Abstract
Objectives
To explore the link between ultrasonographic features of dactylitis in psoriatic arthritis (PsA) and symptoms, digital tenderness and duration of dactylitis.
Methods
Forty-eight cases of PsA dactylitis were investigated using high frequency ultrasound (US) both in grey scale (GS) and Power Doppler (PD), evaluating the presence and the degree of flexor tenosynovitis, peri-tendinous oedema, subcutaneous PD, extensor tendon involvement, GS synovitis and intra-articular PD signal (PDS) of the involved digits. Patients were compared according to the presence of local pain and digital tenderness, the duration of dactylitis and the concomitant treatment.
Results
The presence of pain/tenderness was positively associated with US GS flexor tenosynovitis of grade > 2 (p < 0.001), PD-flexor tenosynovitis (p < 0.001), peri-tendinous oedema (p < 0.001) and subcutaneous PDS (p < 0.001); moreover, it was negatively associated with GS synovitis (p < 0.001) and intra-articular PD (p < 0.001). The same positive and negative association with US findings were found comparing patients with duration of dactylitis shorter or longer than the median (24 weeks) (p < 0.001 for all comparisons).
Conclusions
Pain and digital tenderness are linked to dactylitis duration and earlier lesions are associated with extra synovial inflammatory changes. These findings suggest a hitherto unappreciated extra synovial basis for symptoms in PsA dactylitis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Dactylitis, also known as sausage-digit, defined as diffuse and uniform swelling of the entire digit, is a hallmark of psoriatic arthritis (PsA) occurring in around 40% of cases [1, 2]. It may simultaneously involve multiple digits [3] and represents a clinical marker of disease severity [3, 4]. Dactylitis has been observed in all forms of spondyloarthritis [5, 6], and it has been included in the ClASsification for Psoriatic ARthritis (CASPAR) criteria due to its high specificity in PsA patients [7, 8]. There is a lack of uniformity in the measurement of dactylitis ranging from a dichotomic score to the validated Leeds Dactylitis Index (LDI), which is a score that allows a differentiation between tender and non-tender dactylitis [9,10,11].
Ultrasound (US) and magnetic resonance imaging (MRI) studies showed that dactylitis is a very heterogeneous entity, characterized by the variable association of inflammatory involvement of flexor tendons (tenosynovitis), adjacent soft tissue thickening/edema, synovitis of metacarpophalangeal (MCP), proximal interphalangeal (PIP) and distal interphalangeal (DIP) joints and, to a lesser degree, extensor tendon involvement and enthesitis [12,13,14,15].
Dactylitis may manifest as an acute/tender form (diffuse swelling of the digit, pain reported by the patient and tenderness at physical examination) or a chronic/non-tender form (diffuse swelling but no tenderness, recent pain not reported by the patient), also called “cold dactylitis” [16, 17]. In many clinical and US studies, the dactylitic fingers were not classified as being tender or non-tender and therefore this variant remains poorly understood. The absence of tenderness, despite obvious digital swelling, might have several potential explanations including the specific anatomical locations of inflammation, the relative degree of inflammation and the persistence of subclinical inflammation.
We hypothesized that an anatomical compartmentalization of dactylitic related US abnormalities may explain patient symptoms and may also in turn be linked to the durations of disease.
This cross-sectional study was designed to evaluate the US abnormalities observed in tender versus non tender dactylitis and to explore the association between sonographic features and dactylitis duration.
Patients and methods
All PsA patients with dactylitis consecutively seen at the Rheumatology Research Unit of University “Federico II” of Naples between August 2016 and February 2018 entered the study. Four patients were enrolled in two other Rheumatology Centers (Bologna and Reggio Emilia) and were examined in Naples because they were living there.
The study was conducted in accordance with the Declaration of Helsinki. Written informed consent was taken from all subjects and the study protocol was approved by the local Ethics Committee.
Inclusion criteria were the following: (1) presence of finger dactylitis, (2) dactylitis duration of more than 1 week, (3) fulfillment of CASPAR criteria and (4) age over 18 years. Exclusion criteria were the following: (1) current engagement in heavy manual work, (2) recent history of hand trauma (6 months), (3) concomitant treatment with biological or targeted synthetic DMARDs and (4) previous peri-tendinous or intra-articular corticosteroid injection in the involved finger.
Before US examination, a clinical assessment was performed by clinicians (RS, RM, CS) with longstanding expertise in the field of PsA who recorded the presence of dactylitis, swelling and tenderness of the involved finger. The diagnosis of dactylitis was made by physical examination (diffuse swelling of the digit, with or without tenderness) and was confirmed using the Leeds Dactylitis Instrument (a 10% difference in the ratio of circumference of the affected digit to the contralateral) [10].
A careful history was taken with a special regard to disease characteristics (including age of onset, disease duration and subset of disease). Clinical assessment was performed before US examination by a rheumatologist in each centre, who recorded presence or absence of local finger pain, tenderness and swelling of the finger joint.
The duration of dactylitis was based on the duration of diffuse digital swelling and/or pain as reported by the patients.
Patients were divided into two groups according to the dactylitis duration shorter or longer than the median duration of the sample.
Ultrasound and Power Doppler examination
US examination of the clinically affected finger was performed by a single rheumatologist (NG) trained and experienced in musculoskeletal US (more than 4 years of experience), blinded to the laboratory and clinical data (in particular about pain, tenderness and dactylitis duration), using a MyLab 70XVG machine equipped with a 6–18 MHz linear transducer (Esaote S.p.A., Genoa, Italy).
The US grey-scale (GS) imaging parameters were optimised for maximal image resolution; Power Doppler (PD) settings were standardized at the following values: 500 Hz for pulse repetition frequency, 3 for wall filter, 4 for persistence and color gain between 45 and 55%. The window of the color box was restricted to the areas studied. Measurements were conducted to the depth of 20 mm.
Flexor and extensor tendons, MCP, PIP and DIP joints of the affected fingers were assessed by GS and PD US evaluation in longitudinal and transverse scanning views, in accordance with current guidelines and publications [18, 19]. Joints were examined from both dorsal and volar sides. A generous quantity of gel was used, and compression with the US probe was avoided with the purpose of correctly evaluating vascularization. Each US examination took about 15 min in a darkened room.
All US images were reassessed by a senior rheumatologist (PM) with over 25 years of MSUS experience, to give a consensus result.
The following dactylitis-related sonographic lesions were investigated: flexor tenosynovitis, flexor peri-tendinous PD signal (PDS), peri-tendinous edema, subcutaneous PDS, extensor tendon involvement (including paratenonitis and enthesitis of extensor tendon at proximal-interphalangeal joint), joint synovitis and intra-articular PDS.
According to the Outcome Measures in Rheumatology (OMERACT) definitions, tenosynovitis was defined in grey-scale (GS) as an abnormal anechoic and/or hypoechoic tendon sheath widening [20].
Peri-tendinous oedema was defined as a diffuse hypo/isoechoic thickening of the extratendinous soft tissues around flexor tendon (pseudotenosynovitis) with positive PDS in the subcutaneous tissue, in long axis view [21].
To define the peritenon extensor tendon inflammation, we used the term paratenonitis which indicates a hypoechoic area surrounding a tendon without synovial sheath, with or without peri-tendinous PDS [22, 23]. Enthesitis of extensor tendon at PIP joint was graded as present in GS when it was found hypoechoic and increased thickness of the insertion into the bone, as compared to the body of tendon and to the controlateral joint [24, 25].
Synovitis was defined according to the OMERACT definition as an abnormal hypoechoic intra-articular tissue that is non-displaceable and poorly compressibile and that may exhibit intra-articular PDS [26].
The pathological US findings were graded in GS and PD with 0 when absent and 1 if present. Furthermore, tenosynovitis was assessed using the four-grade semi-quantitative scoring scale on B-mode (grade 0, normal; grade 1, minimal; grade 2, moderate; grade 3, severe) and Doppler mode (grade 0, no peri-tendinous Doppler signal; grade 1, focal peri-tendinous Doppler activity; grade 2, multifocal peri-tendinous Doppler activity; grade 3, diffuse peri-tendinous Doppler activity), as proposed by the OMERACT US group [20].
Joint synovitis was subjectively scored on a four-point semiquantitative scale in GS (0 = absence, 1 = mild, 2 = moderate and 3 = severe) [27]. Intra-articular PD was scored on a semiquantitative scale from 0 to 3 (0 = no intra-articular colour signal; 1 = mild, single vessel signal; grade 2 = moderate, greater than grade 1 to < 50% of the intra-articular area filled with colour signals and grade 3 = ≥ 50% of the intra-articular area filled with colour signals) [27].
Statistical analysis
The statistical analysis was performed using SPSS, version 23. All quantitative variables were expressed in terms of mean ± SD or median and range in case of strong violation of normality, while qualitative variables were expressed as percentages.
Intraobserver and inter-observer reliabilities were obtained in two measurements (at basal and at 3 months from the first US evaluation) using 20 static images of 20 patients. We utilized the Cohen’s kappa coefficient (k) for each sonographic lesion and values > 0.8 were considered as excellent.
Continuous variables were compared using t test or non-parametric tests when appropriate. Non-continuous variables were compared using chi-square test. Statistical tests were performed at a significance level of α = 0.05.
Results
Characteristics of patients
Thirty-seven PsA patients (with a total of 48 dactylitic fingers) were enrolled in the study (16 female and 21 male; mean age ± SD 46.1 ± 13.1 years, range 23–66 years; see demographic characteristics in Table 1). Nail involvement was present in 43% of patients but just in 30% of the fingers affected by dactylitis. Twenty-nine patients had dactylitis in one digit and eight patients had the contemporary or subsequent involvement of two or three digits. In more than half of the patients (19/37), the episode of dactylitis heralded the subsequent diagnosis of PsA.
Ultrasound findings
Intra-observer reliability of the two sonographers (NG and PM) was excellent for all parameters (k > 0.8). The inter-observer reliability depicted by k coefficient was 0.89 (95% CI0.85–0.93) for GS flexor tenosynovitis, 0.84 (95% CI 0.80–0.88) for flexor peri-tendinous PDS, 0.87 (95% CI 0.81–0.93) for peri-tendinous oedema, 0.83 (95% CI 0.75–0.90) for subcutaneous PDS, 0.84 (95% CI 0.76–0.93) for extensor tendon involvement, 0.83 (95% CI 0.80–0.86) for joint synovitis and 0.88 (95% CI 0.84–0.92) for intra-articular PDS.
The frequencies of local pain, tenderness and US abnormalities are reported in Table 2. Joint synovitis was present more frequently at PIP level (94% of cases with joint synovitis) while it was present at MCP and DIP level in 12% and 17% of cases, respectively. Simultaneous synovitis of MCP, PIP and DIP joints was not found in any patient.
Association between local pain, tenderness and US abnormalities
Patients who reported local pain/tenderness had higher prevalence of flexor peri-tendinous PDS (p < 0.001), peri-tendinous soft tissue oedema (p < 0.001) and subcutaneous PDS (p < 0.001). There was a significant association between clinical symptoms and the presence of tenosynovitis degree greater or equal 2 (p < 0.001). Moreover, we found reduced prevalence of US joint synovitis in patients without local pain/tenderness (p < 0.001) (Table 3).
Local pain and tenderness were associated with peri-tendinous oedema: 97% of patients with peri-tendinous oedema plus flexor tenosynovitis reported pain, while 94% of them had tenderness. Pain and tenderness were also associated with subcutaneous PDS: 97% of patients with subcutaneous PDS plus flexor peri-tendinous PDS had pain, while 92% of them presented tenderness. Of the five patients with flexor tenosynovitis plus joint involvement, only one had spontaneous pain and no one had tenderness. No patient with pain or tenderness had an isolated flexor tenosynovitis. Flexor tenosynovitis was associated with tenderness and pain only when it was concomitant with peritendinous oedema or with peritendinous PDS.
Association between the dactylitis duration and US abnormalities
We divided the patients into two groups according to dactylitis duration (shorter or longer than the median, 24 weeks). As shown in Table 4, the patients with shorter dactylitis duration had a significantly higher prevalence of all US parameters except for GS flexor tenosynovitis of grade < 2.
GS flexor tenosynovitis of grade > 2 was seen in 96% of cases with shorter dactylitis duration, while it was seen in 18% of cases with longer dactylitis duration (p < 0.001); regarding flexor peri-tendinous PDS of grade > 2, we observed similar results (p < 0.001). Local pain, tenderness, peri-tendinous oedema and subcutaneous PDS were more frequently detected in cases with a shorter dactylitis duration (p < 0.001). All cases of extensor tendon involvement were present in the group with a dactylitis duration longer than 24 weeks.
On the other hand, the presence of GS synovitis and intra-articular PDS was more frequent in patients with a dactylitis duration longer than 24 weeks (p < 0.001).
Examples of the US characteristics of a finger dactylitis duration shorter or longer than 24 weeks are shown in Figs. 1 and 2.
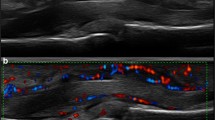
Longitudinal views of the second right finger in a patient with psoriatic dactylitis with duration less than 24 weeks. (a) Ultrasound shows flexor tenosynovitis of grade 2 (white star) with peritendinous oedema (white arrowhead) whereas joint synovitis is absent. (b) Power Doppler signal (PDS) is present around the tendon fibers and in subdermal tissue; intra-articular PDS is absent
Longitudinal views of the third right finger in a patient with psoriatic dactylitis with duration longer than 24 weeks. (a) Ultrasound shows no peritendinous oedema, distension of the flexor tendon sheath (grade 1) and of the capsule (grade 3) at proximal interphalangeal joint level. (b) PDS is absent in the articular, peritendinous and subcutaneous areas
Effects of csDMARDs on US findings
With regard to treatment, only 13 cases (27%) were taking csDMARDs (9 cases on methotrexate and 4 on sulphasalazine).
In order to look at the effect of csDMARDs on pain, tenderness and US findings, we split the cohort into those that had been on csDMARDs for more than 3 months and those who were csDMARDs naïve. We found no statistically significant differences between the two groups (data not shown).
Furthermore, in the group with shorter dactylitis duration (less than 24 weeks), 7 cases were on csDMARDs and 19 cases were DMARDs naïve. In the group with longer dactylitis duration (more than 24 weeks), 6 cases were on csDMARDs and 16 cases were DMARDs naïve. There were no statistically significant differences between the two groups. In Table 5, there is summarized prevalence of clinical and US abnormalities according to dactylitis duration therapy in conventional synthetic DMARDs naïve patients and patients on conventional synthetic DMARDs for more than 3 months. In Table 6, there is summarized prevalence of cases on therapy according to dactylitis duration.
Discussion
The present study showed for the first time that local pain and tenderness in PsA dactylitis is strongly associated with flexor tenosynovitis (grade > 2) and various extra-synovial features including flexor peritendinous PDS, peri-tendinous soft tissue oedema and subcutaneous PDS.
Historically, isolated flexor tenosynovitis was the most commonly reported US abnormality with joint synovitis as a variable coexisting feature [28,29,30]. In these previous studies, symptoms were not recorded, PD was not utilized and the dactylitic fingers were not classified as being tender or non-tender. In our study, in keeping with all previous US reports, flexor tenosynovitis was observed in almost all cases (94%) supporting the key role of this lesion in dactylitis.
Moreover, the current study has shown that peritendinous soft tissue oedema was present in 75% of cases, which was originally shown on MRI in PsA patients [31] and subsequently by other studies [32,33,34]. Fourniè et al. first used US to show diffuse inflammation of the digital soft tissues (reported as subcutaneous and extra-tendinous) in PsA patients with dactylitis, which they described as “pseudo-tenosynovitis” [21]. A recent US study observed that peri-tendinous lesions can differentiate PSA and rheumatoid arthritis (RA) [35]. In our study, pain and tenderness were associated with concomitant flexor tenosynovitis and peritendinous oedema. Therefore, the “pseudo-tenosynovitis” lesion (soft tissue thickening/oedema outside a flexor tendon) appears to be a cardinal one for joint symptoms in dactylitis.
Regarding tenderness, only one previous MRI study analysed the differences between the tender and non-tender forms of dactylitis although the dactylitis duration was not reported [32]. The authors showed that tender dactylitis had more abnormalities (in particular soft-tissue oedema and joint synovitis) than non-tender forms although the association was not strong.
Our data also confirm that synovitis is not required for the sausage-like appearance, as reported previously [3, 28,29,30]. We found a clear sign of joint synovitis (synovial hypertrophy with or without PDS) in 40% of cases, more frequently in cases with a longer dactylitis duration and without pain. It is not surprising that patients with US-detected synovitis had no pain as other studies have already reported a disparity between clinical and US findings for synovitis in other rheumatic diseases [36,37,38]. In particular, pain was not correlated with GS synovitis and intra-articular PD in patients with rheumatoid arthritis.
Moreover, the prevalence of local pain, tenderness, peritendinous oedema and peritendinous PDS decreased in patients with longer dactylitis duration. On the other hand, the prevalence of GS synovitis and intra-articular PDS increased over time. In a MRI study by Olivieri et al. that included 12 dactylitic toes (all tender and with a dactylitis duration shorter than 7 weeks), flexor tenosynovitis and peritendinous soft tissue edema were present in all patients (in most cases of grades 2 or 3). Additionally, joint synovitis (when present) was of grade 1 [33]. Our study for the first time evaluated the variation of US lesions according to dactylitis duration. Our results strongly suggest that US characteristics of dactylitis change over time but this obviously needs testing in longitudinal studies. From these data, we hypothesize that in the initial phase (acute form), dactylitis is symptomatic and tender; this form is characterized at US exam by GS flexor tenosynovitis of grade > 2, PD-flexor tenosynovitis, peritendinous oedema and subcutaneous PDS. In the following phase (chronic form), dactylitis is often asymptomatic and non-tender, while joint synovitis could be present. Longitudinal scans in early disease are needed to formally show that the painful lesion subsides in association with a reduction of peritendinous soft tissue oedema and/or PDS change improvement.
Although the precise pathogenesis of dactylitis is not well understood, it has been suggested by MRI studies that enthesitis is the primary lesion (with extra and intra-articular inflammation caused by proinflammatory mediators from the digital entheseal insertions and adjacent accessory pulleys) [39,40,41]. A prominent role for mechanical stress triggering micro-damage leading to inflammatory responses has been suggested [42, 43].
Further, our study, which is focused on hand dactylitis, cause a high proportion of subclinical inflammation that has been reported at the small joints of feet in psoriasis patients both with and without clinical arthritis [44].
This study has several limitations, the major one being its cross-sectional nature. The duration of symptoms is therefore obtained by medical history. However, we note that the variations observed were associated with significant differences in disease characteristics. A second limitation is related to the fact that treatments may have modified the US characteristics of dactylitis. However, we have observed that US characteristics appear to be significantly modified either by previous treatment with biological DMARDs or by peritendinous corticosteroid injection which were therefore used as exclusion criteria (study in preparation). Also, the patients on treatment still had an active disease such as to have a diagnosis of dactylitis.
Furthermore, in our study, we have not evaluated lesions of volar plate, pulley and extensor tendons at distal phalanx insertion because of limitation of our US machine. In our experience, the study of these structures is better made with probe with higher frequency (22–24 MHz).
In conclusion, we showed an association with inflammation outside synovial cavity and tenosynovial sheath compartments in dactylitis, especially in cases with a shorter dactylitis duration. US findings in symptomatic dactylitis are associated with the presence of peritendinous PDS and peritendinous soft tissue oedema. The chronic (non-tender) form is mainly characterized by joint synovitis at PIP level. These data suggest a link between dactylitis duration, symptoms and US features that could be important to understand the pathogenesis of dactylitis. Longitudinal studies to evaluate this are needed.
References
Gladman DD, Ziouzina O, Thavaneswaran A, Chandran V (2013) Dactylitis in psoriatic arthritis: prevalence and response to therapy in the biologic era. J Rheumatol 40:1357–1359
Kaeley GS, Eder L, Aydin SZ, Gutierrez M, Bakewell C (2018) Dactylitis: a hallmark of psoriatic arthritis. Semin Arthritis Rheum 48:263–273. https://doi.org/10.1016/j.semarthrit.2018.02.002
Brockbank JE, Stein M, Schentag CT, Gladman DD (2005) Dactylitis in psoriatic arthritis: a marker for disease severity? Ann Rheum Dis 64:188–190
Caso F, Costa L, Atteno M, Del Puente A, Cantarini L, Lubrano E et al (2014) Simple clinical indicators for early psoriatic arthritis detection. Springerplus 3:759
Dougados M, van der Linden S, Juhlin R, Huitfeldt B, Amor B, Calin A et al (1991) The European spondylarthropathy study group preliminary criteria for the classification of spondylarthropathy. Arthritis Rheum 34:1218–1227
Peluso R, Costa L, Caso F, Del Puente A, Di Minno MN, Manguso F et al (2016) Dactylitis in enteropathic spondyloarthritis. Clin Exp Rheumatol 34:842–847
Taylor W, Gladman D, Helliwell P, Marchesoni A, Mease P, Mielants H, CASPAR Study Group (2006) Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum 54:2665–2673
Tillett W, Costa L, Jadon D, Wallis D, Cavill C, McHugh J et al (2012) The ClASsification for Psoriatic ARthritis (CASPAR) criteria - a retrospective feasibility, sensitivity, and specificity study. J Rheumatol 39:154–156
Ramiro S, Smolen JS, Landewé R, van der Heijde D, Gossec L (2018) How are enthesitis, dactylitis and nail involvement measured and reported in recent clinical trials of psoriatic arthritis? A systematic literature review. Ann Rheum Dis 77:782–783
Helliwell PS, Firth J, Ibrahim GH, Melsom RD, Shah I, Turner DE (2005) Development of an assessment tool for dactylitis in patients with psoriatic arthritis. J Rheumatol 32:1745–1750
Healy PJ, Helliwell PS (2007) Measuring dactylitis in clinical trials: which is the best instrument to use? J Rheumatol 34:1302–1306
Eshed I, Bollow M, McGonagle DG, Tan AL, Althoff CE, Asbach P et al (2007) MRI of enthesitis of the appendicular skeleton in spondyloarthritis. Ann Rheum Dis 66:1553–1559
Bakewell CJ, Olivieri I, Aydin SZ, Dejaco C, Ikeda K, Gutierrez M, Terslev L, Thiele R, D'Agostino MA, Kaeley GS, OMERACT Ultrasound Task Force (2013) Ultrasound and magnetic resonance imaging in the evaluation of psoriatic dactylitis: status and perspectives. J Rheumatol 40:1951–1957
Tan AL, Fukuba E, Halliday NA, Tanner SF, Emery P, McGonagle D (2015) High-resolution MRI assessment of dactylitis in psoriatic arthritis shows flexor tendon pulley and sheath-related enthesitis. Ann Rheum Dis 74:185–189
Watad A, Eshed I, McGonagle D (2017) Lessons learned from imaging on enthesitis in psoriatic arthritis. Isr Med Assoc J 19:708–711 Review
Ferguson EG, Coates LC (2014) Optimisation of rheumatology indices: dactylitis and enthesitis in psoriatic arthritis. Clin Exp Rheumatol 32:113–117
Coates LC, Helliwell PS (2010) Disease measurement-enthesitis, skin, nails, spine and dactylitis. Best Pract Res Clin Rheumatol 24:659–670
Möller I, Janta I, Backhaus M, Ohrndorf S, Bong DA, Martinoli C, Filippucci E, Sconfienza LM, Terslev L, Damjanov N, Hammer HB, Sudol-Szopinska I, Grassi W, Balint P, Bruyn GAW, D’Agostino MA, Hollander D, Siddle HJ, Supp G, Schmidt WA, Iagnocco A, Koski J, Kane D, Fodor D, Bruns A, Mandl P, Kaeley GS, Micu M, Ho C, Vlad V, Chávez-López M, Filippou G, Cerón CE, Nestorova R, Quintero M, Wakefield R, Carmona L, Naredo E (2017) The 2017 EULAR standardised procedures for ultrasound imaging in rheumatology. Ann Rheum Dis 76:1974–1979
Backhaus M, Burmester GR, Gerber T, Grassi W, Machold KP, Swen WA, Wakefield RJ, Manger B, Working Group for Musculoskeletal Ultrasound in the EULAR Standing Committee on International Clinical Studies including Therapeutic Trials (2001) Guidelines for musculoskeletal ultrasound in rheumatology. Ann Rheum Dis 60:641–649
Naredo E, D'Agostino MA, Wakefield RJ, Möller I, Balint PV, Filippucci E, Iagnocco A, Karim Z, Terslev L, Bong DA, Garrido J, Martínez-Hernández D, Bruyn GAW, on behalf of the OMERACT Ultrasound Task Force* (2013) Reliability of a consensus-based ultrasound score for tenosynovitis in rheumatoid arthritis. Ann Rheum Dis 72:1328–1334
Fournié B, Margarit-Coll N, Champetier de Ribes TL, Zabraniecki L, Jouan A, Vincent V, Chiavassa H, Sans N, Railhac JJ (2006) Extrasynovial ultrasound abnormalities in the psoriatic finger. Prospective comparative power-doppler study versus rheumatoid arthritis. Joint Bone Spine 73:527–531
Gutierrez M, Filippucci E, Salaffi F, Di Geso L, Grassi W (2011) Differential diagnosis between rheumatoid arthritis and psoriatic arthritis: the value of ultrasound findings at metacarpophalangeal joints level. Ann Rheum Dis 70:1111–1114
Wakefield RJ, O'Connor PJ, Conaghan PG, McGonagle D, Hensor EM, Gibbon WW et al (2007) Finger tendon disease in untreated early rheumatoid arthritis: a comparison of ultrasound and magnetic resonance imaging. Arthritis Rheum 57:1158–1164
Terslev L, Naredo E, Iagnocco A, Balint PV, Wakefield RJ, Aegerter P, Aydin SZ, Bachta A, Hammer HB, Bruyn GAW, Filippucci E, Gandjbakhch F, Mandl P, Pineda C, Schmidt WA, D'Agostino MA, on behalf of the Outcome Measures in Rheumatology Ultrasound Task Force (2014) Defining enthesitis in spondyloarthritis by ultrasound: results of a Delphi process and of a reliability reading exercise. Arthritis Care Res (Hoboken) 66:741–748
Filippou G, Di Sabatino V, Adinolfi A, Bertoldi I, Picerno V, Biasi G et al (2013) No enthesis should be overlooked when psoriatic arthritis is suspected: enthesitis of the extensor digitorum tendons. J Rheumatol 40:335
Wakefield RJ, Balint PV, Szkudlarek M, Filippucci E, Backhaus M, D’Agostino MA et al (2005) Musculoskeletal ultrasound including definitions for ultrasonographic pathology. J Rheumatol 32:2485–2487
Szkudlarek M, Court-Payen M, Jacobsen S, Klarlund M, Thomsen HS, Østergaard M (2003) Interobserver agreement in ultrasonography of the finger and toe joints in rheumatoid arthritis. Arthritis Rheum 48:955–962
Olivieri I, Barozzi L, Favaro L, Pierro A, de Matteis M, Borghi C, Padula A, Ferri S, Pavlica P (1996) Dactylitis in patients with seronegative spondylarthropathy. Assessment by ultrasonography and magnetic resonance imaging. Arthritis Rheum 39:1524–1528
Kane D, Greaney T, Bresnihan B, Gibney R, FitzGerald O (1999) Ultrasonography in the diagnosis and management of psoriatic dactylitis. J Rheumatol 26:1746–1751
Wakefield RJ, Emery P, Veale D (2000) Ultrasonography and psoriatic arthritis. J Rheumatol 27:1564–1565
Giovagnoni A, Grassi W, Terilli F, Blasetti P, Paci E, Ercolani P et al (1995) MRI of the hand in psoriatic and rheumatical arthritis. Eur Radiol 5:590–595
Jevtic V, Watt I, Rozman B, Kos-Golja M, Demsar F, Jarh O (1995) Distinctive radiological features of small hand joints in rheumatoid arthritis and seronegative spondyloarthritis demonstrated by contrast-enhanced (Gd-DTPA) magnetic resonance imaging. Skelet Radiol 24:351–355
Olivieri I, Scarano E, Padula A, D'Angelo S, Salvarani C, Cantini F et al (2008) Fast spin echo-T2-weighted sequences with fat saturation in toe dactylitis of spondyloarthritis. Clin Rheumatol 27:1141–1145
Healy PJ, Groves C, Chandramohan M, Helliwell PS (2008) MRI changes in psoriatic dactylitis-extent of pathology, relationship to tenderness and correlation with clinical indices. Rheumatology (Oxford) 47:92–95
Tinazzi I, McGonagle D, Zabotti A, Chessa D, Marchetta A, Macchioni P (2018) Comprehensive evaluation of finger flexor tendon entheseal soft tissue and bone changes by ultrasound can differentiate psoriatic arthritis and rheumatoid arthritis. Clin Exp Rheumatol 36(5):785–790
Pereira DF, Gutierrez M, de Buosi AL, Ferreira FB, Draghessi A, Grassi W et al (2015) Is articular pain in rheumatoid arthritis correlated with ultrasound power Doppler findings? Clin Rheumatol 34:1975–1979
Hirata A, Ogura T, Hayashi N, Takenaka S, Ito H, Mizushina K, Fujisawa Y, Yamashita N, Nakahashi S, Imamura M, Kujime R, Kameda H (2017) Concordance of patient-reported joint symptoms, physician-examined arthritic signs, and ultrasound-detected synovitis in rheumatoid arthritis. Arthritis Care Res 69:801–806
Dejaco C, Stadlmayr A, Duftner C, Trimmel V, Husic R, Krones E, Zandieh S, Husar-Memmer E, Zollner G, Hermann J, Gretler J, Lackner A, Ficjan A, Datz C, Axman R, Zwerina J (2017) Ultrasound verified inflammation and structural damage in patients with hereditary haemochromatosis-related arthropathy. Arthritis Res Ther 19:243
McGonagle D, Gibbon W, Emery P (1998) Classification of inflammatory arthritis by enthesitis. Lancet 352:1137–1140
McGonagle D, Gibbon W, O'Connor P, Green M, Pease C, Emery P (1998) Characteristic magnetic resonance imaging entheseal changes of knee synovitis in spondylarthropathy. Arthritis Rheum 41:694–700
McGonagle D, Lories RJ, Tan AL, Benjamin M (2007) The concept of a “synovio-entheseal complex” and its implications for understanding joint inflammation and damage in psoriatic arthritis and beyond. Arthritis Rheum 56:2482–2491
Tan AL, Tanner SF, Waller ML, Hensor EM, Burns A, Jeavons AP et al (2013) High-resolution [18F]fluoride positron emission tomography of the distal interphalangeal joint in psoriatic arthritis--a bone-enthesis-nail complex. Rheumatology (Oxford) 52:898–904
Lories RJ, Matthys P, de Vlam K, Derese I, Luyten FP (2004) Ankylosing enthesitis, dactylitis, and onychoperiostitis in male DBA/1 mice: a model of psoriatic arthritis. Ann Rheum Dis 63:595–598
Mathew AJ, Bird P, Gupta A, George R, Danda D (2018) Magnetic resonance imaging (MRI) of feet demonstrates subclinical inflammatory joint disease in cutaneous psoriasis patients without clinical arthritis. Clin Rheumatol 37:383–388
Acknowledgments
The authors want to thank Professor Stefano Galletti for his suggestions.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was performed in accordance with the Helsinki Declaration of 1975, as amended in 2000, and the locally applicable laws. Written informed consent was taken from all subjects and the study protocol was approved by the local Ethics Committee.
Conflict of interest
DMG received grants and/or honoraria from Pfizer, MSD, Abbvie, BMS, UCB, Novartis, Celgene and Janssen.
The other authors have declared no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Girolimetto, N., Costa, L., Mancarella, L. et al. Symptomatic psoriatic dactylitis is associated with ultrasound determined extra-synovial inflammatory features and shorter disease duration. Clin Rheumatol 38, 903–911 (2019). https://doi.org/10.1007/s10067-018-4400-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-018-4400-z