Abstract
Global change models predict that high-latitude boreal forests will become increasingly susceptible to fire activity as climate warms, possibly causing a positive feedback to warming through fire-driven emissions of CO2 into the atmosphere. However, fire-climate feedbacks depend on forest regrowth and carbon (C) accumulation over the post-fire successional interval, which is influenced by nitrogen (N) availability. To improve our understanding of post-fire C and N accumulation patterns in boreal forests, we evaluated above- and belowground C and N pools within 70 stands throughout interior Alaska, a region predicted to undergo a shift in canopy dominance as fire severity increases. Stands represented gradients in age and successional trajectory, from black spruce (Picea mariana) self-replacement to species replacement by deciduous species of trembling aspen (Populus tremuloides) and Alaska paper birch (Betula neoalaskana). Stands undergoing deciduous trajectories stored proportionally more of their C and N in aboveground stemwood and had 5–7 times faster rates of aboveground net primary productivity of trees compared to stands undergoing a black spruce trajectory, which stored more of their C and N in the soil organic layer (SOL), a thick layer of mostly undecomposed mosses. Thus, as successional trajectories shift, total C and N pool sizes will remain relatively unchanged, but there will be a trade-off in pool location and a potential increase in C and N longevity due to decreased flammability and decomposition rates of deciduous stemwood. Despite often warmer, drier conditions in deciduous compared to black spruce stands, deciduous stemwood has a C:N around 10 times higher than the black spruce SOL and often remains standing for many years with reduced exposure to fungal decomposers. Thus, a fire-driven shift in successional trajectories could cause a negative feedback to climate warming because of increased pool longevity in deciduous trajectories.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the last 30 years, fire activity has increased in boreal forests in conjunction with climate warming and drying (Soja and others 2007; Balshi and others 2009; Flannigan and others 2009) to a level that has been unprecedented in the last 10,000 years (Kelly and others 2013). Because boreal forests store a large proportion of global terrestrial carbon (C) stocks (McGuire and others 2009; Pan and others 2011), increased fire consumption of vegetation and organic soils and subsequent release of CO2 into the atmosphere are predicted to cause a positive feedback to climate warming (Randerson and others 2006; Bradshaw and others 2009), especially if increased burning mines previously protected “old” C in this huge pool. Although boreal forests have historically been considered C sinks (Pan and others 2011), increased fire could shift the biome to a net C source over multiple disturbance cycles (Bond-Lamberty and others 2007; Turetsky and others 2011). Increased fire activity is also expected to volatilize nitrogen (N) (Harden and others 2002; Neff and others 2005; Boby and others 2010) in this already N-limited ecosystem (Van Cleve and Alexander 1981) because most N is stored in organic soils which are often greater than 50% consumed by fire (Kasischke and others 1995). Reduced N availability may limit forest productivity and regeneration and thus, C accumulation and storage during the post-fire interval (Harden and others 2000).
Changes in the fire regime may also alter patterns of C and N accumulation by triggering patterns of forest regrowth that differ from the pre-fire stand. For example, increased fire frequency and extent can reduce seed availability (Romme and others 1998; Johnstone and Chapin 2006b), while increased fire severity can increase seed germination (Johnstone and Chapin 2006a). These changes in initial establishment parameters, combined with species-specific growth habits and competitive abilities, can alter stand composition and structure, and ultimately the rate and amount of C and N stored during post-fire succession (Weber and Flannigan 1997). Thus, the balance between fire-driven losses of C and N and their re-accumulation rates will determine whether increased fire activity creates a positive, neutral, or negative feedback to the climate system.
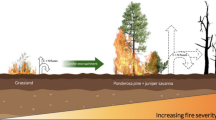
In boreal forests of interior Alaska, recent studies have shown that high severity fires that leave behind a shallow soil organic layer (SOL) favor germination and seedling establishment of deciduous species like trembling aspen (Populus tremuloides Michx.) and Alaska paper birch (Betula neoalaskana Sarg.) over that of black spruce (Picea mariana Mill B.S.P.), an evergreen conifer (Johnstone and others 2010a, b). Light-seeded deciduous species proliferate on high-quality mineral soil seedbeds and are able to outcompete slower-growing black spruce (Johnstone and Kasischke 2005). Because initial patterns of seedling recruitment tend to predict future stand development (Johnstone and others 2004; Johnstone and others 2010a, b), many researchers predict that increased fire severity and consumption of the SOL will trigger a canopy shift from black spruce to deciduous forests, especially on moderate to well-drained sites (for example, Johnstone and others 2010a). This shift could have numerous consequences for ecosystem function because of species-specific effects on productivity, albedo, evapotranspiration, flammability, and wildlife habitat (Chapin and others 2008; Huntington and others 2012).
Our primary objective was to quantitatively evaluate the contribution of two drivers, time after fire and successional trajectory, to the development of above- and belowground C and N pools during post-fire succession. We hypothesized that aboveground C and N pools would increase with time after fire in stands undergoing both black spruce and deciduous (trembling aspen or Alaska paper birch) successional trajectories, but that the rate of increase would be substantially higher in stands proceeding along deciduous trajectories (Figure 1A) because of substantially more stemwood stored in deciduous trees. In contrast, we hypothesized that soil C and N pools would increase fastest in black spruce successional trajectories (Figure 1B) because the cool, moist conditions, dominate moss understory, and slow decomposition rates associated with these stands (Cleve and others 1983a; Viereck and others 1983) would promote the development of a thick SOL horizon. Overall, we predicted that increased stemwood contribution to C and N pools in deciduous trajectories and increased SOL contribution in black spruce trajectories would cause an increase in total C:N ratios with time after fire, but the magnitude of this increase would be greatest in deciduous trajectories (Figure 1C) because of a higher C:N of stemwood. Ultimately, this change in location (above- vs. belowground) of C and N pools with shifting successional trajectory would lead to a similar change in total C and N pools with time after fire (Figure 1D). Understanding how boreal forest C and N pools respond to climate-driven alterations to the fire regime will help us predict the fate of these forests under novel ecological conditions.
Methods
Study Area
This study took place within an 800-km2 area of interior Alaska. This region is bounded to the north by the Brooks Range and altitudinal treeline (67°N), to the south by the Alaskan Range (63°N), to the west by the Dalton Highway (150°W), and to the east by the Alaska/Canadian border (142°W), and includes the Yukon, Tanana, and Kuskokwim River valleys (Hulten 1968). Climate is continental, with long, cold winters (−23°C in January) and warm, dry summers (17°C in July) (Alaska Climate Research Center, http://climate.gi.alaska.edu/). Nearly half (47%) of annual precipitation (286 mm) occurs during the growing season (June–August) and about 35% falls as snow (Hinzman and others 2005), which covers the ground 6–9 months per year (Slaughter and Viereck 1986). The region is underlain by discontinuous permafrost (75–80%) except along floodplains and on south-facing slopes (Osterkamp and Romanovsky 1999). Soils range from poorly drained Gelisols to well-drained, permafrost-free Inceptisols (Dyrness 1982). The continental climate also proves conducive to frequent wildfires. Fire return interval in interior Alaska was about 196 years from 1950 to 2000 and dropped to around 144 years during the 2000s (Kasischke and others 2010). The region is dominated by forests of black spruce, an evergreen, semiserotinous conifer, intermixed with local patches of deciduous trees, including trembling aspen and Alaska paper birch. Tall shrubs, including willows (Salix spp.) and alder (Alnus spp.), occur frequently across all stand types (Alexander and others 2012). Mosses, including feathermosses and Sphagnum spp., often dominate the understory of mature black spruce stands (Viereck and others 1983; Turetsky 2003), whereas heavy leaf litter tends to limit moss cover in deciduous stands (Chapin and others 2006a).
Site Selection
In summers 2008–2011, we surveyed 70 stands varying in age and trajectory for above- and belowground C and N pools. Intermediate-aged stands (20–59 years old) undergoing different trajectories were selected using a combination of fire scar maps (Alaska Geospatial Data Clearinghouse; http://agdc.usgs.gov/data/blm/fire/), satellite imagery, and field reconnaissance. We aimed to sample stands ranging from black spruce to deciduous-dominated within each fire scar, and we only sampled stands that could have originated following fire from a previously black spruce-dominated stand (that is, signs of fire-killed black spruce, regenerating black spruce in the understory, and not on southeast facing slopes, where black spruce typically does not grow). We did not sample poorly drained black spruce stands because they have a longer burn return interval and rarely transition to other tree species. Further details regarding selection criteria of intermediate-aged stands can be found in Alexander and others (2012). We also sampled mature black spruce (sites described in Hollingsworth and others 2006) and deciduous stands (often paired with a nearby mature black spruce stand) using the same selection criteria. To determine stand age of mature stands, a wood core was obtained 20-cm from the base of 10 trees sampled randomly within the center of each stand. Wood samples were dried at 60°C and sanded sequentially with finer grit sizes to obtain a smooth surface. Samples were scanned at high resolution and analyzed for ring count using WinDendro (Regent Instruments, Canada). Stand age was determined to be the average of the 10 trees.
Aboveground C and N Pools and Aboveground Net Primary Productivity (ANPP)
Stand inventories and allometric equations were used to produce estimates of aboveground biomass and net primary productivity (ANPP) of trees and large shrubs (Salix and Alnus spp.). Within each stand, we established a 100-m-long transect, subdivided into five 20-m-long subsections. Along a 10 m portion of each 20 m subsection (note: a few stands in 2008 were measured along a 5-m long section), we measured the diameter at breast height (DBH; 1.4 m tall) or basal diameter (BD; for stems <1.4 m tall) of all live and dead (snags) trees and large shrubs falling within 1 m to either side of the transect. This generated five replicate sub-plots within each stand. Inventories were converted to live or dead biomass and ANPP (crown, stemwood, and total) using allometric equations and annual increment growth (Alexander and others 2012). Snag biomass was computed as the difference between total live biomass and live crown biomass. Snag biomass for alder was estimated directly from allometric equations created from dead shrub raw harvest data.
To convert biomass to C and N pools, %C and %N of foliage and twigs from live trees were determined for five randomly selected trees of each dominant species within each stand using an Elemental Analyzer (Costech Analytical Technologies, Inc., Valencia, CA). Samples were collected from the tops of trees either directly by hand, cutting trees down, or using a slingshot. Prior to analyses, all foliage samples were separated from twigs, cleaned of debris, and ground to a homogenized powder. Twigs were finely chopped by hand, pooled by species for each stand, and ground to a homogenized powder. Stemwood C and N were determined by finely chopping then grinding a thin section of bole (from center to bark) from five representative samples of each dominant tree species. Snag C and N were calculated using %C of 50% and %N of 0.12% based on mean cross-species wood/bark values from a subset of trees collected across the study area.
Belowground C and N Pools
To estimate soil C pools, we sampled leaf litter, soil organic layer (SOL), and upper mineral soil (10-cm depth) at 20 m intervals along each 100 m transect (n = 5) (see Alexander and others 2012 for description of transects). When the SOL was thin (<10 cm), SOL and mineral soils were sampled using a 7.5-cm diameter metal soil corer. For thicker SOLs, samples were obtained by cutting a ‘brownie’ of known dimensions using a soil saw. Leaf litter was collected beneath a 20 × 20 cm quadrat at the same locations. Samples were transported from the field in coolers and stored frozen at the University of Alaska-Fairbanks until the end of the field season when they were shipped frozen back to the University of Florida for processing and analysis.
Following homogenization and removal of coarse materials (>2 mm), subsamples were dried at 60°C (leaf litter/organic + fine roots) or 105°C (mineral soil) for 48 h to determine bulk density and soil moisture. A subsample of each horizon was analyzed with an Elemental Analyzer to determine %C and %N. Total C and N contained within leaf litter, organic, and mineral horizons were calculated as the product of each horizon’s depth, bulk density, and C and N content. Coarse fraction (roots, charcoal, buried wood) C and N pools for organic and mineral soils were based on coarse material mass values and 50%C and 0.25%N. Occasionally, mineral soil samples could not be obtained to 10-cm depth because we hit rock or permafrost. In these instances, C and N pools were computed based on a mean %C and %N and bulk density obtained from other mineral soil samples in the same stand, or if less than one sample was available from that stand, a mean %C and %N and bulk density was obtained from across all stands.
Data Analyses and Statistics
To better understand how successional trajectory influences C and N pools, we subdivided stands into trajectory types (trembling aspen, Alaska paper birch, or black spruce) based on the relative contribution of each species to total stand biomass. Species occurring infrequently (L. laricina, P. balsamifera L., and P. glauca (Moench) Voss) were grouped into a single ‘other’ category, and willows and alders were grouped into a ‘tall shrub’ category. Any species constituting at least 20% of stand biomass was considered to be a dominant stand component. For 48 of 64 stands, black spruce (n = 20), aspen (n = 16), Alaska paper birch (n = 11), or shrubs (n = 1) clearly dominated, having greater than 66% of stand biomass. The other stands grouped into black spruce/shrub (n = 4), black spruce/aspen (n = 3), black spruce/aspen/shrub (n = 3), black spruce/Alaska paper birch (n = 6), and black spruce/Alaska paper birch/shrub (n = 2). The single shrub stand was removed from our analyses, and the stands containing a mix of shrubs and trees were classified as the successional trajectory which contained the greatest biomass.
We first used linear regression (JMP v 11.0) to explore patterns in our data, including aboveground and soil C and N pools and C:N ratios as a function of ‘years since fire’ and soil C and N pools as a function of organic layer depth, bulk density, and %C and %N. Regression lines are included on figures when significant. For response variables potentially having curvilinear trends, we also fit quadratic and exponential models and then compared AICc values from these models to the linear model. In no instance did curvilinear models provide a better fit (ΔAICc < 5). Thus, we always report the more parsimonious linear model.
To improve our understanding of how aboveground and soil C and N pools and C:N ratios vary with ‘years since fire’ among trajectory types, we tested for an interaction effect of trajectory type and ‘years since fire,’ and when significant (P < 0.05), performed Fisher’s post hoc multiple comparison tests to determine differences among slopes (JMP v 11.0). For pools and C:N ratios where there was a significant effect of trajectory but no significant effect of ‘years since fire’ or trajectory * ‘years since fire’ interaction, differences in trajectories were determined using ANOVA. F values and P values were computed based on Type III sums of square. There were no significant effects of any predictor variable on mineral soil C and N pools or C:N ratio; thus, these data are presented only in Online Appendix A.
We tested for significant differences (P < 0.05) in C and N pools, C:N ratio, and ANPP among successional trajectories of similar ages using an ANOVA (JMP v 11.0). To do this, we grouped trajectory types into three age categories: (1) 20–39 ‘years since fire,’ (2) 40–59 ‘years since fire,’ and (3) 60–100 ‘years since fire.’ For these comparisons, we do not report data from stands greater than 100 ‘years since fire’ because of insufficient replication (n = 2, black spruce; n = 1, aspen; n = 0, birch). All response variables were tested for normality and homogeneity of variance prior to analyses and were transformed using square-root or log + 1 transformations when they did not meet these underlying assumptions. We report values for upper (0–10 cm depth) mineral soil horizons in these tables but do not include these values in soil or total C and N pools as there were no differences in mineral soil pools and inclusion in the total pools skew trends driven by differences in organic layer horizons. We also highlight marginally significant (P < 0.10) differences because of the reduced sample size when grouping by ‘years since fire’ categories.
Uni- and multivariate regression models were fit to the data to determine how much of the variation in aboveground and soil C and N pools and C:N ratio could be explained by deciduous importance value (IV) and ‘years since fire’ (JMP v 11.0). To determine the deciduous IV of each stand, we grouped tree and large shrub (Salix spp. and Alnus spp.) species by their leaf phenology (evergreen vs. deciduous) and then calculated an IV of the deciduous component of each stand based on stand inventory data, where IV = relative density + relative basal area + relative frequency. We generated linear models containing each of the explanatory variables independently and in combination. Since our original study design was to sample several forest stands of varying deciduous dominance within the same fire scar, we also modeled our data using ‘fire scar’ as a random effect. However, this did not improve our models, so was left out of future analyses. We selected the ‘best-fit’ model for each dependent variable based on Akaike Information Criteria (AIC) values corrected for small sample sizes (AICc).
Results
Aboveground and Belowground C
Aboveground C pools (live trees + snags) exhibited a significant (P < 0.01 for all) linear increase with ‘years since fire’ in all three successional trajectories (Figure 2A; Table 1), but the rate of increase was 6–8 times higher in stands undergoing trembling aspen and Alaska paper birch trajectories compared to those undergoing black spruce trajectories (P < 0.0001 for both comparisons; Table 2). The rate of increase was similar for the two deciduous trajectories (P = 0.318; Table 2). Crown, stemwood, and total ANPP were consistently higher in deciduous trajectories compared to black spruce, and by 60–100 ‘years since fire,’ total ANPP was 5–7 times greater in deciduous stands (Table 3). Differences in aboveground C pools between successional trajectories were evident in all age categories and were mostly driven by differences in components of live tree biomass, exclusive of foliage, which was always similar among trajectory types (Table 4). By 60–100 ‘years since fire,’ aboveground C pools were 6 times higher in trembling aspen (6430 ± 636 g C m−2) and Alaska paper birch (6324 ± 1072 g C m−2) trajectories compared to stands proceeding along black spruce trajectories (1006 ± 325 g C m−2). When uni- and multivariate models using deciduous IV and ‘years since fire’ as independent variables were fit to aboveground C pools, both univariate variables predicted a significant and positive linear relation with aboveground C pools, but a multivariate model that included these two predictor variables and their interaction provided a better fit based on AICc values (Table 5).
Changes in (A) aboveground, (B) belowground, and (C) total carbon pools with ‘years since fire’ in stands undergoing trembling aspen, Alaska paper birch, and black spruce successional trajectories within interior Alaska. Regression lines are shown for pools exhibiting a significant (P < 0.05) linear trend with ‘years since fire’.
Belowground C pools (leaf litter + organic matter (OM) horizons + downed woody debris) exhibited a significant linear increase (P < 0.0001) with ‘years since fire’ in stands undergoing black spruce successional trajectories, but a significant linear decrease (P = 0.045) and no obvious temporal trend (P = 0.500) in stands undergoing trembling aspen and Alaska paper birch trajectories, respectively (Figure 2B). The rate of increase in belowground C pools in black spruce trajectories was about 3 times faster than the rate of increase of aboveground C pools of that trajectory. Differences in belowground C pools within black spruce stands were driven by increased depth of the soil organic layer, which became more developed with ‘years since fire’ (Table 4; Online Appendix 2). Stands undergoing Alaska paper birch trajectories had relatively high amounts of C stored in shallow (0–5 cm) OM pools, but with little change over time. By 60–100 years since fire, total soil C pools were about 2 and 5 times higher in black spruce trajectories (7395 ± 558 g C m−2) compared to Alaska paper birch (3970 ± 703 g C m−2) and trembling aspen trajectories (1519 ± 305 g C m−2) (Table 4). A univariate model with deciduous IV as the independent variable predicted a significant (P = 0.01; R 2 = 0.19) negative linear correlation with belowground C pools, while belowground pools exhibited a significant increase with ‘years since fire’ (P = 0.0003; R 2 = 0.10) (Table 5). A multivariate model including both independent variables and their interaction also provided a significant fit (P < 0.0001; R 2 = 0.30) but no better fit than the univariate model with only deciduous IV (ΔAICc < 5).
Soil organic layer depth and bulk density, but not soil %C, influenced SOL C pools (Figure 3). For all trajectory types, soil organic layer C pools increased with increasing depth of the organic horizon (P < 0.0001 for all regressions; R 2 = 0.51, 0.62, and 0.55, for aspen, birch, and spruce, respectively) (Figure 3A). Soil C pools in trembling aspen (P < 0.0001; R 2 = 0.33) and Alaska paper birch trajectories (P = 0.0031; R 2 = 0.09) also increased linearly with soil bulk density, but not in black spruce trajectories (P = 0.09; R 2 = 0.02; Figure 3B). Organic soil C pools did not exhibit any obvious patterns in relation to %C (Fig. 3C).
Total soil organic layer (SOL) carbon and nitrogen pools for all soil cores obtained within trembling aspen, Alaska paper birch, and black spruce trajectory types within interior Alaska as a function of (A, D) organic layer depth, (B, E) bulk density, and (C, F) % carbon and % nitrogen. Regression lines are shown for relationships exhibiting a significant (P < 0.05) linear trend.
Total C pools (above- + belowground) increased significantly with ‘years since fire’ across all trajectories types (Figure 2C; Table 1), which did not differ significantly (P = 0.430) in their rate of increase (Table 2). Two mature (70 and 87 ‘years since fire’) Alaska paper birch stands had substantially higher aboveground C pools than other stands because of high contribution of snags (~ 2000 g C m−2) and development of thick SOL (9- and 15-cm depth). Despite a trade-off in location of C pools among trajectory types, there was no significant difference in total C pools among trajectory types during any of the ‘years since fire’ categories, and by 60–100 ‘years since fire,’ all trajectories stored approximately 8000–10,000 g C m−2 in live trees, snags, leaf litter, SOL, and woody debris (Table 4). A univariate model with ‘years since fire’ as the independent variable was best able to predict total C pools (P < 0.0001; R 2 = 0.40; Table 5).
Aboveground and Belowground N
There was a significant linear increase in aboveground N pools with ‘years since fire’ for all successional trajectories (Figure 4A; Table 1). Similar to aboveground C pools, the rate of increase was 7–10 times faster in trembling aspen and Alaska paper birch trajectories than in black spruce trajectories (Table 2). There was no significant difference (P = 0.300) in the rate of increase between the two deciduous trajectories (Table 2). Differences in aboveground N pools between deciduous and black spruce trajectories were evident in all ‘years since fire’ categories but were most pronounced 60–100 ‘years since fire,’ when there was around 6 times less aboveground N in black spruce trajectories (6 ± 2 g N m−2) than in trembling aspen (38 ± 3 g N m−2) and Alaska paper birch (40 ± 7 g N m−2) trajectories (Table 6). A multivariate model including deciduous IV, ‘years since fire,’ and their interaction as independent variables provided a substantially better fit (P < 0.0001; R 2 = 0.54; ΔAICc > 5) of aboveground N pools compared to univariate models using just deciduous IV or ‘years since fire’ (Table 5).
Changes in (A) aboveground, (B) belowground, and (C) total nitrogen pools with ‘years since fire’ in stands undergoing trembling aspen, Alaska paper birch, and black spruce successional trajectories within interior Alaska. Regression lines are shown for pools exhibiting a significant (P < 0.05) linear trend with ‘years since fire.’ For (D) belowground and (E) total nitrogen pools, bar graphs of mean values across successional trajectories (A = aspen, B = birch, S = spruce) are shown because there was a significant effect of trajectory (denoted by different lowercase letters) but no significant effect of ‘years since fire’ or trajectory *‘years since fire’ interaction.
There was no effect of ‘years since fire’ on belowground N pools within any successional trajectory (Figure 4B; Table 1). However, there was a significant (P < 0.0001) effect of successional trajectory. Averaged across all age classes, black spruce (158 ± 14 g N m−2) and Alaska paper birch (145 ± 17 g N m−2) trajectories stored approximately 40% more N in belowground pools than trembling aspen (88 ± 10 g N m−2) trajectories (Figure 4D). These trends were marginally significant (P < 0.10) when compared within ‘years since fire’ categories for stands 20–39 and 60–100 ‘years since fire’ (Table 6) and appear to be a function of a deep SOL in the black spruce trajectory and a combination of a moderately deep SOL and higher SOL %N in the Alaska paper birch trajectory (Online Appendix 2). Similar to belowground C pools, belowground N pools increased with the increasing SOL depth in all trajectories and with bulk density in deciduous trajectories, but not with soil %N (Figure 3F). No univariate or multivariate model with deciduous IV, ‘years since fire,’ and their interaction provided a significant fit for belowground N pools (Table 5).
There was no significant effect of ‘years since fire’ on total N pools (Figure 4C; Table 1), but there was a significant effect of trajectory type (P = 0.004) when averaged across all age classes (Figure 4E; Table 2). Similar to belowground N pools, total N pools were significantly higher (~40%) in black spruce (164 ± 13 g N m−2) and Alaska paper birch (173 ± 19 g N m−2) trajectories compared to trembling aspen (102 ± 9 g N m−2). When considered within individual categories of ‘years since fire,’ these trends were marginally significant for 20–39 ‘years since fire,’ and there were no significant differences in total N pools for other ‘years since fire’ categories (Table 6). No univariate or multivariate model with deciduous IV, ‘years since fire,’ and their interaction provided a significant fit for total N pools (Table 5).
C:N
Trembling aspen trajectories exhibited a significant linear increase in aboveground (Figure 5A) and total (Figure 5C) C:N ratio with ‘years since fire’ (P = 0.049 and 0.002, R 2 = 0.20 and 0.44, respectively), but no temporal trends were observed for Alaska paper birch or black spruce trajectories for aboveground or total C:N or for belowground C:N (Figure 5B) for any trajectory (Table 1). There was a significant trajectory (P = 0.044) effect on belowground C:N when averaged over ‘years since fire,’ where the C:N ratio was higher in trembling aspen (38 ± 2) and black spruce (37 ± 2) trajectories than Alaska paper birch (30 ± 1) (Figure 5D; Table 2). These belowground trends were also evident in stands 60–100 ‘years since fire’ (Table 7). Overall, by 60–100 ‘years since fire,’ total C:N was about twice as high in trembling aspen trajectories (104 ± 21) than black spruce (58 ± 6) and Alaska paper birch (47 ± 9) trajectories (Table 7).
Changes in (A) aboveground, (B) belowground, and (C) total C:N ratio with ‘years since fire’ in stands undergoing trembling aspen, Alaska paper birch, and black spruce successional trajectories within interior Alaska. Regression lines are shown for pools exhibiting a significant (P < 0.05) linear trend with ‘years since fire.’ For belowground C:N ratio, a bar graph of mean values across successional trajectories (A = aspen, B = birch, S = spruce) is shown because there was a significant effect of trajectory (denoted by different lowercase letters) but no significant effect of ‘years since fire’ or trajectory*‘years since fire’ interaction
A univariate model with deciduous IV as the independent variable provided the best-fit model for aboveground C:N (Table 5), which tended to decrease with increasing deciduous IV. A multivariate model with deciduous IV, ‘years since fire,’ and their interaction as independent variables produced a slightly higher R 2 value (0.20), but the AICc values were similar between the two models. Belowground C:N, which decreased with deciduous IV, ‘years since fire,’ and their interaction, was best predicted by a multivariate model with all three independent variables. A multivariate model with deciduous IV and ‘years since fire’ was the best fit for total C:N, which increased with both independent variables.
Discussion
As hypothesized (Figure 1A), aboveground C and N pools exhibited a linear increase with time after fire in all successional trajectories, but the rate of increase was substantially faster in deciduous trajectories, which had 5–7 times faster rates of tree ANPP compared to black spruce trajectories and accumulated 70–80% of their aboveground biomass in stemwood. These findings are comparable to the few other studies in interior Alaska reporting aboveground C and N pools and tree ANPP for these stand types (Van Cleve and others 1983a; Mack and others 2008). Deciduous species annually shed relatively high-quality (that is, low C:N) leaf litter that decomposes faster than the low-quality, more recalcitrant litter produced in black spruce stands, which is typically moss litter mixed with spruce needles (Van Cleve and Alexander 1981; Van Cleve and others 1983b). More and regular inputs of high-quality leaf litter in deciduous stands may equate to higher nutrient availability (Jerabkova and others 2006) and a physical and/or chemical restriction of moss growth (Oechel and Van Cleve 1986; Natalia and others 2008), reducing soil insulation and increasing soil temperature (Légaré and others 2005). Warm soils with high nutrient availability can foster tree growth and accumulation of aboveground C and N pools, especially in stemwood (Légaré and others 2004). Thus, properties of the trees themselves (that is, relatively low C:N litterfall and high soil N in deciduous stands and the opposite in spruce stands) act to reinforce tree ANPP and standing biomass as the stand ages (Gower and others 2000). Because aboveground C and N pools behaved similar for both deciduous trajectories and clearly exhibited distinct differences from the black spruce trajectory over time, we were able to predict reasonably well changes in both aboveground C and N pools using a multivariate linear model with ‘deciduousness,’ years since fire, and the interaction of these predictors.
In contrast to our expectation of increasing pools of both belowground C and N with years since fire across all trajectories (Figure 1B), only belowground C pools in stands proceeding along a black spruce trajectory increased over time. This trend was driven by the development of a deep SOL (often >20 cm depth) such that by 60–100 years since fire, the SOL represented more than 80% of total C pools in the black spruce trajectory compared to around 20 and 40%, respectively, in aspen and birch trajectories. Belowground C pools in the black spruce trajectory increased three times faster (~37 g C m−2 y−1) than that of aboveground C pools (~13 g C m−2 y−1), equating to a belowground C sequestration rate of 5.4 Tg C y−1 across the upland black spruce zone of interior Alaska (14.6 million ha), a rate similar to that reported by Turetsky and others (2011) for the entire black spruce zone (5.5 Tg C y−1). The accumulation of a deep SOL over the successional interval in a black spruce trajectory likely stems from a combination of relatively high moss productivity in mature stands (20–30 g m−2 y−1; Mack and others 2008) and aggradation of dead moss on the soil surface (Harden and others 2000) associated with slow decomposition rates in cold, moist conditions’ characteristic of black spruce stands (Hobbie and others 2000). In contrast, belowground C pools decreased in the trembling aspen trajectory and remained relatively unchanged in the Alaska paper birch trajectory. Deep forest floor horizons are uncommon in deciduous stands because leaf litter inputs impede moss growth (Natalia and others 2008), and labile leaf litter combined with warm, dry conditions in deciduous-dominated stands increase decomposition rates (Van Cleve and others 1983b). In the aspen trajectory, decomposition of the SOL appeared to exceed leaf litter inputs, causing a reduction in SOL depth and belowground C pools over time. Belowground C pools in the paper birch trajectory appeared more balanced, perhaps because Alaska paper birch typically occur on east-facing slopes, so tend to be cooler and moister than south-facing aspen stands (Viereck and others 1983). These differences in abiotic conditions between the two deciduous trajectories could have led to differences in decomposition rates, and consequently, differences in the buildup of the SOL. Overall, the combined effects of ‘deciduousness’ and years since fire interacted to determine changes in belowground C pools, although the strength of the multivariate model to predict these changes was relatively weak, likely because of differences among the two deciduous trajectories.
Despite little temporal variation, belowground N pools were considerably higher in black spruce and paper birch trajectories compared to the aspen trajectory. In stands undergoing a black spruce trajectory, the opposing effects of increasing SOL depth and declining %N and bulk density over time interacted to create relatively high, and mostly stable, belowground N pools. These changes in SOL characteristics likely reflect increased contribution of moss-derived material to the SOL with stand age, as dead mosses tend to be porous with low bulk density (O’Donnell and others 2009) and have a high C:N ratio (~40 C:N; Boby and others 2010). In Alaska paper birch trajectories, relatively high SOL %N (often >2%) combined with occasionally deep SOL (often >15 cm) horizons, led to greater belowground N pools compared to the aspen trajectory. Higher SOL %N in birch trajectories could reflect higher N availability in these stands. The N-fixing shrub, Alnus crispa, is often found in the understory of these stands (Viereck and others 1983) and has been shown to increase soil N availability in Alaskan boreal forests (Wurtz 1995; Rhoades and others 2001). Alternatively, paper birch may be able to access N pools deeper in the mineral soils on some sites, leading to higher %N. Because of differences among the two deciduous trajectories and the lack of a temporal trend, none of the models tested predicted belowground N pools.
We hypothesized that C:N ratio would increase with time after fire across all successional trajectories but be more pronounced in deciduous trajectories (Figure 1C) due to proportionally more influence from stemwood biomass as the stands aged, as stemwood has a high C:N ratio (~335 to 430). Our data partially support this hypothesis. In aspen trajectories, C:N ratios increased linearly with time since fire, coincident with increased stemwood biomass contribution to total C pools (from an average 17% in stands 20–39 years since fire to 65% in those 60–100 years since fire). Alaska paper birch C:N ratio also increased as stemwood contribution to C pools increased from 11% (20–39 years since fire) to 40% (40–59 years since fire), but leveled off after this due to greater accumulation of SOL pools, which tended to have a lower C:N ratio (~23) compared to stemwood. C:N ratio of black spruce trajectories remained relatively constant because %SOL contribution to total pools remained constant with years since fire.
There was a change in the location (above- vs. belowground) of pools with shifting successional trajectory that led to total pools varying similarly with years since fire. Total C pools switched from being mainly in the SOL in a black spruce trajectory (87% at maturity compared to 35 and 15% in birch and aspen, respectively) to mainly in aboveground stemwood in deciduous trajectories (43 and 65% at maturity in birch and aspen, respectively, compared to only 8% in black spruce). As predicted (Figure 1D), the magnitude of this shift was equivalent, such that all three trajectories accumulated C similarly over time. Total N pools remained relatively constant over time because of changes in SOL quality described above, but proportionally less N was stored in the SOL in the deciduous trajectories (52–79% at maturity) compared to the black spruce trajectory (97%). This reduction in the amount of C and N stored in the SOL with a shift from a black spruce trajectory to deciduous trajectories could increase the longevity of these C pools. Deciduous stemwood C:N was about 10 times higher than that of the black spruce SOL, which could slow decomposition rates of this material, as substrate quality is often a major determinant of decomposition rates (Zhang and others 2008). In addition, because stemwood often remains standing after a tree dies, this could further increase longevity of this pool because decomposing fungi are less abundant on standing compared to fallen wood (Rayner and others 1988). SOL decomposability may be reduced by the cold, moist conditions common on the forest floor of black spruce stands, but the relative importance of abiotic conditions versus substrate effects on tissue decomposability across deciduous and black spruce trajectories remain unknown. However, in tundra ecosystems, substrate quality had more pronounced effects than temperature on decomposition rates (Hobbie 1996), suggesting that similar controls may occur in boreal forests.
Conclusion
As fire severity increases, successional trajectories of boreal forests of interior Alaska are likely to shift from black spruce self-replacement to species replacement, where deciduous species occupy areas previously dominated by black spruce. This shift will inevitably lead to a variety of ecosystem-level consequences because of differences in longevity, growth habit, litter quality, and understory microclimatic conditions between coniferous and deciduous functional types. One consequence demonstrated in this and an associated study (Alexander and others 2012) will be a substantial increase in aboveground C and N pools in tree biomass because of high productivity of deciduous trees and accumulation and storage of C and N in stemwood. This increase in aboveground C and N pools within deciduous trajectories will be accompanied by an increase in C:N ratios and an often equivalent decrease in C and N stored in the soil organic layer. Thus, there will be a trade-off in the location of these pools but little change in the total pool. This trade-off associated with a shift in forest composition could increase C and N longevity in this region due to more C and N stored in low-quality deciduous stemwood. Forest flammability will also decrease with increased deciduousness due to decreased fuel loads in the SOL and higher moisture content of deciduous biomass (Chapin and others 2006b), which could further increase longevity of C and N pools. Our findings highlight the role of forest trajectory type in determining rates of C and N accumulation and storage within boreal forests and suggest that a fire-driven shift from black spruce to deciduous trajectories could potentially cause a negative feedback to the climate system through changes in C and N pool longevity.
References
Alexander HD, Mack MC, Goetz S, Beck PSA, Belshe EF. 2012. Implications of increased deciduous cover on stand structure and aboveground carbon pools of Alaskan boreal forests. Ecosphere 3:art45.
Balshi MS, McGuire AD, Duffy P, Flannigan M, Kicklighter DW, Melillo J. 2009. Vulnerability of carbon storage in North American boreal forests to wildfires during the 21st century. Glob Change Biol 15:1491–510.
Boby LA, Schuur EA, Mack MC, Verbyla D, Johnstone JF. 2010. Quantifying fire severity, carbon, and nitrogen emissions in Alaska’s boreal forest. Ecol Appl 20:1633–47.
Bond-Lamberty B, Peckham SD, Ahl DE, Gower ST. 2007. Fire as the dominant driver of central Canadian boreal forest carbon balance. Nature 450:89–92.
Bradshaw CJ, Warkentin IG, Sodhi NS. 2009. Urgent preservation of boreal carbon stocks and biodiversity. Trends Ecol Evolut 24:541–8.
Chapin FS, Trainor SF, Huntington O, Lovecraft AL, Zavaleta E, Natcher DC, McGuire AD, Nelson JL, Ray L, Calef M et al. 2008. Increasing wildfire in Alaska’s boreal forest: pathways to potential solutions of a wicked problem. BioScience 58:531–40.
Chapin FSIII, Hollingsworth T, Murray DF, Viereck LA, Walker MD. 2006a. Floristic diversity and vegetation distribution in the Alaskan boreal forest. New York: Oxford University Press.
Chapin FSIII, Viereck LA, Adams P, Van Cleve K, Fastie CL, Ott RA, Mann D, Johnstone JF. 2006b. Successional processes in the Alaskan boreal forest. Oxford: Alaska’s changing boreal forest Oxford University Press. pp 100–20.
Dyrness CT. 1982. Control of depth to permafrost and soil temperature by the forest floor in black spruce/feathermoss communities. US Dept. of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station.
Flannigan M, Stocks B, Turetsky M, Wotton M. 2009. Impacts of climate change on fire activity and fire management in the circumboreal forest. Glob Change Biol 15:549–60.
Gower ST, Hunter A, Campbell J, Vogel J, Veldhuis H, Harden J, Trumbore S, Norman JM, Kucharik CJ et al. 2000. Nutrient dynamics of the southern and northern BOREAS boreal forests. Ecoscience 7:481–90.
Harden JW, Mack M, Veldhuis H, Gower ST. 2002. Fire dynamics and implications for nitrogen cycling in boreal forests. J Geophys Res Atmos (1984–2012) 107:WFX-4.
Harden JW, Trumbore SE, Stocks BJ, Hirsch A, Gower ST, O’neill KP, Kasischke ES. 2000. The role of fire in the boreal carbon budget. Glob Change Biol 6:174–84.
Hinzman LD, Bettez ND, Bolton WR, Chapin FS, Dyurgerov MB, Fastie CL, Griffith B, Hollister RD, Hope A, Huntington HP et al. 2005. Evidence and implications of recent climate change in northern Alaska and other arctic regions. Clim Change 72:251–98.
Hobbie SE. 1996. Temperature and plant species control over litter decomposition in Alaskan tundra. Ecol Monogr 66:503–22.
Hobbie SE, Schimel JP, Trumbore SE, Randerson JR. 2000. Controls over carbon storage and turnover in high-latitude soils. Glob Change Biology 6:196–210.
Hollingsworth TN, Walker MD, Chapin FSIII, Parsons AL. 2006. Scale-dependent environmental controls over species composition in Alaskan black spruce communities. Can J For Res 36:1781–96.
Hulten E. 1968. Flora of Alaska and neighboring territories: a manual of the vascular plants. Redwood City, CA: Stanford University Press.
Huntington HP, Goodstein E, Euskirchen E. 2012. Towards a tipping point in responding to change: rising costs, fewer options for Arctic and global societies. Ambio 41:66–74.
Jerabkova L, Prescott CE, Kishchuk BE. 2006. Nitrogen availability in soil and forest floor of contrasting types of boreal mixedwood forests. Can J For Res 36:112–22.
Johnstone JF, Chapin FSIII. 2006a. Effects of soil burn severity on post-fire tree recruitment in boreal forest. Ecosystems 9:14–31.
Johnstone JF, Chapin FSIII. 2006b. Fire interval effects on successional trajectory in boreal forests of northwest Canada. Ecosystems 9:268–77.
Johnstone JF, Kasischke ES. 2005. Stand-level effects of soil burn severity on postfire regeneration in a recently burned black spruce forest. Can J For Res 35:2151–63.
Johnstone JF, Chapin FS, Foote J, Kemmett S, Price K, Viereck L. 2004. Decadal observations of tree regeneration following fire in boreal forests. Can J For Res 34(2):267–73.
Johnstone JF, Chapin FS, Hollingsworth TN, Mack MC, Romanovsky V, Turetsky M. 2010a. Fire, climate change, and forest resilience in interior Alaska. Can J For Res 40:1302–12.
Johnstone JF, Hollingsworth TN, Chapin FS, Mack MC. 2010b. Changes in fire regime break the legacy lock on successional trajectories in Alaskan boreal forest. Glob Change Biol 16:1281–95.
Kasischke ES, Christensen Jr NL, Stocks BJ. 1995. Fire, global warming, and the carbon balance of boreal forests. Ecol Appl 5(2):437–51.
Kasischke ES, Verbyla DL, Rupp TS, McGuire AD, Murphy KA, Jandt R, Barnes JL, Hoy EE, Duffy PA, Calef M et al. 2010. Alaska’s changing fire regime-implications for the vulnerability of its boreal forests. Can J For Res 40:1313–24.
Kelly R, Chipman ML, Higuera PE, Stefanova I, Brubaker LB, Hu FS. 2013. Recent burning of boreal forests exceeds fire regime limits of the past 10,000 years. Proc Natl Acad Sci 110:13055–60.
Légaré S, Paré D, Bergeron Y. 2004. The responses of black spruce growth to an increased proportion of aspen in mixed stands. Can J For Res 34:405–16.
Légaré S, Paré D, Bergeron Y. 2005. Influence of aspen on forest floor properties in black spruce-dominated stands. Plant Soil 275:207–20.
Mack MC, Treseder KK, Manies KL, Harden JW, Schuur EA, Vogel JG, Randerson JT, Chapin FSIII. 2008. Recovery of aboveground plant biomass and productivity after fire in mesic and dry black spruce forests of interior Alaska. Ecosystems 11:209–25.
McGuire AD, Anderson LG, Christensen TR, Dallimore S, Guo L, Hayes DJ, Heimann M, Lorenson TD, Macdonald RW, Roulet N. 2009. Sensitivity of the carbon cycle in the Arctic to climate change. Ecol Monogr 79:523–55.
Natalia S, Lieffers VJ, Landhäusser SM. 2008. Effects of leaf litter on the growth of boreal feather mosses: implication for forest floor development. J Veg Sci 19:253–60.
Neff JC, Harden JW, Gleixner G. 2005. Fire effects on soil organic matter content, composition, and nutrients in boreal interior Alaska. Can J For Res 35:2178–87.
O’Donnell JA, Romanovsky VE, Harden JW, McGuire AD. 2009. The effect of moisture content on the thermal conductivity of moss and organic soil horizons from black spruce ecosystems in interior Alaska. Soil Sci 174:646–51.
Oechel WC, Van Cleve K. 1986. The role of bryophytes in nutrient cycling in the taiga. In: Van Cleve K, Chapin FS, Flanagan P, Viereck LA, Dyrness CT, Eds. Forest ecosystems in the Alaskan taiga. Berlin: Springer. p 121–37.
Osterkamp TE, Romanovsky VE. 1999. Evidence for warming and thawing of discontinuous permafrost in Alaska. Permafr Periglac Process 10:17–37.
Pan Y, Birdsey RA, Fang J, Houghton R, Kauppi PE, Kurz WA, Phillips OL, Shvidenko A, Lewis SL, Canadell JG et al. 2011. A large and persistent carbon sink in the world’s forests. Science 333:988–93.
Randerson JT, Liu H, Flanner MG, Chambers SD, Jin Y, Hess PG, Pfister G, Mack MC, Treseder KK, Welp LR et al. 2006. The impact of boreal forest fire on climate warming. Science 314:1130–2.
Rayner AD, Boddy L et al. 1988. Fungal decomposition of wood. Its biology and ecology. New York: Wiley.
Rhoades C, Oskarsson H, Binkley D, Stottlemyer B. 2001. Alder (Alnus crispa) effects on soils in ecosystems of the Agashashok River valley, northwest Alaska. Ecoscience 8:89–95.
Romme WH, Everham EH, Frelich LE, Moritz MA, Sparks RE. 1998. Are large, infrequent disturbances qualitatively different from small, frequent disturbances? Ecosystems 1:524–34.
Slaughter CW, Viereck LA. 1986. Climatic characteristics of the taiga in interior Alaska. In: Van Cleve K, Chapin FS, Flanagan P, Viereck LA, Dyrness CT, Eds. Forest ecosystems in the Alaskan taiga. Berlin: Springer. p 9–21.
Soja AJ, Tchebakova NM, French NH, Flannigan MD, Shugart HH, Stocks BJ, Sukhinin AI, Parfenova EI, Chapin FSIII, Stackhouse PW Jr. 2007. Climate-induced boreal forest change: predictions versus current observations. Glob Planet Change 56:274–96.
Turetsky MR. 2003. The role of bryophytes in carbon and nitrogen cycling. Bryol 106:395–409.
Turetsky MR, Kane ES, Harden JW, Ottmar RD, Manies KL, Hoy E, Kasischke ES. 2011. Recent acceleration of biomass burning and carbon losses in Alaskan forests and peatlands. Nat Geosci 4:27–31.
Van Cleve K, Alexander V. 1981. Nitrogen cycling in tundra and boreal ecosystems. Ecol Bull 33:375–404.
Van Cleve K, Oliver L, Schlentner R, Viereck LA, Dyrness CT. 1983a. Productivity and nutrient cycling in taiga forest ecosystems. Can J For Res 13(5):747–66.
Van Cleve K, Dyrness CT, Viereck LA, Fox J, Chapin FS, Oechel W. 1983b. Taiga ecosystems in interior Alaska. Bioscience 33(1):39–44.
Viereck LA, Dyrness CT, Van Cleve K, Foote MJ. 1983. Vegetation, soils, and forest productivity in selected forest types in interior Alaska. Can J For Res 13:703–20.
Weber MG, Flannigan MD. 1997. Canadian boreal forest ecosystem structure and function in a changing climate: impact on fire regimes. Environ Rev 5(3–4):145–66.
Wurtz TL. 1995. Understory alder in three boreal forests of Alaska: local distribution and effects on soil fertility. Can J For Res 25:987–96.
Zhang D, Hui D, Luo Y, Zhou G. 2008. Rates of litter decomposition in terrestrial ecosystems: global patterns and controlling factors. J Plant Ecol 1:85–93.
Acknowledgements
We would like to thank Kamala Earl, Camilo Mojica, Nils Pederson, Leslie Boby, Mindy Sun, Michael Mahala, and Jennifer Stanley for their invaluable help in the field and laboratory. We appreciate the assistance of the US Army for allowing access to fire scars on military sites. Funding for this research was provided by NASA Ecosystems and Carbon Cycle Grant NNX08AG13G, the DOD Strategic Environmental Research and Development Program (SERDP) under project RC-2109, and the Bonanza Creek Long Term Ecological Research Site program, which is funded by NSF DEB-0620579 and USDA Forest Service, Pacific Northwest Research Station, grant PNW01-JV11261952-231.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
HDA has assisted with study design, performed research, analyzed data, and wrote manuscript; MCM has conceived of and designed the study, contributed to data interpretation, and critically reviewed the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alexander, H.D., Mack, M.C. A Canopy Shift in Interior Alaskan Boreal Forests: Consequences for Above- and Belowground Carbon and Nitrogen Pools during Post-fire Succession. Ecosystems 19, 98–114 (2016). https://doi.org/10.1007/s10021-015-9920-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-015-9920-7