Abstract
Background
Topographical disorientation (TD) refers to a particular condition which determines the loss of spatial orientation, both in new and familiar environments. TD and spatial memory impairments occur relatively early as effect of cognitive decline in aging, even in prodromal stages of dementia, namely mild cognitive impairment (MCI).
Aims
(a) To show that components linked to the recall of familiar spatial knowledge are relatively spared with respect to the learning of unfamiliar ones in normal aging, while they are not in MCI, and (b) to investigate gender differences for their impact on egocentric and allocentric frames of reference.
Method
Forty young participants (YC), 40 healthy elderly participants (HE), 40 elderly participants with subjective memory complaints (SMC), and 40 elderly with probable MCI were administered with egocentric and allocentric familiar tasks, based on the map of their hometown, and with egocentric and allocentric unfamiliar tasks, based on new material to be learned. A series of general linear models were used to analyze data.
Results
No group differences were found on egocentric task based on familiar information. MCI performed worse than the other groups on allocentric tasks based on familiar information (YC = HE = SMC > MCI). Significant differences emerged between groups on egocentric and allocentric tasks based on unfamiliar spatial information (YC > HE = SMC > MCI). A gender difference was found, favoring men on allocentric unfamiliar task.
Conclusion
Familiarity of spatial memory traces can represent a protective factor for retrospective components of TD in normal aging. Conversely, using newly learned information for assessment may lead to overestimating TD severity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Topographical disorientation (TD) is defined by the seminal work by Aguirre and D’Esposito [1] as a particular condition which determines the loss of spatial orientation and is characterized by the difficulty in acquiring spatial information in new and unknown environments, and in encompassing familiar environments such as one’s neighborhoods or one’s house. TD and spatial memory impairments occur relatively early as effect of cognitive decline in aging, and it is possible to observe transient episodes of TD, other than in people suffering from dementia, especially of Alzheimer’s type [2], even in prodromal stages of dementia, defined as mild cognitive impairment (MCI) [3]. Following Aguirre and D’Esposito taxonomy, TD is not a unitary concept, but it is possible to split it into four components: egocentric disorientation, heading/allocentric disorientation, landmark agnosia, and anterograde disorientation.
As pointed out by a recent review [4], in the evaluation of TD it is necessary to take into account that older memories are less prone to be impaired by the effect of aging, compared with the newly learned information. It is noteworthy that we can distinguish between egocentric and heading/allocentric disorientation components which refer to previously learned information (i.e., retrograde amnesia), from egocentric and heading/allocentric disorientation components which refers to all the learning processes in order to acquire new spatial memory traces (i.e., anterograde amnesia). It is worthwhile to note that familiarity/remoteness of spatial memory traces can represent a protective factor for TD in aging [5]. This statement is supported by neuroanatomic evidences that familiar environments seem to be mainly processed by an extended temporal-frontal network, whereas recently learned environments require activation in the parahippocampal cortex and in the parietal and occipital lobes [6].
Most studies requested elderly people to deal with recently learned information, which has not yet had sufficient time to be consolidated and transferred to brain structures other than the hippocampus [7], and this may in part account for the difficulties in the use of egocentric and even more allocentric spatial information. Thus, there could be an overestimation bias when observing a decreased performance in both egocentric and allocentric tasks in elderly with a normal cognition compared with young or adult people.
Although most studies investigating egocentric and allocentric spatial memory employed tasks based on learning new information, the relevance of remote spatial information is supported by a limited but growing number of researches. The seminal works by Evans and Pezdek [8] and Thorndyke and Hayes-Roth [9] introduced the concept of familiarity related to spatial information, showing that environments learned through direct and continuous experience and navigation were better represented and remembered than environments learned through indirect experience (i.e., map study). Behavioral and cognitive studies employing familiar information with young and old participants found similar results, reporting no significant age effect [5, 10,11,12,13,14,15]. Also, neuroscience and neuroimaging studies have provided relevant support for the effect of familiarity in spatial memory [16, 17].
A recent contribution by Lopez and colleagues [5] investigated recent and well-consolidated spatial memory within the egocentric and allocentric frames of reference in young and healthy elderly people. Young and healthy elderly participants were tested on four spatial tasks requiring allocentric and egocentric judgments, based on recent and remote spatial information. Results showed an age effect on both egocentric and allocentric tasks in recently learned spatial information. The age effect was not present in tasks regarding well-consolidated spatial information. A gender effect favoring males was found in performance in allocentric tasks regarding both recent and well-consolidated memory.
All the studies cited so far dealt with familiarity in spatial memory traces in healthy elderly. Several studies investigated egocentric and allocentric spatial memory in people with MCI, using tasks based on new information to be learned, and all of them found a lower performance at least in allocentric spatial memory with respect to healthy elderly [18,19,20,21,22,23,24,25,26]. Moreover, very few studies included also people with subjective memory complaints (SMC) and found that they showed a performance comparable with healthy elderly in tasks based on new information to be learned [e.g., 17]. The SMC is a condition in which individuals complain to have a memory impairment, but no clear impairment on objective psychometric memory test is detectable [27, 28]. The functional meaning of this condition is still debated; nonetheless, it is considered as a subtle marker of a subsequent state of cognitive impairment [29]. No studies, to our knowledge, addressed the topic of familiarity of spatial information in people with SMC and MCI.
The aim of the present study is twofold: the first is to show that components linked to the recall of familiar spatial knowledge (which deficits results in egocentric and allocentric disorientation) are relatively spared with respect to the learning of unfamiliar ones (which deficit results in anterograde amnesia) in normal aging, while they are not in prodromal stage of dementia, namely in MCI. The second aim is to investigate gender differences for their impact on egocentric and allocentric frames of reference. An advantage of men on allocentric frame of reference is expected [5].
Methods and materials
Participants
Forty healthy young and one hundred twenty elderly participants were recruited and took part in the study. All the participants were from the metropolitan area of Bari, Italy. Elderly participants were divided into three groups: forty healthy elderly (HE), forty elderly participants with subjective memory complaints (SMC), forty participants with probable MCI (MCI). Young participants (YC) were university students from introductory courses in psychology. Elderly participants were recruited from senior centers, third-age universities with a support of a proxy informant, generally undergraduate or graduate students, trainees, employers of the centers, and also general practitioners. They were blinded to the hypothesis of the study and signed a consent form for participating. The Ethical Committee of the Institution approved the study protocol, and the whole study was performed following Helsinki Declaration and its later amendments.
Materials and procedure
Neuropsychological tests
All the participants were consecutively enrolled between May 2017 and March 2018. Elderly participants were administered (a) a general anamnesis, carried out by supervised trainees in psychogeriatric care assessment, in order to exclude people with a history of suspected uncompensated systemic/traumatic/psychiatric diseases, or with severe vision/hearing loss, which could have affected cognition, and (b) a standardized neuropsychological battery, in order to establish a diagnosis of SMC or probable MCI, according to the MCI working group of the European Consortium on AD [30]. Global cognitive function was evaluated by the Montreal Cognitive Assessment (MoCA) [31,32,33,34]. A possible occurrence of functional decline usually associated with a severe cognitive impairment and with dementia was evaluated by the activities of daily living (ADL) and instrumental activities of daily living (IADL) [35]. The 15-item version of the Geriatric Depression Scale (GDS) [36] was administered in order to exclude major depressive symptoms. Subjective complaints regarding memory loss were evaluated by the Subjective Memory Complaints questionnaire (SMCq) [28, 37]. Verbal episodic memory was evaluated by the Rey Auditory Verbal Learning Test [38], with both immediate and delayed recall. Executive function was evaluated by the frontal assessment battery (FAB) [39] and the clock-drawing test (CDT) [40]. Regarding the group of MCI participants, 26 were diagnosed as probable amnesic MCI and 14 as probable non-amnesic MCI. No young participants were excluded, while 13 elderly participants were excluded after the general anamnesis for the following reasons: 5 for suspected uncompensated systemic diseases, 2 for suspected uncompensated psychiatric diseases, 2 for severe vision loss, 4 for severe hearing loss.
Spatial tests
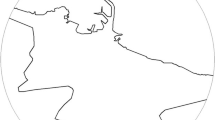
All the participants were administered three spatial tests, two of them used to assess egocentric (Landmark Positioning along a Route (LPR)) and allocentric (Landmark Positioning on a Map (LPM)) spatial memory based on remote topological familiar information. Such tasks were described in detail elsewhere [4, 5]. Briefly, in LPR task, participants had to identify and pinpoint the correct position of eight well-known landmarks of their hometown on a path orally described by the research assistant (see Fig. 1). The score consisted of a single measure over correct position and correct side, ranging from 0 to a maximum of 36 points. In LPM task, participants had to identify and pinpoint the correct position of eight well-known landmarks of their hometown, different from those used in LPR task, on a blind map of the city, keeping in mind metric (i.e., relative distances) as well as categorical (“A is above/below and left/right of B”) spatial relationship between landmarks. The total score for the LPM task ranged from 0 to 56 points (see Fig. 2)
.
The third test, namely the Ego-Allo Task (EAT) [41], was composed of two subtasks and was used to assess egocentric and allocentric spatial memory based on recent and newly learned spatial information, in a table-top, non-topological format. Participants were instructed to memorize the position and the characteristics of three three-dimensional solids (shape and color). In the testing phase, solids were presented on the table, in order to judge distances between them and the observer (egocentric judgment), or between the solids themselves (allocentric judgment). The maximum total score was 8 points for egocentric and 8 points for allocentric judgment, respectively.
The entire procedure was made clear to the participants beforehand. Participants were assessed individually in a well-lit and quiet room without disturbances. Data were collected in one session. The whole assessment lasted a maximum of 2 h. The order of the tasks was the same mentioned in the text. Breaks were allowed upon request.
Statistical analyses
Data were analyzed using R 3.5.1 statistical software. We obtained demographic data (gender, age, and years of education) and scores on neuropsychological and spatial tests. A one-tailed value of p < .05 was determined to be statistically significant. Pearson’s chi-squared analysis was performed to assess for differences in the distribution of gender among the four groups. A series of univariate analyses of variance (ANOVAs) were carried out to compare means from the three diagnostic groups for demographic continuous data, and scores on neuropsychological tests used to establish a diagnosis of SMC and probable MCI. A series of multivariate general linear models (GLMs) were performed in order to compare means on the spatial tests depending on group, gender, and education scores. GLMs were replicated excluding the group of young participants in order to compare means on the spatial tests depending on three diagnostic groups of elderly participants, gender, age, education, and depression scores. Pairwise comparisons were carried out on univariate results of each GLM for each outcome with Tukey’s honestly significant difference (HSD) post hoc tests. Effect size estimates were reported by calculating partial eta squared for each effect on continuous outcome and W for each effect regarding association between categorical variables.
Results
Table 1 reports socio-demographic characteristics of the participants, mean scores and standard deviations on the neuropsychological screening tests for the elderly groups, and statistical tests for their differences. The three elderly groups differed by age: HE was on average younger than MCI, while no differences were found among SMC and the two other groups. The four groups did not show any difference by gender distribution, but differed by schooling years, with YC being on average more educated than HE, which in turn were more educated than SMC and MCI. The three groups of elderly participants showed significant differences on the neuropsychological screening tests. HE showed to be less depressed than SMC, but both groups did not show significant differences in level of depression with respect to MCI. The latter group had a MoCA score significantly lower than HE and SMC. No differences were found among the three groups on ADL and IADL. SMC showed a significant difference in SMCq score with respect to the other groups. MCI obtained significant lower scores in the RAVLT immediate and delayed recall, FAB, and CDT scores.
A multivariate general linear model (GLM) was performed in order to test for the hypothesis that group, gender, and education years could have a significant impact on total score of the LPR task and on total score of the LPM task as outcome variables. Results showed a main effect of group (Wilks’ lambda = 0.624, p < .001). Education years, gender, and the interaction term gender × group were not significant. Follow-up univariate analyses were conducted, as appropriate, to test for main and interactions effects on univariate measures. No main nor interaction effects emerged for group, gender, and education years on total score the LPR task. A main effect of group was found on total score of the LPM task (F(3,151) = 28.57, p < .001, ɳ2p = 0.36). Tukey’s HSD post hoc analysis revealed that MCI had a lower performance in LPM task with respect to the other three groups, which did not show significant differences among them (see Fig. 3)
.
A second multivariate GLM was performed with the same predictors on total score of the egocentric task and on total score of the allocentric task of the EAT as outcome variables. Results showed again a main effect of group (Wilks’ lambda = 0.495, p < .001). Education years, gender, and the interaction term gender × group were not significant. Regarding univariate analyses, a main effect of group emerged for egocentric task (F(3,151) = 34.71, p < .001, ɳ2p = 0.41). Tukey’s HSD post hoc revealed that MCI had a significant lower performance compared with that of HE and SMC, which in turn had a lower performance with respect to YC on the egocentric task (see Fig. 4, left panel). For the allocentric task, a main effect of group (F(3,151) = 23.61, p < .001, ɳ2p = 0.32), a main effect of gender (F(1,151) = 4.40, p < 05, ɳ2p = 0.03), and an interaction effect group × gender (F(3,151) = 3.41, p < .05, ɳ2p = 0.06) were found to be significant. Tukey’s HSD post hoc revealed a similar pattern of results for the four groups on allocentric score, and a slight advantage for man with respect to women. Regarding the interaction effect group × gender, the extent of differences in allocentric score for men with respect to women with MCI was significantly larger than in the other three groups (see Fig. 4, right panel)
.
The two previous GLMs were re-run without the group of YC, in order to include age and GDS scores as additional predictors, for which significant differences among the three elderly groups were found. Analyses yielded the same pattern of results for LPR (no significant effects of any predictor) and LPM (main effect of group F(2,111) = 28.07, p < .001, ɳ2p = 0.34) tasks. Tukey’s HSD post hoc found a significant lower performance for MCI with respect to both HE and SMC for LPM task. Regarding egocentric score of Ego-Allo Task, again a main effect of group was significant (F(2,111) = 26,18, p < .001, ɳ2p = 0.32), with MCI having a lower performance with respect to the other two groups of elderly. Regarding allocentric score of Ego-Allo Task, a main effect of group (F(2,111) = 14.86, p < .001, ɳ2p = 0.21) and a main effect of gender (F(1,111) = 7.21, p < .01, ɳ2p = 0.06) were found to be significant. Again, MCI had a lower performance with respect to the other two groups of elderly, and men obtained higher scores than women. No interaction effects emerged.
Discussion
The present study aimed to give a further contribution to the investigation of recent and remote spatial memory in healthy young participants and elderly people with and without cognitive impairments.
The increase of age seemed to affect allocentric spatial representation of remote and familiar information only in presence of MCI. Egocentric spatial representation of familiar information seems to be spared, even in presence of MCI. This differential vulnerability across egocentric and allocentric representations of familiar environments was shown in previous studies and supported by neuropsychological evidences regarding the preserved egocentric component of spatial memory from neurodegenerative processes [42,43,44]. Thus, information consolidated across a huge number of retrieval episodes seems more solidly preserved in elderly people, and it is likely that the consolidation is preserved in areas other than the hippocampus [6, 45, 46]. Our results are consistent with aforementioned investigations on the effect of familiarity on spatial memory [4, 5, 10,11,12] and substantiate the usefulness of familiar spatial tasks also in case of an initial cognitive impairment.
A progressive decrease in performance on both egocentric and allocentric tasks based on recent and unfamiliar spatial information emerged. It is worthwhile to note that in pathological aging, especially in the MCI condition, participants showed impairments in several spatial tasks, such as map drawing, landmark location, direction pointing, and navigation in both real and virtual environments [e.g., 18, 44]. Recent studies [24, 47] have shown differences in egocentric and allocentric learning and memory in navigational space, being the egocentric component spared and the allocentric one affected in both MCI and AD with respect to control participants. What is common among these studies is the manipulation of recent information. Our results are in line with those findings; indeed, MCI participants showed a decline in the performance on both egocentric and allocentric tasks based on recent information.
To the best of our knowledge, this is the first study that uses ecological tasks in both normal and pathological aging. As shown in a previous research [5, 48], the multiple trace theory (MTT) [7] framework supports the idea that in normal aging, old memories are less prone to disruption than recent memories because of their frequent reinstatement of traces. When information is well established in memory, egocentric representation seems to be fairly accessible for the elderly, even with a probable cognitive impairment. Allocentric ones are compromised only in people with MCI. It is reasonable to support the idea that the magnitude of difference between egocentric and allocentric judgments in normal aging is somewhat attributable to the characteristic of spatial tasks, showing a continuum for tasks based on well-consolidated information. Overall, well-consolidated information seemed to be better preserved in memory and less prone to the impairment with respect to newly learned ones in healthy elderly and in elderly with SMC with respect to elderly with MCI. Regarding SMC participants, we supposed that they are aware of their memory failures, although not recognizable by objective neuropsychological tests. This condition does not require objective sign of cognitive impairment [27], so the results shall be considered plausible also in the light of the MTT.
A more recent theoretical framework, the default–executive coupling hypothesis of aging (DECHA) [49], suggests that autobiographical memory becomes increasingly semanticized over the adult lifespan, and the engagement of such representations is associated with preserved cortical thickness in lateral and anterior temporal lobe regions. Elderly people may then still rely to crystalized cognition in order to efficiently solve goal-directed thoughts or actions.
However, another factor may also explain the observed results. Indeed, the two kinds of tasks (i.e., tasks based on familiar and unfamiliar spatial information) differed also on another relevant dimension, that is, the type of space. Familiar tasks were based on topological information representative of navigational/environmental space, while unfamiliar tasks were based on table-top non-topological information representative of reaching/peripersonal space of the participants. Thus, it could also be plausible that MCI participants showed deficits in processing egocentric representation only in the reaching/peripersonal space, being the egocentric processing in navigational/environmental space unaffected by the cognitive decline. This result is supported by studies assessing spatial judgments in peripersonal and extrapersonal space [41, 50]. For example, Iachini and colleagues [50], in two experiments in which participants had to judge if two balls were to collide or not, found a worst performance in peripersonal than in extrapersonal space. Moreover, another study [51] investigated brain areas in stroke patients associated with visual neglect in peripersonal and extrapersonal space in egocentric and allocentric frames of reference. Results showed that the right parahippocampal gyrus, hippocampus, and thalamus were associated with egocentric peripersonal neglect as measured with a cancelation task. It is plausible that the same brain areas are involved with respect to tasks tapping egocentric reaching/peripersonal space in participants with MCI, although more research is needed on this topic.
An overall gender difference was found for elderly participants, favoring men on allocentric unfamiliar task. The extent of such difference was greater for MCI than for the other two elderly groups. This result partially confirmed those of previous studies, which found an advantage of elderly men on allocentric familiar and unfamiliar tasks [5, 48]. Nori and colleagues [52] reported similar results on young participants, showing that gender differences disappeared when women had the necessary time to acquire spatial information, and that retrieving environmental information through the use of allocentric coordinates was more difficult than the use of egocentric ones. Also, another previous study reported a greater accuracy and speed of men than women in a buildings’ location task [53], and this difference was particularly evident in participants unfamiliar with the environment. It seems that familiarity may be relevant to dissolve gender differences in both young and elderly people, although more research is needed on this topic.
Familiarity of spatial memory traces can thus represent a protective factor for retrospective components of TD in normal aging. Conversely, using newly learned information for assessment may lead to overestimating TD severity as it combines two contributing factors, namely heading/allocentric disorientation and anterograde agnosia.
Limitations
The present study has some limitations due to different features of familiar and unfamiliar tasks employed. The first one is due to a different modality in the acquisition of spatial information: in the case of remote information related to the hometown, it is possible to suppose that an unintentional learning process has happened throughout the entire life of the individual, while in the case of recent information, a form of intentional learning based on direct instruction has happened. This feature is difficult to be overcome, since we do not have control on spatial learning processes occurred during lifetime, and constitutes a somewhat insuperable limitation, due to the intrinsic nature of the concept of familiarity [4]. The second one is related to the format of the information: familiar tasks gather spatial information based on a direct navigation of the environment, while unfamiliar tasks rely on spatial information acquired through a table-top format. The third one is related to the format of response: hometown tasks rely on a non-verbal response, while unfamiliar tasks require a verbal response. The fourth one deals with the charge of each task: indeed, familiar tasks require to represent/process a number of elements (i.e., 8 landmarks for each task) which is more than twice the number of elements to be processed in unfamiliar tasks (i.e., only 3 solids for each task). Lastly, the type of judgment that the two types of tasks require is different: familiar tasks require participants to recall and localize positions between landmarks, while unfamiliar tasks require participants to recall and judge distances between objects. Those weaknesses limit the possibility to draw a direct comparison between familiar and unfamiliar tasks. In order to remediate to the latter two limitations, it would be appropriate to build equivalent tasks for familiar and unfamiliar environments, in order to compare them directly.
Conclusion
The advantage of supplementing neuropsychological evaluation of spatial cognition with tasks based on familiar information is to avoid a penalty for healthy elderly, which may have an initial, but not pathological, decline in working memory as well as in learning abilities. Once that a person with MCI or with an early AD condition has lost the ability to learn new information, we can rely on remote familiar information in order to monitor the progression of the neurodegenerative disorder. This is relevant also in order to assess the residual degree of autonomy patients have in their daily activities and to plan restorative or compensatory interventions. Proposing tools, which rely on remote spatial knowledge, in addition to those based on recent spatial information, may help the clinician to conduct a fair evaluation of spatial abilities closely linked to a functional activity of daily living, i.e., the ability to navigate the environment and to travel from one place to another in one’s own hometown or neighborhood.
Further research should investigate the role of familiarity with spatial information in elderly people with different types of cognitive impairment, i.e., elderly with amnesic and non-amnesic as well as with single and multiple domain MCI, and persons suffering from Alzheimer's disease.
References
Aguirre GK, D’Esposito M (1999) Topographical disorientation: a synthesis and taxonomy. Brain 122(9):1613–1628. https://doi.org/10.1093/brain/122.9.1613
Pai M, Jacobs WJ (2004) Topographical disorientation in community-residing patients with Alzheimer’s disease. Int J Geriatr Psychiatry 19(3):250–255. https://doi.org/10.1002/gps.1081
Petersen RC, Caracciolo B, Brayne C, Gauthier S, Jelic V, Fratiglioni L (2014) Mild cognitive impairment: a concept in evolution. J Intern Med 275(3):214–228. https://doi.org/10.1111/joim.12190
Lopez A, Caffò AO, Bosco A (2018) Topographical disorientation in aging. Familiarity with the environment does matter. Neurol Sci 39(9):1519–1528. https://doi.org/10.1007/s10072-018-3464-5
Lopez A, Caffò AO, Spano G, Bosco A (2019) The effect of aging on memory for recent and remote egocentric and allocentric information. Exp Aging Res 45(1):57–73. https://doi.org/10.1080/0361073X.2018.1560117
Boccia M, Nemmi F, Guariglia C (2014) Neuropsychology of environmental navigation in humans: review and meta-analysis of FMRI studies in healthy participants. Neuropsychol Rev 24(2):236–251. https://doi.org/10.1007/s11065-014-9247-8
Moscovitch M, Rosenbaum RS, Addis DR, Westmacott R, Grady C, McAndrews MP, Levine B, Black S, Winocur G, Nadel L (2005) Functional neuroanatomy of remote episodic, semantic and spatial memory: a unified account based on multiple trace theory. J Anat 207(1):35–66. https://doi.org/10.1111/j.1469-7580.2005.00421.x
Evans GW, Pezdek K (1980) Cognitive mapping: knowledge of real-world distance and location information. J Exp Psychol Hum Learn 6(1):13–24. https://doi.org/10.1037/0278-7393.6.1.13
Thorndyke PW, Hayes-Roth B (1982) Differences in spatial knowledge acquired from maps and navigation. Cogn Psychol 14(4):560–589. https://doi.org/10.1016/0010-0285(82)90019-6
Merriman NA, Ondřej J, Roudaia E, O’Sullivan C, Newell FN (2016) Familiar environments enhance object and spatial memory in both younger and older adults. Exp Brain Res 234(6):1555–1574. https://doi.org/10.1007/s00221-016-4557-0
Campbell JI, Hepner IJ, Miller LA (2014) The influence of age and sex on memory for a familiar environment. J Environ Psychol 40:1–8. https://doi.org/10.1016/j.jenvp.2014.04.007
Kirasic KC (1991) Spatial cognition and behavior in young and elderly adults: implications for learning new environments. Psychol Aging 6(1):10–18. https://doi.org/10.1037/0882-7974.6.1.10
Rosenbaum RS, Winocur G, Binns MA, Moscovitch M (2012) Remote spatial memory in aging: all is not lost. Front Aging Neurosci 4. https://doi.org/10.3389/fnagi.2012.00025
Meneghetti C, Borella E, Fiore F, De Beni R (2013) The ability to point to well-known places in young and older adults. Aging Clin Exp Res 25(2):203–209. https://doi.org/10.1007/s40520-013-0027-8
Muffato V, Della Giustina M, Meneghetti C, De Beni R (2015) Age-related differences in pointing accuracy in familiar and unfamiliar environments. Cogn Process 16(1):313–317. https://doi.org/10.1007/s10339-015-0720-y
Rosenbaum RS, Priselac S, Köhler S, Black SE, Gao F, Nadel L, Moscovitch M (2000) Remote spatial memory in an amnesic person with extensive bilateral hippocampal lesions. Nat Neurosci 3(10):1044–1048. https://doi.org/10.1038/79867
Rosenbaum RS, Gao F, Richards B, Black SE, Moscovitch M (2005) ‘Where to?’ Remote memory for spatial relations and landmark identity in former taxi drivers with Alzheimer’s disease and encephalitis. J Cogn Neurosci 17(3):446–462. https://doi.org/10.1162/0898929053279496
DeIpolyi AR, Rankin KP, Mucke L, Miller BL, Gorno-Tempini ML (2007) Spatial cognition and the human navigation network in AD and MCI. Neurology 69(10):986–997. https://doi.org/10.1212/01.wnl.0000271376.19515.c6
Hort J, Laczó J, Vyhnálek M, Bojar M, Bureš J, Vlček K (2007) Spatial navigation deficit in amnestic mild cognitive impairment. Proc Natl Acad Sci U S A 104(10):4042–4047. https://doi.org/10.1073/pnas.0611314104
Lim TS, Iaria G, Moon SY (2010) Topographical disorientation in mild cognitive impairment: a voxel-based morphometry study. J Clin Neurol 6(4):204–211. https://doi.org/10.3988/jcn.2010.6.4.204
Weniger G, Ruhleder M, Lange C, Wolf S, Irle E (2011) Egocentric and allocentric memory as assessed by virtual reality in individuals with amnestic mild cognitive impairment. Neuropsychologia 49(3):518–527. https://doi.org/10.1016/j.neuropsychologia.2010.12.031
Benke T, Karner E, Petermichl S, Prantner V, Kemmler G (2014) Neuropsychological deficits associated with route learning in Alzheimer disease, MCI, and normal aging. Alzheimer Dis Assoc Disord 28(2):162–167. https://doi.org/10.1097/WAD.0000000000000009
Rusconi ML, Suardi A, Zanetti M, Rozzini L (2015) Spatial navigation in elderly healthy subjects, amnestic and non amnestic MCI patients. J Neurol Sci 359(1–2):430–437. https://doi.org/10.1016/j.jns.2015.10.010
Boccia M, Silveri MC, Sabatini U, Guariglia C, Nemmi F (2016) Neural underpinnings of the decline of topographical memory in mild cognitive impairment. Am J Alzheimers Dis Other Dement 31(8):618–630. https://doi.org/10.1177/1533317516654757
Caffò AO, Lopez A, Spano G, Serino S, Cipresso P, Stasolla F, Savino M, Lancioni GE, Riva G, Bosco A (2018) Spatial reorientation decline in aging: the combination of geometry and landmarks. Aging Ment Health 22(10):1372–1383. https://doi.org/10.1080/13607863.2017.1354973
Mohammadi A, Kargar M, Hesami E (2018) Using virtual reality to distinguish subjects with multiple- but not single-domain amnestic mild cognitive impairment from normal elderly subjects. Psychogeriatrics 18(2):132–142. https://doi.org/10.1111/psyg.12301
Reid LM, MacLullich AMJ (2006) Subjective memory complaints and cognitive impairment in older people. Dement Geriatr Cogn Disord 22(5–6):471–485. https://doi.org/10.1159/000096295
Youn JC, Kim KW, Lee DY, Jhoo JH, Lee SB, Park JH, Choi EA, Choe JY, Jeong JW, Choo IH, Woo JI (2009) Development of the subjective memory complaints questionnaire. Dement Geriatr Cogn Disord 27(4):310–317. https://doi.org/10.1159/000205512
Schmand B, Jonker C, Hooijer C, Lindeboom J (1996) Subjective memory complaints may announce dementia. Neurology 46(1):121–125. https://doi.org/10.1212/WNL.46.1.121
Portet F, Ousset PJ, Visser PJ, Frisoni GP, Nobili F, Scheltens P, Vellas B, Touchon J, MCI Working Group of the European Consortium on Alzheimer’s Disease (EADC) (2006) Mild cognitive impairment (MCI) in medical practice: a critical review of the concept and new diagnostic procedure. Report of the MCI Working Group of the European Consortium on Alzheimer’s disease. J Neurol Neurosurg Psychiatry 77:714–718. https://doi.org/10.1136/jnnp.2005.085332
Bosco A, Spano G, Caffò AO, Lopez A, Grattagliano I, Saracino G, Pinto K, Hoogeveen F, Lancioni GE (2017) Italians do it worse. Montreal cognitive assessment (MoCA) optimal cut-off scores for people with probable Alzheimer’s disease and with probable cognitive impairment. Aging Clin Exp Res 29(6):1113–1120. https://doi.org/10.1007/s40520-017-0727-6
Siciliano M, Chiorri C, Passaniti C, Sant’Elia V, Trojano L, Santangelo G (2019) Comparison of alternate and original forms of the Montreal cognitive assessment (MoCA): an Italian normative study. Neurol Sci 40(4):691–702. https://doi.org/10.1007/s10072-019-3700-7
Kawada T (2019) Montreal cognitive assessment (MoCA) and its memory tasks for detecting mild cognitive impairment. Neurol Sci 40(3):633–633. https://doi.org/10.1007/s10072-018-3616-7
Li X, Jia S, Zhou Z, Jin Y, Zhang X, Hou C, Zheng W, Rong P, Jiao J (2018) The role of the Montreal cognitive assessment (MoCA) and its memory tasks for detecting mild cognitive impairment. Neurol Sci 39(6):1029–1034. https://doi.org/10.1007/s10072-018-3319-0
Katz S (1983) Assessing self-maintenance: activities of daily living, mobility, and instrumental activities of daily living. J Am Geriatr Soc 31(12):721–727. https://doi.org/10.1111/j.1532-5415.1983.tb03391.x
Brink TL, Yesavage JA, Lum O, Heersema PH, Adey M, Rose TL (1982) Screening tests for geriatric depression. Clin Gerontol 1(1):37–43. https://doi.org/10.1300/J018v01n01_06
Lin Y, Shan P, Jiang W, Sheng C, Ma L (2019) Subjective cognitive decline: preclinical manifestation of Alzheimer’s disease. Neurol Sci 40(1):41–49. https://doi.org/10.1007/s10072-018-3620-y
Schmidt M (1996) Rey auditory verbal learning test: a handbook. Western Psychological Services, Los Angeles
Appollonio I, Leone M, Isella V, Piamarta F, Consoli T, Villa ML, Forapani E, Russo A, Nichelli P (2005) The frontal assessment battery (FAB): normative values in an Italian population sample. Neurol Sci 26(2):108–116. https://doi.org/10.1007/s10072-005-0443-4
Spinnler H (1987) Standardizzazione e taratura italiana di test neuropsicologici. Ital J Neurol Sci 6:21–120
Ruggiero G, Iachini T, Ruotolo F, Senese VP (2011) Spatial memory: the role of egocentric and allocentric frames of reference. In J.B. Thomas (Ed.), Spatial memory: visuospatial processes, cognitive performance and developmental effects (Chapter 2, pp. 51–75). New York: Nova Science Publishers
Klencklen G, Després O, Dufour A (2012) What do we know about aging and spatial cognition? Reviews and perspectives. Ageing Res Rev 11(1):123–135. https://doi.org/10.1016/j.arr.2011.10.001
Serino S, Cipresso P, Morganti F, Riva G (2014) The role of egocentric and allocentric abilities in Alzheimer’s disease: a systematic review. Ageing Res Rev 16(1):32–44. https://doi.org/10.1016/j.arr.2014.04.004
Winocur G, Moscovitch M, Sekeres M (2007) Memory consolidation or transformation: context manipulation and hippocampal representations of memory. Nat Neurosci 10(5):555–557. https://doi.org/10.1038/nn1880
Hirshhorn M, Newman L, Moscovitch M (2011) Detailed descriptions of routes traveled, but not map-like knowledge, correlates with tests of hippocampal function in older adults. Hippocampus 21(11):1147–1151. https://doi.org/10.3389/fnagi.2012.00025
Colombo D, Serino S, Tuena C, Pedroli E, Dakanalis A, Cipresso P, Riva G (2017) Egocentric and allocentric spatial reference frames in aging: a systematic review. Neurosci Biobehav Rev 80:605–621. https://doi.org/10.1016/j.neubiorev.2017.07.012
Parizkova M, Lerch O, Moffat SD, Andel R, Mazancova AF, Nedelska Z, Vyhnalek M, Hort J, Laczó J (2018) The effect of Alzheimer’s disease on spatial navigation strategies. Neurobiol Aging 64:107–115
Lopez A, Caffò AO, Bosco A (2019) Memory for familiar locations: the impact of age, education and cognitive efficiency on two neuropsychological allocentric tasks. Assessment. https://doi.org/10.1177/1073191119831780
Spreng RN, Lockrow AW, DuPre E, Setton R, Spreng KA, Turner GR (2018) Semanticized autobiographical memory and the default–executive coupling hypothesis of aging. Neuropsychologia 110:37–43. https://doi.org/10.1016/j.neuropsychologia.2017.06.009
Iachini T, Ruotolo F, Vinciguerra M, Ruggiero G (2017) Manipulating time and space: collision prediction in peripersonal and extrapersonal space. Cognition 166:107–117. https://doi.org/10.1016/j.cognition.2017.03.024
Ten Brink AF, Biesbroek JM, Oort Q, Visser-Meily JMA, Nijboer TCW (2019) Peripersonal and extrapersonal visuospatial neglect in different frames of reference: a brain lesion-symptom mapping study. Behav Brain Res 356:504–515. https://doi.org/10.1016/j.bbr.2018.06.010
Nori R, Piccardi L, Maialetti A, Goro M, Rossetti A, Argento O, Guariglia C (2018) No gender differences in egocentric and allocentric environmental transformation after compensating for male advantage by manipulating familiarity. Front Neurosci 12:204. https://doi.org/10.3389/fnins.2018.00204
Iachini T, Ruotolo F, Ruggiero G (2009) The effects of familiarity and gender on spatial representation. J Environ Psychol 29(2):227–234. https://doi.org/10.1016/j.jenvp.2008.07.001
Acknowledgments
The present study is part of the PhD program of the second author, under the supervision of the last author.
Funding
The first author was supported by the project “Epidemiology of Topographical Disorientation and Mild Cognitive Impairment in a South Italian elderly population”—Action Co-founded by Cohesion and Development Fund 2007–2013—APQ Research Puglia Region “Regional programme supporting smart specialization and social and environmental sustainability—FutureInResearch” (Grant Code CEY4SQ4).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Caffò, A.O., Lopez, A., Spano, G. et al. The differential effect of normal and pathological aging on egocentric and allocentric spatial memory in navigational and reaching space. Neurol Sci 41, 1741–1749 (2020). https://doi.org/10.1007/s10072-020-04261-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-020-04261-4