Abstract
Introduction
Rheumatologists practising in the UK National Health Service (NHS) are likely to treat migrant patients from sub-Saharan Africa. This study aimed to conduct a literature review about rheumatoid arthritis prevalence in Africa and understand the experiences of patients with rheumatological conditions, about their past healthcare in sub-Saharan Africa and their transition of care to the United Kingdom (UK).
Methods
A systematic search and a pilot study using semi-structured interviews to gain the views of migrants from sub-Saharan Africa with rheumatological conditions was conducted.
Results
Thirty-two studies reported on the prevalence of rheumatoid arthritis in Africa. Studies were small and out-of-date, and there was significant heterogeneity in prevalence rates. For the qualitative study, seven participants were recruited. Four themes were highlighted: (i) the physical and emotional impact of rheumatological conditions on participants; (ii) limited rheumatology care in sub-Saharan Africa with high costs, limited access to specialists, lack of investigations and treatments, the use of traditional medicines and poor communication by clinicians; (iii) barriers to rheumatology care in the UK such as migrants’ poor understanding of rheumatological conditions and NHS entitlements; (iv) and ways to improve access to care such as patient, public and general practitioner education.
Conclusion
This study has highlighted the paucity of rheumatoid arthritis prevalence data in Africa and described, for the first time, the migrant’s perspective of rheumatology care in sub-Saharan Africa and the transition of care to the UK. This description begins to allow healthcare providers in the UK to tailor management for this migrant population.
Key Points • Rheumatological conditions are common in Africa, but there is a paucity of epidemiological data regarding the prevalence of specific conditions such as rheumatoid arthritis. • UK clinicians need to be mindful when treating migrants that access to rheumatologists and specialist investigations and treatment is limited in sub-Saharan Africa and that there is often limited public and patient understanding of rheumatological conditions. • Migrants continue to lack understanding of their NHS entitlements and fear data sharing with immigration services which can be a barrier to seeking care. • This study has exposed the lack of understanding about rheumatological conditions by the public and some general practitioners which needs to be addressed with effective education and awareness campaigns. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatological conditions are a leading cause of morbidity and disability worldwide [1]. With over 150 rheumatological diseases they present with a broad symptomatic spectrum including non-inflammatory musculoskeletal disorders and systemic inflammatory conditions such as rheumatoid arthritis, ankylosing spondylitis and systemic lupus erythematosus. Sub-Saharan Africa (SSA) comprises all countries which lie south of the Sahara Desert. Rheumatological diseases are common in SSA, and as a result, the burden of musculoskeletal conditions is 2.5 times higher than in Europe [1]. Non-inflammatory musculoskeletal diagnoses rates are high, with osteoarthritis affecting up to 82.7% of over 65 s in South Africa [2] and an average 62% lifetime prevalence of lower back pain in Africa [3]. However, a paucity of data means the prevalence of inflammatory arthritis such as rheumatoid arthritis (RA) is less clear [4].
Healthcare for rheumatological conditions is limited in SSA. Diagnosing rheumatological diseases can be difficult and relies mostly on clinical expertise. This is problematic as less than 150 rheumatologists currently serve 1 billion people in SSA [5] and most countries have no formal rheumatology training program [6]. Treatment such as immune-modulating biologic therapy is extremely expensive. Many countries in SSA are low and middle-income with gross domestic products (GDPs) ranging from US$936 per capita in Tanzania to US$6951 in South Africa, compared with US$39,720 in the United Kingdom (UK) [7]. Some treatments come with significant risks, side effects and blood test monitoring requirements which are not always practical in a lower resource setting [8]. As a result of poor access to appropriate healthcare, patients with rheumatological symptoms are often diagnosed late, undertreated and develop high levels of disability and comorbidities [9].
There are currently 1.2 million people in the UK who have migrated from SSA [10]. In the UK, healthcare is provided by the publicly funded “National Health Service” (NHS). The literature suggests there are significant barriers to these migrants receiving healthcare in the UK [11], including language, culture and faith differences, lack of awareness, lack of advocacy and fear of cost and eligibility of healthcare services.
Rheumatologists practising in the UK National Health Service (NHS) are likely to be exposed to these patients. This study aims to gain a better understanding of the provision of rheumatology healthcare in SSA and the transition of this care to the UK in order to help UK healthcare providers to tailor management to the migrant patient. A literature review about RA prevalence in Africa and a pilot study using semi-structured interviews to gain the views of migrants from SSA with rheumatological conditions was conducted.
Methods
Literature review of rheumatoid arthritis prevalence in Africa
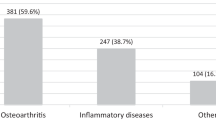
There is substantial evidence that rates of non-inflammatory musculoskeletal diagnoses such as osteoarthritis are high in Africa [2]. There is less data available about the prevalence of systemic inflammatory rheumatological conditions. As the most common systemic inflammatory rheumatological condition, RA was chosen as the subject of the literature review to provide an insight into the prevalence of inflammatory arthritis in Africa.
The limited data available suggests that the prevalence of RA in Africa is higher than previously thought [4, 12]. In 2012, Dowman et al. published a meta-analysis of 21 studies which estimated RA prevalence in Africa as 0.42%, affecting 4.3 million people [12]. There were, however, substantial limitations to the meta-analysis because the studies were small, out-of-date (most from the 1970s and 1980s) and concentrated in South Africa, Nigeria and Uganda. One further systematic review was published in 2015 but the prevalence rates were so heterogenous they could not be included in meta-analyses [4]. For example, RA prevalence per country ranged from 0.6 (95% CI 0.4–0.8) in the Democratic Republic of Congo (DRC) to 2.54% (95% CI − 0.43–5.52) in South Africa [4].
Lack of good quality epidemiological data on rheumatological conditions in Africa has a range of reasons [13]. Firstly, research in this area has been grossly under-resourced. As a result, any studies have been small and often poorly planned with problems in study design and the use of different classification criteria preventing direct comparisons [2, 14]. Secondly, the clinical care settings in which research is undertaken are often disparate so comparison is difficult. For example, some hospitals in South Africa are resourced with specialist-trained clinicians who have access to tests and treatments, whilst many patients in remote areas of other African countries will have no access to even general physicians or basic blood tests [15]. Finally, Africa covers a large geographical area and includes patients from a wide range of cultural, ethnic and financial backgrounds, making results difficult to generalise.
In an attempt to standardise epidemiological methodology, the World Health Organisation (WHO) and the International League of Associations for Rheumatology (ILAR) instigated a Community Oriented Programme for Control of Rheumatic Disease (COPCORD) in 1981 [16, 17]. Although this methodology has been adopted in other low- and middle-income countries, only a few centres in Africa have been able to instigate COPCORD [18,19,20].
Given the poor understanding about the prevalence of RA in Africa and the significant time since the last thorough review, a systematic search was performed on 31/07/2019. Search criteria, methodology and PRISMA flow chart showing the search results are included in Appendix 1.
Qualitative pilot study using semi-structured interviews to gain the views of migrants from sub-Saharan Africa with long-term rheumatological conditions
Full ethical approval was obtained from North West-Liverpool East Research Ethics Committee on 24/05/2019 (REC Reference: 19/NW/0271, HRA approval 29/05/2019).
Participant recruitment
Over an 8-month period, potential participants were identified by the direct care team, comprising clinicians from two rheumatology outpatient departments in London. Participants were included if they were (i) aged 18 or older, (ii) migrant from SSA in the last 10 years, (iii) had a confirmed rheumatological condition by a rheumatologist in the UK (including RA, psoriatic arthritis, ankylosing spondylitis, systemic lupus erythematosus, osteoarthritis) and(iv) their symptoms started in their country of origin. The exclusion criteria were as follows: (i) unable to communicate in English or (ii) experienced a flare of their rheumatological condition around the time of the interview. When patients expressed an interest to take part in the study, they received a Participant Information Sheet (PIS) with a copy of the consent form. At least 24 h following receipt of the PIS, potential interviewees were contacted by telephone by the first author (Z RL) to arrange an appropriate time and place to conduct the interviews.
Semi-structured interviews
Initially, a draft interview topic guide, based on a literature review [5, 21,22,23,24,25] and including informal discussions with patients was trialled with one patient who confirmed the questions were relevant and appropriate, and the length of the interview was reasonable. The data from this first interview was included in the main study.
The interviews took place in the respective rheumatology outpatient clinics and over the telephone. Following fully informed, written consent each participant provided socio-demographic information. Then the audio-recorded 1:1 interviews were conducted (Appendix 2: Interview topic guide) and took between 25 and 55 min.
Data review and analysis
Thematic inductive analysis was applied to the transcribed interviews (Table 1). The data is not guided by any pre-existing theoretical framework, providing rich and detailed accounts that generate unanticipated insights [26].
Results
Literature search
The results of the literature search are presented in Table 2.
Qualitative study
Recruitment was terminated after 8 months. Thirty-three patients were approached, and 26 did not meet the inclusion and exclusion criteria. No patients declined to take part in the study. Three were interviewed in the clinic, and four by telephone. Two participants were recruited from clinic 1, and five from clinic 2.
Demographics
Participant demographics are shown in Table 3.
Themes
Thematic analysis identified four main themes each with a number of sub-themes, shown in Table 4.
The number (n=) of participants who reported a particular subtheme is included in the text, called single counting [27]. Accounts from the interview transcripts are included in italic to illustrate and provide a direct explanation of the subtheme (Appendix 3). Under each account, the allocated number for each patient and gender is shown.
Theme 1: Impact of the condition on the participant
Physical impact
Pain was described using emotive language like ‘suffer’, ‘unbearable’ and ‘horrendous’ (n = 6) (Appendix 3, Table 1, Account 1 and 2). This rheumatic pain led to functional disability (n = 5) (Appendix 3, Table 1, Account 3 and 4) and dependence on others (n = 2) (Appendix 3, Table 1, Account 5).
Emotional impact
The accounts revealed a detailed emotional impact of their condition (n = 6). Four experienced low mood (Appendix 3, Table 1, Account 6). Lack of understanding of their condition by doctors and the public led to frustration and anger (n = 2) (Appendix 3, Table 1, Account 7) and embarrassment (n = 3). As a coping mechanism one changed her physical appearance in order to ‘re-define’ her self-identity (Appendix 3, Table 1, Account 8). Limited psychological and social support was reported in their country of origin (Appendix 3, Table 1, Account 9 and 10).
Financial impact
The need to seek private care had a negative effect on their finances (n = 4) (Appendix 3, Table 1, Account 11) and resulted in inability to access medications (n = 1) (Appendix 3, Table 1, Account 12) and the need to move the country (n = 3) (Appendix 3, Table 1, Account 14).
Social impact
A majority of interviewees (n = 6) described how their families had been supportive (Appendix 3, Table 1, Account14) and took on the ‘primary carer role’ (n = 4). Participants felt frustration at being a burden to their families(n = 2) (Appendix 3, Table 1, Account15). Their condition resulted in social isolation (n = 1) (Appendix 3, Table 1, Account 16) and breakdown of a marriage (n = 1) (Appendix 3, Table 1, Account17).
Theme 2: Limited rheumatology care in sub-Saharan Africa compared with that in the UK
Healthcare service provision structure
Healthcare was either paid for at point of care (n = 5) or a medical aid insurance system was in place (n = 2) in interviewees’ country of origin. The private system had benefits with low waiting times for a doctor’s review (n = 4) (Appendix 3, Table 2, Account 1 and 2) and investigations (n = 4) (Appendix 3, Table 2, Account 3).
Difficulty to access specialists was highlighted by the interviewees (n = 6), either because there were no rheumatologists (n = 4) (Appendix 3, Table 2, Account 4 and 5) or because it was difficult to access those in the country (n = 2) (Appendix 3, Table 2, Account 6).
Specialist medications were often unavailable in participants’ country of origin (n = 3) (Appendix 3, Table 2, Account 7 and 8), and one interviewee was told a specialist immunological test she needed was unattainable.
The lack of appropriate medication resulted in one patient receiving long term steroids and developing severe osteoporosis (Appendix 3, Table 2, Account 9).
Traditional medicines
Traditional medicine emerged as an important aspect of participants’ beliefs and care (n = 5). One estimated that over 90% of people use traditional medicine, even if they also seek conventional medical attention. The majority took herbal remedies (n = 4) made up by family members (n = 2) or bought from traditional medicine sellers (n = 2).
People took these medications due to belief in their efficacy (n = 4) (Appendix 3, Table 2, Account 10 and 11), low cost (n = 2) (Appendix 3, Table 2, Account 12), easy accessibility (n = 1) (Appendix 3, Table 2, Account 13) and in desperation due to their suffering, lack of understanding of their condition and inability to find effective treatments (n = 2) (Appendix 3, Table 2, Account 14).
Understanding by doctors
All those who saw a rheumatologist in their country of origin (n = 3) felt they were proficient (Appendix 3, Table 2, Account 15 and 16).
However, limited rheumatological knowledge by general physicians was identified (n = 6), due to limited understanding that there are different types of arthritis (n = 4) (Appendix 3, Table 2, Account 17 and 18). Participants reported this limited knowledge was because doctors had no experience with rheumatological cases and typically managed acute infectious diseases (Appendix 3, Table 2, Account 19, 20, 21 and 22).
This meant participants often sought advice from multiple doctors and care was fragmented (n = 4) (Appendix 3, Table 2, Account 23) leading to confusion and frustration (n = 2) (Appendix 3, Table 2, Account 24).
In the UK, rheumatologists were observed to have good rheumatological knowledge (n = 7) (Appendix 3, Table 2, Account 25 and 26), but general practitioners (GP) did not (n = 2) (Appendix 3, Table 2, Account 27).
Doctor–patient communication
Although the importance of patient education was highlighted (n = 3) (Appendix 3, Table 2, Account 28), it seemed there was limited information in their country of origin (n = 3) (Appendix 3, Table 2, Account 29).
In contrast, in the UK, most reported they had received good explanations (n = 5) (Appendix 3, Table 2, Account 30 and 31) and commented on UK doctors’ kindness (n = 4), using words like ‘compassion’, ‘sincerity’, ‘openness’, ‘commitment’, ‘interest’, ‘concern’ and ‘empathy (Appendix 3, Table 2, Account 32 and 33).
Theme 3: Barriers to accessing rheumatology care in the UK
Public understanding of rheumatological conditions
There was a stated lack of understanding by the public about different types of arthritis (n = 4) (Appendix 3, Table 3, Account 1 and 2) and by the patients of what RA is (n = 3) (Appendix 3, Table 3, Account 3). Participants explained that this lack of understanding may in part be due to their own limited education and the fact that rheumatological conditions, such as RA, are relatively rare (n = 3).
Processes to access NHS care
Interviewees mentioned they did not understand how the NHS referral processes work (n = 3) (Appendix 3, Table 3, Account 4), and what NHS care they were entitled to (n = 2). Furthermore, some NHS workers also seemed unclear what care migrants are entitled to (n = 2) (Appendix 3, Table 3, Account 5 and 6). Two interviewees reported fear of data sharing with immigration services (Appendix 3, Table 3, Account 7).
Theme 4: Ways to improve the transition of care for migrants from sub-Saharan Africa
Early referral and investigation of symptoms
Two participants suggested that care could be improved by expediting investigations and reported a long wait for their investigations, which led to increased anxiety (Appendix 3, Table 4, Account 1). They described that GP referral to specialist rheumatology services was too slow occasionally (n = 3) (Appendix 3, Table 4, Account 2 and 3) or the NHS waiting time was too long (n = 2) (Appendix 3, Table 4, Account 4).
Education
Participants emphasised that education both to patients and the public was extremely important and suggested this as a way to improve care (n = 4) (Appendix 3, Table 4, Account 5 and 6). Available educational materials in the UK were easy to access and clear (n = 2) (Appendix 3, Table 4, Account 7). One participant who had attended support groups found the advice provided helpful. Three explained that they could not relate culturally to other patients (n = 2) (Appendix 3, Table 4, Account 8 and 9) or due to a ‘negative’ atmosphere (n = 1) (Appendix 3, Table 4, Account 10).
Discussion
The literature review has highlighted the paucity of good quality epidemiological data reporting on RA prevalence in Africa. Thirty-two studies were identified which were small and out of date. Although there was significant heterogeneity in prevalence rates, they were similar to the UK [51]. The most recent meta-analysis estimated that RA affects 0.42% of the African population [5]. If we assume that RA affects 0.42% of the 1.2 million migrants from SSA living in the UK [11], we can estimate that about 5000 migrants from SSA in the UK suffer from RA. It is therefore important for UK health providers to have an insight into rheumatology care in Africa and understand barriers to accessing care in the UK. The qualitative study has described, for the first time, the significant impact rheumatological conditions have on these patients in their country of origin, the limited care they receive in SSA and some of the barriers they face when accessing NHS care.
The significant physical and psychological impact of rheumatological conditions identified on this study is in keeping with the literature. Clinical depression has been diagnosed in up to 39% of patients with RA [52], and the participants were often living with high disease activity which exacerbates low mood [53]. The emotional impact of RA is particularly relevant in SSA where access to social and psychological support is limited [54]. Consequently, when treating migrants from SSA, NHS rheumatologists need to be particularly aware of the psychological impact these long-term rheumatological conditions have and provide appropriate support, as patient support groups were not felt to be helpful for a majority of interviewees.
In most African countries healthcare is paid for at point of use so affordability is likely to be an issue for a substantial proportion of the population. Given that there are less than 150 rheumatologists in SSA [13] (Fig. 1), and most work in major cities, it is unsurprising that participants in this study were unable to consult with specialists reliably in their country of origin. General physicians were felt to have limited understanding about the different types of arthritis and lacked experience in managing chronic rheumatological conditions which was a primary driving factor for interviewees to seek care from other countries. This lack of understanding is likely to be due to limited rheumatology training. In a recent survey of 69 primary care physicians in Kenya, 67% had received less than 2 weeks of rheumatology training [6]; and in Nigeria in 2017, only 10 of 30 medical schools included rheumatology on their curriculum [58]. Specific tests and medications were not available to some interviewees because medications for rheumatic disease are not on the national ‘formulary’ of most sub-Saharan African countries [58]. Biologic treatment is expensive and adequate monitoring is not possible as patients have to travel long distances for routine monitoring of blood tests [25]. As a result, biologic therapy use is low and out of 60 RA patients in a rheumatology unit in Kenya none had been initiated on biologic therapy and only 12% were in disease remission [62]. These findings suggest that the majority of patients are undertreated [63] and is indicative of treatment throughout SSA [5, 14, 15, 64,65,66] where patients with RA live with high levels of disability and comorbidities [9].
Number of rheumatologists per 100,000 people in certain sub-Saharan African countries. This figure shows that the number of rheumatologists per 100,000 people in all countries is grossly below the WHO recommended ratio of one rheumatologist per 100,000 people. Most rheumatologists are in South Africa where there are 85 rheumatologists, providing paediatric and adult services for nearly 56 million people [8]. Zambia, with a population of 13 million people, has two rheumatologists working (part-time), one a paediatrician, and the other a physician [55]. Namibia has 1 rheumatologist for 2.5 million people [56]. In West Africa, Senegal has 4 rheumatologists serving 16 million people [57], Cameroon has 13 rheumatologists serving 23 million people [58] and Nigeria has 30 rheumatologists but they serve 170 million people [58]. In East Africa, Sudan has 4 rheumatologists [5], all working in the capital city and serving more than 40 million people (10). Uganda has 1 rheumatologist for 43 million people [59]. In Ethiopia two rheumatologists are currently in training to provide services for 105 million people [60]. In Kenya there are 6 rheumatologists as a result of an international training drive but they still serve 50 million people [61].
The importance of traditional medicines was highlighted. In many countries in SSA, illness is believed to be natural, cultural or social in origin and so treatment is aimed at not just the physical aspects but also spiritual, moral and social aspects of being well [67]. Traditional medicine use was driven in part by lack of understanding about their rheumatological condition and inability to find effective conventional treatments. Such remedies are also both cheaper and more easily accessible than conventional medicine. As an example, the ratio of traditional healers to the population is 1: 500 compared with that of 1:40000 medical doctors in SSA [68]. The literature suggests migrants continue to use traditional medicines when they have left their country of origin [69]. Many patients do not disclose their traditional medicine use to healthcare professionals [67], so UK clinicians are advised to directly ask migrants during consultations about traditional medicine use because belief in the efficacy of alternative therapies can delay patients seeking care and affect medication compliance.
Migrants access to healthcare relies on their knowledge about their entitlements to care and trust in the healthcare system [70]. In the UK, migrants are eligible for free emergency treatment but are required to pay for non-urgent specialist care [71]. The interviewees in this study did not report any financial burden during the interviews. In contrast the literature suggests that financial constraints limit healthcare access by many migrants [72]. Lack of understanding about entitlements both by the public and NHS staff was identified as a barrier to care. Many migrants do not realise that primary care is free and so delay seeking advice [72]. When migrants are aware of their eligibility, they can be asked by NHS staff to produce documents which are not required [70]. Previous studies have identified confusion about entitlements, language barriers and financial fears that lead to delays in treatment and create a sense of helplessness [11, 72, 73]. Sharing of non-clinical data by NHS England with immigration authorities was implemented as a tool to identify those breaching immigration law but is believed to create fear among migrants accessing NHS care [70]. This study strengthens the case that more needs to be done to provide relevant advice to migrants about their entitlements and specifically what information will be shared with immigration services.
Participants stated that some GPs in the UK lacked clinical rheumatology expertise and investigations took a long time, resulting in delayed diagnosis and treatment. This has been highlighted in The British Society of Rheumatology National Early Inflammatory Arthritis Audit (BSR NEIAA) report, that primary care referral is frequently delayed, with only 41% meeting the target to refer possible inflammatory arthritis within 3 days [74]. The recommendations suggest the promotion of GP education that may help to improve access to rheumatology services for all patients, including migrants [74].
Education was identified as a potential solution to improve accessibility to care in the UK. Participants explained they had little understanding about their condition prior to arrival in the UK and that high quality and detailed information was positive for their psychological wellbeing. Patient education is accepted as a vital aspect of rheumatology care in the UK [74], and this study highlights the importance of ensuring migrants have access to up-to-date information even if they have been living with the condition for a long time.
Study limitations
Most participants were from Cameroon or South Africa and all had postgraduate degrees and had the funds to move to the UK, so they may not represent fully the migrant population. Migrants who are unable to speak English and those who do not access healthcare were not included. This pilot interview study may be replicated with non-English-speaking migrants in other settings, such as in the community and may result in the identification of new themes.
Conclusion
This study has highlighted the paucity of good quality epidemiological data on RA prevalence in Africa and has described, for the first time, the patient’s perspective of rheumatology health care in SSA and the transition of care to the UK. These findings contribute to the understanding of rheumatology care in Africa and begin to allow healthcare providers in the UK to tailor management for this migrant population. A larger main study across different centres would be welcomed in order to extend the knowledge obtained by this pilot study.
References
Ade A, Gabriel SE (2010) Addressing musculoskeletal health inequity in Africa. Arthritis Care Res 62(4):439–441
Meyers OL, Jessop S, Klemp P (1982) The epidemiology of rheumatic disease in a rural and an urban population over the age of 65 years. S Afr Med J 62(12):403–405
Louw QA, Morris LD, Grimmer-Somers K (2007) The Prevalence of low back pain in Africa: a systematic review. BMC Musculoskelet Disord 8(105)
Usenbo A, Kramer V, Young T, Musekiwa A (2015) Prevalence of arthritis in Africa: a systematic review and meta-analysis. PLoS One 10(8):e0133858
Elshafie AI, Elkhalifa AD, Elbagir S, Aledrissy MIE, Elagib EM, Nur MAM, Weitoft T, Rönnelid J (2016) Active rheumatoid arthritis in central Africa: a comparative study between Sudan and Sweden. J Rheumatol 43(10):1777–1786
Colmegna I, Bartlett SJ, Oyoo OG (2011) The ILAR-East Africa initiative: current needs and progress in the globalization of rheumatology. Clin Rheumatol 30(2):251–253
World Bank Group, “GDP per capita (current US$) | Data,” 2019. [Online]. Available: https://data.worldbank.org/indicator/NY.GDP.PCAP.CD. Accessed: 27-Jan-2019
Mody GM (2017) Rheumatology in Africa-challenges and opportunities. Arthritis Res Ther 19(1):49
Botha-Scheepers S, Mohammed AGA, Gcelu A, Hodkinson B (2018) AB0400 high prevalence of comorbidities in patients with rheumatoid arthritis in South Africa. Ann Rheum Dis 77(2):1366.1–1366
Office for National Statistics. Population of the UK by country of birth and nationality - Office for National Statistics, July 2017–June 2018, 2018. [Online]. Available: https://www.ons.gov.uk/peoplepopulationandcommunity/populationandmigration/internationalmigration/datasets/populationoftheunitedkingdombycountryofbirthandnationality. Accessed: 29-Jan-2019
Brandenberger J, Tylleskär T, Sontag K, Peterhans B, Ritz N (2019) A systematic literature review of reported challenges in health care delivery to migrants and refugees in high-income countries - the 3C model. BMC Public Health (19, 1):755
Dowman B, Campbell RM, Zgaga L, Adeloye D, Chan KY (2012) Estimating the burden of rheumatoid arthritis in Africa: a systematic analysis. J Glob Health 2(2):020406
Tikly M, McGill P (2016) Epidemiology: the challenge of practicing rheumatology in Africa. Nat Rev Rheumatol 12(11):630–631
Njobvu PD, Trollip S, Chipeta J, Bucala R (2013) Clinical and diagnostic features of Rheumatoid Arthritis (RA) in Zambians. Clin Rheumatol 32(2 SUPPL. 1):S127–S128
Solomon A, Christian BF, Dessein PH, Stanwix AE (2005) The need for tighter rheumatoid arthritis control in a South African public health care center. Semin Arthritis Rheum 35(2):122–131
Wigley R (2012) Copcord 30 years progress in the distribution and control of rheumatic disease (RD). Intern Med J 42(SUPPL.2):22
Darmawan J (2007) Recommendations from the community oriented program for control of rheumatic disease for data collection for the measurement and monitoring of health in developing countries. Clin Rheumatol 26(6):853–857
Ekwom PE, Oyoo GO, Ongore D (2013) Prevalence of musculoskeletal pain in Nairobi, Kenya: results of aphase 1, Stage 1 COPCORD study. Clin Rheumatol 32(2 SUPPL. 1):S121
Courage UU, Stephen DP, Lucius IC, Ani C, Oche AO, Emmanuel AI, Olufemi AO (2017) Prevalence of musculoskeletal diseases in a semi-urban Nigerian community: results of a cross-sectional survey using COPCORD methodology. Clin Rheumatol 36(11):2509–2516
Wigley R (2013) COPCORD 30 years progress. Int J Rheum Dis 16(SUPPL. 1):109–110
Ochieng BMN (2013) Black African migrants: the barriers with accessing and utilizing health promotion services in the UK. Eur J Pub Health 23(2):265–269
Shangase P, Egbe CO (2014) Barriers to accessing HIV Services for Black African Communities in Cambridgeshire, the United Kingdom. J Community Health 40(1):20–26
Scuccimarri R et al (2016) Barriers encountered in the diagnosis of pediatric rheumatic diseases in Kenya: a focus group study. J Rheumatol 43(6):1162
Kumar B (2017) Global health inequities in rheumatology. Rheumatology 56(1):4–5
Kalla AA, Tikly M (2003) Rheumatoid arthritis in the developing world. Best Pract Res Clin Rheumatol 17(5):863–875
Braun V, Clarke V (2006) Using thematic analysis in psychology. Qual Res Psychol 3(2):77–101
Seale C (1999) The quality of qualitative research: generalizing from qualitative research. Qual Qual Res:106–119
Shaper AG, Shaper L (1958) Analysis of medical admissions to Mulago Hospital, 1957. East Afr Med J 35(12):647–678
Greenwood BM (1968) Autoimmune disease and parasitic infections in Nigerians. Lancet 292(7564):380–382
Greenwood BM (1969) Polyarthritis in Western Nigeria. I. Rheumatoid arthritis. Ann Rheum Dis 28(5):488–496
Anderson IF (1970) Rheumatoid arthritis in the bantu. S Afr Med J 44(43):1227–1229
Muller AS, Valkenburg HA, Greenwood BM (1972) Rheumatoid arthritis in three west African populations. East Afr Med J 49(2):75–83
Beighton P, Solomon L, Valkenburgt HA (1975) Rheumatoid arthritis in a rural South African Negro population. Ann Rheum Dis 34:136–141
Solomon L, Robin G, Valkenburg HA (1975) Rheumatoid arthritis in an urban South African Negro population. Ann Rheum Dis 34(2):128–135
Moolenburgh JD, Moore S, Valkenburg HA, Erasmus MG (1984) Rheumatoid arthritis in Lesotho. Ann Rheum Dis 43(1):40–43
Moolenburgh JD, Valkenburg HA, Fourie PB (1986) A population study on rheumatoid arthritis in Lesotho, southern Africa. Ann Rheum Dis 45(8):691–695
Brighton SW, de la Harpe AL, van Staden DJ, Badenhorst JH, Myers OL (1988) The prevalence of rheumatoid arthritis in a rural African population. J Rheumatol 15(3):405–408
Bwanahali K, Mbuyi M, Kapita B (1991) Osteoarthrosis, gout and arthritis rheumatoid in internal medicine in Kinshasa. Rev Rhum Mal Osteoartic 58(2):105–111
Silman AJ et al (1993) Absence of rheumatoid-arthritis in a rural Nigerian population. J Rheumatol 20(4):618–622
Bileckot R, Mouaya A, Bileckot R, Mouaya A, Makuwa M (1998) Prevalence and clinical presentations of arthritis in HIV-positive patients seen at a rheumatology department in Congo-Brazzaville. Rev Rhum (Engl Ed) 65(10):549–554
Abdel-Nasser SA, Abdel-Tawab R, Mahmoud J (2009) The prevalence of rheumatoid arthritis in rural Egypt: a WHO-ILAR-COPCORD study. Rheumatology 48(SUPPL. 1)
Singwe-Ngandeu M, Meli J, Ntsiba H, Nouédoui C, Yollo AV, Sida MB, Muna WF (2007) Rheumatic diseases in patients attending a clinic at a referral hospital in Yaounde, Cameroon. East Afr Med J 84(9):404–409
Malemba JJ, Mbuyi-Muamba JM, Mukaya J, Bossuyt X, Verschueren P, Westhovens R (2012) The epidemiology of rheumatoid arthritis in Kinshasa, Democratic Republic of Congo—a population-based study. Rheumatology (United Kingdom) 51(9):1644–1647
Halland AM, Louw IC, Jelsma J (2012) The prevalence and impact of musculoskeletal complaints at primary health care facilities in Cape Town: a COPCORD study. Int J Rheum Dis 15(SUPPL. 1):136
Slimani S, Ladjouze-Rezig A (2014) Prevalence of rheumatoid arthritis in an urban population of Algeria: a prospective study. Rheumatology (United Kingdom) 53(3):571–573
Ouedraogo D-D et al (2014) Clinical spectrum of rheumatologic diseases in a department of rheumatology in Ouagadougou (Burkina Faso). Clin Rheumatol 33(3):385–389
Nzambi DJP, Mbaya LA, Mulenge, Mpemble E, Malemba JJ, Mukaya TJL (2013) Prevalence of musculoskeletal disorders in a rural area of the Democratic Republic of Congo (DRC). Clin Rheumatol 32(2 SUPPL. 1):S149
Dahou-Makhloufi C et al (2018) Low rheumatoid arthritis prevalence in Algeria: A mediterranean singularity? Clin Exp Rheumatol 36(Supplement 109):S83–S84
Olivier N, Burger J, Joubert R, Lubbe M, Naudé A, Cockeran M (2018) Chronic disease list conditions in patients with rheumatoid arthritis in the private healthcare sector of South Africa. Rheumatol Int 38(5):837–844
Teclessou JN et al (2018) Connective tissue diseases in the hospital setting in Lome: a retrospective study of 231 cases. Les Connect. en milieu Hosp. a Lome etude Retrosp. 231 cas, vol. 30, p 176
Abhishek A, Doherty M, Kuo CF, Mallen CD, Zhang W, Grainge MJ (2017) Rheumatoid arthritis is getting less frequent-results of a nationwide population-based cohort study. Rheumatology (U K) 56(5):736–744
Dickens C, Jackson J, Tomenson B, Hay E, Creed F (2003) Association of depression and rheumatoid arthritis. Psychosomatics 44(3):209–215
Elenkov IJ (2008) Neurohormonal-cytokine interactions: implications for inflammation, common human diseases and well-being. Neurochem Int 52(1):40–51
Sankoh O, Sevalie S, Weston M (2018) Mental health in Africa. Lancet Glob Health 6(9):e954–e955
Chipeta J, Njobvu P, McGill PE, Bucala R (2014) Progress made towards enhancement of rheumatology education and practice in Zambia: review of an ILAR-supported project. Clin Rheumatol 33(10):1367–1372
“The joint, muscle and bone fixer - the Namibian.” [Online]. Available: https://www.namibian.com.na/164956/archive-read/The-joint-muscle-and-bone-fixer. Accessed: 20-Dec-2019
Ndongo S, Lekpa FK, Ka MM, Ndiaye N, Diop TM (2009) Presentation and severity of rheumatoid arthritis at diagnosis in Senegal. Rheumatology 48(9):1111–1113
Elizabeth H. Prospects for treating patients with arthritis in African countries with few rheumatologists. [Online]. Available: the-rheumatologist.org/article/prospects-treating-patients-arthritis-african-countries-rheumatologists. [Accessed: 06-Dec-2019]
“Arthritis Uganda has one specialist.” [Online]. Available: https://www.newvision.co.ug/new_vision/news/1279739/arthritis-uganda-specialist. [Accessed: 20-Dec-2019]
“(No Title).” [Online]. Available: https://rheumatologyforall.org/our-work/. [Accessed: 20-Dec-2019]
Genga EK, Moots RJ, Oyoo OG, OOtieno F (2016) Building a rheumatology team for East Africa. Rheumatology:kew432
Singwe-Ngandeu M, Ntandzi T, Sa’aLontsi S, Ngoufack C (2013) Treatment of rheumatoid arthritis in a developing country in the era of biologic therapies: a hospital based study in Yaounde, Cameroon. Clin Rheumatol 32(2 SUPPL. 1):S132–S133
Owino BO, Oyoo GO, Otieno CF (2009) Socio-demographic and clinical aspects of rheumatoid arthritis. East Afr Med J 86(5):204–211
Chipeta J, Njobvu P.D., and Bucala R (2013) Spectrum of paediatric rheumatological conditions in Sub-Saharan Africa. Clin Rheumatol 32(2 SUPPL. 1):S134
El Zorkany B et al (2013) Suboptimal management of rheumatoid arthritis in the Middle East and Africa: could the EULAR recommendations be the start of a solution? Clin Rheumatol 32(2):151–159
Bedaiwi M et al (2019) Disease burden and treatment challenges of psoriatic arthritis in Africa and the Middle East. Rheumatol Int 39(8):1321–1329
James PB, Wardle J, Steel A, Adams J (2018) Traditional, complementary and alternative medicine use in Sub-Saharan Africa: a systematic review. BMJ Glob Health 3(5):895
Abdullahi AA (2011) Trends and challenges of traditional medicine in Africa. Afr J Tradit Complement Altern Med 8(5 SUPPL):115–123
Tamhane A, McGwin G Jr, Redden DT, Hughes LB, Brown EE, Westfall AO, Conn DL, Jonas BL, Smith EA, Brasington RD, Moreland LW, Bridges SL Jr, Callahan LF (2014) Complementary and alternative medicine use in African Americans with rheumatoid arthritis. Arthritis Care Res 66(2):180–189
O’Donnell K et al (2019) Transparency is needed on NHS charges for migrants and data sharing agreements. The BMJ Opinion. [Online]. Available: https://blogs.bmj.com/bmj/2019/05/13/creating-a-hostile-environment-for-migrants-transparency-is-needed-on-nhs-charges-and-data-sharing-agreements-in-nhs-england/. [Accessed: 05-Dec-2019]
Gov.uk (2019) NHS entitlements: migrant health guide - GOV.UK. https://www.gov.uk/guidance/nhs-entitlements-migrant-health-guide. [Online]. Available: https://www.gov.uk/guidance/nhs-entitlements-migrant-health-guide. [Accessed: 02-Dec-2019]
Poduval S, Howard N, Jones L, Murwill P, Mckee M, Legido-Quigley H (2015) Experiences among undocumented migrants accessing primary care in the United Kingdom: a qualitative study. Int J Health Serv 45(2):320–333
Agudelo-Suárez AA, Gil-González D, Vives-Cases C, Love JG, Wimpenny P, Ronda-Pérez E (2012) A metasynthesis of qualitative studies regarding opinions and perceptions about barriers and determinants of health services’ accessibility in economic migrants. BMC Health Serv Res 12(1)
B. S. of Rheumatology (2019) National Early Inflammatory Arthritis Audit (NEIAA) 1st Annual Report. [Online]. Available: https://www.rheumatology.org.uk/practice-quality/audits/neiaa. Accessed: 15-Dec-2019
Acknowledgements
We thank the participants for their kindness to give their time to contribute to this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Zoe Rutter-Locher. The first draft of the manuscript was written by Zoe Rutter-Locher and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Rheumatology in Africa
Electronic supplementary material
ESM 1
(DOCX 54 kb)
Rights and permissions
About this article
Cite this article
Rutter-Locher, Z., Galloway, J. & Lempp, H. Rheumatology care of migrants from sub-Saharan Africa: a literature review and qualitative pilot study of patients’ perspectives. Clin Rheumatol 40, 3429–3438 (2021). https://doi.org/10.1007/s10067-020-05099-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05099-z