Abstract
Objectives
The aim of this study was to assess the relationship between erosive tooth wear (ETW) and possible etiological factors in a group of dental students.
Materials and methods
A total of 126 dental students from a public dental school were included in this study. A questionnaire was used to investigate the possible etiological factors related to ETW. The Basic Erosive Wear Examination (BEWE) criteria were used to examine the status of ETW. A univariable and multivariable logistic regression models were used to assess the relationship between the presence of ETW and explanatory variables.
Results
Univariable analysis revealed that taking acidic foods and alcohol more than 4–5 times per week increases the odds of ETW by 6.043 and 2.532 times, respectively, and taking dairy products, fruit juice, and milk more than 4–5 times per week decreases the likelihood of ETW by 61%, 66%, and 80%, respectively. The results of multivariable regression analysis showed that the frequency of consumption of especially acidic foods significantly increased the risk of ETW (OR = 9.981, 95% CI 3.577–27.849, p < 0.001).
Conclusions
Although the ETW status of dental students, who are the future dentists, varies depending on different possible etiological factors, especially the consumption of acidic foods has increased the risk of ETW approximately 10 times.
Clinical relevance
The findings highlight the high relevance of ETW, especially with acidic food consumption, and the importance of controlling potential etiological factors in dental students.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Erosive tooth wear (ETW) is described as the loss of hard tissues of teeth owing to various acidic factors without any bacterial involvement. In clinical examination, eroded tooth surfaces differ from the original tooth morphology in some specific features [1]. Intrinsic acids caused by endogenous acids such as gastroesophageal reflux or extrinsic acids usually caused by various nutritional habits, especially acidic beverages, or industrial/environmental chemicals, can lead to ETW [2]. Based on the origin of the erosion-causing acids, a distinction between ETW originating from intrinsic and extrinsic acids can be made [2, 3]. However, since the etiology of ETW, which is an oral biological health problem, can be multifactorial, this clinical distinction must be made carefully.
Besides any aesthetic consequences, the severity and longevity of ETW may lead to hypersensitivity, pulpal inflammation, and finally the loss of the affected teeth. The etiological and host-related factors are the most important determinants of the severity and form of ETW. Although host-related factors such as salivary flow rate, buffering capacity, pH, and/or components establish the progression of defects, etiological factors such as personal lifestyle, habits and/or behaviors, medical conditions, environmental exposures, or inborn or inherited characteristics are the main determinants of ETW [2, 4].
Extrinsic acid exposures to dental hard tissues include consumption of acidic foods such as citrus fruits, acidic beverages, wine, and salad dressings; usage of medications such as acetylsalicylic acid and vitamin C supplements; long-term occupational exposures in places such as battery, galvanizing, or ammunition factories; and/or professional swimmers and wine tasters. Patients with eating disorders are also known to consume more acidic foods [5, 6].
ETW caused by intrinsic acids is mostly seen in some gastrointestinal and psychosomatic disorders in which acidic gastric fluid enters the oral cavity, such as bulimia nervosa and gastroesophageal reflux. Besides, chronic alcohol consumption is also a determinant for ETW due to alcohol’s effects on the degradation mechanisms [7, 8].
Studies on the possible etiological factors of ETW in different populations have been conducted in different countries and regions. Today, practitioners are given numerous different diagnostic materials, methods, and indexes for qualitative and quantitative evaluation of ETW [9]. One of the most commonly used indexes is the Basic Erosive Wear Examination (BEWE). The BEWE, which was introduced in 2008, was developed to provide a simpler way to follow and record the severity and progression of ETW in clinical use and to enhance awareness of erosive lesions [10]. Since its publicity, the scientific authorities have approved the BEWE for clinical usage and research, in spite of infrequent data concerning reliability and validity [10].
Considering the importance of oral and dental health and the high prevalence of oral and dental diseases, the efforts of dentists to ensure the oral and dental health of people are very important. In this context, dental students, the future dental professionals, will play an important role in improving public oral and dental health [11, 12]. Oral health professionals’ behaviors related to oral and dental health reflect their understanding of oral and dental preventive measures, which have an important effect on their delivery of oral health care and the health of patients [13, 14]. While dental students have also been found to have a generally positive attitude towards oral health in this regard [15], their own oral health behaviors need to evolve if they are to serve as positive role models for their patients, families, and friends [12, 16, 17]. In addition, the diet-related behaviors of dental students and changes in these behaviors are also reflected to the behaviors and habits of their patients [18]. Therefore, it is very important for dental students to be conscious about oral and dental health, as they will provide health services in the future [14].
The objective of this study was to investigate the ETW status of senior dental students in the School of Dentistry, Marmara University, and to identify the possible etiological factors associated with their ETW status.
Materials and methods
Study group
The present study was approved by the Ethical Committee of Marmara University School of Dentistry with protocol number 2018/186. The study was carried out in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. A total of 130 senior dental students were invited to the study. Before starting, the participants were informed about the study and signed informed consent forms.
The inclusion criteria required for participants are to be a senior student at the School of Dentistry, Marmara University; to sign an informed-consent form; and to complete the questionnaire fully and correctly. Participants with unsigned informed consent forms and incorrectly and/or incompletely filled questionnaires were excluded.
Calibration and training of examiners
The study consisted of a questionnaire and a clinical examination. All data collection was carried out by two researchers: researcher 1 (BG) and researcher 2 (BS). Researcher 1’s role in the study was to assess ETW by performing an intraoral examination of the participants, and researcher 2’s role was to explain the research to the participants, to evaluate their suitability for the research, and to apply the questionnaire. Prior to the study, researcher 1 was trained for clinical evaluation of ETW by the BEWE scores. Subsequently, the assessment was tested on 20 individuals. The outcomes from these participants were not added to the main study database. This pilot study allowed the calibration of researcher 1 for the assessment of ETW using BEWE scores, resulting in an intra-examiner Kappa value of 0.95. To determine the reproducibility of the BEWE criteria, almost 20% of the students were re-examined.
Questionnaire
Of the 130 invited, four did not accept, and finally, 126 senior dental students aged between 21 and 34 participated in the study. Researcher 2 assessed the relevance of the inclusion criteria of the participants and presented the study together with an informed consent form and a questionnaire related to the potential etiological factors behind the ETW status as recommended by Lussi and Carvalho [19]. The questionnaire included closed-ended questions concerning demographic information, education level of the parents, frequency of some types of drinks and foods (including acidic foods, dairy products, fruit juice, acidic drinks, carbonated drinks, sports drinks, tea, milk, and alcohol), usage of straw during consumption of acidic beverages, frequency of smoking and swimming in pools, general health (including presence of gastroesophageal reflux, regurgitation, rumination, anorexia, and bulimia nervosa), usage of some medications (including acetylsalicylic acid, psychotropics, antiemetics, and vitamin C tablets), and oral hygiene habits. The questionnaire was completed prior to the intraoral examination.
Clinical examination
After the questionnaire was completed, a clinical examination of the participants was performed by researcher 1. To minimize bias, it was preferred that researcher 1 not be aware of the questionnaire results.
The clinical examination was conducted with a standard dental probe (PCP11) and a front-surface dental mirror (#5), and gauze to dry the teeth surfaces luminated by an overhead dental reflector in the pediatric dentistry clinic of Marmara University School of Dentistry. The caries status of the participants was evaluated with the DMFT index, which was approved by the World Health Organization (WHO) [20].
The ETW status of the individuals was assessed and recorded using BEWE criteria [21]. All third molars and primary and supernumerary teeth were excluded. The four-level score stages of the severity of ETW are: no ETW (0), initial loss of surface texture (1), hard tissue loss < 50% of the surface area (2), and hard tissue loss ≥ 50% of the surface area (3) [21]. The buccal/labial, palatinal/lingual, and occlusal/incisal surfaces of all teeth were examined to detect the BEWE score of each tooth, sextant, and individual. Each participant was assigned to a group in accordance with the greatest code determined for any tooth, forming two categories: No erosive tooth wear (BEWE = 0): all tooth surfaces had a code “0”, and erosive tooth wear (BEWE ≥ 1): at least one tooth surface had a code “1” or higher.
Statistical analysis
Categorical variables were expressed as frequencies and percentages (%). Continuous variables were expressed as mean ± standard deviation (SD), median (IQR) as appropriate. Intra-examiner agreement was evaluated using Cohen’s kappa. The Shapiro–Wilk test was used to assess the normality assumption for the continuous variables. Categorical variables were evaluated by the chi-square test or Fisher exact test as appropriate. The comparison between two groups (BEWE = 0 and BEWE ≥ 1) for the non-normally distributed continuous variables was assessed by the Mann–Whitney U test. Odds ratios (95% confidence intervals) of the independent parameters were calculated with univariable and multivariable logistic regression models to predict the presence of ETW. Since the more frequently and traditionally used significance levels such as 0.05 may fail to identify variables known to be important for regression analysis [22], multivariable logistic regression analysis was built by performing stepwise variable selection on those variables with a univariate p value < 0.25 [23]. The Hosmer and Lemeshow tests were computed to detect goodness of fit in the multivariable logistic regression models, and a non-significant p value indicated a good fit. All statistical analyses were conducted using SPSS version 19.0 software (IBM Corp., Armonk, NY, USA). p values of less than 0.05 were considered to indicate statistical significance.
Results
One hundred twenty-six senior dental students participated in this study. Participants were aged between 21 and 34, and their mean age was 22.95 ± 1.5. Of a total of 126 students, 80 (63.5%) were female and 46 (36.5%) were male. There was a statistically significant difference in BEWE scores between females and males (chi2 = 5.31, p = 0.021). While 65.1% of all students (58 female and 24 male) were free of ETW (BEWE code 0), 34.9% of all students (22 female and 22 male) had a BEWE code greater than or equal to 1. Clinical examination of teeth using the BEWE scores in this study showed that gender was found to be a significant determinant in ETW. As the income of the participants that were examined was unknown, socioeconomic and sociocultural factors were built on the parents' education levels. Although the mother’s educational status did not have a significant effect on the ETW (p = 0.243), a statistically significant relationship was found between the father’s educational status and BEWE codes (p = 0.001). Table 1 shows the distribution of ETW status of dental students by age, gender, and parental education level.
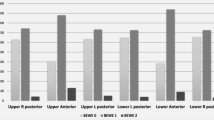
When the distribution of ETW status according to tooth types was examined, it was seen that BEWE 1 and 2 codes mostly affected the right and left permanent first molars in the lower jaw (27% and 25.4%, respectively), and the right permanent first molars and left permanent second molars in the upper jaw (22.2% and 22.2%, respectively). While the teeth least affected by ETW in the upper jaw were the right and left permanent lateral incisors (2.4% and 3.2%, respectively), the least affected teeth in the lower jaw were the right permanent canines, and right permanent lateral and central incisors (8.7%, 8.7%, and 8.7%, respectively). When evaluating the whole mouth, it can be said that the teeth most affected by ETW were the permanent first molars (between 21.4% and 27%), and the least affected teeth were the permanent lateral incisors (between 2.4% and 9.5%).
Table 2 presents the weekly consumption frequency of foods and drinks, and some habits according to the presence of ETW. Statistically significant differences in the presence of ETW were found with respect to the weekly consumption frequency of acidic foods, dairy products, fruit juice, milk, and alcohol (p < 0.001, p = 0.031, p = 0.026, p < 0.001, and p = 0.026, respectively). In addition, no statistically significant effect was found on the ETW status of acidic drinks, carbonated drinks, sports drinks, tea/coffee consumption frequency, and straw use while consuming acidic/carbonated drinks (p = 0.354, p = 0.082, p = 0.326, p = 0.067, and p = 0.415, respectively).
Table 3 shows the distribution of senior dental students with respect to BEWE scores and according to habits that may be associated with ETW. Statistically significant differences were observed between the frequency of weekly swimming in a pool, fluoride-containing toothpaste usage, dental visit frequency, toothbrush type, and hypersensitivity and presence of ETW (p < 0.001, p = 0.017, p < 0.001, p < 0.001, and p = 0.004, respectively). There was no statistical difference in terms of DMFT scores (p = 0.163). In addition, no statistically significant difference was observed between the frequency of toothbrushing, the use of electrical toothbrush, the presence of bruxism, and BEWE scores (p = 0.724, p = 0.303, and p = 0.706, respectively).
No statistically significant associations were found between the status of ETW and medical conditions (gastroesophageal reflux, regurgitation, anorexia/blumia nervosa/rumination) (p > 0.05). When the relationship between usage of medications and ETW was examined, a statistically significant relationship was observed between only usage of vitamin C tablets and ETW (p = 0.014) (Table 4).
We established univariable and multivariable logistic regression analyses to further examine the association of these risk factors with ETW. Univariable analysis revealed that taking acidic foods and alcohol more than 4–5 times per week increases the odds of ETW by 6.043 and 2.532 times, respectively. In addition, taking dairy products, fruit juice, and milk more than 4–5 times per week decreases the likelihood of ETW by 61%, 66%, and 80%, respectively, compared to those who take them less than 1–3 times a week. According to age- and gender-adjusted multivariable model, the odds of ETW was found to be ten fold higher in individuals taking acidic foods more than 4–5 times per week when compared to those who takes less than 1–3 times per week (p < 0.001, 95% CI for OR 3.577–27.849) (Table 5).
Discussion
This study contributes information about the distribution of ETW and its possible related etiological variables among senior dental students. The outcomes of this investigation should be evaluated with attention due to its limitations and possible risk of bias. A suitable sampling method was applied but was unable to reach a wider population due to time and cost limitations and pandemic conditions. The main problem with estimating the relationship between ETW and possible etiological factors is that it is difficult to compare various research and publications. There are large variations in scoring systems, sample sizes, types of recordings, the selection of the reference values and sample groups, standards, and choice of results. The selection of a scoring system, in particular, has an effect on the outcome of a study and might be a determinant of the status of ETW. In addition, differences in nutritional habits observed in different countries and even different cities lead to changes in the etiological factors of ETW [24]. Besides defining ETW as a chemical–mechanical process that results in cumulative loss of enamel and/or dentine not caused by microorganisms, it is difficult to pinpoint the main etiological factor of ETW observed on tooth surfaces [25]. In particular, it is important to make a differential diagnosis of whether the wear on the occlusal surfaces of the teeth occurs chemically and/or mechanically. Although the highest scoring surface of a tooth determines the score of that tooth according to the BEWE [21], the fact that not being able to evaluate separately the degree of affection of the different surfaces in the study appears to be a limitation.
According to the most recent studies, ETW is a widespread condition, and there is evidence that its incidence is continuously increasing, particularly among younger adult age groups. It is also reported that males are more affected by ETW than females [26]. Socioeconomic and sociocultural factors have an effect on nutritional and oral hygiene habits. Low socioeconomic level and income can lead to erosive lesions affecting the dentine or pulp due to the increase of consumption of acidic fruits and carbohydrate products. In addition, it is known that children whose parents have high socioeconomic levels have higher living standards, healthier dietary habits, and more regular oral hygiene habits [27, 28]. Parents’ educational levels might have an impact on their daily decisions concerning their children's nutritional habits. Therefore, a lack of parental education might raise the likelihood of ETW in their children [29]. In parallel with this information, the results of the current study showed that the frequency of ETW in males was statistically significantly higher than in females. In addition, a significant relationship was found between the father’s educational status and the presence of ETW.
The main cause of ETW is thought to be the frequent and regular consumption of acidic foods [30, 31]. But there is no clear relationship between acid exposure and the formation of ETW for all evaluated resources [32,33,34]. Therefore, whether chronic exposure to different acid sources has a real impact on ETW status and severity is still under investigation [24]. It is critical to be able to recognize the wide range of foods, beverages, and medications that have highly erosive potential [35]. Jarvinen et al. [36] reported that consumption of acidic citrus fruits and products derived from them increased the risk of ETW up to 37 times. It is reasonably safe to assume that any nutritional food or drink containing added fruit will be acidic and therefore have an erosive potential [35, 37]. It is known that high consumption of fruits, fruit juices, and soft drinks by children and young adults due to their taste preferences is responsible for the increased risk of ETW [35, 38, 39]. Besides, it is known that the frequency of consumption of acidic foods and beverages is more important than the type of food or beverage in terms of ETW risk [40]. A longitudinal study of 55 adults over 6 years observed that frequent consumption of nutritional acids was related to ETW progression [41]. Similarly, in this study, a statistically significant relationship was found between the frequency of acidic foods and fruit juice consumption and the ETW. The main reason why milk and other dairy products such as yoghurt and cheese with acidic pH values do not cause ETW is the calcium and casein they contain [37, 42]. The results of this study also show that the increase in the frequency of consumption of milk and dairy products has a statistically significant effect on the ETW. The results of the multivariable regression analysis of the study showed that the increase in the frequency of consumption of acidic foods elavates the risk of ETW by approximately 10 times. Frequent daily acid intake is an important factor in the occurrence of severe ETW, affecting 1–3% of the total population [43, 44]. In this direction, Martignon et al. [44] showed that the increase in the frequency of daily acid intake with diet was statistically significantly associated with the presence and severity of ETW in young adults aged 18–25.
Incorrect tooth brushing habits or the use of toothpaste containing high abrasives have long been implicated as etiological factors of tooth wear [45]. Abrasive tooth wear is caused by factors such as toothbrush type, filament stiffness, and tooth brushing method, force, time, and frequency [36]. Several studies conducted have reported synergistic effects of erosive and abrasive tooth wear [46, 47]. Enamel and dentine, which are softened by erosive attacks, can be affected by toothpaste or even tooth brushing without toothpaste [46, 47]. Tooth wear due to toothpaste abrasion reaches dangerous levels in cases of ETW, acting additively or synergistically [48]. When analysing individuals’ oral hygiene habits, in the present study, it was observed that the usage of fluoride-containing toothpaste, type of toothbrush, presence of hypersensitivity, and frequency of dental visits presented a statistically significant relationship with ETW scores.
Lussi et al. [37] reported that some medications containing vitamin C caused ETW by statistically significantly reducing the enamel surface hardness values. Similarly, in the current study, it was observed that the use of vitamin C tablets had a significant effect on the ETW. In spite of this, no significant relation was found between the presence of gastroesophageal reflux, regurgitation, and usage of acetylsalysilic acid, psychotropics, and antiemetics, which are known to be associated with ETW [5, 6, 49, 50]. We are of the opinion that the existence of this relationship can only be determined by evaluations conducted in populations containing more individuals with these diseases and medication usages.
Transforming knowledge about health problems into healthy behaviors remains a challenging task in health promotion. Given that most chronic disorders and conditions are caused by lifestyle and behavioral factors, increasing knowledge is beneficial to the extent that it subsequently leads to behavior change [51]. Studies have shown that dental students with clinical experience have more knowledge about oral health than preclinical dental students who have no experience with patients, and this knowledge is reflected in their attitudes and behaviors [52]. Hong et al. [53] showed that dental students have more accurate knowledge and more positive attitudes about ETW than medical and non-medical students. It was also stated that the attitudes of dental students toward ETW were in significant and positive correlation with their knowledge of this subject. As a limitation, questions about the level of knowledge of dental students on ETW were not included in the questionnaire. Only the effects of nutritional and oral hygiene behaviors, different medical conditions and medication usage, and various sociodemographic factors on the presence and level of ETW were examined. Therefore, there is a need for studies that investigate the relationship between all of these parameters in larger sample groups by evaluating both the level of knowledge of dental students about ETW and how and at what level they reflect this knowledge in their behavior and attitudes, as well as evaluating the ETW status of these students.
Conclusions
The results of this study confirmed that there is a relationship between ETW and some nutritionally consumed foods and beverages, some oral hygiene habits, and usage of vitamin C tablets. Considering that dental students, who are the dentists of the future, reflect their own behaviors and habits to their patients, the consumption of acidic foods increases the risk of ETW approximately 10 times. On the contrary, milk and dairy product consumption can help prevent or control ETW.
References
Ganss C, Lussi A (2014) Diagnosis of erosive tooth wear. Monogr Oral Sci 25:22–31. https://doi.org/10.1159/000359935
Kanzow P, Wegehaupt FJ, Attin T, Wiegand A (2016) Etiology and pathogenesis of dental erosion. Quintessence Int 47:275–278. https://doi.org/10.3290/j.qi.a35625
Ganss C, Lussi A, Schlueter N (2014) The histological features and physical properties of eroded dental hard tissues. Monogr Oral Sci 25:99–107. https://doi.org/10.1159/000359939
Schlueter N, Amaechi BT, Bartlett D, Buzalaf M, Carvalho TS, Ganss C, Hara AT, Huysmans M, Lussi A, Moazzez R, Vieira AR, West NX, Wiegand A, Young A, Lippert F (2020) Terminology of erosive tooth wear: Consensus report of a workshop organized by the ORCA and the cariology research group of the IADR. Caries Res 54:2–6. https://doi.org/10.1159/000503308
Silva NC, Ventura TMO, Oliveira BP, Dos Santos NM, Pelá VT, Buzalaf MAR, Rodrigues JA (2021) Proteomic profile of the acquired enamel pellicle of professional wine tasters with erosive tooth wear. Eur J Oral Sci 129:e12779. https://doi.org/10.1111/eos.12779
Lussi A, Jaeggi T, Zero D (2004) The role of diet in the aetiology of dental erosion. Caries Res 38:34–44. https://doi.org/10.1159/000074360
Teixeira L, Manso MC, Manarte-Monteiro P (2017) Erosive tooth wear status of institutionalized alcoholic patients under rehabilitation therapy in the north of Portugal. Clin Oral Investig 21:809–819. https://doi.org/10.1007/s00784-016-1823-2
Moazzez R, Bartlett D (2014) Intrinsic causes of erosion. Monogr Oral Sci 25:180–196. https://doi.org/10.1159/000360369
Bartlett D, Harding M, Sherriff M, Shirodaria S, Whelton H (2011) A new index to measure tooth wear–methodolgy and practical advice. Community Dent Health 28:182–187. https://doi.org/10.1922/CDH_2552Bartlett06
Wohlrab T, Flechtenmacher S, Krisam J, Saure D, Wolff D, Frese C (2019) Diagnostic value of the basic erosive wear examination for the assessment of dental erosion on patients, dental photographs, and dental casts. Oper Dent 44:E279–E288. https://doi.org/10.2341/18-127-C
Khami MR, Virtanen JI, Jafarian M, Murtomaa H (2007) Prevention-oriented practice of Iranian senior dental students. Eur J Dent Educ 11:48–53. https://doi.org/10.1111/j.1600-0579.2007.00436.x
Peker K, Uysal O, Bermek G (2010) Dental training and changes in oral health attitudes and behaviors in Istanbul dental students. J Dent Educ 74:1017–1023. https://doi.org/10.1002/j.0022-0337.2010.74.9.tb04958.x
Rabiei S, Mohebbi SZ, Patja K, Virtanen JI (2012) Physicians’ knowledge of and adherence to improving oral health. BMC Public Health 12:855. https://doi.org/10.1186/1471-2458-12-855
Yao K, Yao Y, Shen X, Lu C, Guo Q (2019) Assessment of the oral health behavior, knowledge and status among dental and medical undergraduate students: a cross-sectional study. BMC Oral Health 19:26. https://doi.org/10.1186/s12903-019-0716-6
Brusokaite J, Januleviciute I, Kukleris A, Zekonis G (2003) Evaluation of dental health of dental students at Kaunas University of Medicine. Stomatologija Baltic Dent Maxillofac J 5:133–136
Kumar H, Behura SS, Ramachandra S, Nishat R, Dash KC, Mohiddin G (2017) Oral health knowledge, attitude, and practices among dental and medical students in Eastern India - A comparative study. J Int Soc Prev Community Dent 7:58–63. https://doi.org/10.4103/jispcd.JISPCD_30_17
Dagli RJ, Tadakamadla S, Dhanni C, Duraiswamy P, Kulkarni S (2008) Self reported dental health attitude and behavior of dental students in India. J Oral Sci 50:267–272. https://doi.org/10.2334/josnusd.50.267
Taylor GW, Stumpos ML, Kerschbaum W, Inglehart MR (2014) Educating dental students about diet-related behavior change: does experiential learning work? J Dent Educ 78:64–74. https://doi.org/10.1002/j.0022-0337.2014.78.1.tb05658.x
Lussi A, Carvalho TS (2014) Erosive tooth wear: a multifactorial condition of growing concern and increasing knowledge. Monogr Oral Sci 25:1–15. https://doi.org/10.1159/000360380
World Health Organization (2013) Oral health surveys basic methods. WHO Press, Geneva, pp 47–74
Bartlett D, Ganss C, Lussi A (2008) Basic Erosive Wear Examination (BEWE): a new scoring system for scientific and clinical needs. Clin Oral Investig 12:S65-68. https://doi.org/10.1007/s00784-007-0181-5
Bendel RB, Afifi AA (1977) Comparison of stopping rules in forward “stepwise” regression. J Am Stat Assoc 72:46–53. https://doi.org/10.1080/01621459.1977.10479905
Bursac Z, Gauss CH, Williams DK, Hosmer DW (2008) Purposeful selection of variables in logistic regression. Source Code Biol Med 3:17. https://doi.org/10.1186/1751-0473-3-17
Schlueter N, Luka B (2018) Erosive tooth wear - a review on global prevalence and on its prevalence in risk groups. Br Dent J 224:364–370. https://doi.org/10.1038/sj.bdj.2018.167
Carvalho TS, Colon P, Ganss C, Huysmans MC, Lussi A, Schlueter N, Schmalz G, Shellis RP, Tveit AB, Wiegand A (2015) Consensus report of the European Federation of Conservative Dentistry: erosive tooth wear–diagnosis and management. Clin Oral Investig 19:1557–1561. https://doi.org/10.1007/s00784-015-1511-7
Wetselaar P, Vermaire JH, Visscher CM, Lobbezoo F, Schuller AA (2016) The prevalence of tooth wear in the Dutch adult population. Caries Res 50:543–550. https://doi.org/10.1159/000447020
Vieira Pedrosa BR, de Menezes VA (2020) Prevalence of erosive tooth wear and related risk factors in adolescents: An integrative review. J Dent Child (Chic) 87(1):18–25
Racki DNO, Dalla Nora Â, Comim LD, Zenkner JEDA, Alves LS (2020) Erosive tooth wear among South Brazilian adolescents, and its association with sociodemographic variables. Braz Oral Res 33:e119. https://doi.org/10.1590/1807-3107bor-2019.vol33.0119
Maharani DA, Zhang S, Gao SS, Chu CH, Rahardjo A (2019) Dental Caries and the Erosive Tooth Wear Status of 12-Year-Old Children in Jakarta, Indonesia. Int J Environ Res Public Health 16:2994. https://doi.org/10.3390/ijerph16162994
Johansson AK, Lingström P, Birkhed D (2002) Comparison of factors potentially related to the occurrence of dental erosion in high- and low-erosion groups. Eur J Oral Sci 110:204–211. https://doi.org/10.1034/j.1600-0447.2002.11211.x
Barbour ME, Lussi A (2014) Erosion in relation to nutrition and the environment. Monogr Oral Sci 25:143–154. https://doi.org/10.1159/000359941
Shellis RP, Featherstone JD, Lussi A (2014) Understanding the chemistry of dental erosion. Monogr Oral Sci 25:163–179. https://doi.org/10.1159/000359943
Schlueter N, Tveit AB (2014) Prevalence of erosive tooth wear in risk groups. Monogr Oral Sci 25:74–98. https://doi.org/10.1159/000359938
Hong DW, Lin XJ, Wiegand A, Yu H (2020) Does delayed toothbrushing after the consumption of erosive foodstuffs or beverages decrease erosive tooth wear? A systematic review and meta-analysis. Clin Oral Investig 24:4169–4183. https://doi.org/10.1007/s00784-020-03614-9
O’Toole S, Mullan F (2018) The role of the diet in tooth wear. Br Dent J 224:379–383. https://doi.org/10.1038/sj.bdj.2018.127
Järvinen VK, Rytömaa II, Heinonen OP (1991) Risk factors in dental erosion. J Dent Res 70:942–947. https://doi.org/10.1177/00220345910700060601
Lussi A, Megert B, Shellis RP, Wang X (2012) Analysis of the erosive effect of different dietary substances and medications. Br J Nutr 107:252–262. https://doi.org/10.1017/S0007114511002820
Brug J, Tak NI, te Velde SJ, Bere E, de Bourdeaudhuij I (2008) Taste preferences, liking and other factors related to fruit and vegetable intakes among schoolchildren: results from observational studies. Br J Nutr 99:S7–S14. https://doi.org/10.1017/S0007114508892458
Grimm GC, Harnack L, Story M (2004) Factors associated with soft drink consumption in school-aged children. J Am Diet Assoc 104:1244–1249. https://doi.org/10.1016/j.jada.2004.05.206
O’Toole S, Bernabé E, Moazzez R, Bartlett D (2017) Timing of dietary acid intake and erosive tooth wear: A case-control study. J Dent 56:99–104. https://doi.org/10.1016/j.jdent.2016.11.005
Lussi A, Schaffner M (2000) Progression of and risk factors for dental erosion and wedge-shaped defects over a 6-year period. Caries Res 34:182–187. https://doi.org/10.1159/000016587
Buzalaf MAR, Magalhães AC, Rios D (2018) Prevention of erosive tooth wear: targeting nutritional and patient-related risks factors. Br Dent J 224:371–378. https://doi.org/10.1038/sj.bdj.2018.173
Bartlett DW, Lussi A, West NX, Bouchard P, Sanz M, Bourgeois D (2013) Prevalence of tooth wear on buccal and lingual surfaces and possible risk factors in young European adults. J Dent 41:1007–1013. https://doi.org/10.1016/j.jdent.2013.08.018
Martignon S, López-Macías AM, Bartlett D, Pitts N, Usuga-Vacca M, Gamboa LF, O’Toole S (2019) The use of index teeth vs. full mouth in erosive tooth wear to assess risk factors in the diet: A cross-sectional epidemiological study. J Dent 88:103164. https://doi.org/10.1016/j.jdent.2019.07.002
Hara AT, Barlow AP, Eckert GJ, Zero DT (2014) Novel in-situ longitudinal model for the study of dentifrices on dental erosion-abrasion. Eur J Oral Sci 122:161–167. https://doi.org/10.1111/eos.12108
Ganss C, Hardt M, Blazek D, Klimek J, Schlueter N (2009) Effects of toothbrushing force on the mineral content and demineralized organic matrix of eroded dentine. Eur J Oral Sci 117:255–260. https://doi.org/10.1111/j.1600-0722.2009.00617.x
Körner P, Inauen DS, Attin T, Wegehaupt FJ (2020) Erosive/Abrasive Enamel Wear While Using a Combination of Anti-Erosive Toothbrush/-Paste. Oral Health Prev Dent 18:53–60. https://doi.org/10.3290/j.ohpd.a43352
Wiegand A, Schwerzmann M, Sener B, Magalhaes AC, Roos M, Ziebolz D, Imfeld T, Attin T (2008) Impact of toothpaste slurry abrasivity and toothbrush filament stiffness on abrasion of eroded enamel - an in vitro study. Acta Odontol Scand 66:231–235. https://doi.org/10.1080/00016350802195041
Wilder-Smith CH, Materna A, Martig L, Lussi A (2017) Longitudinal study of gastroesophageal reflux and erosive tooth wear. BMC Gastroenterol 17:113. https://doi.org/10.1186/s12876-017-0670-1
Zalewska I, Trzcionka A, Tanasiewicz M (2021) A Comparison of Etiology-Derived and Non-Etiology-Derived Indices Utilizing for Erosive Tooth Wear in People with Eating Disorders. The Validation of Economic Value in Clinical Settings. Coatings 11:471
Rimal RN (2000) Closing the knowledge-behavior gap in health promotion: the mediating role of self-efficacy. Health Commun 12:219–237. https://doi.org/10.1207/S15327027HC1203_01
Al-Wesabi AA, Abdelgawad F, Sasahara H, El Motayam K (2019) Oral health knowledge, attitude and behaviour of dental students in a private university. BDJ Open 5:16. https://doi.org/10.1038/s41405-019-0024-x
Hong DW, Lin XJ, Wiegand A, Yu H (2020) Knowledge of and attitudes towards erosive tooth wear among students of two Chinese universities. BMC Oral Health 20:110. https://doi.org/10.1186/s12903-020-01105-7
Author information
Authors and Affiliations
Contributions
Conceptualization: Berkant Sezer, Begüm Giritlioğlu, and Betül Kargül. Methodology: Berkant Sezer and Betül Kargül. Formal analysis and investigation: Berkant Sezer, Begüm Giritlioğlu, Duygu Sıddıkoğlu, and Betül Kargül. Writing—original draft preparation: Berkant Sezer. Writing—review and editing: Berkant Sezer, Duygu Sıddıkoğlu, Adrian Lussi, and Betül Kargül. Supervision: Adrian Lussi and Betül Kargül.
Corresponding author
Ethics declarations
Ethics approval
The present study was approved by the Ethical Committee of Marmara University School of Dentistry with protocol number 2018/186. The study was carried out in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Before starting the study, the participants were informed about the study and signed informed consent forms.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sezer, B., Giritlioğlu, B., Sıddıkoğlu, D. et al. Relationship between erosive tooth wear and possible etiological factors among dental students. Clin Oral Invest 26, 4229–4238 (2022). https://doi.org/10.1007/s00784-022-04425-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04425-w