.4% and 23.9% were caused by Pseudomonas aeruginosa and other GNB, respectively. Moreover, bacteremias caused by carbapenemase-producing Enterobacterales were excluded. Bacteremia relapse and mortality were similar between the three groups, adjusted by a propensity score analysis. Of note, the short treatment group included patients treated for up to 10 days. However, it might be controversial to assume that this is a short duration.
The last one is a prospective observational study that compared the duration of AT with a median of 7 days (IQR: 7–7) versus 14 days (IQR: 14–14) in 74 patients with cancer and HSCT presenting GNB bacteremia [32]. The inclusion criteria were the same as those in the present study. However, a small percentage of patients with bacteremia caused by Pseudomonas aeruginosa were included and only around 60% were neutropenic. The groups were comparable in terms of baseline characteristics, clinical presentation, severity, epidemiology, and microbiology. As in our study, 30-day mortality and bacteremia relapse were low and similar, with a significant reduction in length of hospital stay in those who received short AT.
These are the major findings of the present study: first, besides being neutropenic, a considerable number of patients was severely immunosuppressed, presented complicated bacteremia, and had a high APACHE II score. Notwithstanding that, mortality was low and in no case related to bacteremia, which suggests that early implementation of adequate empirical AT and source control of bacteremia is crucial. Second, in almost half of the patients that received 7-day AT, antibiotics were discontinued before neutrophil recovery, which shows that this approach could be safe. Third, in most of the patients who received AT for 7 days, DT was monotherapy, highlighting the importance of de-escalating antibiotics in high-risk neutropenic patients with a clinical response [33]. Fourth, one-third of patients in the short treatment group received antibiotics in prolonged infusion as the strategy implemented by some centers over the last years [34]. This variable could have had an impact on the result. However, there is still no conclusive data available to determine whether this strategy offers clinical benefits in high-risk neutropenic patients with GNB bacteremia [35]. Finally, patients who received short treatment had a 9-day median duration of hospital stay since bacteremia. Of note, hospital stay ≥ 10 days is known to be an independent risk factor for developing KPC-carbapenemase producing Enterobacterales bacteremia [9].
Our study has some limitations that should be considered. First, patients received different AT, which could induce a bias in the result. However, both cohorts were prescribed adequate AT, with a similar proportion of combination therapy and monotherapy. In addition, empirical AT was based on IDSA and ECIL recommendations. Second, some variables that could have influenced the outcomes, such as septic shock and higher Charlson comorbidity score, were more frequent in patients who received 14-day AT. Given that DT was a personalized decision, we cannot rule out that the study investigators might have decided to prolong the AT based on these factors. Third, although we did not observe statistical differences in relapse rates between the groups, the study might have been underpowered to detect this outcome. Fourth, although a significant rate of MDR-GNB bacteremias was included, the number of carbapenemase-producing Enterobacterales was low. Therefore, we cannot conclude that the findings obtained can be applied to these microorganisms. Finally, since the number of patients who presented with septic shock was low, we cannot extrapolate the final results to high-risk neutropenic patients with hemodynamic failure.
The strengths of our study rely on its prospective and multicenter design, carried out in healthcare facilities specialized in the treatment of high-risk neutropenic patients. Likewise, a homogeneous and comparable population was included in terms of clinical, microbiological, and outcome variables in cohorts allocated to the same duration of AT.
In conclusion, this study showed that AT for 7 days might be adequate for selected high-risk neutropenic patients with Enterobacterales bacteremia who received adequate empirical AT, with clinical response. Furthermore, this strategy was associated with a significant reduction in length of hospital stay, and this might have potential benefits, such as the reduction of healthcare costs and the emergence of MDR-GNB. Randomized studies are needed to support these findings and confirm the efficacy and safety of short courses of AT in this population.
Data availability
No datasets were generated or analyzed during the current study.
References
Kuderer NM, Dale DC, Crawford J, Cosler LE, Lyman GH (2006) Mortality, morbidity, and cost associated with febrile neutropenia in adult cancer patients. Cancer 106(10):2258–2266. https://doi.org/10.1002/cncr.21847
Bos MM, Smeets LS, Dumay I, de Jonge E (2013) Bloodstream infections in patients with or without cancer in a large community hospital. Infection 41(5):949–958. https://doi.org/10.1007/s15010-013-0468-1
Dandoy CE, Ardura MI, Papanicolaou GA, Auletta JJ (2017) Bacterial bloodstream infections in the allogeneic hematopoietic cell transplant patient: new considerations for a persistent nemesis. Bone Marrow Transplant 52(8):1091–1106. https://doi.org/10.1038/bmt.2017.14
Trecarichi EM, Pagano L, Candoni A, Pastore D, Cattaneo C, Fanci R, Nosari A, Caira M, Spadea A, Busca A, Vianelli N, Tumbarello M, HeMABISRegistry—SEIFEM Group, Italy (2015) Current epidemiology and antimicrobial resistance data for bacterial bloodstream infections in patients with hematologic malignancies: an Italian multicentre prospective survey. Clin Microbiol Infect. 21(4):337–43. https://doi.org/10.1016/j.cmi.2014.11.022
Averbuch D, Tridello G, Hoek J et al (2017) Antimicrobial resistance in gram-negative rods causing bacteremia in hematopoietic stem cell transplant recipients: intercontinental prospective study of the infectious diseases working party of the european bone marrow transplantation group. Clin Infect Dis 65(11):1819–1828. https://doi.org/10.1093/cid/cix646
Cruz-Vargas SA, García-Muñoz L, Cuervo-Maldonado SI et al (2023) Molecular and clinical data of antimicrobial resistance in microorganisms producing bacteremia in a multicentric cohort of patients with cancer in a Latin American country. Microorganisms 11(2):359. https://doi.org/10.3390/microorganisms11020359
Castanheira M, Deshpande LM, Mendes RE, Canton R, Sader HS, Jones RN (2019) Variations in the occurrence of resistance phenotypes and carbapenemase genes among Enterobacteriaceae isolates in 20 years of the SENTRY antimicrobial surveillance program. Open Forum Infect Dis 6(Suppl 1):S23–S33. https://doi.org/10.1093/ofid/ofy347
Thomas GR, Corso A, Pasterán F et al (2022) Increased detection of carbapenemase-producing enterobacterales bacteria in Latin America and the Caribbean during the COVID-19 pandemic. Emerg Infect Dis 28(11):1–8. https://doi.org/10.3201/eid2811.220415
Herrera F, Torres D, Laborde A et al (2023) Development of a clinical score to stratify the risk for carbapenem-resistant Enterobacterales bacteremia in patients with cancer and hematopoietic stem cell transplantation. Antibiotics (Basel) 12(2):226. https://doi.org/10.3390/antibiotics12020226
Freifeld AG, Bow EJ, Sepkowitz KA, Boeckh MJ, Ito JI, Mullen CA, Raad II, Rolston KV, Young JA, Wingard JR, Infectious Diseases Society of America (2011) Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 52(4):e 56-93. https://doi.org/10.1093/cid/cir073
Heinz WJ, Buchheidt D, Christopeit M, von Lilienfeld-Toal M, Cornely OA, Einsele H, Karthaus M, Link H, Mahlberg R, Neumann S, Ostermann H, Penack O, Ruhnke M, Sandherr M, Schiel X, Vehreschild JJ, Weissinger F, Maschmeyer G (2017) Diagnosis and empirical treatment of fever of unknown origin (FUO) in adult neutropenic patients: guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Medical Oncology (DGHO). Ann Hematol 96(11):1775–1792. https://doi.org/10.1007/s00277-017-3098-3
Averbuch D, Orasch C, Cordonnier C, Livermore DM, Mikulska M, Viscoli C, Gyssens IC, Kern WV, Klyasova G, Marchetti O, Engelhard D, Akova M; ECIL4, a joint venture of EBMT, EORTC, ICHS, ESGICH/ESCMID and ELN. European guidelines for empirical antibacterial therapy for febrile neutropenic patients in the era of growing resistance: summary of the 2011 4th European Conference on Infections in Leukemia. Haematologica. 2013;98(12):1826–35. https://doi.org/10.3324/haematol.2013.091025. Erratum in: Haematologica. 2014;99(2):400.
Averbuch D, Cordonnier C, Livermore DM, Mikulska M, Orasch C, Viscoli C, Gyssens IC, Kern WV, Klyasova G, Marchetti O, Engelhard D, Akova M; ECIL4, a joint venture of EBMT, EORTC, ICHS, ESGICH/ESCMID and ELN. Targeted therapy against multi-resistant bacteria in leukemic and hematopoietic stem cell transplant recipients: guidelines of the 4th European Conference on Infections in Leukemia (ECIL-4, 2011). Haematologica. 2013;98(12):1836–47. https://doi.org/10.3324/haematol.2013.091330.
Gudiol C, Aguilar-Guisado M, Azanza JR et al (2020) Executive summary of the consensus document of the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC), the Spanish Network for Research in Infectious Diseases (REIPI) and the Spanish Society of Haematology and Haemotherapy (SEHH) on the management of febrile neutropenia in patients with hematological malignancies. Enferm Infecc Microbiol Clin (Engl Ed) 38(4):174–181. https://doi.org/10.1016/j.eimc.2019.01.013
Yahav D, Franceschini E, Koppel F, et al. Bacteremia Duration Study Group. Seven versus 14 days of antibiotic therapy for uncomplicated gram-negative bacteremia: a noninferiority randomized controlled trial. Clin Infect Dis. 2019;69(7):1091–1098. https://doi.org/10.1093/cid/ciy1054.
von Dach E, Albrich WC, Brunel AS et al (2020) Effect of C-reactive protein-guided antibiotic treatment duration, 7-day treatment, or 14-day treatment on 30-day clinical failure rate in patients with uncomplicated gram-negative bacteremia: a randomized clinical trial. JAMA 323(21):2160–2169. https://doi.org/10.1001/jama.2020.6348
Molina J, Montero-Mateos E, Praena-Segovia J, et al. SHORTEN trial team. Seven-versus 14-day course of antibiotics for the treatment of bloodstream infections by Enterobacterales: a randomized, controlled trial. Clin Microbiol Infect. 2022;28(4):550–557. https://doi.org/10.1016/j.cmi.2021.09.001.
Turjeman A, von Dach E, Molina J, et al. Duration of antibiotic treatment for Gram-negative bacteremia – Systematic review and individual participant data (IPD) meta-analysis. eClinicalMedicine. 2022;55:101750. https://doi.org/10.1016/j.eclinm.2022.101750.
Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008;36(5):309-32. https://doi.org/10.1016/j.ajic.2008.03.002. Erratum in: Am J Infect Control. 2008;36(9):655.
Nesher L, Rolston KV (2013) Neutropenic enterocolitis, a growing concern in the era of widespread use of aggressive chemotherapy. Clin Infect Dis 56(5):711–717. https://doi.org/10.1093/cid/cis998
Friedman ND, Kaye KS, Stout JE, McGarry SA, Trivette SL, Briggs JP, Lamm W, Clark C, MacFarquhar J, Walton AL, Reller LB, Sexton DJ (2002) Health care-associated bloodstream infections in adults: a reason to change the accepted definition of community-acquired infections. Ann Intern Med 137(10):791–797. https://doi.org/10.7326/0003-4819-137-10-200211190-00007
Korvick JA, Bryan CS, Farber B, Beam TR Jr, Schenfeld L, Muder RR, Weinbaum D, Lumish R, Gerding DN, Wagener MM et al (1992) Prospective observational study of Klebsiella bacteremia in 230 patients: outcome for antibiotic combinations versus monotherapy. Antimicrob Agents Chemother 36(12):2639–2644. https://doi.org/10.1128/AAC.36.12.2639
Singer M, Deutschman CS, Seymour CW et al (2016) The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA 315(8):801–810. https://doi.org/10.1001/jama.2016.0287
Harris PNA, Tambyah PA, Lye DC, et al. MERINO Trial Investigators and the Australasian Society for Infectious Disease Clinical Research Network (ASID-CRN). Effect of piperacillin-tazobactam vs meropenem on 30-day mortality for patients with E coli or Klebsiella pneumoniae bloodstream infection and ceftriaxone resistance: a randomized clinical trial. JAMA. 2018;320(10):984–994. https://doi.org/10.1001/jama.2018.12163. Erratum in: JAMA. 2019;321(23):2370.
Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, Harbarth S, Hindler JF, Kahlmeter G, Olsson-Liljequist B, Paterson DL, Rice LB, Stelling J, Struelens MJ, Vatopoulos A, Weber JT, Monnet DL (2012) Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 18(3):268–281. https://doi.org/10.1111/j.1469-0691.2011.03570.x
Perez F, Adachi J, Bonomo RA. Antibiotic-resistant gram-negative bacterial infections in patients with cancer. Clin Infect Dis. 2014;59 Suppl 5(Suppl 5):S335–9. https://doi.org/10.1093/cid/ciu612.
Protocolos de PCR-Multiplex para Gram negativos. Available at: http://antimicrobianos.com.ar/2023/06/protocolo-de-pcr-para-la-deteccion-del-gen-ctx-m/. Accessed 20 Feb 2024.
Leffler DA, Lamont JT (2015) Clostridium difficile infection. N Engl J Med 372(16):1539–1548. https://doi.org/10.1056/NEJMra1403772
McDonald LC, Gerding DN, Johnson S, Bakken JS, Carroll KC, Coffin SE, Dubberke ER, Garey KW, Gould CV, Kelly C, Loo V, Shaklee Sammons J, Sandora TJ, Wilcox MH (2018) Clinical practice guidelines for clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis 66(7):987–994. https://doi.org/10.1093/cid/ciy149
Metais A, Torregrosa Diaz JM, GallegoHernanz MP, Pichon M, Desmier D, Roblot F, Rammaert B (2022) Efficacy of antibiotic short course for bloodstream infections in acute myeloid leukemia patients with febrile neutropenia: A retrospective comparative study. J Infect 84(1):1–7. https://doi.org/10.1016/j.jinf.2021.10.017
Ranganath N, Yetmar ZA, McCandless AR, Rivera CG, Lahr BD, Tande AJ, Shah AS. Evaluating antimicrobial duration for Gram-negative bacteremia in patients with neutropenia due to hematologic malignancy or hematopoietic stem cell transplantation. Transpl Infect Dis. 2023:e14085. https://doi.org/10.1111/tid.14085.
Herrera F, Torres D, Carena A et al (2023) Short course of antibiotic therapy for gram-negative bacilli bacteremia in patients with cancer and hematopoietic stem cell transplantation: less is possible. Microorganisms 11(2):511. https://doi.org/10.3390/microorganisms11020511
Verlinden A, Jansens H, Goossens H, Anguille S, Berneman ZN, Schroyens WA, Gadisseur AP. Safety and efficacy of antibiotic de-escalation and discontinuation in high-risk hematological patients with febrile neutropenia: a single-center experience. Open Forum Infect Dis. 2021;9(3):ofab624. https://doi.org/10.1093/ofid/ofab624.
Ram R, Halavy Y, Amit O, Paran Y, Katchman E, Yachini B, Kor S, Avivi I, Ben-Ami R (2018) Extended vs bolus infusion of broad-spectrum β-lactams for febrile neutropenia: an unblinded, randomized trial. Clin Infect Dis 67(8):1153–1160. https://doi.org/10.1093/cid/ciy258.Erratum.In:ClinInfectDis.2018;67(7):1151
Laporte-Amargos J, Ulldemolins M, Puig-Asensio M, Tebé C, Castro S, Carratalà J, Gudiol C (2023) Prolonged vs short-term infusion of β-lactam antibiotics for the treatment of febrile neutropenia: a systematic review and meta-analysis. J Infect S0163–4453(23):00368–00377. https://doi.org/10.1016/j.jinf.2023.06.023
Acknowledgements
We thank Valeria Melia, scientific translator at CEMIC-CONICET Research Unit, for English edition of the manuscript. We thank other members of ROCAS study: Patricia Costantini, Martín Luck and Marcelo Bronzi (Instituto de Oncología, Dr. Angel Roffo), Andrea Nenna and María Laura Chaves (Hospital Municipal de Oncología, Marie Curie), José Benso and Verónica Fernández (Hospital Italiano de San Justo), Viviana Vilches (Hospital Universitario Austral) and Fernando Poletta (CEMIC).
Funding
This work was partially supported by Norberto Quirno Foundation (a non-profit entity) scholarship. The Foundation has not participated in the study design, sample collection and analysis and data interpretation, or in the decision to submit the manuscript for publication. Only the authors had full access to the study data files.
Author information
Authors and Affiliations
Consortia
Contributions
F.H, D.T. and A.A.C. conceived and designed the study. F.H. wrote the manuscript. C.G. reviewed and edited the manuscript. A.L.; R.J.; L.T.; N.M.; M.L.P; N.S.; L.B.; M.L.G.I; J.J.E.; S.L.; L.B. and I.R.R. recruited patients and collected clinical data. F.N.; M.P., R.M.; M.B., M.V., M.R.; R.C.; F.P., A.C. and M.R. performed microbiological analysis. D.T. and A.A.C. developed the database and analyzed and interpreted data.
Corresponding author
Ethics declarations
Institutional review board statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the CEMIC Ethics Committee on 30 April 2014 (Approval number 880) and the ethics committees from the different participating institutions.
Informed consent statement
Patient participation in this study was voluntary, and a written informed consent was required until December 2016. Since then, patient consent was waived by the Ethics Committees due to this study continued as a registry of statistic, epidemiological, and scientific data, according to the point 2, article 7 of de law 25326, Protection of Personal Data.
Competing of interests
F.H. has participated in advisory boards and/or received speaker honoraria and grants from Gilead, Knight Therapeutics, Merck, Sharp & Dohme (MSD), TEVA and Pfizer. D.T. has participated in advisory boards and/or received speaker honoraria from Gilead, Knight Therapeutics, MSD and GlaxoSmithKline. R.J. has participated in advisory boards and/or received speaker honoraria from Gilead and Pfizer. L.T. has received speaker honoraria from MSD. C.G. has participated in advisory boards and/or received speaker honoraria from Gilead, MSD, Pfizer and Mundipharma. S.L. has received speaker honoraria from MSD and Pfizer. F. P. has received speaker honoraria from MSD and Pfizer. AAC is an employee of ROCHE Diagnostics with the role of Regional Medical Lead for Latin America. All other authors report no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Study group members are listed in the acknowledgment section.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Herrera, F., Torres, D., Laborde, A. et al. Seven-day antibiotic therapy for Enterobacterales bacteremia in high-risk neutropenic patients: toward a new paradigm. Eur J Clin Microbiol Infect Dis 43, 1741–1751 (2024). https://doi.org/10.1007/s10096-024-04885-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-024-04885-w