Abstract
Malignant lymphoma of the cranial vault is a rare entity and the tumor growth patterns are not well understood. Here we report two cases of malignant lymphoma involving the scalp and epidural space with slight changes in the intervening skull. A 63-year-old woman presented with a scalp mass in her right frontal area. Computed tomography (CT) and magnetic resonance (MR) imaging demonstrated mass lesions in the scalp and epidural space with slight osteolytic changes in the intervening skull. She underwent resection of the lesions. A 53-year-old man presented with a mass in his right frontal area. CT and MR imaging showed mass lesions in the scalp and epidural space without changes in the skull. He underwent resection of the lesions. The histopathological diagnosis was diffuse large B cell lymphoma in both cases. Microscopic examination of the intervening skull found that the bone marrow was diffusely replaced by lymphoma cells, and lymphoma cells extended to the extra- or intra-cranial space along the emissary veins without destruction of the cortical or trabecular bone. These histopathological findings explain the radiological findings and provide the clues to elucidate the mechanism of extension of cranial vault lymphoma.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Malignant lymphoma of the cranial vault is rare. 59 cases have been reported so far [1–7]. Recently, two systematic reviews pointed out that this entity might involve both scalp and epidural spaces without significant destruction of the skull in the large proportion of the many cases. However, no reports have demonstrated the pathological findings of the intervening skull.
Here we report two cases of malignant lymphoma of the cranial vault with minimal changes in the intervening skull and show the pathological findings of the skull.
Case reports
Case 1
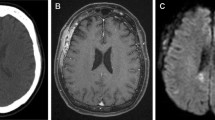
A 63-year-old woman had a 4-month history of mass lesion in her right frontal area (Fig. 1a). The skin over the lesion was normal, and she had no neurological deficits. Her cervical lymph nodes were swollen. Testing for antibody to human immunodeficiency virus was negative. Bone computed tomography (CT) revealed a mass lesion in the right frontal scalp with slight osteolytic changes in the skull (Fig. 1b). Magnetic resonance (MR) imaging showed lesions in both the scalp and epidural space as hypointense on T1-weighted imaging and hyperintense on T2-weighted imaging (Fig. 1c, d). These masses were homogeneously enhanced by gadolinium (Gd) (Fig. 1e). The diploe was partially enhanced (Fig. 1e). Preoperative CT scan showed neither hepatomegaly nor splenomegaly. Biopsy from iliac bone marrow and cervical lymph node showed no evidence of lymphoma cells. 67Ga whole-body scintigraphy showed uptake of 67Ga in the cranial mass, not in bilateral cervical lymph nodes. Preoperative diagnosis was meningioma, metastatic skull tumor, or mesenchymal tumor. Based on preoperative diagnosis, the lesions in the scalp, skull, and epidural space were resected. Intraoperatively, a mass lesion was found under the galea aponeurotica. Tumor did not invade to the surface of galea aponeurotica, and periosteum and aponeurotica adhered tightly to the tumor. As a result, the origin of the tumor was not identified, intraoperatively. After removal of scalp lesion, many small holes were found in the skull. The involved skull was removed. A tumor was found on the dura without invasion to the arachnoid or brain parenchyma. This tumor was subtotally resected. The patient received subsequent chemotherapy with cyclophosphamide, adriamycin, vincristine, and prednisolone (CHOP). The patient remained free from signs of disease recurrence at a 9-year follow-up examination.
Macroscopic and radiological findings of case 1. a Photograph demonstrating the gradually growing subcutaneous mass in the right frontal area. b Computed tomography scan of the bone demonstrating the mass lesion in the right frontal scalp with slight osteolytic changes of the skull. Magnetic resonance images demonstrating the extra-cranial and intra-cranial masses as hypointense on T1-weighted imaging (c) and hyperintense on T2-weighted imaging (d). These masses were homogeneously enhanced by gadolinium (e)
Case 2
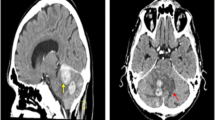
A 53-year-old man had 2-month history of mass lesion in his right frontal area (Fig. 2a). The skin over the lesion was normal, and he had no neurological deficits. Testing for antibody to human immunodeficiency virus was negative. Brain and bone CT revealed a mass lesion in the right frontal scalp and epidural space without changes in the skull (Fig. 2b, c). MR imaging revealed extra- and intra-cranial masses as hypointense on T1-weighted imaging and hyperintense on T2-weighted imaging (Fig. 2d, e). These masses were heterogeneously enhanced by Gd (Fig. 2f). 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET) showed uptake of FDG in the cranial mass lesion (maximum standardized uptake value (SUVmax) 9.5) and the bilateral cervical lymph nodes (SUVmax 3.5). The result of FDG-PET did not provide a definitive finding for differentiating lymphoma from non-specific inflammatory changes. Physical examination revealed no evidence of lymphadenopathy. Preoperative CT scan showed neither hepatomegaly nor splenomegaly. Biopsy from iliac bone marrow showed no evidence of lymphoma cells. Preoperative differential diagnosis was meningioma, metastatic skull tumor, mesenchymal tumor or malignant lymphoma. The lesions located in the scalp, skull, and epidural space were resected. Intraoperatively, a mass lesion was firmly attached to the galea aponeurotica and subaponeurotic tissue, and the origin of the tumor was not identified. After removal of this lesion, many small holes were found in the skull. A tumor was found on the dura, but without invasion to the arachnoid or brain parenchyma. Intraoperative pathological analysis did not rule out the possibility of malignant mesenchymal tumor, and we decided to perform debulking surgery. This tumor involved the superior sagittal sinus so subtotal resection was performed. The patient received subsequent chemotherapy with rituximab and CHOP (R-CHOP). The patient had local recurrence 7 months after surgery and received 40 Gy of radiation therapy to the tumor bed. Complete response was obtained after radiation therapy, and he is free from second recurrence 17 months after surgery.
Macroscopic and radiological findings of case 2. a Photograph demonstrating the gradually growing subcutaneous mass in the right frontal region. Computed tomography scans of the brain (b) and bone (c) demonstrating the mass lesion in the right frontal scalp and epidural space without changes in the skull. Magnetic resonance images demonstrating the extra-cranial and intra-cranial masses as hypointense on T1-weighted imaging (d) and hyperintense on T2-weighted imaging (e). These masses were heterogeneously enhanced by gadolinium (f)
Pathological investigations
After fixation in 10 % formalin overnight, the resected tissues were embedded in paraffin with routine methods and 3-µm thick sections were cut. Bone tissues were decalcified in 10 % formic acid solution for 3 weeks before being embedded in paraffin. The sections were stained with hematoxylin and eosin. Immunohistochemistry was performed using an automated immunostainer (Roche, Tokyo, Japan) following the manufacturer’s instructions with antigen retrieving treatments. The antibodies used were as follows: CD3 (DAKO, polyclonal, 1:50 dilution), CD20 (DAKO, L26, 1:100 dilution), CD79a (DAKO, L26, 1:100 dilution), CD10 (Leica, L26, 1:100 dilution), Bcl-2 (DAKO, L26, 1:100 dilution), Bcl-6 (DAKO, L26, 1:20 dilution), MUM-1 (DAKO, L26, 1:100 dilution).
Pathological findings
The tumors in both cases consisted of large lymphoid cells with large nucleus and prominent nucleolus, and these cells proliferated diffusely (Fig. 3a, b). Immunohistochemical analysis revealed that the tumor cells were positive for CD20 (Fig. 3c) and negative for CD3. The histological diagnosis was diffuse large B cell lymphoma (DLBCL) in both cases. Histological examination of the intervening skull found minimal destruction of the cortical and trabecular bone in both cases (Fig. 3d), and bone marrow was replaced by lymphoma cells (Fig. 3g, h). Mild to moderate reparative changes with osteolysis and hyperostosis were identified. These changes in the trabecular bones with mild fibrosis of the bone marrow were slightly more conspicuous in case 2. Tumor cell extension to the extra-cranial space along the emissary veins was often observed in both cases (Fig. 3d–f, arrows).
Histological findings of the tumor and intervening skull specimens. a, b H&E staining of the resected bone demonstrating diffuse infiltrative growth of large atypical lymphoid cells with prominent nucleus and nucleolus which had proliferated diffusely. c Immunohistochemical analysis for CD 20 demonstrating that the tumor cells were positive for B lymphocytic marker. H&E staining of the emissary vein in case 1 (d, e) and case 2 (f), demonstrating tumor cells extending along the emissary vein (arrows). g, h H&E staining of the diploe demonstrating that the bone marrow was replaced by lymphoma cells without destruction of the trabecular bones. Scale bars 10 µm in a–c, 200 µm in d, 75 µm in e, 50 µm in f, and 100 µm in g and h
As there have been few reports demonstrating the expression of the prognostic markers in the cranial vault lymphoma, we examined the expression of CD10, Bcl-6 and MUM-1. Both cases showed negative for CD10 and MUM-1. Bcl-6 expression was negative in case 1 and positive in case 2 (Table 1). Ki-67 labeling index was quite high in both cases (Table 1).
Discussion
In this report, we described two cases with cranial vault lymphoma. It was difficult to decide whether these cases were primary or secondary infiltration of systemic disease. There were no physical or radiological findings of systemic disease, except swelling cervical lymph node in case 1 and abnormal uptake of 18FDG in cervical lymph node of case 2. Considering the absence of lymphoma cells in the specimen from cervical lymph node in case 1 and relatively lower 18FDG uptake of cervical lymph node in case 2, both cases in this report were considered to be primary cranial vault lymphoma.
According to a previous report [1], findings of the skull in 2 cases with primary and 3 cases with secondary infiltration of systemic disease were described in detail. Both of primary and secondary skull lesions exhibit identical features, including permeative skull lesion without destruction of cortical bone, and multifocal lesions. However, patients with secondary infiltration of systemic disease had poorer prognosis compared with primary cranial vault lymphoma [1].
Previous cases have demonstrated the radiological findings of malignant lymphoma of the cranial vault [1, 2, 8]. Dural or cranial vault lymphomas involve both the extra- and intra-cranial spaces [1–16], and the two cases in this report also showed tumor extension to both extra- and intra-cranial spaces. In addition, the intervening skull shows characteristic findings in malignant lymphoma [1, 2, 8, 9, 11, 14, 16–19]. Bone CT showed osteolytic changes in 74 % of cases and hyperostotic changes in 5 %, whereas 18 % had normal cranial vault [2]. Such absence of bone erosion or osteolysis may be a characteristic finding of malignant lymphoma of the cranial vault [1, 8, 9, 11, 18]. Since bone CT in our two cases showed tumor extension to both the extra- and intra-cranial spaces and slight changes in the intervening skull, our cases illustrate typical radiological findings of malignant lymphoma of the cranial vault.
These radiological characteristics are well understood, but how lymphoma cells extend to the scalp, intervening skull, and epidural space remains unknown. We reviewed the intraoperative and histopathological findings of the intervening skull to elucidate the structural and histological changes in the intervening bones and the mechanisms of tumor growth and invasion of malignant lymphomas. Our histopathological analysis of the intervening skull showed lymphoma cells infiltrated the intra-diploe space by extending along the emissary veins without destroying the overall bone structure. We consider that this permeating growth pattern is important finding for understanding the mechanisms of tumor growth or invasion of malignant lymphoma of the cranial vault. Similarly, lymphoma have a permeative growth pattern into the subarachnoid space along Virchow–Robinson space of perforating vessels [9, 12], periosteum of femur [20], and long bone [21].
Case 2 had early recurrence immediately after completion of R-CHOP chemotherapy. He received salvage radiation therapy and had remission of recurrent disease. Some previous reports demonstrated outcomes of CHOP chemotherapy with or without rituximab in the treatment of cranial vault DLBCL [4–7, 11–13, 22]. Despite variable treatment strategies and small number of cases, all of the cases with target lesion had good response to the therapy [7, 11, 13, 22] and did not develop the recurrence for 6 months to 3 years [4–6, 12, 13]. However, there were cases, in which aggressive recurrence had developed even during initial chemotherapy [7]. To predict such cases, we analyzed the expression profiles of predictive markers for prognosis. According to the previous report, the expression pattern of CD10, Bcl-6 and MUM-1 can be predictive of survival in DLBCL [23]. Based on the expression profile, DLBCL can be classified into germinal B cell like (GCB) group and non-GCB group, and the former had better overall survival rate than that of the latter [23]. Cases 1 and 2 were classified into non-GCB and GCB group, respectively. This result did not correspond to their clinical course. To elucidate the significance of this classification in the cranial vault lymphoma, long-term follow-up and accumulation of cases are indispensable.
In conclusion, malignant lymphoma of the cranial vault showed a permeating growth pattern along the space of the bone marrow and emissary veins without destruction of the skull. Minimum changes in the intervening skull on CT reflected this growth pattern.
References
da Rocha AJ, da Rocha TM, da Silva CJ, Paes RP, Bruniera P, Chiattone CS (2010) Cranial vault lymphoma: a systemic review of five patients. J Neurooncol 100:9–15
El Asri AC, Akhaddar A, Baallal H, Boulahroud O, Mandour C, Chahdi H, Mikdame M, Gazzaz M, El Mostarchid B, Boucetta M (2012) Primary lymphoma of the cranial vault: case report and a systemic review of the literature. Acta Neurochir (Wien) 154:257–265
Khalid M, Siddiqui MA, Qaseem SM, Ahmad M, Khalid S (2010) Multifocal primary lymphoma of the cranial vault in a non-immunocompromised adolescent. JBR-BTR 93:296–298
Salunke P, Garg R, Bal A, Kedia S, Bindal S (2013) Primary malignant non-Hodgkin’s lymphoma of the skull vault in an immunocompetent patient. Neurol India 61:201–204
Ko MJ, Hwang SN, Park YS, Nam TK (2013) Primary malignant lymphoma of the cranial vault with extra- and intracranial extension. Brain Tumor Res Treat 1(1):32–35
Muin IA, Safari HM, Hasimah YN (1997) Primary non-Hodgkin’s lymphoma of the crnial vault mimicking a meningioma: a case report. Med J Malaysia 52(1):86–88
Horstman AL, Go RS, Bottner WA (2004) Cranial vault lymphoma. Haematologia 89(3):e42
Ochiai H, Kawano H, Miyaoka R, Kawano N, Shimao Y, Kawasaki K (2010) Primary diffuse large B-cell lymphomas of the temporoparietal dura mater and scalp without intervening skull bone invasion. Neurol Med Chir (Tokyo) 50:595–598
Yuki K, Kodama Y, Onda J, Emoto K, Kirimoto K, Yamane T, Sasaki N, Nanba K, Kamada N (1989) Metastatic malignant lymphoma of the skull with extradural and extracranial extension. Case report. Neurol Med Chir (Tokyo) 29:762–766
Tomaszek DE, Tyson GW, Stang P, Bouldin T (1984) Contiguous scalp, skull, and epidural Hodgkin’s disease. Surg Neurol 21:182–184
Holtas S, Monajati A, Utz R (1985) Computed tomography of malignant lymphoma involving the skull. J Comput Assist Tomogr 9:725–727
Morioka T, Tashima T, Nishio S, Nishie E, Fukui M, Okamura T (1994) Malignant lymphoma of the scalp at the site of a previous blunt trauma: report of two cases. Surg Neurol 42:117–120
Nishimoto T, Yuki K, Sasaki T, Imada Y, Murakami T, Kodama Y (2003) A case of subcutaneous malignant lymphoma with dura mater lesion. No Shinkei Geka 31:43–47 (Japanese)
Fukushima Y, Oka H, Utsuki K, Nakahara K, Fujii K (2007) Primary malignant lymphoma of the cranial vault. Acta Neurochir (Wien) 149:601–604
Viella LM, Blanco-Salazar A, Caballero R, Borbolla-Escoboza R (2007) Aggressive lymphoma involving intracranial epidural region. J Neurooncol 83:181–182
Gonzalez-Bonet LG, Gutierrez-Herrera JM, Gallego JM, Barcia JA (2008) Primary immunoblastic B-cell lymphoma of the cranial vault. Acta Neurochir (Wien) 150:507–508
Yoon S, Paek SH, Park S, Kim DG, Jung H (2008) Non-Hodgkin lymphoma of the cranial vault with retrobulbar metastasis mimicking a subacute hematoma. J Neurosurg 108:1018–1020
Razaei-Kalantari K, Samimi K, Jafari M, Karimi MA, Ansari K, Davoodi M, Nabi-Meybodi M, Gorjian M (2012) Primary diffuse large B cell lymphoma of the cranial vault. Iran J Radiol 9:88–92
Kosugi S, Kume M, Sato J, Sakuma I, Moroi J, Izumi K, Sato Y, Nakamura N, Takahashi M, Miura I (2013) Diffuse large B-cell lymphoma with mass lesions of skull vault and ileocecum. J Clin Exp Hematop 53:215–219
Abdelwahab IF, Hoch B, Hermann G, Bianchi S, Klein MJ, Springfield DS (2007) Primary periosteal lymphoma-rare and unusual- Skeletal Radiol 36:335–339
Mulligan ME, McRae GA, Murphey MD (1999) Imaging features of primary lymphoma of bone. AJR Am J Roentgenol 173:1691–1697
Martin J, Ramesh A, Kamaludeen M, Udhaya Ganesh K, Martin JJ (2012) Primary non-Hodgkin’s lymphoma of the scalp and cranial vault. Case Rep Neurol Med 2012:616813
Christine PH, Dennis DW, Timothy CG, Randy DG, Jan D, German O, Konrad M, Elias C, Rita MB, Elaine SJ, Zenggang P, Pedro F, Lynette MS, Brunangelo F, Alison HB, Andreas R, Louis MS, Joseph MC, James OA, Wing CC (2004) Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 103:275–282
Conflict of interest
The authors have no conflicting remarks.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tashiro, R., Kanamori, M., Suzuki, H. et al. Diffuse large B cell lymphoma of the cranial vault: two case reports. Brain Tumor Pathol 32, 275–280 (2015). https://doi.org/10.1007/s10014-015-0225-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10014-015-0225-5