Abstract
In the boreal forest of Alaska, increased fire severity associated with climate change is expanding deciduous forest cover in areas previously dominated by black spruce (Picea mariana). Needle-leaf conifer and broad-leaf deciduous species are commonly associated with differences in tree growth, carbon (C) and nutrient cycling, and C accumulation in soils. Although this suggests that changes in tree species composition in Alaska could impact C and nutrient pools and fluxes, few studies have measured these linkages. We quantified C, nitrogen, phosphorus, and base cation pools and fluxes in three stands of black spruce and Alaska paper birch (Betula neoalaskana) that established following a single fire event in 1958. Paper birch consistently displayed characteristics of more rapid C and nutrient cycling, including greater aboveground net primary productivity, higher live foliage and litter nutrient concentrations, and larger ammonium and nitrate pools in the soil organic layer (SOL). Ecosystem C stocks (aboveground + SOL + 0–10 cm mineral soil) were similar for the two species; however, in black spruce, 78% of measured C was found in soil pools, primarily in the SOL, whereas aboveground biomass dominated ecosystem C pools in birch forest. Radiocarbon analysis indicated that approximately one-quarter of the black spruce SOL C accumulated prior to the 1958 fire, whereas no pre-fire C was observed in birch soils. Our findings suggest that tree species exert a strong influence over C and nutrient cycling in boreal forest and forest compositional shifts may have long-term implications for ecosystem C and nutrient dynamics.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Climate change has been linked to an increase in the occurrence, extent, and severity of wildfires across many forested ecosystems (for example, Flannigan and others 2009b; Rocca and others 2014). Although many ecosystems are adapted to fire disturbances, changes to the fire regime can introduce novel opportunities for ecosystem reorganization that facilitate long-term changes in plant-soil-microbial feedbacks and ecosystem structure and function (Johnstone and others 2010b). Across interior Alaska, changes to the fire regime are shifting boreal forest tree species composition from needle-leaf conifer black spruce (Picea mariana (Mill.) B.S.P.)-dominated ecosystems to greater deciduous broad-leaf species cover. Differences in tree species composition are known to coincide with important characteristics in the boreal biome, including aboveground productivity, biogeochemical cycling, and the soil environment (Van Cleve and others 1983a). However, few studies have quantified how these new successional trajectories may catalyze long-term shifts in ecosystem structure and nutrient dynamics.
Black spruce has dominated Alaska’s boreal landscape for the last 6000 years (Lynch and others 2002; Chapin and others 2006) and historically self-replaced following wildfires (Johnstone and others 2010a). In recent decades, increased frequency of large fires (Kasischke and others 2010; Kelly and others 2013) that burn deeply into the soil organic layer (SOL) (Turetsky and others 2011) have been observed. Severe fires leave behind a shallow residual SOL or expose mineral soil (Boby and others 2010) that provide a high-quality seedbed on which broad-leaf deciduous species are able to outcompete black spruce and establish at high rates (Greene and others 2007; Johnstone and others 2010b). This change in species establishment post-fire represents an abrupt shift in forest successional trajectories in Interior Alaska (Johnstone and others 2010a, b; Beck and others 2011) that may have long-term implications that unfold throughout stand development.
Within forested biomes, needle-leaf conifers and broad-leaf deciduous tree species are often associated with differences in plant-soil-microbial feedbacks that impact C and nutrient availability and distribution (Hobbie 1992; Vesterdal and others 2013). Needle-leaf conifers are typically adapted to nutrient-poor environments and maximize nutrient use by growing slowly, investing in long-lived foliage that is rich in recalcitrant compounds and low in nutrients, and by resorbing a large proportion of nutrients prior to leaf senescence (Melillo and others 1982; Flanagan and Van Cleve 1983; Aerts 1995). These characteristics contribute to slow decomposition rates and accumulation of C and nutrients in the SOL, which further reinforce nutrient limitation. In contrast, broad-leaf deciduous species generally exhibit higher net primary productivity (NPP) (Van Cleve and others 1983a; Lambers and Poorter 1992; Alexander and others 2012), have a greater nutrient demand, and produce more foliar litter containing higher nutrient concentrations (Van Cleve and others 1983a; Bond-Lamberty and others 2006). Deciduous species also often exhibit faster rates of C and nutrient turnover in soils (Bauhus and others 1998; Vance and Chapin 2001), which can result in larger C and nutrient stocks in tree biomass relative to organic soil pools (Van Cleve and others 1983a; Alexander and Mack, in press).
Compared to other forested biomes, the linkage between tree species composition and plant-soil-microbial feedbacks in boreal forest is unique because of the close association between overstory tree species and forest floor moss cover. In black spruce stands, the forest floor is often composed of a nearly continuous mat of feather and sphagnum moss. This moss can account for as much as 20% of ecosystem NPP and consists of recalcitrant litter that is slow to decompose, leading to high rates of SOL accumulation (Hobbie and others 2000; Turetsky and others 2010). Thick moss layers also have a low bulk density and thermal conductivity, and high water holding capacity that keep soils cool (O’Donnell and others 2009; Turetsky and others 2012). This reduces microbial activity and rates of nitrogen (N) mineralization (Vance and Chapin 2001; Gornall and others 2007) and limits permafrost thaw during the summer months (Van Cleve and others 1983a; Jorgenson and others 2010). These SOL characteristics in black spruce stands have historically created a resilient ecosystem where wildfires did not burn deeply into the SOL unless the fires were exceptionally severe (Dyrness and Norum 1983; Shetler and others 2008) and where black spruce were able to re-establish on the charred residual SOL by large seed releases from semi-serotinous cones (Johnstone and others 2010b). In poorly drained areas underlain by permafrost, this pattern is likely to continue, but in upland black spruce sites that are moderately well drained, more severe fires are expected to consume the SOL and allow for high rates of broad-leaf deciduous tree species establishment (Johnstone and others 2010b). Unlike black spruce, deciduous forests in the boreal zone have only patchy moss distribution on the forest floor (Turetsky and others 2010), contributing to the development of a warmer, drier soil environment.
Soil nutrient dynamics have also been shown to differ between boreal needle-leaf conifer and broad-leaf deciduous forests. For instance, broad-leaf deciduous species exhibit shorter SOL turnover times and higher N and phosphorus (P) mineralization rates relative to spruce species (Flanagan and Van Cleve 1983). The boreal ecosystem is highly nutrient limited, especially by N (Van Cleve and others 1983b; Hobbie and others 2002), but P limitation also occurs at high latitudes (Bowman and others 1993; Hobbie and others 2002). Further, base cation deficiencies have been identified in some regions of the boreal zone (Quesnel and Côté 2009). If broad-leaf deciduous forests in the boreal region cycle nutrients more quickly than black spruce, as is suggested from previous studies, then we may expect greater opportunity for nutrient loss in stands that shift to deciduous dominance, which could further constrain NPP and alter ecosystem nutrient dynamics.
In this study, we quantified C and nutrient pools and fluxes in spatially interspersed, mid-successional approximately 45-year-old stands of black spruce, and Alaska paper birch (Betula neoalaskana, Sarg.) that established following a single fire event in interior Alaska. As noted previously, other studies have investigated interspecific differences in C and nutrient cycling in Alaska. These studies often characterize tree species relationships across heterogeneous landscapes differing in time since fire, topographic position, and deciduous-conifer mixtures. Here, we focus instead on a single fire event within one site, to refine our mechanistic understanding of the relative influence of black spruce and Alaska paper birch on successional development and ecosystem C and nutrient dynamics. Alaska paper birch is a broad-leaf deciduous species able to establish across a wide range of environmental conditions, including in cooler soils on north-facing slopes that are currently commonly dominated by black spruce (Viereck and others 1983). Therefore, we may expect greater establishment of Alaska paper birch in these areas following severe fire events. Building on the conceptual framework developed by Johnstone and others (2010a), we hypothesized that black spruce and Alaska paper birch successional trajectories would shape contrasting patterns of C and nutrient cycling and distribution. We expected birch stands to exhibit characteristics consistent with more rapid plant-soil-microbial feedbacks than spruce stands, consisting of higher aboveground net primary productivity (ANPP), faster decomposition and nutrient turnover, and a warmer, shallower SOL. In contrast, we expected black spruce to display high SOL accumulation, deeper, cooler soils, and slower decomposition and nutrient turnover rates. Refining our understanding of how abrupt changes associated with a shifting fire regime impact forest successional processes and species-specific ecosystem nutrient dynamics will allow for stronger predictions of long-term changes in C storage, nutrient availability, and ecosystem structure and function.
Methods
Site Information
Research was conducted in the Tanana Valley State Forest near Fairbanks, Alaska. The forest in this area established following the Murphy Dome Fire in 1958 and is comprised primarily of black spruce and Alaska paper birch commonly found in mono-specific stands. This site is in a region of discontinuous permafrost where upland soils are typically silty loams (Van Cleve and others 1983a). We established study plots in three locations (hereafter called blocks) within this forest where we identified adequately sized, adjacent stands dominated by black spruce and Alaska paper birch. Within each block, five 10 m x 10 m plots were established for each tree species, resulting in a total of 15 plots dominated by each tree species across the study site. This block design allowed us to quantify C and nutrient pools and fluxes in the two forest types while also capturing some of the spatial heterogeneity across the site. Two blocks were located on one access road and were approximately 900 m apart (Block A, N64°52′54.0″, W148°23′29.4″; Block B, N64°52′34.9″, W148°22′57.4″). This portion of the site is comprised primarily of Steese silt loam, with some Saulich peat underlying the spruce in Block A (U.S. Department of Agriculture 2013). The third block (Block C) was on a different road, at a distance of about 3000–4000 m (overland) from the other two blocks (N64°53′42.4″, W148°27′03.3″). Block C soils are classified as Histic pergelic cryaquepts (U.S. Department of Agriculture 2013). All blocks had a northeasterly aspect with a mean slope of 7° ± 3 (SD) in black spruce and 11° ± 2 (SD) in birch.
In black spruce stands, the SOL surface was dominated by the feather moss species Hylocomium splendens and Pleurozium schreberi, and in birch, the SOL was most commonly composed of birch leaf litter, with little moss cover. Mean annual air temperature (measured every 30 minutes) across the site was −3.2°C in 2012–2013. During winter 2012–2013, the onset of snow cover and snow-out dates were similar in black spruce and paper birch stands, with continuous snow cover present from mid-October to mid-May.
Aboveground Stand Characteristics
We measured the diameter at breast height (DBH, 1.4 m) of all live and standing dead trees at least 1.4 m tall and basal diameter (BD) on trees less than 1.4 m tall, and all tall shrubs. Dead trees with an angle of greater than 45° from the forest floor were categorized as standing dead. We then applied allometric equations reported by Alexander and others (2012) to all trees and equations developed by Berner and others (2015) for tall shrubs to estimate total and component-specific biomass. Total C and N pool sizes were determined using C and N concentrations reported here and values from H. Alexander (unpublished). Forest age and tree ANPP (hereafter ANPPtree) were estimated from one stem cross section collected at DBH from a dominant tree adjacent to each study plot. Detailed methods for biomass, tree aging, and ANPPtree can be found in Appendix 1 in supplementary material.
Foliar nutrient concentrations and specific leaf area were determined from fresh foliage collected from the upper canopy of each plot. Plot subsamples were analyzed for percent C and N, as well as total P, calcium (Ca), magnesium (Mg), and potassium (K) (methods detailed in Appendix 1 in supplementary material). Annual litter production was estimated by collecting all litter that fell into collection baskets over the course of one year. Baskets were installed at three random locations within each plot. In the spruce stands, 50 cm × 50 cm wooden frames with fiberglass mesh-lined bottoms (18 × 16 openings per square inch of mesh) were placed on the ground, and in birch plots, 62.2 cm × 45.4 cm fiberglass mesh-lined laundry baskets were installed. Litter was collected from the birch stands following leaf fall in September and October 2012, and in all plots in June and July 2013. Collected litter was dried and sorted into foliar litter (by species), wood, seeds, cones, and other miscellaneous components, then re-dried and weighed. A subsample of the birch foliar litter collected in September 2012 was ground and analyzed for C, N, P, and base cations using the same methods outlined for fresh foliage. Spruce needles collected in July 2013 were also analyzed for C and N. To minimize nutrient leaching from senesced foliage, nutrient analysis was conducted only on material that had been in baskets for less than one month. Because of equipment limitations, black spruce foliar litter P, Ca, Mg, and K was analyzed on replicated samples collected across only one study block (Block A) in 2011. Woody debris was quantified using the line intercept method (Brown 1974), detailed in Appendix 1 in supplementary material.
Soil Characterization
Soil temperature was monitored from September 2012 to July 2013 using two Thermochron iButtons (Maxim Integrated Products, San Jose, CA) installed 10 cm below the SOL surface in each plot. A difference in SOL depth between the stand types resulted in the sensors being in the SOL in black spruce and in the mineral soil in birch. High rock content prevented an accurate assessment of depth to permafrost, so we do not report data here.
To assess soil C and nutrient pools and fluxes, we collected the SOL and top 10 cm of mineral soil from three random locations within each study plot in August 2012. First, an intact, approximately 10 cm × 10 cm sample of the SOL was collected using a knife and pruners. Dimensions and depths of each horizon (green moss, dead (brown) moss, and fibric) were recorded and charcoal was noted if visible. A mineral soil core (6.8 cm diameter) was then extracted from immediately below the SOL sampling location. In June 2013, we collected additional soil samples from one location per species within each study block (6 locations total, 3 per species) to be used only to estimate SOL age and elemental concentrations of deeper (0–100 cm) mineral soil. The SOL was removed as an intact sample and underlying mineral soils were then collected in 10 cm increments using a diamond-tipped, gas powered rotary corer (9.5 cm internal diameter, Rau and others 2011). All samples were then frozen until analysis.
In the lab, soils collected in 2012 were thawed at room temperature (23–25°C) and vascular plants and live (green) moss were removed from the top of SOL samples. Green moss was weighed and dried and spruce SOLs were cut horizontally to separate the dead moss layer (when present) from the fibric horizon. No humic horizon was observed in any SOL profiles. Birch SOLs were processed as a single layer. An intact subsample of each SOL sample representing the entire depth of the sample was removed using a knife and pruners. Mineral soil cores typically did not stay intact, so a spatula was used to collect a subsample containing material from the entire depth of the core. These subsamples were used for estimation of potential net N mineralization and nitrification, and carbon dioxide (CO2) respiration rates (methods to follow). Next, rocks, roots larger than 2 mm in diameter, and any large pieces of minimally decayed wood were removed. Roots and wood were dried at 60°C and rock volume was estimated by water displacement in a graduated cylinder. All remaining SOL and mineral material was thoroughly homogenized and the three samples collected within each plot were combined, generating one composite sample per plot, per horizon. Composite samples were analyzed for gravimetric moisture content, C, N, and exchangeable base cation concentrations, Mehlich P, and pH. Initial pool sizes of ammonium (NH4 +) and nitrate (NO3 −) used to calculate potential N mineralization and nitrification were also determined from these samples. Finally, a subsample of each 0–10 cm mineral soil plot composite was further composited into one sample per species, per block, for analysis of total element concentrations. See Appendix 1 in supplementary material for detailed analytical methods. We used the SOL samples collected in 2013 to estimate the age of the C within the SOL profile (Mack and others 2011), to quantify the amount of C that had accumulated since the fire and confirm that observed charcoal layers within the profiles in spruce sites were related to the last stand-replacing fire. The deeper mineral soils collected in 2013 were analyzed for total C and N, and near-total element concentrations. Methodological details are provided in Appendix 1 in supplementary material.
Pools of NH4 +, NO3 −, and phosphate (PO4 2−) were estimated in the field using ion exchange resins (BioRad, Hercules, CA). In August 2012, one anion and one cation resin bag containing about 3 g of moist resin was deployed in the SOL and upper mineral soils within each plot. All birch resins and SOL spruce resins were removed and replaced in June 2013 and bags from all locations were swapped in August 2013. Mineral bags in spruce plots were inaccessible in June 2013 because the ground was frozen. Resins were rinsed and refrigerated prior to extraction in a 0.1 M HCl and 2 M sodium chloride mixture and analysis on an autoanalyzer (detailed methods provided in Appendix 1 in supplementary material).
Soil C and N Fluxes
Estimates of soil CO2 respiration (90-day) and potential net N mineralization and nitrification rates (30- and 90-day) were determined on all SOL and 0–10 cm mineral soil samples. One composite sample was created for each horizon for each plot and time period (see Appendix 1 in supplementary material for detailed methods). Samples were brought to approximate field capacity using deionized water, then placed in the dark at 15°C. This temperature was intended to maximize microbial activity, but stay within the range these soils might experience in the field (Lavoie and others 2011). Soil CO2 respiration rates were measured every eight hours for the first two weeks, then at least weekly for the remainder of the 90-day experiment. Within the last 20 days of the experiment, 14CO2 was captured for samples from two plots of each species within each block, to estimate the age of C being respired (Schuur and others 2009). Potential net N mineralization and nitrification were calculated for the samples used in the 90-day CO2 respiration experiment and for a second set of samples processed in the same way, but incubated for 30 days. Extraction details are provided in Appendix 1 in supplementary material.
Statistics
To identify significant differences between species and assess variance among blocks, we conducted analysis of variance tests using two model designs. First, we used a nested model with species as the main effect and block nested within species (JMP Pro 9.0, SAS Institute, Cary NC) to test how within-species variability across blocks might affect our ability to detect overall species differences. For most analyses, differences between species were present and no significant differences were observed between blocks of the same species, suggesting low variability across the blocks. In the instances where there was a significant block effect, a post hoc Tukey test was used to confirm that overall species effects were not driven by any one block acting as a large outlier. Because no large differences were observed, we simplified our model to include only the species main effect and report the overall species effect here (Underwood 1997). Prior to the analysis of variance testing, datasets were transformed when needed to meet normality and homoscedasticity assumptions. For datasets that could not be transformed to comply with normality assumptions, a nonparametric Wilcoxon test with a 2-sample normal approximation was used.
Results
Aboveground
Ring counts of trees across the study site suggested a mean age at DBH of 45 years (Table 1, Appendix 2 in supplementary material), which is consistent with tree establishment following the known 1958 fire for both stand types. Within birch plots, birch trees accounted for 97% of live tree aboveground biomass, while black spruce comprised 79% of the aboveground biomass in spruce plots (Table 1, Appendix 2 in supplementary material). Birch was the most common “other” tree species observed in black spruce stands. Stem density was over seven times higher in spruce stands (7.31 stems m−2) compared to birch (0.93 stems m−2). However, spruce tree diameters were relatively small, resulting in significantly smaller basal area and less than half the total tree biomass found in birch stands (5.30 vs. 11.33 kg m−2, Table 1). Birch ANPPtree was also more than double that found in spruce plots (Table 1).
Fresh birch foliage exhibited concentrations of N, P, Ca, and Mg, that were at least double that found in current year black spruce needles (Table 2). A higher concentration of K was also observed in birch foliage, whereas black spruce showed a higher C concentration and larger C:N ratio (Table 2). Foliar litter N, P, Mg, and K concentrations were also higher in birch, whereas black spruce contained a higher Ca concentration, as well as a larger C:N ratio. Comparing live foliage and foliar litter nutrient concentrations suggested that paper birch resorbed 64% of N and 14% of foliar P from foliage prior to leaf fall, whereas black spruce resorbed 33% of foliar N and 71% of P. Specific leaf area of birch foliage was 3.5-fold that of spruce (158 ± 4 vs. 43 ± 1 cm2 g−1 for birch and spruce, respectively, P < 0.0001). Total litter inputs in birch stands were more than double that in spruce (242.8 ± 14.9 vs. 93.8 ± 10.8 g m−2 y−1, Figure 1A). This pattern was driven by larger inputs of foliar litter (178.4 ± 3.9 vs. 72.2 ± 8.0, P < 0.0001) and wood (52.2 ± 15.0 vs. 16.2 ± 4.7 g m−2 y−1, P = 0.01) in birch plots. Interestingly, in both stand types around 77% of total inputs were from foliage and about 17% from wood, although for black spruce only 60% of the litter was spruce needles, with the rest being birch and shrub leaves. Total foliar inputs of C, N, Mg, and K were all four to six times larger in birch plots relative to spruce, and Ca inputs were also statistically larger (Figure 1B).
Annual litter input and composition in black spruce and Alaska paper birch stands (A), and input rates of nutrients in foliar litter in each stand type (B). Carbon input values correspond to the axis on the right side of B. Values are presented as mean (SE) for each indicated component, (n = 15 plots per species). Significant differences between tree species for each variable are indicated with *** (P < 0.0001), determined using a nonparametric Wilcoxon test.
Soils
The soil temperature 10 cm below the SOL surface was higher in birch soils relative to black spruce in September–October 2012 and June–July 2013 (Figure 2). Once soils dipped below freezing, species differences were no longer detectable. The SOL in black spruce soils was deeper (16.4 vs. 7.6 cm, Table 3), exhibited a larger mass (9.7 ± 0.3 vs. 5.2 ± 0.9 kg organic material (OM) m−2, P < 0.0001), and contained larger C and N stocks than paper birch soils (4.8 ± 0.3 vs. 2.4 ± 0.1 kg C m−2 and 139.2 ± 10.4 vs. 93.3 ± 5.6 g N m−2, Figure 3B, D). No difference in SOL bulk density was observed (Table 3). Radiocarbon analysis of the SOL indicated that the uppermost approximately 9–12 cm of the SOL in black spruce stands was composed of C that accumulated in the years since the 1958 fire and accounted for about 3.6 ± 0.6 kg C m−2 of the observed SOL C stocks. Soil organic layer C stocks measured across all black spruce plots averaged 4.8 kg C m−2, suggesting that approximately 75% of the observed SOL C accumulated in the years since the fire and 25% of the C survived the 1958 fire and is a legacy of the previous forest. In the birch SOL, bulk soil Δ14C values indicated that the bulk C pool was no greater than 26 years old, indicating all SOL C has accumulated post-fire and that the SOL turns over relatively rapidly in the deciduous stands.
Monthly soil temperature, mean (SE), n = 15 plots per species, collected September 2012–July 2013. Note that low variability among plots of the same species make the error bars difficult to detect. Significant differences between black spruce and paper birch for each month indicated by *** (P < 0.0001) determined using a Wilcoxon test for nonparametric data.
Measured forest stand C and N stocks (A, C) and the distribution of stocks among the measured forest stand pools (B, D). Mineral soil includes the uppermost 10 cm. Values are reported mean (SE), n = 15 plots per species. For A and C, error bars and statistical analyses are for the total summed ecosystem stocks. Significant differences between species for each stock and pool are indicated by **P < 0.001 and ***P < 0.0001.
Birch SOL samples contained higher concentrations of N, Mehlich P, and exchangeable Ca and Mg, and showed higher pH, whereas a larger C:N ratio was observed in the black spruce SOL (Table 3). Despite higher concentrations of exchangeable Ca and Mg in paper birch, the larger SOL mass in black spruce offset this, resulting in no difference in the total pool size of either base cation in the SOL (data not shown). Within the uppermost 10 cm of mineral soil, black spruce contained higher concentrations of C and N, a larger C:N ratio, and higher pH than birch, whereas birch soils showed greater concentrations of Mehlich P and exchangeable K (Table 3). There was a trend for higher total elemental P in black spruce soils, but the small sample size prevented statistical analysis. Total element concentrations for the deeper mineral soils, collected from 0 to 100 cm, showed greater variability among blocks than between species, suggesting that adjacent stands of the two different species established on similar parent material (Appendix 3 in supplementary material).
Accumulation of resin bound NH4 + was higher in both SOL and mineral soils of birch stands relative to spruce (179 ± 78 vs. 84 ± 35 μg NH4–N g resin−1 for SOL and 116 ± 37 vs. 22 ± 8 μg NH4–N g resin−1 for mineral soil, Figure 4A). A similar pattern was observed for resin bound PO4 2− (802 ± 180 vs. 144 ± 63 PO 24 –P g resin−1 for SOL and 1251 ± 216 vs. 140 ± 68, Figure 4E). Nitrate accumulation was also higher in the SOL in birch soils (3 ± 2 vs. 0.3 ± 0.3 NO3–N g resin−1), whereas no difference was observed in mineral soil (Figure 4C). For the 90-day incubation, there was a trend for higher potential net N mineralization and nitrification in organic and mineral birch soils (Figure 4B, D); however, these differences were only significant for N mineralization in mineral soil (78.1 ± 17.7 vs. 4.4 ± 2.1 μg N g soil N−1 d−1) and nitrification in the SOL (38.0 ± 16.5 vs. 0.9 ± 0.4 μg N g soil N−1 d−1). A similar pattern was observed for the 30-day incubation (data not shown). No difference in cumulative CO2–C released or the Δ14C value of the CO2–C was observed between species for either soil layer (Table 4). The ratio of C mineralized per unit N mineralized was significantly higher in birch mineral soils, whereas no difference was evident between the tree species in the SOL (Table 4).
Annual anion exchange resin NH4 + (A), NO3 − (C), and PO4 2− (E) accumulation and potential net N mineralization (B) and nitrification (D) for organic and 0–10 cm mineral soils of black spruce and Alaska paper birch. Significant differences between the species for each N or P metric and soil layer are indicated by *P < 0.05, **P < 0.001, ***P < 0.0001. A Wilcoxon test for nonparametric data was used to analyze these datasets.
Ecosystem C and N
Measured ecosystem C stocks (aboveground + SOL + 0–10 cm mineral soil) did not differ between black spruce and Alaska paper birch stands at our site (11.3 ± 1.9 vs. 11.1 ± 1.6 kg C m−2, P = 0.7145, Figure 3A). The distribution of C among biomass pools, however, was markedly different between the stand types, as indicated by interspecific differences for every measured pool (Figure 3B). In black spruce forest, the soil (SOL + mineral soil) was the dominant C pool (8.9 ± 0.5 kg C m−2), whereas in paper birch, live tree stem biomass was the largest C pool (5.2 ± 0.3 kg C m−2). Measured ecosystem N stocks were higher in black spruce relative to paper birch (364.0 ± 22.2 vs. 283.2 ± 14.9 g N m−2, Figure 3C). For both forest types, soils held the greatest proportion of ecosystem N (Figure 3D), but the larger mass of the black spruce SOL and the higher C and N concentration in black spruce mineral soils resulted in a larger N pool in spruce soils (Figure 3C). Paper birch contained larger C and N pools in all measured aboveground components, with the exception of live foliage (Figure 3B, D).
Discussion
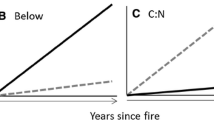
Our results illustrate large interspecific differences in plant-soil-microbial feedbacks in boreal forest that strongly influence C and nutrient turnover and distribution. In general, these findings supported our hypotheses: differences in C and nutrient pool sizes and potential N mineralization were consistent with relatively slow biogeochemical cycling in black spruce forest and more rapid C and nutrient turnover in birch stands. Small fluxes and low nutrient availability were coupled with slow tree growth and large soil OM pools in spruce, whereas large fluxes, small nutrient pool sizes, and more rapid growth characterized stands of the same age that were dominated by birch (Figure 5). Our findings suggest that moving toward greater deciduous dominance across boreal Alaska may accelerate C and nutrient cycling and shift surface soil C to aboveground pools.
Past fire history also contributed to observed differences in the SOL and measured ecosystem C stocks. In black spruce stands, about 25% of the SOL C survived the previous fire. This finding, along with the absence of pre-fire SOL C in birch stands, suggests that the current black spruce forest established on a residual SOL and that the birch-dominated stands established in areas that burned more severely. Excluding the black spruce pre-fire SOL C from our ecosystem C pool estimates indicated that SOL C accumulation in the spruce stands was still larger than in birch soils and averaged 87 g m−2 y−1 for spruce, which is generally consistent with previous studies (for example, Trumbore and Harden 1997; O’Donnell and others 2011; Turetsky and others 2011; Hicks Pries and others 2012). Re-calculating ecosystem C pools using a 25% reduction in black spruce SOL C stocks did not change our finding of no difference in total ecosystem C pools; however, these results highlight the importance of fire legacy in shaping ecosystem C dynamics and the linkage between fire legacy and boreal forest tree species composition.
Aboveground Dynamics
Many of our findings coincide with expectations stemming from known functional traits of black spruce and Alaska paper birch. The adjacent location of these contrasting forests suggests that black spruce and paper birch strongly interact with the SOL environment and influence nutrient turnover rates that feedback to influence nutrient uptake and growth. Low ANPPtree and relatively small aboveground live tree biomass in black spruce is common (Gower and others 2001; Bond-Lamberty and others 2004; Alexander and others 2012) and can be attributed to adaptation to nutrient-poor environments (Lambers and Poorter 1992), the cool summer soil temperatures (Van Cleve and others 1981, 1983b), and the low soil N availability we observed (Reich and others 1997). In contrast, the higher ANPPtree in birch likely drove the much larger aboveground live tree biomass in birch stands observed here and elsewhere (Van Cleve and others 1983b; Viereck and others 1983; Alexander and others 2012), and suggests greater nutrient uptake by this species to support high growth rates. The higher concentrations of N, P, and base cations in birch live foliage also suggest greater nutrient acquisition by this species and exhibit a pattern consistent with other studies of deciduous broad-leaf and black spruce forest in boreal ecosystems (Troth and others 1976; Van Cleve and others 1981, 1983b; Légaré and others 2005).
Although we did not directly measure moss NPP in this study, we can estimate how incorporating mosses in the black spruce stands would affect total ANPP values (ANPP for birch stands was not calculated because of sparse moss coverage). Following Turetsky and others (2010) who found that around 20% of total ANPP can be attributed to mosses in upland sites, and using our mean black spruce ANPPtree value, we estimated the mean ANPPtree+moss in black spruce stands to be approximately 240 g m−2 y−1. Although this is larger than our reported ANPPtree value (192.5 g m−2 y−1, Table 1) by about 48 g m−2 y−1, this total ANPP is still only about half the ANPP observed in birch (463.9 g m−2 y−1, Table 1), supporting our general finding of much higher ANPP in birch forest relative to black spruce.
Fluxes
The larger foliar litter C and nutrient inputs in birch, in conjunction with less SOL accumulation post-fire observed for this species (Figure 5), indicates more rapid SOL turnover than observed in spruce. We had expected to see higher potential C mineralization in paper birch soils that would indicate a larger relatively fast cycling C pool and support our expectation of higher decompositions rates. This would have also supported our observed smaller SOL C pools in this species. No interspecific differences were detected, however. Côté and others (2000) found little difference between mixed Alaska paper birch/aspen (Populus tremuloides Michx) and white spruce/balsam fir stands (Picea glauca (Moench) Voss/Abies balsamea (L.) Mill.) during a 100-day incubation and attributed this to the inverse relationship between OM quality and quantity. In our study, the large size of the SOL C pool in spruce may have had a similar absolute amount of relatively fast cycling C as found in birch, despite this fast cycling C being a much smaller proportion of the total C pool. The similarity in the Δ14C values of respired CO2 suggests a similar age of C being released from both stand types, which could also indicate the fast C cycling pool as the primary C source. It is also possible that the in situ, cooler soils observed in spruce forest had inhibited microbial activity and allowed for higher accumulation of a labile C pool which was released when we increased the soil temperature to 15°C for our incubation (Lavoie and others 2011). A longer-term incubation study would provide further insights into the decomposability of SOL C in the two stand types and the relationship between decomposition and soil C accumulation. Although our deeper (0–100 cm) mineral soil elemental concentrations did not show a trend for differences in the parent material beneath our study species, it is possible that small-scale variation in soils including soil type, moisture content, and temperature between adjacent sites could affect potential respiration rates. Bauhus and others (1998) observed differential patterns in CO2 respiration between birch and conifer species on clay vs. till soils and attributed this to factors including soil C concentration, the location of roots, and impacts of soil drainage and porosity on the microbial community.
More rapid nutrient cycling in both the SOL and upper mineral soils of Alaska paper birch is supported by our N data, showing a trend for higher potential net N mineralization and significantly higher net nitrification, larger resin-sorbed NH4 + and NO3 − pools in the SOL, and greater N mineralization and NH4 + pools in the top 10 cm of mineral soil. Higher N cycling rates in birch soils could be influenced by the higher N concentration and lower C:N ratio of birch foliar litter relative to spruce (Paré and Bergeron 1996; Côté and others 2000), higher pH in the SOL (Vance and Chapin 2001), differences in microbial community composition (Högberg and others 2007), and variation in microbial activity resulting from warmer summer soil temperatures (Vance and Chapin 2001). Relatively higher concentrations of P and base cations in birch foliar litter, larger annual foliar litter inputs, and the smaller SOL also suggest more rapid re-cycling of these nutrients in birch stands relative to spruce. Higher P mineralization in aspen/balsam poplar (Populus balsamifera L.) relative to white spruce-dominated stands has been previously documented (Lindo and Visser 2003) and both aspen (Légaré and others 2005) and mixed aspen/birch (Troth and others 1976) stands often have higher base cation concentrations than black spruce. Higher soil pH in deciduous stands was also documented in these studies. In temperate forests, others have linked larger soil P availability with higher pH, resulting from increased microbial activity and great mobilization of metals (DeForest and others 2012). Greater uptake of available soil nutrient pools could stem from higher fine root biomass in birch soils. Although we did not measure fine root biomass, others have shown higher biomass in birch stands (Yuan and Chen 2010) and a positive correlation between root biomass and resin-sorbed P concentrations (Huang and Schoenau 1997).
Moss and Belowground Pools
The presence of moss and SOL C that survived the 1958 fire in black spruce stands appears to influence observed patterns in soil C and nutrient pools and emphasizes the important linkage between mosses and black spruce in the boreal biome. Large SOL accumulation in black spruce stands has commonly been documented (for example, Troth and others 1976; Van Cleve and others 1981; Harden and others 1997; Rapalee and others 1998; Vesterdal and others 2013) and has been linked directly to the recalcitrance of moss tissue (Turetsky and others 2010), and indirectly to environmental factors via moss impacts on the soil thermal and hydrological regime (Gornall and others 2007). Despite higher concentrations of N, Mehlich P, and exchangeable Ca and Mg in the birch SOL, the large moss accumulation in spruce (likely driven by both moss NPP and relatively slow decomposition) contributed to larger soil N and Mehlich P pools and similar pool sizes for Ca and Mg. In addition to the direct effects resulting from moss mass, some mosses are associated with biological N fixation (DeLuca and others 2007; Markham 2009) and show high adsorption of P (Chapin and others 1987) and base cations (Rühling and Tyler 1970), which could further contribute to the observed accumulation of these elements in spruce soils relative to birch. Finally, our radiocarbon data indicates that a portion of the SOL in spruce was deposited prior to the 1958 fire, and therefore, the SOL mass is in part independent from the OM deposited during the development of this forest, thereby highlighting the importance of black spruce regeneration in facilitating long-term SOL C storage in boreal ecosystems. In the top 10 cm of mineral soils, higher C and N concentrations led to larger C and N pools. These higher concentrations could be the result of greater leaching and sorption of dissolved OM in spruce mineral soils (Borken and others 2011; Fröberg and others 2011). Additionally, higher fine root biomass in black spruce soils could also contribute to larger C and N stored beneath this species, as has been shown previously for black spruce relative to deciduous species (Yuan and Chen 2010; Hansson and others 2013).
Implications for Shifting Successional Trajectories
Climate warming and increases in wildfire extent and severity are expected to continue (Balshi and others 2009; Flannigan and others 2009a; Mann and others 2012) and expand deciduous forest establishment across interior Alaska (Beck and others 2011). Our results indicate that shifting from black spruce to Alaska paper birch-dominated regeneration following severe fire events could result in dramatic changes to the structure and function of boreal ecosystems. The legacy of fire and the development of contrasting, species-specific linkages between forest productivity, C and nutrient pools, and the soil environment may cause landscape-scale shifts in the distribution of ecosystem C and N pools and nutrient cycling rates (Figure 5).
Although measured ecosystem C stocks were similar for black spruce and Alaska paper birch in our mid-successional stands, the partitioning of C into above versus shallow belowground pools could lead to changes in C accumulation rates, mean residence times, and ultimately feedbacks to the global climate system. Rapid tree growth in Alaska paper birch may result in greater C sequestration aboveground during early to mid-succession; however, deciduous species exhibit reductions in NPP at a younger age than black spruce (Viereck and others 1983; Yarie and Billings 2002). Mortality and subsequent decomposition of birch tree biomass may therefore occur sooner after forest maturation, which could decrease C storage more quickly in birch relative to the longer-lived black spruce trees containing greater C stocks in soils. Further, smaller SOL accumulation and near absence of moss cover in birch could result in relatively small C accumulation in soils, a pool with some C fractions displaying decadal to millennial turnover times (Trumbore 1997). Without moss, soil may also become warmer and drier, and expose underlying permafrost C to loss, a feedback pathway that may enhance climate warming. A more deciduous landscape will also increase surface albedo and evapotranspiration (Baldocchi and others 2000; Chapin and others 2000; Amiro and others 2006; Terrier and others 2013), and may increase the fire return interval as a result of higher leaf moisture in deciduous broad-leaf species and a shallower SOL (Chapin and others 2008; Terrier and others 2013), which may act as negative feedback to climate warming.
A shift toward greater deciduous cover also has implications for N cycling and ecosystem productivity. Although birch stands displayed smaller total N stocks than black spruce, N mineralization and nitrification rates were higher, suggesting that a greater fraction of N, especially NO3 −, is vulnerable to losses via leaching. Despite N being a limiting nutrient in boreal forest, N losses have been shown to exceed inputs (Jones and others 2005). Additionally, continued high tree growth rates in our mid-successional stands could further increase N stocks stored in aboveground woody tissue and reduce soil N pools, further constraining NPP. However, rapid turnover of large, nutrient rich, annual foliar litter inputs might also allow for higher nutrient availability to be sustained if tree uptake remains high.
Conclusions
Our results indicate that a shift toward greater deciduous dominance following high severity fires may cause large changes in plant-soil-microbial feedbacks and ecosystem structure. Although our study focused on forest regeneration following a single fire event, our findings are likely to represent the type of impacts shifting successional trajectories will create across Alaska’s boreal landscape. By quantifying C and nutrient pools and fluxes, and improving our understanding of potential shifts in C and nutrient distribution, this research can contribute to improved predictions for how changes in the wildfire regime will affect boreal ecosystems.
References
Aerts R. 1995. The advantages of being evergreen. Trends Ecol Evol 10:402–7.
Alexander HD, Mack MC. A canopy shift in interior Alaskan boreal forests: consequences for above—and belowground carbon and nitrogen pools during post—fire succession. Ecosystems (in press).
Alexander HD, Mack MC, Goetz S, Beck PSA, Belshe EF. 2012. Implications of increased deciduous cover on stand structure and aboveground carbon pools of Alaskan boreal forests. Ecosphere 3:1–21.
Amiro BD, Orchansky AL, Barr AG, Black TA, Chambers SD, Chapin FS, Gouldenf ML, Litvakg M, Liu HP, McCaughey JH, McMillan A, Randerson JT. 2006. The effect of post-fire stand age on the boreal forest energy balance. Agric For Meteorol 140:41–50.
Baldocchi D, Kelliher FM, Black TA, Jarvis P. 2000. Climate and vegetation controls on boreal zone energy exchange. Glob Change Biol 6:69–83.
Balshi MS, McGuirez AD, Duffy P, Flannigan M, Walsh J, Melillo J. 2009. Assessing the response of area burned to changing climate in western boreal North America using a multivariate adaptive regression splines (MARS) approach. Glob Change Biol 15:578–600.
Bauhus J, Paré D, Côté L. 1998. Effects of tree species, stand age and soil type on soil microbial biomass and its activity in a southern boreal forest. Soil Biol Biochem 30:1077–89.
Beck PSA, Goetz SJ, Mack MC, Alexander HD, Jin YF, Randerson JT, Loranty MM. 2011. The impacts and implications of an intensifying fire regime on Alaskan boreal forest composition and albedo. Glob Change Biol 17:2853–66.
Berner LT, Alexander HD, Loranty MM, Ganzlin P, Mack MC, Davydov SP, Goetz SJ. 2015. Biomass allometry for alder, dwarf birch, and willow in boreal forest and tundra ecosystems of far northeastern Siberia and north-central Alaska. For Ecol Manage 337:110–18.
Boby LA, Schuur EAG, Mack MC, Verbyla D, Johnstone JF. 2010. Quantifying fire severity, carbon, and nitrogen emissions in Alaska’s boreal forest. Ecol Appl 20:1633–47.
Bond-Lamberty B, Gower ST, Wang C, Cyr P, Veldhuis H. 2006. Nitrogen dynamics of a boreal black spruce wildfire chronosequence. Biogeochemistry 81:1–16.
Bond-Lamberty B, Wang CK, Gower ST. 2004. Net primary production and net ecosystem production of a boreal black spruce wildfire chronosequence. Glob Change Biol 10:473–87.
Borken W, Ahrens B, Schulz C, Zimmermann L. 2011. Site-to-site variability and temporal trends of DOC concentrations and fluxes in temperate forest soils. Glob Change Biol 17:2428–43.
Bowman WD, Theodose TA, Schardt JC, Conant RT. 1993. Constraints of nutrient availability on primary production in 2 alpine tundra communities. Ecology 74:2085–97.
Brown JK. 1974. Handbook for inventorying downed woody material. Ogden, Utah: USDA Forest Service, Intermountain Forest and Range Experiment Station.
Chapin FSIII, Trainor SF, Huntington O, Lovecraft AL, Zavaleta E, Natcher DC, McGuire AD, Nelson JL, Ray L, Calef M, Fresco N, Huntington H, Rupp TS, Lo Dewilde, Naylor RL. 2008. Increasing wildfire in Alaska’s boreal forest: pathways to potential solutions of a wicked problem. Bioscience 58:531–40.
Chapin FS, McGuire AD, Randerson J, Pielke R, Baldocchi D, Hobbie SE, Roulet N, Eugster W, Kasischke E, Rastetter EB, Zimov SA, Running SW. 2000. Arctic and boreal ecosystems of western North America as components of the climate system. Glob Change Biol 6:211–23.
Chapin FS, Oechel WC, Van Cleve K, Lawrence W. 1987. The role of mosses in the phosphorus cycling of an Alaskan black spruce forest. Oecologia 74:310–15.
Chapin FSI, Oswood MW, Van Cleve K, Viereck LA, Verbyla DL, Eds. 2006. Alaska’s changing boreal forest. New York: Oxford University Press.
Côté L, Brown S, Paré D, Fyles J, Bauhus J. 2000. Dynamics of carbon acid nitrogen mineralization in relation to stand type, stand age and soil texture in the boreal mixedwood. Soil Biol Biochem 32:1079–90.
DeForest JL, Smemo KA, Burke DJ, Elliott HL, Becker JC. 2012. Soil microbial responses to elevated phosphorus and pH in acidic temperate deciduous forests. Biogeochemistry 109:189–202.
DeLuca TH, Zackrisson O, Gentili F, Sellstedt A, Nilsson M-C. 2007. Ecosystem controls on nitrogen fixation in boreal feather moss communities. Oecologia 152:121–30.
Dyrness CT, Norum RA. 1983. The effects of experimental fires on black spruce forest floors in interior Alaska. Can J For Res 13:879–93.
Flanagan PW, Van Cleve K. 1983. Nutrient cycling in relation to decomposition and organic-matter quality in taiga ecosystems. Can J For Res 13:795–817.
Flannigan M, Stocks B, Turetsky M, Wotton M. 2009a. Impacts of climate change on fire activity and fire management in the circumboreal forest. Glob Change Biol 15:549–60.
Flannigan MD, Krawchuk MA, de Groot WJ, Wotton BM, Gowman LM. 2009b. Implications of changing climate for global wildland fire. Int J Wildland Fire 18:483–507.
Fröberg M, Hansson K, Kleja DB, Alavi G. 2011. Dissolved organic carbon and nitrogen leaching from scots pine, Norway spruce and silver birch stands in southern Sweden. For Ecol Manage 262:1742–7.
Gornall JL, Jónsdóttir IS, Woodin SJ, Van der Wal R. 2007. Arctic mosses govern below-ground environment and ecosystem processes. Oecologia 153:931–41.
Gower ST, Krankina O, Olson RJ, Apps M, Linder S, Wang C. 2001. Net primary production and carbon allocation patterns of boreal forest ecosystems. Ecol Appl 11:1395–411.
Greene DF, Macdonald SE, Haeussler S, Domenicano S, Nöel J, Jayen K, Charron I, Gauthier S, Hunt S, Gielau ET, Bergeron Y, Swift L. 2007. The reduction of organic-layer depth by wildfire in the North American boreal forest and its effect on tree recruitment by seed. Can J For Res 37:1012–23.
Hansson K, Helmisaari H-S, Sah SP, Lange H. 2013. Fine root production and turnover of tree and understorey vegetation in Scots pine, silver birch and Norway spruce stands in SW Sweden. For Ecol Manage 309:58–65.
Harden JW, O’Neill KP, Trumbore SE, Veldhuis H, Stocks BJ. 1997. Moss and soil contributions to the annual net carbon flux of a maturing boreal forest. J Geophys Res Atmos 102:28805–16.
Hicks Pries CE, Schuur EAG, Crummer KG. 2012. Holocene carbon stocks and carbon accumulation rates altered in soils undergoing permafrost thaw. Ecosystems 15:162–73.
Hobbie SE. 1992. Effects of plant species on nutrient cycling. Trends Ecol Evol 7:336–9.
Hobbie SE, Nadelhoffer KJ, Högberg P. 2002. A synthesis: the role of nutrients as constraints on carbon balances in boreal and arctic regions. Plant Soil 242:163–70.
Hobbie SE, Schimel JP, Trumbore SE, Randerson JR. 2000. Controls over carbon storage and turnover in high-latitude soils. Glob Change Biol 6:196–210.
Högberg MN, Högberg P, Myrold DD. 2007. Is microbial community composition in boreal forest soils determined by pH, C-to-N ratio, the trees, or all three? Oecologia 150:590–601.
Huang WZ, Schoenau JJ. 1997. Seasonal and spatial variations in soil nitrogen and phosphorus supply rates in a boreal aspen forest. Can J Soil Sci 77:597–612.
Johnstone JF, Chapin FS, Hollingsworth TN, Mack MC, Romanovsky V, Turetsky M. 2010a. Fire, climate change, and forest resilience in interior Alaska. Can J For Res 40:1302–12.
Johnstone JF, Hollingsworth TN, Chapin FSIII, Mack MC. 2010b. Changes in fire regime break the legacy lock on successional trajectories in Alaskan boreal forest. Glob Change Biol 16:1281–95.
Jones JB, Petrone KC, Finlay JC, Hinzman LD, Bolton WR. 2005. Nitrogen loss from watersheds of interior Alaska underlain with discontinuous permafrost. Geophys Res Lett 32:L02401.
Jorgenson MT, Romanovsky V, Harden J, Shur Y, O’Donnell J, Schuur EAG, Kanevskiy M, Marchenko S. 2010. Resilience and vulnerability of permafrost to climate change. Can J For Res 40:1219–36.
Kasischke E, Verbyla D, Rupp TS, McGuire AD, Murphy KA, Jandt R, Barnes JL, Hoy EE, Duffy PA, Calef M, Turetsky MR. 2010. Alaska’s changing fire regime—implications for the vulnerability of its boreal forests. Can J For Res 40:1313–24.
Kelly R, Chipman ML, Higuera PE, Stefanova I, Brubaker LB, Hu FS. 2013. Recent burning of boreal forests exceeds fire regime limits of the past 10,000 years. Proc Natl Acad Sci USA 110:13055–60.
Lambers H, Poorter H. 1992. Inherent variation in growth rate between higher plants: a search for physiological causes and ecological consequences. Adv Ecol Res 23:187–261.
Lavoie M, Mack MC, Schuur EAG. 2011. Effects of elevated nitrogen and temperature on carbon and nitrogen dynamics in Alaskan arctic and boreal soils. J Geophys Res Biogeosci 116:G03013.
Légaré S, Paré D, Bergeron Y. 2005. Influence of aspen on forest floor properties in black spruce-dominated stands. Plant Soil 275:207–20.
Lindo Z, Visser S. 2003. Microbial biomass, nitrogen and phosphorus mineralization, and mesofauna in boreal conifer and deciduous forest floors following partial and clear-cut harvesting. Can J For Res 33:1610–20.
Lynch JA, Clark JS, Bigelow NH, Edwards ME, Finney BP. 2002. Geographic and temporal variations in fire history in boreal ecosystems of Alaska. J Geophys Res Atmos 108:8-1–-17.
Mack MC, Bret-Harte MS, Hollingsworth TN, Jandt RR, Schuur EAG, Shaver GR, Verbyla DL. 2011. Carbon loss from an unprecedented arctic tundra wildfire. Nature 475:489–92.
Mann DH, Rupp TS, Olson MA, Duffy PA. 2012. Is Alaska’s boreal forest now crossing a major ecological threshold? Arct Antarct Alp Res 44:319–31.
Markham JH. 2009. Variation in moss-associated nitrogen fixation in boreal forest stands. Oecologia 161:353–9.
Melillo JM, Aber JD, Muratore JF. 1982. Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–6.
O’Donnell JA, Harden JW, McGuire AD, Kanevskiy MZ, Jorgenson MT, Xu XM. 2011. The effect of fire and permafrost interactions on soil carbon accumulation in an upland black spruce ecosystem of interior Alaska: implications for post-thaw carbon loss. Glob Change Biol 17:1461–74.
O’Donnell JA, Romanovsky VE, Harden JW, McGuire AD. 2009. The effect of moisture content on the thermal conductivity of moss and organic soil horizons from black spruce ecosystems in interior Alaska. Soil Sci 174:646–51.
Paré D, Bergeron Y. 1996. Effect of colonizing tree species on soil nutrient availability in a clay soil of the boreal mixedwood. Can J For Res 26:1022–31.
Quesnel PO, Côté B. 2009. Prevalence of phosphorus, potassium, and calcium limitations in white spruce across Canada. J Plant Nutr 32:1290–305.
Rapalee G, Trumbore SE, Davidson EA, Harden JW, Veldhuis H. 1998. Soil carbon stocks and their rates of accumulation and loss in a boreal forest landscape. Global Biogeochem Cycles 12:687–701.
Rau BM, Melvin AM, Johnson DW, Goodale CL, Blank RR, Fredriksen G, Miller WW, Murphy JD, Todd DE Jr, Walker RF. 2011. Revisiting soil carbon and nitrogen sampling: quantitative pits versus rotary cores. Soil Sci 176:273–9.
Reich PB, Grigal DF, Aber JD, Gower ST. 1997. Nitrogen mineralization and productivity in 50 hardwood and conifer stands on diverse soils. Ecology 78:335–47.
Rocca ME, Miniat CF, Mitchell RJ. 2014. Introduction to the regional assessments: climate change, wildfire, and forest ecosystem services in the USA. For Ecol Manage 327:265–8.
Rühling A, Tyler G. 1970. Sorption and retention of heavy metals in woodland moss Hylocomium splendens (Hedw.) Br. et Sch. Oikos 21:92–7.
Schuur EAG, Vogel JG, Crummer KG, Lee H, Sickman JO, Osterkamp TE. 2009. The effect of permafrost thaw on old carbon release and net carbon exchange from tundra. Nature 459:556–9.
Shetler G, Turetsky MR, Kane ES, Kasischke E. 2008. Sphagnum mosses limit total carbon consumption during fire in Alaskan black spruce forests. Can J For Res 38:2328–36.
Terrier A, Girardin MP, Périé C, Legendre P, Bergeron Y. 2013. Potential changes in forest composition could reduce impacts of climate change on boreal wildfires. Ecol Appl 23:21–35.
Troth JL, Deneke FJ, Brown LM. 1976. Upland aspen/birch and black spruce stands and their litter and soil properties in interior Alaska. Forest Science 22:33–44.
Trumbore SE. 1997. Potential responses of soil organic carbon to global environmental change. Proc Natl Acad Sci USA 94:8284–91.
Trumbore SE, Harden JW. 1997. Accumulation and turnover of carbon in organic and mineral soils of the BOREAS northern study area. J Geophys Res Atmos 102:28817–30.
Turetsky MR, Bond-Lamberty B, Euskirchen E, Talbot J, Frolking S, McGuire AD, Tuittila ES. 2012. The resilience and functional role of moss in boreal and arctic ecosystems. New Phytol 196:49–67.
Turetsky MR, Kane ES, Harden JW, Ottmar RD, Manies KL, Hoy E, Kasischke ES. 2011. Recent acceleration of biomass burning and carbon losses in Alaskan forests and peatlands. Nat Geosci 4:27–31.
Turetsky MR, Mack MC, Hollingsworth TN, Harden JW. 2010. The role of mosses in ecosystem succession and function in Alaska’s boreal forest. Can J For Res 40:1237–64.
U.S. Department of Agriculture NRCS. 2013. Web Soil Survey, soil map—greater Nenana Area, Alaska, and North Star Area, Alaska.
Underwood AJ. 1997. Experiments in ecology. Cambridge: Cambridge University Press. p 504p.
Van Cleve K, Barney R, Schlentner R. 1981. Evidence of temperature control of production and nutrient cycling in 2 interior Alaska black spruce ecosystems. Can J For Res 11:258–73.
Van Cleve K, Dyrness CT, Viereck LA, Fox J, Chapin FS, Oechel W. 1983a. Taiga ecosystems in interior Alaska. Bioscience 33:39–44.
Van Cleve K, Oliver L, Schlentner R, Viereck LA, Dyrness CT. 1983b. Productivity and nutrient cycling in taiga forest ecosystems. Can J For Res 13:747–66.
Vance ED, Chapin FS. 2001. Substrate limitations to microbial activity in taiga forest floors. Soil Biol Biochem 33:173–88.
Vesterdal L, Clarke N, Sigurdsson BD, Gundersen P. 2013. Do tree species influence soil carbon stocks in temperate and boreal forests? For Ecol Manage 309:4–18.
Viereck LA, Dyrness CT, Van Cleve K, Foote MJ. 1983. Vegetation, soils, and forest productivity in selected forest types in interior Alaska. Can J For Res 13:703–20.
Yarie J, Billings S. 2002. Carbon balance of the taiga forest within Alaska: present and future. Can J For Res 32:757–67.
Yuan ZY, Chen HYH. 2010. Fine root biomass, production, turnover rates, and nutrient contents in boreal forest ecosystems in relation to species, climate, fertility, and stand age: literature review and meta-analyses. Crit Rev Plant Sci 29:204–21.
Acknowledgements
We thank Camilo Mojica, Samantha Miller, Demetra Panos, Bethany Avera, Simon McClung, Alicia Sendrowski, Peter Ganzlin, Melanie Jean, and many additional undergraduate assistants at the University of Florida for help in the field and laboratory. Grace Crummer and Julia Reiskind provided analytical assistance, Heather Alexander generously shared unpublished tree C and N concentration data, and Rosvel Bracho provided guidance in setting up the incubation experiment and processing acquired data. We also thank Jamie Hollingsworth for logistical help. Funding for this research was provided by the Department of Defense’s Strategic Environmental Research and Development Program (SERDP) under project RC-2109. This study was also supported by the Bonanza Creek Long Term Ecological Research Program, which is jointly funded by the National Science Foundation and the U.S. Department of Agriculture Forest Service. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
AMM led the field, laboratory, and data analysis and drafted the manuscript with guidance from MCM. MCM, JFJ, and EAGS developed the research questions and study approach. ADM and HG provided advice on data collection and all authors provided input to this manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Melvin, A.M., Mack, M.C., Johnstone, J.F. et al. Differences in Ecosystem Carbon Distribution and Nutrient Cycling Linked to Forest Tree Species Composition in a Mid-Successional Boreal Forest. Ecosystems 18, 1472–1488 (2015). https://doi.org/10.1007/s10021-015-9912-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-015-9912-7