Abstract
Rescue behavior is a kind of prosocial response that involves the provision of help to a stressed individual. This behavior has been observed in domestic dogs assisting their owners when they pretended to be trapped. Given the role of the hormone oxytocin as a facilitator for prosocial behavior, we aimed to evaluate the effects of its intranasal administration on the rescue behavior of dogs directed to their owners. In addition, we used the Monash Dog Owner Relationship Scale (MDORS) to assess whether the dog–owner bond was associated with this behavior. After receiving either oxytocin or saline, dogs participated in a stressed condition in which their owner pretended to be stressed inside of a box, or a control one, in which the owner was in a calm state. Dogs released their owners more frequently in the stressed condition. Contrary to our expectations, dogs who received oxytocin were less likely to open the box and took longer to do so than those that received saline. Regarding the dog–owner bond, dogs in the stressed condition who received oxytocin exhibited a lower rate and a higher latency of openings the more intense the bond was, while the opposite pattern was observed in dogs in the control condition who received saline. In conclusion, dogs would rescue their owners when they pretended to be trapped and stressed. Both oxytocin administration and the bond with the owner appear to modulate this behavior, but further studies are needed to inquire into the involved mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Domestic dogs have benefited humans for centuries, both as companion animals and by fulfilling various working roles such as herding or guarding, and more recently, aiding disabled individuals, searching and rescuing missing people and detecting explosives, among others (Coppinger and Coppinger 2001; Serpell 2017). Dogs are usually extensively trained to carry out these activities (e.g., Svartberg 2002). However, to a lesser extent, some evidence suggest that dogs may be able to collaborate with people spontaneously, particularly when they understand their goals or needs (e.g., Bräuer, Schönefeld and Call 2013; Piotti and Kaminski 2016).
Rescue behavior is a prosocial response that refers to the actions of one individual to help another who is stressed or in danger to terminate this negative state (Nowbahari and Hollis 2010). Recent studies have investigated whether dogs are able to display such behavior towards their owners when they pretend to be stressed. To this end, they have employed the trapped-other paradigm, adapted from the one that Ben-Ami Bartal, Decety and Mason (2011) used to study this behavior in rats (Carballo et al. 2020; Sanford et al. 2018; Van Bourg et al. 2020).
The first of these studies was from Sanford et al. (2018), who evaluated whether dogs released their owners when they asked for help while trapped in a small room. To free their owners, dogs had to move a door which was held by magnets. The owners either said “Help” in a distressed tone and made crying sounds (experimental condition) or said it in a neutral tone and hummed (control condition). Results showed that almost half of the dogs opened the door, but there were no differences between conditions in the number of openings nor their latency. In addition, no differences between conditions were found in stress behaviors or heart rate variation. However, a subsequent analysis focusing only on dogs that opened the door, revealed a lower latency of openings in the experimental condition in which the owner pretended to be stressed, which suggests the emotional state of the owner may have had some effect on this behavior. Moreover, those dogs that opened, decreased their stress behaviors during the task compared to their baseline, while those that did not open exhibited an inverse pattern. These results suggest a connection between the dogs stress levels and their rescue behavior.
In another study, Carballo et al. (2020) investigated whether dogs were able to rescue their owners who pretended to be trapped inside of a box and emitted signs of stress for 3 consecutive trials. In the experimental condition, the owner was instructed to behave as he would do in a real emergency (e.g., calling the dog, yelling for help, hitting the walls of the box), while in the control condition the owner was told to stay calm inside of the box. The authors registered the openings rate and its latency as well as behavioral and physiological indicators of the dogs’ stress. Results indicate that dogs opened more often and faster when the owners were stressed, suggesting they were sensitive to their emotional state. Regarding stress indicators, dogs’ heart rate in the experimental condition was higher during all trials compared to the baseline, while control dogs showed a decrease in heart rate across task trials. However, no differences were found in other indicators such as stress-related behaviors or salivary cortisol levels. Considering these findings, it is unclear whether dogs got stressed based on the negative emotional state of their owners or got aroused by the situation. On the other hand, the authors highlighted that learning and prior experiences play a major role on this behavior. Specifically, they found that dogs trained for search and rescue were faster opening the box than pet dogs. This result suggests that trained dogs may have generalized their previous learning to this novel task. Moreover, in both groups opening latency decreased across trials, which indicates that opening behavior was more efficient the more experience the dogs had with the task.
Finally, Van Bourg et al. (2020) emphasized the importance of the dogs having the instrumental ability to complete the task and thus carry out the rescue behavior. They used the trapped-other paradigm and dogs participated in semi-random order in three conditions: (1) distress test: in which the owner asked for help in a distressed tone, (2) reading test: in which the owner read a magazine and said “Help” in a neutral tone, (3) food test: in which there was food instead of the owner inside of the box. The latter condition assessed whether dogs were able to open the box when they were motivated to do so. About a third of the dogs opened the box, but no differences were found between the distress and reading conditions. However, when considering only those dogs that opened in the food condition, it was found that they opened more often when the owner was stressed than when he was reading. Therefore, having the ability to open the device appears to be key to exhibit rescue behavior in this paradigm. Moreover, they surveyed previous experiences and found that practice opening objects in their daily lives was a strong predictor of rescue behavior in this task. In line with the findings of Carballo et al. (2020), prior experience appears to be relevant and may be generalizable to new situations. Nevertheless, this result is based on the owner’s opinion and more objective measures of the dogs’ previous abilities manipulating objects are needed to further clarify this issue. On the other hand, dogs exhibited more stress signals during the distress test than during the reading or food tests, which suggests they expressed an emotional state similar to their owners. It is important to note that this is the only one of these studies that found differences in stress behaviors across conditions. All in all, in the aforementioned studies, dogs were able to release their owners when they were trapped and exhibited stress signals, and this behavior may be facilitated by the dogs’ ability to open the device.
It has been proposed that the mechanism underlying rescue behavior is emotional contagion (Carballo et al. 2020; Sanford et al. 2018; Van Bourg et al. 2020), which implies that an individual experiences the same emotional state or activation of another (de Waal 2008). If such is the case, the dog would experience an emotional state of stress similar to that of their trapped owner. This hypothesis is supported by some evidence. For instance, when the owner emitted stress signs there was an increase in the dogs’ heart rate (Carballo et al. 2020) and stress behaviors (Van Bourg et al. 2020). However, neither Carballo et al. (2020) nor Sanford et al. (2018) found differences in stress behaviors and there were no differences in salivary cortisol between conditions (Carballo et al. 2020). These discrepancies may be due to methodological differences across studies. In particular, the score used by Van Bourg et al. (2020) did not include some behaviors which are often used to measure stress in dogs such as lip licking, shaking, ears down and tail down (e.g., Beerda et al. 1997). It is possible that this score, similarly to the increase in heart rate, may reflect a global effect of activation rather than a stress response. These controversial findings put the emotional contagion hypothesis in doubt as the mechanism underlying rescue behavior and highlight the need for further investigations to clarify the factors involved in this response.
In this sense, a candidate to modulate rescue behavior is the neuro-hormone oxytocin (OT) as it is known to be related to prosocial behavior and emotional contagion in several species (for reviews: Kikusui et al. 2019; Robinson et al. 2019). In dogs, it is yet unknown the impact of OT on rescue behavior, but it has been observed that OT administration increases some sociocognitive responses directed towards people such as gazing (Barrera et al. 2018; Nagasawa et al. 2015) and social orientation (Romero et al. 2014), as well as the following of communicative cues (Macchitella et al. 2017; Oliva et al. 2015, 2019; for a review, Kis, Ciobica and Topál 2017a). Considering these findings, the aim of the present study was to evaluate the effects of intranasal OT administration on the rescue behavior of dogs when their owners appeared to be trapped. Following the hypothesis that OT increases prosocial behavior and emotional contagion towards stressed individuals, we predict that OT administration will increase rescue behavior in dogs.
On the other hand, the bond between the helper and the affected individual has been shown to facilitate prosocial behavior in various species (for a review, Rault 2019). The only study that included the relationship between dogs and their owners when analyzing rescue behavior was Sanford et al. (2018). To this end, they considered gazing duration during an unsolvable task as an indicator of the closeness of the dog–owner bond. Results showed that dogs that opened in the experimental condition were the ones that gazed more towards the owner, compared to those that did not open. The authors suggested that closely bonded dogs may have opened more in the experimental condition because they were more attentive to their owners’ emotional states than those with a less intense bond. However, although gazing is important for bonding, this study did not include a more direct standardized measure of the dog–owner bond (e.g., obtained through a behavioral test or owner questionnaire). Moreover, as gazing is related to attention (Bayliss et al. 2011), the measurement of gazing time may have partly captured the attention the dog paid to the owner and not exclusively the nature of their bond. Taking this into account, in the present study, we evaluated whether the level of the bond between dogs and owners was associated with rescue behavior. To this aim, the owners received a Spanish translation of the Monash Dog Owner Relationship Scale (MDORS) (Dwyer, Bennett and Coleman 2006). This scale measures the dog–owner bond through three subscales that evaluate dog–owner interaction, the perceived emotional closeness, and the perceived cost of this relationship (Dwyer et al. 2006). We expected to find a positive correlation between the level of the bond and rescue behavior.
In sum, we aim to evaluate the effects of OT administration on rescue behavior as well as whether the level of the dog–owner bond influences this prosocial behavior. These findings will further extend the knowledge regarding the proximal mechanisms that regulate dogs’ prosocial rescue behavior towards people.
Method
Ethical statement
The protocol that involved the evaluation of the dogs and their owners was approved by the Institutional commission for the care and use of laboratory animals (CICUAL) at the Medical Research Institute, IDIM UBA-CONICET (Res. Nro. 108–19) and complied with the current Argentine law of animal protection (Law 14.346). All owners involved were adults (> 18 years old) and expressed their consent for the participation of their dogs in the present study.
Subjects
The sample included 48 healthy pet dogs of various breeds and mixes (21 males and 27 females, 34 neutered and 14 intact; mean age: 4.69, SD ± 2.83 years). We evaluated 11 additional dogs that could not be included in the final sample as their owners did not properly follow the instructions during the task. Only 4 dogs had a basic obedience level of training, while the rest were untrained, and none had specific training for this task (see Table 1 for further details).
Apparatus
We constructed a wooden box of 1 m2 base and 1.75 height with a transparent Plexiglas’ door that allowed the dog to see the owner inside. Dogs could open the box by pawing and nuzzling at a small slit between the door and the frame, as well as by pushing a rock that held the door shut (see Fig. 1).
Tests were held outdoors in a 10 × 15 m field belonging to CONICET in Bahía Blanca, Argentina. There was a small waiting room in which the dog and the experimenters (Es) could stay when needed (see Procedure). Two cameras (SONY DCR-SR88 and SONY DCR 308) were placed on tripods at opposite sides of the field to capture the situation from different angles.
Procedure
Owners took their dogs to the testing field and let them explore freely to get familiarized with the place and the box. After this 10 min habituation phase, the dog received either 16 IU of OT (Syntocinon Spray, Novartis) or saline solution according to the treatment assigned to them. One experimenter (E1) applied two puffs of spray (OT or saline) in each nostril while the owner was present and held the dog if needed. The other experimenter (E2) and the owner were blind to the treatment the dog received. This was done 40 min before behavioral testing. This interval is the time OT takes to reach central levels (MacDonald et al. 2011; Quintana et al. 2015) and has been frequently used in prior literature (for a review, Kis et al. 2017a).
During the 40 min wait, the owners completed the MDORS and, once they finished, E1 gave them the instructions for the task. Meanwhile, the dogs were free to explore the field. The owners and Es did not interact with them, avoiding physical and eye contact, as it could increase their endogenous OT levels (e.g., Handlin et al. 2011; Nagasawa et al. 2015; Rehn et al. 2014a).
The rescue task followed the same procedure as in Carballo et al. (2020). Before starting, E2 took the dog to the waiting room. Then, the owner entered the box and E2 placed the rock that held the door shut. After the owner was locked inside of the box, E2 freed the dog and entered the room, calling out “now!” (in Spanish, “¡ya!”) which marked the beginning of the first trial. Only the dog and the owner were outdoors during trials.
Dogs from each treatment (OT and saline) were semi-randomly assigned to two conditions according to their sex and breed group:
Stressed owner (SO): Once E2 signaled the start of the trial, the owners began to ask for help while pretending to be trapped inside of the box. Owners were instructed to act like they would do in a real emergency situation (i.e., yell for help, call the dog, pretend to cry, hit the walls of the box). During the trial, E1 stayed in the waiting room and registered how many times and in what moments the owner called the dog’s name.
Calm calling owner (CCO): The owners stayed calm inside of the box and called the dog in a neutral tone, avoiding eye contact. As owners in the SO group were instructed to behave as they would in a real emergency, the frequency and timing of their calls were variable. To account for this, each owner in the CCO condition was paired to a previous owner in the SO condition and instructed to calmly call his dog the same amount of times and at the same moments that the previous SO did. To do this, they were provided with a chronometer and a piece of paper indicating when to call their dogs.
There were 3 trials lasting a maximum of 2 min or until the dog opened the box. For each trial, an opening was considered when the dog moved the door more than 45° or put his head completely inside of the box.
If the dog opened the door before the 2 min period had passed, the owner got out and verbally congratulated the dog. If after 2 min the dog had not opened, E2 returned, took the dog back to the waiting room and then allowed the owner to get out of the box.
There was an intertrial interval of 1 min. During this time, the dogs stayed in the waiting room with E2, while the owner and E1 waited outside. Each dog was evaluated in only one condition (SO or CCO) and received only one treatment (OT or saline).
Data coding
Rescue task
We measured the following behaviors directed to the box: (1) total openings rate: total number of openings across trials, divided by the number of trials completed for each dog. We used the rate of openings instead of the total number of openings because 6 trials had to be discarded due to technical problems. (2) Latency of openings: time (s) from the start of each trial until the opening (if the dog did not open the door, we coded the maximum possible latency of 120 s). (3) Contact with the box: time (s) the dogs physically contacted the box (touching it with their snout, sniffing at it or pushing and scratching with their paws) during each trial. (4) Proximity to the box: time (s) the dogs spent with their front paws less than 50 cm from the box during each trial.
We also recorded the following behavioral indicators of dogs stress during each trial: (1) ears down: time (s) dogs had their ears folded down towards their head, (2) tail down: time (s) dogs held their tails lower than normal or between their legs; (3) vocalizations: number of barks and whines. We considered one vocalization event each time the noises the dog emitted were paused for at least 1 s; (4) lip licking: number of times the dog licked any part of its snout.
For all variables, we divided the duration or frequency of the behavior by the total duration of the trial as it was not the same in all trials (i.e., the duration of the trial varied according to when the door was opened).
Dogs whose anatomy made it impossible to assess their ears or tail (i.e., droopy ears, cropped ears, docked tail) were excluded from the analysis of such indicators (9 for ears down and 9 for tail down).
One observer coded 100% of the videos while a second observer analyzed 20% of them. Both were blind to the treatment the dogs had received. Inter-observer reliability was excellent (rs > 0.95, ps < 0.001, N = 10).
MDORS
We considered the total score of the MDORS as a measure of the level of the dog–owner bond. This score comprises a Likert scale that ranges from a very low (1) to a very high (5) bond level. This measure was obtained by adding all the items of the scale and inverting the value of the ones referring to the perceived cost of the relationship. We also coded the subscales: dog–owner interaction, perceived emotional closeness and perceived cost.
Data analysis
Rescue task
We constructed a Generalized Lineal Model (GLM) for the total openings rate. The distribution was set to normal and related to the fixed factors through the identity link function. The model included the following fixed factors: condition (SO, CCO), treatment (OT, saline), sex (male, female), neutering status (neutered, intact), owner’s sex (male, female) and age as a covariable. The model also included the two-way interactions between condition and the factors treatment, sex, neutering status and age. Non-significant factors were removed step by step according to the backwards method. The factors condition, treatment and their interaction were preserved due to their theoretical relevance.
To analyze the variables latency of openings, contact with the box, proximity to the box, ears down and tail down, we constructed Mixed Generalized Linear Models (GLMM) for each dependent variable. Latency of openings had a gamma distribution with a log link function, while the other variables followed normal distributions with identity link functions. The models included as fixed factors: condition (SO, CCO), treatment (OT, saline), trial (1, 2, 3), sex (male, female), neutering status (neutered, intact), owner’s sex (male, female) and age. The model also included the two-way interactions between condition and the factors treatment, trial, sex, neutering status and age. Non-significant factors were removed step by step according to the backwards method. The factors condition, treatment, trial and their interaction were preserved due to their theoretical relevance. For all models, the random effects structure included intercepts to account for variability across subject’s ID. We used the Satterthwaite approximation to estimate the degrees of freedom due to different cluster sizes in the between and within-participants factors. Post hoc comparisons were conducted using paired contrasts (sequential adjusted Bonferroni) when required. Furthermore, we calculated the effect size (Cohen’s d) from the parameters estimated by the final models using Stata 13 Software.
The variables vocalizations and lip lickings were not analyzed due to the elevated number of zeros in the data (see descriptive statistics in Table 3).
MDORS
To assess whether the level of the bond was associated with the opening behavior and indicators of dogs stress, subjects were divided into 4 groups according to their treatment and condition (SO–OT, SO–saline, CCO–OT, and CCO–saline). For each group we carried out a Spearman’s correlation test between the total score of the scale and its subscales (dog–owner interaction, perceived emotional closeness, perceived costs) and the variables total openings rate, latency of openings, contact with the box, proximity to the box, ears down and tail down. Vocalizations and lip lickings were not included due to the elevated number of zeros in the data. We computed the effect size (Cohen’s d) using Stata Software.
All tests were two tailed (α = 0.05) and the data were analyzed with SPSS 22.0.
Results
Rescue task
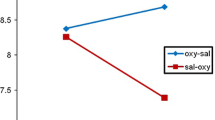
For the total openings rate measure, the final model included condition, treatment, and their interaction. We found a main effect of condition (X2 (1) = 5.34, p = 0.021, d = 0.39) showing that the total openings rate was higher for dogs in the SO condition than those in the CCO. In addition, a significant effect of treatment was found (X2 (1) = 8.23, p = 0.003, d = 0.36) indicating that dogs who received saline released their owners more frequently than dogs who received OT (see Fig. 2). No effects of the condition by treatment interaction were observed (p > 0.05).
Mean and SEM of the total openings rate for each group. SO–OT: dogs of the stressed owner condition that received oxytocin; SO–saline: dogs of the stressed owner condition that received saline; CCO–OT: dogs of the calm calling owner condition that received oxytocin; CCO–saline: dogs of the calm calling owner condition that received saline
Regarding the latency of openings, the final model included condition, treatment, trial, and the interactions of condition by treatment, condition by trial and treatment by trial. These factors were included in the final model of all the variables reported unless stated otherwise.
The effect of condition approached significance (F (1,44) = 3.92, p = 0.054, d = 0.57) as dogs in the CCO condition tended to have higher latencies to open the box. Furthermore, dogs that received OT took longer to open the box than those that received saline (F (1,44) = 9.28, p = 0.004, d = 0.88). Finally, we found a main effect of trial (F (2,87) = 14.77, p < 0.001, d = 0.95) on the latency to open the box. Pairwise comparison showed that dogs took longer to open the box in the first trial compared to the second (t(118) = 4.01, p < 0.001) and third (t(120) = 4.15, p < 0.001) trials (see Fig. 3). No other significant differences were found (ps > 0.05).
Mean and SEM of the latency of openings in each trial for each group. SO–OT: dogs of the stressed owner condition that received oxytocin; SO–saline: dogs of the stressed owner condition that received saline; CCO–OT: dogs of the calm calling owner condition that received oxytocin; CCO–saline: dogs of the calm calling owner condition that received saline
Concerning the contact with the box, we observed a main effect of condition (F (1,44) = 15.29, p < 0.001, d > 1) as dogs in the SO condition stayed longer in contact with the box than dogs in the CCO condition. Furthermore, there was a main effect of trial (F (2,89) = 4.90, p = 0.010, d = 0.54) as dogs in the second trial spent more time in contact with the box than in the third trial (t(89) = 3.03, p = 0.010). In addition, this increment in the second trial approached significance compared to the first trial (t(90) = 2.20, p = 0.060) (see Table 2). No other significant differences were found (ps > 0.05).
Regarding proximity to the box, the final model included the aforementioned factors, as well as sex as a fixed factor and the interaction of condition by sex. We observed a main effect of condition (F (1,41) = 13.54, p = 0.001, d > 1) as dogs in the SO condition stayed longer in proximity to the box than dogs in the CCO condition (see Table 2). Furthermore, we observed a main effect of trial (F (2,88) = 12.57, p < 0.001, d = 0.16), dogs in the first and second trial spent more time near the box than in the third trial (trial 1 vs 3: t(88) = 4.62, p < 0.001; trial 2 vs 3: t(87) = 3.96, p < 0.001). An interaction effect between condition and trial (F (2,88) = 4.57, p = 0.013, d = 0.61), indicates that dogs in the SO condition spent more time in proximity to the box in the first (t(111) = 4.41, p < 0.001) and third (t(110) = 2.68, p = 0.009) trials than dogs in the CCO condition. Finally, we found a sex by condition interaction effect (F (1,41) = 6.92, p = 0.012, d = 0.76). In the SO condition females spent more time in proximity to the box than males (t(42) = 2.96, p = 0.005). In addition, females spent more time in proximity in the SO condition than in the CCO condition (t(42) = 4.76, p < 0.001), while no differences were found for male. No other significant differences were found (ps > 0.05).
Concerning the stress-related behavioral measurements (ears and tail down) (see Table 3), we observed that dogs in the SO condition spent significantly more time with their ears down than dogs in the CCO condition (F (1,43) = 6.34, p = 0.016, d = 0.72). No other significant differences were found (ps > 0.05).
MDORS
Considering the behaviors directed to the box, for dogs in the SO–OT group, we found a significant negative correlation between the MDORS total score and the total openings rate (rho = -0.702, p = 0.024, N = 10, d = 0.29), as well as a positive correlation with the latency of openings (rho = 0.643, p = 0.045, N = 10, d = 0.35). These findings suggest that dogs in the SO–OT group who had a stronger bond with their owners opened the box less frequently and took longer to do so. Furthermore, the score of the dog–owner interaction subscale had a significant negative correlation with the total openings rate (rho = -0.712, p = 0.021, N = 10, d = 0.27) and a positive correlation with the latency of openings (rho = 0.677, p = 0.031, N = 10, d = 0.32). Thus, dogs in the SO group who interact more with their owners in their daily lives, opened the box less and took longer to solve the task when they did it (see Table 4).
For dogs in the SO–saline group, we found a significant positive correlation between the proximity to the box and both the MDORS total score (rho = 0.586, p = 0.035, N = 13, d = 0.33) and the perceived emotional closeness subscale (rho = 0.617, p = 0.025, N = 13, d = 0.31). Thus, dogs in the SO–saline who had a stronger bond and a higher perceived emotional closeness with their owners, remained longer in proximity to the box (see Table 4).
Regarding stress behaviors, for dogs in the CCO–OT group, we found a significant negative correlation between tail down and both the MDORS total score (rho = – 0.726, p = 0.007, N = 12, d = 0.22) and the dog–owner interaction subscale (rho = – 0.825, p = 0.001, N = 12, d = 0.14). Thus, dogs in the CCO–OT group who had a stronger bond and greater interaction with their owners, stayed less time with their tail down (see Table 4).
Finally, we found a significant and positive correlation for dogs in the CCO–saline group between the MDORS total score and the openings rate (rho = 0.766, p = 0.002, N = 13, d = 0.23), and a significant negative correlation with the latency to open the box (rho = – 0.677, p = 0.013, N = 13, d = 0.32). Thus, contrary to dogs in the SO–OT group, in the CCO condition dogs that received saline and had stronger bonds with their owners, opened the box more frequently and took less time to release their owners. Furthermore, the scores of the perceived emotional closeness subscale correlated significantly and positively with the total openings rate (rho = 0.787, p = 0.001, N = 13, d = 0.21) and negatively with the latency of openings (rho = – 0.643, p = 0.018, N = 13, d = 0.35). Hence, dogs with a closer affective bond with their owners opened the box more frequently and faster than dogs with a more distant bond. Finally, the perceived emotional closeness subscale had a significant positive correlation with tail down (rho = 0.634, p = 0.020, N = 13, d = 0.29). Thus, dogs in the CCO–saline group who had a stronger emotional closeness with their owners, remained longer with their tail down (see Table 4). No other significant results were found (ps > 0.05).
Discussion
The aim of this study was to analyze the effects of intranasal OT administration on the rescue behavior of dogs towards their owners using the trapped-other paradigm. In addition, we analyzed if the level of the dog–owner bond was associated with this prosocial behavior. Results show that dogs had a higher rate of openings, tended to open faster and stayed longer in proximity and contact with the box when their owners pretended to be trapped and emitted stress signs compared to when they were calm. In line with previous studies (Carballo et al. 2020; Sanford et al. 2018; Van Bourg et al. 2020) these findings indicate that dogs are able to rescue their owners without prior training. The observed rescue behavior suggests dogs are sensitive to the emotional state of their owners in this situation. This finding is consistent with what has been observed in other protocols in which dogs were able to discriminate different human emotions, even from cues presented in only one sensory modality (e.g., Albuquerque et al. 2016; D’Aniello et al. 2018; Müller et al. 2015).
Besides requiring the ability to discriminate human emotional states, it has been proposed that rescue behavior is modulated by emotional contagion. According to this hypothesis, in this paradigm dogs would open the door because they experienced stress due to their owners’ anguish (Carballo et al. 2020; Sanford et al. 2018; Van Bourg et al. 2020). To assess this, we examined the dogs’ stress behaviors during the task. We found that dogs spent more time with their ears down in the stressed owner condition than when the owner was calm. This result is consistent with Van Bourg et al. (2020), who found more stress behaviors in the distress test compared to the reading and food ones. These findings suggest that dogs’ rescue behavior may be motivated by a basic empathetic mechanism, similar to what has been found in other paradigms (e.g., Huber et al. 2017; Romero, Konno and Hasegawa 2013; Yong and Ruffman 2014). However, out of all of the stress indicators we measured (i.e., ears and tail down, vocalizations, lip licking), ears down was the only one that evidenced stress during the task. Moreover, in previous studies using similar tasks, stress behaviors and salivary cortisol did not differ according to the owner being stressed or calm (Carballo et al.2020; Sanford et al. 2018). Therefore, evidences of dogs being stressed during the rescue task and thus experiencing emotional contagion are inconclusive.
However, it is important to remark that, to this date, only in Carballo et al. (2020) and this study, differences between the experimental and control conditions were found considering the whole sample. On the contrary, in prior studies, the differences between groups were only observed when analyzing the dogs that opened the door during the task (Sanford et al. 2018) or the dogs who were able to open when there was food inside of the box (Van Bourg et al. 2020). One possibility is that the protocol used in this and Carballo et al. (2020) studies is more sensitive to evocate this response. Thus, the higher rate of openings may be due to the owner’s spontaneous calls for help which may be more salient to the dogs than the standardized help requests used in other studies (Sanford et al. 2018; Van Bourg et al. 2020). Indeed, spontaneous calls for help may be more familiar to the animals and, as it was observed in another study of helping behavior, it may facilitate the dogs’ understanding of the owners’ intent to communicate their need for help (Bräuer et al. 2013). Although this protocol’s feature increases variability in the owners’ acting, it allows for a more ecological simulation of the rescue situation. Another possibility is that the studies differed in the behaviors required to open the box. While in this and Carballo et al. (2020) studies the dogs had to move a rock to open the door, in Sanford et al. (2018) dogs had to press a magnet lock and in Van Bourg et al. (2020) they had to move a foam board. These responses varied in complexity and familiarity and thus they may have carried different degrees of difficulty for the dogs. This could have influenced the dogs’ performance and thus affected the measured dependent variables such as latency, opening rate and stress behaviors, contributing to the heterogeneity of the results across experiments. In this regard, we believe that a strength of this study is that it used a protocol which has already been successfully applied (Carballo et al. 2020). Future investigations should take into account the level of complexity of the behavioral responses required to perform the rescue task.
On the other hand, we observed an effect of learning during the task, as the latency to open the door decreased across trials, replicating prior results by Carballo et al. (2020). This suggests that experience during the task favors rescue behavior and is in line with findings indicating that prior experiences may facilitate the expression of this behavior (Carballo et al. 2020; Van Bourg et al. 2020). Furthermore, the time dogs spent in contact and proximity to the box decreased across trials. This could imply an effect of habituation to the situation or an effect of fatigue, but in either case it did not interfere with adequately solving the task.
Controlling for effects of sex, we found that female dogs spent more time next to the box than males in the stressed owner condition. This could be due to sex differences in sociocognitive abilities. In this sense, is has been observed that females are more inclined to interact with humans in some social tasks compared to males (for a review, Scandurra et al. 2018). For instance, during a sociability task, females spent more time close and in physical contact with a person than males (Lore and Eisenberg 1986). In addition, when exposed to the chemosignals produced by the sweat of a happy person, female dogs were more socially oriented (proximity and gazing) towards a stranger than male dogs (D’Aniello et al. 2021). Furthermore, this finding is in accordance with prior results of trapped-other studies in which female rats were faster to release their stressed conspecific than males and were also more active than males during this situation (Ben-Ami Bartal et al. 2011). The authors suggested that these differences in helping behavior may be due to a higher empathic ability in females. However, these results should be taken with caution, as sex differences were not observed in the other variables.
Regarding OT administration, contrary to our expectations, it negatively impacted rescue behavior. We found that dogs who received OT exhibited a lower rate of openings and a higher latency to do so. This effect appeared in the stressed owner condition as well as in the calm calling owner one, so it seems to be relatively independent from the emotional state expressed by the owner. We found two possible explanations for this decrease in the observed responses.
First, besides the previously mentioned effects on affiliative behavior, OT has been reported to have anxiolytic properties in various species (for a review, Buttner 2016; Onaka and Takayanagi 2019). In dogs, these effects include a decrease in heart rate accompanied by an increase in heart rate variability which is related to reduced stress levels (Kis et al. 2015; Kovács et al. 2016), and a decrease of yawning, considered as a stress indicator (Kis et al. 2020). Furthermore, when presented with pictures of angry humans, dogs decreased their gaze (Kis et al. 2017b) and eye fixation (Somppi et al. 2017). In the trapped-other paradigm employed here, OT administration may have made the situation less threatening to the dogs, thus affecting their prosocial behavior. This hypothesis is supported by findings of Ben-Ami Bartal et al. (2016), who observed the administration of the anxiolytic midazolam worsened rescue behavior in rats. However, considering that in the present study, OT administration decreased openings, increased latency in both conditions (stressed and calm calling owner) and had no significant effects on any of the stress indicators, it is unlikely for this reduction to be due to the anxiolytic effects of OT.
Second, Yamagishi et al. (2020) have recently shown that high OT levels may deteriorate rescue behavior. They observed that OT administration affected this behavior in rats housed in pairs, but not in those housed alone. The authors speculated that endogenous OT levels were higher for those rats living in pairs compared to ones living alone. Consequently, OT administration in that group may have led to excessively elevated OT levels that may have worsened rescue behavior. Unfortunately, this hypothesis could not be corroborated as endogenous OT levels were not analyzed. In the present study, rescue behavior was directed to the dogs’ owners, so dogs endogenous OT levels may have been increased due to the familiarity of the victim. In line with this, there is evidence that OT levels increase when dogs interact with their owners (Handlin et al. 2011; Hritcu et al. 2019; Nagasawa et al. 2015; Odendaal and Meintjes 2003; but see Powell et al. 2019; Marshall-Pescini et al. 2019). Therefore, endogenous OT concentrations may have interacted with the exogenous administration deteriorating rescue behavior. Given that there were no measurements of endogenous OT levels, this hypothesis, although plausible, is speculative. Further studies should compare rescue behavior of dogs towards familiar and unfamiliar people as well as measure endogenous OT levels.
The second aim of this study was to assess whether rescue behavior could be associated to the level of the dog–owner bond, which was measured with the MDORS. Results indicate that dogs who received saline and whose owners reported a stronger bond and a higher emotional closeness spent more time near the box in the stressed owner condition. This suggests that a greater bond may be associated with an increase in the dogs’ interest towards their stressed owners, which is in line with prior studies indicating that strongly bonded individuals exhibit more helping behavior (de Waal 2008; Rault 2019). In support of that hypothesis, Sanford et al. (2018) found that dogs who opened in the stressed owner condition, had a closer bond with their owners than those that did not. However, it must be noted that in our study, the facilitating effect of the dog–owner bond was observed in the time they spent in proximity to the box but not on opening behavior.
The opposite pattern was observed in dogs in the stressed owner condition who received OT, as they exhibited both a lower rate and a higher latency of openings the stronger the bond was. This decrease in rescue behavior would be in line with Yamagishi et al. (2020) and the aforementioned hypothesis of the interaction between endogenous OT levels and its exogenous administration. Although there are some contradictory results (Powell et al. 2019; Marshall-Pescini et al. 2019), prior studies associated the level of the dog–owner bond with higher endogenous OT concentrations in dogs (Handlin et al. 2012) and humans (Nagasawa et al. 2009). For instance, the total score of the MDORS was positively correlated with the average plasmatic concentration of OT, while the perceived costs subscale was negatively correlated with such concentration (Handlin et al. 2012). The authors indicated that dogs whose owners reported a higher level of bond and perceived a lower cost of the relationship were the ones with higher OT levels. In line with this, one possible explanation for the present results is that those dogs who had a more intense bond with their owners had higher endogenous OT levels than those with a less intense bond. When those dogs received the administration of exogenous OT, their OT levels may have increased even more, crossing a threshold from which rescue behavior was deteriorated. Nevertheless, further studies are needed to support this hypothesis.
On the other hand, for dogs in the calm calling owner condition that received saline a stronger bond as well as a higher emotional closeness reported by the owner were associated with a higher number of openings and a shorter latency to do so. It is important to remark, as suggested by Sanford et al. (2018), that the motivation behind the openings may differ between the stressed and the calm calling owner conditions. In this case, the opening may be related to feelings of anxiety due to the separation from the owner. Therefore, considering the subgroup of dogs in the calm calling owner condition that opened the door, this behavior may have aimed to restore social contact with the owners. In this sense, it has been observed that dogs exhibit proximity seeking behaviors when their owner is absent (Palmer and Custance 2008; Prato-Previde et al. 2003) and establish more physical contact with the owner, rather than an unfamiliar person, after a short separation (Rehn et al. 2014b; Topál et al. 1998).
In addition, Rehn et al. (2014b) reported a positive association between physical contact upon owner return and the dog–owner interaction subscale of the MDORS. This suggests that seeking to reestablish social contact with the owner may be modulated by the level of the bond. In our protocol, dogs were in the waiting room separated from their owners before starting each trial. Furthermore, during the task the reappearance of the owner was incomplete, as they could see them inside of the box but could not engage in physical contact. Both of these separations may have triggered some anxiety and thus encouraged opening behavior, which would be greater according to the level of the dog–owner bond. In support of this hypothesis, in the calm calling owner condition, dogs that received saline and had a stronger bond with their owners remained more time with their tail down, which suggests they may have been more stressed due to the separation than dogs with weaker bonds. Interestingly, OT administration appears to revert this effect, as dogs in the calm calling owner condition who received OT and had a stronger bond with their owners stayed less time with their tail down. This could be due to the anxiolytic effects of OT (Buttner 2016). However, due to the low frequency of the tail down behavior, these data should be taken with caution. In addition, owners in the calm calling condition called the dogs’ name neutrally. The higher response of those intensely bonded dogs may have been due to them being more attentive to their owner and his calls than those with more distant bonds (Sanford et al. 2018).
It is important to consider that a limitation of this work is that the level of the bond was evaluated through an owner-based questionnaire and thus the answers depended on his subjective assessment. It would be advisable for future studies to include a behavioral standardized test such as Ainsworth’s Strange Situation Test to evaluate the dog–owner bond and its influences on rescue behavior.
Another general limitation of trapped-other studies in dogs is that owners are pretending to be stressed and their stress signs are feigned, though in this case they were instructed to act as naturally as they could. These studies may not accurately reflect what would happen in a real emergency situation. However, Van Bourg et al. (2020) analyzed the level of the owners’ vocalizations with a sincerity score measured by an observer and found it did not predict opening behavior. The authors concluded that the artificial nature of the task does not prevent the assessment of dogs’ rescue behavior. Nevertheless, due to its artificiality, the experimental situation does not include all of the sensory modalities of a real situation, particularly olfactory cues. Although dogs are able to detect human emotions in absence of such cues (e.g., Albuquerque et al. 2016; Müller et al. 2015), is it possible the stimulus is less intense than it would be in a real emergency situation in which those cues are present. This may affect the number and intensity of the dogs’ responses, explaining, for instance, the relatively minor stress behaviors observed. Future studies could manipulate the stress of the owner (i.e., using psychological stressors such as difficult mathematical problems) to examine its impact on rescue behavior.
In sum, dogs released their owners when they pretended to be trapped inside of a box displaying signs of stress. Contrary to our expectations, OT administration decreased opening behavior regardless of the emotional state of the owner. In addition, the level of the dog–owner bond was differentially associated to rescue behavior according to the condition (stressed vs calm calling owner) and treatment (OT or saline) the dogs received. Therefore, although both OT concentrations and the level of the bond appear to be related to rescue behavior, the involved mechanisms are still unclear and further studies are needed. From a broad perspective, the present findings highlight the complexity of studying OT effects on dogs’ social cognition given the multiple roles of this hormone.
References
Albuquerque N, Guo K, Wilkinson A, Savalli C, Otta E, Mills D (2016) Dogs recognize dog and human emotions. Biol Lett 12(1):20150883. https://doi.org/10.1098/rsbl.2015.0883
Barrera G, Dzik V, Cavalli C, Bentosela M (2018) Effect of intranasal oxytocin administration on human-directed social behaviors in shelter and pet dogs. Front Psychol 9:2227. https://doi.org/10.3389/fpsyg.2018.02227
Bayliss AP, Bartlett J, Naughtin CK, Kritikos A (2011) A direct link between gaze perception and social attention. J Exp Psychol Hum Percept Perform 37(3):634–644. https://doi.org/10.1037/a0020559
Beerda B, Schilder MB, van Hooff JA, de Vries HW (1997) Manifestations of chronic and acute stress in dogs. Appl Anim Behav Sci 52(3–4):307–319. https://doi.org/10.1016/S0168-1591(96)01131-8
Ben-Ami Bartal I, Decety J, Mason P (2011) Empathy and pro-social behavior in rats. Science 334(6061):1427–1430. https://doi.org/10.1126/science.1210789
Ben-Ami Bartal I, Shan H, Molasky NM, Murray TM, Williams JZ, Decety J, Mason P (2016) Anxiolytic treatment impairs helping behavior in rats. Front Psychol 7:850. https://doi.org/10.3389/fpsyg.2016.00850
Bräuer J, Schönefeld K, Call J (2013) When do dogs help humans? Appl Anim Behav Sci 148:138–149. https://doi.org/10.1016/j.applanim.2013.07.009
Buttner AP (2016) Neurobiological underpinnings of dogs’ human-like social competence: How interactions between stress response systems and oxytocin mediate dogs’ social skills. Neurosci Biobehav Rev 71:198–214. https://doi.org/10.1016/j.neubiorev.2016.08.029
Carballo F, Dzik V, Freidin E, Damián JP, Casanave EB, Bentosela M (2020) Do dogs rescue their owners from a stressful situation? A behavioral and physiological assessment. Anim Cogn 23(2):389–403. https://doi.org/10.1007/s10071-019-01343-5
Coppinger R, Coppinger L (2001) Dogs: A startling new understanding of canine origin, behavior & evolution. Simon and Schuster
D’Aniello B, Semin GR, Alterisio A, Aria M, Scandurra A (2018) Interspecies transmission of emotional information via chemosignals: from humans to dogs (Canis lupus familiaris). Anim Cogn 21(1):67–78. https://doi.org/10.1007/s10071-017-1139-x
D’Aniello B, Fierro B, Scandurra A, Pinelli C, Aria M, Semin GR (2021) Sex differences in the behavioral responses of dogs exposed to human chemosignals of fear and happiness. Anim Cogn. https://doi.org/10.1007/s10071-021-01473-9
De Waal FBM (2008) Putting the altruism back into altruism: the evolution of empathy. Annu Rev Psychol 59(1):279–300. https://doi.org/10.1146/annurev.psych.59.103006.093625
Dwyer F, Bennett PC, Coleman GJ (2006) Development of the Monash dog owner relationship scale (MDORS). Anthrozoös 19(3):243–256. https://doi.org/10.2752/089279306785415592
Handlin L, Hydbring-Sandberg E, Nilsson A, Ejdebäck M, Jansson A, Uvnäs-Moberg K (2011) Short-term interaction between dogs and their owners: effects on oxytocin, cortisol, insulin and heart rate—an exploratory study. Anthrozoös 24(3):301–315. https://doi.org/10.2752/175303711X13045914865385
Handlin L, Nilsson A, Ejdebäck M, Hydbring-Sandberg E, Uvnäs-Moberg K (2012) Associations between the psychological characteristics of the human–dog relationship and oxytocin and cortisol levels. Anthrozoös 25(2):215–228. https://doi.org/10.2752/175303712X13316289505468
Hritcu LD, Horhogea C, Ciobica A, Spataru MC, Spataru C, Kis A (2019) Conceptual replication of canine serum oxytocin increase following a positive dog-human interaction. Revista de Chimie 70(5):1579–1581. https://doi.org/10.37358/rc.19.5.7172
Huber A, Barber AL, Faragó T, Müller CA, Huber L (2017) Investigating emotional contagion in dogs (Canis familiaris) to emotional sounds of humans and conspecifics. Anim Cogn 20(4):703–715
Kikusui T, Nagasawa M, Nomoto K, Kuse-Arata S, Mogi K (2019) Endocrine regulations in human-dog coexistence through domestication. Trends Endocrinol Metab 30(11):793–806. https://doi.org/10.1016/j.tem.2019.09.002
Kis A, Hernádi A, Kanizsár O, Gácsi M, Topál J (2015) Oxytocin induces positive expectations about ambivalent stimuli (cognitive bias) in dogs. Horm Behav 69:1–7. https://doi.org/10.1016/j.yhbeh.2014.12.004
Kis A, Ciobica A, Topál J (2017a) The effect of oxytocin on human-directed social behaviour in dogs (Canis familiaris). Horm Behav 94:40–52. https://doi.org/10.1016/j.yhbeh.2017.06.001
Kis A, Hernádi A, Miklósi B, Kanizsár O, Topál J (2017b) The way dogs (Canis familiaris) look at human emotional faces is modulated by oxytocin. An eye-tracking study. Front Behav Neurosci 11:210. https://doi.org/10.3389/fnbeh.2017.00210
Kis A, Tóth K, Kanizsár O, Topál J (2020) The effect of oxytocin on yawning by dogs (Canis familiaris) exposed to human yawns. Appl Anim Behav Sci 223:104916. https://doi.org/10.1016/j.applanim.2019.104916
Kovács K, Kis A, Kanizsár O, Hernádi A, Gácsi M, Topál J (2016) The effect of oxytocin on biological motion perception in dogs (Canis familiaris). Anim Cogn 19(3):513–522. https://doi.org/10.1007/s10071-015-0951-4
Lore RK, Eisenberg FB (1986) Avoidance reactions of domestic dogs to unfamiliar male and female humans in a kennel setting. Appl Anim Behav Sci 15(3):261–266. https://doi.org/10.1016/0168-1591(86)90096-1
Macchitella L, Stegagno T, Giaconella R, di Sorrentino EP, Schino G, Addessi E (2017) Oxytocin improves the ability of dogs to follow informative pointing: a neuroemotional hypothesis. Rendiconti Lincei 28(1):105–115. https://doi.org/10.1007/s12210-016-0579-6
MacDonald E, Dadds MR, Brennan JL, Williams K, Levy F, Cauchi AJ (2011) A review of safety, side-effects and subjective reactions to intranasal oxytocin in human research. Psychoneuroendocrinology 36(8):1114–1126. https://doi.org/10.1016/j.psyneuen.2011.02.015
Marshall-Pescini S, Schaebs FS, Gaugg A, Meinert A, Deschner T, Range F (2019) The role of oxytocin in the dog–owner relationship. Animals 9(10):792. https://doi.org/10.3390/ani9100792
Müller CA, Schmitt K, Barber AL, Huber L (2015) Dogs can discriminate emotional expressions of human faces. Curr Biol 25(5):601–605. https://doi.org/10.1016/j.cub.2014.12.055
Nagasawa M, Kikusui T, Onaka T, Ohta M (2009) Dog’s gaze at its owner increases owner’s urinary oxytocin during social interaction. Horm Behav 55(3):434–441. https://doi.org/10.1016/j.yhbeh.2008.12.002
Nagasawa M, Mitsui S, En S, Ohtani N, Ohta M, Sakuma Y, Kikusui T (2015) Oxytocin-gaze positive loop and the coevolution of human-dog bonds. Science 348(6232):333–336. https://doi.org/10.1126/science.1261022
Nowbahari E, Hollis KL (2010) Rescue behavior: Distinguishing between rescue, cooperation and other forms of altruistic behavior. Commun Integrative Biol 3(2):77–79. https://doi.org/10.4161/cib.3.2.10018
Odendaal JS, Meintjes RA (2003) Neurophysiological correlates of affiliative behaviour between humans and dogs. Vet J 165(3):296–301. https://doi.org/10.1016/S1090-0233(02)00237-X
Oliva JL, Rault JL, Appleton B, Lill A (2015) Oxytocin enhances the appropriate use of human social cues by the domestic dog (Canis familiaris) in an object choice task. Anim Cogn 18(3):767–775. https://doi.org/10.1007/s10071-015-0843-7
Oliva JL, Mengoli M, Mendonça T, Cozzi A, Pageat P, Chabaud C, Bienboire-Frosini C (2019) Working smarter not harder: oxytocin increases domestic dogs’ (Canis familiaris) accuracy, but not attempts, on an object choice task. Front Psychol. https://doi.org/10.3389/fpsyg.2019.02141
Onaka T, Takayanagi Y (2019) Role of oxytocin in the control of stress and food intake. J Neuroendocrinol 31(3):e12700. https://doi.org/10.1111/jne.12700
Palmer R, Custance D (2008) A counterbalanced version of Ainsworth’s Strange Situation Procedure reveals secure-base effects in dog–human relationships. Appl Anim Behav Sci 109(2–4):306–319. https://doi.org/10.1016/j.applanim.2007.04.002
Piotti P, Kaminski J (2016) Do dogs provide information helpfully? PLoS ONE 11(8):e0159797. https://doi.org/10.1371/journal.pone.0159797
Powell L, Edwards KM, Bauman A, Guastella AJ, Drayton B, Stamatakis E, McGreevy P (2019) Canine endogenous oxytocin responses to dog-walking and affiliative human-dog interactions. Animals 9(2):51. https://doi.org/10.3390/ani9020051
Prato-Previde E, Spiezio C, Sabatini F, Custance DM (2003) Is the dog-human relationship an attachment bond? An observational study using Ainsworth’s strange situation. Behaviour 140(2):225–254. https://doi.org/10.1163/156853903321671514
Quintana DS, Alvares GA, Hickie IB, Guastella AJ (2015) Do delivery routes of intranasally administered oxytocin account for observed effects on social cognition and behavior? A two-level model. Neurosci Biobehav Rev 49:182–192. https://doi.org/10.1016/j.neubiorev.2014.12.011
Rault JL (2019) Be kind to others: Prosocial behaviours and their implications for animal welfare. Appl Anim Behav Sci 210:113–123. https://doi.org/10.1016/j.applanim.2018.10.015
Rehn T, Lindholm U, Keeling L, Forkman B (2014) I like my dog, does my dog like me? Appl Anim Behav Sci 150:65–73. https://doi.org/10.1016/j.applanim.2013.10.008
Rehn T, Handlin L, Uvnäs-Moberg K, Keeling LJ (2014) Dogs’ endocrine and behavioural responses at reunion are affected by how the human initiates contact. Physiol Behav 124:45–53. https://doi.org/10.1016/j.physbeh.2013.10.009
Robinson KJ, Bosch OJ, Levkowitz G, Busch KE, Jarman AP, Ludwig M (2019) Social creatures: Model animal systems for studying the neuroendocrine mechanisms of social behaviour. J Neuroendocrinol. https://doi.org/10.1111/jne.12807
Romero T, Konno A, Hasegawa T (2013) Familiarity bias and physiological responses in contagious yawning by dogs support link to empathy. PLoS One 8(8):e71365. https://doi.org/10.1371/journal.pone.0071365
Romero T, Nagasawa M, Mogi K, Hasegawa T, Kikusui T (2014) Oxytocin promotes social bonding in dogs. Proc Natl Acad Sci 111(25):9085–9090. https://doi.org/10.1073/pnas.1322868111
Sanford EM, Burt ER, Meyers-Manor JE (2018) Timmy’s in the well: Empathy and prosocial helping in dogs. Learn Behav 46(4):374–386. https://doi.org/10.3758/s13420-018-0332-3
Scandurra A, Alterisio A, Di Cosmo A, D’Aniello B (2018) Behavioral and perceptual differences between sexes in dogs: an overview. Animals 8(9):151. https://doi.org/10.3390/ani8090151
Serpell J (2017) The domestic dog. Cambridge University Press, Cambridge
Somppi S, Törnqvist H, Topál J, Koskela A, Hänninen L, Krause CM, Vainio O (2017) Nasal oxytocin treatment biases dogs’ visual attention and emotional response toward positive human facial expressions. Front Psychol 8:1854. https://doi.org/10.3389/fpsyg.2017.01854
Svartberg K (2002) Shyness–boldness predicts performance in working dogs. Appl Anim Behav Sci 79(2):157–174. https://doi.org/10.1016/S0168-1591(02)00120-Xd
Topál J, Miklósi Á, Csányi V, Dóka A (1998) Attachment behavior in dogs (Canis familiaris): a new application of Ainsworth’s (1969) strange situation test. J Compar Psychol 112(3):219. https://doi.org/10.1037/0735-7036.112.3.219
Van Bourg J, Patterson JE, Wynne CDL (2020) Pet dogs (Canis lupus familiaris) release their trapped and distressed owners: Individual variation and evidence of emotional contagion. PLoS One 15(4):e0231742. https://doi.org/10.1371/journal.pone.0231742
Yamagishi A, Okada M, Masuda M, Sato N (2020) Oxytocin administration modulates rats’ helping behavior depending on social context. Neurosci Res 153:56–61. https://doi.org/10.1016/j.neures.2019.04.001
Yong MH, Ruffman T (2014) Emotional contagion: Dogs and humans show a similar physiological response to human infant crying. Behav Proc 108:155–165. https://doi.org/10.1016/j.beproc.2014.10.006
Acknowledgements
We would like to express our special gratitude to all the owners who kindly participated in this experiment. We also thank Gabriel Granada who helped us finding the facilities to run the tests. Finally, we appreciate the valuable collaboration of Camila Cavalli and Cynthia Roxana Romero.
Funding
This work was supported by AGENCIA (PICT 2014 Nº 0883), CONICET (PIP 2013 Nº 11220130100182) and Universidad Nacional del Sur: PGI 24/B243.
Author information
Authors and Affiliations
Contributions
M.V. Dzik and F. Carballo: conceptualization, methodology, formal analysis, investigation, and writing—review and editing. E. Casanave: conceptualization and writing—review and editing. M. Bentosela: conceptualization, methodology, investigation, formal analysis, writing—review and editing, and supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dzik, M.V., Carballo, F., Casanave, E. et al. Effects of oxytocin administration and the dog–owner bond on dogs’ rescue behavior. Anim Cogn 24, 1191–1204 (2021). https://doi.org/10.1007/s10071-021-01512-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-021-01512-5