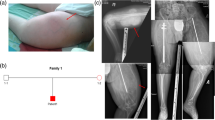
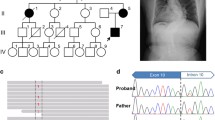
Abstract
Introduction
Osteogenesis imperfecta (OI) is a well-known heritable disorder of connective tissue characterized by skeletal fragility and low bone mass. Nearly 90% of patients with OI have disease variants in COL1A1 and COL1A2 that encode for the α1 and α2 chains of type I collagen.
Materials and methods
A retrospective analysis of 185 probands who were diagnosed with OI in Shanghai Jiao Tong University Affiliated Sixth People’s Hospital from March 2005 to December 2019 was performed.
Results
A total of 140 mutations in COL1A1 and 45 mutations in COL1A2 were identified, of which 18 variations were novel. In the phenotype analysis, there were more sporadic cases than familial OI cases in China (54.6% vs. 45.4%, P < 0.001). A total of 98.9% of patients presented with a fracture history. The most common fracture sites were extremity long bones (femur, tibia-fibula and radius-ulna accounted for 36.6%, 17.1% and 11.7%, respectively). Patients with OI types III and IV, especially type III, had a higher proportion of dentinogenesis imperfecta (DI) than patients with OI type I (55% vs. 28%, P < 0.001). Interestingly, G767S and D1219N in COL1A1 and G337S in COL1A2 were the most frequent (3.52%, 2.11% and 8.89%, respectively), which seem to be hotspot mutations in the COL1A1 and COL1A2 genes in Chinese patients.
Conclusions
This study describes the mutations in the main pathogenic genes, COL1A1 and COL1A2, and the clinical characteristics of osteogenesis imperfecta in China. Furthermore, these findings help reveal the genetic basis of Asian OI patients and contribute to genetic counselling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteogenesis imperfecta (OI; MIM 166200, 166210, 259420, and 166220) is the most prevalent form of brittle fracture in children, with an approximate incidence of 1/15,000 to 20,000 births [1]. In addition, other clinical phenotypes, including blue or grey sclera, dentinogenesis imperfecta (DI), short stature, hyperlaxity of ligaments and skin, and progressive conductive hearing loss, are common in OI patients [2].
Nearly 90% of patients with OI have disease variants in COL1A1 and COL1A2 that encode the α1 and α2 chains of type I collagen, a major protein of skin and bone matrix [3]. There are two common mutation mechanisms: nonsense mutation and frameshift mutation in COL1A1, which can reduce the amount of type I collagen synthesis by half through nonsense mediated mRNA degradation (NMD), resulting in insufficient single dose of type I collagen; triple helix missense mutation in COL1A1 and COL1A2 can affect the triple helix synthesis of collagen and its relationship with the outer matrix through dominant negative effect, resulting in three helix structure variation of type I collagen and severe phenotype [4,5,6]. Over the past decade, many genes have been found to be responsible for OI, highlighting the genetic heterogeneity of the disease. However, there is no clear relationship between the type of gene mutation and clinical manifestation [3, 7,8,9,10,11]. Short stature often accompanies the whole growth and development stage of patients. Previous studies have shown that final height in individuals with OI is below the normal mean of the general population [12, 13]. Nevertheless, few studies have systematically analysed the clinical phenotypes of patients with different genotypes in OI. To our knowledge, there are few such studies performed in Asia [14,15,16].
In the present study, we collected clinical data on 185 probands with OI from different Chinese families who carried mutations in the COL1A1 or COL1A2 genes to further illuminate genotype–phenotype correlations.
Materials and methods
Subjects
This study is a retrospective review of 185 probands followed at the Department of Osteoporosis and Bone Disease, Shanghai Jiao Tong University Affiliated Sixth People's Hospital (Shanghai, China) from 2005 to 2019 who had clinical features of OI and known or novel mutations in the related genes. These probands included 84 familial cases and 101 sporadic cases belonging to 185 unrelated families. The 185 probands in the study included 117 patients previously published in journals [2, 17]. The study was approved by the Ethics Committee of Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, and informed consent was obtained from all the patients or from patients’ parents if they were under 18 years old.
Analysis for gene mutations
Genomic DNA was extracted from 2 mL peripheral blood samples using the QuickGene DNA Whole Blood Kit (Kurabo Industries Ltd, Osaka, Japan) and a Nucleic Acid Isolation system (QuickGene-610L; Autogen, Inc, Holliston, MA, USA). Sanger sequencing was used to analyse the COL1A1 and COL1A2 genes of 185 probands and the mutation sites of their parents. All exons of the COL1A1 and COL1A2 genes, including the exon–intron boundaries, were amplified by polymerase chain reaction using an ABI 3730 automated sequencer and the Big Dye Terminator Sequencing protocol (ABI). The genomic sequences (NG_007400.1 and NG_007405.1) and mRNA sequences (NM_000088.4 and NM_000089.4) of the COL1A1 and COL1A2 genes were used as reference sequences. DNA mutation numbering was based on the cDNA sequence using the A of the ATG translation initiation start site as nucleotide + 1. Sequencing results were compared with the OI variant databases (https://oi.gene.le.ac.uk/). Variants were defined as novel if not available here. Mutations were classified as haploinsufficiency (frame shifts owing to small insertions or deletions, point mutations that create termination codons, and some splice-site mutations) or helical mutation (the substitution of glycine by another amino acid in the triple-helical domain of either the α1 or the α2 chain) [18].
Bone densitometry
The bone mineral density (BMD; g/cm2) of the lumbar spine (L1-L4) and proximal hip was measured by dual energy X-ray absorptiometry (DXA) (Lunar, Madison, WI, USA; Hologic, Boston, MA, USA). All DXA scans were conducted by a specially trained specialist. BMD results were converted to age- and gender-specific Z-score-matched normal Chinese children. Of the 185 probands, 139 underwent the examination, and none of the patients received prior bisphosphonate treatment in this study.
Clinical characteristics
Detailed clinical features were collected through a medical examination. According to Sillence [19], patients were clinically classified into four types (types I–IV). Due to the retrospective review, some phenotypic data are incomplete, resulting in variation in the number of observations for the different clinical characteristics.
Height was defined as the vertical dimension between the top of the head and soles of the feet. All height measurements were recorded as standard deviations (SD) in relation to age- and sex-specific reference data (Z-score) supplied by the Chinese National Centers for Disease Control and Prevention [20]. In children, body length was measured from the heels to the top of the head in the supine position when they were too young to stand. Data on fractures were collected as the total number of fractures, and fracture sites were also recorded for further analysis. The age of onset of OI was defined as the age at which OI related symptoms, such as fracture, hearing loss, dentinogenesis imperfecta, etc., first appeared. Scleral hue was judged clinically, and all hues in the blue-grey scale were digitally recorded as ‘blue’ compared with ‘white’. The diagnosis of DI was based on clinical examination, performed by a dentist specialized in this condition.
Serum levels of alkaline phosphatase (ALP), calcium (Ca) and phosphate (P) were measured by automated analyzers. Serum levels of beta cross-linked carboxy-terminal telopeptide of type I collagen (β-CTX), 25-hydroxyvitamin D (25OHD) and intact parathyroid hormone (PTH) were determined using an automated Roche electrochemiluminescence system (E170; Roche Diagnostic GmbH, Mannheim, Germany). All these serum biochemical parameters were measured in the central clinical laboratory of Shanghai Jiao Tong University Affiliated Sixth People’s Hospital.
Statistical analyses
Statistical analyses were performed in SPSS for Windows version 20.0 (SPSS Inc, Chicago, IL). The distribution of continuous variables was determined by the Kolmogorov–Smirnov test. The continuous variables of normal distribution were expressed as the mean ± standard deviation and compared using Student’s t-test. Nonnormally distributed continuous variables, presented as medians (25th and 75th percentiles), were compared by the Mann–Whitney U-test. In a covariance analysis, serum levels of biochemical indices were compared in different groups after adjusting for age and gender. The chi-square test was used to analyse categorical variables. A P value < 0.05 was considered statistically significant.
Results
Patient characteristics
The study cohort consisted of 185 probands (113 males, 72 females) aged 2 months to 74 years (median age 12 years). A total of 84 probands had familial OI (45.4%), and 101 were sporadic cases. Most of the probands were diagnosed due to fragility fractures in their daily activities, and the median number of fractures was 5 at the first visit. Table 1 summarizes the presentation of features at the time of initial visits of diagnosis. The average age at presentation was 16 years (7, 24; 25th, 75th percentile; N = 185). A total of 98.9% of patients presented with a fracture history. A total of 132 probands presented with blue sclerae (71.4%), 65 probands suffered from dentinogenesis imperfecta (35.1%), 12 probands suffered from scoliosis (6.5%), and 14 patients had hearing loss (7.5%), including 7 cases of hearing loss in both ears and 7 cases of unilateral hearing loss. Moreover, the total number of fractures in specific locations recorded in this study was 820, and the most common fracture sites were extremity long bones (femur, tibia-fibula and radius-ulna accounted for 36.6%, 17.1% and 11.7%, respectively, Fig. 1).
COL1A1 and COL1A2 mutations
Gene mutational analysis of the 185 pedigrees revealed 140 mutations in COL1A1 and 45 mutations in COL1A2 (Table 4). In this study, 101 probands with OI had de novo mutations in the COL1A1 or COL1A2 gene, and 84 families showed autosomal dominant inheritance. Among the family cases, 47 fathers and 37 mothers carried mutant genes and developed the disease, respectively. According to the Type 1 Collagen Mutation Database, 19 gene mutations (10.3%) had not been previously reported, which included 4 missense mutations, 2 splicing mutations, 4 deletions, 4 duplications in COL1A1 and 4 missense mutations, and 1 insertion in COL1A2 (Table 2). Mutation frequencies were analysed by individual.
In addition, the 140 mutation sites detected in COL1A1 were distributed throughout nearly all the exons and flanking intronic sequences, but no mutation was detected outside exons 5–11 or 20–27 in COL1A2. Nearly a third of the probands (33.0%; 61/185) had a substitution mutation of the glycine within the Gly-X–Y triplet domain of the triple helix, of which 36 were in COL1A1 and 25 were in COL1A2. Substitutions of a serine amino acid were the most common in the present study. Among all the point mutations, G767S, D1219N in COL1A1 and G337S in COL1A2 were the most frequent (3.52%, 2.11% and 8.89%, respectively).
Genotype–phenotype correlation
According to the Sillence classification, this study included 108 (58.4%) patients with type I, 44 (23.8%) patients with type III, 33 (17.8%) patients with type IV OI. There was no remarkable difference in gender between different genes (P = 0.491) and there was no difference in age of onset and visit between patients with qualitative mutations and those with quantitative mutations (P = 0.217, Table 3). Compared with patients with haploinsufficiency, patients with helical glycine mutations had lower BMD at the lumbar spine (Z-score, − 2.3 ± 0.8, P = 0.016) and femoral neck (Z-score, − 1.8 ± 1.0, P = 0.042), lower positive family history (28% vs. 59%, P = 0.001), lower hearing loss (0% vs. 7%, P = 0.044) and higher rates of fractures (5.2 ± 2.3, P < 0.001). In addition, there was no significant difference in serum levels of Ca, P, PTH, ALP, β-CTX and height among different types of OI. In the COL1A1 mutation group, the most common fracture site was the femur (n = 198; 31.1%), followed by tibia/fibula (n = 118; 18.5%) radius/ulna (n = 70; 11.0%) and elbow (n = 61; 9.6%). In the COL1A2 mutation group, the most common fracture site was the femur (n = 102; 55.7%), followed by radius/ulna (n = 26; 14.2%) and tibia/fibula (n = 22; 12.0%) (Table 4).
Discussion
The present study with a large sample size investigated the genotype–phenotype relationship in patients with OI in China. We found 185 mutations in COL1A1 and COL1A2, including 18 novel mutations.
In the phenotype analysis, there were more sporadic cases than familial OI cases in China (55.1% vs. 44.9%, P < 0.001). Although the proportion of familial OI cases is still lower than that of South Korea [21], whose inhabitants are also descended from Mongolia, it is higher than that of our previous research [17]. As we assumed before, the low proportion of family OI patients in China is the product of the national family planning policy, and with the implementation of the open two child policy in China, family OI patients have increased. In our study, OI type I (58.4%) was the most common type. A total of 70.6% of the probands presented with blue sclerae, and the colour of the sclera faded with age. The height of patients with OI was lower than that of the normal population, which confirmed that multiple fractures can cause limb shortening, while there was no significant difference among patients with different genes mutations. In terms of extraskeletal manifestation, blue sclerae was a common feature of patients with OI, in which there was no difference.
Similar to previous studies, patients with types III and IV OI, especially type III, had a higher proportion of DI than patients with type I (55% vs. 28%, P < 0.001), which may be explained by the fact that bone and dentin were similar in the composition of the extracellular matrix [2, 22]. Hearing loss is a common secondary feature of OI in adults, often with mixed conductive and sensorineural deficiency [23]. In the present study, 7.5% of probands had hearing loss, but the proportion was lower than reports in Scotland (23%) [24] and Denmark (24%) [25], which may be because the majority of the patients were children. In addition, a relationship between hearing loss and genotype was not found in this study.
In the genotype analysis, substitutions of glycine by serine were the most common in COL1A1 and COL1A2, and the number of mutations observed in COL1A1 was almost three times that of COL1A2 [2]. In the present study, missense mutations were dominant in this cohort, especially in COL1A2, but the proportion in COL1A1 was lower than reports in Canada (47.3%) [18], Sweden (60.9%) [26], Vietnam (67.6%) [27], and India (85.7%) [28], which is similar to other studies in China [2, 22]. Moreover, this study found 21 novel variations in COL1A1 and COL1A2, which contributed to extending the gene mutant spectrum of OI and revealing its pathogenesis. Of the 18 novel mutations, 4 missense mutations, 1 splicing mutation, 4 deletions, 4 duplications in COL1A1 and 4 missense mutations, and 1 insertion in COL1A2 were identified. In addition, the mutation of c.2299G > A (p. Gly767Ser), c.3655G > A (p. Asp1219Asn) in COL1A1 and c.1009G > A (p. Gly337Ser) in COL1A2 were detected in many Chinese OI families.
Regarding the relationship of phenotype and genotype, it was confirmed that the clinical symptoms of patients with helical mutation were more serious than those of patients with haploinsufficiency. In this study, there was no difference in age of onset and visit between patients with qualitative mutations and those with quantitative mutations. Compare with patients with haploinsufficiency, patients with helical mutation had more fractures (5.2 ± 2.3, P < 0.001). According to the Sillence classification, we compared the relevant clinical characteristics of I, III and IV types. The gender distribution was slightly skewed, with a larger proportion of males than females. Although there was no significant difference in the age of treatment between the subtypes, there was a significant difference in the age of onset. Interestingly, our study found that, unlike previous studies, patients with type III OI had a higher age of onset than those with lighter types I and IV OI, which may be because type III patients included in this study were older at the time of first visit than those in other studies [22, 29, 30]. Although the clinical symptoms of type III patients are more serious and their life span is significantly shortened due to respiratory tract infection and skull fracture, patients over 10 years old are better [31, 32]. In contrast to Rauch [33], this study did not find a phenomenon in which OI patients with mutations close to the carboxyl-terminal end of type 1 collagen may not present blue sclerae. Moreover, there was no significant difference in height, BMD, number of fractures or clinical classification, suggesting that the specific type of COL1A1 or COL1A2 gene mutation was not the main factor affecting the stature of patients, which was consistent with previous research results [12]. In addition, this study also compared the clinical characteristics of children and adults with type I collagen gene mutations. The results showed that the fracture rate decreased, which suggested that patients with osteogenesis imperfecta may have some degree of disease relief with increasing age.
Although this research expanded the spectrum of mutations in COL1A1 and COL1A2 and revealed a significant correlation between genotype and phenotype of OI, there were still some limitations. This was a cross-sectional study, which did not reveal the relationship of genotype and efficacy of drug therapy in patients with OI.
In conclusion, we identified 185 mutations of COL1A1 and COL1A2 in Chinese OI, 18 of which were novel. Among the 185 unrelated Chinese patients, sporadic cases were more common than familial OI cases in China (54.6% vs. 45.4%). A total of 98.9% of patients presented with a fracture history, and the most common fracture sites were extremity long bones. Interestingly, G767S and D1219N in COL1A1 and G337S in COL1A2 were the most frequent in Chinese patients. This study reflects the mutation of the main pathogenic genes, COL1A1 and COL1A2, and the clinical characteristics of OI in China. Furthermore, this finding helps reveal the genetic basis of Asian OI patients and contributes to genetic counselling.
References
Forlino A, Marini JC (2016) Osteogenesis imperfecta. Lancet 387:1657–1671
Zhang H, Yue H, Wang C, Hu W, Gu J, He J, Fu W, Hu Y, Li M, Zhang Z (2016) Clinical characteristics and the identification of novel mutations of COL1A1 and COL1A2 in 61 Chinese patients with osteogenesis imperfecta. Mol Med Rep 14:4918–4926
Lindahl K, Barnes AM, Fratzl-Zelman N, Whyte MP, Hefferan TE, Makareeva E, Brusel M, Yaszemski MJ, Rubin CJ, Kindmark A, Roschger P, Klaushofer K, McAlister WH, Mumm S, Leikin S, Kessler E, Boskey AL, Ljunggren O, Marini JC (2011) COL1 C-propeptide cleavage site mutations cause high bone mass osteogenesis imperfecta. Hum Mutat 32:598–609
Rauch F, Glorieux FH (2004) Osteogenesis imperfecta. Lancet 363:1377–1385
Forlino A, Cabral WA, Barnes AM, Marini JC (2011) New perspectives on osteogenesis imperfecta. Nat Rev Endocrinol 7:540–557
Marini JC, Forlino A, Bächinger HP, Bishop NJ, Byers PH, Paepe A, Fassier F, Fratzl-Zelman N, Kozloff KM, Krakow D, Montpetit K, Semler O (2017) Osteogenesis imperfecta. Nat Rev Dis Primers 3:17052
Roughley PJ, Rauch F, Glorieux FH (2003) Osteogenesis imperfecta–clinical and molecular diversity. Eur Cell Mater 5:41–47
Maioli M, Gnoli M, Boarini M, Tremosini M, Zambrano A, Pedrini E, Mordenti M, Corsini S, D’Eufemia P, Versacci P, Celli M, Sangiorgi L (2019) Genotype-phenotype correlation study in 364 osteogenesis imperfecta Italian patients. Eur J Hum Genet 27:1090–1100
Venturi G, Tedeschi E, Mottes M, Valli M, Camilot M, Viglio S, Antoniazzi F, Tatò L (2006) Osteogenesis imperfecta: clinical, biochemical and molecular findings. Clin Genet 70:131–139
Benusiené E, Kucinskas V (2003) COL1A1 mutation analysis in Lithuanian patients with osteogenesis imperfecta. J Appl Genet 44:95–102
Marini JC, Forlino A, Cabral WA, Barnes AM, San Antonio JD, Milgrom S, Hyland JC, Körkkö J, Prockop DJ, De Paepe A, Coucke P, Symoens S, Glorieux FH, Roughley PJ, Lund AM, Kuurila-Svahn K, Hartikka H, Cohn DH, Krakow D, Mottes M, Schwarze U, Chen D, Yang K, Kuslich C, Troendle J, Dalgleish R, Byers PH (2007) Consortium for osteogenesis imperfecta mutations in the helical domain of type I collagen: regions rich in lethal mutations align with collagen binding sites for integrins and proteoglycans. Hum Mutat 28:209–221
Barber LA, Abbott C, Nakhate V, Do AND, Blissett AR, Marini JC (2019) Longitudinal growth curves for children with classical osteogenesis imperfecta (types III and IV) caused by structural pathogenic variants in type I collagen. Genet Med 21:1233–1239
Jain M, Tam A, Shapiro JR, Steiner RD, Smith PA, Bober MB, Hart T, Cuthbertson D, Krischer J, Mullins M, Bellur S, Byers PH, Pepin M, Durigova M, Glorieux FH, Rauch F, Lee B, Sutton VR (2019) Growth characteristics in individuals with osteogenesis imperfecta in North America: results from a multicenter study. Genet Med 21:275–283
Liu Y, Asan MD, Lv F, Xu X, Wang J, Xia W, Jiang Y, Wang O, Xing X, Yu W, Wang J, Sun J, Song L, Zhu Y, Yang H, Wang J, Li M (2017) Gene mutation spectrum and genotype-phenotype correlation in a cohort of Chinese osteogenesis imperfecta patients revealed by targeted next generation sequencing. Osteoporos Int 28:2985–2995
Ohata Y, Takeyari S, Nakano Y, Kitaoka T, Nakayama H, Bizaoui V, Yamamoto K, Miyata K, Yamamoto K, Fujiwara M, Kubota T, Michigami T, Yamamoto K, Yamamoto T, Namba N, Ebina K, Yoshikawa H, Ozono K (2019) Comprehensive genetic analyses using targeted next-generation sequencing and genotype-phenotype correlations in 53 Japanese patients with osteogenesis imperfecta. Osteoporos Int 30:2333–2342
Essawi O, Symoens S, Fannana M, Darwish M, Farraj M, Willaert A, Essawi T, Callewaert B, De PA, Malfait F, Coucke PJ (2018) Genetic analysis of osteogenesis imperfecta in the Palestinian population: molecular screening of 49 affected families. Mol Genet Genomic Med 6:15–26
Zhang ZL, Zhang H, Ke YH, Yue H, Xiao WJ, Yu JB, Gu JM, Hu WW, Wang C, He JW, Fu WZ (2012) The identification of novel mutations in COL1A1, COL1A2, and LEPRE1 genes in Chinese patients with osteogenesis imperfecta. J Bone Miner Metab 30:69–77
Rauch F, Lalic L, Roughley P, Glorieux FH (2010a) Relationship between genotype and skeletal phenotype in children and adolescents with osteogenesis imperfecta. J Bone Miner Res 25:1367–1374
Sillence DO, Rimoin DL (1978) Classification of osteogenesis imperfect. Lancet 1:1041–1042
Li H, Ji CY, Zong XN, Zhang YQ (2009) Zhonghua. Er Ke Za Zhi 47:487–492
Lee KS, Song HR, Cho TJ, Kim HJ, Lee TM, Jin HS, Park HY, Kang S, Jung SC, Koo SK (2006) Mutational spectrum of type I collagen genes in Korean patients with osteogenesis imperfecta. Hum Mutat 27:599
Li LJ, Lyu F, Song YW, Wang O, Jiang Y, Xia WB, Xing XP, Li M (2019) Genotype-phenotype relationship in a large cohort of osteogenesis imperfecta patients with COL1A1 mutations revealed by a new scoring system. Chin Med J (Engl) 132:145–153
Swinnen FK, Casselman JW, De Leenheer EM, Cremers CW, Dhooge IJ (2013) Temporal bone imaging in osteogenesis imperfecta patients with hearing loss. Laryngoscope 123:1988–1995
Paterson CR, Monk EA, McAllion SJ (2001) How common is hearing impairment in osteogenesis imperfecta? J Laryngol Otol 115:280–282
Hald JD, Folkestad L, Swan CZ, Wanscher J, Schmidt M, Gjørup H, Haubek D, Leonhard CH, Larsen DA, Hjortdal JØ, Harsløf T, Duno M, Lund AM, Jensen JB, Brixen K, Langdahl B (2018) Osteogenesis imperfecta and the teeth, eyes, and ears-a study of non-skeletal phenotypes in adults. Osteoporos Int 29:2781–2789
Lindahl K, Åström E, Rubin CJ, Grigelioniene G, Malmgren B, Ljunggren Ö, Kindmark A (2015) Genetic epidemiology, prevalence, and genotype-phenotype correlations in the Swedish population with osteogenesis imperfecta. Eur J Hum Genet 23:1042–1050
Ho Duy B, Zhytnik L, Maasalu K, Kändla I, Prans E, Reimann E, Märtson A, Kõks S (2016) Mutation analysis of the COL1A1 and COL1A2 genes in Vietnamese patients with osteogenesis imperfecta. Hum Genomics 10:27
Stephen J, Shukla A, Dalal A, Girisha KM, Shah H, Gupta N, Kabra M, Dabadghao P, Hasegawa K, Tanaka H, Phadke SR (2014) Mutation spectrum of COL1A1 and COL1A2 genes in Indian patients with osteogenesis imperfecta. Am J Med Genet A 164A:1482–1489
Kobayashi T, Nakamura Y, Suzuki T, Yamaguchi T, Takeda R, Takagi M, Hasegawa T, Kosho T, Kato H (2018) Efficacy and safety of denosumab therapy for osteogenesis imperfecta patients with osteoporosis-case series. J Clin Med 7:479
Mrosk J, Bhavani GS, Shah H, Hecht J, Krüger U, Shukla A, Kornak U, Girisha KM (2018) Diagnostic strategies and genotype-phenotype correlation in a large Indian cohort of osteogenesis imperfecta. Bone 110:368–377
Kovero O, Pynnönen S, Kuurila-Svahn K, Kaitila I, Waltimo-Sirén J (2006) Skull base abnormalities in osteogenesis imperfecta: a cephalometric evaluation of 54 patients and 108 control volunteers. J Neurosurg 105:361–370
Rohrbach M, Giunta C (2012) Recessive osteogenesis imperfecta: clinical, radiological, and molecular findings. Am J Med Genet C Semin Med Genet 160C:175–189
Rauch F, Lalic L, Roughley P, Glorieux FH (2010b) Genotype-phenotype correlations in nonlethal osteogenesis imperfecta caused by mutations in the helical domain of collagen type I. Eur J Hum Genet 18:642–647
Acknowledgements
This work was funded by the National Key R&D Program of China (2018YFA0800801), Shanghai Key Clinical Center for Metabolic Disease, Shanghai Health Commission Grant (2017ZZ01013), National Natural Scieuce Foundation of China (81870618) and the Emerging Frontier Technology Joint Research Project of Shanghai Shenkang Hospital Development Center (SHDC2018120). We would like to thank all the staff and patients who have contributed to this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The present study was approved by the Ethics Committee of Shanghai Jiao Tong University Affiliated Sixth People's Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Xi, L., Zhang, H. & Zhang, ZL. Clinical and genetic analysis in 185 Chinese probands of osteogenesis imperfecta. J Bone Miner Metab 39, 416–422 (2021). https://doi.org/10.1007/s00774-020-01163-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-020-01163-5