Abstract
Objective
To assess the impact of various local pathologies on facial alveolar bone dimensions at tooth sites.
Materials and methods
Cone-beam computed tomography images of 60 patients were analyzed. Healthy teeth and teeth with local pathologies (i.e., endodontically treated, periodontally diseased teeth, and teeth with periapical lesions) were included. The thickness of the facial alveolar bone was measured at five locations: (1) the bone crest (W0), (2) 25% (W25), (3) 50% (W50), (4) 75% (W75) of the distance from the bone crest to the root apex (A), and (5) in the A region (W100). The results were considered statistically significant at p < 0.0008 (adjustment according to the statistical correction for multiple testing).
Results
A total of 1174 teeth (707 healthy and 467 with the local pathologies) were assessed. Periodontally diseased maxillary premolars and anterior teeth in the mandible in the W0 position, as well as maxillary molars in the W25 position, tended to have a lower facial bone thickness when compared to the healthy teeth (0.68 mm vs. 0.84 mm, p = 0.008; 0.47 mm vs. 0.55 mm, p = 0.004; and 1.27 mm vs. 1.72 mm; p = 0.009, respectively). In contrast, the observed tendency pointed towards thicker facial bone wall for the periodontally diseased mandibular anterior teeth in the W50 position (0.74 vs. 0.52, p = 0.001). Healthy maxillary molars tended to display a thicker facial alveolar bone compared to the teeth with local pathologies in the W25, W50, and W75 positions (p = 0.001, p = 0.005, and p = 0.004, respectively).
Conclusions
The present analysis has indicated that local pathologies are commonly associated with a compromised socket morphology.
Clinical relevance
The facial bone thickness was particularly reduced at periodontally diseased teeth, which may challenge implant therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Following tooth extraction, the alveolar ridge undergoes marked dimensional alterations leading to the reduction of the overall ridge volume and change in the ridge shape [1]. A number of studies have demonstrated that substantial dimensional changes of the alveolar ridge occur during the first 6 months following tooth extraction, resulting in horizontal and vertical bone loss of 29–63% (corresponding to 2.5–4.5 mm) and 11–22% (corresponding to 0.8–1.5 mm), respectively [2]. These changes were shown to be more pronounced at the buccal aspect compared to its lingual/palatal counterpart [3, 4].
The magnitude of alveolar bone resorption was found to be influenced by such factors as location of the tooth (anterior vs. posterior region), [5] the presence of severe bone loss at the time of the extraction, [6,7,8] and the thickness of the alveolar buccal bone plate [9, 10]. In particular, reduction of the ridge volume in anterior sites was found to be more pronounced than in posterior regions (− 34% vs. − 18%, respectively) [5]. Tooth extraction at compromised sites (e.g., the presence of severe bone loss at the time of extraction) was associated with slower healing and cortication [6, 8] and a greater reduction in volume compared to the changes at non-compromised sites [7]. Moreover, significantly higher vertical bone loss was found at the sites with a buccal bone thickness of < 1 mm compared to a wall thickness of 1 mm or more (7.5 mm (62%) vs. 1.1 mm (9%), respectively) [9].
Based on three-dimensional radiographic assessments, the thickness of the facial alveolar bone in the anterior teeth was reported to range between 0.5 and 1.5 mm, with the majority of cases being associated with a wall thickness of ≤ 1 mm (57% in the mandible [11] and 63% [12] to 69% [9] in the maxilla) [9, 11,12,13,14,15,16,17,18,19]. These findings corroborate those obtained by direct clinical measurements, where 87% of teeth in the anterior maxilla had a facial alveolar bone wall thickness of ≤ 1 mm, with a mean value amounting to 0.8 mm [20].
When considering the posterior regions, for both the maxilla and the mandible, a steady increase of the facial bone thickness from the premolar to molar regions was observed [11, 21,22,23]. In the bone crest area, a facial bone thickness of < 1 mm was found in more than half of the cases of the premolars (mandible: 64% to 81%, maxilla: 58% to 66%) and in 12% to 88% of the cases of the maxillary and mandible molars [11, 21, 23].
However, it should be pointed out that these currently available investigations reported on the thickness of the facial alveolar bone at healthy tooth sites. Information regarding the facial bone thickness at compromised tooth sites is scarce.
Therefore, the present analysis aimed to investigate the effects of the presence of local pathologies (e.g., endodontic periapical lesions, endodontic treatment, and periodontal disease) on the facial alveolar bone dimensions in various regions of the maxilla and the mandible.
Material and methods
This retrospective analysis included 60 patients (29 male and 31 female) who attended the Department of Oral Surgery, Heinrich Heine University, Düsseldorf, Germany, between January 2009 and August 2013 and were referred for cone-beam computerized tomography (CBCT) examinations for various indications. The mean age of the enrolled patients was 48.8 years (range: 15–84 years).
A data extraction template was generated and used for the pseudonymous acquisition of demographic study variables and tooth site characteristics recorded during CBCT scan analysis. The study protocol was in accordance with the Helsinki Declaration as revised in 2013 and was approved by the local ethics committee (Study Nr. 4301).
CBCT evaluation
Sixty CBCT scans (field of view size: 8 × 8 and 12 × 8.5; voxel size: 0.12–0.3 mm, focal spot: 0.5 mm; image acquisition: single 360° rotation; scan time: 15 s; generator voltage: 50–90 kVp) obtained using a Pax Duo 3D scanner (VATECH Co., Ltd., Hwaseong, South Korea) were investigated. A total of 1174 teeth (707 healthy and 467 with the local pathologies) were analyzed using the software provided by the same company (Ez3D2009, VATECH Co., Ltd., Hwaseong, South Korea). With regard to the multi-rooted teeth, the measurements were performed for both the mesial and distal roots of the mandibular molars and the mesial and distal buccal roots of the maxillary molars. Accordingly, a total of 1395 roots (847 healthy and 548 with the local pathologies) were included into the analysis (Table 1). According to the presence or absence of local tooth pathologies, the following groups were distinguished (Table 1):
Healthy teeth (H);
Endodontically treated teeth (Endo);
Periodontally diseased teeth (i.e., teeth presenting with the radiographic distance between the cementoenamel junction (CEJ) to the crestal bone ≥ 3.5 mm) (Perio);
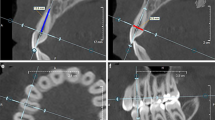
Teeth with periapical lesions (i.e., periapical translucence ≥ 1.0 mm) (PL) (Fig. 1a);
Periodontally diseased and endodontically treated teeth (Perio + Endo);
Endodontically treated teeth with periapical lesions (Endo + PL);
Periodontally diseased teeth with periapical lesions (Perio + PL) (Fig. 1b);
Teeth with one of the local pathologies and their combinations (i.e., Endo; Perio, PL; Perio + Endo, Endo + PL, Perio + PL) (pathology).
According to location, the teeth were divided into the anterior (incisors and canines), premolar, and molar segments of the maxilla and mandible.
To perform the measurements, sagittal slices of the examined roots were positioned through the long axis (LA) of each tooth and displayed with a 250% zooming factor. The sagittal cross-sectional slices were used to measure the alveolar bone thickness.
The following landmark lines were identified (Fig. 1):
A line crossing the long axis of the tooth (LA);
A line marking the radiographic cemento-enamel junction (CEJ).
The following dimensions were measured (Fig. 2):
the distance from the radiographic CEJ to the facial alveolar bone crest (BC) (CEJ-BC);
the distance from the BC to the apex of the tooth (BC-A);
facial bone thickness was measured in five different positions perpendicular to the LA:
- (i)
at the BC (W0);
- (ii)
at the position of 25% of the BC-A distance (W25);
- (iii)
at the position of 50% of the BC-A distance (W50);
- (iv)
at the position of 75% of the BC-A distance (W75);
- (v)
at the apex of the root (W100).
Dimensions measured in the study: CEJ–BC-the distance from the radiographic cemento-enamel junction (CEJ) to the facial alveolar bone crest (BC); BC–A-the distance from the BC to the apex of the tooth (a). Locations of facial bone thickness measurements: W0: at the BC; W25: at the position of 25% BC–A distance; W50: at the position of 50% of the BC–A distance; W75: at the position of 75% of the BC–A distance; W100: at the A (W100)
Measurements were not recorded in areas with the presence of artifacts, crowding, or improper alignment of the teeth.
All measurements were performed by one calibrated examiner. Intra-examiner reproducibility was assessed using randomly selected CBCT scans of three teeth. The calculated mean variability between the repeated measurements at baseline and 48 h was 1.5% (1.8%, 2.3%, and 0.5%, respectively).
Statistical analysis
Statistical analysis was performed using SPSS Statistics 23 software. The mean values, standard deviations (SDs), and medians were calculated for the facial bone thickness in different positions (W0, W25, W50, W75, and W100). The measurements at the respective positions at a tooth root were considered as statistical unit. No statistical correction was performed to address the random factor patient.
Prior to analysis, all teeth were sorted by location into the anterior, premolar, and molar segments of the maxilla and the mandible. Groups with at least 15 teeth roots were included in the statistical analysis. This resulted in statistical comparison among the healthy anterior teeth and premolars vs. the Perio and Pathology groups, and the healthy posterior teeth vs. the Perio, Endo, and Pathology groups in the maxilla. In the mandible, healthy anterior, premolar, and posterior teeth were compared to the teeth in the Perio and Pathology groups.
A Mann-Whitney U test was used to assess the influence of the presence or absence of pathological tooth conditions as well as the type of pathology on the thickness of the facial bone. According to the statistical correction for multiple testing (Bonferroni method), results were considered statistically significant at p < 0.0008.
Results
Maxilla
The mean values, standard deviations, and medians of the evaluated parameters in the maxilla are presented in Table 2.
Anterior region
The mean values of the CEJ–BC distance in the H group and the Pathology group amounted to 2.51 mm and 4.39 mm, respectively. The longest distances were registered for the Perio (4.53 mm) and the Perio + Endo (5.26 mm) groups. Accordingly, the lowest mean values for the BC–A distances were detected in the latter groups in comparison to the H group (Perio: 10.16 mm, Perio + Endo: 8.32 mm, and H: 11.24 mm).
The general tendency demonstrated the lowest values in facial bone thickness in the W0 area, which increased in the W25 region. Facial bone thickness in the W50 and W75 positions showed similar values but these were slightly lower compared to the W25 position. The highest values for facial bone thickness were registered at the W100 position. The comparison of alveolar facial bone thickness in H group and for teeth with one of the investigated local pathologies revealed no significant differences.
Premolar region
The mean of the CEJ–BC distance was 2.21 mm in the H group and 4.08 mm in the Pathology group. In the latter group, the longest distance (5.10 mm) was found in the Perio + PL group, followed by 4.80 mm in the Perio group. The BC–A distance in the Pathology group displayed lower values in comparison to the H group (8.76 mm and 10.29 mm, respectively). Particularly, the Perio + PL group exhibited the lowest values of the BC–A distance (6.65 mm).
With regard to the facial bone thickness for healthy teeth and teeth with local pathologies, the lowest values were registered in the W0 region. These increased in the W25 position and showed a tendency to reduce in the W50 and W75 areas. The highest thickness of the facial bone wall was registered in the W100 area. When comparing healthy teeth to the teeth with local pathologies, the observed tendency pointed toward a lower bone thickness at the W0 level in the Perio group (Perio: 0.68 mm and H: 0.84 mm, p = 0.008).
Molar region
The CEJ–BC distance of the posterior teeth in the H group was 2.18 mm and was lower when compared to the distance noted for the molars in the Pathology group (4.74 mm). The highest CEJ–BC distances were found in the Perio and Perio + Endo groups (8.05 mm and 5.45 mm, respectively). Accordingly, the BC–A distance in the H group was found to be greater compared to that found in the Pathology group (9.64 mm and 7.36 mm, respectively).
For the healthy maxillary molars, a gradual increase of the bone thickness was observed from the W0 to W75 positions. In the apex region, the facial bone wall displayed lower thickness compared to the W25–W75 regions. For the teeth with one of the local pathologies, the general trend showed comparable bone thickness in the W25, W50, and W75 positions, with the lowest values in the W0 area and the highest in the W100 region.
The observed tendency revealed a higher facial bone thickness at the molar sites in the H group for the W25, W50, and W75 positions in comparison to the Pathology group (p = 0.001, p = 0.005, and p = 0.004, respectively). Additionally, in the H group, the facial bone tended to be thicker compared to the Perio group in the W25 region (1.72 mm vs. 1.27 mm, p = 0.009).
Mandible
Table 3 presents the findings of the evaluated parameters in the mandible.
Anterior region
The CEJ–BC distance in the healthy dentition was 2.47 mm. The corresponding value for the teeth in the Pathology group was 4.49 mm, with the highest value detected for periodontally diseased teeth (Perio: 4.62 mm, Perio + PL: 5.10 mm, and Perio + Endo: 4.20 mm). Similarly, the BC–A distance was lower for the Pathology group compared to the healthy teeth (9.82 mm and 11.96 mm, respectively).
Generally, the mean facial bone thickness in the H group was slightly reduced in the W25 and W50 regions compared to the W0 region, whereas it gradually increased in the W75 and W100 areas. For teeth exhibiting one of the local pathologies, the overall tendency showed a gradual increase in mean facial bone thickness from the W0 to W100 position.
The bone thickness in the Perio group tended to be thinner than in the healthy sites. In particular, mean bone thickness in the W0 area was higher around the healthy teeth (H: 0.55 mm and Perio: 0.47 mm, p = 0.004), while in the W50 position, higher values were registered for the Perio group (Perio: 0.74 mm and H: 0.52 mm, p = 0.001).
Premolar region
The mean CEJ–BC distance was 2.43 mm in the H group and 4.10 mm for the Pathology group. In the latter group, the Perio group exhibited the highest CEJ–BC distance (4.45 mm). The BC–A distance was higher for healthy teeth compared to the teeth with one of the local pathologies (12.18 mm and 10.82 mm, respectively).
Regarding facial bone thickness in healthy teeth and teeth with one of the local pathologies, a steady increase from W0 to W100 was detected. Between the two groups, no significant differences in bone thickness were found at any position. Likewise, the investigated pathologies were not found to have an influence on facial bone thickness dimensions.
Posterior region
The CEJ–BC distance in healthy teeth was 2.23 mm, and it was 4.46 mm for teeth in the Pathology group. The highest values of the CEJ-CB distance were registered in the Perio + Endo and Perio + PL groups (5.70 mm and 8.15 mm, respectively). The mean values for the BC–A distance were lower for the Pathology group when compared to the healthy dentition (9.57 mm and 11.35 mm, respectively) and were particularly reduced in the Perio + PL and Perio + Endo groups (4.17 mm and 8.02 mm, respectively).
Based on the general tendency, facial bone thickness in healthy teeth and in teeth with local pathologies gradually increased from the W0 to the W100 area. The comparison between healthy teeth and teeth with local pathologies, as well as the types of local pathology with respect to bone thickness in various regions, revealed no significant differences.
Discussion
This retrospective investigation was aimed at assessing the impact of various local pathologies (i.e., endodontic treatment, periodontal disease, and the presence of periapical lesions) on facial alveolar bone dimensions at tooth sites.
Based on the CBCT findings, it was noted that periodontal disease had a tendency to influence the facial bone thickness for the maxillary premolars, molars, and anterior mandibular teeth. In the W0 area, specifically, the periodontally diseased maxillary premolars, anterior teeth in the mandible, and maxillary molars in the W25 position exhibited a lower facial bone thickness compared to the healthy teeth (0.68 vs. 0.84, p = 0.008; 0.47 mm vs. 0.55 mm, p = 0.004; and 1.27 mm vs. 1.72 mm, p = 0.009, respectively). Moreover, it should be noted that the periodontally diseased teeth showed the highest CEJ-BC values and, accordingly, the lowest BC-A values. Together, these findings illustrate that periodontitis is associated not only with the surrounding alveolar bone loss [24] but, based on our findings, also leads to the reduced facial bone thickness in the aforementioned regions.
In contrast, periodontally diseased anterior mandibular teeth in the W50 area tended to have thicker facial alveolar bone walls (0.74 mm vs. 0.52 mm, p = 0.001, respectively) when compared to the H group. This finding can be explained by the different general tendencies noted for the healthy teeth and teeth with pathologies, leading to the assumption that the W50 position in the Perio group may correspond to the W75 position of the healthy teeth. Besides, healthy maxillary molars tended to display thicker facial alveolar bone walls than the teeth with local pathologies in the W25, W50, and W75 positions. This finding implies that apical periodontitis, which is characterized by bone destruction in the periapical tissues [25], as well as marginal periodontitis, leads to a reduced thickness of the facial bone in the maxillary molars. To the authors’ knowledge, this is the first study to evaluate facial bone thickness in the presence of local tooth pathologies. Therefore, our results could not be compared to those previously reported.
According to our findings, the CEJ–BC distance in the healthy tooth sites varied from 2.18 to 2.51 mm, which is in line with the previous investigations, in which the CEJ–BC distance for anterior and premolar teeth ranged from 1.6 to 3.48 mm [13,14,15,16, 19, 26,27,28] and the distance for maxillary and mandibular molars ranged from 1.69 to 2.45 mm [23]. For teeth with one of the local pathologies, the corresponding distance varied from 4.08 to 4.72 mm, with the highest values noted for periodontally diseased teeth. However, we identified no studies that investigated the CEJ–BC distance for teeth with local pathologies.
In agreement with the previous studies, our results showed a steady facial bone increase from the anterior toward the posterior regions in the maxilla and the mandible [12, 21, 23]. Considering the healthy anterior teeth, the mean thickness of the facial bone wall at the BC was 0.45 mm and 0.55 mm for the maxilla and the mandible, respectively. These values are within the range reported by previous authors, who observed that facial bone thickness in the anterior teeth varied from 0.4 to 1.59 mm (measured at a distance of 1–3 mm apically to the BC) [11, 13,14,15, 29]. Additionally, the decrease in mean bone thickness from the W25 toward the W75 position in the anterior maxillary teeth [15, 17, 26, 27] and the gradual bone thickness increase from the bone crest to the apex in the anterior mandible is consistent with previous observations [11].
In the current investigation, the mean alveolar bone thickness in healthy premolars at the BC varied from 0.51 to 0.84 mm for the mandible and the maxilla. These measurements corroborate the previously reported data, in which the facial alveolar bone thickness ranged between 0.3 and 1.72 mm for maxillary premolars and between 0.5 and 1.21 mm for mandibular premolars [15, 21, 23, 28, 29]. With regard to the molar regions, the present study found similar mean facial bone thickness at the BC for the maxillary and mandibular molars (mean 0.97 mm), which is in line with the previously reported facial bone thickness for the maxilla and the mandible at the BC or 1 mm below (range: 0.90–2.45 mm in the maxilla [11, 21, 23]; range: 0.92–1.08 mm in the mandible [11, 23]).
The results of our study revealed a gradual facial bone thickness increase from the coronal to apical part in the mandibular premolars and molars, which is consistent with the findings of the previous CBCT investigation [21]. The overall tendency noted for the maxilla showed reduced facial bone dimensions in the W50 and W75 positions (compared to W25) for the premolars and reduced bone dimensions at the root apex for the molars (compared to the W25–75 positions). This observation is comparable to the previously reported data, in which posterior maxillary teeth (premolars and molars) showed a gradual decrease in facial bone thickness measured 1, 3, and 5 mm below the BC [21]. In addition, based on the aforementioned study, first maxillary premolars and the mesial roots of first molars exhibited significantly thinner facial bone walls than all other roots in the posterior maxilla [21].
The present statistical analysis did not perform statistical correction to address potential correlation of measurements coming from the same subject. Estimation of an individual correction factor was of questionable reliability because pathologies were not equally distributed among the patients and because facial bone thickness also varies considerably with respect to the respective measurement location. Also, non-parametric mixed effects designs would have required rather homogenous groups and were therefore not eligible for the present investigation. However, the present study design was chosen to address the overall differences in facial bone thickness at different positions among patients exhibiting different pathologies, and these differences could be expressed through the non-parametric comparison.
CBCT is a widely used imaging technique that facilitates diagnosis and improves treatment planning in implant dentistry [30]. The accuracy of linear measurements is crucial for the basic preoperative implant planning [31]. Voxel size plays a significant role in image quality as it defines the spatial resolution of the CBCT images, with a lower voxel size providing a higher degree of detail [32]. In the current investigation, we used a voxel size ranging between 0.12 and 0.3 mm. As shown in previous studies, linear measurements using CBCT images with different voxel sizes (0.2 mm vs. 0.3 mm vs. 0.4 mm) provided comparable accuracy, and smaller voxels did not result in greater precision of vertical or horizontal bone measurements [33, 34]. In line with these findings, a comparison of four voxel sizes (0.15 mm, 0.2 mm, 0.25 mm, and 0.3 mm) for detection of external root resorption revealed similar diagnostic efficacy [34]. Besides, a recent systematic analysis concluded that a voxel size of 0.3 to 0.4 mm is adequate to provide CBCT images of acceptable diagnostic quality for planning implant treatment [35].
Facial bone thickness prior to extraction was shown to be a major factor affecting the degree of vertical dimension change of the alveolar crest following tooth extraction and immediate implant placement [10, 36]. Particularly, facial bone thickness of < 1 mm was associated with significantly higher vertical bone resorption compared to sites exhibiting bone wall thickness > 1 mm [10, 29]. Furthermore, as our findings demonstrated, the presence of local tooth pathologies has an impact on the facial bone thickness. Consequently, considering the fact that the majority of the teeth are being extracted due to the presence of local pathology, the assessment of the facial bone wall dimensions prior to tooth extraction may help to anticipate the degree of bone loss and evaluate the necessity of a bone augmentation procedure prior or simultaneous to implant placement.
Conclusions
The present analysis has indicated that local pathologies are commonly associated with a compromised socket morphology. The facial bone thickness was particularly reduced at Perio sites, which may challenge implant therapy.
References
Hammerle CH, Araujo MG, Simion M (2012) Evidence-based knowledge on the biology and treatment of extraction sockets. Clin Oral Implants Res 23(Suppl 5):80–82
Tan WL, Wong TL, Wong MC, Lang NP (2012) A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin Oral Implants Res 23 Suppl 5:1–21
Araujo MG, Silva CO, Misawa M, Sukekava F (2015) Alveolar socket healing: what can we learn? Periodontol 68(1):122–134
Botticelli D, Berglundh T, Lindhe J (2004) Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol 31(10):820–828
Araujo MG, da Silva JCC, de Mendonça AF, Lindhe J (2015) Ridge alterations following grafting of fresh extraction sockets in man. A randomized clinical trial. Clin Oral Implants Res 26(4):407–412
Ahn JJ, Shin HI (2008) Bone tissue formation in extraction sockets from sites with advanced periodontal disease: a histomorphometric study in humans. Int J Oral Maxillofac Implants 23(6):1133–1138
Aimetti M, Manavella V, Corano L, Ercoli E, Bignardi C, Romano F (2018) Three-dimensional analysis of bone remodeling following ridge augmentation of compromised extraction sockets in periodontitis patients: a randomized controlled study. Clin Oral Implants Res 29(2):202–214
Bertl K, Kukla EB, Albugami R, Beck F, Gahleitner A, Stavropoulos A (2018) Timeframe of socket cortication after tooth extraction: a retrospective radiographic study. Clin Oral Implants Res 29(1):130–138
Chappuis V, Engel O, Reyes M, Shahim K, Nolte LP, Buser D (2013) Ridge alterations post-extraction in the esthetic zone: a 3D analysis with CBCT. J Dent Res 92(12 Suppl):195s–201s
Tomasi C, Sanz M, Cecchinato D, Pjetursson B, Ferrus J, Lang NP, Lindhe J (2010) Bone dimensional variations at implants placed in fresh extraction sockets: a multilevel multivariate analysis. Clin Oral Implants Res 21(1):30–36
López-Jarana P, Díaz-Castro CM, Falcão A, Falcão C, Ríos-Santos JV, Herrero-Climent M (2018) Thickness of the buccal bone wall and root angulation in the maxilla and mandible: an approach to cone beam computed tomography. BMC Oral Health 18(1):194
Braut V, Bornstein MM, Belser U, Buser D (2011) Thickness of the anterior maxillary facial bone wall-a retrospective radiographic study using cone beam computed tomography. Int J Periodontics Restorative Dent 31(2):125–131
Januário AL, Duarte WR, Barriviera M, Mesti JC, Araújo MG, Lindhe J (2011) Dimension of the facial bone wall in the anterior maxilla: a cone-beam computed tomography study. Clin Oral Implants Res 22(10):1168–1171
Ghassemian M, Nowzari H, Lajolo C, Verdugo F, Pirronti T, D'Addona A (2012) The thickness of facial alveolar bone overlying healthy maxillary anterior teeth. J Periodontol 83(2):187–197
Vera C, De Kok IJ, Reinhold D, Limpiphipatanakorn P, Yap AK, Tyndall D, Cooper LF (2012) Evaluation of buccal alveolar bone dimension of maxillary anterior and premolar teeth: a cone beam computed tomography investigation. Int J Oral Maxillofac Implants 27(6):1514–1519
Nowzari H, Molayem S, Chiu CH, Rich SK (2012) Cone beam computed tomographic measurement of maxillary central incisors to determine prevalence of facial alveolar bone width >/=2 mm. Clin Implant Dent Relat Res 14(4):595–602
Kim YJ, Park JM, Kim S, Koo KT, Seol YJ, Lee YM, Rhyu IC, Ku Y (2016) New method of assessing the relationship between buccal bone thickness and gingival thickness. J Periodontal Implant Sci 46(6):372–381
Fuentes R, Flores T, Navarro P, Salamanca C, Beltrán V, Borie E (2015) Assessment of buccal bone thickness of aesthetic maxillary region: a cone-beam computed tomography study. J Periodontal Implant Sci 45(5):162–168
El Nahass H, N Naiem S (2015) Analysis of the dimensions of the labial bone wall in the anterior maxilla: a cone-beam computed tomography study. Clin Oral Implants Res 26(4):e57–e61
Huynh-Ba G, Pjetursson BE, Sanz M, Cecchinato D, Ferrus J, Lindhe J, Lang NP (2010) Analysis of the socket bone wall dimensions in the upper maxilla in relation to immediate implant placement. Clin Oral Implants Res 21(1):37–42
Temple KE, Schoolfield J, Noujeim ME, Huynh-Ba G, Lasho DJ, Mealey BL (2016) A cone beam computed tomography (CBCT) study of buccal plate thickness of the maxillary and mandibular posterior dentition. Clin Oral Implants Res 27(9):1072–1078
Braut V, Bornstein MM, Lauber R, Buser D (2012) Bone dimensions in the posterior mandible: a retrospective radiographic study using cone beam computed tomography. Part 1--analysis of dentate sites. Int J Periodontics Restorative Dent 32(2):175–184
Zekry A, Wang R, Chau AC, Lang NP (2014) Facial alveolar bone wall width - a cone-beam computed tomography study in Asians. Clin Oral Implants Res 25(2):194–206
Papapanou PN, Sanz M, Buduneli N, Dietrich T, Feres M, Fine DH, Flemmig TF, Garcia R, Giannobile WV, Graziani F, Greenwell H, Herrera D, Kao RT, Kebschull M, Kinane DF, Kirkwood KL, Kocher T, Kornman KS, Kumar PS, Loos BG, Machtei E, Meng H, Mombelli A, Needleman I, Offenbacher S, Seymour GJ, Teles R, Tonetti MS (2018) Periodontitis: consensus report of workgroup 2 of the 2017 world workshop on the classification of periodontal and Peri-implant diseases and conditions. J Periodontol 89(Suppl 1):S173–s182
Braz-Silva PH, Bergamini ML, Mardegan AP, De Rosa CS, Hasseus B, Jonasson P (2019) Inflammatory profile of chronic apical periodontitis: a literature review. Acta Odontol Scand 77(3):173–180
Khoury J, Ghosn N, Mokbel N, Naaman N (2016) Buccal bone thickness overlying maxillary anterior teeth: a clinical and radiographic prospective human study. Implant Dent 25(4):525–531
Farahamnd A, Sarlati F, Eslami S, Ghassemian M, Youssefi N, Jafarzadeh Esfahani B (2017) Evaluation of impacting factors on facial bone thickness in the anterior maxillary region. J Craniofac Surg 28(3):700–705
Rojo-Sanchis J, Viña-Almunia J, Peñarrocha-Oltra D, Peñarrocha-Diago M (2017) Facial alveolar bone width at the first and second maxillary premolars in healthy patients: a cone beam computed tomography study. J Oral Implantol 43(4):261–265
Misawa M, Lindhe J, Araujo MG (2016) The alveolar process following single-tooth extraction: a study of maxillary incisor and premolar sites in man. Clin Oral Implants Res 27(7):884–889
Bornstein MM, Horner K, Jacobs R (2017) Use of cone beam computed tomography in implant dentistry: current concepts, indications and limitations for clinical practice and research. Periodontol 73(1):51–72
Nickenig HJ, Eitner S (2007) Reliability of implant placement after virtual planning of implant positions using cone beam CT data and surgical (guide) templates. J Craniomaxillofac Surg 35(4–5):207–211
Watanabe H, Honda E, Tetsumura A, Kurabayashi T (2011) A comparative study for spatial resolution and subjective image characteristics of a multi-slice CT and a cone-beam CT for dental use. Eur J Radiol 77(3):397–402
Torres MG, Campos PS, Segundo NP, Navarro M, Crusoé-Rebello I (2012) Accuracy of linear measurements in cone beam computed tomography with different voxel sizes. Implant Dent 21(2):150–155
Nikneshan S, Aval SH, Bakhshalian N, Shahab S (2014) Mohammadpour M4, Sarikhani S5. Accuracy of linear measurement using cone-beam computed tomography at different reconstruction angles. Imaging Sci Dent 44(4):257–262
Fokas G, Vaughn VM, Scarfe WC, Bornstein MM (2018) Accuracy of linear measurements on CBCT images related to presurgical implant treatment planning: a systematic review. Clin Oral Implants Res 29(Suppl 16):393–415
Ferrus J, Cecchinato D, Pjetursson EB, Lang NP, Sanz M, Lindhe J (2010) Factors influencing ridge alterations following immediate implant placement into extraction sockets. Clin Oral Implants Res 21(1):22–29
Funding
This study was funded by the authors’ own departments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study protocol was in accordance with the Helsinki Declaration as revised in 2013 and was approved by the local ethics committee (Study Nr. 4301).
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The article does not contain any studies with human participants.
Informed consent
For the systematic review, formal consent is not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ramanauskaite, A., Becker, K., Kassira, H.C. et al. The dimensions of the facial alveolar bone at tooth sites with local pathologies: a retrospective cone-beam CT analysis. Clin Oral Invest 24, 1551–1560 (2020). https://doi.org/10.1007/s00784-019-03057-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-03057-x