Abstract
Genetic brain channelopathies result from inherited or de novo mutations of genes encoding ion channel subunits within the central nervous system. Most neurological channelopathies arise in childhood with paroxysmal or episodic symptoms, likely because of a transient impairment of homeostatic mechanisms regulating membrane excitability, and the prototypical expression of this impairment is epilepsy. Migraine, episodic ataxia and alternating hemiplegia can also occur, as well as chronic phenotypes, such as spinocerebellar ataxias, intellectual disability and autism spectrum disorder. Voltage-gated and ligand-gated channels may be involved. In most cases, a single gene may be associated with a phenotypical spectrum that shows variable expressivity. Different clinical features may arise at different ages and the adult phenotype may be remarkably modified from the syndrome onset in childhood or adolescence. Recognizing the prominent phenotypical traits of brain channelopathies is essential to perform appropriate diagnostic investigations and to provide the better care not only in the paediatric setting but also for adult patients and their caregivers. Herein, we provide an overview of genetic brain channelopathies associated with epilepsy, highlight the different molecular mechanisms and describe the different clinical characteristics which may prompt the clinician to suspect specific syndromes and to possibly establish tailored treatments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The term ‘channelopathy’ encompasses a wide spectrum of disorders underpinned by genetic or acquired dysfunction of ion channels [1].
Ion channels are proteins located across the lipid bilayer of cell membranes, wherein they form a pore to facilitate the inflow or outflow of ions according to their electrochemical gradient.
Each channel exhibits a selective permeability to specific ions, whose trafficking is gated by the channel state: open, inactivated closed and resting closed. The gating may be controlled by transmembrane electric potential (voltage-gated channels), specific extracellular ligand molecules (ligand-gated channels), or else different mechanisms such as intracellular second messengers, light, temperature and mechanical stimuli. Ion pumps similarly modulate ion trafficking, albeit with a different mechanism, i.e. ions are moved against gradient through active transport to maintain the resting potential. Most ion channels exhibit a heteromeric structure with a pore-forming subunit and accessory regulatory subunits. Ion pumps are similar heteromeric proteins with a catalytic main subunit as well as auxiliary and regulatory subunits.
Genetic channelopathies have been described in diverse systemic disorders, consistent with the widespread expression of ion channels and ion pumps and with their role in regulating the resting potential, signal transduction, size, growth, motility and apoptosis of all eukaryotic cells. There exists a tissue selectivity of ion channels; hence, most genetic channelopathies are confined to the system wherein the related ion channel is mainly expressed. Indeed, most of ion channel genes are enriched in the brain, wherein their dysfunction is especially detrimental to neurons, by affecting the generation, suppression and spread of the action potential [2, 3]. The transient and recurrent impairment of membrane excitability may lead to paroxysmal symptoms, especially seizures, but their occurrence may vary over time due to the different expression of ion channels at different ages [4]. Indeed, the history of epilepsy genetics has long been dominated by inherited channelopathies. The first gene found in familial epilepsy was CHRNA4, encoding for a subunit of the ligand-gated nicotinic acetylcholine receptor and causing autosomal dominant frontal lobe epilepsy [5]. Thereafter, a growing number of mutations affecting ion channel genes have been identified in neurological disorders, including familial and sporadic epilepsies.
Specific syndromes associated with single ion channel genes are well described; however, the large-scale application of next generation sequencing (NGS) has revealed the genotype-phenotype correlation may be extremely challenging. Ion channel genes initially associated only with benign phenotypes (e.g. KCNQ2, SCN2A) have also been demonstrated to be implicated in severe epileptic encephalopathies.
To be able to diagnose a specific genetic aetiology in epilepsy carries substantial benefit in both children and adults. Families are often relieved to find a cause usually after a distressing diagnostic odyssey. Unnecessary invasive testing can be avoided and appropriate familial counselling can be established [6]. Furthermore, the comprehension of the underlying molecular biology raises the possibility of delivering precision medicine by using drugs that reverse or counteract the specific ion channel dysfunction [7].
Herein, we overview the molecular pathogenesis and the phenotype of genetic brain encephalopathies associated with epilepsy, highlighting the different clinical characteristics which may prompt the clinician to suspect specific syndromes and discussing the specific clinical challenges at different ages of presentation.
Methods
We performed a PubMed/Medline search for articles up to and including May 2019 using Boolean logic with the following terms: ‘brain AND channel*’, ‘seizure AND channel*’, ‘epilepsy AND channel*’, ‘encephalopathy AND channel*’ with no language restrictions. We integrated the items retrieved by the automated query with selected references manually searched on PubMed, Embase, the Human Gene Mutation Database (HGMD, http://www.hgmd.cf.ac.uk/ac/index.php), Google (https://www.google.com) and Google Scholar (https://scholar.google.com) search engines.
We then analysed the available data for the two main types of ion channels.
Voltage-gated channels
Sodium channels
Voltage-gated sodium channels (‘VGSCs’ or ‘Nav channels’) are heteromeric complexes consisting of a large central pore-forming alpha subunit and two smaller auxiliary beta subunits. A superfamily of genes clustering on chromosome 2 encodes for different isoforms of the alpha subunit (Nav1.1-Nav1.9). All alpha subunits comprise four homologous domains (I–IV), each containing six transmembrane segments (S1–S6). The S5–S6 segments of each domain form the pore, whilst the S4 segment acts as a voltage sensor. The depolarization of the cell membrane leads to a conformational change of the channel, resulting in a sodium inflow which triggers action potentials in nerve, muscle and other excitable cells [8].
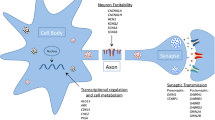
Mutations in three alpha subunit genes, all highly expressed in the central nervous system (SCN1A, SCN2A, SCN8A), have been shown to cause epilepsy in humans [9] (Fig. 1).
SCN1A encodes for the alpha 1 subunit (Nav1.1), whose expression sharply increases from 4 to 5 months of age [10]. Mutations of this gene yield distinct phenotypes sharing a peculiar fever sensitivity. Pathogenic variants of SCN1A were initially found in genetic epilepsy with febrile seizures plus (GEFS+), an autosomal dominant disorder characterized by variable intra-familial phenotypical expression. Affected family members may exhibit febrile seizures persisting beyond 6 years of age as well as focal or generalized seizures, but with normal cognitive functioning [9] (Figs. 1 and 2). De novo heterozygous SCN1A mutations were later identified in a more severe phenotype, Dravet syndrome [11]. Indeed SCN1A is the most frequently mutated gene in epileptic encephalopathies; de novo mutations have been identified in 2.8% of trios investigated by exome sequencing in the Epi4k study [12] and SCN1A was the only gene that reached exome-wide significance in the Epi25 study of ultra-rare variants in epilepsy (odds ratio = 18.4, comparing individuals with developmental and epileptic encephalopathies to healthy controls) [13].
The mutations identified in GEFS+ are missense with either gain or loss-of-function effect. Dravet syndrome-related mutations may be truncating mutations, splice-site mutations, frameshift intragenic deletions or gene duplications leading to haploinsufficiency or they may be missense mutations affecting the pore-forming region of the protein [14]. The underlying pathogenesis has been hypothesized to result from a reduction of the sodium current density in inhibitory interneurons rather than in pyramidal cells, leading to a net hyperexcitatory effect [15].
Dravet syndrome typically develops with the sudden onset of febrile seizures at 5–8 months of age in previously healthy children. Unlike simple febrile convulsions, these febrile seizures are often unilateral and very prolonged, sometimes leading to status epilepticus [16]. In about a third of cases, such an abrupt onset occurs after a vaccination [17] and indeed most cases formerly interpreted as alleged ‘vaccine encephalopathy’ were patients with previously undiagnosed Dravet syndrome [18]. During the first year of life, both febrile and afebrile seizures occur. From the second year of age, an overt encephalopathy is established. Developmental delay is accompanied by polymorphic seizure types including generalized tonic-clonic, alternating unilateral clonic, focal with impaired awareness, brief myoclonic, tonic (rare) or myoclonic non-convulsive status epilepticus described as ‘obtundation status’ [16, 19, 20]. Increased body temperature remains the most important seizure precipitant, whether caused by fever, warm baths, ambient warmth or physical exercise [21]. Seizures are also typically precipitated by the use of anti-epileptic drugs that are sodium channel blockers (e.g. carbamazepine, phenytoin, lamotrigine). The EEG shows generalized spike-wave and polyspike-wave activity with multifocal discharges as well as a progressive slowing of the background activity. Photosensitivity is common, may be very early (median age at onset, 1.25 years) and transient [22]. Some older patients may auto-induce seizures by voluntary eye closure [16, 21]. Children with Dravet syndrome typically start to walk and talk at a normal age but soon develop a wide-based ataxic gait and an impairment of language production skills, in spite of relative preservation of comprehension [16]. They may also have behavioural disturbances, impaired attention and autistic traits [23]. After 5 years of age, epilepsy tends to improve with stabilization of intellectual functioning and behaviour [16, 19, 24]. There is a high rate of early mortality, especially due to accidents, drowning, severe status epilepticus, infections and sudden unexplained death in epilepsy (SUDEP) [16].
In adulthood, seizures become less aggressive and temperature-sensitive (Table 1). Myoclonic and absence seizures disappear and convulsive seizures occur mainly in sleep along with tonic seizures. The EEG may show multifocal interictal discharges (Online Resource 1). Cerebellar features become more prominent, especially ataxia, dysarthria and intention tremor [25, 26]. Adults with Dravet syndrome can also develop characteristic levodopa-responsive extrapyramidal signs, such as axial dystonia and anterocollis with a crouching gait [27]. Gait disturbance may also be exacerbated by the onset of an axonal motor neuropathy [28].
Familial hemiplegic migraine type 3 (FHM3) is an alternative SCN1A allelic disorder associated with missense mutations; patients with FHM3 may rarely exhibit concomitant seizures [29, 30] (Figs. 1 and 2).
The SCN2A gene encodes for the NaV1.2 channel, which is highly expressed in the initial axonal segments of excitatory hippocampal and cortical neurons from the birth [31]. Mutations of SCN2A were initially found in families with ‘Benign familial neonatal-infantile seizures’ (BFNIS), characterized by self-limiting epilepsy arising in early infancy and a favourable cognitive outcome [32, 33]. Thereafter, de novo SCN2A mutations have been increasingly recognized as a major cause of epileptic encephalopathies [32, 34] and linked to non-syndromic intellectual disability [35, 36] and autism spectrum disorders [36, 37]. De novo mutations of SCN2A have been identified in 0.8% of probands with epileptic encephalopathies in the Epi4k study [12].
Most patients with epilepsy and SCN2A mutations exhibit seizures from the first week of life, possibly due to the timing of NaV1.2 channel expression, which peaks during early development and is then gradually replaced by NaV1.6 (SCN8A) during the first months of life [31]. Less frequently, epilepsy arises during childhood, accompanied by cognitive regression and autistic features [32, 33] (Fig. 1). Recent findings suggest children with neonatal-onset seizures mainly have gain-of-function missense mutations and a good response to sodium channel blocking anti-epileptic drugs, whilst those with later onset epilepsy mostly have loss-of-function missense or truncating variants and do not respond to sodium channel blockers [33]. In patients with autism or intellectual disability, most identified mutations are truncating [37]. Although the NaV1.2 channel appears to be mainly involved in childhood disorders, an SCN2A mutation with incomplete penetrance has also been recently identified in two siblings with juvenile onset myoclonic seizures and ataxia [38]. Follow-up data on the SCN2A encephalopathy suggest patients may develop episodic ataxia from infancy/puberty [39] or suffer persisting seizures, intellectual disability, autistic traits and stereotypies in adulthood [32] (Table 1, Figs. 1 and 2).
Heterozygous missense mutations of SCN8A have recently been associated with a spectrum of epilepsies, ranging from autosomal dominant familial benign infantile seizures (inherited mutations) to epileptic encephalopathies (de novo mutations in 0.8% of the Epi4k cohort) [12, 40, 41] (Fig. 1). The Epi25 study has recently disclosed a remarkable enrichment of ultra-rare variants in people with epilepsy compared with healthy subjects (odds ratio = 13.8), pinpointing SCN8A as a top ranking gene in epilepsy [13].
The SCN8A gene product (sodium channel subunit Nav1.6) is increasingly expressed from the neonatal period, progressively replacing the NaV1.2 channel in initial axonal segments. Accordingly, most patients with SCN8A mutations develop epilepsy in infancy (median age = 4 months) [40]. Patients with familial benign infantile seizures exhibit self-limited focal and generalized seizures up to 2 years of age; thereafter, epilepsy remits and neurodevelopment is normal in almost all cases [42]. Some of these patients may later develop episodes of paroxysmal dyskinesia from puberty [43] Conversely, those with SCN8A-related encephalopathy suffer global developmental delay and multiple intractable seizure types, which include focal, tonic with autonomic signs, clonic, myoclonic, absence and epileptic spasms [40, 44, 45]. Sleep disorders and frequent startles related to an hyperalert state have been reported [46]. An intermediate phenotype with treatable epilepsy and mild cognitive impairment has also been described [47]. Genotype-phenotype correlation is poor as patients harbouring the same mutation may have different clinical presentations [40]. Evidence from animal models of SCN8A-related encephalopathy suggests a high risk of fatal cardiac arrhythmias [48]. A movement disorder characterized by dystonic/dyskinetic attacks may develop both in patients with benign familial infantile seizures and in those with an epileptic encephalopathy, independently from the seizure course [40, 42]. There are also isolated case reports of adults with intellectual disability, ataxia and extrapyramidal signs [47] (Table 1). There is clinical evidence of a possible targeted efficacy of sodium channel blockers in SCN8A-related epilepsy, especially if the mutations are known to cause gain of function [49].
Potassium channels
Potassium channels are a large family of ubiquitous transmembrane proteins, each composed of four α subunits, which interlock to constitute the pore-forming region, and different accessory subunits. According to their structure and kinetics, potassium channels can be categorized as voltage-gated (Kv) channels, calcium-activated potassium channels, inwardly rectifying (Kir) channels and tandem pore domain (K2P) channels [50].
Most human potassium channelopathies involve the voltage-gated family (Kv). These channels feature six transmembrane domains per alpha subunit with a voltage sensor on the fourth transmembrane segment (S4). The channels open in response to neuronal depolarization, modulating the frequency and duration of action potential [50].
Mutations affecting the Kv channels encoded by KCNQ2 [51] and KCNQ3 [52] were initially identified in patients with autosomal dominant benign familial neonatal seizures, characterized by normal neurodevelopment and time-limited seizures starting at 2–8 days of life and remitting spontaneously within 12 months. Akin to many other channelopathies, the phenotypic spectrum has recently expanded to include severe phenotypes, namely families with drug-resistant seizures and intellectual disability harbouring KCNQ2 [53] and KCNQ3 [54] mutations as well as sporadic cases with a specific KCNQ2-epileptic encephalopathy [55] (Fig. 1).
The latter exhibit a characteristic epilepsy course, with drug-resistant tonic asymmetric, focal and clonic seizures in the first week of life, progressively disappearing in early infancy and remitting by the age of 1–3 years. There is a dynamic electroencephalographic pattern, with neonatal burst-suppression evolving to multifocal epileptiform activity during follow-up. In adulthood, patients suffer intellectual disability of variable severity, hypotonia and spastic tetraparesis.
[55] (Table 1). The pathophysiology of the divergence between the favourable seizure prognosis and the dismal neurodevelopmental outcome is possibly due to the different role of KCNQ2 channels in epileptogenesis and cognitive functioning. In neonates, potassium channels are the main source of inhibition; mutations of KCNQ2 expose the newborn brain to a powerful excitatory drive, which is then mitigated by the growing role of GABAergic inhibitory transmission in early childhood [56]. The neurodevelopmental impairment may instead be related to the critical role of potassium channels in synaptic efficacy and learning-related gene expression in the cerebral cortex and hippocampus [57].
The severe encephalopathic phenotype is likely attributable to a dominant negative effect of KCNQ2 mutations. The vast majority of mutations are missense and appear to cluster in four putative functional hotspots which would affect the conformation of the tetrameric subunits of the channel [58, 59]. Of note, to identify KCNQ2 variants in severe epilepsies may assist the clinician in the therapeutic choice. The potassium channel opener retigabine may partially reverse the KCNQ2 loss of function and reduce seizure frequency [58], yet its production has been discontinued in 2017 due to safety issues. Sodium channel blockers appear effective in the active phase of epilepsy, possibly because sodium channels and KCNQ channels colocalize and interact at critical locations of the neuronal membrane [55, 59].
KCNQ2 mutations found in benign familial neonatal seizures are heterogeneous, including deletions in 20q13 affecting the whole gene and contiguous segments, and are randomly distributed throughout the channel structure [58].
Calcium-activated potassium channels are also implicated in epilepsy. These channels are located in axons and at presynaptic terminals of excitatory neurons. They are activated in response to the calcium influx during an action potential, thus hyperpolarizing the cell and limiting neuronal excitability. However, the functional effect of mutations affecting these channels is unclear. Gain-of-function mutations of KCNMA1 (KCa1.1 channel) have been described in children with paroxysmal nonkinesigenic dyskinesia and early onset absences [60].
Mutations affecting inwardly rectifying potassium (Kir) channels have been demonstrated in patients with epilepsy. In particular, KCNB1, encoding for the main delayed rectifier channel of the hippocampus and cortex (Kv2.1), is an emerging gene in neurodevelopmental disorders. Mutations in this gene may produce a phenotypical spectrum including non-syndromic intellectual disability and an epileptic encephalopathy characterized by epileptic spasms, severe developmental delay, autism spectrum disorder and chronic drug-resistant epilepsy [61] .
De novo KCNJ11 pathogenic variants affecting the inwardly rectifying Kir6.2 channel have also been associated with epilepsy and intellectual disability in patients with neonatal diabetes mellitus [62].
Specific severe epileptic phenotypes can result from mutations of KCNT1, which encodes a sodium-activated potassium channel subunit involved in the slow hyperpolarization that follows repetitive neuronal firing. Inherited missense mutations were initially reported in a severe form of autosomal dominant nocturnal frontal lobe epilepsy (ADNFLE, see below, also known as autosomal dominant sleep-related hypermotor epilepsy) with intellectual disability and behavioural disturbances. De novo gain-of-function mutations have subsequently been found in about 40% of patients with malignant migrating partial seizures of infancy (MMPSI); in this syndrome, patients suffer nearly continuous multifocal seizures with migrating ictal EEG discharges in the first 6 months of life and then develop persistent drug-resistant seizures with acquired microcephaly, intellectual disability, axial hypotonia, pyramidal signs, extrapyramidal signs with athetoid movements and strabismus [63, 64] (Table 1, Figs. 1 and 2).
Further studies have demonstrated that the penetrance of KCNT1 mutations is not complete and that different affected family members harbouring the same inherited variant may develop either autosomal dominant frontal lobe epilepsy or MMPSI. Clinical evidences suggest KCNT1 may be considered an actionable gene, as about 30% of patients with both KCNT1-related phenotypes benefit from quinidine, an anti-arrhythmogenic potassium channel blocker, in terms of seizure control [63, 65]. Isolated cases of SUDEP and cardiac arrhythmias in patients with KCNT1 mutations have been reported [66]. Even though the neurological phenotype in adults has not been described, a pathogenic KCNT1 mutation has been reported in a 38-year-old patient with life-threatening Brugada syndrome by a cardiology team [67]; interestingly, this patient was diagnosed after sudden loss of consciousness interpreted as possible syncope or isolated seizure, raising the possibility of a mild epilepsy phenotype in adult age (Table 1).
None of these genes has been encountered in the Epi4k [12] and Epi25 [13] studies, mutations of potassium channels are likely less common than those of sodium channels.
Calcium channels
Voltage-gated calcium channels are composed of an α1 pore-forming subunit and several different subunits and they have a critical role in neuronal regulation of intracellular calcium concentration. Channel opening may result from either large membrane depolarization (high voltage-activated channels: HVA) or from small voltage changes (low voltage-activated channels: LVA), allowing the inflow of calcium ions according to their electrochemical gradient. Intracellular calcium modulates gene transcription, neurotransmitter release, neurite outgrowth and enzyme activity [68].
The HVA Cav2.1 (P/Q-type) channel, encoded by CACNA1A, is involved in neurological phenotypes. Heterozygous mutations of CACNA1A have been found in patients with epilepsy, ataxia or migraine (Table 1). Loss-of-function variants have been associated with two overlapping phenotypes, namely childhood absence epilepsy and episodic ataxia type 2. Patients with CACNA1A-related absence seizures may develop ataxic symptoms [69] and those with episodic ataxia have cerebellum-related paroxysmal symptoms (i.e. unsteadiness, vertigo, nystagmus) and may in turn develop tonic-clonic and absence seizures in 7% of cases [70]. Conversely, gain-of-function mutations may lead to familial hemiplegic migraine type 1, characterized by sudden onset of hemimotor and hemisensory symptoms lasting up to 72 h which can accompany, outlast or occur independently from a typical migrainous attack. These patients often have cerebellar symptoms and may develop unprovoked seizures in childhood and migraine-provoked seizures in adulthood [71] (Figs. 1 and 2). Late-onset parkinsonism has also been reported [72].
Biallelic CACNA1A mutations have been disclosed in patients with early onset seizures, intellectual disability, autistic features and progressive cerebral and optic nerve atrophy [73]. Moreover, CACNA1A de novo missense mutations have been identified in 1% of cases from a large cohort of epileptic encephalopathies [74].
Cerebellar dysfunction is a frequent phenotypical trait in patients harbouring CACNA1A mutations. The Cav2.1 channel is indeed especially important in cerebellar networks. In the mice model, CACNA1A deletions impair both the excitatory drive of the granule cells on Purkinje cells and the neurotransmitter release by the Purkinje cells themselves, resulting in seizures and ataxia [75, 76]. In turn, the cerebellar dysfunction may influence cognitive functions. Disruption of the cerebellar projections to the prefrontal, frontal and limbic cortex may impair complex cognitive processes, interfering both with the default mode network and with motor memory learning and consolidation [77,78,79].
Not every CACNA1A mutation causes seizures: Patients with spinocerebellar ataxia type 6 (SCA6), caused by expansion of the CAG trinucleotide repeat sequence in this gene, do not develop epilepsy but rather a late-onset progressive ataxia, sometimes accompanied by pyramidal and extrapyramidal signs as well as peripheral neuropathy and autonomic disturbance [80].
Genes encoding for LVA channels, especially CACNA1H, are currently regarded as susceptibility genetic loci for human idiopathic generalized epilepsies in children and adolescents; this is not surprising considering that the murine model of absence epilepsy (Generalized Absence Epilepsy Rat of Strasbourg) exhibits CACNA1H variants [81].
HCN channels
Hyperpolarization-activated, cyclic nucleotide-gated (HCN) channels are a class of voltage-gated ion channels permeable to potassium and, to a lesser extent, sodium. Their kinetics are unique, as the channel is activated by cell hyperpolarization, remain open at negative voltages and their opening is potentiated by binding of cAMP. The brain isoform (HCN1) yields an inward depolarizing current named Ih (‘hyperpolarization-activated’ current) which drives the neuronal membrane potential back toward the threshold for calcium and sodium channel activation, hence modulating the neuronal excitability [82, 83].
The phenotypical spectrum of HCN1 mutations spans from early onset encephalopathies to benign idiopathic generalized epilepsies [84,85,86,87]. Patients with sporadic mutations develop a neonatal/infantile onset epileptic encephalopathy in about a third of cases; in these subjects, epilepsy arises as prolonged febrile or afebrile seizures at a median age of 7 months and then progresses with drug-resistant tonic, tonic-clonic and seizures and intellectual disability. On the contrary, 42% of sporadic cases and the vast majority of familial cases exhibit milder phenotypes, such as transient febrile seizures, GEFS+, drug-responsive idiopathic generalized epilepsies with normal or borderline cognitive functioning [85] (Figs. 1 and 2).
Encephalopathic phenotypes are associated with pathogenic missense variants clustering within or close to transmembrane domains, milder phenotypes segregate with missense variants outside transmembrane domains, in the intracellular N- and C-terminal parts of the channel [85].
Ion pumps
Mutations affecting the function of ionic pumps can dysregulate the membrane potential and yield neuronal hyperexcitability. Neurological paroxysmal disorders can especially result from pathogenic variants of ATP1A2, which encodes the α2 subunit of the A1A2 glial sodium–potassium ATPase pump. Indeed, ATP1A2 is the gene responsible of familial hemiplegic migraine type 2 (FHM2); up to 60% of patients with FHM2 may experience seizures, that usually start during childhood as febrile or afebrile seizures, sometimes preceding the first hemiplegic migraine attack, and have a benign evolution (i.e. benign familial infantile convulsions) [88, 89] (Figs. 1 and 2).
Ligand-gated channels
Nicotinic acetylcholine receptors
The first monogenic pathogenic variant to be identified in the history of epilepsy genetics affected a ligand-gated channel, namely the alpha-4 subunit of the nicotinic acetylcholine receptor encoded by CHRNA4 in patients with autosomal dominant nocturnal frontal lobe epilepsy (ADNFLE) [5] (Fig. 1). Beforehand, the ADNFLE syndrome had already been extensively described, yet the underlying pathophysiology was unknown. The disclosure of CHRNA4 mutations in these patients highlighted the importance of ion channels in human epilepsy and paved the way for the concept of brain channelopathies [90]. Nowadays, we know ADNFLE may result from mutations affecting both the alpha (CHRNA2, CHRNA4) and the beta (CHRNB2) subunits of the receptor with a penetrance of about 70–80%. Most mutations are located in the pore-forming domain and act through a gain of function mechanism, although loss-of-function variants have also been reported [91, 92]. Seizures arise from about 10 years of age and typically occur in clusters during slow-wave sleep [91, 92]. They are stereotyped, frequent and brief. Seizure semiology comprises hyperkinetic or tonic seizures, paroxysmal arousals, dystonia-like attacks and epileptic nocturnal wanderings. Similar seizure phenotypes may be associated with mutations of unrelated genes such as KCNT1 (see above), CRH and DEPDC5 (reviewed in [93]). In childhood and adolescence, the differential diagnosis includes parasomnias, especially because seizures in ADNFLE may be accompanied by normal EEG recordings. However, parasomnias usually tend to spontaneously disappear in adult age; on the contrary, seizures in ADNFLE continue in adulthood if they are not recognized and they are drug-resistant in a third of cases [93] (Table 1). Many reports document sodium channel blockers as drugs of choice in ADNFLE, yet topiramate, acetazolamide, nicotine transdermal patches and fenofibrate have also been efficacious in small case series (reviewed in [93, 94]).
GABA receptors
Mutations affecting GABA receptors have also been linked to epilepsy, not surprisingly, considering GABA is the main inhibitory neurotransmitter of the central nervous system. Most of the reported mutations affect genes for the GABA-A receptor, which is composed of five subunits. Missense mutations affecting either the alpha (GABRA1, GABRA6), beta (GABARB3), or gamma (GABARG2) subunit have been found in sporadic and familial idiopathic generalized epilepsies, especially in childhood absence epilepsy and juvenile myoclonic epilepsy [95]. However, GABRA1A is also a major causative gene for heterogeneous epileptic encephalopathies, including Ohtahara syndrome, infantile spasms, Dravet syndrome-like phenotypes and myoclonic astatic epilepsy [95] (Fig. 1). Red flags for suspecting GABRA1A mutations are infantile-onset epilepsy (from 1 day to 15 months of age), prominent tonic-clonic and myoclonic seizures, generalized spike-and-wave activity and a photoparoxysmal response. Such features can occur both in patients with idiopathic epilepsies and in those with epileptic encephalopathy [95] (Table 1, Fig. 2). No clear genotype-phenotype correlation has emerged, although the phenotype appears to be similar within single families [95]. Interestingly, GABA-A receptor has also been demonstrated to be involved in Angelman syndrome, a complex neurodevelopmental disorder characterized by ataxia, cortical tremor, sociable behaviour, seizures and intellectual disability. Angelman syndrome is mostly caused by deletion of the maternally derived chromosome 15q11-q13 region, which includes not only the causative UBE3A gene, but also the beta(3)-alpha(5)-gamma(3) GABA(A) receptor subunit gene cluster; post-mortem cortical tissues exhibit an abnormal shift in GABA-A receptor subunits compared with healthy controls [96].
NMDA receptors
The ligand-gated NMDA receptor is another ion channel involved in neurodevelopmental disorders. The channel is composed of two glycine-binding NR1 subunits and two glutamate-binding NR2 subunits. It mediates excitatory neurotransmission between neurons allowing inflow of cations [97]. Mutations affecting the NR2 subunits (i.e. GRIN2A, GRIN2B) appear to be especially detrimental for neurodevelopment. The phenotypical spectrum of GRIN2A mutations includes patients with isolated intellectual disability, idiopathic focal epilepsy or epileptic encephalopathies. Interestingly, language disorders are common and range from mild speech impairment with no seizures to the epilepsy aphasia spectrum (atypical rolandic epilepsy, continuous spikes and waves during slow-wave sleep and Landau-Kleffner-Syndrome) [98]. In a child with GRIN2A encephalopathy, the targeted approach with the NMDA receptor antagonist memantine has successfully reduced seizure frequency [99], yet this observation in a single case remains to be confirmed in additional cases. Gain-of-function GRIN2B mutations cause West syndrome, as well as childhood onset focal epilepsy in association with intellectual disability. Cognitive skills depend upon the degree of channel function impairment [100]. In adults, GRIN2A mutations have been anecdotally reported in patients with schizophrenia and GRIN2B mutations in Alzheimer disease [101], yet such findings need to be confirmed in larger case series..
Conclusion
The clinical spectrum of genetic brain channelopathies ranges from life-threatening infantile-onset epileptic encephalopathies to mild adult-onset epilepsies. Epilepsy is the archetypical neurological phenotype with an extremely heterogeneous and variable presentation throughout life. In most cases, seizures are only part of complex encephalopathic phenotypes with widespread neurological signs often with a progressive course, attributable to the dysfunction of ion channels over the whole central nervous system, including the cerebral cortex and the cerebellum.
Technological advances currently allow extensive screening in single patients with suspected genetic epilepsy using gene panel analyses or exome sequencing. Despite this, the clinician’s assessment remains of utmost importance. Recognizing the prominent phenotypical traits of brain channelopathies is essential to best employ molecular analyses. To identify single pathogenic variants may in turn guide the clinician in designing a tailored treatment. Understanding the spectrum of brain channelopathies may also prompt the adult neurologist to perform molecular investigations in patients who could not benefit from modern diagnostic technologies in their childhood.
Still the genetic diagnosis should be followed by the recognition of the potentially changing neurological phenotype at different ages, to provide better care not only in the paediatric setting but also for adult patients and their caregivers, especially in the delicate transition of care of people with epilepsy.
References
Kim J-B (2014) Channelopathies. Korean J Pediatr 57:1–18. https://doi.org/10.3345/kjp.2014.57.1.1
Kew JNC, Davies CH (2010) Ion channels: from structure to function, 2nd edn. Oxford University Press, Oxford New York
Seitter H, Koschak A (2018) Relevance of tissue specific subunit expression in channelopathies. Neuropharmacology 132:58–70. https://doi.org/10.1016/j.neuropharm.2017.06.029
Ryan DP, Ptáček LJ (2010) Episodic neurological channelopathies. Neuron 68:282–292. https://doi.org/10.1016/j.neuron.2010.10.008
Steinlein OK, Mulley JC, Propping P, Wallace RH, Phillips HA, Sutherland GR, Scheffer IE, Berkovic SF (1995) A missense mutation in the neuronal nicotinic acetylcholine receptor α4 subunit is associated with autosomal dominant nocturnal frontal lobe epilepsy. Nat Genet 11:201–203. https://doi.org/10.1038/ng1095-201
Berkovic SF (2015) Genetics of epilepsy in clinical practice. Epilepsy Curr 15:192–196. https://doi.org/10.5698/1535-7511-15.4.192
Balestrini S, Sisodiya SM (2018) Pharmacogenomics in epilepsy. Neurosci Lett 667:27–39. https://doi.org/10.1016/j.neulet.2017.01.014
Catterall WA (2014) Structure and function of voltage-gated sodium channels at atomic resolution: voltage-gated sodium channels at atomic resolution. Exp Physiol 99:35–51. https://doi.org/10.1113/expphysiol.2013.071969
Escayg A, Goldin AL (2010) Sodium channel SCN1A and epilepsy: mutations and mechanisms: sodium channel SCN1A and epilepsy. Epilepsia 51:1650–1658. https://doi.org/10.1111/j.1528-1167.2010.02640.x
Cheah CS, Westenbroek RE, Roden WH, Kalume F, Oakley JC, Jansen LA, Catterall WA (2013) Correlations in timing of sodium channel expression, epilepsy, and sudden death in Dravet syndrome. Channels 7:468–472. https://doi.org/10.4161/chan.26023
Claes L, Del-Favero J, Ceulemans B et al (2001) De novo mutations in the sodium-channel gene SCN1A cause severe myoclonic epilepsy of infancy. Am J Hum Genet 68:1327–1332. https://doi.org/10.1086/320609
Epi4K Consortium, Epilepsy Phenome/Genome Project (2013) De novo mutations in epileptic encephalopathies. Nature 501:217–221. https://doi.org/10.1038/nature12439
Feng Y-CA, Howrigan DP, Abbott LE et al (2019) Ultra-rare genetic variation in the epilepsies: a whole-exome sequencing study of 17,606 individuals. Am J Hum Genet 105:267–282. https://doi.org/10.1016/j.ajhg.2019.05.020
Marini C, Scheffer IE, Nabbout R, Suls A, de Jonghe P, Zara F, Guerrini R (2011) The genetics of Dravet syndrome: genetics of Dravet syndrome. Epilepsia 52:24–29. https://doi.org/10.1111/j.1528-1167.2011.02997.x
Yu FH, Mantegazza M, Westenbroek RE, Robbins CA, Kalume F, Burton KA, Spain WJ, McKnight G, Scheuer T, Catterall WA (2006) Reduced sodium current in GABAergic interneurons in a mouse model of severe myoclonic epilepsy in infancy. Nat Neurosci 9:1142–1149. https://doi.org/10.1038/nn1754
Dravet C (2011) The core Dravet syndrome phenotype: core Dravet syndrome. Epilepsia 52:3–9. https://doi.org/10.1111/j.1528-1167.2011.02994.x
McIntosh AM, McMahon J, Dibbens LM, Iona X, Mulley JC, Scheffer IE, Berkovic SF (2010) Effects of vaccination on onset and outcome of Dravet syndrome: a retrospective study. Lancet Neurol 9:592–598. https://doi.org/10.1016/S1474-4422(10)70107-1
Reyes IS, Hsieh DT, Laux LC, Wilfong AA (2011) Alleged cases of vaccine encephalopathy rediagnosed years later as Dravet syndrome. Pediatrics 128:e699–e702. https://doi.org/10.1542/peds.2010-0887
Scheffer IE (2012) Diagnosis and long-term course of Dravet syndrome. Eur J Paediatr Neurol EJPN Off J Eur Paediatr Neurol Soc 16(Suppl 1):S5–S8. https://doi.org/10.1016/j.ejpn.2012.04.007
Losito E, Kuchenbuch M, Chemaly N et al (2017) Age-related “sleep/nocturnal” tonic and tonic clonic seizure clusters are underdiagnosed in patients with Dravet syndrome. Epilepsy Behav EB 74:33–40. https://doi.org/10.1016/j.yebeh.2017.05.037
Verbeek NE, Wassenaar M, van Campen JS et al (2015) Seizure precipitants in Dravet syndrome: what events and activities are specifically provocative compared with other epilepsies? Epilepsy Behav 47:39–44. https://doi.org/10.1016/j.yebeh.2015.05.008
Verbeek N, Kasteleijn-Nolst Trenité D, Wassenaar M et al (2017) Photosensitivity in Dravet syndrome is under-recognized and related to prognosis. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 128:323–330. https://doi.org/10.1016/j.clinph.2016.11.021
Sinoo C, de Lange IM-L, Westers P et al (2019) Behavior problems and health-related quality of life in Dravet syndrome. Epilepsy Behav EB 90:217–227. https://doi.org/10.1016/j.yebeh.2018.11.029
Guerrini R (2012) Dravet syndrome: the main issues. Eur J Paediatr Neurol EJPN Off J Eur Paediatr Neurol Soc 16(Suppl 1):S1–S4. https://doi.org/10.1016/j.ejpn.2012.04.006
Genton P, Velizarova R, Dravet C (2011) Dravet syndrome: the long-term outcome. Epilepsia 52(Suppl 2):44–49. https://doi.org/10.1111/j.1528-1167.2011.03001.x
Akiyama M, Kobayashi K, Yoshinaga H, Ohtsuka Y (2010) A long-term follow-up study of Dravet syndrome up to adulthood. Epilepsia 51:1043–1052. https://doi.org/10.1111/j.1528-1167.2009.02466.x
Fasano A, Borlot F, Lang AE, Andrade DM (2014) Antecollis and levodopa-responsive parkinsonism are late features of Dravet syndrome. Neurology 82:2250–2251. https://doi.org/10.1212/WNL.0000000000000521
Gitiaux C, Chemaly N, Quijano-Roy S, Barnerias C, Desguerre I, Hully M, Chiron C, Dulac O, Nabbout R (2016) Motor neuropathy contributes to crouching in patients with Dravet syndrome. Neurology 87:277–281. https://doi.org/10.1212/WNL.0000000000002859
Russell MB, Ducros A (2011) Sporadic and familial hemiplegic migraine: pathophysiological mechanisms, clinical characteristics, diagnosis, and management. Lancet Neurol 10:457–470. https://doi.org/10.1016/S1474-4422(11)70048-5
Castro M-J, Stam A, Lemos C, de Vries B, Vanmolkot KR, Barros J, Terwindt GM, Frants RR, Sequeiros J, Ferrari MD, Pereira-Monteiro JM, van den Maagdenberg A (2009) First mutation in the voltage-gated Na v 1.1 subunit gene SCN1A with co-occurring familial hemiplegic migraine and epilepsy. Cephalalgia 29:308–313. https://doi.org/10.1111/j.1468-2982.2008.01721.x
Liao Y, Deprez L, Maljevic S, Pitsch J, Claes L, Hristova D, Jordanova A, Ala-Mello S, Bellan-Koch A, Blazevic D, Schubert S, Thomas EA, Petrou S, Becker AJ, de Jonghe P, Lerche H (2010) Molecular correlates of age-dependent seizures in an inherited neonatal-infantile epilepsy. Brain J Neurol 133:1403–1414. https://doi.org/10.1093/brain/awq057
Howell KB, McMahon JM, Carvill GL, Tambunan D, Mackay MT, Rodriguez-Casero V, Webster R, Clark D, Freeman JL, Calvert S, Olson HE, Mandelstam S, Poduri A, Mefford HC, Harvey AS, Scheffer IE (2015) SCN2A encephalopathy: a major cause of epilepsy of infancy with migrating focal seizures. Neurology 85:958–966. https://doi.org/10.1212/WNL.0000000000001926
Wolff M, Johannesen KM, Hedrich UBS et al (2017) Genetic and phenotypic heterogeneity suggest therapeutic implications in SCN2A-related disorders. Brain. https://doi.org/10.1093/brain/awx054
Nakamura K, Kato M, Osaka H, Yamashita S, Nakagawa E, Haginoya K, Tohyama J, Okuda M, Wada T, Shimakawa S, Imai K, Takeshita S, Ishiwata H, Lev D, Lerman-Sagie T, Cervantes-Barragán DE, Villarroel CE, Ohfu M, Writzl K, Gnidovec Strazisar B, Hirabayashi S, Chitayat D, Myles Reid D, Nishiyama K, Kodera H, Nakashima M, Tsurusaki Y, Miyake N, Hayasaka K, Matsumoto N, Saitsu H (2013) Clinical spectrum of SCN2A mutations expanding to Ohtahara syndrome. Neurology 81:992–998. https://doi.org/10.1212/WNL.0b013e3182a43e57
de Ligt J, Willemsen MH, van Bon BWM, Kleefstra T, Yntema HG, Kroes T, Vulto-van Silfhout A, Koolen DA, de Vries P, Gilissen C, del Rosario M, Hoischen A, Scheffer H, de Vries BB, Brunner HG, Veltman JA, Vissers LE (2012) Diagnostic exome sequencing in persons with severe intellectual disability. N Engl J Med 367:1921–1929. https://doi.org/10.1056/NEJMoa1206524
Rauch A, Wieczorek D, Graf E et al (2012) Range of genetic mutations associated with severe non-syndromic sporadic intellectual disability: an exome sequencing study. Lancet 380:1674–1682. https://doi.org/10.1016/S0140-6736(12)61480-9
D’Gama AM, Pochareddy S, Li M et al (2015) Targeted DNA sequencing from autism spectrum disorder brains implicates multiple genetic mechanisms. Neuron 88:910–917. https://doi.org/10.1016/j.neuron.2015.11.009
Huang Q, Yu L, Ma M, Qi H, Wu Y (2019) Novel SCN2A mutation in a family associated with juvenile-onset myoclonus: case report. Medicine (Baltimore) 98:e14698. https://doi.org/10.1097/MD.0000000000014698
Schwarz N, Bast T, Gaily E et al (2019) Clinical and genetic spectrum of SCN2A-associated episodic ataxia. Eur J Paediatr Neurol EJPN Off J Eur Paediatr Neurol Soc 23:438–447. https://doi.org/10.1016/j.ejpn.2019.03.001
Larsen J, Carvill GL, Gardella E, Kluger G, Schmiedel G, Barisic N, Depienne C, Brilstra E, Mang Y, Nielsen JE, Kirkpatrick M, Goudie D, Goldman R, Jähn JA, Jepsen B, Gill D, Döcker M, Biskup S, McMahon J, Koeleman B, Harris M, Braun K, de Kovel CG, Marini C, Specchio N, Djémié T, Weckhuysen S, Tommerup N, Troncoso M, Troncoso L, Bevot A, Wolff M, Hjalgrim H, Guerrini R, Scheffer IE, Mefford HC, Møller RS, EuroEPINOMICS RES Consortium CRP (2015) The phenotypic spectrum of SCN8A encephalopathy. Neurology 84:480–489. https://doi.org/10.1212/WNL.0000000000001211
Gardella E, Marini C, Trivisano M, Fitzgerald MP, Alber M, Howell KB, Darra F, Siliquini S, Bölsterli BK, Masnada S, Pichiecchio A, Johannesen KM, Jepsen B, Fontana E, Anibaldi G, Russo S, Cogliati F, Montomoli M, Specchio N, Rubboli G, Veggiotti P, Beniczky S, Wolff M, Helbig I, Vigevano F, Scheffer IE, Guerrini R, Møller RS (2018) The phenotype of SCN8A developmental and epileptic encephalopathy. Neurology 91:e1112–e1124. https://doi.org/10.1212/WNL.0000000000006199
Gardella E, Becker F, Møller RS et al (2016) Benign infantile seizures and paroxysmal dyskinesia caused by an SCN8A mutation: BFIS, PKD, and SCN8A mutation. Ann Neurol 79:428–436. https://doi.org/10.1002/ana.24580
Gardella E, Becker F, Møller RS, Schubert J, Lemke JR, Larsen LH, Eiberg H, Nothnagel M, Thiele H, Altmüller J, Syrbe S, Merkenschlager A, Bast T, Steinhoff B, Nürnberg P, Mang Y, Bakke Møller L, Gellert P, Heron SE, Dibbens LM, Weckhuysen S, Dahl HA, Biskup S, Tommerup N, Hjalgrim H, Lerche H, Beniczky S, Weber YG (2016) Benign infantile seizures and paroxysmal dyskinesia caused by an SCN8A mutation. Ann Neurol 79:428–436. https://doi.org/10.1002/ana.24580
Trivisano M, Pavia GC, Ferretti A et al (2019) Generalized tonic seizures with autonomic signs are the hallmark of SCN8A developmental and epileptic encephalopathy. Epilepsy Behav 96:219–223. https://doi.org/10.1016/j.yebeh.2019.03.043
Denis J, Villeneuve N, Cacciagli P, Mignon-Ravix C, Lacoste C, Lefranc J, Napuri S, Damaj L, Villega F, Pedespan JM, Moutton S, Mignot C, Doummar D, Lion-François L, Gataullina S, Dulac O, Martin M, Gueden S, Lesca G, Julia S, Cances C, Journel H, Altuzarra C, Ben Zeev B, Afenjar A, Barth M, Villard L, Milh M (2019) Clinical study of 19 patients with SCN8A-related epilepsy: two modes of onset regarding EEG and seizures. Epilepsia 60:845–856. https://doi.org/10.1111/epi.14727
Hammer MF, Wagnon JL, Mefford HC, Meisler MH (1993) SCN8A-related epilepsy with encephalopathy. In: Adam MP, Ardinger HH, Pagon RA et al (eds) GeneReviews®. University of Washington, Seattle
Johannesen KM, Gardella E, Encinas AC, Lehesjoki AE, Linnankivi T, Petersen MB, Lund ICB, Blichfeldt S, Miranda MJ, Pal DK, Lascelles K, Procopis P, Orsini A, Bonuccelli A, Giacomini T, Helbig I, Fenger CD, Sisodiya SM, Hernandez-Hernandez L, Krithika S, Rumple M, Masnada S, Valente M, Cereda C, Giordano L, Accorsi P, Bürki SE, Mancardi M, Korff C, Guerrini R, von Spiczak S, Hoffman-Zacharska D, Mazurczak T, Coppola A, Buono S, Vecchi M, Hammer MF, Varesio C, Veggiotti P, Lal D, Brünger T, Zara F, Striano P, Rubboli G, Møller RS (2019) The spectrum of intermediate SCN8A-related epilepsy. Epilepsia 60:830–844. https://doi.org/10.1111/epi.14705
Frasier CR, Wagnon JL, Bao YO et al (2016) Cardiac arrhythmia in a mouse model of sodium channel SCN8A epileptic encephalopathy. Proc Natl Acad Sci U S A 113:12838–12843. https://doi.org/10.1073/pnas.1612746113
Boerma RS, Braun KP, van de Broek MPH et al (2016) Remarkable phenytoin sensitivity in 4 children with SCN8A-related epilepsy: a molecular neuropharmacological approach. Neurotherapeutics 13:192–197. https://doi.org/10.1007/s13311-015-0372-8
Kuang Q, Purhonen P, Hebert H (2015) Structure of potassium channels. Cell Mol Life Sci 72:3677–3693. https://doi.org/10.1007/s00018-015-1948-5
Biervert C, Schroeder BC, Kubisch C, Berkovic SF, Propping P, Jentsch TJ, Steinlein OK (1998) A potassium channel mutation in neonatal human epilepsy. Science 279:403–406
Charlier C, Singh NA, Ryan SG, Lewis TB, Reus BE, Leach RJ, Leppert M (1998) A pore mutation in a novel KQT-like potassium channel gene in an idiopathic epilepsy family. Nat Genet 18:53–55. https://doi.org/10.1038/ng0198-53
Dedek K, Fusco L, Teloy N, Steinlein OK (2003) Neonatal convulsions and epileptic encephalopathy in an Italian family with a missense mutation in the fifth transmembrane region of KCNQ2. Epilepsy Res 54:21–27
Miceli F, Striano P, Soldovieri MV, Fontana A, Nardello R, Robbiano A, Bellini G, Elia M, Zara F, Taglialatela M, Mangano S (2015) A novel KCNQ3 mutation in familial epilepsy with focal seizures and intellectual disability. Epilepsia 56:e15–e20. https://doi.org/10.1111/epi.12887
Weckhuysen S, Mandelstam S, Suls A et al (2012) KCNQ2 encephalopathy: emerging phenotype of a neonatal epileptic encephalopathy. Ann Neurol 71:15–25. https://doi.org/10.1002/ana.22644
Ben-Ari Y, Holmes GL (2006) Effects of seizures on developmental processes in the immature brain. Lancet Neurol 5:1055–1063. https://doi.org/10.1016/S1474-4422(06)70626-3
Wang J, Li Y (2016) KCNQ potassium channels in sensory system and neural circuits. Acta Pharmacol Sin 37:25–33. https://doi.org/10.1038/aps.2015.131
Millichap JJ, Park KL, Tsuchida T, Ben-Zeev B, Carmant L, Flamini R, Joshi N, Levisohn PM, Marsh E, Nangia S, Narayanan V, Ortiz-Gonzalez XR, Patterson MC, Pearl PL, Porter B, Ramsey K, McGinnis E, Taglialatela M, Tracy M, Tran B, Venkatesan C, Weckhuysen S, Cooper EC (2016) KCNQ2 encephalopathy: features, mutational hot spots, and ezogabine treatment of 11 patients. Neurol Genet 2:e96. https://doi.org/10.1212/NXG.0000000000000096
Pan Z (2006) A common ankyrin-G-based mechanism retains KCNQ and NaV channels at electrically active domains of the axon. J Neurosci 26:2599–2613. https://doi.org/10.1523/JNEUROSCI.4314-05.2006
Du W, Bautista JF, Yang H et al (2005) Calcium-sensitive potassium channelopathy in human epilepsy and paroxysmal movement disorder. Nat Genet 37:733–738. https://doi.org/10.1038/ng1585
de Kovel CGF, Syrbe S, Brilstra EH et al (2017) Neurodevelopmental disorders caused by de novo variants in KCNB1 genotypes and phenotypes. JAMA Neurol. https://doi.org/10.1001/jamaneurol.2017.1714
Gloyn AL, Diatloff-Zito C, Edghill EL, Bellanné-Chantelot C, Nivot S, Coutant R, Ellard S, Hattersley AT, Robert JJ (2006) KCNJ11 activating mutations are associated with developmental delay, epilepsy and neonatal diabetes syndrome and other neurological features. Eur J Hum Genet 14:824–830. https://doi.org/10.1038/sj.ejhg.5201629
McTague A, Appleton R, Avula S, Cross JH, King MD, Jacques TS, Bhate S, Cronin A, Curran A, Desurkar A, Farrell MA, Hughes E, Jefferson R, Lascelles K, Livingston J, Meyer E, McLellan A, Poduri A, Scheffer IE, Spinty S, Kurian MA, Kneen R (2013) Migrating partial seizures of infancy: expansion of the electroclinical, radiological and pathological disease spectrum. Brain 136:1578–1591. https://doi.org/10.1093/brain/awt073
Lim CX, Ricos MG, Dibbens LM, Heron SE (2016) KCNT1 mutations in seizure disorders: the phenotypic spectrum and functional effects. J Med Genet 53:217–225. https://doi.org/10.1136/jmedgenet-2015-103508
Milligan CJ, Li M, Gazina EV, Heron SE, Nair U, Trager C, Reid CA, Venkat A, Younkin DP, Dlugos DJ, Petrovski S, Goldstein DB, Dibbens LM, Scheffer IE, Berkovic SF, Petrou S (2014) KCNT1 gain of function in 2 epilepsy phenotypes is reversed by quinidine: KCNT1 and human epilepsy. Ann Neurol 75:581–590. https://doi.org/10.1002/ana.24128
Møller RS, Heron SE, Larsen LHG, Lim CX, Ricos MG, Bayly MA, van Kempen M, Klinkenberg S, Andrews I, Kelley K, Ronen GM, Callen D, McMahon J, Yendle SC, Carvill GL, Mefford HC, Nabbout R, Poduri A, Striano P, Baglietto MG, Zara F, Smith NJ, Pridmore C, Gardella E, Nikanorova M, Dahl HA, Gellert P, Scheffer IE, Gunning B, Kragh-Olsen B, Dibbens LM (2015) Mutations in KCNT1 cause a spectrum of focal epilepsies. Epilepsia 56:e114–e120. https://doi.org/10.1111/epi.13071
Juang J-MJ, Lu T-P, Lai L-C et al (2014) Disease-targeted sequencing of ion channel genes identifies de novo mutations in patients with non-familial Brugada syndrome. Sci Rep 4. https://doi.org/10.1038/srep06733
Simms BA, Zamponi GW (2014) Neuronal voltage-gated calcium channels: structure, function, and dysfunction. Neuron 82:24–45. https://doi.org/10.1016/j.neuron.2014.03.016
Imbrici P, Jaffe SL, Eunson LH, Davies NP, Herd C, Robertson R, Kullmann DM, Hanna MG (2004) Dysfunction of the brain calcium channel CaV2.1 in absence epilepsy and episodic ataxia. Brain J Neurol 127:2682–2692. https://doi.org/10.1093/brain/awh301
Sintas C, Carreño O, Fernàndez-Castillo N, Corominas R, Vila-Pueyo M, Toma C, Cuenca-León E, Barroeta I, Roig C, Volpini V, Macaya A, Cormand B (2017) Mutation spectrum in the CACNA1A gene in 49 patients with episodic ataxia. Sci Rep 7:1–9. https://doi.org/10.1038/s41598-017-02554-x
Zangaladze A, Asadi-Pooya AA, Ashkenazi A, Sperling MR (2010) Sporadic hemiplegic migraine and epilepsy associated with CACNA1A gene mutation. Epilepsy Behav 17:293–295. https://doi.org/10.1016/j.yebeh.2009.12.017
Bruun M, Hjermind LE, Thomsen C et al (2015) Familial hemiplegic migraine type 1 associated with parkinsonism: a case report. Case Rep Neurol 7:84–89. https://doi.org/10.1159/000381827
Reinson K, Õiglane-Shlik E, Talvik I et al (2016) Biallelic CACNA1A mutations cause early onset epileptic encephalopathy with progressive cerebral, cerebellar, and optic nerve atrophy. Am J Med Genet A 170:2173–2176. https://doi.org/10.1002/ajmg.a.37678
Myers CT, McMahon JM, Schneider AL et al (2016) De novo mutations in SLC1A2 and CACNA1A are important causes of epileptic encephalopathies. Am J Hum Genet 99:287–298. https://doi.org/10.1016/j.ajhg.2016.06.003
Maejima T, Wollenweber P, Teusner LUC, Noebels JL, Herlitze S, Mark MD (2013) Postnatal loss of P/Q-type channels confined to rhombic-lip-derived neurons alters synaptic transmission at the parallel fiber to purkinje cell synapse and replicates genomic Cacna1a mutation phenotype of ataxia and seizures in mice. J Neurosci 33:5162–5174. https://doi.org/10.1523/JNEUROSCI.5442-12.2013
Mark MD, Maejima T, Kuckelsberg D, Yoo JW, Hyde RA, Shah V, Gutierrez D, Moreno RL, Kruse W, Noebels JL, Herlitze S (2011) Delayed postnatal loss of P/Q-type calcium channels recapitulates the absence epilepsy, dyskinesia, and ataxia phenotypes of genomic Cacna1A mutations. J Neurosci 31:4311–4326. https://doi.org/10.1523/JNEUROSCI.5342-10.2011
Alonso I, Marques JM, Sousa N, Sequeiros J, Olsson IA, Silveira I (2008) Motor and cognitive deficits in the heterozygous leaner mouse, a Cav2.1 voltage-gated Ca2+ channel mutant. Neurobiol Aging 29:1733–1743. https://doi.org/10.1016/j.neurobiolaging.2007.04.005
Krienen FM, Buckner RL (2009) Segregated fronto-cerebellar circuits revealed by intrinsic functional connectivity. Cereb Cortex 19:2485–2497. https://doi.org/10.1093/cercor/bhp135
Galliano E, Gao Z, Schonewille M, Todorov B, Simons E, Pop AS, D'Angelo E, van den Maagdenberg A, Hoebeek FE, de Zeeuw CI (2013) Silencing the majority of cerebellar granule cells uncovers their essential role in motor learning and consolidation. Cell Rep 3:1239–1251. https://doi.org/10.1016/j.celrep.2013.03.023
Fujioka S, Sundal C, Wszolek ZK (2013) Autosomal dominant cerebellar ataxia type III: a review of the phenotypic and genotypic characteristics. Orphanet J Rare Dis 8:14. https://doi.org/10.1186/1750-1172-8-14
Zamponi GW, Lory P, Perez-Reyes E (2010) Role of voltage-gated calcium channels in epilepsy. Pflüg Arch - Eur J Physiol 460:395–403. https://doi.org/10.1007/s00424-009-0772-x
Biel M, Wahl-Schott C, Michalakis S, Zong X (2009) Hyperpolarization-activated cation channels: from genes to function. Physiol Rev 89:847–885. https://doi.org/10.1152/physrev.00029.2008
Benarroch EE (2013) HCN channels: function and clinical implications. Neurology 80:304–310. https://doi.org/10.1212/WNL.0b013e31827dec42
Lucariello M, Vidal E, Vidal S, Saez M, Roa L, Huertas D, Pineda M, Dalfó E, Dopazo J, Jurado P, Armstrong J, Esteller M (2016) Whole exome sequencing of Rett syndrome-like patients reveals the mutational diversity of the clinical phenotype. Hum Genet 135:1343–1354. https://doi.org/10.1007/s00439-016-1721-3
Marini C, Porro A, Rastetter A, Dalle C, Rivolta I, Bauer D, Oegema R, Nava C, Parrini E, Mei D, Mercer C, Dhamija R, Chambers C, Coubes C, Thévenon J, Kuentz P, Julia S, Pasquier L, Dubourg C, Carré W, Rosati A, Melani F, Pisano T, Giardino M, Innes AM, Alembik Y, Scheidecker S, Santos M, Figueiroa S, Garrido C, Fusco C, Frattini D, Spagnoli C, Binda A, Granata T, Ragona F, Freri E, Franceschetti S, Canafoglia L, Castellotti B, Gellera C, Milanesi R, Mancardi MM, Clark DR, Kok F, Helbig KL, Ichikawa S, Sadler L, Neupauerová J, Laššuthova P, Šterbová K, Laridon A, Brilstra E, Koeleman B, Lemke JR, Zara F, Striano P, Soblet J, Smits G, Deconinck N, Barbuti A, DiFrancesco D, LeGuern E, Guerrini R, Santoro B, Hamacher K, Thiel G, Moroni A, DiFrancesco J, Depienne C (2018) HCN1 mutation spectrum: from neonatal epileptic encephalopathy to benign generalized epilepsy and beyond. Brain 141:3160–3178. https://doi.org/10.1093/brain/awy263
EuroEPINOMICS RES Consortium, Nava C, Dalle C et al (2014) De novo mutations in HCN1 cause early infantile epileptic encephalopathy. Nat Genet 46:640–645. https://doi.org/10.1038/ng.2952
DiFrancesco JC, Castellotti B, Milanesi R, Ragona F, Freri E, Canafoglia L, Franceschetti S, Ferrarese C, Magri S, Taroni F, Costa C, Labate A, Gambardella A, Solazzi R, Binda A, Rivolta I, di Gennaro G, Casciato S, D'Incerti L, Barbuti A, DiFrancesco D, Granata T, Gellera C (2019) HCN ion channels and accessory proteins in epilepsy: genetic analysis of a large cohort of patients and review of the literature. Epilepsy Res 153:49–58. https://doi.org/10.1016/j.eplepsyres.2019.04.004
De Fusco M, Marconi R, Silvestri L et al (2003) Haploinsufficiency of ATP1A2 encoding the Na+/K+ pump alpha2 subunit associated with familial hemiplegic migraine type 2. Nat Genet 33:192–196. https://doi.org/10.1038/ng1081
Vanmolkot KRJ, Kors EE, Hottenga J-J, Terwindt GM, Haan J, Hoefnagels WA, Black DF, Sandkuijl LA, Frants RR, Ferrari MD, van den Maagdenberg A (2003) Novel mutations in the Na+, K+-ATPase pump gene ATP1A2 associated with familial hemiplegic migraine and benign familial infantile convulsions. Ann Neurol 54:360–366. https://doi.org/10.1002/ana.10674
Hirose S, Okada M, Kaneko S, Mitsudome A (2000) Are some idiopathic epilepsies disorders of ion channels?: A working hypothesis. Epilepsy Res 41:191–204
Provini F, Plazzi G, Tinuper P et al (1999) Nocturnal frontal lobe epilepsy. A clinical and polygraphic overview of 100 consecutive cases. Brain J Neurol 122(Pt 6):1017–1031
Steinlein OK, Kaneko S, Hirose S (2012) Nicotinic acetylcholine receptor mutations. In: Noebels JL, Avoli M, Rogawski MA et al (eds) Jasper’s basic mechanisms of the epilepsies, 4th edn. National Center for Biotechnology Information (US), Bethesda
Nobili L, Proserpio P, Combi R, Provini F, Plazzi G, Bisulli F, Tassi L, Tinuper P (2014) Nocturnal frontal lobe epilepsy. Curr Neurol Neurosci Rep 14:424. https://doi.org/10.1007/s11910-013-0424-6
Puligheddu M, Melis M, Pillolla G et al (2017) Rationale for an adjunctive therapy with fenofibrate in pharmacoresistant nocturnal frontal lobe epilepsy. Epilepsia 58:1762–1770. https://doi.org/10.1111/epi.13863
Johannesen K, Marini C, Pfeffer S et al (2016) Phenotypic spectrum of GABRA1: from generalized epilepsies to severe epileptic encephalopathies. Neurology 87:1140–1151. https://doi.org/10.1212/WNL.0000000000003087
Roden WH, Peugh LD, Jansen LA (2010) Altered GABAA receptor subunit expression and pharmacology in human Angelman syndrome cortex. Neurosci Lett 483:167–172. https://doi.org/10.1016/j.neulet.2010.08.001
Furukawa H, Singh SK, Mancusso R, Gouaux E (2005) Subunit arrangement and function in NMDA receptors. Nature 438:185–192. https://doi.org/10.1038/nature04089
Carvill GL, Regan BM, Yendle SC, O'Roak BJ, Lozovaya N, Bruneau N, Burnashev N, Khan A, Cook J, Geraghty E, Sadleir LG, Turner SJ, Tsai MH, Webster R, Ouvrier R, Damiano JA, Berkovic SF, Shendure J, Hildebrand MS, Szepetowski P, Scheffer IE, Mefford HC (2013) GRIN2A mutations cause epilepsy-aphasia spectrum disorders. Nat Genet 45:1073–1076. https://doi.org/10.1038/ng.2727
Pierson TM, Yuan H, Marsh ED, Fuentes-Fajardo K, Adams DR, Markello T, Golas G, Simeonov DR, Holloman C, Tankovic A, Karamchandani MM, Schreiber JM, Mullikin JC, PhD for the NISC Comparative Sequencing Program, Tifft CJ, Toro C, Boerkoel CF, Traynelis SF, Gahl WA (2014) GRIN2A mutation and early-onset epileptic encephalopathy: personalized therapy with memantine. Ann Clin Transl Neurol 1:190–198. https://doi.org/10.1002/acn3.39
Lemke JR, Hendrickx R, Geider K, Laube B, Schwake M, Harvey RJ, James VM, Pepler A, Steiner I, Hörtnagel K, Neidhardt J, Ruf S, Wolff M, Bartholdi D, Caraballo R, Platzer K, Suls A, de Jonghe P, Biskup S, Weckhuysen S (2014) GRIN2B mutations in west syndrome and intellectual disability with focal epilepsy: GRIN2B mutations in epilepsy. Ann Neurol 75:147–154. https://doi.org/10.1002/ana.24073
Andreoli V, De Marco EV, Trecroci F et al (2014) Potential involvement of GRIN2B encoding the NMDA receptor subunit NR2B in the spectrum of Alzheimer’s disease. J Neural Transm Vienna Austria 1996 121:533–542. https://doi.org/10.1007/s00702-013-1125-7
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The Authors state that this work has not received any funding and declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Online resource 1
EEG changes at different ages in an adult patient with Dravet syndrome and drug-resistant seizures (personal observation, routine EEG bipolar montage, high-pass filter 1.6 Hz, low-pass filter 70 Hz, amplitude sensitivity 100 microV/cm). A. Mild slowing of background activity at 7–7.5 Hz, short electrographic seizure arising in the right frontotemporal regions (20 years of age). B. Bilateral diffuse slow activity at 5.5–6 Hz with rare sharp waves in the right frontotemporal regions (25 years of age). C. Slow background activity at 6–7 Hz with abundant interictal discharges, especially in frontotemporal regions bilaterally and over the midline channels (26 year of age). D. Mild slowing of background activity at 7–7.5 Hz, persisting sharp waves in the right temporal regions (32 years of age) (PNG 3612 kb)
Rights and permissions
About this article
Cite this article
Bartolini, E., Campostrini, R., Kiferle, L. et al. Epilepsy and brain channelopathies from infancy to adulthood. Neurol Sci 41, 749–761 (2020). https://doi.org/10.1007/s10072-019-04190-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-019-04190-x