Abstract
Objectives
Bisphosphonate-associated osteonecrosis of the jaw is a severe side effect in patients receiving nitrogen-containing bisphosphonates (N-BPs). One characteristic is its high recurrence rate; therefore, basic research for new therapeutic options is necessary. N-BPs inhibit the farnesylpyrophosphate synthase in the mevalonate pathway causing a depletion of the cellular geranylgeranyl pool, resulting in a constriction of essential functions of different cell lines. Geranylgeraniol (GGOH) has been proven to antagonise the negative biological in vitro effects of bisphosphonates.
Material and methods
This study analyses the influence of the isoprenoids eugenol, farnesol, R-limonene, menthol and squalene on different functions of zoledronate-treated human umbilicord vein endothelial cells (HUVEC), fibroblasts and osteogenic cells. In addition to the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl 2H-tetrazolium bromide (MTT) vitality test, the migration capacity was analysed by scratch wound assay and the morphological architecture of the treated cells by phallacidin staining.
Results
In contrast to GGOH, none of the other tested isoprenoids were able to prevent cells from having negative zoledronate effects.
Conclusions
Despite structural analogy to GGOH, the investigated isoprenoids are not able to prevent the N-BP effect. The negative impact of zoledronate on fibroblasts, HUVEC and osteogenic cells is due to inhibition of protein geranylgeranylation since the substitution of squalene and farnesyl did not have any effect on viability and wound healing capacity whereas GGOH did reduce the negative impact.
Clinical relevance
These data suggest the importance and exclusiveness of the mevalonate pathway intermediate GGOH as a potential therapeutic approach to bisphosphonate-associated osteonecrosis of the jaws.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bisphosphonates (BPs) are therapeutics used to inhibit bone resorption, for example, in the management of osteoporosis, Paget’s disease, multiple myeloma and cancer metastases to the bone. Bisphosphonate-associated osteonecrosis of the jaws (BP-ONJ) is one of the most often seen complications in patients treated with intravenous nitrogen-containing BPs (N-BPs) such as zoledronate [1–5].
Although the underlying pathophysiology of BP-ONJ is not well understood, a number of theories have been proposed. Many different tissues and cell types appear to be involved. Reduced bone remodelling as a consequence of inhibited osteogenic cells and osteoclasts is one of the most common theories [6]. Some authors describe the inhibition of angiogenesis and neovascularization resulting in an avascular osteonecrosis [7–10]. There is also evidence for reduced oral wound healing caused by inhibitory effects on soft tissue [7, 11, 8]. However, most authors believe in a multifactorial genesis with additional trigger factors [12].
Therapy consists of conservative management with antibacterial mouth rinse such as chlorhexidine or systematic antibiotic therapy and surgical interventions such as debridement and resections of the affected regions with a secure wound closure [13]. Other possibilities such as hyperbaric oxygen and soft laser therapy have been discussed controversially [13].
Previously, geranylgeraniol (GGOH) has been described as a potential new therapeutic way to prevent and treat BP-ONJ [14]. N-BPs inhibit farnesyl pyrophosphate synthase (FPPS) in the mevalonate pathway (MVP) (Fig. 1). The consequence is a suppressed biosynthesis of the isoprenoid lipids farnesyl pyrophosphate (FPP) and geranylgeraniol pyrophosphate (GGPP), which are required for posttranslational prenylation of small GTP-binding proteins of the Ras superfamily. Lipophilic modification of these G proteins is necessary for membrane interaction and biological activities like basic intracellular signalling processes [15–19]. The N-BP-induced reduction of cell viability and migration capacity in fibroblasts, osteogenic cells and human umbilicord vein endothelial cells (HUVECs) can be reduced or even neutralised by simultaneous treatment with GGOH. Marcuzzi et al. found that GGOH and several other natural isoprenoids are able to antagonise the effect of N-BPs on monocytes. Therefore, the aim of the present study was to investigate the effect of the isoprenoids (Fig. 2) farnesol (FOH), eugenol (Eu), R-(+) limonene (Li), menthol (MOH) and squalene (Sq) in comparison with GGOH on different cell lines treated with zoledronate (Zol). The isoprenoids could potentially enter the MVP and therefore avoid the inhibitory effects caused by the N-BP [20]. Cell viability, migration capacity and cell architecture of HUVECs, fibroblasts and osteogenic cells were examined.
Mevalonate pathway (MVP). The conversion from hydroxymethylglutaryl coenzyme A (HMG-CoA) to mevalonate, which is catalysed by HMG-CoA reductase, is the first step in the MVP. The condensation of two units of the follow-up intermediate isopentenylpyrophosphate (IPP, C5) and one unit of its stereoisomer dimethylallylpyrophosphate (DMAPP, C5) is catalysed by farnesylpyrophosphate (FPP) synthase, the target of nitrogen-containing bisphosphonates. Two molecules of FPP (C15) can be metabolised in a condensation reaction to squalene (C30; Sq), a precursor of cholesterol and steroid biosynthesis. On the other hand, geranylgeranyl pyrophosphate synthase can catalyse the conversion from FPP to geranylgeranyl pyrophosphate (GGPP) (C20). The isoprenoids FPP and GGPP are essential for posttranslationation prenylation of small GTPases. [39]. Non-N-BP non-nitrogen-containing bisphosphonate, N-BP nitrogen-containing bisphosphonate, FOH farnesyl, GGOH geranylgeraniol
Material and methods
Cell culture
Human gingival fibroblasts (Lonza, Basel, Switzerland: No. CC-7049), human umbilical vein endothelial cells (HUVECs; Lonza, Basel, Switzerland: No. CC-2517) and human osteogenic cells (PromoCell, Heidelberg, Germany: No. C-12720) were cultured in an incubator with 5 % CO2 and 95 % air at 37 °C. Cells were passaged at regular intervals depending on their growth characteristics using 0.25 % trypsin (Seromed Biochrom, Berlin, Germany). Fibroblasts were grown in Stroma Cell Growth Medium (Lonza, Basel, Switzerland) with 1 % penicillin-streptomycin-neomycin antibiotic mixture (PSN), 10 % fetal calf serum (FCS) and 500 ng basic fibroblast growth factor (bFGF) per 500 ml medium. HUVECs were cultured in an endothelial basal medium supplemented with 1 μg/ml hydrocortisone, 12 μg/ml bovine brain extract, 50 μg/ml gentamycine, 50 ng/ml amphothericin-B, 10 ng/ml epidermal growth factor (EGF) and 10 % FCS. Osteogenic cells were cultured in a solution composed of Dulbecco’s modified Eagle’s medium (DMEM) with 1 % PSN, 1 % l-glutamine and 10 % FCS.
Bisphosphonates/natural isoprenoids
Zometa (zoledronate) from Novartis Pharma (Basel, Switzerland) was used. Geranylgeraniol ≥85 % (GGOH); farnesol 95 %, mixture of isomers (FOH); squalene ≥98 % (Sq) R-(+) limonene (Li); (±) menthol (MOH) and eugenol (EU) were purchased from Sigma Aldrich (Saint Louis, MO, USA).
The chemical structures of the different isoprenoids are shown in Fig. 1. According to the previous study [14], testing geranylgeraniol for antagonising effects of bisphosphonates 50 μM were used.
Cell viability by MTT assay
Cell viability of the three cell lines was evaluated by 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl 2H-tetrazolium bromide (MTT) colorimetric assay (Sigma Aldrich, Saint Louis, MO, USA). Viable cells ferment tetrazolium bromide to formazan that can be photometrically measured at 550 nm after cell lysis. For the MTT assay, cells of the respective cell lines were transferred into 12-well plates (fibroblasts 40,000 cells/well; HUVEC 40,000 cells/well; osteogenic cells 40,000 cells/well); after 24 h, all cell lines were incubated with isoprenoids (GGOH, Eu, FOH, Li, Sq or MOH) in increasing concentrations (0, 10, 25, 50 and 100 μM) with and without Zol (50 μM) for 72 h. The experiment was performed in triplicate for each cell line, each isoprenoid and each concentration.
Scratch wound assay
The influence of GGOH, Eu, FOH, Li, Sq and MOH on the wound healing capacity of Zol-treated cells was investigated by scratch wound assay. The cells were seeded on 12-well plates (fibroblasts 100,000 cells/well; HUVEC 100,000 cells/well; osteogenic cells 100,000 cells/well). At 80 % cell confluence, a scratch wound was performed using a sterile P10 pipette tip. Immediately after setting the wound, all cell lines were incubated with the previously mentioned isoprenoids (0 and 10 μM) with and without Zol (50 μM). The size of the gap was measured microscopically immediately (0 h set to 100 %), 8 h, 24 h and 48 h later. The experiment was performed three times for each cell line and isoprenoid.
Cytoskeletal morphology
The cell architectures of fibroblasts, HUVEC and osteogenic cells that were treated with the different isoprenoids (0 and 10 μM) without or in combination with Zol (50 μM) for 48 h were analysed. The cells were stained with a fluorescent dye for F-actin called 7-nitrobenz-2-oxa-1.3-diazole-(NBD) phallacidin (Bodipy FL pallacidin, Molecular Probes, Leiden, Netherlands). For this, cells were fixed in 4 % paraformaldehyde, permeabilised with 0.1 % Triton X-100 and washed twice with PBS. After blocking with 1 % BSA for 30 min, the cells were incubated with NBD phallacidin for 20 min in the dark. After washing twice, samples were incubated for 2 min with the DNA fluorescent dye bisbenzimide H (Sigma Aldrich, Saint Louis, MO, USA), followed by microscopic examination with a Zeiss (AxioVision) microscope (Göttingen, Germany) using a fluorescent light source [14].
Statistical analysis
Continuous variables are expressed as mean ± SD (standard deviation). Comparisons between groups were performed via analysis of variance (ANOVA, post hoc test: Tukey) for experiments with more than two subgroups. The software SPSS 17.0 for Windows was used for calculations. A p value <0.05 was considered as statistically significant.
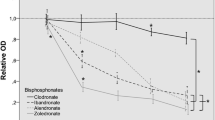
The bar in the middle of each box plot represents the median. The box itself describes the 1st and 3rd quartile. The whiskers show all values within the 1.5 interquartile ranges.
Results
MTT assay
Fibroblasts
Incubation with 50 μM Zol caused a significant reduction of fibroblast viability compared to the negative control (from 100 to 5.65 % with p < 0.001) (Fig. 3a). In every tested concentration, GGOH caused a significant enhancement of viability on Zol-treated cells (p < 0.001). Addition of 10 and 25 μM GGOH equalised the effect of Zol, and 50 and 100 μM even produced a significantly higher viability compared to the control (+36.38 %, p < 0.001 and +27.30 %, p = 0.011). The other tested isoprenoids had no positive effect on Zol-treated fibroblasts.
a–c 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl 2H-tetrazolium bromide (MTT) viability—assay for fibroblasts (a), human umbilicord vein endothelial cells (b) and osteogenic cells (c) influenced by geranylgeraniol (a), eugenol (b), farnesol (c), limonene (d), menthol (e) or squalene (f) at different concentrations with and without 50 μM zoledronate compared to a control set to 100 %. GGOH enhances viability of zoledronate-treated cells. For fibroblasts, this effect is significant in all tested concentrations (a(a)), for HUVECs in a concentration of 10 μM (b(a)) and for osteogenic cells (c(a)) in a concentration of 10, 25 and 100 μM GGOH. None of the other tested isoprenoids shows these preventing effects on cell viability (a(b–f), b(b–f) and c(b–f)
Isoprenoid effects on fibroblasts without Zol
The addition of 10 μM GGOH had a negative effect on the viability (from 100 to 69.04 %, p = 0.003); higher GGOH concentrations had no impact compared to the untreated control. Incubation with 25 μM Eu increased cell viability compared to the control (+39 %, p < 0.001). FOH showed a negative effect in each tested concentration. The addition of 50 μM Li increased the viability (+29.78 %, p = 0.004). MOH and Sq decreased the fibroblast viability at 25 μM and higher concentrations (MOH 25 μM from 100 to 76 %, p = 0.021; Sq 25 μM from 100 to 65.95 %, p < 0.001).
HUVEC—human umbilicord vein endothelial cells
Incubation with 50 μM Zol significantly reduced cell viability compared to the control (from 100 to 25.07 %, p < 0.001) (Fig. 3b). The addition of 10 μM GGOH to Zol-treated HUVEC showed a significant increase in viability from 25.07 to 86.75 % (p < 0.001). The difference of this experimental approach was not statistically significant compared to the control. The substitution of higher GGOH concentration showed no positive effect on Zol-treated cells. Other tested isoprenoids were not able to increase the viability of Zol-affected HUVEC.
Isoprenoid effects on HUVEC without Zol
Incubation with 10- and 25-μM GGOH had no significant effect on the control group, and higher concentrations caused a significant decrease in viability (50 μM GGOH (from 100 to 21.05 %) with p < 0.001 and 100 μM GGOH (from 100 to 1.38 %) with p < 0.001). The addition of 50 μM and higher concentrations of Eu reduced the viability (50 μM from 100 to 56.44 %, p < 0.001). FOH supplementation caused a significant decrease of cell viability in comparison to the control culture in a concentration of 25 μM or higher (25 μM from 100 to 57.24 % with p < 0.001, 50 μM from 100 to 16.91 % with p < 0.001, 100 μM from 100 to 0.99 % with p < 0.001). In cells incubated with Li, only a high concentration (100 μM) showed a significant reduction (from 100 to 5.16 % with p < 0.001). Treatment with MOH had no influence on viability. Remarkable is the upward trend at 10 μM of this isoprenoid (from 100 to 126.99 % with p = 0.062). Sq showed no difference compared to the control culture.
Osteogenic cells
The viability of Zol-treated osteogenic cells was significantly reduced compared to the control culture (from 100 to 32.33 %, p < 0.001) (Fig. 3c). The substitution of GGOH had a positive effect, which was significant for 10 μM (from 32.33 to 64.60 %, p = 0.002), 25 μM (from 32.33 to 63.74 %, p = 0.002) and 100 μM (from 32.33 to 58.64 %, p = 0.018). The other tested isoprenoids were not able to increase the viability of Zol-treated cells.
Isoprenoid effects on osteogenic cells without Zol
Incubation with 50 and 100 μM GGOH had a negative impact compared to the control culture (50 μM from 100 to 73.6 %, p < 0.001 and 100 μM from 100 to 74.52 %, p < 0.001). FOH significantly reduced viability in each tested concentration. Li had a negative influence above 25 μM (25 μM from 100 to 64.89 %, p = 0.001). Treatment with Eu, MOH and Sq showed no effect on cell viability compared to the control sample.
Scratch wound assay
Fibroblasts
In untreated cells, the size of the gap was reduced from 100 to 54.73 % after 8 h (Fig. 4a). After 24 h, the gap was nearly closed with 0.42 %. The reduction in wound healing capacity for Zol-treated fibroblasts became statistically significant after 24 h compared to the control (p < 0.001). At this moment, the gap had a size of 43.68 %; after 48 h, it still had 22.71 % of the initial size. Co-treatment with GGOH enhanced wound healing ability after 24 h compared to incubation with Zol alone (p < 0.001). The size of the wound was 6.54 % at this point in time and was completely closed after 48 h. Next to GGOH, the co-treatment with Eu (24 h 22.69 % and 48 h 10.53 %) had a positive effect on wound closure compared to the incubation with Zol alone. The difference to fibroblasts treated with Zol alone was significant after 24 h (p < 0.001). But the positive effect of co-treatment was significantly higher for GGOH than for Eu (p = 0.003). The other tested isoprenoids had no effect on the wound healing capacity of zoledronate-treated cells.
a–c Scratch wound assay of fibroblasts (a), human umbilicord vein endothelial cells (b) and osteogenic cells (c) influenced by 10 μM of geranylgeraniol (a), eugenol (b), farnesol (c), limonene (d), menthol (e) or squalene (f) with and without 50 μM zoledronate (Zol) in percentage to the initial dehiscence. After 24 h, the size of the artificial wound in the monolayer of zoledronate-treated fibroblasts is significantly reduced by the isoprenoids GGOH (a(a)) and Eu (a(b)). Only GGOH is able to enhance the wound healing capacity of zoledronate-affected HUVECs and osteogenic cells. Whereas the positive effect on osteogenic cells appears after 24 h (c(a)), there is already a significant influence of GGOH on zoledronate-treated HUVECs after 8 h (b(a))
Isoprenoid effects on fibroblasts without Zol
None of the investigated isoprenoids had any influence on wound healing of fibroblasts.
HUVEC—human umbilicord vein endothelial cells
After 8 h, the gap compared to the control group was reduced from 100 to 51.54 %, after 24 h to 9.91 %, and after 48 h, the wound in the cell layer was completely closed (Fig. 4b). Treatment with Zol reduced wound closure to 69.87 % after 8 h (p = 0.019).
The additional treatment with GGOH accelerated wound healing to a gap size of 50.00 % after 8 h (p = 0.006). After 24 h, the size of the gap was only reduced to 53.14 % in HUVECs treated with Zol (p < 0.001 compared to the control culture). The addition of GGOH caused a significant enhancement to the wound healing capacity (8.72 % at 24 h) compared to incubation with Zol alone (p < 0.001). Furthermore, there was no difference to the untreated control. After 48 h, the wound in the cell layer of the control was completely closed; at this particular time, the gap of Zol-treated HUVECs still had 36.82 % of the original size. The additional treatment with GGOH led to a complete wound closure (p < 0.001 compared to zoledronate-treated cells). None of the other tested isoprenoids showed a positive influence on wound healing at any time.
Isoprenoid effects on HUVEC without Zol
Whereas GGOH and Sq showed no influence, the single addition of Eu caused a significant reduced wound healing capacity compared to the control at any time (the size of the gap after 8 h, 69.85 % with p = 0.036; 24 h, 48.09 % and 48 h, 24.43 %, both with p < 0.001). FOH-treated cells showed a decreased wound healing capacity compared to control after 24 h (32.30 % with p = 0.003). Li caused a significant reduction at any time (the size of the gap after 8 h, 70.0 % with p = 0.033; 24 h, 36.52 % with p < 0.001; 48 h, 20.87 % with p = 0.006). MOH showed a significantly reduced wound healing capacity after 24 h (41.24 % with p < 0.001), and after 48 h, there was a negative trend compared to the untreated control (16.32 % with p = 0.064).
Osteogenic cells
The artificial wound in the osteogenic cell culture was reduced to 86.43 % after 8 h, 26.25 % after 24 h, and after 48 h, the wound was completely closed (Fig. 4c). Additional treatment with Zol reduced the wound healing to 53.33 % after 24 h (p < 0.001) and to 25.00 % after 48 h (p < 0.001). Substitution of GGOH antagonised the Zol effect, with 27.61 % (p < 0.001) after 24 h and a complete wound closure after 48 h (p < 0.001). Substitution of Li had a temporary positive effect on wound healing after 24 h compared to Zol-treated osteogenic cells (size of the gap, 40.69 % with p = 0.054). The other isoprenoids had no positive effect on Zol-affected osteogenic cells at any time point.
Isoprenoid effects on osteogenic cells without Zol
The addition of FOH had a negative effect on wound healing after 24 h (51.43 % with p < 0.001) and 48 h (23.81 % with p < 0.001) as well as the addition of MOH (24 h, 43.56 % with p = 0.005 and 48 h, 22.15 % with p < 0.001). Whereas Eu showed a reduced wound healing after 24 h only (the size of the gap, 39.22 % with p = 0.026), the addition of GGOH, Li and Sq had no influence compared to the control.
Cytoskeletal morphology
In contrast to the untreated controls, which showed a sub-confluent layer with well-spread cells and a characteristic morphology, Zol had a destroying influence on architecture and number of all tested cell types. Cell density is considerably reduced, so that there are only a few contact points between the individual cells (Fig. 5a–c control).
Cell architecture of fibroblasts (a), HUVEC (b) and osteogenic cells (c) without isoprenoid (control) or treated with 10 μM of geranylgeraniol (GGOH), eugenol (Eu), farnesol (FOH), R-(+) limonene (Li); (+) menthol (MOH) or squalene (Sq) in the absence of zoledronate (−Zol) or with 50 μM zoledronate (+Zol). Cell architecture is disturbed by zoledronate in all tested cell types. GGOH is the only isoprenoid that prevents cells from this negative effect. Cells without zoledronate are not visibly affected
Cell bodies of HUVEC (Fig. 5b, +Zol) and osteogenic cells (Fig. 5c, +Zol) have an atrophic and spindle-shaped morphology with long hair-like cell fibres, while the architecture of fibroblasts is rather triangular (Fig. 5a, +Zol). GGOH was the only isoprenoid able to prevent cells from these effects. The other isoprenoids did not affect actin skeletal organisation in cell cultures not exposed to Zol (Fig. 5a–c, −Zol)
Discussion
In the absence of causal treatment options for BP-ONJ, the administration of GGOH has been suggested as a new therapeutic option [14]. The natural isoprenoid GGOH, which is metabolised to geranylgeranyl pyrophosphate (GGPP), is the substrate for prenylation of most GTP-binding proteins. In the present study, other isoprenoids have been tested in order to identify their ability and effectiveness to bypass the N-BP-induced block in the MVP, and the results have been compared to the effect of GGOH.
In addition to sesquiterpene FOH and triterpene Sq, both intermediates of the MVP, the three monoterpenes Eu, Li and MOH have been investigated (Fig. 1). A limit of the present study is that many isoprenoids have been tested and therefore, only few concentrations of the isoprenoids were used. Eu was the only compound which showed a protective influence on Zol-treated cells, but this effect was limited to the wound healing capacity of fibroblasts as of 24 h and was significantly lower than the positive influence of GGOH (p < 0.001). As well, neither viability nor cell architecture of any type was restored by Eu. None of the other isoprenoids showed antagonising effects to Zol, not even at the concentration of 100 μM. These data suggest that only the diterpene GGOH is able to prevent a N-BP-induced reduction in wound healing capacity, viability and destruction of the internal framework of actin filaments in gingival fibroblasts, HUVECs and osteogenic cells.
This observation suggests that the lack of protein geranylgeranylation, rather than protein farnesylation, mainly provoke the N-BP-induced suppression of elemental cell functions in the tested cell lines. There is evidence that only a little FPP (15-carbon chain), the phosphorylation product of FOH, is metabolised further to GGPP (20-carbon chain). The main part of FPP is used for protein farnesylation [19, 21]. The farnesyl group is known to modify Ras proteins and lamin B, whereas the majority of GTP-binding proteins depend on geranylgeranylation [18]. Geranylgeranylated proteins appear to be of paramount importance in cell cycle progression, in contrast to farnesylated proteins [22]. In agreement with the present findings, Van Beek et al. already showed that GGOH and not FOH was able to reverse N-BP effects on osteoclast-mediated bone resorption [23]. Another study demonstrated that GGOH and not FOH was able to overcome the Zol-induced block in prostate cancer cells. In this cell type, the independence of Ras GTPase in zoledronate’s underlying mode of action was shown [24]. In contrast, there is evidence that different natural isoprenoids (geranylgeraniol, geraniol, farnesol and menthol) are capable of reducing the pro-inflammatory effects in LPS-stimulated monocytes which were simultaneously treated with N-BPs [20]. Other data show a complete prevention of the effects caused by the hydroxymethylglutaryl coenzyme A (HMG-CoA) reductase inhibitor lovastatin on the MVP (statins’ point of action is shown in Fig. 2) in mesangial cells by GGOH and FOH. Most likely, the relevance of the two forms of protein prenylation is cell specific [23].
The addition of the triterpene Sq, a downstream intermediate of FFP in the MVP (Fig. 2), did not reverse the N-BP-induced effects in this study. Although reduced effects of N-BP on serum cholesterol levels were reported [25], the depletion of Sq and a consequently decreased de novo synthesis of steroids do not seem to be overly important in the prevention of cell viability, migration capacity and cell architecture in the tested cell lines compared to the lack of protein prenylation.
In addition to the intermediates of the MVP (Fig. 1a), three natural isoprenoids with monoterpene structure have been analysed (Fig. 1b). Monoterpenes are the main ingredients of essential oils and are found almost everywhere in the plant kingdom [26]. In this in vitro study, d-Li was tested, one of the most common terpenes in nature, which is an essential part of many citrus oils with characteristic fragrance [27]. In addition to the effect of Eu, the major part of clove oil was analysed. Because of its anti-inflammatory, analgesic and locally anaesthetic effect, it is widely used in dentistry [28]. MOH is another monoterpene often used in medical treatment due to its antiseptic, analgesic and cooling effects [29]. As mentioned, none of the three selected terpenes was able to overcome the Zol-induced effects in fibroblasts, HUVEC and osteogenic cells.
Next to this, the effect on cells of the immune system would be interesting to address since bisphosphonates do have a positive effect on those cells [30].
Several studies have investigated the chemotherapeutic potential of diverse monoterpenes that interact and suppress the MVP. Initially, isoprenyl transferases (farnesyl transferase and geranylgeranyl transferase, shown in Fig. 2) were considered to be the main target; they were believed to be inhibited by these isoprenoid compounds, and their loss of function was thought to be responsible for the decreased prenylation of small GTPases [31]. Further investigations found out that the inhibition of protein prenylation required very high monoterpene concentrations (1.5 to 10 mM) [32]. The HMG-CoA reductase inhibitor lovastatin is known to reduce protein prenylation due to a depletion of the farnesyl and geranylgeranyl diphosphonate pool [33]. The consequence is an up-regulation of unprenylated proteins of the Ras superfamily [34]. In concentrations from 0.25 to 0.75 mM, a monoterpene-mediated inhibition of the lovastatin causing up-regulation of these nascent proteins was demonstrated. This supports the theory of a regulatory function of the monoterpenes in the expression of small GTPases independent of protein prenylation [35].
Furthermore, an isoprenoid-mediated down-regulation of HMG-CoA reductase at the post-transcriptional level has been reported [36]. The mevalonate pathway of tumour cells seems to be more sensitive to exogenous isoprenoids because their HMG-CoA reductase activity is resistant to complex sterol-mediated feedback regulation, which is the primary mechanism in somatic cells [37].
In addition to the chemotherapeutic approach, suppressed bone resorption generated by monoterpenes was observed [38]. The fact that neither GGOH nor FOH was able to reverse the monoterpene-induced effects on suppressed formation of osteoclasts in vitro supports the theory of a monoterpene effect independent of cellular isoprenoid synthesis [26].
Conclusion
The negative impact of Zol on fibroblasts, HUVEC and osteogenic cells is due to inhibition of protein geranylgeranylation since the substitution of Sq and FOH did not have any effect on viability and wound healing capacity of zoledronate-treated cells whereas GGOH did reduce the negative impact of Zol. The hypothesis that natural isoprenoids other than GGOH can restore N-BP-induced restriction of cell viability, the internal framework of actin filaments and the wound healing capacity of the tested cell lines could not be confirmed.
At present, GGOH seems to be the only natural isoprenoid that is able to overcome the effects of Zol on gingival fibroblasts, HUVEC and osteogenic cells. To date, GGOH seems to be the only substance that could possibly influence the development of BP-ONJ in patients with bisphosphonate treatment and the treatment outcome in patients with BP-ONJ.
References
Kuhl S, Walter C, Acham S, Pfeffer R, Lambrecht JT (2012) Bisphosphonate-related osteonecrosis of the jaws—a review. Oral Oncol 48(10):938–947. doi:10.1016/j.oraloncology.2012.03.028
Walter C, Al-Nawas B, du Bois A, Buch L, Harter P, Grotz KA (2009) Incidence of bisphosphonate-associated osteonecrosis of the jaws in breast cancer patients. Cancer 115(8):1631–1637. doi:10.1002/cncr.24119
Walter C, Al-Nawas B, Frickhofen N, Gamm H, Beck J, Reinsch L, Blum C, Grotz KA, Wagner W (2010) Prevalence of bisphosphonate associated osteonecrosis of the jaws in multiple myeloma patients. Head Face Med 6:11. doi:10.1186/1746-160X-6-11
Walter C, Al-Nawas B, Grotz KA, Thomas C, Thuroff JW, Zinser V, Gamm H, Beck J, Wagner W (2008) Prevalence and risk factors of bisphosphonate-associated osteonecrosis of the jaw in prostate cancer patients with advanced disease treated with zoledronate. Eur Urol 54(5):1066–1072. doi:10.1016/j.eururo.2008.06.070
Walter C, Grotz KA, Kunkel M, Al-Nawas B (2007) Prevalence of bisphosphonate associated osteonecrosis of the jaw within the field of osteonecrosis. Support Care Cancer 15(2):197–202. doi:10.1007/s00520-006-0120-z
Mashiba T, Mori S, Burr DB, Komatsubara S, Cao Y, Manabe T, Norimatsu H (2005) The effects of suppressed bone remodeling by bisphosphonates on microdamage accumulation and degree of mineralization in the cortical bone of dog rib. J Bone Mineral Metab 23(Suppl):36–42
Walter C, Klein MO, Pabst A, Al-Nawas B, Duschner H, Ziebart T (2010) Influence of bisphosphonates on endothelial cells, fibroblasts, and osteogenic cells. Clin Oral Investig 14(1):35–41. doi:10.1007/s00784-009-0266-4
Walter C, Pabst A, Ziebart T, Klein M, Al-Nawas B (2011) Bisphosphonates affect migration ability and cell viability of HUVEC, fibroblasts and osteoblasts in vitro. Oral Dis 17(2):194–199. doi:10.1111/j.1601-0825.2010.01720.x
Ziebart T, Pabst A, Klein MO, Kammerer P, Gauss L, Brullmann D, Al-Nawas B, Walter C (2011) Bisphosphonates: restrictions for vasculogenesis and angiogenesis: inhibition of cell function of endothelial progenitor cells and mature endothelial cells in vitro. Clin Oral Investig 15(1):105–111. doi:10.1007/s00784-009-0365-2
Pabst AM, Ziebart T, Ackermann M, Konerding MA, Walter C (2013) Bisphosphonates’ antiangiogenic potency in the development of bisphosphonate-associated osteonecrosis of the jaws: influence on microvessel sprouting in an in vivo 3D Matrigel assay. Clin Oral Investig 18(3):1015–1022. doi:10.1007/s00784-013-1060-x
Pabst AM, Ziebart T, Koch FP, Taylor KY, Al-Nawas B, Walter C (2012) The influence of bisphosphonates on viability, migration, and apoptosis of human oral keratinocytes—in vitro study. Clin Oral Investig 16(1):87–93. doi:10.1007/s00784-010-0507-6
Walter C, Laux C, Sagheb K (2013) Radiologic bone loss in patients with bisphosphonate-associated osteonecrosis of the jaws: a case-control study. Clin Oral Investig 18(2):385–390. doi:10.1007/s00784-013-0974-7
Grötz KA, Piesold JU, Al-Nawas B (2012) S3-Leitlinie: Bisphosphonat-assoziierte Kiefernekrose (BP-ONJ) und andere Medikamenten-assoziierte Kiefernekrosen. Leitlinie der Deutschen Gesellschaft für Mund-, Kiefer- und Gesichtschirurgie. Accessed Dec. 2012
Ziebart T, Koch F, Klein MO, Guth J, Adler J, Pabst A, Al-Nawas B, Walter C (2011) Geranylgeraniol—a new potential therapeutic approach to bisphosphonate associated osteonecrosis of the jaw. Oral Oncol 47(3):195–201. doi:10.1016/j.oraloncology.2010.12.003
Reszka AA, Rodan GA (2003) Mechanism of action of bisphosphonates. Curr Osteoporos Rep 1(2):45–52
Walker K, Olson MF (2005) Targeting Ras and Rho GTPases as opportunities for cancer therapeutics. Curr Opin Genet Dev 15(1):62–68. doi:10.1016/j.gde.2004.11.001
Virtanen SS, Vaananen HK, Harkonen PL, Lakkakorpi PT (2002) Alendronate inhibits invasion of PC-3 prostate cancer cells by affecting the mevalonate pathway. Cancer Res 62(9):2708–2714
Casey PJ (1992) Biochemistry of protein prenylation. J Lipid Res 33(12):1731–1740
Shipman CM, Croucher PI, Russell RG, Helfrich MH, Rogers MJ (1998) The bisphosphonate incadronate (YM175) causes apoptosis of human myeloma cells in vitro by inhibiting the mevalonate pathway. Cancer Res 58(23):5294–5297
Marcuzzi A, Tommasini A, Crovella S, Pontillo A (2010) Natural isoprenoids inhibit LPS-induced-production of cytokines and nitric oxide in aminobisphosphonate-treated monocytes. Int Immunopharmacol 10(6):639–642. doi:10.1016/j.intimp.2010.03.008
Crick DC, Andres DA, Waechter CJ (1997) Novel salvage pathway utilizing farnesol and geranylgeraniol for protein isoprenylation. Biochem Biophys Res Commun 237(3):483–487. doi:10.1006/bbrc.1997.7145
Vogt A, Qian Y, McGuire TF, Hamilton AD, Sebti SM (1996) Protein geranylgeranylation, not farnesylation, is required for the G1 to S phase transition in mouse fibroblasts. Oncogene 13(9):1991–1999
Van Beek ER, Lowik CW, Papapoulos SE (2002) Bisphosphonates suppress bone resorption by a direct effect on early osteoclast precursors without affecting the osteoclastogenic capacity of osteogenic cells: the role of protein geranylgeranylation in the action of nitrogen-containing bisphosphonates on osteoclast precursors. Bone 30(1):64–70
Nogawa M, Yuasa T, Kimura S, Kuroda J, Segawa H, Sato K, Yokota A, Koizumi M, Maekawa T (2005) Zoledronic acid mediates Ras-independent growth inhibition of prostate cancer cells. Oncol Res 15(1):1–9
Guney E, Kisakol G, Ozgen AG, Yilmaz C, Kabalak T (2008) Effects of bisphosphonates on lipid metabolism. Neuroendocrinol Lett 29(2):252–255
Dolder S, Hofstetter W, Wetterwald A, Muhlbauer RC, Felix R (2006) Effect of monoterpenes on the formation and activation of osteoclasts in vitro. J Bone Miner Res 21(4):647–655. doi:10.1359/jbmr.060111
Matura M, Goossens A, Bordalo O, Garcia-Bravo B, Magnusson K, Wrangsjo K, Karlberg AT (2002) Oxidized citrus oil (R-limonene): a frequent skin sensitizer in Europe. J Am Acad Dermatol 47(5):709–714
Pramod K, Ansari SH, Ali J (2010) Eugenol: a natural compound with versatile pharmacological actions. Nat Prod Commun 5(12):1999–2006
Patel T, Ishiuji Y, Yosipovitch G (2007) Menthol: a refreshing look at this ancient compound. J Am Acad Dermatol 57(5):873–878. doi:10.1016/j.jaad.2007.04.008
Hagelauer N, Pabst AM, Ziebart T, Ulbrich H, Walter C (2014) In vitro effects of bisphosphonates on chemotaxis, phagocytosis, and oxidative burst of neutrophil granulocytes. Clin Oral Investig. doi:10.1007/s00784-014-1219-0
Crowell PL, Chang RR, Ren ZB, Elson CE, Gould MN (1991) Selective inhibition of isoprenylation of 21-26-kDa proteins by the anticarcinogen d-limonene and its metabolites. J Biol Chem 266(26):17679–17685
Hardcastle IR, Rowlands MG, Barber AM, Grimshaw RM, Mohan MK, Nutley BP, Jarman M (1999) Inhibition of protein prenylation by metabolites of limonene. Biochem Pharmacol 57(7):801–809
Wang IK, Lin-Shiau SY, Lin JK (2000) Suppression of invasion and MMP-9 expression in NIH 3T3 and v-H-Ras 3T3 fibroblasts by lovastatin through inhibition of ras isoprenylation. Oncology 59(3):245–254
Holstein SA, Wohlford-Lenane CL, Hohl RJ (2002) Isoprenoids influence expression of Ras and Ras-related proteins. Biochemistry 41(46):13698–13704
Holstein SA, Hohl RJ (2003) Monoterpene regulation of Ras and Ras-related protein expression. J Lipid Res 44(6):1209–1215. doi:10.1194/jlr.M300057-JLR200
Peffley DM, Gayen AK (2003) Plant-derived monoterpenes suppress hamster kidney cell 3-hydroxy-3-methylglutaryl coenzyme a reductase synthesis at the post-transcriptional level. J Nutr 133(1):38–44
Mo H, Elson CE (2004) Studies of the isoprenoid-mediated inhibition of mevalonate synthesis applied to cancer chemotherapy and chemoprevention. Exp Biol Med (Maywood) 229(7):567–585
Muhlbauer RC, Lozano A, Palacio S, Reinli A, Felix R (2003) Common herbs, essential oils, and monoterpenes potently modulate bone metabolism. Bone 32(4):372–380
Thurnher M, Nussbaumer O, Gruenbacher G (2012) Novel aspects of mevalonate pathway inhibitors as antitumor agents. Clin Cancer Res 18(13):3524–3531. doi:10.1158/1078-0432.CCR-12-0489
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hagelauer, N., Ziebart, T., Pabst, A.M. et al. Bisphosphonates inhibit cell functions of HUVECs, fibroblasts and osteogenic cells via inhibition of protein geranylgeranylation. Clin Oral Invest 19, 1079–1091 (2015). https://doi.org/10.1007/s00784-014-1320-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-014-1320-4