Abstract
Mangroves and seagrasses are important nurseries for many marine species, and this function is linked to the complexity and context of these habitats in coastal seascapes. It is also connected to bathymetric features that influence habitat availability, and the accessibility of refuge habitats, but the significance of terrain variation for nursery function is unknown. To test whether seafloor terrain influences nursery function, we surveyed fish assemblages from mangrove and seagrass habitats in 29 estuaries in eastern Australia with unbaited underwater cameras and quantified the surrounding three-dimensional terrain with a set of complementary surface metrics (that is, depth, aspect, curvature, slope, roughness) applied to sonar-derived bathymetric maps. Terrain metrics explained variability in assemblages in both mangroves and seagrasses, with differing effects for the entire fish assemblage and nursery species composition, and between habitats. Higher depth, plan curvature (concavity or convexity) and roughness (backscatter) were negatively correlated with abundance and diversity in mangroves and positively linked to abundance and diversity in seagrass. Mangrove nursery species (6 species) were most abundant in forests adjacent to flats with concave holes, rough substrates and low-moderate depths, whereas seagrass nursery species (3 species) were most abundant in meadows adjacent to deep channels with soft mounds and ledges. These findings indicate that seafloor terrain influences nursery function and demonstrate contrasting effects of terrain variation in mangroves and seagrass. We suggest that incorporating three-dimensional terrain into coastal conservation and restoration plans could help to improve outcomes for fisheries management, but contrasting strategies might be needed for different nursery habitats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Highlights
-
Fish assemblages in coastal nurseries are linked to seafloor terrain variation
-
Distinct effects of terrain on nursery function in mangrove and seagrass habitats
-
Nursery paradigms must consider the three-dimensional context of coastal habitats
Introduction
Landscapes consist of a mosaic of ecosystems that provide a diversity of habitat functions for mobile organisms (Nagelkerken and others 2015; Liquete and others 2016). The ecological significance of many habitats is shaped by their capacity to provide these important functions, including food provision, protection from predators and safe reproduction sites, and is typically greatest in structurally complex ecosystems, as they support a diversity of ecological niches (Tews and others 2004; Borland and others 2021). The spatial distribution of ecosystems, and the habitat functions they confer, shape the diversity and distribution of species in most landscapes and can have pervasive effects on the growth and survival of juvenile animals (Baguette and Van Dyck 2007; Sheaves 2009). Habitats that support high densities of juveniles and facilitate elevated rates of growth and survival, and contribute disproportionally to the recruitment of juveniles into adult populations, are considered to function as ecological nurseries (Beck and others 2001; Barbier and others 2011; Whitfield and Pattrick 2015). The importance of nursery habitats as foraging and refugia sites has been documented for the juveniles of a diversity of species in coastal seascapes, and positive effects of habitat size, complexity and context on nursery function have been widely documented (Nagelkerken and others 2008; Whitfield 2017; Bradley and others 2020). Nursery habitats occur in seascapes that are also characterised by a diversity of three-dimensional terrain features (for example, intertidal flats with high curvature, subtidal channels with steep slopes and rocky pinnacles with high rugosity), and these seafloor features likely modify the availability of nurseries and the accessibility of subtidal refuge habitats, but it is not clear how terrain variability affects nursery function (Nagelkerken and others 2015; Borland and others 2021).
The spatial distribution of animals in most landscapes is shaped by the composition (that is, substrate characteristics such as roughness and hardness) and complexity (that is, topographic variation such as slope and rugosity) of terrain features (Simonson and others 2014; Pygas and others 2020). Animal abundance and diversity is positively correlated with terrain complexity in most seascapes, as the distribution of many species (for example, cetaceans, fish, turtles and crustaceans) is typically concentrated over complex bathymetric features (for example, reefs, pinnacles, seamounts and artificial structures) (Rex and others 2006; Pittman and others 2007b; Schlacher and others 2010; Rees and others 2014; Borland and others 2022a). Prominent terrain features often harbour diverse faunal assemblages because they support an abundance of food (for example, photosynthetic organisms and invertebrate prey species) (Cameron and others 2014; Rees and others 2018), provide settlement and sheltering sites (Sabatés and others 2007; Bejarano and others 2011) and offer refugia from various forms of disturbance (for example, hydrodynamic forces and fishing pressure) (Pittman and Brown 2011; Stamoulis and others 2018). These functions of complex terrain features are analogous to many of the services that are provided by nursery habitats (Sheaves and others 2015; Whitfield and Pattrick 2015), and for this reason areas of high bathymetric relief are often viewed as enhanced nurseries in continental shelf and rocky reef seascapes (Giannoulaki and others 2011; Farmer and others 2017; Pirtle and others 2017). Seafloor terrain variation in coastal seascapes might also modify the ecological values of nursery habitats, like mangroves and seagrasses, but this concept has not been widely explored in studies of the nursery role hypothesis.
Coastal seascapes comprise a heterogeneous mix of many marine plant communities, including mangroves, seagrasses, algal beds, saltmarshes and tidal freshwater marshes, which provide nursery habitats for a diversity of estuarine and marine species (Sheaves and others 2015; Whitfield 2017). Many biotic (for example, food availability, predation risk and resource competition) and abiotic (for example, water quality, sediment characteristics and hydrodynamic conditions) factors combine to shape the ecological values of coastal ecosystems as nursery habitats, and many of these can be optimised in large habitat patches, which confer high structural complexity, and are also well connected to alternative habitats (Beck and others 2001; Baker and Sheaves 2005; Whitfield and Pattrick 2015). Mangrove forests and seagrass meadows are prominent nursery habitats in many inshore seascapes, and these ecosystems are characterised by a diversity of structurally complex habitat features, such as roots, rhizomes, stems, leaves and debris, which provide refuge from predators and frequently support an abundance of food (Nagelkerken and others 2000; Sheaves and others 2015). Many studies have identified the importance of connections between these ecosystems and neighbouring habitats, including coral reefs, oyster reefs and other nursery habitats, which also provide fish with suitable refuge sites, foraging areas and spawning grounds (Connolly and Hindell 2006; Pittman and others 2007a; Heck Jr and others 2008; Sheaves 2009; Hyndes and others 2014; Nagelkerken and others 2015). The ecological significance of mangroves and seagrasses as nursery habitats is also modified by tidal regimes, which determine the frequency and duration of their availability for aquatic organisms (Igulu and others 2014; Whitfield 2017). As tides recede from inter-tidal habitats, nursery species must move to alternative deeper habitats where they are typically more vulnerable to predators (Sheaves 2005; Unsworth and others 2009). Subtidal habitats with high structural complexity, such as rock-bars, pinnacles and channel edges, can provide supplementary habitats for nursery species at low tide, but there is no data to describe how changes in the bathymetric characteristics of these terrain features influence the ecological roles of nursery habitats across coastal seascapes (Dorenbosch and others 2004; Lugendo and others 2007; Alp and Pichon 2020; Bradley and others 2020).
Here, we investigated the importance of seafloor terrain variation for the ecological roles of mangrove and seagrass habitats that provide important nursery functions for a diversity of marine species in eastern Australia (Sheaves and others 2016; Hayes and others 2020). Terrain features in the subtidal zones of these seascapes can provide similar habitat functions to mangroves and seagrass for marine animals and might therefore serve as supplementary nursery habitats (Wedding and others 2008; Pittman and Brown 2011; Borland and others 2022a), but this hypothesis has not been tested with empirical data. Furthermore, many species use mangroves and seagrass habitats as juveniles (that is, nursery species), whilst other transient species (that is, non-nursery species) also occupy these ecosystems opportunistically (for example, during foraging and reproductive migrations) (Harborne and others 2017). The contrasting effects of seascape context and complexity on assemblages of nursery and non-nursery species are widely appreciated (Harborne and others 2016; Berkstrom and others 2020), but it is not known whether variation in seafloor terrain has consistent effects on the composition of these species in nursery habitats. The primary aim of this research was to identify whether variation in the seafloor terrain of coastal seascapes affects the nursery function of intermittently available mangrove and seagrass habitats. Although it is well recognised that there are many measurable components of nursery function (for example, juvenile density, growth and survival), the intent of this study was to focus on the influence of seafloor terrain on particular aspects of this function: nursery species richness, abundance and composition (following Cheminée and others 2021; Dance and others 2021). The secondary objective of this research was to identify whether the composition of nursery species (that is, species with known juvenile-habitat associations in mangrove and seagrass nurseries) and the entire fish assemblage (that is, nursery and non-nursery species combined) respond to different terrain features in mangrove and seagrass nurseries. We predict that three-dimensional variation in seafloor terrain will have distinct effects on the composition, abundance and diversity of nursery species and that these effects will differ to the entire assemblage in mangrove and seagrass ecosystems and influence the value of these habitats as nurseries in coastal seascapes.
Methods
Study Area
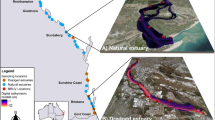
To investigate whether variation in seafloor terrain affects the ecological value of nursery habitats, fish assemblages were sampled from mangrove and seagrass habitats in 29 estuaries in eastern Australia (Figure S1). The focal estuaries support abundant mangrove forests and seagrass meadows and are characterised by substantial variation in the three-dimensional complexity of their terrain features, ranging from shallow sand-bar dominated estuaries (for example, Baffle Creek) to deeper estuaries with abundant subtidal rock bars (for example, Maroochy River) (Figure 1, Table S1) (Gilby and others 2018; Henderson and others 2019; Borland and others 2022a, b).
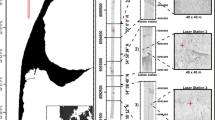
Estuaries demonstrating variation in the bathymetric context (that is, depth gradients measured as metres below HAT) of remote underwater video stations (RUVS) deployments in mangrove (brown circles) and seagrass (green circles) ecosystems (A and B); and a conceptual illustration of terrain metrics used to describe variation in seafloor relief (for example, depth), complexity (for example, slope), morphology (for example, curvature, aspect) and composition (for example, backscatter) in this study (C).
Fish Surveys
Fish assemblages from mangrove and seagrass habitats were surveyed using unbaited remote underwater video stations (RUVS), which were comprised of a high-definition GoPro camera mounted on a 3 kg weight (Gilby and others 2018; Henderson and others 2019). RUVS are widely used in ecological studies that examine the nursery value of different ecosystems for fish because they efficiently sample fish species from all size classes (Leslie and others 2017; Parsons and others 2018), and the absence of bait minimises the likelihood of attracting species to the cameras from other ecosystems (Sheaves and others 2016; Bradley and others 2017). RUVS were deployed at 180 sites in mangrove and seagrass habitats across the 29 estuaries. Our intent was to survey five sites in each habitat in each estuary (following Gilby and others 2018; Henderson and others 2019; Swadling and others 2019), but this was not always possible because some estuaries did not support seagrass or contained patches that were simply too small to ensure independent sampling. To standardise all RUVS for time and account for the potential confounding effects of tide, depth, water quality and seascape context, they were deployed: (1) for 30 min; (2) within 2 h of high tide; (3) in water depths between one and two metres; (4) from the mouth of each estuary to the point at which salinity reaches 30 psu (Practical Salinity Units) (that is, the marine extent of each estuary); and (5) in isolated mangrove and seagrass patches, which were separated by more than 500 m, but this was not always possible (90% efficiency) because of the scarcity of seagrass in some estuaries (Sheaves and others 2016; Brook and others 2018; Gilby and others 2018). RUVS were positioned over bare sediment parallel to mangroves and seagrass patches to capture species entering and exiting the nursery habitat (Henderson and others 2021). Data on fish abundance and diversity were obtained from video footage using the standard MaxN statistic (Ellis and Demartini 1995; Murphy and Jenkins 2010; Gladstone and others 2012). The MaxN was recorded for every species that swam within about 1 m of the camera to account for variable water clarity (following Gilby and others 2020). Fish were then categorised as nursery or non-nursery species based on their known utilisation of mangrove and seagrass habitats as juveniles (sensu Berkstrom and others 2020) (Table S2).
Quantifying Seafloor Terrain
To describe the three-dimensional terrain features of estuarine seascapes, and test for effects of terrain variation on fish assemblages, high-resolution (= 1 m−1) digital bathymetric models (DBMs) were created for each estuary using sonar data obtained from surveys with an acoustic sounder (Lowrance HDS 7–Gen 3). The bathymetry and bottom composition (that is, backscatter: E1 hardness, E2 roughness) of each estuary were mapped by logging the along-shore and cross-shore variation in terrain features on overlapping latitudinal and longitudinal transects across the sampled extent of each estuary, which were traversed at a maximum speed of 8 km/h−1. Sonar data were processed in ReefMaster 2.0, corrected to the highest astronomical tide (HAT) using tide data collected from pressure sensors (INW Smart Sensor) that were also deployed in each estuary (Young and others 2010; Li and others 2017; Borland and others 2022b). Terrain metrics were then quantified from DBMs to describe variation in the bathymetry surrounding the sampling sites in each estuary, using the Spatial Analyst and Benthic Terrain Modeler packages in ArcGIS (Walbridge and others 2018). Nine terrain metrics that are consistent predictors of the effects of terrain on fish assemblages in a diversity of marine seascapes were selected to describe variation in the: relief (that is, average depth), complexity (that is, slope and rugosity), morphology (that is, plan curvature; profile curvature; and aspect: northness and eastness) and composition (that is, backscatter: roughness and hardness) of seafloor terrain (Figure 1, Table 1) (Borland and others 2021). All terrain metrics were indexed by quantifying their mean values within a 500-m buffer of each sampling location; this scale was chosen because it encompasses the daily home ranges of many estuarine fishes and has been widely used in other studies of fish-seascape relationships in the study area (Brook and others 2018; Henderson and others 2019; Borland and others 2022a).
Data Analysis
Multivariate generalised linear models (manyGLMs) were constructed in the mvabund package in R to test whether variation in seafloor terrain complexity shapes the composition of fish assemblages and the abundance and diversity of nursery species in mangrove and seagrass habitats (Wang and others 2012). These models also included latitude to account for the large geographical extent of the study area, and estuary as fixed effect because manyGLMs cannot incorporate random factors. Prior to analysis, terrain metrics were tested for co-linearity using Pearson’s correlation coefficient, and subsequently, eastness (correlated with northness), profile curvature (correlated with plan curvature), rugosity (correlated with slope) and hardness (correlated with roughness) were removed from analyses (r2 = ≥ 0.7). Best-fit models were selected using reverse stepwise simplification, based on Akaike Information Criterion (AIC). The “p.uni” function within the mvabund package was then used to identify indicator species that were correlated with significant factors within the best-fit manyGLM model. Non-metric multidimensional scaling ordinations (nMDS) were then used to visualise effects of significant factors identified by the manyGLM on the composition of fish assemblages and on the abundance and diversity of nursery species, in mangrove and seagrass habitats. Generalised linear mixed models (GLMMs) were then constructed using the glmmTMB package in R to further investigate, and illustrate, the effects of significant predictors identified by each manyGLM on fish abundance and species richness (Brooks and others 2017). GLMMs were fitted with natural splines, with three or fewer polynomial functions, to encapsulate potential nonlinear effects of terrain using the splines package in R, and included estuary as a random factor (Brooks and others 2017; Geraci and others 2021). The relationship between significant indicator species (as identified by “p.uni” analyses) and variables from the best-fit manyGLMs were then plotted using the predict.manyglm function within the mvabund package in R (Wang and others 2012). Univariate and multivariate GLM(M)s were checked for normally distributed residuals, homogeneity of variance, outliers and over-dispersion and were fitted with negative binomial distributions, with log link functions.
Results
Effects of Seafloor Terrain on Fish Assemblages in Mangrove and Seagrass Habitats
The composition of fish assemblages in both mangroves and seagrass was associated with terrain features surrounding these habitats, but the nature of these correlations differed for the entire fish assemblage (that is, nursery and non-nursery species) versus the composition of nursery species (Figure 2, Tables 2 and 3). The composition of the entire fish assemblage varied between estuaries (8 species) and with changes in the plan curvature (5 species), slope (2 species) and depth (1 species) of terrain features, but the effects of depth differed between mangrove and seagrass habitats (Figures 2 and S2, Tables 2 and S3). By contrast, the composition of nursery species was linked to changes in plan curvature (5 species), roughness (2 species) and average depth (1 species) in mangroves and varied between estuaries (6 species) and with changes in roughness (2 species) and depth (1 species) in seagrass (Figures 2 and S3, Tables 3 and S4).
Non-metric multi-dimensional scaling ordinations (nMDS) illustrating the effects of seafloor terrain variation on the entire fish assemblage (that is, nursery and non-nursery species) in A mangrove and B seagrass habitats, and the effects of terrain changes on nursery species in C mangrove and D seagrass habitats. Black lines illustrate the correlation of significant terrain metrics with variation in assemblage composition (identified by manyGLM analysis); dotted lines illustrate correlations between indicator species and significant terrain metrics (identified by “p.uni” analysis); and text in bold illustrates significant interactions between terrain metrics and habitat (that is, a categorical factor delineating either mangrove or seagrass ecosystems).
Effects of Seafloor Terrain on Nursery Species in Mangrove and Seagrass Habitats
The abundance and diversity of nursery species was highest at mangrove sites near terrain features with: low-moderate average depths (that is, 3 m), and either gradually (abundance) or sharply (diversity) declined at both lower and higher depths; high roughness (that is, 3.8), and gradually increased with increasing roughness; and low plan curvature (that is, − 0.008), and sharply declined with increasing or decreasing plan curvature (Figure 3, Table 4). By contrast, the abundance and diversity of nursery species in seagrass was highest at sites near terrain features with: high average depths (that is, 4.1 m) and gradually increased with increasing depths; and low roughness (that is, 1.9) and sharply decreased with increasing roughness (Figure 3, Table 4).
In mangrove forests, variation in the depth, roughness and plan curvature of terrain features were linked to changes in the abundance of six nursery species (Figure 4, Table S4). The abundance of common silverbiddy (Gerres subfasciatus) sharply decreased with increasing depths and was highest at sites with low depths (that is, 1.3 m) (Figure 4, Table S4). The abundance of grass emperor (Lethrinus laticaudis) gradually decreased with increasing roughness, was highest at sites with low roughness (that is, 1.2) and were not present when roughness was low-moderate (that is, > 2) (Figure 4, Table S4). The abundance of common silverbiddy sharply increased with increasing roughness and was highest at sites with high roughness (that is, 3.8) (Figure 4, Table S4). The abundance of striped scat (Selenotoca multifasciata), goldlined rabbitfish (Siganus lineatus), moses perch (Lutjanus russelli) and pacific seabream (Acanthopagrus pacificus) sharply decreased with increasing plan curvature, were highest at sites with low plan curvature (that is, − 0.02) and were not present when plan curvature was low–moderate (that is, > 0) (Figure 4, Table S4). The abundance of grass emperor sharply increased with increasing plan curvature, was highest at sites with high plan curvature (that is, 0.02) and was not present when plan curvature was moderate–high (that is, < 0.005) (Figure 4, Table S4).
In seagrass meadows, variation in the depth and roughness of terrain features were linked to changes in the abundance of three nursery species (Figure 5, Table S4). The abundance of tarwhine (Rhabdosargus sarba) gradually increased with increasing depth, was highest at sites with greater depths (that is, 4.1 m) and were not present when depths were low-moderate (< 3.7 m) (Figure 5, Table S4). The abundance of luderick (Girella tricuspidata) and sand whiting (Sillago ciliata) decreased sharply with increasing roughness and were highest at sites with lower roughness (that is, 1.9) (Figure 5, Table S4).
Discussion
Mangrove and seagrass ecosystems provide a variety of habitat functions for coastal organisms and are considered to be important nurseries for the juveniles of many species (Whitfield 2017; Lefcheck and others 2019), but these habitats are also located in diverse seascapes with variable bathymetry and changes in terrain complexity, which could mediate their roles as nurseries (Borland and others 2021). Our findings demonstrate, for the first time, that variation in the three-dimensional terrain features of coastal seascapes shapes the composition of fish assemblages in mangrove and seagrass nurseries. There was, however, a large diversity of responses, as the significance of terrain for fish assemblages differed between species that use mangrove and seagrass ecosystems as nurseries (that is, nursery species) and the entire fish assemblage (that is, nursery and non-nursery species), and between the different nursery habitats. Fish assemblages in both mangrove and seagrass habitats were affected by changes in the depth, plan curvature (that is, convexity or concavity) and slope of terrain features in the surrounding seascape. By contrast, nursery species were linked to the depth and roughness (that is, backscatter) of adjacent terrain features in both mangrove and seagrass habitats; and plan curvature in mangrove forests. The abundance and diversity of mangrove nursery species were highest in forests near rough terrain features with concave holes in shallow-moderate depths, whereas seagrass nursery species were most abundant and diverse in meadows near deep channels with soft sediment mounds and ledges. These findings align with previous research highlighting the importance of seafloor terrain for the distribution of fish species, composition of fish assemblages and provision of potential nursery habitats in deep reef and shelf seascapes (Farmer and others 2017; Pirtle and others 2017; Pygas and others 2020; Borland and others 2021) and also demonstrate that the ecological significance of seafloor terrain can transcend habitat boundaries and structure nursery function in coastal seascapes.
Mangrove forests are nursery habitats for a diversity of species, but often grow high on intertidal zones where they are only available for short periods of each tidal cycle, and fish must retreat to subtidal structure when tides recede (Igulu and others 2014; Baker and others 2015). Our results show, for the first time, that the composition of nursery species in mangrove forests was modified by variation in plan curvature (5 species), roughness (2 species) and depth (1 species). Nursery species abundance, richness and the abundance of common silverbiddy were negatively correlated with high average depths. Shallow mangrove forests contain high densities of juveniles from many species because predation risk is typically lower here than in deeper water, and these areas provide safe locations for juveniles to feed and disperse regardless of tidal state, or the availability of submerged vegetation (Ellis and Bell 2004; Rypel and others 2007; Rotherham and others 2011; Munsch and others 2016). The abundance of two nursery species, grass emperor and common silverbiddy, was correlated with terrain roughness (that is, backscatter). Grass emperor occupy mangroves with soft sediment terrain (that is, low roughness) because these substrates harbour a high abundance of their invertebrate prey (for example, crustaceans and molluscs) (Salini and others 1994; Brewer and others 1995). By contrast, common silverbiddy move between mangroves and subtidal habitats tidally, and we suggest that hard subtidal terrain features (for example, reefs with high backscatter) provide supplementary nursery habitats for this species when mangrove forests are exposed at low tide (Fowler and Booth 2013; Morton and Gladstone 2014). Nursery species abundance, richness and the abundance of four species (that is, striped scat, goldlined rabbitfish, moses perch and pacific seabream) were positively correlated with the presence of concave terrain features. Curvature in the seafloor can provide a degree of protection from hydrodynamic forces and predation pressure, and concave terrain features (for example, holes and undercuts) might provide nursery species with refugia that help to minimise energy expenditure and risk and maximise growth, when mangroves are not available (Johnston and Sheaves 2007; Pavlov and others 2008; Pirtle and others 2017). By contrast, the abundance of grass emperor was positively correlated with the presence of convex terrain features in the seascape. Juvenile grass emperor migrate tidally between seagrass meadows and mangrove forests to feed and shelter, and their association with convex terrain features (for example, bars and mounds) possibly reflects the accumulation of sediment in, and around, subtidal seagrass meadows (Wilson 1998; Kimball and others 2018; Quang Le and others 2020). Variation in the terrain of estuaries is, therefore, inherently linked to the nursery function of intertidal mangroves because topographically complex terrain features provide important subtidal refuge habitats that are critical components of coastal seascape nurseries (Nagelkerken and others 2015).
Seagrass meadows are nursery habitats that support the juveniles of many species in high abundance, but many nursery species that use seagrass also undertake regular tidal and/or diurnal feeding migrations into adjacent soft sediments habitats (McDevitt-Irwin and others 2016; Whitfield 2017). Our findings demonstrate that the composition of nursery species in seagrass meadows was affected by variation in depth (1 species) and roughness (2 species). Nursery species abundance and the abundance of tarwhine were positively correlated with depth. Deep terrain features, such as tidal channels, are used as migration pathways and dispersal conduits by juveniles and larvae, which frequently settle in adjacent seagrass meadows that offer protection from hydrodynamic forces and a diversity of feeding opportunities (Heithaus 2004; Gullström and others 2008; Ford and others 2010; Kough and others 2017). Nursery species richness and the abundance of two species, sand whiting and luderick, were negatively correlated with roughness. Seagrass meadows that are interspersed with soft sediments (that is, low roughness) are often hotspots for juvenile fish diversity because these areas provide complementary foraging grounds for generalist zoobenthivores (for example, sand whiting) and omnivores (for example, luderick) (Raubenheimer and others 2005; Hadwen and others 2007). The larvae of some seagrass nursery species (for example, sand whiting) also settle over shallow soft sediments (for example, intertidal flats and tidal pools) before later migrating to seagrass, because these areas support few predators and abundant food (for example, nematodes) (Burchmore and others 1988; Krück and others 2009). The three-dimensional characteristics of surrounding estuarine seascapes is, therefore, likely connected to the ecological significance of seagrass nurseries because complex terrain features provide complementary habitats and support similar habitat functions (for example, foraging sites, predator refuges and larval settlement locations) to many seagrass meadows (Hyndes and others 2014).
The ecological values of coastal nursery habitats depend on their position in the wider seascape, and the spatial distribution of fish assemblages has been linked to the two-dimensional proximity, area and connectivity (that is, seascape context) of complementary nursery habitats like mangrove forests, seagrass meadows and coral reefs (Harborne and others 2016; Berkstrom and others 2020). We show that the significance of nursery habitats can also be modified by the three-dimensional characteristics of the seafloor and demonstrate that a variety of terrain features combine to shape the distribution of fish assemblages in mangrove and seagrass ecosystems. These results support the findings of other research, which has suggested that subtidal terrain features, such as shallow sand banks, steep rocky ledges and outcrops, and curved banks and gullies, provide nursery habitats for a diversity of species and often mirror habitat functions (for example, foraging grounds, predator refuges and spawning locations) that are provided by intertidal nurseries (Rochette and others 2010; Farmer and others 2017; Pirtle and others 2017). The synergistic roles of connected intertidal and subtidal habitats in underpinning nursery functions for a diversity of species is now widely accepted (see reviews by Nagelkerken 2009; Sheaves 2009; Olds and others 2016; Pittman 2018). Recognition of the significance of inter-habitat linkages culminated in the development of the “seascape nursery” concept (sensu Nagelkerken and others 2015), which advances the nursery habitat paradigm considerably, but still conceptualises nurseries as two-dimensional mosaics of interlinked benthic habitats. Given the ecological consequences of seafloor terrain variation for nursery habitat function (see Borland and others 2021), we suggest that the seascape nursery concept could be expanded to incorporate variation in the three-dimensional structure of the seafloor, with potential wider implications for the spatial conservation of nursery habitats and the management of fisheries for nursery species.
Conclusion
We report strong effects of three-dimensional terrain features on the ecological function of nursery habitats in coastal seascapes and demonstrate that bathymetric variability has distinct effects on species, depending on life stages and their utilisation of mangrove and seagrass nurseries as juvenile habitat. Our findings show that the use of nursery habitats by a diversity of fish species is affected by a suite of terrain attributes in the surrounding seascape. The results of this study demonstrate that the ecological consequences of terrain variation transcend the boundaries of ecosystems and can differ between mangrove and seagrass habitats. Given the significance of seafloor variation for nursery function, we recommend that nursery habitats and seascapes should be evaluated in three dimensions to better incorporate heterogeneity in the complexity, morphology and relief of undersea terrain. The key challenge now is for research to examine how three-dimensional terrain and two-dimensional seascape context combine to influence nursery function, such as the growth and survival of juveniles, and the consistency of these effects across a diversity of seascapes and habitat patches. We suggest that integrating seafloor terrain into the seascape nursery concept should help to improve our understanding of nursery habitat function and might have wider implications for habitat prioritisation decisions in coastal conservation and restoration.
Data Availability
Data available from the USC Research Bank (https://doi.org/10.25907/00138).
References
Alp M, Pichon CL. 2020. Getting from sea to nurseries: considering tidal dynamics of juvenile habitat distribution and connectivity in a highly modified estuarine riverscape. Ecosystems 24:583–601.
Baguette M, Van Dyck H. 2007. Landscape connectivity and animal behavior: functional grain as a key determinant for dispersal. Landscape Ecology 22:1117–29.
Baker R, Sheaves M. 2005. Redefining the piscivore assemblage of shallow estuarine nursery habitats. Marine Ecology Progress Series 291:197–213.
Baker R, Sheaves M, Johnston R. 2015. Geographic variation in mangrove flooding and accessibility for fishes and nektonic crustaceans. Hydrobiologia 762:1–14.
Barbier EB, Hacker SD, Kennedy C, Koch EW, Stier AC, Silliman BR. 2011. The value of estuarine and coastal ecosystem services. Ecological Monographs 81:169–93.
Beck MW, Heck KL, Able KW, Childers DL, Eggleston DB, Gillanders BM, Halpern B, Hays CG, Hoshino K, Minello TJ, Orth RJ, Sheridan PF, Weinstein MR. 2001. The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. Bioscience 51:633–41.
Bejarano S, Mumby PJ, Sotheran I. 2011. Predicting structural complexity of reefs and fish abundance using acoustic remote sensing (RoxAnn). Marine Biology 158:489–504.
Berkstrom C, Eggertsen L, Goodell W, Cordeiro CAMM, Lucena MB, Gustafsson R, Bandeira S, Jiddawi N, Ferreira CEL. 2020. Thresholds in seascape connectivity: the spatial arrangement of nursery habitats structure fish communities on nearby reefs. Ecography 43:882–96.
Blaber SJM, Blaber TG. 1980. Factors affecting the distribution of juvenile estuarine and inshore fish. Journal of Fish Biology 17:143–62.
Borland HP, Gilby BL, Henderson CJ, Leon JX, Schlacher TA, Connolly RM, Pittman SJ, Sheaves M, Olds AD. 2021. The influence of seafloor terrain on fish and fisheries: a global synthesis. Fish and Fisheries 22:707–34.
Borland HP, Gilby BL, Henderson CJ, Connolly RM, Gorissen B, Ortodossi NL, Rummell AJ, Pittman SJ, Sheaves M, Olds AD. 2022a. Dredging fundamentally reshapes the ecological significance of 3D terrain features for fish in estuarine seascapes. Landscape Ecology.
Borland HP, Gilby BL, Henderson CJ, Connolly RM, Gorissen B, Ortodossi NL, Rummell AJ, Pittman SJ, Sheaves M, Olds AD. 2022b. Dredging transforms the seafloor and enhances functional diversity in urban seascapes. Science of the Total Environment, 831:154811.
Bradley M, Baker R, Sheaves M. 2017. Hidden components in tropical seascapes: deep-estuary habitats support unique fish assemblages. Estuaries and Coasts 40:1195–206.
Bradley M, Nagelkerken I, Baker R, Sheaves M. 2020. Context dependence: a conceptual approach for understanding the habitat relationships of coastal marine fauna. Bioscience 70:986–1004.
Brewer DT, Blaber SJM, Salini JP, Farmer MJ. 1995. Feeding ecology of predatory fishes from Groote Eylandt in the Gulf of Carpentaria, Australia, with special reference to predation on penaeid prawns. Estuarine, Coastal and Shelf Science 40:577–600.
Brook TW, Gilby BL, Olds AD, Connolly RM, Henderson CJ, Schlacher TA. 2018. The effects of shoreline armouring on estuarine fish are contingent upon the broader urbanisation context. Marine Ecology Progress Series 605:195–206.
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM. 2017. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. The R Journal 9:400.
Burchmore J, Pollard D, Middleton M, Bell J, Pease B. 1988. Biology of four species of Whiting (Pisces: Sillaginidae) in Botany Bay, NSW. Journal of Marine and Freshwater Research 39:709–27.
Cameron MJ, Lucieer V, Barrett NS, Johnson CR, Edgar GJ. 2014. Understanding community-habitat associations of temperate reef fishes using fine-resolution bathymetric measures of physical structure. Marine Ecology Progress Series 506:213–29.
Cheminée A, Le Direach L, Rouanet E, Astruch P, Goujard A, Blanfuné A, Bonhomme D, Chassaing L, Jouvenel JY, Ruitton S, Thibaut T, Harmelin-Vivien M. 2021. All shallow coastal habitats matter as nurseries for Mediterranean juvenile fish. Scientific Reports 11.
Connolly RM, Hindell JS. 2006. Review of nekton patterns and ecological processes in seagrass landscapes. Estuarine, Coastal and Shelf Science 68:433–44.
Dance MA, Rooker JR, Kline RJ, Quigg A, Stunz GR, Wells RJD, Lara K, Lee J, Suarez B. 2021. Importance of low-relief nursery habitat for reef fishes. Ecosphere 12.
Dorenbosch M, Verweij MC, Nagelkerken I, Jiddawi N, Van Der Velde G. 2004. Homing and daytime tidal movements of juvenile snappers (Lutjanidae) between shallow-water nursery habitats in Zanzibar, western Indian Ocean. Environmental Biology of Fishes 70:203–09.
Ellis WL, Bell SS. 2004. Conditional use of mangrove habitats by fishes: depth as a cue to avoid predators. Estuaries 27:966–76.
Ellis DM, Demartini EE. 1995. Evaluation of a Video Camera Technique for Indexing Abundances of Juvenile Pink Snapper, Pristipomoides-Filamentosus, and Other Hawaiian Insular Shelf Fishes. Fishery Bulletin 93:67–77.
Farmer NA, Heyman WD, Karnauskas M, Kobara S, Smart TI, Ballenger JC, Reichert MJM, Wyanski DM, Tishler MS, Lindeman KC, Lowerre-Barbieri SK, Switzer TS, Solomon JJ, McCain K, Marhefka M, Sedberry GR. 2017. Timing and locations of reef fish spawning off the southeastern United States. PLoS One 12.
Ford JR, Williams RJ, Fowler AM, Cox DR, Suthers IM. 2010. Identifying critical estuarine seagrass habitat for settlement of coastally spawned fish. Marine Ecology Progress Series 408:181–93.
Fowler AM, Booth DJ. 2013. Seasonal dynamics of fish assemblages on breakwaters and natural rocky reefs in a temperate estuary: consistent assemblage differences driven by sub-adults. PLoS One 8:e75790.
Geraci ML, Colloca F, Di Maio F, Falsone F, Fiorentino F, Sardo G, Scannella D, Gancitano V, Vitale S. 2021. How is artificial lighting affecting the catches in deep water rose shrimp trawl fishery of the Central Mediterranean Sea? Ocean & Coastal Management 215:105970.
Giannoulaki M, Pyrounaki MM, Liorzou B, Leonori I, Valavanis VD, Tsagarakis K, Bigot JL, Roos D, De Felice A, Campanella F, Somarakis S, Arneri E, Machias A. 2011. Habitat suitability modelling for sardine juveniles (Sardina pilchardus) in the Mediterranean Sea. Fisheries Oceanography 20:367–82.
Gilby BL, Olds AD, Connolly RM, Maxwell PS, Henderson CJ, Schlacher TA. 2018. Seagrass meadows shape fish assemblages across estuarine seascapes. Marine Ecology Progress Series 588:179–89.
Gilby BL, Olds AD, Duncan CK, Ortodossi NL, Henderson CJ, Schlacher TA. 2020. Identifying restoration hotspots that deliver multiple ecological benefits. Restoration Ecology 28:222–32.
Gladstone W, Lindfield S, Coleman M, Kelaher B. 2012. Optimisation of baited remote underwater video sampling designs for estuarine fish assemblages. Journal of Experimental Marine Biology and Ecology 429:28–35.
Gullström M, Bodin M, Nilsson PG, Öhman MC. 2008. Seagrass structural complexity and landscape configuration as determinants of tropical fish assemblage composition. Marine Ecology Progress Series 363:241–55.
Hadwen WL, Russell GL, Arthington AH. 2007. Gut content- and stable isotope-derived diets of four commercially and recreationally important fish species in two intermittently open estuaries. Marine and Freshwater Research 58:363–75.
Halliday IA, Young WR. 1996. Density, biomass and species composition of fish in a subtropical Rhizophora stylosa Mangrove forest. Marine and Freshwater Research 47:609–15.
Harborne AR, Nagelkerken I, Wolff NH, Bozec Y-M, Dorenbosch M, Grol MGG, Mumby PJ. 2016. Direct and indirect effects of nursery habitats on coral-reef fish assemblages, grazing pressure and benthic dynamics. Oikos 125:957–67.
Harborne AR, Selwyn JD, Lawson JM, Gallo M. 2017. Environmental drivers of diurnal visits by transient predatory fishes to Caribbean patch reefs. Journal of Fish Biology 90:265–82.
Hayes MA, McClure EC, York PH, Jinks KI, Rasheed MA, Sheaves M, Connolly RM. 2020. The differential importance of deep and shallow seagrass to nekton assemblages of the great barrier reef. Diversity 12.
Heck KL Jr, Carruthers TJB, Duarte CM, Randall Hughes A, Kendrick G, Orth RJ, Williams SW. 2008. Trophic transfers from seagrass meadows subsidize diverse marine and terrestrial consumers. Ecosystems 11:1198–210.
Heithaus MR. 2004. Fish communities of subtropical seagrass meadows and associated habitats in Shark Bay, western Australia. Bulletin of Marine Science 75:79–99.
Henderson CJ, Gilby B, Schlacher TA, Connolly R, Sheaves M, Flint N, Borland HP, Olds AD. 2019. Contrasting effects of mangroves and armoured shorelines on fish assemblages in tropical estuarine seascapes. ICES Journal of Marine Science 76:1052–061.
Henderson CJ, Gilby BL, Stone E, Borland HP, Olds AD. 2021. Seascape heterogeneity modifies estuarine fish assemblages in mangrove forests. ICES Journal of Marine Science 78:1108–116.
Hyndes GA, Nagelkerken I, McLeod RJ, Connolly RM, Lavery PS, Vanderklift MA. 2014. Mechanisms and ecological role of carbon transfer within coastal seascapes. Biological Reviews 89:232–54.
Igulu MM, Nagelkerken I, Dorenbosch M, Grol MG, Harborne AR, Kimirei IA, Mumby PJ, Olds AD, Mgaya YD. 2014. Mangrove habitat use by juvenile reef fish: meta-analysis reveals that tidal regime matters more than biogeographic region. PLoS One 9:e114715.
Johnston R, Sheaves M. 2007. Small fish and crustaceans demonstrate a preference for particular small-scale habitats when mangrove forests are not accessible. Journal of Experimental Marine Biology and Ecology 353:164–79.
Kimball ME, Boswell KM, Rozas LP, Berwaldt EK, Richards AR. 2018. Swimming abilities of juvenile estuarine fishes: implications for passage at water control structures. Wetlands Ecology and Management 26:383–90.
Kough AS, Cronin H, Skubel R, Belak CA, Stoner AW. 2017. Efficacy of an established marine protected area at sustaining a queen conch Lobatus gigas population during three decades of monitoring. Marine Ecology Progress Series 573:177–89.
Krück NC, Chargulaf CA, Saint-Paul U, Tibbetts IR. 2009. Early post-settlement habitat and diet shifts and the nursery function of tidepools during Sillago spp. recruitment in Moreton Bay. Australia. Marine Ecology Progress Series 384:207–19.
Laegdsgaard P, Johnson CR. 1995. Mangrove habitats as nurseries: unique assemblages of juvenile fish in subtropical mangroves in eastern Australia. Marine Ecology Progress Series 126:67–81.
Le Quang D, Yin Fui S, Tanaka K, Suratman S, Sano Y, Shirai K. 2020. Feeding habitats of juvenile reef fishes in a tropical mangrove-seagrass continuum along a Malaysian shallow-water coastal lagoon. Bulletin of Marine Science 96:469–86.
Lefcheck JS, Hughes BB, Johnson AJ, Pfirrmann BW, Rasher DB, Smyth AR, Williams BL, Beck MW, Orth RJ. 2019. Are coastal habitats important nurseries? A meta-analysis. Conservation Letters 12.
Leslie T, James NC, Potts WM, Rajkaran A. 2017. The relationship between habitat complexity and nursery provision for an estuarine-dependent fish species in a permanently open South African Estuary. Estuarine, Coastal and Shelf Science 198:183–92.
Li D, Tang C, Xia C, Zhang H. 2017. Acoustic mapping and classification of benthic habitat using unsupervised learning in artificial reef water. Estuarine, Coastal and Shelf Science 185:11–21.
Liquete C, Cid N, Lanzanova D, Grizzetti B, Reynaud A. 2016. Perspectives on the link between ecosystem services and biodiversity: the assessment of the nursery function. Ecological Indicators 63:249–57.
Lugendo BR, Nagelkerken I, Kruitwagen G, Van Der Velde G, Mgaya YD. 2007. Relative importance of mangroves as feeding habitats for fishes: a comparison between mangrove habitats with different settings. Bulletin of Marine Science 80:497–512.
McDevitt-Irwin JM, Iacarella JC, Baum JK. 2016. Reassessing the nursery role of seagrass habitats from temperate to tropical regions: a meta-analysis. Marine Ecology Progress Series 557:133–43.
Morton JK, Gladstone W. 2014. Changes in rocky reef fish assemblages throughout an estuary with a restricted inlet. Hydrobiologia 724:235–53.
Munsch SH, Cordell JR, Toft JD. 2016. Fine-scale habitat use and behavior of a nearshore fish community: nursery functions, predation avoidance, and spatiotemporal habitat partitioning. Marine Ecology Progress Series 557:1–15.
Murphy HM, Jenkins GP. 2010. Observational methods used in marine spatial monitoring of fishes and associated habitats: a review. Marine and Freshwater Research 61:236–52.
Nagelkerken I, Van Der Velde G, Gorissen MW, Meijer GJ, Van’t Hof T, Den Hartog C. 2000. Importance of mangroves, seagrass beds and the shallow coral reef as a nursery for important coral reef fishes, using a visual census technique. Estuarine, Coastal and Shelf Science 51:31–44.
Nagelkerken I, Blaber SJM, Bouillon S, Green P, Haywood M, Kirton LG, Meynecke JO, Pawlik J, Penrose HM, Sasekumar A, Somerfield PJ. 2008. The habitat function of mangroves for terrestrial and marine fauna: a review. Aquatic Botany 89:155–85.
Nagelkerken I, Sheaves M, Baker R, Connolly RM. 2015. The seascape nursery: a novel spatial approach to identify and manage nurseries for coastal marine fauna. Fish and Fisheries 16:362–71.
Nagelkerken I. 2009. Evaluation of nursery function of mangroves and seagrass beds for tropical decapods and reef fishes: Patterns and underlying mechanisms. Ecological Connectivity among Tropical Coastal Ecosystems, pp 357–99.
Olds AD, Connolly RM, Pitt KA, Pittman SJ, Maxwell PS, Huijbers CM, Moore BR, Albert S, Rissik D, Babcock RC, Schlacher TA. 2016. Quantifying the conservation value of seascape connectivity: a global synthesis. Global Ecology and Biogeography 25:3–15.
Parsons DM, MacDonald I, Buckthought D, Middleton C. 2018. Do nursery habitats provide shelter from flow for juvenile fish? PLoS One 13:e0186889.
Pavlov DS, Mikheev VN, Lupandin AI, Skorobogatov MA. 2008. Ecological and behavioural influences on juvenile fish migrations in regulated rivers: a review of experimental and field studies. Hydrobiologia 609:125.
Pirtle JL, Shotwell SK, Zimmermann M, Reid JA, Golden N. 2017. Habitat suitability models for groundfish in the Gulf of Alaska. Deep-Sea Research Part II: Topical Studies in Oceanography 165:303–21.
Pittman SJ, Brown KA. 2011. Multi-scale approach for predicting fish species distributions across coral reef seascapes. PLoS One 6:e20583.
Pittman SJ, Caldow C, Hile SD, Monaco ME. 2007a. Using seascape types to explain the spatial patterns of fish in the mangroves of SW Puerto Rico. Marine Ecology Progress Series 348:273–84.
Pittman SJ, Christensen JD, Caldow C, Menza C, Monaco ME. 2007b. Predictive mapping of fish species richness across shallow-water seascapes in the Caribbean. Ecological Modelling 204:9–21.
Pittman SJ. 2018. Introducing seascape ecology. In: Pittman SJ, Ed. seascape ecology, . Malaysia: WILEY Blackwell.
Pygas DR, Ferrari R, Figueira WF. 2020. Review and meta-analysis of the importance of remotely sensed habitat structural complexity in marine ecology. Estuarine, Coastal and Shelf Science 235:106468.
Raubenheimer D, Zemke-White WL, Phillips RJ, Clements KD. 2005. Algal macronutrients and food selection by the omnivorous fish girella tricuspidata. Ecology 86:2601–610.
Rees MJ, Jordan A, Price OF, Coleman MA, Davis AR. 2014. Abiotic surrogates for temperate rocky reef biodiversity: implications for marine protected areas. Diversity and Distributions 20:284–96.
Rees MJ, Knott NA, Neilson J, Linklater M, Osterloh I, Jordan A, Davis AR. 2018. Accounting for habitat structural complexity improves the assessment of performance in no-take marine reserves. Biological Conservation 224:100–10.
Rex MA, Etter RJ, Morris JS, Crouse J, McClain CR, Johnson NA, Stuart CT, Deming JW, Thies R, Avery R. 2006. Global bathymetric patterns of standing stock and body size in the deep-sea benthos. Marine Ecology Progress Series 317:1–8.
Rochette S, Rivot E, Morin J, Mackinson S, Riou P, Le Pape O. 2010. Effect of nursery habitat degradation on flatfish population: Application to Solea solea in the Eastern Channel (Western Europe). Journal of Sea Research 64:34–44.
Rotherham D, Chapman MG, Underwood AJ, Gray CA, Johnson DD. 2011. Untangling spatial and temporal variation in abundances of estuarine fish sampled with multi-mesh gillnets. Marine Ecology Progress Series 435:183–95.
Rypel AL, Layman CA, Arrington DA. 2007. Water depth modifies relative predation risk for a motile fish taxon in Bahamian tidal creeks. Estuaries and Coasts 30:518–25.
Sabatés A, Olivar MP, Salat J, Palomera I, Alemany F. 2007. Physical and biological processes controlling the distribution of fish larvae in the NW Mediterranean. Progress in Oceanography 74:355–76.
Salini JP, Blaber SJM, Brewer DT. 1994. Diets of trawled predatory fish of the Gulf of Carpentaria, Australia, with particular reference to predation on prawns. Australian Journal of Marine and Freshwater Research 45:397–411.
Schlacher TA, Williams A, Althaus F, Schlacher-Hoenlinger MA. 2010. High-resolution seabed imagery as a tool for biodiversity conservation planning on continental margins. Marine Ecology 31:200–21.
Sheaves M. 2005. Nature and consequences of biological connectivity in mangrove systems. Marine Ecology Progress Series 302:293–305.
Sheaves M. 2009. Consequences of ecological connectivity: the coastal ecosystem mosaic. Marine Ecology Progress Series 391:107–15.
Sheaves M, Baker R, Nagelkerken I, Connolly RM. 2015. True value of estuarine and coastal nurseries for fish: incorporating complexity and dynamics. Estuaries and Coasts 38:401–14.
Sheaves M, Johnston R, Baker R. 2016. Use of mangroves by fish: new insights from in-forest videos. Marine Ecology Progress Series 549:167–82.
Simonson WD, Allen HD, Coomes DA. 2014. Applications of airborne lidar for the assessment of animal species diversity. Methods in Ecology and Evolution 5:719–29.
Stamoulis KA, Delevaux JMS, Williams ID, Poti M, Lecky J, Costa B, Kendall MS, Pittman SJ, Donovan MK, Wedding LM, Friedlander AM. 2018. Seascape models reveal places to focus coastal fisheries management. Ecological Applications 28:910–25.
Swadling DS, Knott NA, Rees MJ, Davis AR. 2019. Temperate zone coastal seascapes: seascape patterning and adjacent seagrass habitat shape the distribution of rocky reef fish assemblages. Landscape Ecology 34:2337–352.
Tews J, Brose U, Grimm V, Tielbörger K, Wichmann MC, Schwager M, Jeltsch F. 2004. Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. Journal of Biogeography 31:79–92.
Unsworth RKF, Garrard SL, De León PS, Cullen LC, Smith DJ, Sloman KA, Bell JJ. 2009. Structuring of Indo-Pacific fish assemblages along the mangrove-seagrass continuum. Aquatic Biology 5:85–95.
Walbridge S, Slocum N, Pobuda M, Wright JD. 2018. Unified geomorphological analysis workflows with Benthic Terrain Modeler. Geosciences 8:94–117.
Wang Y, Naumann U, Wright ST, Warton DI. 2012. mvabund– an R package for model-based analysis of multivariate abundance data. Methods in Ecology and Evolution 3:471–74.
Wedding LM, Friedlander AM, McGranaghan M, Yost RS, Monaco ME. 2008. Using bathymetric lidar to define nearshore benthic habitat complexity: Implications for management of reef fish assemblages in Hawaii. Remote Sensing of Environment 112:4159–165.
Whitfield AK. 2017. The role of seagrass meadows, mangrove forests, salt marshes and reed beds as nursery areas and food sources for fishes in estuaries. Reviews in Fish Biology and Fisheries 27:75–110.
Whitfield AK, Pattrick P. 2015. Habitat type and nursery function for coastal marine fish species, with emphasis on the Eastern Cape region, South Africa. Estuarine, Coastal and Shelf Science 160:49–59.
Wilson GG. 1998. A description of the early juvenile colour patterns of eleven Lethrinus species (Pisces: Lethrinidae) from the Great Barrier Reef. Australia. 50:55–83.
Young MA, Iampietro PJ, Kvitek RG, Garza CD. 2010. Multivariate bathymetry-derived generalized linear model accurately predicts rockfish distribution on Cordell Bank, California, USA. Marine Ecology Progress Series 415:247–61.
Acknowledgements
This research was funded by the Queensland Department of Agriculture and Fisheries (DAF1498CQ8-3), Healthy Land and Water and the Sea World Research and Rescue Foundation. The authors would like to thank Olivia Kimber, Jesse Mosman, Felicity Osborne, Sarah Thackwray and Nicholas Yabsley for assistance in the field and laboratory.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions: HB, BLG and AO conceived the idea; BLG, RC, MS and AO resourced the project; HB, BLG, CH, BG, NO and AO collected the data; HB, BLG, AR and AO analysed the data; and HB, BLG, CH, RC, BG, NO, AR, IN, SP, MS and AO wrote and drafted the manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visithttp://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Borland, H.P., Gilby, B.L., Henderson, C.J. et al. Seafloor Terrain Shapes the Three-dimensional Nursery Value of Mangrove and Seagrass Habitats. Ecosystems 26, 442–456 (2023). https://doi.org/10.1007/s10021-022-00767-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-022-00767-4