Abstract
The primary objective of this study was to evaluate the impact of colonization pressure on intensive care unit (ICU)-acquired multidrug resistant bacteria (MDRB). All patients hospitalized for more than 48 h in the ICU were included in this prospective observational study. MDRB were defined as methicillin resistant Staphylococcus aureus, Pseudomonas aeruginosa resistant to ceftazidime or imipenem, Gram-negative bacilli producing extended-spectrum beta-lactamases (ESBL), and all strains of Acinetobacter baumannii and Stenotrophomonas maltophilia. Colonization pressure was daily calculated in the three participating ICUs. Univariate and multivariate analyses were used to determine risk factors for ICU-acquired MDRB. Two hundreds and four (34%) of the 593 included patients acquired an MDRB during their ICU stay. Multivariate analysis identified colonization pressure as an independent risk factor for ICU-acquired MDRB (OR (95% CI) 4.18 (1.03–17.01), p = 0.046). Other independent risk factors for ICU-acquired MDRB were mechanical ventilation (3.08 (1.28–7.38), p = 0.012), and arterial catheter use (OR, 3.04 (1.38–6.68), p = 0.006). ICU-acquired MDRB were associated with increased mortality, duration of mechanical ventilation, and ICU stay. However, ICU-acquired MDRB was not independently associated with ICU-mortality. Colonization pressure is an independent risk factor for acquiring MDRB in the ICU.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multidrug resistant bacteria (MDRB) are common in critically ill patients, and are frequently reported to be responsible for ICU-acquired infections. Based on the results of the large multinational EPIC II study, 35% of infectious episodes diagnosed in 7,087 patients were related to MDRB [1]. Further, the EUROBACT multinational study showed that MDRB were responsible for 48% of hospital-acquired bacteremia episodes [2]. A more recent large multinational observational study, aiming at assessing the incidence of ventilator-associated lower respiratory tract infections, reported that MDRB were identified as causative pathogens in 61% of patients with ventilator-associated pneumonia (VAP) or ventilator-associated tracheobronchitis [3].
Infections related to MDRB are associated with higher mortality rates, longer duration of mechanical ventilation, and ICU stay [4, 5]. Potential explanations for the high mortality rate in patients with infections related to MDRB include inappropriate initial antibiotic treatment, drug toxicity, and subsequent resistance [6].
The main risk factors for resistance are prior exposure to antibiotics, prolonged hospital and ICU length of stay, invasive devices, comorbidities and local epidemiology [7–10]. Identifying risk factors for ICU-acquired MDRB might be helpful to improve preventive strategies and outcome of critically ill patients. Colonization pressure, defined as the ratio of patients colonized with MDRB relative to all patients, has been reported to be a risk factor for ICU-acquired methicillin-resistant Staphylococus aureus (MRSA) [11], vancomycin resistant enterococcus (VRE) [12], Clostridium difficile [13], Acinetobacter baumannii [14], and Pseudomonas aeruginosa [15]. However, none of these studies evaluated the impact of colonization pressure related to all MDRB on the risk for acquiring these bacteria in the ICU. Therefore, we hypothesized that higher colonization pressure related to MDRB would be associated with increased risk for ICU-acquired MDRB.
The primary aim of this prospective observational study is to determine if colonization pressure for all MDRB is a risk factor for ICU-acquired colonization or infection related to MDRB.
Material and methods
Study design
This prospective observational study was performed during a 13-month period (from January 2007 to January 2008), in three 10-bed medical and surgical ICUs at the University Hospital of Lille, France.
Study Population
All adult patients admitted to the ICU for >48 h were included. Exclusion criteria were length of stay ≤ 48 h, age less than 18 years, and ICU readmission.
Infection control policy included hospitalization in single-bed rooms, adequate hand hygiene, achieved by using an alcohol-based hand rub formulation before and after each patient contact, routine screening for MDRB, written antibiotic treatment protocols, continuous surveillance of nosocomial infections and adequate cleaning of ICU rooms.
Routine screening for MDRB was performed for all patients at ICU admission and weekly thereafter. This screening included nasal and anal swabs. In addition, tracheal aspirates were performed in intubated or tracheotomized patients. Microbiological cultures of other specimens were performed according to clinical status.
In all patients, isolation techniques were used at ICU admission until receipt of screening results. Thereafter, these techniques were performed in all patients with infection or colonization due to MDRB. Preventive isolation techniques were applied in all immunosuppressed patients, during the whole ICU stay. These techniques included protective gowns, gloves, and mask usage.
Nurse to patient ratio was 1:3 in the three participating units.
Data collection and definitions
All data were prospectively collected. MDRB were defined based on our institution’s definition, as MRSA, ceftazidime or imipenem resistant Pseudomonas aeruginosa, ESBL-producing GNB, and all strains of Acinetobacter baumannii, and Stenotrophomonas maltophilia [16].
Daily occupancy rate was defined as the ratio of hospitalized patients to the total number of operational beds per ICU. In patients with MDRB, occupancy rate was defined as the mean of daily occupation rate, from ICU admission until the last MDRB acquisition. In patients with no MDRB, occupancy rate was defined as the mean of daily occupation rate during the whole ICU stay.
Daily colonization index was defined as the ratio of patients with MDRB (infected and/or colonized) to the total number of patients. In patients with MDRB, colonization pressure was determined as the mean of colonization index, from ICU admission until the last MDRB acquisition. In patients with no MDRB, colonization pressure was determined as the mean of colonization index during the whole ICU stay.
In patients with MDRB, percentage of days with antibiotics was defined as the ratio of days with antibiotics, from ICU admission until the last MDRB acquisition. In patients with no MDRB, percentage of days with antibiotics was defined as the ratio of days with antibiotics during the whole ICU stay.
VAP was defined by the presence of new or progressive pulmonary infiltrate, associated with two of the following findings: temperature ≥38.5 °C or <36 °C; leukocyte count ≥10 000/μL or <1500/μL; and purulent sputum or tracheal aspirate. Microbiological confirmation of pneumonia was required, and defined as bronchoalveolar lavage or tracheal aspirate with ≥104 CFU/mL and ≥106 CFU/mL, respectively [17]. Only first episodes of VAP diagnosed > 48 h of mechanical ventilation were taken into account. Other infections were defined according to the modified Centers for Disease Control criteria [18].
The primary objective was to determine the impact of MDRB colonization pressure on the risk for ICU-acquired MDRB colonization or infection. Secondary objective was the impact of ICU-acquired MDRB on outcome.
Statistical analysis
SPSS 11.5 software (SPSS, Chicago, IL, USA) was used for data analysis. Distribution of quantitative variables was tested. Normally distributed and skewed quantitative variables are presented as mean ± SD, and median (interquartile range), respectively. Results of qualitative variables are presented as numbers (percentage). All p values are two-tailed. The statistical significance was set at p < 0.05.
Univariate analysis was used to determine variables associated with acquisition of MDRB during ICU stay. Categorical variables were compared using Pearson chi-square, or Fischer exact test. Quantitative variables were compared using Student’s t-test or Mann–Whitney U test, as appropriate. Exposure to all risk factors for MDRB was taken into account until the last acquisition of MDRB, or ICU discharge, whichever happened first. Multivariate analysis was used to determine variables independently associated with acquisition of MDRB. All predictors showing an association at p <0.1 with infection or colonization caused by MDRB in univariate analysis were included in the multivariate logistic regression analysis. Potential interactions were tested, and goodness of fit was assessed by the Hosmer-Lemeshow test. Cox proportional hazards univariate and multivariate models were also used to determine risk factors for ICU-acquired MDRB.
To determine the impact of ICU-acquired MDRB on outcome, duration of mechanical ventilation, ICU length of stay, and ICU mortality were compared between patients with MDRB, and patients with no MDRB. Risk factors for ICU mortality were determined using univariate analysis. All predictors showing an association at p <0.1 with ICU mortality in univariate analysis were included in the multivariate logistic regression analysis. Potential interactions were tested, and goodness of fit was assessed by the Hosmer-Lemeshow test.
Results
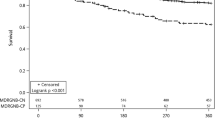
Five hundred ninety-three consecutive patients were included during the study period (Fig. 1), of which 204 (34%) acquired MDRB (colonization and/or infection) during their ICU stay. The most common MDRB were P. aeruginosa (38%), A. baumannii (22%), ESBL-GNB (18%), MRSA (15%), and S. maltophilia (7%). ICU-acquired infection related to MDRB occurred in 111 patients (19%). VAP and ICU-acquired bacteremia were the most common ICU-acquired infections related to MDRB. Prior colonization related to MDRB was significantly higher in patients with ICU-acquired infections related to MDRB compared with those who had infections related to other bacteria (86 of 111 (77%) versus 44 of 108 (41%), p < 0.001, OR (95% CI) 5 (2.8-9)). No outbreak occurred during the study period, but only endemic transmission of MDRB. No significant difference was found in rate of ICU-acquired MDRB between the three study ICUs (89 of 226 (39%), 63 of 181 (35%), and 73 of 186 (39%), p = 0.58). Patient characteristics are presented in Tables 1 and 2.
Risk factors for ICU-acquired MDRB by univariate analysis
At ICU admission, age, SAPS II, LOD score, transfer from other wards, prior antibiotic treatment, surgery, and infection at admission were identified as risk factors for MDRB (Table 1).
During ICU stay, colonization pressure, use and duration of intravascular and urinary catheters, sedation, mechanical ventilation, and ICU length of stay were identified as risk factors for MDRB by univariate analysis (Table 2). Percentage of days with antimicrobials, including pipercillin-tazobactm, fourth generation cephalosporins, carbapenem, fluoroquinolones, aminoglycoside, and glycopeptides was significantly higher in patients with ICU-acquired MDRB, compared with those with no MDRB (Table 3).
Colonization pressure for different ICU-acquired MDRB was also significantly higher in patients with ICU-acquired MDRB compared with those with no MDRB (Fig. 2).
Risk factors for ICU-acquired MDRB by multivariate analysis
Colonization pressure, need for mechanical ventilation and the presence of an arterial catheter were independently associated with ICU-acquired MDRB (Table 4).
Risk factors for ICU-acquired MDRB by univariate and multivariate Cox proportional hazards models
Colonization pressure was also identified as a risk factor for ICU-acquired MDRB by univariate and multivariate Cox proportional hazards models (Table 5).
Impact of ICU-acquired MDRB on outcome
Total duration of mechanical ventilation, length of ICU stay, and ICU mortality rate were significantly higher in patients with ICU-acquired MDRB, compared with those with no ICU-acquired MDRB (Table 2). However, ICU-acquired MDRB was not independently associated with ICU-mortality (Table 6).
Discussion
Our results suggest that colonization pressure is independently associated with ICU-acquired MDRB. Mechanical ventilation and arterial catheter use were also identified as independent risk factors for ICU-acquired MDRB. Further, acquisition of MDRB in the ICU was associated with increased duration of mechanical ventilation, ICU length of stay, and mortality rate. However, ICU-acquired MDRB was not independently associated with ICU mortality.
The strengths of our study are the large number of included patients, the daily calculation of colonization pressure in all participating units and patients, and the fact that this study is the first to evaluate the relationship between colonization pressure and all ICU-acquired MDRB. Previous studies identified colonization pressure as an independent risk factor for specific MDRB. However, none of these studies evaluated the relationship between colonization pressure and all ICU-acquired MDRB. Colonization pressure was previously identified as an independent risk factor for VRE acquisition in a medical ICU with non-individual rooms [12], and in an ICU setting with individualized rooms [19]. Single center and multicenter studies have shown MRSA-related colonization pressure to be independently associated with MRSA acquisition, not only in the ICU setting but also in medicine wards [20, 21]. As far as Gram-negative bacilli in ICU are concerned, two recent studies established colonization pressure as a risk factor for carbapenem-resistant A. baumannii [14] and multiresistant P. aeruginosa [15].
Our results could be explained by the higher risk for cross-transmission of MDRB in units where colonization pressure is high. However, molecular typing was not performed to confirm this hypothesis. Cross-transmission of MDRB might have occurred directly between two patients via health-workers or indirectly via environment. Previous studies clearly showed that the risk for ICU-acquired MDRB was significantly higher when prior room occupant was colonized or infected with MDRB [22–24].
Clinical implications of our findings include the potential usefulness of cohorting patients with MDRB, and the importance of screening for MDRB at ICU admission and during ICU stay. Based on our results and those of previous studies, one could argue that cohorting of staff or of ICU patients with MDRB might be beneficial in reducing transmission of ICU-acquired MDRB. Whilst no interventional study has shown beneficial effect of such an intervention, European Society of Clinical Microbiology and Infectious Diseases guidelines recommend patient cohorting during outbreaks only [25], and European Centers for Disease Control recommends staff cohorting in all settings, and patient cohorting in outbreaks only [26]. However, additional measures such as hand hygiene, environment cleaning, isolation measures, and antibiotic stewardship should be used to reduce cross-transmission of MDRB. Another potential implication of our findings is that preventive strategies aiming at reducing cross-transmission of MDRB should be enhanced in ICUs with high colonization pressure.
The present study clearly argues for a systematic screening of MDRB among ICU patients, thus isolation contact measures could be performed in these patients to stop the spread of MDRB. Recent studies reported conflicting results regarding the efficiency of contact isolation measures [27, 28]. However, recent recommendations clearly encourage physicians to isolate patients with MDRB [25].
Mechanical ventilation and arterial catheter use were identified as independent risk factors for ICU-acquired MDRB. Further, ICU-acquired MDRB are significantly associated with negative impact on outcome. These results are in accordance with other reports [5, 29, 30].
In addition to the above-discussed limitations, this study was performed in a single center, which precludes generalization of its results to other centers. Second, actual workload, compliance with hand hygiene, and room cleansing protocols were not evaluated. Third, our definition of MDRB included all A. baumannii, and S. maltophilia strains. However, if another definition, taking into account only resistant strains of these bacteria, had been used, different study results might have been obtained. Finally, our study was focused on all MDRB, with different virulence, risk factors and prevalence. However, analysis of specific MDRB showed similar results regarding the relationship between colonization pressure and ICU-acquired MDRB.
Conclusion
Colonization pressure is independently associated with ICU-acquired MDRB. Our results suggest that cohorting staff or critically ill patients with MDRB and enhancement of preventive measures in ICUs with high colonization pressure might be beneficial to reduce cross-transmission of MDRB. Further large interventional multicenter studies are required to confirm our findings.
Abbreviations
- ICU:
-
Intensive care unit
- MDRB:
-
Multidrug resistant bacteria
- MRSA:
-
Methicillin resistant Staphylococcus aureus
- VRE:
-
Vancomycin resistant enterococcus
References
Vincent J-L, Rello J, Marshall J, Silva E, Anzueto A, Martin CD et al (2009) International study of the prevalence and outcomes of infection in intensive care units. JAMA 302:2323–2329
Tabah A, Koulenti D, Laupland K, Misset B, Valles J, Bruzzi De Carvalho F et al (2012) Characteristics and determinants of outcome of hospital-acquired bloodstream infections in intensive care units: The EUROBACT International Cohort Study. Intensive Care Med 38:1930–1945
Martin-Loeches I, Povoa P, Rodríguez A, Curcio D, Suarez D, Mira J-P et al (2015) Incidence and prognosis of ventilator-associated tracheobronchitis (TAVeM): a multicentre, prospective, observational study. Lancet Res Med 3:859–868
Melsen WG, Rovers MM, Groenwold RHH, Bergmans DCJJ, Camus C, Bauer TT et al (2013) Attributable mortality of ventilator-associated pneumonia: A meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis 13:665–671
Bassetti M, De Waele JJ, Eggimann P, Garnacho-Montero J, Kahlmeter G, Menichetti F et al (2015) Preventive and therapeutic strategies in critically ill patients with highly resistant bacteria. Intensive Care Med 41:776–795
Barbier F, Lisboa T, Nseir S (2016) Understanding why resistant bacteria are associated with higher mortality in ICU patients. Intensive Care Med 42:2066–2069
Garnacho-Montero J, Dimopoulos G, Poulakou G, Akova M, Cisneros JM, De Waele J et al (2015) Task force on management and prevention of Acinetobacter baumannii infections in the ICU. Intensive Care Med 41:2057–2075
Karam G, Chastre J, Wilcox MH, Vincent J-L (2016) Antibiotic strategies in the era of multidrug resistance. Crit Care 20:136
Micek ST, Wunderink RG, Kollef MH, Chen C, Rello J, Chastre J et al (2015) An international multicenter retrospective study of Pseudomonas aeruginosa nosocomial pneumonia: impact of multidrug resistance. Crit Care 19:219
Martin-Loeches I, Torres A, Rinaudo M, Terraneo S, de Rosa F, Ramirez P D, E, Fernández-Barat L, Li Bassi GL FM. Resistance patterns and outcomes in intensive care unit (ICU)-acquired pneumonia. J Infect 2015;70:213–22
Williams VR, Callery S, Vearncombe M, Simor AE (2009) The role of colonization pressure in nosocomial transmission of methicillin-resistant Staphylococcus aureus. Am J Infect Control 37:106–110
Bonten MJ, Slaughter S, Ambergen AW, Hayden MK, van Voorhis J, Nathan C et al (1998) The role of “colonization pressure” in the spread of vancomycin-resistant enterococci: an important infection control variable. Arch Intern Med 158:1127–1132
Lawrence SJ, Puzniak LA, Shadel BN, Gillespie KN, Kollef MH, Mundy LM (2007) Clostridium difficile in the intensive care unit: epidemiology, costs, and colonization pressure. Infect Control Hosp Epidemiol 28:123–130
Arvaniti K, Lathyris D, Ruimy R, Haidich A-B, Koulourida V, Nikolaidis P et al (2012) The importance of colonization pressure in multiresistant Acinetobacter baumannii acquisition in a Greek intensive care unit. Crit Care 16:R102
Cobos-Trigueros N, Solé M, Castro P, Torres JL, Hernández C, Rinaudo M et al (2015) Acquisition of Pseudomonas aeruginosa and its resistance phenotypes in critically ill medical patients: role of colonization pressure and antibiotic exposure. Crit Care 19:218
Ledoux G, Six S, Lawson R, Labreuche J, Blazejewski C, Wallet F (2016) Impact of a targeted isolation strategy at ICU-admission on ICU-acquired infection related to multidrug-resistant bacteria. Clin Microbiol Infect 22:888.e11–e18
American Thoracic Society; Infectious Diseases Society of America (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171:388–416
Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM (1988) CDC definitions for nosocomial infections, 1988. Am J Infect Control 16:128–140
Drees M, Snydman DR, Schmid CH, Barefoot L, Hansjosten K, Vue PM et al (2008) Prior environmental contamination increases the risk of acquisition of vancomycin-resistant enterococci. Clin Infect Dis 46:678–685
Merrer J, Santoli F, Appéré de Vecchi C, Tran B, De Jonghe B, Outin H (2000) Colonization pressure and risk of acquisition of methicillin-resistant Staphylococcus aureus in a medical intensive care unit. Infect Control Hosp Epidemiol 21:718–23
Bloemendaal ALA, Fluit AC, Jansen WMT, Vriens MR, Ferry T, Argaud L et al (2009) Acquisition and cross-transmission of Staphylococcus aureus in European intensive care units. Infect Control Hosp Epidemiol 30:117–124
Nseir S, Blazejewski C, Lubret R, Wallet F, Courcol R (2011) Durocher a. Risk of acquiring multidrug-resistant Gram-negative bacilli from prior room occupants in the intensive care unit. Clin Microbiol Infect 17:1201–1208
Passaretti CL, Otter JA, Reich NG, Myers J, Shepard J, Ross T et al (2013) An evaluation of environmental decontamination with hydrogen peroxide vapor for reducing the risk of patient acquisition of multidrug-resistant organisms. Clin Infect Dis 56:27–35
Blazejewski C, Wallet F, Rouzé A, Le Guern R, Ponthieux S, Salleron J et al (2015) Efficiency of hydrogen peroxide in improving disinfection of ICU rooms. Crit Care 19:30
Tacconelli E, Cataldo MA, Dancer SJ, De Angelis G, Falcone M, Frank U et al (2014) ESCMID guidelines for the management of the infection control measures to reduce transmission of multidrug-resistant Gram-negative bacteria in hospitalized patients [Internet]. Clin Microbiol Infect 20(1):1–55
European Center for Disease Prevention and Control (2011) Annual Epidemiological Report 2011. Reporting on 2009 surveillance data and 2010 epidemic intelligence data. Stockholm: ECDC, 2011. http://www.ecdc.europa.eu. Accessed 13 December 2016
Cohen CC, Cohen B, Shang J (2015) Effectiveness of contact precautions against multidrug-resistant organism transmission in acute care: a systematic review of the literature. J Hosp Infect 90:275–284
Kullar R, Vassallo A, Turkel S, Chopra T, Kaye KS, Dhar S (2016) Degowning the controversies of contact precautions for methicillin-resistant Staphylococcus aureus: A review. Am J Infect Control 44:97–103
Lucet J-C, Bouadma L, Zahar J-R, Schwebel C, Geffroy A, Pease S et al (2010) Infectious risk associated with arterial catheters compared with central venous catheters. Crit Care Med 38:1030–1035
Mantzarlis K, Makris D, Manoulakas E, Karvouniaris M, Zakynthinos E (2013) Risk factors for the first episode of Klebsiella pneumoniae resistant to carbapenems infection in critically ill patients: a prospective study. Biomed Res Int 850547
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
SN Bayer, Basilea (advisory board); other authors, none.
Funding
None.
Ethical approval
The local IRB (Comité de Protection des Personnes Nord) approved the study.
Informed consent
No informed consent was required by the local IRB, because of the non-interventional design of the study.
Rights and permissions
About this article
Cite this article
Masse, J., Elkalioubie, A., Blazejewski, C. et al. Colonization pressure as a risk factor of ICU-acquired multidrug resistant bacteria: a prospective observational study. Eur J Clin Microbiol Infect Dis 36, 797–805 (2017). https://doi.org/10.1007/s10096-016-2863-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-016-2863-x